Abstract
Background
Emerging evidence suggests a link between migraine and selenium (Se). Se is related to systemic low-grade inflammation and thyroid function, which in their turn are also linked to migraine. Thus, we aimed to explore these relationships and hypothesized that higher Se levels would be related to decreased risk of migraine with an influence of systemic low-grade inflammation and thyroid function.
Methods
In a prospective analysis of the Brazilian Longitudinal Study of Adult Health (ELSA-Brasil), measurements of serum Se levels, dietary Se intake, high-sensitivity C-reactive protein (hs-CRP) and thyroid function hormones at baseline (2008–2010) and migraine incidence (2013–2014) were assessed. Serum Se was quantified by inductively coupled plasma mass spectrometry, a validated Food Frequency Questionnaire estimated dietary Se intake and hs-CRP was measured by the quantitative nephelometry method. Diagnosis of migraine without aura (MWO) and migraine with aura (MWA) followed International Classification of Headache Disorders, 3rd edition, criteria, while chronic migraine (CM) was defined according to headache attack frequency. Modified Poisson regression models estimated risk ratios (RR) for migraine subtypes, along with 95% confidence intervals, according to serum Se levels (continuous or quartiles).
Results
Among 2355 adults (mean age: 52.4 ± 9.1 years, 48.8% female), 20.0% were diagnosed with overall migraine between 2013–14. The mean ± SD follow-up time was 4.1 ± 0.37 years. Compared to no migraine, the overall migraine group had significantly lower median (interquartile range) serum Se levels (70.8 μg/l (60.4–82.8) vs. 177.0 μg/l (149.0–220.7), p = 0.017) and dietary Se intake (170.9 μg/l (147.4–200.3) vs. 177.0 μg/l (149.0–220.7), p = 0.018), while no between-group differences for hs-CRP and thyroid function hormones was found. There was a significant positive association between Se levels and dietary Se intake, while both were negatively associated with hs-CRP levels. Compared to the lowest quartile of serum Se levels, the highest quartile was associated with a lower risk of overall migraine (RR = 0.56 (0.31–0.99), p = 0.046) in the models adjusted for sex, age, body mass index, race, household income, schooling, marital status, smoking status, alcohol consumption, and use of migraine prophylactic medication, thyroid function hormones and hs-CRP. In the sex-stratified analysis considering the same confounders, a decreased risk of MWO was observed among males (RR =0.53 (0.29–0.94), p = 0.026) and CM among females (RR = 0.71 (0.51–0.98), p = 0.038) within the highest quartile of serum Se.
Conclusions
In the ELSA-Brasil Study, diet-related higher Se levels were associated with a lower risk of migraine regardless of systemic low-grade inflammation and thyroid hormones, with migraine type- and sex-specific associations. Further studies are warranted to confirm the involvement of Se in the risk of migraine and migraine progression.
This is a visual representation of the abstract.
Introduction
Migraine pathophysiology encompasses endogenous and environmental factors (1). Dietary factors are among the most common environmental factors associated with migraine (2).
A growing body of evidence has suggested an association of the essential micronutrient selenium (Se) with migraine (3–7). Se is an essential metalloid that participates in several enzymological pathways and plays crucial roles in health, disease and aging (8). One of the most studied biological roles of Se is its anti-inflammatory and antioxidant properties by composing the selenoproteins family that maintains an optimum cellular antioxidant state (8).
The main assumption of a possible protective role of Se on migraine is through dietary Se sources, which allegedly could counteract the pro-inflammatory mechanisms and higher oxidative stress observed in migraine (6,9,10). Higher levels of high-sensitivity C-reactive protein (hs-CRP), a non-specific biomarker of systemic low-grade inflammation, have been linked to migraine (11,12). Conversely, a meta-analysis of randomized controlled trials showed that Se supplementation significantly and dose-dependently reduced serum hs-CRP levels (13).
Despite previous studies have suggested a link between selenium, inflammation and migraine (4–7), no study has so far measured serum Se, dietary Se intake and inflammatory markers adopting a prospective design. Besides, Se is constitutional of key enzymes of thyroid function, such as the thioredoxin reductases, thioredoxin-glutathione reductase, iodothyronine deiodinases family (14), while clinical and epidemiological evidence have shown bidirectional associations between migraine and hypothyroidism (15–21).
To address this critical research gap, this study aimed to estimate prospectively the risk of migraine based on the exposure to serum Se levels and whether serum Se would be associated with dietary Se, hs-CRP and thyroid function hormones. Furthermore, we aimed to test whether the relationship between Se levels and migraine would be modified by hs-CRP and thyroid function hormones. Given the higher prevalence of migraine in women, we wondered whether there would be sex-specific associations.
We hypothesized that serum Se levels would be positively associated with dietary Se intake and lower hs-CRP levels, and higher Se status would be associated with a decreased risk of migraine, especially in women, in a Brazilian prospective cohort, the Brazilian Longitudinal Study of Adult Health (ELSA-Brasil).
Methods
Study design and participants
This is a prospective study with secondary, post-hoc analysis of a subset of participants from the ELSA-Brasil study. In brief, the ELSA-Brasil study is a large ongoing multicenter cohort, designated to investigate several lifestyle and risk factors associated with major non-communicable disorders factors in a sample of 15,105 civil servants (35–74 years of age) from six metropolitan areas of Brazil: Belo Horizonte, Porto Alegre, Rio de Janeiro, Salvador, São Paulo and Vitória (22). While the primary aim of ELSA-Brasil is to investigate cardiometabolic diseases, the study has also examined other comorbidities, including headache disorders, lifestyle factors such as diet, and an array of laboratory tests such as serum metals/metalloids and hs-CRP (22).
From 5061 individuals from the São Paulo Investigation Center of ELSA-Brasil, we considered 2763 with full data on headache disorders, serum Se, and dietary Se intake at baseline (2008–2010). Among these, we excluded participants with a diagnosis of definite migraine or secondary headaches at baseline (n = 266) and serum hs-CRP ≥ 20 mg/l (n = 95). Finally, the remaining participants with a previous medical history of major cardiovascular diseases, stroke, heart surgery, hepatological, or hematological diseases that could affect systemic inflammation and interfere with the anti-inflammatory effects of Se were also excluded (n = 47). Ultimately, we analyzed data from 2355 participants (Figure 1). Of note, probable migraine cases were not excluded from the main analysis.
