Abstract
Background: The role of cerebrospinal fluid (CSF) diversion in treating idiopathic intracranial hypertension (IIH) is disputed.
Method: We conducted a 10-year, retrospective case note review to evaluate the effects of CSF diversion in IIH. Symptoms, signs and details of shunt type, complications and revisions were documented at baseline, 6, 12 and 24 months post-operatively.
Results: Fifty-three IIH patients were shunted [predominantly lumboperitoneal (92%)]. The most common symptom pre-surgery was headache (96%). Post-operatively, significantly fewer patients experienced declining vision and visual acuity improved at 6 (p = 0.001) and 12 months (p = 0.016). Headache continued in 68% at 6 months, 77% at 12 months and 79% at 2 years post-operatively. Additionally, post-operative low-pressure headache occurred in 28%. Shunt revision occurred in 51% of patients, with 30% requiring multiple revisions.
Conclusion: We conclude that CSF diversion reduces visual decline and improves visual acuity. Unfortunately, headache remained in the majority of patients and low-pressure headache frequently complicated surgery. Over half of the patients required shunt revision with the majority of these requiring multiple revisions. We suggest that CSF shunting should be conducted as a last resort in those with otherwise untreatable, rapidly declining vision. Alternative treatments, such as weight reduction, may be more effective with less associated morbidity.
Keywords
Introduction
Idiopathic intracranial hypertension (IIH) is characterised by elevated intracranial pressure (ICP) in the absence of intracerebral space occupation or venous sinus thrombosis (1). The aetiology of IIH is not fully characterised (2–4). The condition is associated with significant morbidity from chronic headaches and visual loss which is severe and permanent in up to 25% of cases (5). The overall incidence of IIH is 1–3 per 100,000, but among female patients with obesity the incidence rises to 20 per 100,000 (2).
The evidence base for treatment in IIH is limited. The Cochrane Database of Systematic Reviews 2005 concluded that there was insufficient evidence to determine which treatments were beneficial and which could have harmful outcomes (6). Over 90% of IIH patients are obese (7) and the efficacy of therapeutic weight loss has been recently demonstrated (8). Difficulties in achieving compliance with dietary programmes are, however, recognised. Medical therapy such as diuretics and carbonic anhydrase inhibitors (acetazolamide) aim to reduce CSF secretion, but their efficacy is unclear (9). In patients with declining visual function, despite medical therapy, surgical CSF diversion or decompression of the optic nerve sheath with a fenestration procedure have been employed. In some centres, CSF diversion is also used to manage chronic headaches in IIH despite there being no immediate threat to vision (10). Reduction in ICP has also been demonstrated following intracranial venous shunting, in cases of venous sinus stenosis associated with IIH (11).
Reducing ICP by surgical CSF diversion is achieved most commonly by lumboperitoneal shunting (LPS) (87%) (12), reflecting the minimally invasive nature of this type of surgery. Ventricular shunts, such as ventriculoperitoneal shunts (VPSs), ventriculoatrial shunts and ventriculojugular shunts are less frequently utilised (9%, 2% and 2%, respectively) due to the potential difficulty of placing the proximal catheter in the normally small or even slit-like ventricles of IIH patients (12,13).
Early reports hailed CSF shunting as the treatment of choice in IIH and claimed superior efficacy to medical treatment (13). Certainly, symptomatic improvement in IIH is invariably documented in those with functioning shunts (10) and low initial morbidity is reported following surgery (10). There are, however, conflicting reports of shunt failure and revision rates. This may reflect the small size of previous cohorts studied (17–37 subjects) (10,12,14–17). Additionally, the number of patients enjoying visual recovery, typically the principal indication for CSF diversion in IIH, is very variable, between 10 and 71% (15,16). Later shunt failure is reported to occur in up to 77% of patients usually reflecting shunt obstruction (65% of revisions) or overdrainage, causing low-pressure headaches (15% of revisions) (12). Less commonly, lumbar radiculopathy, shunt migration, disconnection and abdominal discomfort necessitate shunt revision (10).
