Abstract
Background
Neck pain is common in migraine patients, occurring during all migraine phases and between attacks. It can be a migraine symptom, trigger or a coexisting condition, and is associated with greater disability and poorer treatment response. There is evidence that neck pain associated with headaches can be frequently incorrectly diagnosed as a cervical disorder rather than migraine, resulting in a lack of appropriate treatment. Accurately assessing the connection between neck pain and migraine is crucial for effective treatment.
Methods
This narrative review aims to summarise existing research on the role and contribution of neck pain in migraine, both as a symptom and a trigger, and outlines future research needed to deepen our understanding of this relationship. It also proposes a structured approach for assessing neck pain in migraine and a treatment algorithm, offering guidance for clinical evaluation and treatment. For this purpose, a comprehensive narrative review was conducted using PubMed, covering preclinical, clinical, neurophysiological and imaging evidence on migraine and neck pain.
Results
Migraine patients frequently exhibit cervical dysfunction, tenderness and altered posture, with overlapping neuroanatomical pathways of the neck and trigeminal systems, suggesting shared mechanisms of nociception and migraine initiation. Clinical assessment involves a thorough history, physical exam and exclusion of secondary causes. Standard migraine therapies, such as amitriptyline and onabotulinumtoxinA, may help reduce neck pain and non-pharmacological treatments, such as physical therapy, acupuncture and behavioural strategies, show some promise. However, evidence on neck pain relief is limited.
Conclusions
Accurately distinguishing whether neck pain is a symptom, trigger or comorbid condition in migraine is essential for guiding effective treatment strategies. Both pharmacological and non-pharmacological approaches may help manage migraine-associated neck pain. However, few studies have assessed the effects of acute or preventive migraine therapies, particularly calcitonin gene-related peptide-targeted treatments, on neck pain, highlighting a significant gap in our current knowledge. Future research should evaluate the effectiveness of these therapies on neck pain, both alone and in combination with non-pharmacological interventions.
This is a visual representation of the abstract.
Introduction
Neck pain is highly prevalent in migraine patients and can be present in any phase of a migraine attack, including the prodrome, headache and postdrome, and interictally between attacks (1,2). Neck pain may arise as a symptom of migraine or as a result of comorbid conditions and is associated with higher levels of migraine disability, poorer pharmacological treatment prognosis and a higher frequency of migraine attacks (3,4). Afferent nociceptors from the trigeminovascular system and upper cervical nerves converge on the second-order neurons in the trigeminocervical complex (TCC) (5). This shared neuroanatomy supports a pathophysiological link between neck pain and migraine (Figure 1).

Shared neuroanatomical and pathophysiology of migraine and neck pain. The trigeminocervical complex (TCC) consists of the trigeminal nucleus caudalis, which receives inputs from the trigeminal ganglia (TG), and the trigeminocervical nucleus, which receives convergent inputs from the TG and the C1–C3 dorsal root ganglia (DRG). Sensory information, such as nociception, is transmitted to the thalamus by second-order neurons in the TCC via the spinothalamic tract. The TG and upper cervical (C1–C3) DRG innervate the musculature, vasculature, and meninges of the head, including the greater occipital nerve (GON). Additionally, the upper cervical DRG innervate the neck muscles.
Identifying the origin of neck pain is crucial for accurate diagnosis and effective treatment. Patients often assume that neck pain is the cause of their headaches, leading to an underdiagnosis of migraine (6,7). Additionally, neck pain can trigger or contribute to the worsening of attacks (4,8). Similarly, migraine can worsen comorbid neck conditions (9). A clearer understanding of neck pain in the context of migraine is essential to guide clinical assessment and improve patient outcomes.
Recently, there has been a push to improve our understanding of neck pain as a migraine symptom and trigger, with several systematic and narrative reviews highlighting the frequency and severity of neck pain in patients with migraine and the gaps in our knowledge of its pathophysiology and treatment (1,10,11). Therefore, this review examines epidemiological studies of neck pain in migraine, debates neck pain as a symptom or trigger and provides practical guidance for clinical evaluation and treatment recommendations.
Search strategy and selection criteria
This is a narrative literature review. The reviewers were divided into three groups to cover the extensive literature and focus on their respective areas of expertise, working independently to perform literature searches from the PubMed database using the search term “migraine” and “neck pain” or “cervical disability” in combination with the terms “epidemiology”, “prevalence”, “premonitory symptom”, “prodrome”, “trigger”, “pathophysiology”, “assessment” or “treatment”. Additionally, papers from references cited in relevant articles were identified. No publication date restrictions were applied, but original articles published in the last 10 years were preferentially selected. Reviews were selected when space did not allow for a more comprehensive discussion. Letters to the editors and articles published in languages other than English were excluded. Each group selected the relevant articles and resolved the conflicts with each other through serial discussions. TR and EC supervised the main working groups. After the initial round of discussions, all researchers evaluated the full texts of the relevant studies in detail regarding research quality and generated the exact study reference list. The senior reviewers (PPR and EC) approved the final reference list.
Epidemiology
Neck pain is a common symptom among individuals with primary headache disorders, including migraine and tension-type headache. In a population-based cross-sectional study conducted by Ashina et al. (12), the one-year prevalence of neck pain was significantly higher in patients with migraine (76.2%) and in those with migraine and tension-type headache (89.3%) compared to individuals without headache (p < 0.05). Similarly, a systematic meta-analysis by Al-Khazali et al. (1) reported a pooled relative frequency of neck pain of 77.0% (95% confidence interval (CI) = 69.0–86.4) in the migraine group, compared to 23.2% (95% CI = 18.6–28.5) in non-headache controls. Neck pain was more frequent in patients with chronic migraine (87.0%, 95% CI = 77.0–93.0) than in those with episodic migraine (77.0%, 95% CI = 69.0–84.0) (1). However, despite this high prevalence, Liang et al. (7) identified cervical musculoskeletal dysfunction in only approximately 40% of migraine patients, suggesting a multifactorial origin of neck pain in migraine.