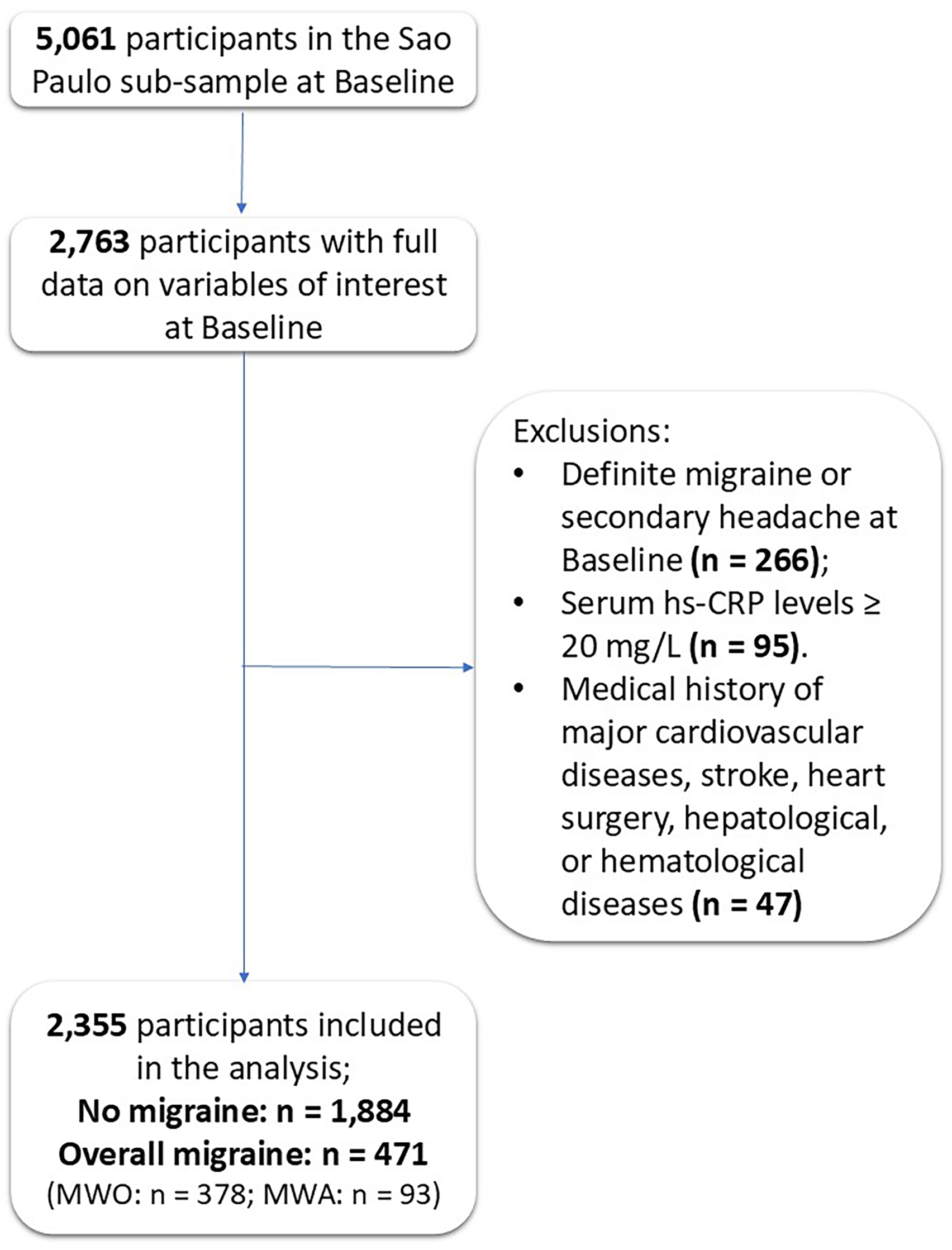
Participants’ flow in the study. Se = selenium; hs-CRP = high-sensitivity C-reactive protein; MWO = migraine without aura; MWA = migraine with aura.
Approval from the local institutional review board (CEP-HU/USP:#659/06), the National Research Ethics Committee (CAAE: #08109612.7.1001.0076) and signed informed consent were provided previously to the beginning of the study. This study complies with the STROBE guidelines for reporting data from observational research.
Primary outcome and exposures
The primary outcome was the risk of migraine, based on the information collected at Wave 2 (2013–2014) in the ELSA-Brasil study, while serum Se levels at baseline (exposure) was set as exposure/predictor. Serum Se concentration was set as a predictor because it represents an objective measure of Se status (8).
Migraine diagnosis
All participants who answered “yes” to the question “In the last 12 months, did you have a headache?” at the ELSA-Brasil baseline evaluation were invited to answer a detailed headache questionnaire based on the International Classification of Headache Disorders, 2nd edition (ICHD-II) (23) previously validated in Brazil (24). For migraine diagnosis, the questionnaire showed a sensitivity of 90.2%, specificity of 57.9%, a positive predictive value of 65.7% and a negative predictive value of 86.8% (Cohen’s κ = 0.47) (24). The questionnaire investigates pain frequency, duration, quality, location, intensity, triggering factors and accompanying symptoms, such as nausea or vomiting. The inventory of aura symptoms included the same headache questionnaire used at baseline and included questions on visual, somatosensory or motor symptoms for the diagnosis of migraine with aura (MWA). Migraine without aura (MWO) and MWA diagnoses were based on the 3rd version of the International Headache Society (his) criteria (25). Regarding headache frequency, participants with migraine were asked whether their headache attack frequency in the past month was <15 or ≥15 days/month. For the purpose of this study, the definition of chronic migraine (CM) was operationalized through a headache frequency ≥ 15 days/month, as adopted by other prospective cohort studies (26).
Participants experiencing no headache or other primary non-migraine headaches in the past 12 months were classified as “No Migraine”. An expert medical doctor from the ELSA-Brasil study conducted the interviews by telephone and determined the diagnoses based on the International Classification of Headache Disorders, 3rd edition (ICHD-3).