There is a current lack of consensus as to the benefit of CSF diversion in IIH. Additionally, the complication rates, an important factor when appraising an invasive therapy, are disputed. To help resolve this debate we sought to characterise the effects of CSF diversion in a large cohort of IIH subjects.
Method
We retrospectively studied the medical records of all patients with IIH who had undergone CSF shunting over a 10-year period (April 1998–April 2008) at the University Hospital Birmingham NHS Foundation Trust, Birmingham, UK. IIH was diagnosed according to the updated modified Dandy criteria (1), with all patients having normal MRI and normal magnetic resonance venography (MRV). Subjects with alternative aetiology to explain the elevated ICP were excluded (e.g. hydrocephalus, malignancy and cerebral venous sinus thrombosis).
Demographic details including age, gender, ethnicity, weight, lumbar puncture (LP) pressure and drug history (acetazolamide and other diuretic therapy) were recorded prior to initial shunt insertion. The time from diagnosis of IIH to initial shunt insertion was recorded. Patients were reviewed at neuro-ophthalmology and neurology clinics at baseline (pre-shunt) and at 6, 12 and 24 months post-shunting. The presence or absence of symptoms [visual deficit (indicating a subjective loss or distortion of the visual field or acuity), diplopia, visual obscurations and tinnitus] was documented. Headache occurrence and phenotype were noted. Visual loss was further categorised into those who were improving, stable or deteriorating. Examination findings of papilloedema (presence or absence), logarithm of the minimum angle of resolution (logMAR) visual acuity and Ishihara colour vision were recorded. Abnormality on visual field assessment (Humphrey 24-2 and Goldman perimetry) was noted. Missing data were documented.
Statistical analysis
Statistical analysis was performed using SPSS version 15 (SPSS, Inc., Chicago, IL, USA) and Prism for Windows version 5.0 (GraphPad Software, Inc., La Jolla, CA, USA) software packages. Categorical data were analysed using the McNemar's test. Numeric data were analysed using a paired t-test for parametric data and the Wilcoxon signed-rank test for non-parametric data. Early and late shunted patients were compared using a Mann–Whitney U test (non-parametric data). Data were summarised using means and standard deviations or medians and ranges. Missing data were excluded from the analysis and paired analyses were performed on individuals with data at both time points. The level at which the results were judged significant was p < 0.05. Right and left eye data correlated significantly, therefore right eye data are quoted.
Results
Subject characteristics

LP: lumbar puncture, LPS: lumboperitoneal shunt, VPS: ventriculoperitoneal shunt. aMean ± standard deviation. bType of primary shunt inserted. cMedian and range.
Characteristics pre- and post-shunting

LogMAR: logarithm of the minimum angle of resolution.
Patients with a visual deficit who described ongoing visual deterioration. bMean ± standard deviation (number of patients). P values indicate change from baseline (0 months). Dichotomous variables were analysed with a McNemar's test. Non-parametric continuous variables were analysed with a Wilcoxon signed-rank test.
Prior to surgery, established visual loss was reported in 77% of subjects and 65% of these described the visual loss as deteriorating. The majority of patients (91%) had more than one symptom prior to surgery and only one patient was shunted exclusively for headache symptoms (2%). Prescription of medical therapy for IIH was recorded in 83% of patients prior to shunting. Acetazolamide therapy was most frequently prescribed (66%) for a mean of 17.0 ± 17.6 months (median 12.0, range 0.5–72.0 months) prior to surgery.
The lumboperitoneal shunt was the most common primary shunt inserted (92%). The number of patients describing visual deficits (77%) did not significantly alter following shunt insertion (63% at 6 months, p = 0.375; 55% at 12 months, p = 0.289; and 30% at 24 months, p = 0.125). In contrast, those patients with deteriorating visual deficits (65% at baseline) significantly improved at 6 and 12 months following shunt insertion (23%, p = 0.004 and 22%, p = 0.021, respectively) although by 24 months this was no longer significant (50%, p = 0.219) (Figure 1A). There was a significant reduction in those patients describing visual obscurations (78% at baseline) at 6, 12 and 24 months (24%, p = 0.002; 25%, p < 0.001; and 32%, p = 0.004, respectively) (Figure 1B). Tinnitus showed a non-significant reduction following shunt surgery [80% affected at baseline, 19% at 6 months (p = 0.125), 33% at 12 months (p = 1.000) and 57% at 24 months (p = 1.000)], possibly reflecting poor documentation. Diplopia did not significantly improve (34% at baseline; 11% at 6 months, p = 0.375; 30% at 12 months, p = 0.272; and 25% at 24 months, p = 1.000).