Patients with migraine experience neck pain in 39.7% of their attacks (13). In a prospective observational study by Calhoun et al. (14), 113 patients with episodic or chronic migraine were asked to record symptoms associated with their migraine attacks. Exclusion of patients with cervicogenic headache and/or fibromyalgia minimised potential confounding. Neck pain emerged as a more frequently reported symptom than nausea (p < 0.0001), regardless of the intensity of headache pain at the time of treatment. A positive linear correlation was identified between attack frequency and the presence of neck pain (r = 0.32) (14). Chronic migraine was also associated with an increased risk of disability due to neck pain, as assessed by the Neck Disability Index, compared to episodic migraine, with relative risks of 2.5 (95% CI = 1.1–6.1) for mild, 3.7 (95% CI = 1.5–8.8) for moderate and 5.1 (relative risk = 5.1; 95% CI = 2.1–11.9) for severe disability related to neck pain (3). Finally, a multi-country cross-sectional study found that patients with migraine who experience neck pain during their attacks report higher prevalence rates of depression (40.2% vs. 28.2%, p < 0.001) and anxiety (41.2% vs. 29.2%, p < 0.001) compared to those without neck pain. They also experience greater work productivity losses and lower quality of life (p < 0.001) (15). It is currently unclear whether there is a causal relationship between neck pain and anxiety, depression and productivity; however, the findings suggest that neck pain could contribute to impaired emotional and social well-being. Therefore, effective management of neck pain could have a significant impact on the overall quality of life in individuals with migraine
Pathophysiology
Premonitory symptom or trigger?
Neck pain or stiffness commonly precedes the headache phase of migraine; however, its role as either an extracranial premonitory symptom or a migraine attack trigger is debated. Neck pain or stiffness is a frequently reported premonitory symptom of spontaneous migraine attacks, with prevalence estimates ranging from 6.9% to 49.7% (16–21). The prevalence of premonitory neck pain varies by study design. Prospective studies using headache diaries have reported higher rates (41.9–49.7%) of neck pain or stiffness as a premonitory symptom (19,20), whereas retrospective questionnaires or interviews where patients were prompted to recall premonitory symptoms, including neck pain and stiffness, show lower prevalence rates (6.9–35%) (16,17,21,22). For example, in a large multicentre retrospective study in China, only 6.9% of 4821 migraine patients reported neck stiffness as a premonitory symptom (21). The observed discrepancy underscores the potential limitations of retrospective studies, which may underestimate symptoms due to recall bias. Furthermore, neck stiffness is also a frequent premonitory symptom in pharmacologically triggered migraine attacks, with a prevalence ranging from 27.5% to 61% (23–26). However, these studies lack placebo controls, and it is unclear whether neck stiffness is a direct consequence of pharmacological provocation or would occur independently.
As a premonitory symptom, neck pain is reported by migraine patients to present as stiffness or cramp-like (26.5% of patients) or electric shock-like (21.1%) pain, which is felt at the upper cervical region (95.2%) and is unilateral (82.5%) (18). It often occurs within the two hours preceding the headache phase and is more common in migraine without aura than with aura (16,17). This differs from mechanical neck pain, which tends to be present in the cervical spine and shoulder areas and is provoked or aggravated by movements of the neck and palpation of the cervical muscles (27). Additionally, studies indicate that 50–60% of migraine patients with neck pain cannot be attributed to co-morbid cervical musculoskeletal disorders (1,7). Therefore, in combination with the difference in presentation, it is likely that neck pain that occurs during the premonitory and headache phases of a migraine attack arises from central sensitisation, rather than musculoskeletal dysfunction.
Neck pain is also frequently reported as a migraine trigger. In a retrospective survey of 1207 migraine patients, 38.4% self-identified neck pain as an occasional trigger and 10.6% as a frequent trigger (28). Migraine attacks in the occipital and neck regions are significantly correlated with neck pain as a self-reported trigger (13). However, patients may overestimate this association. For example, although 80% of participants in one study strongly endorsed neck pain as a migraine trigger, only 38.7% experienced it before headache onset (29).
Experimental evidence supports a potential triggering role for neck pain. Endurance tests targeting neck flexor and extensor muscles induced migraine-like headaches in 45% of migraine patients, compared to 16% of controls, with an average onset of 18.5 hours post-exercise (30). Similarly, manual palpation of myofascial trigger points in the medial temporal, suboccipital, occipital and superior trapezius regions provoked migraine-like attacks in 29.6% of patients (31). Furthermore, stimulating the C1 nerve evokes periorbital and frontal pain in migraine patients but occipital pain in non-migraine controls; however, no quantification or comparison of pain intensity was performed (8). Injection of hypertonic saline into the trapezius muscles of headache-free controls and low-frequency electrical stimulation of the C2 nerve of migraine patients evokes pain in the occipital and parietal regions, mirroring migraine location associated with neck pain as a trigger (8,13,32). The induction of migraine-like attacks or pain localisation in migraine patients significantly above headache-free controls is reflective of a sensitised TCC. However, these data suggest that it is possible that activating the TCC via the upper cervical nerves rather than via dysregulation of the ascending/descending pain pathways could contribute to the initiation of a migraine attack.