Serum Se levels: exposure
Before sample analysis, plasma samples were diluted 1:20 with a solution containing 0.5% v/v HNO3 and 0.005% v/v Triton X-100. The quantification of serum Se levels was carried out using Inductively Coupled Plasma Mass Spectrometry equipped with Quadrupole Ion Deflector (NexION® 2000; PerkinElmer, Shelton, CT, USA) according to Batista et al. (27). We tested the accuracy and precision of the data by measuring several reference serum samples obtained from the Institut National de Santé Publique du Quebec (Quebec City, QC, Canada). Results always agreed with target values (t-test, 95%). Results below the respective limit of detection (LOD) were replaced by LOD/√2. Serum Se levels were expressed in micrograms per litre (μg/l).
Dietary Se intake
In the present study, the consumption of food rich in selenium was based on the data from a food frequency questionnaire (28) collected at baseline (2008–2010) in the ELSA-Brasil. We estimated Se in the diet from the regular consumption of the following foods in the last six months: nuts, meat, fish and eggs. Daily nutrient and caloric intake were calculated using the Nutrition Data System for Research (NDSR) software, previously validated in the ELSA-Brasil study (29). Ultimately, all of them were corrected by the total caloric value (TCV) to compound the final variable: dietary Se intake.
The correction was performed to adjust for extreme values in the participants’ responses regarding their dietary intake. We first applied the natural logarithm to the variables TCV and Se (denoted as log TCV and log dietary Se intake, respectively). We then used a linear regression model, where the dependent variable (response) was log dietary Se intake and the independent variable (predictor) was log TCV. The model is expressed as:
We also calculated the mean of log TCV across all participants in the database. We calculated the correction constant using the estimated coefficients and the mean of the log-transformed TCV. We then used the residuals from the regression model and added them to the correction constant. To return to the original units of dietary Se intake (undoing the log transformation), we applied the exponential function to the adjusted values.
Quantification of hs-CRP
Blood samples were collected from participants in the six research centers following an average overnight fasting period of 12 hours. The samples were stored in dry tubes. To ensure consistent and reliable results, they were processed and analyzed in a central laboratory of the ELSA-Brasil study. The hs-CRP levels were measured using the quantitative nephelometry method (BN II; Siemens Muich, Germany) (30). The results were reported in milligrams per liter (mg/l).
Quantification of thyroid function hormones
Thyroid-stimulating hormone (TSH) and free-thyroxine (T4) levels were measured using a third-generation immunoenzymatic assay (Roche Diagnostics, Mannheim, Germany) (31). TSH and T4 are expressed in milli-international units per liter (mIU/l) and nanograms per deciliter (ng/dl), respectively.
Other covariates
Covariates included sociodemographic factors, previous medical history of primary CVD conditions (self-reported since baseline and at Wave 2 through telephone interviews) and use of medication with prophylactic actions for migraine. Sociodemographic variables encompassed sex assigned at birth (female or male), age (years), household income (<US$1,245, US$1245–3,319 and > US$3319), educational level (elementary, high school or college), race determined by self-identified skin color (White, Black, Brown or Pardo, Others – Indigenous, Asian) and marital status (married, separated, single, widow/widower or other).
Smoking status (no, former, current) and alcohol consumption (no, former, current) and body mass index (BMI) were selected as lifestyle and health factors covariates.
Regarding the medications with prophylactic effects on migraine (yes/no), we included medications with evidence level A and B, according to the American Academy of Neurology's guidelines, based on the medication data collected at Wave 2 (32). The medications included for each evidence level were: Level A: antiseizure drugs (divalproex sodium, sodium valproate, topiramate), beta-blockers (propranolol, metoprolol, timolol); Level B: antidepressants (amitriptyline, venlafaxine) and beta-blockers (atenolol, nadolol).
The main cardiometabolic risk factors of this sample were assessed through standardized anthropometric and laboratory procedures following strict quality control 39, including measurements of blood pressure, fasting glucose, total cholesterol and its fractions, low (LDL) and high (HDL) density lipoproteins, triglycerides and glycosylated hemoglobin (HbA1C). Hypertension was diagnosed based on a prior clinical history of the condition, a systolic blood pressure of ≥140 mmHg, a diastolic blood pressure of ≥90 mmHg or the use of antihypertensive medication. Diabetes was defined by a previous medical diagnosis, use of glucose-lowering drugs, fasting plasma glucose ≥126 mg/dl, two-hour plasma glucose ≥200 mg/dl or HbA1C ≥6.5%. Dyslipidemia was diagnosed according to NCEP ATP III criteria, with dyslipidemia defined as LDL cholesterol ≥130 mg/dl or the use of lipid-lowering medication (33).
Statistical analysis
Descriptive statistics for sociodemographic, clinical and laboratory data are presented as absolute values and percentages (%), mean ± standard deviation (SD), or median with interquartile range (IQR). Inspecting the histograms and Kernel density plots verified adherence to the assumption of a normal distribution. Comparison between groups (No migraine: reference, Overall migraine, MWA, MWO) was conducted using a chi-squared test, t-test for independent samples and the Wilcoxon rank-sum test, depending on whether variables were categorical, continuous or exhibited non-normal distribution. We also compared serum Se levels, dietary Se intake, thyroid function hormones and hs-CRP levels between overall migraine status and sex groups.
To explore the relationships between the constant value of the variables of interest in the total sample, linear regression models were built, and the marginal effects of statistically significant associations were plotted to visually illustrate the predicted values of dependent variables based on independent variables. Since these response variables exhibited positive, right-skewed distributions, generalized linear models with a Gamma family for non-normally distributed variables were employed.
To capture complex, non-linear relationships between the serum Se levels and migraine risk and for detecting changes in the slope of association that a linear model cannot fit, we further modeled these relationships using restricted cubic splines with 6 knots positioned at the 5th, 23rd, 41st, 59th, 77th, and 95th percentiles of LE8 scores, following Harrelĺs method (34). The reference value for serum Se levels was set at the samplés median (72.3 μg/l). The restricted cubic splines models were adjusted for sociodemographic variables. To estimate the p-values of the linear and non-linear patterns of the restricted cubic splines, we employed the delta-based Wald test.
