Abstract
Background
The diagnosis, treatment and monitoring of patients with idiopathic intracranial hypertension (IIH) are highly complex processes that require interdisciplinary collaboration with respect to neurology, ophthalmology, neuroradiology, neurosurgery and endocrinology. Accordingly, there is a consensus among international guidelines that the management of these aspects of care should be the responsibility of specialized centers that are equipped with appropriate facilities. The objective of the Austrian Network for Idiopathic Intracranial Hypertension (AN4IH) is to establish a national network of excellence and to provide comprehensive recommendations for the structure and operation of specialized IIH centers (AN4IH centers), including an integrated, interdisciplinary diagnostic and treatment pathway.
Methods
This consensus was developed by an interdisciplinary panel of experts convened by Austrian neurologists, (neuro)ophthalmologists, neuroradiologists, neurosurgeons and endocrinologists. The process adhered to a formal consensus methodology.
Results
The AN4IH consensus provides a comprehensive, integrated, interdisciplinary framework addressing care infrastructure, urgency stratification, diagnostics, treatment and monitoring, as well as considerations related to family planning and pregnancy in patients with IIH. The AN4IH consensus is explicitly intended as a supplement and extension to existing international guidelines.
Conclusions
The management of IIH necessitates a structured, interdisciplinary approach to optimize patient outcomes. Through formal consensus methodology, the AN4IH provides expert – and where available evidence – based recommendations for specialized care centers, emphasizing standardized diagnostic pathways, urgency stratification and tailored treatment protocols. By fostering collaboration and institutionalizing best practices, the AN4IH model represents a significant advancement in delivering comprehensive, patient-centered care for this complex neurological disorder and encourages participants to create a secure, quality-controlled shared database for the collection of all clinical and paraclinical data, alongside the establishment of a biobank for the storage of biosamples.
This is a visual representation of the abstract.
Keywords
Introduction
Idiopathic intracranial hypertension (IIH, formerly known as pseudotumor cerebri or benign intracranial hypertension) is a condition of unknown etiology characterized by elevated intracranial pressure (ICP) that typically – but not exclusively – affects young women with obesity (1). The annual incidence is estimated at 4–21 cases per 100,000 individuals, though this figure is likely an underestimation due to the rising prevalence of obesity and limited awareness of IIH (2,3).
As the (differential-)diagnosis, treatment and monitoring of patients with IIH (pwIIH) are highly complex, interdisciplinary collaboration between neurology, ophthalmology, neuroradiology, neurosurgery and endocrinology is required. Accordingly, major international guidelines recommend that these aspects of care be managed in specialized centers equipped with appropriate interdisciplinary infrastructure (4–9).
To date, Austria, similar to many countries in the Western hemisphere, lacks a standardized care structure for pwIIH, and no dedicated national network exists to meet these multidisciplinary requirements. The establishment of the Austrian Network for Idiopathic Intracranial Hypertension (AN4IH) addresses this gap by creating a nationwide, interdisciplinary network of specialized centers committed to uniform quality standards, thereby ensuring state-of-the-art care for patients with IIH.
The aim of this consensus statement, developed by members of AN4IH, is to provide comprehensive recommendations for the organizational structure of IIH care centers, as well as for the diagnosis, treatment and monitoring of pwIIH. The AN4IH consensus is intended as a supplement and extension – not a replacement – of current international guidelines.
Furthermore, we encourage other countries/headache centers to adapt and extract pathways from this consensus to harmonize diagnosis, treatment and future scientific collaboration on a broader scale.
Methods
This document has been developed by an interdisciplinary panel of experts in management of pwIIH comprising each a neurologist, (neuro)ophthalmologist, neuroradiologist, neurosurgeons and endocrinologist from four University centers in Austria. The committee identified the scope and topics formulating clinical questions covering diagnosis, treatment and monitoring of pwIIH including quality standards for interdisciplinary care delivery structure, conduction, interpretation and reporting of clinical and paraclinical findings, as well as aspects of pregnancy/family planning.
The formulation and agreement of the recommendations were done using the modified Nominal Group Technique, which is a highly structured procedure, based on iterative ratings with feedback, to reach consensus in a small group of experts on topics for which expert opinion is relevant (10). The topics were discussed within the expert committee members and other invited discussants during multiple online meetings starting in September 2024. As a result, the first set of statements was drafted by the core working group (GB, BP, WM, KN, ML, SL and GB) and then circulated to the expert committee for a first round of voting through email, using a percentage agreement scale, with a required pre-defined 80% level of agreement. The revised statements/recommendations were submitted for agreement in a further round of voting through email and finalized at a focused workshop held in Innsbruck in March 2025 (see supplementary material, File S1). The definitiveness of each statements/recommendation reflects the grade of agreement reached during the consensus process, rather than the strength of empirical evidence, and should therefore be interpreted as consensus-based guidance unless otherwise supported by data or references. The final manuscript was reviewed and approved by all members of the expert committee.
Results and recommendations
Quality standards: target population and care infrastructure
The target population addressed by the care infrastructure established through the AN4IH includes post-pubertal pwIIH or a suspected diagnosis thereof. This encompasses patients presenting with symptomatic or incidentally detected papilledema, as well as individuals suffering from chronic and therapy-refractory headaches that cannot be attributed to other primary or secondary headache disorders according to the International Classification of Headache Disorders, 3rd edition (ICHD-3) (11).
As with any other highly complex neurological disease, diagnostic evaluation, as well as the treatment and monitoring of patients within this target group, should be carried out at specialized IIH centers that are equipped with an appropriate interdisciplinary framework. Each center should be composed of qualified and experienced specialists in neurology, (neuro-)ophthalmology, neuroradiology, neurosurgery and endocrinology. The interdisciplinary organization of the center should be coordinated by the neurology department, taking into account local personnel and structural capacities. Each center should provide a point of contact or organizational structure for referring physicians, designed to ensure efficient coordination of diagnostic work-up, diagnosis, treatment and monitoring. This structure should guarantee prompt access for patients in the target group to an integrated, interdisciplinary diagnostic and therapeutic pathway, appropriate to the urgency of their clinical presentation (Figure 1).