Symptoms and signs following shunt surgery: (A) visual deficit, (B) visual obscurations, (C) headache, (D) papilloedema and (E) visual acuity. The p values indicate change from baseline: *p < 0.05, **p < 0.01 and ***p < 0.001.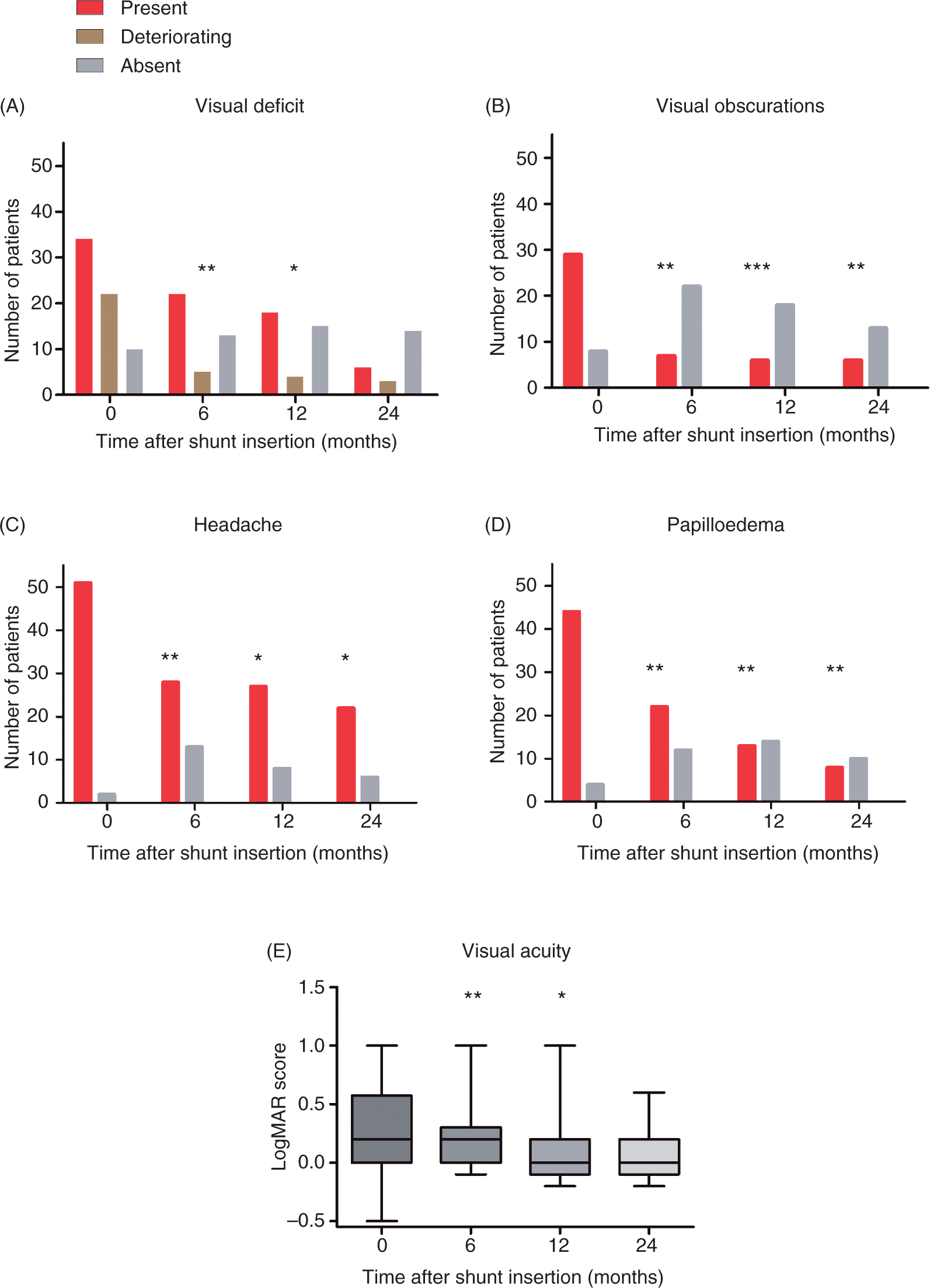
Importantly, there was a significant reduction in the number of patients experiencing headache, attributed to raised ICP (96% symptomatic at baseline; 63% at 6 months, p = 0.002; 77% at 12 months, p = 0.016; and 79% at 24 months, p = 0.031, respectively) (Figure 1C). Of concern, 28% of patients (15 out of 53) suffered with low-pressure headaches as a result of shunt surgery, all these cases having undergone LPS.