As the primary outcome analysis, we utilized modified Poisson regression with robust variance estimators to estimate the risk ratio (RR) with 95% confidence interval (CI) of migraine diagnoses (“no” set as reference and “yes”) at follow-up based on quartiles (Q) of serum Se levels at baseline (Q1st “lowest” set as reference). Separate 2 × 2 models for overall migraine, MWO, MWA and CM were fitted. Adjusted models controlled for the effects of the sociodemographic variables age (continuous), sex (“female” as reference, and “male”), race self-reported skin color (“white” as reference, “brown”, “black” and “others”), household income (“>US$3319” as reference, “US$1245–3319” and “<US$1245”), schooling (“College” as reference, “High School” and “Elementary”), marital status (“married” as reference, “single”, “separated/divorced” and “widow[er]”), BMI (continuous), smoking and alcohol consumption (for both, “never” was as reference, “former” and “current”), use of migraine prophylactic medication (“no” as reference and “yes”), the presence of cardiometabolic risk factors (“no” as reference and “yes”) hypertension, diabetes and dyslipidemia, as well as continuous values of inflammatory systemic markers such as hs-CRP, TSH and T4.
To test whether sex was an effect modifier, sex interactions were computed across models and male was set as reference. Moreover, to further explore the relationship between exposure and outcome regarding sex, sex-stratified analyses were conducted.
The Poisson regression assumptions were verified, and regression diagnostics were evaluated accordingly. To test whether the model significantly improved fit over the null model, the model's deviance was computed. The models significantly improved fit over their respective null models (deviance's p values ranging from 0.813 to 0.999). Pearson and deviance residuals and Pregibońs dbeta plots were built to identify observations with high influence on estimated parameters and overall fit. The models showed high residuals values and influential observations (dbeta values more than times the average values) of serum Se levels (n = 57). Multicollinearity among independent variables was assessed using variance inflation factor (VIF). All models showed VIF values less < 3, ruling out multicollinearity among independent variables across models.
Finally, as the sample showed influential observations, that is, outliers with extremely high values of serum Se levels, a sensitivity analysis was conducted by excluding influential observations of serum Se levels (n = 57).
Data were analyzed in Stata, version 17.0 (StataCorp LLC, College Station, TX, USA). A two-tailed p < 0.05 was considered statistically significantfor all tests.
Results
The flow of participants in the present analysis is demonstrated in Figure 1. Of 2355 participants (mean age: 52.4, SD ± 9.1 years), 48.8% (1150/2355) were female. At the inter-wave assessment, the overall migraine incidence was 20.0% (471/2355) at Wave 2. The incidence was higher in females (28.5%, 328/1150) than in males (11.8%, 143/1205). The incidence of MWO, MWA and CM was 16.0%, 3.9% and 3.2%, respectively.
Sociodemographic data, laboratory results, clinical characteristics, and lifestyle factors at baseline, stratified by migraine status, are presented in Table 1. Compared to the no migraine group, participants with migraine were predominantly female, younger, belonged to lower socioeconomic strata, had mostly middle levels of educational attainment, consumed less alcohol and had a worse lipidemic profile (Table 1).
Sociodemographic characteristics of 2355 participants from the ELSA-Brasil study – Sao Paulo center at baseline.

BMI = body mass index; MWO = migraine without aura; MWA = migraine with aura.
Due to minor missing data for medication use (0.3%), income (0.5%) and race (1.1%), the percentages for these variables do not reach 100%.
The median (IQR) serum Se levels in the total sample were 73.2 μg/l (61.4–87.2). Females showed higher serum Se levels than males (74.0 (63.2–90.4) vs 70.4 μg/l (59.2–84.6), p < 0.001). The overall migraine group showed lower median (IQR) values of serum Se levels (70.8 μg/l (60.4–82.8) vs. 72.5 μg/l (61.9–88.3), p = 0.018) and dietary Se intake (170.9 μg/day (147.4, 200.3) vs. 177.0 μg/day (149.0–220.7), p = 0.017), while there was no difference between groups for hs-CRP, TSH or T4 levels. Females showed higher levels of hs-CRP (1.58 μg/day (0.75–3.86) vs 1.23 μg/day (0.65–2.60), p < 0.001) and lower levels of T4 (1.17 ng/dl (1.06–1.30) vs 1.21 ng/dl (1.11–1.33), p < 0.001) than males.
The adjusted linear regression models in the total sample showed significant positive associations between serum Se levels and dietary Se intake (β = 0.107, 95% CI = 0.097–0.118), p < 0.001). Both serum Se levels (β = −0.514, 95% CI = −0.960 to −0.068), p = 0.024) and dietary Se intake (β = −1.753, 95% CI = −3.303 to −0.203), p = 0.027) were inversely associated with hs-CRP (Figures 2(a)–(c)). Serum Se levels showed no association with TSH (β = 0.02, 95% CI = −0.36 to 0.41), p = 0.909) and T4 (β = −4.25, 95% CI = −10.58 to 2.07), p = 0.187) (Figures 2(d)–(e)), even when the migraine and no migraine subgroups were analyzed separately.

Margins plot of linear regression models for associations between (a) serum selenium (Se) levels and serum hs-CRP, (b) dietary Se intake and hs-CRP, (c) dietary and serum Se, (d), TSH and serum Se, and (e) free T4 and serum Se in 2355 participants of the ELSA-Brasil study. The models were adjusted for sex, age, body mass index, race, household income, schooling, marital status, smoking status, alcohol consumption and use of migraine prophylactic medication. hs-CRP: high-sensitivity C-reactive protein; T4 = free-thyroxine; TSH = thyroid-stimulating hormone.
The continuous values of serum Se levels showed a non-linear relationship with the risk of overall migraine (p-value for non-linear < 0.001) (Figure 3). However, based on Wald´s test for comparisons of splines linearity, we identified a linear inverse relationship after the fourth knot, corresponding to serum Se levels above 90 μg/l (Figure 3).