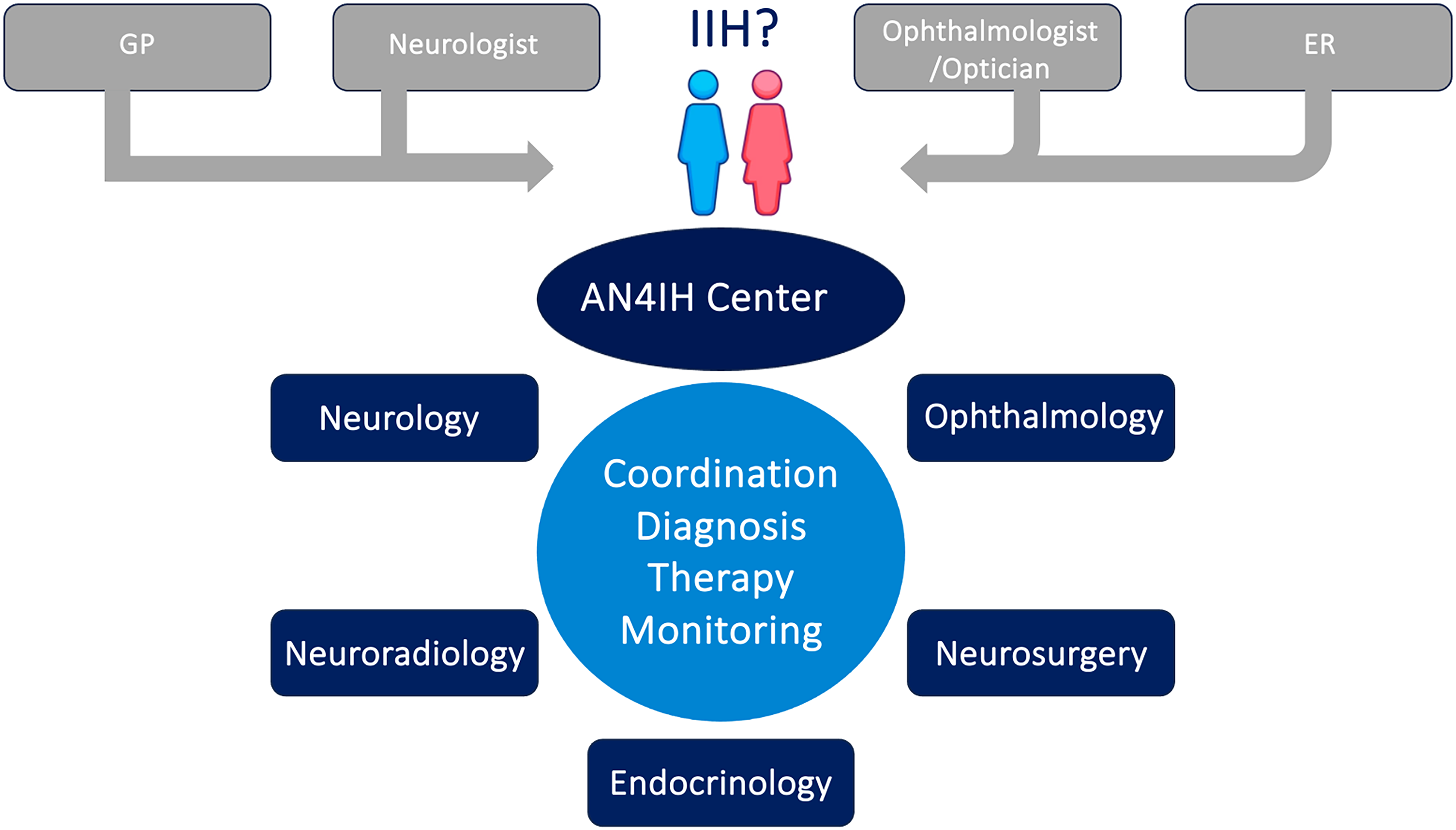
AN4IH center structure and referrals. ER = emergency room. GP = general practitioner. IIH = idiopathic intracranial hypertension.
To meet the complex demands of interdisciplinary care and ensure optimal treatment quality, each center should establish an IIH Board. This board, coordinated by neurology, should include at least one medical specialist with documented expertise in each of the following disciplines: neurology, (neuro-)ophthalmology, neuroradiology, neurosurgery and endocrinology. The primary function of the IIH Board is to determine the indication for invasive therapeutic interventions (see section on treatment). Such interventions should only be performed following interdisciplinary approval by the IIH Board – emergency situations being the sole exception (e.g. urgent cerebrospinal fluid (CSF) drainage or lumbar puncture (LP) in the setting of imminent vision loss). The frequency of IIH Board meetings should be adapted to local needs and capacities but must allow for ad hoc convening to evaluate acute cases and prevent delays in the initiation of urgently required invasive procedures.
Quality standards: diagnostics and diagnosis
The diagnostic evaluation of patients within the target group should be guided by the urgency of the clinical presentation and the available findings. Decisions regarding the diagnostic pathway should be made by medical specialists with appropriate expertise. As an initial step, a primary screening – or, if not feasible, a structured review of available findings – should be performed. This should include, at a minimum, visual acuity testing, perimetry and fundoscopy (Figure 2).

AN4IH center initial urgency stratification for diagnostic pathway. ER = emergency room. GP = general practitioner. IIH = idiopathic intracranial hypertension.
The organization of this initial screening is at the discretion of each center; however, the establishment of a centralized point of initial contact (e.g. telephone hotline or dedicated email address) is recommended.