Furthermore, interventions targeting cervical nerves, such as greater occipital nerve (GON) blockade and electroacupuncture, may modestly reduce headache frequency and intensity (33,34). A meta-analysis of GON blockade in chronic migraine found that it significantly reduced pain intensity (−1.29 visual analogue scale) and number of headache days (−4.45 days), compared to placebo (34). Electroacupuncture of the C2 nerve reportedly reduced the pain intensity (7.4 ± 1.9 to 4.9 ± 2.9, numerical rating scale for pain), Headache Impact Test-6 (64.1 ± 6.3 to 57.8 ± 7.9) and headache days (18 ± 9.0 to 14 ± 10.2) after three months (33). These findings suggest that upper cervical nerve activation could directly contribute to migraine initiation (35). However, it is possible that these procedures act by modulating the TCC to reduce sensitisation, rather than directly preventing upper cervical nerve activation triggering an attack.
Overall, these studies suggest that neck pain could be both a premonitory symptom and a potential migraine trigger mediated by distinct processes (discussed below in “Neuroanatomical and pathophysiological insights”). Further research into the temporal and causal relationships between neck pain and migraine is needed to clarify the contributions of neck pain. Studies systematically comparing the induction and intensity of migraine and pain between migraine patients and headache-free controls following upper cervical nerve activation are needed to determine whether neck pain may act as a trigger. Additionally, placebo-controlled human migraine provocation studies should incorporate the assessment of both premonitory and ictal symptoms, with specific inclusion of neck pain in symptom questionnaires across both phases to better delineate the temporal relationship.
Neuroanatomical and pathophysiological insights
Migraine is associated with higher interictal pericranial and extracranial tenderness, including the neck muscles, with tenderness scores significantly higher in chronic compared to episodic migraine patients (36,37). This sensitivity becomes more pronounced immediately before, during and after migraine attacks (37). Adult migraine patients exhibit reduced pressure pain thresholds in the suboccipital, frontal, temporalis, trapezius, sternocleidomastoid, anterior scalene and levator scapulae muscles during the interictal period, corresponding to migraine laterality (38–41).
Compared to headache-free control groups, migraine patients are reported to demonstrate altered cervical function, including reduced extensor and flexor neck muscle strength (42), increased antagonist muscle co-activation (42), abnormal forward head posture (43), and decreased cervical range of motion and angular velocity (44). Cervical dysfunction correlates with headache frequency, with lower muscle endurance and reduced flexion-rotation test mobility for patients with chronic migraine, compared to episodic, or comorbid chronic neck pain (45,46). However, not all migraine patients who report neck pain experience cervical dysfunction. An Australian cross-sectional study found that episodic and chronic migraine patients with neck pain could be sorted into two clusters: those with cervical function similar to headache-free and neck pain-free controls, and dysfunction comparable to controls with idiopathic neck pain (7). Interestingly, a recent meta-analysis indicates that the level of evidence supporting cervical dysfunction in migraine patients is low and heterogeneous, and may not be clinically detectable or relevant (47). This heterogeneity could be due to the aforementioned proposed two clusters of migraine patients (7). Active myofascial trigger points are also prevalent in migraine patients (93.9%), particularly in the temporal and suboccipital regions, and are predominantly located ipsilateral to the headache side (31,43). These trigger points correlate with anxiety scores and headache intensity but not with attack frequency, duration and chronicity, or whether patients primarily experienced unilateral or bilateral attacks (48). When working with a migraine patient with severe anxiety, consider examining trigger points as part of the assessment. Furthermore, treatments such as physical therapy and relaxation techniques, that target the trigger points, could be beneficial in addressing the migraine symptoms and anxiety, to overall be more effective.
The high prevalence of neck pain and dysfunction in migraine may be due to shared neuroanatomical pathways and overlapping pathophysiological mechanisms (Figure 1). The upper cervical (C1–C3) dorsal root ganglia (DRG) innervate both the neck and pericranial muscles, vasculature and meninges in the posterior of the head (35,49). Sensory inputs from the trigeminal ganglia (TG) and C1–C3 DRG converge in the TCC, express migraine-relevant neuropeptides, like calcitonin gene-related peptide (CGRP) and pituitary adenylate cyclase-activating peptide (i.e. PACAP) (50–52) and synapse with second-order neurons that project to the thalamus via the spinothalamic tract (5). Therefore, activation of these cervical nerves is proposed to contribute to the induction and maintenance of headache alongside neck pain, particularly the afferents like the GON, which innervate the pericranial muscles and posterior meninges (8,32,35). Additionally, rodent studies demonstrate bidirectional interactions between trigeminal and cervical neurons in the TCC, where GON activation increases trigeminal neuron firing and trigeminal afferent stimulation enhances GON C-fibre neuron excitability (53,54). This is consistent with studies in migraine patients, where pressure at the C1–C3 levels reproduces migraine-like head pain (55) and GON blocks reduce trigeminally-mediated nociceptive responses (56). These bidirectional interactions may explain the close temporal relationship between neck pain and migraine onset.