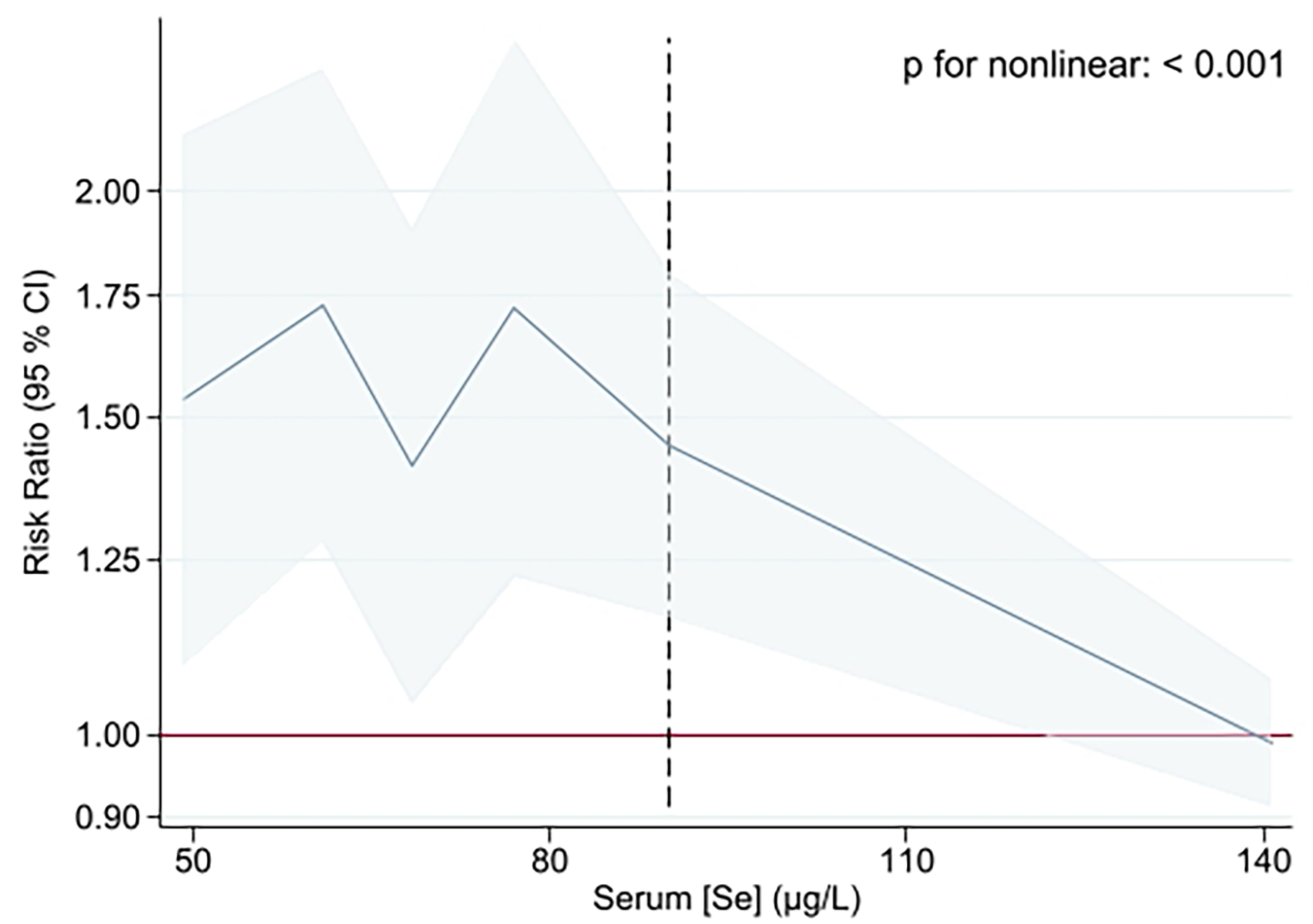
Restricted cubic splines of the associations between serum Se levels and migraine in 2355 participants of the ELSA-Brasil study. Se: Selenium; Reference group: No migraine (n = 1884); The X-axes were trimmed at the 0.5th to 99.5th percentile of serum Se; the model was adjusted for sex, age, body mass index, race, household income, schooling, marital status, smoking status, alcohol consumption and use of migraine prophylactic medication. The dashed vertical line represents the value (90 μg/l) above which the association exhibits an inverse linear pattern. CI = confidence interval.
Regarding the primary outcome, the highest quartile (Q4) of serum Se levels was associated with a decreased risk of overall migraine (RR = 0.53 (0.30–0.93), p = 0.037) compared to the lowest quartile (Q1), even after the adjustments for confounders (Table 2). There was no significant sex interaction effect in the models with overall migraine (p-interaction = 0.592), MWO (p-interaction = 0.815) and MWA (p-interaction = 0.339) set as the outcome.
Risk ratio (RR) with 95% confidence interval (CI) for migraine by quartiles (Qs) of serum selenium (Se) among 2355 participants from the ELSA-Brasil study after four-year follow-up.