Patients presenting with moderate to severe papilledema (Frisén grade ≥3) and/or functionally relevant visual impairment should ideally undergo the AN4IH diagnostic pathway as promptly as possible, and in any case within a maximum of one week (Table 1).
Overview of the AN4IH diagnostic pathway.

CCT = coronary computed tomography; CTA = computed tomograpy angiography.
Patients with mild papilledema (Frisén grades 1–2) and no functionally relevant visual impairment should complete the diagnostic pathway within a maximum of four weeks.
In patients without papilledema (Frisén grade 0) and without functionally relevant visual impairment, but in whom IIH is suspected based on (typically chronic, therapy-refractory) headaches or paraclinical findings, the diagnostic pathway should be completed within a maximum of 12 weeks. In this subgroup, the decision to perform LP should be made with careful consideration of the risk–benefit ratio.
The interdisciplinary AN4IH diagnostic pathway is aligned with major international guidelines (Table 1) (4–7).
Medical history and neurological examination
Evaluation for suspected IIH within the AN4IH diagnostic pathway is fundamentally based on a thorough history aimed at characterizing key clinical features of elevated ICP. This includes visual disturbances (transient visual obscurations, peripheral visual field defects), headache characteristics (type, frequency, intensity, localization/lateralization), associated symptoms (nausea, vomiting, photophobia, phonophobia, allodynia, tinnitus, diplopia) and their temporal evolution. Particular emphasis should be placed on weight history (overweight and obesity, rapid weight gain), cardiometabolic parameters, medication history (especially analgesic overuse, vitamin A derivatives/retinoids, tetracyclines, nitrofurantoin, human growth hormone, lithium, corticosteroids, phenothiazines), pregnancy or signs of endocrine abnormalities (e.g. hypo-/hyperthyroidism, hypoparathyroidism, iron deficiency, hypervitaminosis A, hypercortisolism/polycystic ovary syndrome (PCOS), hypocortisolism/Addison's disease, hyperaldosteronism), evidence of right heart failure or pulmonary hypertension, and sleep-related symptoms suggestive of obstructive sleep apnea. Neurological examination should focus on identifying visual field deficits and cranial nerve abnormalities, particularly involving the abducens nerve.
Ophthalmologic examination
At AN4IH centers, ophthalmologic assessment must include best-corrected visual acuity using standardized optotypes (e.g. Snellen chart, number rows or ETDRS), visual field testing with static threshold perimetry (30° radius) and/or kinetic perimetry with multiple isopters, fundoscopy graded using the Frisén scale (stages 0–5) and photographic documentation where feasible. All findings should be interpreted by ophthalmologists with relevant expertise within the IIH center structure (12,13).
Optical coherence tomography (OCT) using spectral-domain or swept-source techniques must be performed according to OSCAR-IB quality control criteria (14). A scan centered on the optic nerve head should assess peripapillary retinal nerve fiber layer (pRNFL) thickness, a sensitive marker of papilledema. However, as pRNFL may appear “pseudo-normalized” due to pressure-induced axonal atrophy, a supplementary macular volume scan is required to quantify the thickness of the ganglion cell layer (GCL), or GCL plus inner retinal layers (GCIPL, GCL+, GCL++). The GCL reflects neurons responsible for central vision and remains unaffected by swelling, making it reliable for early detection of optic nerve damage – even in the presence of active papilledema. Integrating pRNFL and GCL data enables a comprehensive assessment of both papilledema and neuroaxonal damage. OCT imaging of the optic disc, especially using enhanced depth imaging (EDI) or swept-source OCT, also aids in differentiating pseudopapilledema (e.g. due to optic disc drusen or congenital anomalies). A protrusion of Bruch's membrane in the papillary region may also suggest elevated retrobulbar pressure.
Ultrasound of the optic nerve can support the diagnosis of papilledema. B-scan ultrasonography with a 10-MHz probe can detect the classic “bat sign”, indicating perineural CSF accumulation. Unlike conventional OCT, B-scan ultrasound is also sensitive to deeply located optic disc drusen. Quantitative assessment of the optic nerve sheath diameter (ONSD) using standardized A-scan ultrasound is performed with an 8-MHz probe positioned at the eye's temporal equator in primary gaze (15,16). At least two measurements within 3 mm of the posterior sclera should be taken, with the highest value documented. An ONSD ≥5.0 mm strongly suggests elevated ICP; values <4.5 mm are considered normal in adults; intermediate values are non-specific. A ≥10% reduction in ONSD during abduction (30° test) can further support elevated pressure. However, ultrasound is operator-dependent and requires high expertise.
A critical component of the ophthalmological evaluation is the final integrated assessment to facilitate interdisciplinary communication, which should be documented within the AN4IH center structure. This summary should address functional visual impairment (visual acuity and/or field), grading of papilledema (fundoscopy including Frisén scale, pRNFL) and grading of neuroaxonal damage (pRNFL and GCL), including any longitudinal changes.