Neck pain that arises as a premonitory symptom and after headache onset is likely a result of central sensitisation (57). Persistent TCC activation by trigeminal afferents induces neuroplastic changes, resulting in hyperexcitable neurons with enlarged receptive fields and lower activation thresholds, leading to efferent-mediated allodynia and hyperalgesia in the neck (53,58). Dysregulation of ascending and descending pain pathways, involving the locus coeruleus and periaqueductal grey, also lowers the activation threshold of TCC neurons and may underlie neck pain during the interictal period and prodromal phases of migraine (59,60). Functional imaging studies have shown increased activation of the spinal trigeminal nucleus, where upper cervical nerves synapse, in the hours preceding a migraine attack, suggesting that premonitory neck pain, like other early migraine symptoms, may result from central sensitisation (61,62). In line with this, migraine patients who experience premonitory neck pain have greater stiffness and reduced pressure pain thresholds interictally, compared to migraine patients who do not have premonitory neck pain (63). Interestingly, migraine patients with neck pain report a significantly higher number of premonitory symptoms (64), suggesting a potentially greater degree of central sensitisation in this subgroup. Altered inflammatory profiles could also play a role. Migraine patients with muscle tenderness have elevated pro-inflammatory (e.g interleukin-6, toll-like receptor 2, interferon beta, chemokine (C-C motif) ligand 2 and 8) and reduced anti-inflammatory (e.g. interleukin-10 receptor subunit alpha and colony-stimulating factor 1 receptor) gene expression in the calvarial periosteum (65).
Overall, neck pain in migraine is likely due to shared neuroanatomical pathways and bidirectional interactions between trigeminal and upper cervical afferents within the TCC, which contribute to both headache induction and maintenance. Central sensitisation, neuroplastic changes in the TCC and altered inflammatory profiles could underlie the presence of neck pain across different migraine phases, particularly as a premonitory symptom. Further research is needed to strengthen our understanding of the exact underlying pathophysiology and delineate the contribution of each of these proposed mechanisms to neck pain in the different migraine phases. Therefore, preclinical studies using inflammatory and central sensitisation models to determine their relative contributions to cervical allodynia and nerve activation are needed.
Clinical assessment
Neck pain is a common symptom experienced across various clinical scenarios that are not necessarily mutually exclusive. Symptoms may occur only in relation to migraine or chronically without relation to the migraine pain itself. The neck pain that patients seek care for versus what pain they may experience on a regular basis may vary based on severity, onset, duration and/or impact. The Neck Pain Task Force classifies neck pain into four grades that may prompt patients to seek health care: Grade I: no signs of major pathology with no or little interference with daily activities; Grade II: no signs of major pathology with interference with daily activities; Grade III: neck pain with neurological signs or symptoms (radiculopathy); and Grade IV: neck pain with major pathology (instability or infection) (66). The Neck Pain Task Force defines the neck as the region from the superior nuchal line and occipital protuberance to the spine of the scapula, superior border of the clavicle and suprasternal notch (66).
The evaluation of neck pain and its relation to migraine requires a careful clinical history and physical exam. After establishing that the patient endorses neck pain, standardised assessment tools can help quantify severity and impact. The numeric pain rating scale (NPRS), an 11-point continuum from 0 (absence of pain) to 10 (most extreme imaginable pain), and neck disability index (NDI) may better elucidate the extent to which this issue needs to be addressed during the patient visit (10,67). Pooled analysis of eight studies found the NPRS of neck pain in individuals with migraine was higher by 4 points compared to headache-free controls, with a mean NPRS higher by 5.7 in those with chronic migraine compared to those with episodic migraine (10). The NDI, the most used questionnaire for neck pain-associated disability, is composed of 10 self-assessed items scored on a point scale from 0 to 5. Higher values signify greater disability, across activities including pain intensity, personal care (washing, dressing, etc), lifting, reading, headache, concentration, work, driving, sleep, and recreation. The NDI disability score breakdown is: 0–4 none, 5–14 mild, 15–24 moderate, 25–34 severe and ≥35 complete (10,68,69). Taking this score into context, the higher the NDI, the higher the potential impact of neck pain on migraine. These measures (NPRS and NDI) are of note, but do not assist in the differential diagnosis for the specific origin of pain.
Neck pain may present as a prodromal symptom before a headache attack (22). During the early headache phase, pain may begin in the occipital and neck region for up to 40% of patients before radiating frontally, although neck pain may also manifest interictally (13). Migraine patients with neck pain may demonstrate increased neck stiffness and reduced pain thresholds in the neck (63). Clinical evaluation should further assess for red flag symptoms, including constitutional symptoms, trauma, infectious symptoms, upper motor neuron lesion symptoms, cardiopulmonary symptoms, rheumatological symptoms and genetic conditions, when appropriate. A comprehensive approach to clinical evaluation of neck pain symptoms in relation to migraine is shown in Figure 2 and medical comorbidities to assess for and rule out when necessary are provided in Table 1 (70–73). Other headache disorders with neck pain to consider are explored in Table 2.

Clinical algorithm for neck pain assessment. When neck pain is identified during evaluation, assess the associated neck pain intensity via the Numeric Pain Rating Scale (NPRS) and degree of disability via the Neck Disability Index (NDI). Assess how the pain is related to migraine as a prodrome or subjective trigger, whether it is an ictal or an interictal phenomenon to help guide management decisions. Of note, neck pain as an interictal phenomenon as it relates to migraine could separately be chronic neck pain unrelated to migraine and should undergo proper assessment and management otherwise. For any red flag symptoms or when suspecting conditions aside from migraine, evaluate for any associated underlying medical comorbidities (Table 1) or alternative differential diagnoses (Table 2).
Differential diagnoses associated with neck pain.

Abbreviations: CT = computed tomography; CTA = computed tomography angiography; CT = magnetic resonance imaging.
Other headache disorders with neck pain.

Abbreviations: CT = computed tomography; CT = magnetic resonance imaging.