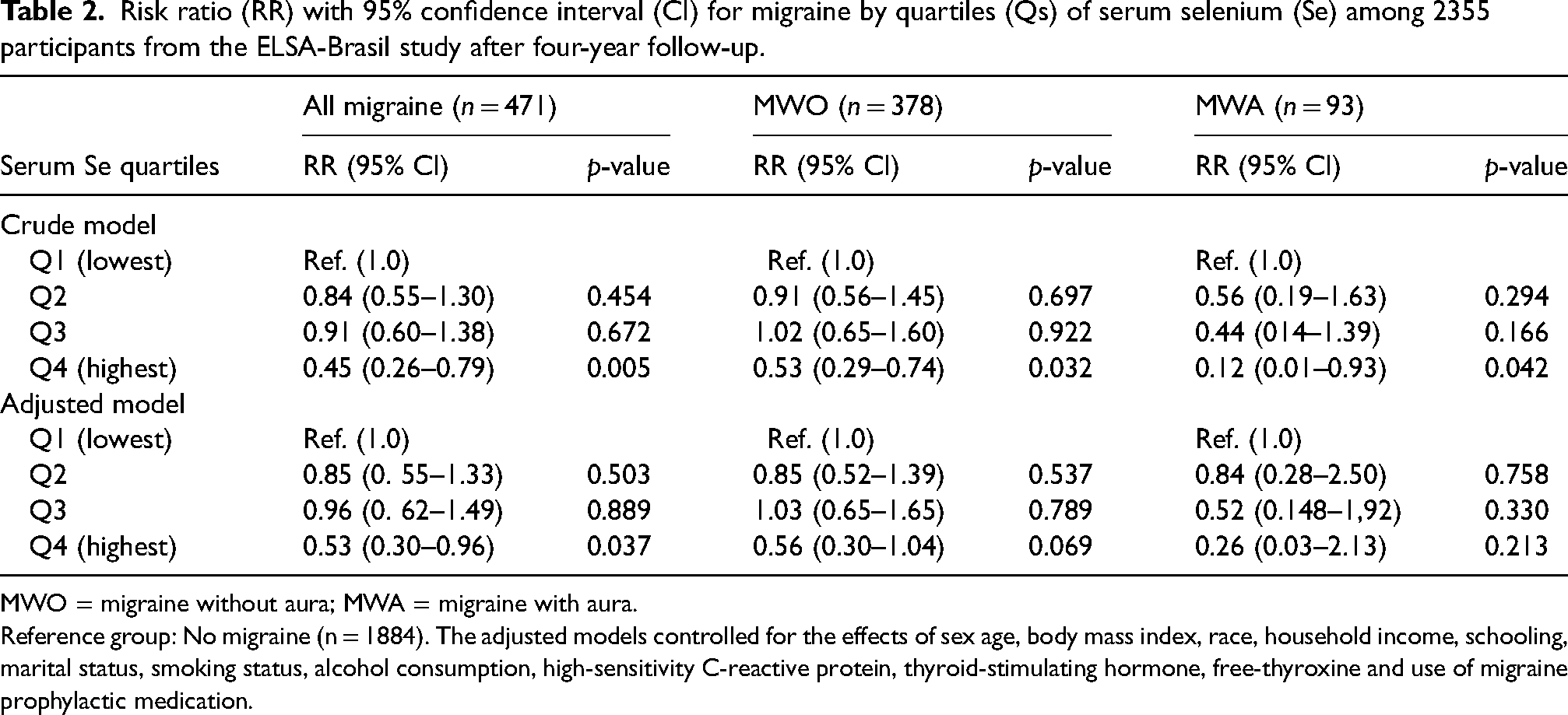
MWO = migraine without aura; MWA = migraine with aura.
Reference group: No migraine (n = 1884). The adjusted models controlled for the effects of sex age, body mass index, race, household income, schooling, marital status, smoking status, alcohol consumption, high-sensitivity C-reactive protein, thyroid-stimulating hormone, free-thyroxine and use of migraine prophylactic medication.
Sex-stratified analysis considering the same multivariate models showed a decreased risk of overall migraine (RR = 0.41 (0.22–0.76), p = 0.009) and MWO (RR = 0.43 (0.22–0.83), p = 0.012) associated with the highest quartile (Q4) of serum Se levels among males only (Table 3).
Sex-stratified risk ratio (RR) with 95% confidence interval (CI) for migraine by quartiles (Qs) of serum selenium (Se) among 2355 participants from the ELSA-Brasil study after four-year follow-up.

Reference group: No migraine (males: n = 1062, females: n = 822). The adjusted models controlled for the effects of age, body mass index, race, household income, schooling, marital status, smoking status, alcohol consumption, high-sensitivity C-reactive protein, thyroid-stimulating hormone, free-thyroxine and use of migraine prophylactic medication.
There was a decreased risk of CM associated with the highest quartile (Q4) of serum Se levels in the adjusted models of the total migraine sample (RR = 0.71 (0.51–0.98), p = 0.041) and among females (RR = 0.59 (0.38–0.91), p = 0.019) was associated with the highest quartile (Q4) of serum Se levels (Table 4). Sex was a significant effect modifier in this model (p-interaction = 0.003).
Risk ratio (RR) with 95% confidence interval (CI) for chronic migraine by quartiles (Qs) of serum selenium (Se), with sex-stratified analysis among 471 participants with migraine from the ELSA-Brasil study after four-year follow-up

Reference group: headache attack frequency < 15 days/month (total n = 396; Males: n = 123; Females: n = 273). The adjusted models controlled for the effects of sex (for total analysis only), age, body mass index, race, household income, schooling, marital status, smoking status, alcohol consumption, high-sensitivity C-reactive protein, thyroid-stimulating hormone, free-thyroxine and use of migraine prophylactic medication.
In the sensitivity analysis, after excluding influential observations (n = 57), the significant associations across migraine types remained significant in the unadjusted models only. This indicates that influential observations with extreme values of serum Se levels had an impact on the models.
Discussion
In the present study, we evaluated the risk of migraine in relation to selenium status exposure over a four-year follow-up period. We provided the first evidence for sex-specific effects of Se status on the risk of migraine, according to aura symptoms and chronification. A higher Se status was associated with decreased risk of overall migraine and MWO in men and CM in women. Furthermore, our hypothesis was confirmed regarding the association between serum Se and dietary Se intake, while both were related to lower hs-CRP levels.
Although Se is recognizably functional for key enzymes of thyroid function (16) and clinical and epidemiological studies have pointed associations between migraine and hypothyroidism (17–23), as well as with high hs-CRP levels (13,14), our analysis indicates that Se was not associated to thyroid function hormones and hs-CRP, nor did they affect the observed link between Se and migraine. Thus, the relationship between Se status and migraine seems to be independent of systemic low-grade inflammation and thyroid function hormones.
The findings here are in the same direction as pre-clinical (5) and clinical studies (3,4,35) that explored the relationship between serum Se levels and migraine, investigated the therapeutic effects of Se supplementation and measured changes in the activity of Se-containing enzymes and oxidative capacity markers. Se supplementation significantly reduced oxidative stress biomarkers and lowered headache frequency, severity and disability scores compared to placebo (4). Therefore, it is likely that Se may exert preventive effects on migraine by recruiting other anti-inflammatory or antioxidant processes via migraine-specific molecular mechanisms.