Magnetic resonance imaging (MRI) and MR venography
Key differential diagnoses to exclude comprise cerebral venous sinus thrombosis (CVST), dural arteriovenous fistulas (dAVF) and intracranial space-occupying lesions. Cerebral MRI (cMRI) and MR venography (MRV) are the methods of choice. If contraindicated, cerebral CT with venous CT angiography (CTV) may be used as an alternative. Beyond differential diagnosis, MRI and MRV may reveal indirect signs suggestive of IIH and should therefore be performed before LP. Common findings in IIH include bilateral transverse sinus stenosis (TSS) without CVST, reflecting elevated ICP with secondary venous compression. Other suggestive features include tortuous or elongated optic nerves with perineural CSF distension (optic nerve sheath dilation), posterior globe flattening (PGF) and a flattened pituitary gland (“empty sella” or “partial empty sella”) (17). Although none of these findings are pathognomonic, the presence of at least three of four features yields a sensitivity of 64% and specificity of 90–100% (18). High-quality imaging is essential for diagnostic sensitivity. AN4IH centers should therefore perform MRI/MRV using the following minimum sequences: 3D T2-weighted imaging (slice thickness 2–3 mm, maximum 5 mm), 3D fluid-attenuated inversion recovery (FLAIR) (isovoxel ≤4–5 mm), axial T2*-GRE or SWI (2–5 mm), 3D T1-weighted imaging pre- and post-gadolinium (isovoxel) and MRV. If gadolinium is contraindicated, phase-contrast MRV or time-of-flight (TOF) venous angiography should be used. Optional additional sequences may include coronal and axial T2 fat-suppressed (from orbits to sella) to optimize optic nerve visualization. Interpretation of MRI/MRV should be performed by expert neuroradiologists using the standardized Korsbaek criteria, for which the components should be explicitly graded in the final report (17).
Cerebrospinal fluid analysis and measurement of CSF opening pressure
LP serves two roles in the diagnostic pathway: to exclude inflammatory/infectious differential diagnoses and to objectively confirm elevated ICP. The procedure must be performed under sterile conditions by clinicians with experience in the procedure using traumatic or atraumatic spinal needles (20–22 G). If unsuccessful, CT-guided LP by skilled specialists is recommended.
CSF opening pressure (CSF-OP) should be measured immediately after dural puncture (before sampling for diagnostics), using a standardized manometer with the patient in the left lateral decubitus position, neck relaxed and legs extended. CSF-OP is recorded in cmCSF. A value <25 cmCSF is considered normal; values ≥25 cmCSF are considered pathological in adults, although there is a diagnostic gray zone including values up to 30 cmCSF in healthy adults. Since CSF-OP may fluctuate, a repeat measurement or continuous monitoring may be warranted in patients with suggestive clinical features and borderline readings.
CSF analysis should include cell count, total protein, glucose, lactate, albumin quotient and immunoglobulin quotients. At least two reserve tubes or 5–10 ml should be collected for further diagnostics if needed. After sampling, CSF pressure should be remeasured. If the OP exceeds 25 cmCSF, therapeutic drainage should be performed immediately, reducing pressure to 10–15 cmCSF or until low-pressure symptoms appear (maximum drained volume 50 ml). Both the total volume drained and CSF closing pressure (CSF-CP) should be documented.
Differential diagnosis: idiopathic vs. secondary intracranial hypertension
In addition to previously discussed differentials, conditions such as obstructive sleep apnea syndrome (OSAS), endocrine disorders (e.g. hypo-/hyperthyroidism, hypoparathyroidism, iron deficiency, hypervitaminosis A, hypercortisolism/polycystic overial syndrome, hypocortisolism/Addison's disease, hyperaldosteronism) and specific medications (e.g. tetracyclines, vitamin A derivatives, human growth hormone, nitrofurantoin, nalidixic acid, danazol, lithium, tamoxifen, indomethacin, phenothiazines, alpha-interferon, cyclosporin, cimetidine, amiodarone) have been associated with elevated intracranial pressure (19,20). These associations, however, lack confirmed causality, and many factors overlap with established IIH risk factors such as obesity, making it difficult in some cases to distinguish between idiopathic and secondary intracranial hypertension.
Diagnosis and diagnostic criteria
Following completion of the interdisciplinary AN4IH diagnostic pathway, the diagnosis should be established according to the criteria of Korsbaek (17,21).
A diagnosis of IIH should be made when CSF is normal and at least two of the following three core criteria are met:
Presence of papilledema Detection of at least three of the four characteristic MRI signs of IIH CSF-OP ≥25 cmCSF
Additionally, all relevant differential diagnoses – specifically CVST, dAVF and intracranial space-occupying lesions – should be ruled out, and there must be no alternative explanation for the symptoms and findings.
Quality standards: Treatment
Following completion of the AN4IH diagnostic pathway, patients diagnosed with IIH should be treated in accordance with the AN4IH treatment algorithm, which aligns with major international guidelines (Table 2) (4–7).
Overview of the AN4IH treatment algorithm.

Only in cases of confirmed sinus venous stenosis with a relevant pressure gradient on conventional digital subtraction angiography at centers with explicitly high expertise.
Cave: avoid first line treatment facilitating weight gain.
BMI = body mass index; CGRP = calcitonin gene-related peptide; CSF = cerebrospinal fluid; GIP = glucose-dependent insulinotropic polypeptide; GLP = glucagon-like peptide ; LP = lumbar puncture; NSAID = non-steroidal anti-inflammatory drug.
The primary therapeutic objective is the prompt reduction of intracranial pressure and resolution of papilledema, which is the main risk factor for permanent visual impairment in IIH. The urgency and intensity of treatment should be clearly based on the severity of papilledema, the presence and potential progression of visual impairment (as assessed by visual acuity and perimetry), and the presence of neurological deficits, typically abducens nerve palsy. The AN4IH treatment algorithm defines three escalation levels. All treatment decisions should ideally be made in interdisciplinary consultation by specialists with appropriate expertise.
Patients with mild papilledema (Frisén grade ≤2), without functionally relevant visual impairment and without focal neurological deficits, should be treated according to stage 1 of the AN4IH pathway. This includes therapeutic LP, performed within the diagnostic setting, providing temporary relief for one to two weeks, and non-pharmacological weight reduction with dietary and lifestyle advice in all patients with a body mass index (BMI) ≥25, targeting at least a 10% reduction from baseline. In patients with BMI ≥27, pharmacological therapy with glucagon-like peptide-1 (GLP-1) receptor agonists or dual glucose-dependent insulinotropic polypeptide (GIP)/-1 receptor agonists can be added off-label. These should ideally be prescribed after endocrinologic evaluation by endocrinologists or physicians experienced in their use.