Prior to initiation of migraine treatment, it is essential to evaluate for other primary headache disorders with neck pain after excluding secondary causes. This may be done via careful clinical history, physical exam and headache diary (74). Several key headache disorders that warrant consideration include: tension-type headaches, cervicogenic headache, temporomandibular disorder, post-traumatic headache and spontaneous intracranial hypotension (Table 2). Tension-type headache frequently presents with bilateral band-like headache and neck pain with no nausea, photophobia or phonophobia (75). Cervicogenic headache exhibits restricted neck range of motion, pain originating from the posterior occiput, headache provoked by mechanical pressure, ipsilateral shoulder and arm discomfort, and often a male predominance (76). Temporomandibular disorder is characterised by headache, bruxism and neck pain, with tenderness in associated muscle groups and temporomandibular joint (77). Post-traumatic headache is characterised by headaches that develop or worsen within seven days of head injury. Often, there may be accompanying post-concussive symptoms, such as cardiovascular (orthostatic hypotension, dysautonomia), visual disturbances, vestibular symptoms or neck pain (78). Spontaneous intracranial hypotension, though highly misdiagnosed and underdiagnosed, characteristically presents with orthostatic headache, variable in location, with neck pain present in approximately 43% of patients (79). Additional details regarding other headache disorders associated with neck pain are provided in Table 2.
Musculoskeletal disorders, whether spine or the extremities, are characterised at their basic level with, collectively, impaired joint, movement and muscle function. For example, early arthritis of the hip will present with joint pain, restricted motion and weakness of the gluteal muscles. The same occurs in cervical musculoskeletal disorders. Thus, to confirm a cervical musculoskeletal disorder in headache, the physical examination must include tests of movement, tests of segmental dysfunction and tests of muscle function at a minimum (7,80,81).
The physical exam should be guided by understanding the quality, intensity and associated disability of neck pain (82,83). The International consensus on the most useful physical examination tests used by physiotherapists for patients with headache highlight 11 physical exam tests they consider clinically useful: manual joint palpation, the cranio-cervical flexion test, the cervical flexion-rotation test, active range of cervical movement, head forward position, trigger point palpation, muscle tests of the shoulder girdle, passive physiological intervertebral movements, reproduction and resolution of headache symptoms, screening of the thoracic spine, and combined movement tests (84). First, start with basic posture evaluation (Figure 3(a)–(c)), then evaluate the range of motion (Figure 3(d)–(i)) of the cervical spine. Once completed, evaluate the upper cervical flexion and extension with C1–C2 rotation with the flexion-rotation test to assess the TCC. Use of the total tenderness score to evaluate cephalic and neck tenderness may help track musculoskeletal dysfunction. Palpate across the cephalic muscle groups (frontalis, temporalis, coronoid process of the mandible, masseter) and then the neck (mastoid process, sternocleidomastoid, trapezius and neck muscle insertions).

Systematic physical examination techniques for neck pain. The initial observation phase focuses on assessing posture (a–c), with examples of (a) normal posture, (b) forward head posture and (c) thoracic kyphosis. The physician (ST) manipulates the patient's head and neck as depicted. Range of motion (ROM) tests (d–i) are then conducted to identify movement limitations and symptomatic patterns: (d) neck forward flexion, (e) neck extension, (f, g) lateral rotation, and (h, i) lateral flexion/head tilt. Written consent to use personal images from the patient has been obtained. NSAIDs = non-steroidal anti-inflammatory drugs.
Manual palpation should also be performed at the cervical facet joints to assess for upper cervical musculoskeletal dysfunction (7,80,85). Continue on to sensory testing to elucidate the degree of central sensitisation in a patient's pain condition. This may include quantitative sensory testing (i.e. QST) assessing touch, vibration, proprioception, sensitivity to pinprick, pressure and temperature stimuli. Lastly, repetitive stimulation to evaluate a patient's pain response over a short period of time may also reveal the degree of central sensitisation (temporal summation, or wind-up testing) (86).
Treatment
Effects of migraine treatments on neck pain in migraine (acute and preventive)
Several pharmacological agents have demonstrated efficacy for migraine prophylaxis, including beta-blockers, angiotensin receptor blockers, antidepressants, antiepileptics, drugs targeting CGRP pathways and onabotulinumtoxinA (BTX-A). The decision to initiate preventive treatment and the choice of the therapeutic agent is tailored to the individual patient's profile (87). However, data are sparse regarding the efficacy of these treatments in alleviating neck disability.
Currently, there are no data available concerning the relief of neck pain during migraine attacks. Systemic non-steroidal anti-inflammatory drugs (NSAIDs), which are effective for treating acute migraine attacks, have also shown efficacy in managing spinal pain (88). This dual benefit highlights their potential utility for managing neck pain in the context of migraine. While triptans are effective for typical migraine symptoms, their impact on associated neck pain remains uncertain and warrants further investigation (89).
Amitriptyline has shown effectiveness in preventing both episodic and chronic migraine (90). Furthermore, in a randomised clinical trial, it was found to significantly reduce the intensity of idiopathic chronic neck pain (p < 0.0001) (91). While these findings do not allow for definitive conclusions, they suggest a potential avenue for managing neck pain in patients with migraine. BTX-A, widely used for chronic migraine prevention, is injected into 31–39 sites across seven muscle groups in the head and neck following the PREEMPT protocol (92). It has been independently studied for upper back myofascial pain syndrome with conflicting results; however, one randomised trial reported a significant reduction in pain intensity during weeks 5–8 (p < 0.05) and a significant increase in pain-free days per week between weeks 5 and 12 (p = 0.036) (93). In chronic migraine patients specifically, one study including 134 participants showed that BTX-A reduced neck disability from severe to mild (−16.5 points median change, p < 0.001) and halved neck pain intensity (p < 0.001) (94). Despite these encouraging findings, no treatment has demonstrated consistent efficacy for acute neck pain relief during migraine attacks, and additional studies are needed to determine whether existing migraine medications can be tailored to improve associated cervical symptoms. However, these studies suggest that in patients with significant neck pain, consider prioritising agents with dual indications (e.g. amitriptyline, BTX-A) as first-line treatments.