Fundus examination showed a significant reduction in the number of patients with papilloedema from 92% at baseline to 65% (p = 0.002) at 6 months, 48% (p = 0.003) at 12 months and 44% (p = 0.008) at 24 months after shunt surgery (Figure 1D). The best corrected LogMAR visual acuity (Figure 1E) significantly improved from 0.31 ± 0.36 at baseline to 0.21 ± 0.31 at 6 months (p = 0.001) and 0.10 ± 0.30 at 12 months (p = 0.016). A one-line improvement on the LogMAR chart equates to 0.1. By 24 months the significant improvement in LogMAR score was lost (score 0.08 ± 0.25, p = 0.109). No improvement in Ishihara colour vision was noted post-shunting (90 ± 19% correctly identified at baseline, 91 ± 21% correct at 6 months (p = 0.574), 79 ± 34% correct at 12 months (p = 0.340) and 87 ± 28% correct at 24 months (p = 0.715). Changes in perimetry could not be quantified due to missing data.
Primary shunt insertion occurred at a mean of 14.6 ± 38.8 months (median 6.0, range 0.0–279.0 months) following the diagnosis of IIH with 17 patients undergoing shunt insertion within 1 month of diagnosis (shunted early). Patients who were shunted early had similar baseline characteristics and symptoms to those shunted after 1 month of diagnosis (delayed shunting). The outcome in those shunted early was not statistically different to those with delayed shunting with the exception of deteriorating vision, which was significantly more common among those shunted early [30% vs. 17% (early vs. late shunts) at 6 months, p = 0.013 and 38% vs. 10% at 12 months, p = 0.004]. Visual deficits were also more common among those shunted early [91% vs. 50% (early vs. late shunts) at 6 months, p = 0.056 and 89% vs. 41% at 12 months, p = 0.040]. For these results, insufficient data at 24 months prevented meaningful analysis.
The mean weight at the preoperative assessment was 102.6 ± 26.8 kg. Following surgery, despite dietary advice, there was no significant decrease in weight (110.5 ± 31.6 kg at 6 months, p = 0.352; 93.7 ± 11.7 kg at 12 months, p = 0.463; 101.5 ± 14 kg at 24 months, p = 0.080).
Shunt revisions

Median (range), n indicates number.
The primary reason for revision surgery was shunt blockage (44.4%) and 22.2% of patients had more than one indication for revision (Table 3). There were no recorded cases of optic nerve sheath fenestration carried out during the study period.
Discussion
We have carried out the largest (n = 53) retrospective study of IIH patients undergoing CSF diversion to date, to more accurately ascertain the benefits and limitations of surgery. The most common symptoms in patients being shunted were headaches (96%), tinnitus (80%) and visual obscurations (78%). CSF shunting significantly improved papilloedema, visual obscurations and visual acuity. While the number of patients with visual deficits did not reduce after surgery, the number with ongoing visual deterioration did decrease significantly. This may indicate that the visual deficits which were not improved by surgery reflect irreversible optic nerve damage.