Patients with mild papilledema (Frisén grade 1–2) and no visual impairment should additionally receive pharmacological therapy to lower ICP. Acetazolamide, at a dose of 250–1000 mg per day, is the first-line agent based on data from a multicenter, randomized, double-blind, placebo-controlled trial (22). Alternatives include topiramate, 25–50 mg once daily (preferably in the evening to minimize side effects, with caution in women of childbearing potential), or furosemide, 40–80 mg per day, although the evidence supporting these is weaker. None of these medications are officially approved for IIH, and so special attention must be paid to informing patients about indications, risks, and side effects. To improve tolerability and reduce risk of adverse effects (e.g. metabolic acidosis, electrolyte imbalance) medications should be initiated at low doses and titrated based on tolerance and response. In patients without papilledema (Frisén grade 0), ICP-lowering medication is generally not indicated due to the absence of risk to vision.
Patients with moderate to severe papilledema (Frisén grade ≥3) and/or functionally relevant visual impairment should be treated according to Stage 2 of the AN4IH pathway. In addition to the stage 1 measures, this includes intensified weight loss efforts. All patients with BMI ≥27 should receive pharmacological weight reduction therapy. If a weight loss of at least 10% from baseline is not achieved within three months, or if the medication is not tolerated, evaluation for switching to a more potent pharmacological weight reduction therapy or bariatric surgery should be undertaken (23). Acetazolamide therapy may be escalated to 4000 mg/day and combined with topiramate up to 100 mg. Experimental studies suggest a synergistic effect of this combination in lowering ICP (24). In patients with visual impairment, therapeutic LP should be performed with a target CSF-CP 10–15 cmCSF and a maximum drainage volume of 50 ml. The therapeutic goal is rapid reduction of papilledema (Frisén grade ≤2) and improvement or at least stabilization of visual function. If these goals are achieved, medications may be gradually tapered toward stage 1 levels. If not, an urgent interdisciplinary review by an IIH Board is required to determine the next steps and potentially escalate to stage 3.
Patients with moderate to severe papilledema (Frisén grade ≥3) or progressive visual impairment despite full implementation of stage 2 should be managed according to stage 3 after case discussion by the IIH Board. Stage 3 includes further intensification of ICP-lowering pharmacotherapy using a triple combination of acetazolamide, topiramate, and furosemide, up to the respective maximum or tolerable doses. Invasive interventions should also be considered at this stage. Available options may include placement of a lumbar CSF drainage system (target pressure 10–15 cmCSF), a ventriculoperitoneal shunt (VP shunt) with a pressure-regulating valve, or bilateral optic nerve sheath fenestration. In centers with proven expertise, venous sinus stenting may also be considered as an experimental option, provided that a high-grade sinus venous stenosis with a significant pressure gradient (≥8 mmHg) is confirmed by conventional digital subtraction angiography (DSA) with venous manometry. Treatment decisions in stage 3 should be individualized and made in an interdisciplinary IIH Board meeting based on patient- and disease-specific characteristics, as well as center expertise and experience. As in the previous stages, the therapeutic goal is a rapid reduction of papilledema (Frisén grade ≤2) and improvement or stabilization of visual function. If these goals are achieved, therapy should be de-intensified stepwise with a gradual tapering of medications toward stage 2, and lumbar drainage devices may be removed if appropriate.
The secondary therapeutic goal is the management of headaches, which, although not life-threatening, can impair quality of life as severely as visual symptoms (25). It is important to note that headache frequency and severity correlate poorly with papilledema severity and ICP, and vary significantly between individuals (26). Persistent or isolated headaches, despite adequate treatment of papilledema, are therefore not an indication for invasive therapy (27). However, emerging evidence suggests that headache phenotype is an independent predictor of outcome and therapeutic response. More than 50% of patients exhibit a migrainous phenotype according to ICHD-3 criteria, while others present with tension-type or undefined headache phenotypes (28). Headache treatment within the AN4IH pathway should be independent of the therapeutic stage and guided primarily by headache severity, frequency, and phenotype (“treat to the phenotype”). Accurate characterization of the headache during medical history-taking is critical, aiming to assign the patient to one or more phenotypes and to communicate this clearly to them. Immediately after diagnosis, the initial focus should be on identifying an effective acute therapy. Following the “treat to the phenotype” principle, migraine-like headaches should be treated with standard migraine therapies, and tension-type headaches treated with therapies appropriate for that phenotype. The goal is to establish effective acute treatment for each identified phenotype. Particular attention should also be paid by physicians and patients to identifying signs of medication overuse headache.
If headaches persist for more than three months with high frequency or substantial impact, prophylactic treatment should be initiated regardless of the concurrent stage within AN4IH treatment pathway, with the aim of reducing headache frequency and associated disability. Independent of phenotype, non-pharmacological measures should be recommended, including aerobic endurance exercise (≥60 minutes on ≥3 days/week), progressive muscle relaxation (e.g. Jacobson technique), biofeedback and possibly pain psychology consultation. If these are insufficient, pharmacological prophylaxis may be considered, again following the “treat to the phenotype” approach and tailored individually by neurologists experienced in headache management (29–31).
For the migraine-like phenotype, preventive therapies approved for migraine should be used, with topiramate (due to its additional benefit in reducing papilledema) and monoclonal antibodies against calcitonin gene-related peptide (CGRP) being prioritized, particularly given their favorable side-effect profiles (as flunarizine and beta-blockers often promote weight gain) (32). Other potentially effective options include botulinum toxin A, amitriptyline, venlafaxine, duloxetine, beta-blockers, sartans and oral CGRP receptor antagonists (gepants). For the tension-type phenotype, first-line therapy is amitriptyline. Second-line options include duloxetine, venlafaxine or tizanidine, with recommendations adjusted from standard guidelines to account for the varying effects of these agents on body weight gain (30).