Effects of non-pharmacological treatments on neck pain in migraine
An increasing body of evidence is emerging on the efficacy of non-pharmacological treatments as complements or alternatives to pharmacological approaches in both acute and prophylactic migraine management. These treatments primarily aim to alleviate musculoskeletal tension and cervical hypersensitivity, thereby reducing migraine frequency and severity. Their effectiveness varies according to the individual and the specific nature of the associated neck pain. Common non-pharmacological approaches include physical therapy, acupuncture, behavioural therapies and neuromodulation.
Physical therapy
Physical therapy encompasses a range of non-pharmacological interventions used in migraine management, including aerobic exercise, strengthening/resistance training and manual therapies, which have shown emerging or moderate evidence supporting their effectiveness in both migraine and cervical musculoskeletal dysfunction. Although some studies suggest that exercise may exacerbate pain during migraine attacks, substantial evidence supports its benefits in both acute and preventive migraine treatment (95–97). A meta-analysis involving 508 patients with migraine showed a significant decrease in pain intensity (n = 166; standardised mean difference (SMD) = 1.25; 95% CI = 0.47–2.04), frequency (n = 214; SMD = 0.76; 95% CI 0.32–1.2) and duration of migraine (n = 106; SMD = 0.41; 95% CI 0.03–0.8) with aerobic exercise. A second meta-analysis conducted by Varangot-Reille et al. (97) confirmed significant differences for aerobic training with a moderate clinical effect for pain intensity (n = 233; SMD = −0.65; 95% CI = −1.07 to −0.22) and headache frequency (n = 288; SMD = −0.72; 95% CI = −1.22 to −0.23). Benefits have been observed when aerobic exercise is implemented either alone or alongside pharmacological treatment. A randomised controlled trial involving 52 individuals with migraine, co-existing tension-type headache and neck pain reported significant reductions in pain intensity in the aerobic exercise group (p = 0.045) (95). The analgesic effects of exercise are considered to be mediated through activation of endogenous opioid and endocannabinoid systems (98).
Other physical therapy interventions focus more specifically on the cervical and shoulder regions and may alleviate pain through both neurophysiological and local neuromuscular mechanisms (98). These include modulation of nociceptive input within the trigeminocervical complex and local adaptations that enhance muscle function and reduce peripheral sensitisation (98). Within this framework, strength/resistance training has been investigated, but its efficacy remains uncertain due to conflicting findings (84). A randomised controlled trial investigating an eight-week craniocervical muscle-strengthening program in 42 migraine patients found no significant improvement in migraine-related outcomes (99).
The role of spinal manipulation therapy (SMT) is also debated (97,100). A systematic review of randomised clinical trials provided moderate evidence supporting SMT in reducing the frequency of chronic headache, suggesting SMT may be a potential therapeutic option for migraine, cervicogenic headache and chronic tension-type headache (100). Nevertheless, the data were too heterogeneous in terms of clinical characteristics, control groups and outcome measures to draw strong conclusions.
Yoga therapy, which includes postures that stretch the neck, shoulders and back muscles, has shown promise. A study that randomised 72 individuals with migraine to a yoga intervention demonstrated reductions in pain frequency(p < 0.001) and intensity (p < 0.001), migraine burden, and improvement in quality of life (101). Despite promising results, the small sample sizes and the presence of co-existing headache conditions, such as tension-type headache, limit the ability to generalise these findings.
Overall, aerobic exercise should be recommended in most patients due to its robust evidence base and dual benefit on migraine and neck pain. Manual therapies and yoga may be considered in individuals with significant cervical involvement or coexisting tension-type headache. Interventions targeting the cervical region should be tailored based on symptom profile, comorbidities, response to initial treatment, and should be performed by experts as they can cause worsening of symptoms.
Acupuncture
Acupuncture has also proven beneficial for both migraine management and non-specific musculoskeletal pain, including back and neck pain. A large meta-analysis involving 4985 migraine patients reported a significant reduction in migraine frequency compared to no acupuncture (n = 2199; SMD −0.56; 95% CI −0.65 to −0.48) and compared to sham acupuncture (n = 1646; SMD −0.18 95% CI −0.28 to −0.08; I2 = 47%), with moderate quality evidence (102). Acupuncture also reduced migraine frequency significantly more than drug prophylaxis after treatment (n = 739; SMD −0.25; 95% CI −0.39 to −0.10), but the significance was not maintained at follow-up (102). However, specific studies focusing on the effects of acupuncture on migraine-related neck pain remain limited. Additionally, the variability in acupuncture protocols may lead to inconsistent results. Furthermore, the small sample sizes and lack of blinding in some studies may limit the generalisability and strength of the findings.