Conversely, actively declining vision prior to surgery significantly improved, potentially indicating reversible optic nerve damage. These data suggest that progressive visual deterioration is a more treatable symptom than an established visual deficit and should therefore guide the decision to proceed to shunting. Importantly, visual acuity did show a significant improvement following shunting, indicating the site restoring potential of this procedure.
Although headache attributed to elevated ICP was reduced by shunting procedures, low-pressure headaches (affecting 28% of subjects) represented a significant source of morbidity post-shunting. Low-pressure headaches, secondary to shunt overdrainage, occurred exclusively in those patients with LPS. Programmable, switchable or otherwise adjustable valves, which enable the rate of CSF drainage to be modified for the individual patient, may potentially reduce low-pressure headaches (18–20). Additionally, morbidity from post-LP shunt cough headache may diminish if a VPS is utilised in those with cerebellar tonsillar herniation, identified preoperatively (21). Seventy-nine per cent of patients continued to complain of headache 2 years post-shunting, further highlighting the limitations of CSF shunting in managing headache. Persistent headache following treatment for IIH has been previously noted (67% of subjects) and likely represents a range of headache phenotypes, including pain attributed to high CSF pressure, migraine and analgesic overuse headache or potentially a combination of factors (22,23). Further headache characterisation and treatment evaluation is needed to improve management of this disabling symptom in IIH.
Our data highlighted that the timing for shunt insertion, following a diagnosis of IIH, was very variable (median 6 months with a range of 0–279 months). Interestingly, patients who were shunted early (within 1 month of diagnosis) were more likely to describe ongoing visual deterioration than those shunted after 1 month of diagnosis. This potentially reflects a group of IIH patients with a more aggressive disease course, rather than the ineffectiveness of shunt insertion.
Despite the improvements in vision noted following shunting, over half of the patients required shunt revision (51%) and 30% required multiple revisions. In previous reports, shunt revision occurred in 77% of IIH patients (12). Despite this, our revision rate remains high and commits many patients to multiple operations. The revision rate may be lowered if the underlying IIH were effectively treated following primary shunt surgery, consequently preventing the need for further surgery, i.e. primary shunt insertion is used to ‘buy time’ to successfully treat the underlying condition. The effectiveness of acetazolamide is debated (9), but weight loss is of proven benefit (8) and should be the focus of therapy post-shunting. Akin to other studies (10) we found that shunt revision typically occurred within a year of initial shunt surgery (86%).
We acknowledge that the study was not controlled or prospectively designed and this limits interpretation. Additionally, patient loss to follow-up and incomplete medical documentation reduced the data set collected and may have influenced the results. Further, detailed prospective studies of shunting in IIH are needed to clarify at what stage of disease deterioration patients should be shunted. Additionally, the risks and benefits of CSF shunting need to be compared to cerebral venous sinus stenting in IIH (11).
In summary, our data highlight that shunting is worthwhile to arrest progressive visual deterioration and improve visual acuity in IIH. Shunting does not restore established visual loss. The frequent need for multiple shunt revisions and the poor outcome for managing headache are, however, a significant disadvantage. We suggest that shunt surgery should be reserved for those patients with visual decline that is so rapid that time does not allow alternative therapeutic strategies, such as weight loss, or alternatively should be conducted when other therapies have failed. As shunt lifespan is often less than a year, the therapeutic focus following primary shunting should be to treat the underlying IIH, principally by encouraging and facilitating weight loss, to prevent the need for future shunt surgery. We recommend that headache should not be the sole indication for shunting. Headache is typically a major concern for IIH subjects but, in view of the high frequency of low-pressure headaches and the large number of patient still complaining of headache 2 years post-shunting, shunting to treat headache is unlikely to be successful and this should be made clear to patients.
Footnotes
Funding
This study was funded by the West Midlands Neuroscience Teaching and Research Fund, UK; Alexandra Sinclair is a MRC Clinical Training Fellow (London, UK).