Quality standards: Monitoring
PwIIH following completion of the AN4IH diagnostic pathway and undergoing treatment according to the AN4IH treatment algorithm should be monitored in accordance with the AN4IH monitoring pathway, which follows the major international guidelines (Table 3) (4–7).
Overview of the AN4IH monitoring pathway.
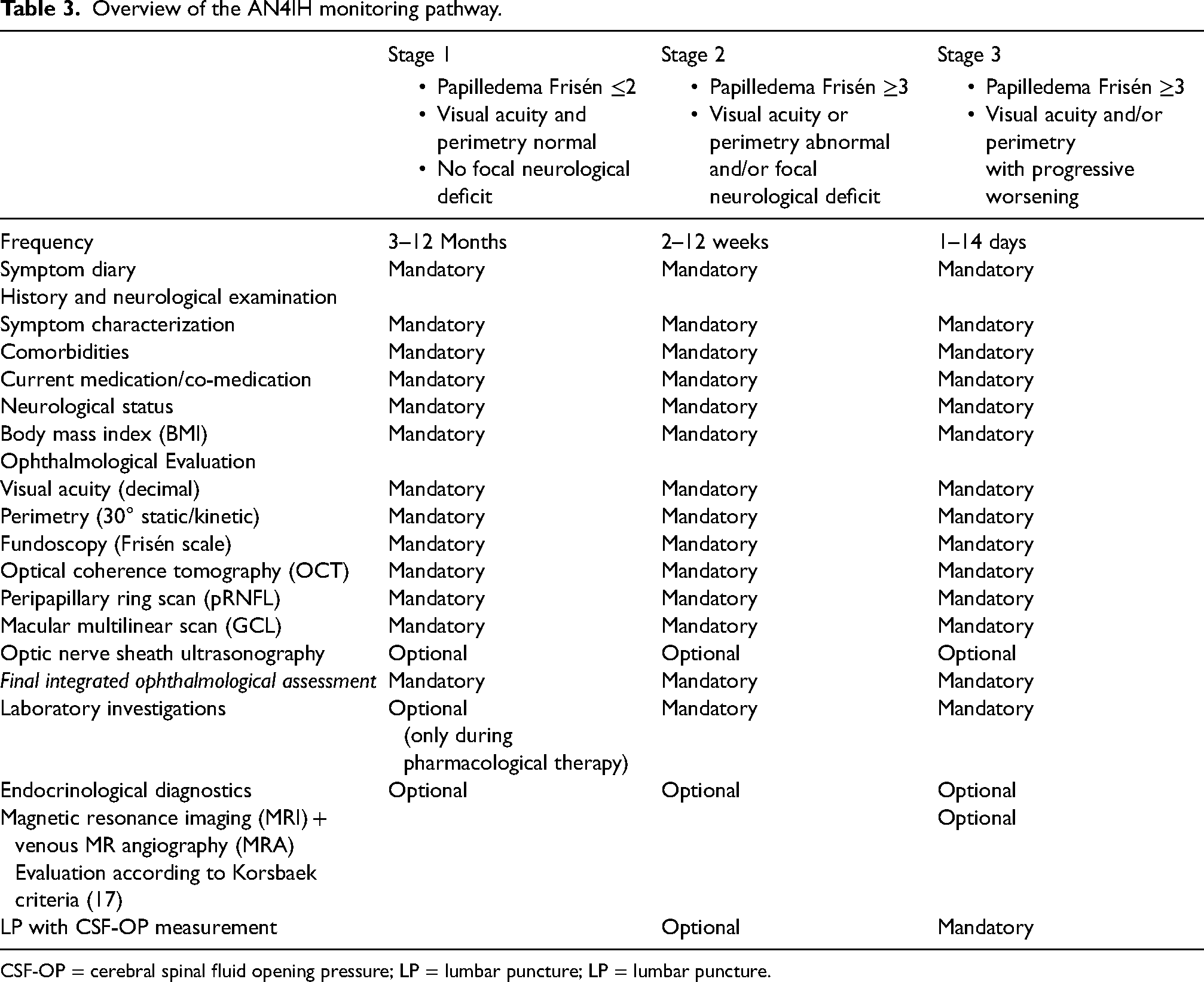
CSF-OP = cerebral spinal fluid opening pressure; LP = lumbar puncture; LP = lumbar puncture.
Monitoring within the AN4IH framework is primarily based on thorough clinical history-taking to assess symptom progression, as well as possible side effects and complications of the administered pharmacological therapies. In addition to history-taking, the use of a symptom diary – such as the AN4IH IIH Symptom Calendar – is considered essential because it allows for systematic longitudinal assessment and objective evaluation of treatment effects and disease dynamics, at the same time as improving treatment adherence and physician-patient communication (33). Particular attention should also be paid to monitoring weight progression and evaluating compliance with and success of weight reduction measures. Neurological examination should focus on detecting visual field deficits and cranial nerve abnormalities, particularly involving the abducens nerve.
Ophthalmological monitoring in the AN4IH pathway should include visual acuity testing, perimetry, fundoscopy with Frisén scale grading, and OCT of the pRNFL and the GCL (or alternatively GCIPL, GCL+ or GCL++). Optional use of optic nerve sheath ultrasonography may provide a sensitive marker for changes in ICP under therapy; however, due to significant inter-rater variability, a high level of expertise in both performance and interpretation is required. Again, a mandatory component is the final integrated and documented assessment to facilitate interdisciplinary communication. This assessment should address the presence of functionally relevant impairment (visual acuity and/or perimetry), the grading of papilledema (based on Frisén scale and pRNFL) and neuroaxonal damage (pRNFL and GCL), along with their respective dynamics over time.