Behavioural therapies
Behavioural therapies involve psychological approaches designed to help patients manage pain by addressing stress, anxiety, coping strategies and pain perception. Common methods include cognitive-behavioural therapy (CBT), relaxation training and mindfulness-based interventions. Their effectiveness in migraine remains heterogeneous, with limited and sometimes contradictory data (103). Nonetheless, emerging evidence suggests that these approaches can improve quality of life and reduce disability. A more recent meta-analysis showed reduction of migraine/headache attack frequency for CBT (n = 839, SMD −0.33; 95% CI −0.55 to −0.11), relaxation (n = 1091, SMD −0.31; 95% CI −0.50 to −0.11) and mindfulness-based therapies (n = 227, SMD −0.30; 95% CI −0.54 to −0.05) but with a low strength of evidence (104). However, the specific response to behavioural interventions on neck pain has not been specifically assessed. Additionally, mindfulness practices have been proposed as valuable tools for managing chronic pain. A prospective study of mindfulness-based interventions in 133 patients with chronic pain, including migraine and neck pain, showed significant improvements in pain intensity and functional limitations. However, patients with chronic headache experienced the smallest improvements compared to those with other pain conditions (105). Consider behavioural interventions in patients with high levels of stress or anxiety contributing to neck pain as migraine symptom.
Neuromodulation
Various neuromodulation techniques have shown promising efficacy in both acute and preventive treatment of migraine (106). Specifically, remote electrical neuromodulation (i.e. REN) was effective for acute migraine treatment, with a large effect size (relative risk = 2.14, 95% CI = 1.34–3.40). For prevention, transcutaneous supra-orbital electrical nerve stimulation (i.e. TENS) and high-frequency transcranial magnetic stimulation (i.e. TMS) applied to the left primary motor cortex showed, respectively, a small (−0.494, 95% CI = −0.799 to −0.188) and a moderate effect size (−0.533, 95% CI = −0.940 to −0.126). Despite these results, their clinical applicability remains limited due to heterogeneity in stimulation parameters, and methodological concerns including variable blinding and outcome reporting (106). Although these approaches may reduce migraine frequency and intensity, specific evidence addressing their efficacy in treating the neck-related components of migraine is currently lacking. Neuromodulation can be considered in migraine management when pharmacological options are limited but should not be used specifically to target neck pain until further evidence is available.
Practical integration and clinical decision-making
Figure 4 presents a clinical algorithm to guide treatment selection for neck pain in the context of migraine. It integrates both pharmacological and non-pharmacological options.
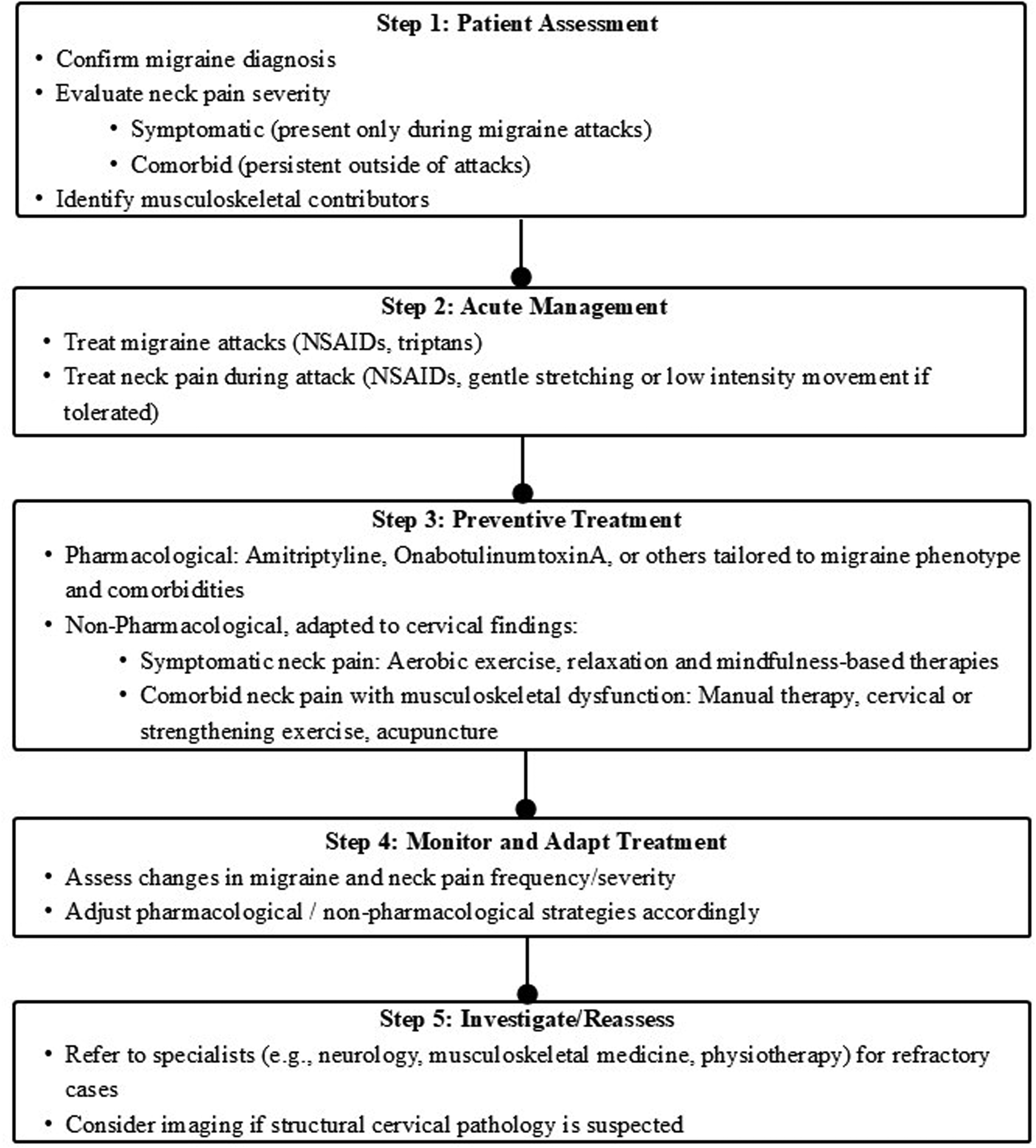
Algorithm for neck pain treatment.