We uncovered a previously unreported sex-specific association between Se status and susceptibility to migraine and migraine progression. Reports on sex-specific effects on serum selenium levels have yielded contradictory findings, with dietary habits exerting a significant role (36,37). It is well-documented that sex hormones exert regulatory actions in Se metabolism, ultimately affecting Se status and its biological functions (36). The intestinal absorption of selenomethionine, which is a major form of Se present in food, was shown to be higher in healthy women (∼96%) than men (∼76%) (38–40). Estrogen and testosterone differentially affect trans-selenation (36). Trans-selenation refers to the enzymatic steps in the conversion of selenomethionine into selenocysteine, the most important active compound of selenoproteins (41). Women have higher activity of selenoprotein glutathione peroxidase 3 than men (42). Finally, the testis has a high uptake rate of Se, which can reduce Se bioavailability (36). These sex differences in Se metabolism may explain the reduced risk of overall migraine in men and migraine progression in women. This warrants further investigation.
Our findings partially align with two cross-sectional studies assessing the National Health and Nutrition Examination Survey (NHANES). One study (n = 12,964; 47.3% female) reported a U-shaped association between dietary Se intake and migraine prevalence in obese individuals, with optimal intake at ∼90 μg/day (6). In the other analysis (n = 9849; 50.7% female), there was also a non-linear relationship, however, Se intake ≥101.9 μg/day was related to a lower odds of migraine in the 20–50 years group (7). Although we also observed a non-linear association between serum Se and migraine, we detected a linear inverse association between serum Se and migraine above a serum Se level of 90 μg/l. Methodological differences between the ELSA-Brasil and NHANES may explain this discrepancy, including study design (longitudinal vs. cross-sectional), adjustment strategies (BMI- and age-adjusted vs. stratified), treatment of unrealistic dietary intake (employment of corrections vs. no correction) and exposure assessment (objective serum Se vs. self-reported dietary Se intake) (6,7).
Furthermore, we selected serum Se as a predictor of migraine risk in our models rather than dietary Se intake because it reflects Se status better than self-report diet, which is subject to several limitations, such as recall bias and variation in diet composition estimates.
The main implication of this study is two-fold. First, our findings provide evidence to support maintaining dietary Se within recommended levels for migraine prevention and management. Clinicians can assess Se status using validated dietary questionnaires or, if feasible, laboratory quantification of serum Se levels. Studies assessing the risk of other diseases and health outcomes (e.g. some cancers, type-2 diabetes, mortality) associated with Se status have suggested that preventive effects or lower mortality rates associated with Se supplementation may benefit populations with baseline serum Se levels below 122 μg/l only, whereas no additional effects or even detrimental metabolic adverse effects have been reported when supplementing people with baseline levels already higher than 122 μg/l (8). Conversely, increasing Se concentration up to 130–150 μg/l was associated with the lowest mortality rates in people with low baseline Se levels (8). Speculation on the underlying mechanisms for these health-related effects of Se supplementation has accounted for the plateau of glutathione’s antioxidant capacity at these serum Se levels (8). Notably, the average serum Se levels in the highest quartile associated with lower migraine risk in the ELSA-Brasil cohort were 129.5 μg/l, which fell around the values of other health-enhancing outcomes found elsewhere (8,41,43). Furthermore, the inverse linear pattern of association between serum Se levels and migraine observed here (90 μg/l) was set right above the threshold of Se deficiency (85 μg/l) (8,44), which strengthens the idea that treating Se deficiency could also reduce migraine burden. As the link between Se and health/disease may exhibit a hormetic pattern (7,8,36), further research is needed to establish its efficacy and optimal use of Se intake or supplementation in migraine prevention and management based on the previous Se status.
Second, this study sheds light on the possible role of Se-mediated mechanisms in the pathophysiology of migraine. Selenium holds a major biological role by composing selenoproteins, which are key to redox regulation, cellular homeostasis, and antioxidant capacity (14). Of note, selenoproteins of the “glutathione family” and Se supplementation can modulate the expression and activity of eicosanoid enzymes and their associated pathways, as well as NO synthase, inducing the formation of prostaglandins E2 and NO (14), respectively, which constitute two “classical” migraine triggers (45). Besides, Se might help mitigate oxidative stress secondary to heavy metals accumulation associated with migraine chronification, such as higher iron deposition in the brain (46). Mitochondrial dysfunction is another factor implicated in migraine (47). Se-containing enzymes play a role in maintaining mitochondrial health by reducing oxidative stress (14). The impaired function of these enzymes may contribute to mitochondrial dysfunction, further linking Se to migraine mechanisms (35,47,48). Thus, this study paves the way for future pre-clinical and clinical research to deepen the understanding of the role of Se in migraine pathophysiology.
In our sample, both migraine and non-migraine subgroups showed higher dietary Se intake (170 μg/day or higher) than the recommended dietary allowance (RDA) of 55 μg/day for adults (43,49,50). Conversely, despite serum Se levels consistently correlating with Se intake, the average serum Se levels (∼72 μg/l) were below the desirable range and fell short of the 85 μg/l threshold, which is the cutoff acceptable to define Se deficiency (8,44). A possible explanation for this conflicting finding may lie in the sources of Se (i.e. animal, vegetable or supplement sources), or an inaccurate estimate of Se in the diet. For example, Se found in seafoods binds to heavy metals such as mercury (Hg), which lowers Se bioavailability from such foods. The predominance of animal sources of Se would result in lower Se bioavailability (51). In the ELSA-Brasil study's food questionnaire (28), the “Nuts” category combines several species (peanuts, cashews, almonds, Brazil nuts and pistachios) without distinguishing between their vastly different Se concentrations. Brazil nuts can contain over 130 times more Se than cashews (51), but cashews are more commonly consumed in the general population. Consequently, total Se intake estimates could be significantly overestimated if participants primarily consumed nut types with lower Se concentration.
Still, the most abundant well-known dietary source of Se is the Brazil nuts (Bertholletia excelsa), which have concentrations around 50.0 μg/g, reaching up to 512 μg/g, depending on the local soil composition (52–54). One single nut (∼6 g raw weight) can suffice the RDA of Se for an adult (53). Brazil nuts are native to the Amazon rainforest, which occupies most of the northern region of Brazil. In this region, its intake is common and serum Se levels of local populations range from normal to high (142.1–2029.3 μg/l) (37,55). Because most of the Se species found Brazil nuts are selenomethionine (75%–96%), the less toxic form of selenoproteins, this population has not reported selenosis (55).