Routine blood count and metabolic panel assessments should be performed under ongoing pharmacological therapy to detect potential adverse effects or complications at an early stage. Follow-up imaging with cMRI and MRV is not routinely required within the AN4IH monitoring pathway, but may be indicated in cases of diagnostic uncertainty, suspicion of secondary pathology, or prior to initiating invasive therapies. The same applies to LP for CSF-OP measurement, which, apart from its use in therapeutic drainage, should only be considered when diagnostic uncertainty exists or discrepancies arise between the various components of standard monitoring.
The frequency of monitoring is guided by the AN4IH treatment stage, and thus, by the severity of papilledema, the presence and progression of visual or neurological deficits, as well as the duration of disease stability, the current dosage of ICP-lowering medication and the interval since the last dose adjustment. In AN4IH treatment stage 1, monitoring should occur every 3–12 months. In stage 2, the recommended interval is every 2–12 weeks, while, in stage 3, follow-up should be scheduled every 1–14 days. Close interdisciplinary communication and decision-making within the IIH Board are essential at stages 2 and 3.
Pregnancy and family planning
Given that women of childbearing age represent the majority of pwIIH, considerations related to pregnancy and family planning are of critical importance. IIH does not typically impair fertility directly; however, associated factors such as obesity and PCOS may negatively affect reproductive outcomes and increase the risk of pregnancy-related complications (34). Therefore, following diagnosis – and no later than upon achieving disease stability – endocrinological screening should be performed, and, if necessary, appropriate therapeutic interventions initiated. GLP-1 RA or GIP/GLP1-RA should be stopped in case of family planning or unplanned pregnancy, and intensive lifestyle and nutritional advice should be refreshed.
Pregnancy-related weight gain inherently may increase the risk of IIH worsening or relapse, especially in cases of suboptimal control of ICP. For this reason, pwIIH should be advised to limit weight gain during pregnancy to less than 15 kg, and close monitoring in accordance with the AN4IH monitoring pathway should occur every 4–8 weeks.
Therapeutic options during pregnancy are limited because many pharmacological treatments are contraindicated or not approved for use. Although acetazolamide is not licensed for use in pregnancy, its teratogenic potential appears to be low. While high doses have shown teratogenic effects in rodent studies, no increased risk of congenital malformations has been observed in humans. Acetazolamide may therefore be considered in pregnancy at low doses (≤500 mg) based on an individualized risk–benefit assessment and after thorough patient counseling, especially in the absence of better-studied alternatives (35). In contrast, topiramate is associated with an increased risk of birth defects, most notably a sixfold higher risk of cleft lip and/or palate (36). Furthermore, its use in the second and third trimesters has been linked to reduced birth weight and potential cognitive impairments in the child. Topiramate is therefore contraindicated during pregnancy and should only be used with caution in IIH patients with plans for conception. Its use is also contraindicated in women of reproductive age not using highly effective contraception. Therapeutic administration of topiramate should only be considered after careful risk–benefit evaluation and comprehensive patient counseling (37).
In severe cases, invasive interventions such as therapeutic LP, lumbar CSF drainage or VP shunt may be considered, but should only be performed following interdisciplinary case review and indication by the IIH Board.
From an obstetric perspective, pwIIH with stable disease (treatment stage 1) are not at increased risk during vaginal delivery. Therefore, the mode of delivery should be determined primarily by obstetric criteria and patient preference (38). In contrast, for pwIIH with unstable disease (treatment stage ≥2), cesarean section should be considered to avoid transient elevations in intracranial pressure during the expulsive phase of labor (38).
Comprehensive interdisciplinary care involving close collaboration with obstetrics and gynecology is essential to minimize the risk of complications for both mother and child.
Considerations and perspectives
These recommendations represent the consensus of the AN4IH network based on currently available evidence and expert opinion where data are lacking. They are intended as guidance for clinical decision-making, not as prescriptive standards of care. Individual patient characteristics, comorbidities and local resources may necessitate deviation from these recommendations. Clinicians should apply their own judgement and consider alternative approaches where appropriate. Statements identified as consensus-based should not be interpreted as universally applicable or evidence-graded. As such, they should be evaluated and validated in academically led studies, which is a clear future direction of our network.
Treatment of IIH without papilledema (IIH-WOP) was not specifically addressed in the AN4IH consensus because, according to the applied Korsbæk criteria, it does not represent a distinct entity; however, it is implicitly encompassed within stage 1 of the treatment and monitoring plan (see supplementary material, File S1).
A highly relevant aspect for implementation in clinical practice is availability of interdisciplinary IIH care to ensure access for all pwIIH – in Austria and beyond. The present consensus recommendation provides a foundational framework to support this objective. As a “living” consensus document, it will be re-evaluated annually by the AN4IH network and updated as necessary.
Based on this framework, continuous education and networking among neurologists, ophthalmologists, neuroradiologists, neurosurgeons and endocrinologists working in both inpatient and outpatient settings across is essential. These activities will be facilitated by AN4IH through a series of national and regional events.
The AN4IH network envisions that intra- and/or extramural centers, either independently or as part of structured networks, will offer all components of an integrated diagnostic and treatment concept, thereby fulfilling the structural and organizational prerequisites for the care of pwIIH. In the long term, meeting these requirements is intended to lead to formal certification as an AN4IH center.
Another key goal of the AN4IH network is the establishment of an analogous care structure for prepubertal patients with confirmed or suspected IIH (39). To this end, the recruitment and integration of (neuro-)pediatric network partners is being actively pursued.
A critical gap in the current care landscape for pwIIH lies in the absence of dedicated concepts for outpatient and inpatient rehabilitation. Such programs are urgently needed to maintain or restore patients’ ability to work and participate socially. Addressing this need would constitute a crucial step in reducing the substantial socioeconomic burden associated with IIH.
Equally important is the clarification of the currently inconsistent reimbursement policies for diagnostic, therapeutic and monitoring services – even within Austria. The present consensus recommendation serves as an important tool in facilitating dialogue with relevant stakeholders and policy representatives.