Unmet needs
The interplay between migraine and neck pain suggests the potential existence of two overlapping subgroups of patients in whom these conditions coexist but arise from different underlying mechanisms. The first subgroup may include patients for whom neck pain is primarily a symptom of migraine, driven by central mechanisms such as trigeminocervical complex sensitisation and migraine-related nociceptive activation. In these patients, neck pain is closely linked temporally to migraine attacks and may respond predominantly to migraine-specific treatments. The second subgroup may comprise patients with migraine who have a comorbid cervical musculoskeletal dysfunction, such as joint hypomobility, muscle tenderness or altered cervical motor control, that may independently contribute to neck pain. In this group, neck pain may persist beyond migraine attacks and requires targeted musculoskeletal assessment and management.
Accurate identification of these subgroups demands comprehensive clinical evaluation, including detailed history taking to characterise the temporal relationship of neck pain with migraine attacks, physical examination assessing cervical joint and muscle function, and evaluation of central sensitisation markers. Patients in this subgroup may present with:
Persistent or recurrent neck pain closely temporally associated with migraine attacks; Positive physical examination findings indicative of cervical joint or muscle impairments; Elevated neck disability index scores; Increased migraine attack frequency or progression to chronic migraine; Evidence of central sensitisation (e.g. cutaneous allodynia); Comorbid anxiety or depression potentially exacerbating symptom burden.
Recognising the predominant pathophysiological mechanisms in each patient is essential to tailor more precise and effective treatment strategies.
Currently, pharmacological treatments for these patients are the same as those prescribed to general migraine patients, as the presence and relief of neck pain in migraine have not been specifically studied. Identifying which mechanisms predominate in each patient could guide more personalised and effective treatment strategies. Randomised clinical trials are urgently needed to evaluate the impact of both acute and preventive migraine treatments on neck pain. These trials would help determine whether certain pharmacological therapies are more effective for patients with coexisting neck pain and migraine, guiding specific treatment recommendations. Additionally, non-pharmacological treatments may also need refinement to address musculoskeletal tension and cervical hypersensitivity more effectively. A comprehensive musculoskeletal evaluation would facilitate the adaptation of physical therapy, behavioural interventions or neuromodulation to the specific needs of these patients, potentially improving outcomes.
Conclusions
The pathophysiological mechanisms underlying neck pain in migraine are not fully understood. Current evidence suggests that cross-talk between cervical and trigeminal afferents in TCC and central sensitisation are involved and may differentially contribute to neck pain as a symptom or trigger (53,54,107). Further preclinical and clinical studies examining the interaction between neck pain and migraine are needed. Furthermore, given the high prevalence of neck pain in migraine and the frequent misdiagnosis of migraine as a cervical disorder (1,6), comprehensive and accurate clinical assessment is essential for ensuring correct diagnosis and effective management. However, the efficacy of both acute and preventive therapies in addressing migraine-associated neck pain remains poorly characterised. Studies evaluating the effectiveness of pharmacological and non-pharmacological interventions in treating neck pain, particularly those targeting the CGRP system where data is sparse, should be prioritised.
Article highlights
Neck pain is highly prevalent in patients with migraine but is often incorrectly diagnosed as cervical disorders, resulting in a lack of appropriate treatment.
Shared neuroanatomy may underlie the bilateral relationship between neck pain and migraine. However, further research is needed.
Careful clinical history and physical exam are essential to ensure the correct diagnosis of neck pain in migraine. Within, we propose an assessment and treatment algorithm.
Standard migraine therapies, such as amitriptyline and onabotulinumtoxinA, may reduce migraine-associated neck pain. However, the efficacy of newer acute and preventive therapies targeting CGRP remains poorly characterised.
Footnotes
Author contributions
All authors contributed to the article and approved the final manuscript submitted for publication.
Data availability
The data used for this article may be obtained from the authors with a reasonable request.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: VW has received honoraria as a speaker for AbbVie. AB has received funding for conferences from TEVA and AbbVie. EC has received honoraria from Novartis, Chiesi, TEVA, Lundbeck, Medscape Dr Reddy's, Eli Lilly and Organon. PP-R reports, within the prior 36 months, having received honoraria as a consultant and speaker for: AbbVie, Dr Reddy's, Eli Lilly, Lundbeck, Medscape, Novartis, Organon, Pfizer and Teva. Her research group has received research grants from Novartis, Teva, AbbVie, EraNET Neuron, RIS3CAT FEDER, AGAUR, ISCIII and International Headache Society; and has received funding for clinical trials from Amgen, Eli Lilly, Lundbeck, Novartis, Pfizer and Teva. She is the Honorary Secretary of the International Headache Society. She is the founder of ![]() . PP-R does not own stocks from any pharmaceutical company.
. PP-R does not own stocks from any pharmaceutical company.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: TAR acknowledges receipt of the International Headache Society Fellowship from the International Headache Society. EC’s salary has been partially funded by Río Hortega grant Acción Estratégica en Salud 2017–2020 from Instituto de Salud Carlos III (CM20/00217) and Juan Rodés fellowship, Subprograma Estatal de Incorporación de la Acción Estratégica en Salud 2023 (JR23/00065).