Notably, the one-year migraine prevalence in the North region is 8.5% (against Braziĺs average prevalence of 15.2%), the lowest prevalence among all regions in Brazil (56). In contrast, our São Paulo-based cohort (20.5% migraine prevalence) (56) rarely consumes Brazil nuts due to cost and cultural factors. Therefore, it is likely that participants with higher Se status in the ELSA-Brasil cohort may represent Se-rich food consumers (e.g. Brazil nut consumers) and presumably less likely to have migraine or frequent headache attacks. On the other hand, it is also important to point out that despite the high levels of selenium found in Brazil nuts, maintaining a balanced diet that includes a variety of other selenium-containing foods, such as fish, seafood, meat, eggs and whole grains, may achieve adequate daily selenium intake (8).
Strengths and limitations
This study's greatest methodological strength is its prospective design, which establishes temporal relationships with pre-outcome exposure assessment. Furthermore, the ELSA-Brasil study encompasses a large sample size and thorough data collection, including headache data via validated IHS-based interviews high-quality lab assessments, and validated dietary habit questionnaires with corrections for unrealistic reporting. Moreover, this study provides a unique opportunity to evaluate migraine, a highly prevalent outcome in the ELSA-Brasil cohort, while considering the role of selenium-rich foods such as Brazil nuts as a potential protective factor for this condition.
It is important to acknowledge several limitations of this study. In this sample, all participants with definite migraine at the follow-up assessment presented probable migraine diagnosis at baseline. This challenges the assumption of Se-induced effects on migraine incidence. Considering the cohort characteristics in terms of age (i.e. on average older than the age range of migraine prevalence peak) and history probable migraine diagnosis, the most plausible interpretation is that higher Se status seems to protect against the evolution from probable migraine to migraine diagnosis in men, while it prevents the progression from episodic to chronic migraine in women. Still, chronic migraine was defined based on the single criterion regarding headache attack frequency ≥15 days/month. This may result in overdiagnosis of chronic migraine. Notwithstanding, considering that disease-related burden is higher in migraine than probable migraine, as well as the headache attacks frequency increases (57), these limitations do not invalidate the potential beneficial influence of Se in reducing migraine burden. A cautious interpretation is advised regarding the presence of influential observations of serum Se levels, which could translate into inflated estimates in the regression models. However, as these observations felt within the physiological range of serum Se (i.e. 165–409 μg/l) (8,43,49), they could demonstrate indeed the effect of high Se status on migraine prevention. Our sample’s geographical and demographic characteristics constrain the findings’ generalizability. The ELSA-Brasil cohort does not represent the Brazilian general population (22). Furthermore, the cohort consists of participants with an average age that is higher than the prevalence peak for migraine, which typically occurs around 30–40years of age (56). By including participants aged 34–75years, we exclude a younger demographic where migraine prevalence is notably high. Consequently, the older average age of our sample may obscure prior migraine diagnoses at younger ages. Finally, the food frequency questionnaire is susceptible to biases, including desirability and recall bias, as well as the version used at the time of ELSA-brasil baseline may not reflect Se composition of current nutrient composition tables. However, we employed corrections for unrealistic reporting of dietary patterns. These limitations should be considered when interpreting the results from the ELSA-Brasil study.
In conclusion, findings from the ELSA-Brasil study indicate that higher circulating selenium levels, reflecting dietary intake, were associated with a lower risk of migraine and migraine progression, with evidence of sex-specific effects. Above the deficiency threshold, serum selenium showed a linear inverse association with migraine. These results support the hypothesis that lifestyle modifications, including tailored dietary interventions, may play a role in migraine prevention. Importantly, future studies should incorporate updated compositional analyses and rigorous safety assessments to determine whether selenium-rich dietary strategies can be translated into effective and safe preventive approaches. Moreover, additional investigations are needed to confirm and elucidate the pathophysiological mechanisms linking selenium status to migraine.
Clinical implications
Higher diet-mediated selenium status was related to lower systemic low-grade inflammation.
Higher selenium status was associated with a lower risk of migraine.
The protection of higher selenium status exhibited migraine type- and sex-specific effects.
Higher selenium status was associated with lower MWO in men and CM in women.
These effects were independent of systemic inflammation and thyroid function hormones.
Footnotes
Acknowledgments
We are deeply grateful to all volunteers in the ELSA-Brasil.
Author contributions
Conception and design: ABO and ACG; Acquisition of data: FBJ, PAL, ISS, IMB and ACG; Analysis and interpretation of data: ABO, MFPP, PAL, ISS, IMB and ACG; Drafting the manuscript: ABO, MFPP, PAL, ISS, IMB and ACG; Revising it for intellectual content: ABO, FBJ, MFPP, PAL, ISS, IMB and ACG; Final approval of the completed manuscript: ABO, FBJ, MFPP, PAL, ISS, IMB and ACG
Data availability
The data is available from the corresponding author with reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical statement
Consent to participate: The participants signed informed consent were provided previously to the beginning of the study.
The ELSA-Brasil baseline study and the four-year follow-up were funded by the Brazilian Ministry of Health (Science and Technology Department) and the Brazilian Ministry of Science and Technology (Brazilian Funding Authority for Studies and Projects – FINEP, and the Brazilian National Research Council – CNPq). Grants of baseline: Rio Grande do Sul State (01 06 0010.00), Bahia State (01 06 0212.00), Espírito Santos State (01 06 0300.00), Minas Gerais State (01 06 0278.00), São Paulo State (01 06 0115.00) and Rio de Janeiro State (01 06 0071.00); grants of four-year follow-up: Rio Grande do Sul State (01 10 0643-03), Bahia State (01 10 0742-00), Espírito Santos State (01 12 0284-00), Minas Gerais State (01 10 0746-00), São Paulo State (01 10 0773- 00) and Rio de Janeiro State (01 11 0093-01).
Arão B Oliveira received a postdoctoral scholarship from FAPESP (Grant #2023/03011-5).
Funding
The study was funded by the São Paulo Research Support Foundation of the State of São Paulo (FAPESP) (Grants #2020/09468-9, #2018/24069-3 and #2021/05059-0).