During the development of this consensus, several evidence gaps in the diagnosis, treatment and monitoring of pwIIH were identified. These gaps are intended to be addressed progressively through research conducted within the AN4IH network and the data it generates. A key initial step in this process is the establishment of a secure, quality-controlled shared database for the collection of all clinical and paraclinical data, as well as a biobank for the collection of biological samples.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251391374 - Supplemental material for Diagnosis, treatment and monitoring of idiopathic intracranial hypertension: Consensus recommendations of the Austrian IIH network (AN4IH)
Supplemental material, sj-docx-1-cep-10.1177_03331024251391374 for Diagnosis, treatment and monitoring of idiopathic intracranial hypertension: Consensus recommendations of the Austrian IIH network (AN4IH) by Gabriel Bsteh, Berthold Pemp, Wolfgang Marik, Klaus Novak, Michael Leutner, Stefan Leis, Gregor Broessner and in Cephalalgia
Footnotes
Acknowledgments
We thank all the AN4IH contributors for helping to create this consensus. The named individuals were not compensated for their help.
Group information Austrian Network for Idiopathic Intracranial Hypertension (AN4IH)
AN4IH investigators in alphabetical order: Aminfar, Haleh (Department of Ophthalmology, Medical University of Graz, Graz, Austria); Broessner, Gregor (Department of Neurology, Medical University of Innsbruck, Innsbruck, Austria); Bsteh, Gabriel (Department of Neurology, Medical University of Vienna, Vienna, Austria); Eppinger, Sebastian (Department of Neurology, Medical University of Graz, Graz, Austria); Ganser, Bernhard (Department of Neurology, Paracelsus Medical University of Salzburg, Salzburg, Austria); Georgi, Thomas (Department of Ophthalmology, Medical University of Graz, Graz, Austria); Grams, Astrid (Department of Neuroradiology, Medical University of Innsbruck, Innsbruck, Austria);
Hatipoglu Majernik Gökce (Department of Neurosurgery, Paracelsus Medical University of Salzburg, Salzburg, Austria); Hochfellner, Daniel (Division of Endocrinology, Department of Internal Medicine, Medical University of Graz, Graz, Austria); Kaya, Emre (Department of Neuroradiology, Paracelsus Medical University of Salzburg, Salzburg, Austria); Keindl, Theresia (Department of Ophthalmology, Paracelsus Medical University of Salzburg, Salzburg, Austria); Kerschbaumer, Johannes (Department of Neurosurgery, Medical University of Innsbruck, Innsbruck, Austria); Leis, Stefan (Department of Neurology, Paracelsus Medical University of Salzburg, Salzburg, Austria); Leutner, Michael (Division of Endocrinology and Metabolism, Department of Internal Medicine 3, Medical University of Vienna, Vienna, Austria); Marik, Wolfgang (Department of Neuroradiology, Medical University of Vienna, Vienna, Austria); Novak, Klaus (Department of Neurosurgery, Medical University of Vienna, Vienna, Austria); Pemp, Berthold (Department of Ophthalmology, Medical University of Vienna, Vienna, Austria); Riedl, Michaela (Division of Endocrinology, Department of Internal Medicine, Medical University of Vienna, Vienna, Austria); Sourij, Harald (Division of Endocrinology, Department of Internal Medicine, Medical University of Graz, Graz, Austria); Stechemesser, Lars (Division of Endocrinology, Department of Internal Medicine, Paracelsus Medical University of Salzburg, Salzburg, Austria); Tschoner, Alexander (Division of Endocrinology, Department of Internal Medicine, Medical University of Innsbruck, Innsbruck, Austria); Velez-Escola Luisa (Department of Ophthalmology, Medical University of Innsbruck, Innsbruck, Austria).
Author contributions
Gabriel Bsteh and Gregor Broessner: drafting of the manuscript, supervision of the guideline development. Berthold Pemp, Wolfgang Marik, Klaus Novak, Michael Leutner and Stefan Leis: critical revision of manuscript for intellectual content, contribution to guideline development. All authors have approved the final version of the manuscript submitted for publication.
Data availability statement
The manuscript does not contain any original data.
Declaration of conflicting interests
Gabriel Bsteh has participated in meetings sponsored by, received speaker honoraria or travel funding from Biogen, BMS, Heidelberg Engineering, Janssen, Lilly, Medwhizz, Merck, Neuraxpharm, Novartis, Roche, Sanofi, Teva and Zeiss, and also received honoraria for consulting Adivo Associates, Biogen, BMS, Janssen, Merck, Novartis, Roche, Sanofi and Teva. He has received unrestricted research grants from BMS and Novartis. He serves on the Executive Committee of the European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS) and the Board of Directors of the International Multiple Sclerosis VisualSystem Consortium (IMSVISUAL).
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Berthold Pemp has received honoraria for speaking and consulting from Novartis.
Wolfgang Marik declares no conflict of interest relevant to this study.
Klaus Novak declares no conflict of interest relevant to this study.
Michael Leutner has received honoraria and research grants from: Abbott, Astra Zeneca, Bayer, Boehringer-Ingelheim, Daiichi Sankyo, Novartis and Novo Nordisk.
Stefan Leis has participated in meetings sponsored by, received speaker honoraria or travel funding from Allergan AbbVie, Lilly, Merz, Neuraxpharm, Novartis, Pfizer and TEVA, and also received honoraria for consulting Allergan AbbVie, Lilly, Lundbeck and Orion.
Gregor Broessner has participated in meetings sponsored by, received speaker honoraria or travel funding from Lilly, Novartis, Pfizer, Organon, Lundbeck, Allergan and Teva, and also received honoraria for consulting Lilly, Novartis, Pfizer, Organon, Lundbeck and Teva. He has received unrestricted research grants from TEVA.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
