Abstract
C-reactive protein (CRP), a marker of inflammation, has been associated with cardiovascular disease. Risk of cardiovascular disease is increased in migraineurs with aura. Results from a clinical report, case-control and a cohort study suggest that CRP is elevated in migraineurs compared with non-migraineurs. We examined the proposed association in a case-control study nested within two large population-based studies. The relationship between migraine and CRP (high-sensitivity CRP) was studied in 5906 men and women aged 55.0 ± 8.5 years in the Reykjavik Study and 1345 men and women aged 27.7 ± 5.5 years from the Reykjavik Study for the Young. A modified version of the International Headache Society's criteria was used to categorize people into migraineurs (two or more symptoms) or non-migraineurs. Migraineurs with visual or sensory symptoms were further defined as having migraine with aura (MA) or without aura (MO). Multivariable-adjusted CRP levels were similar in migraineurs and nonmigraineurs for men (0.83 vs. 0.79 mg/l, P = 0.44) and for women (0.87 vs. 0.87 mg/l, P = 0.90). When further stratified by migraine aura and age, no differences were found between non-migraineurs, MO and MA among men. In women, CRP levels were borderline higher in those with MO compared with non-migraineurs and those with MA (1.01 mg/l vs. 0.81 and 0.75 mg/l, P = 0.08 and P = 0.08) in age group 19-34 years, but significantly lower in age group 60-81 years (0.52 mg/l vs. 1.07 and 1.01 mg/l, P = 0.007 and P = 0.03). CRP levels were not increased among migraine sufferers compared with non-migraineurs. Older women migraineurs without aura had lower CRP values than nonmigraineurs and migraineurs with aura.
Introduction
C-reactive protein (CRP), a marker of inflammation, has been associated with risk of cardiovascular disease (1). CRP has been suggested to be abnormal among migraineurs, possibly through repeated vascular inflammation (2). The risk of stroke and coronary heart disease is greater in migraineurs than in others, especially for those with aura (3, 4). There are limited data on the relationship between CRP and migraine.
Welch et al. performed a retrospective review of 60 migraineurs (90% female) with complex clinical features, who were referred to secondary or tertiary clinics (2). The results indicated that 43% of the patients had elevated CRP (defined as > 3 mg/l), with a suggestion of a higher proportion in patients without aura (16/29, 55%) compared with patients with aura (10/31, 32%).
A case–control study of 50 young adult patients with migraine and 50 controls (78% female) showed that the median CRP level was 1.42 mg/l in migraineurs and 0.90 mg/l in controls (P = 0.03) (5). CRP was higher in the patients without aura than in controls: 2.11 mg/l vs. 0.90 mg/l (P = 0.0002).
Compared with women with no migraine history, women with a self-reported history of migraine had a modestly increased multivariable-adjusted odds ratio for elevated CRP in a large cohort study of female health professionals, aged ≥ 45 years [1.13, 95% confidence interval (CI) 1.05, 1.22](6). Among current migraineurs, age-adjusted CRP was higher in the women without aura, compared with women with aura (4.08 vs. 3.86 mg/l).
Thus, the results from these three studies, a clinical report, a case–control study and one large cohort study of female health professionals, suggest that CRP is modestly elevated in migraineurs compared with controls. The results further suggest that the elevation is more evident for migraineurs without aura than those with aura.
In this study we further examined this potential relationship between CRP and migraine, with and without aura, in a case–control study nested within two large population-based cohorts of women and men ranging in age from 19 to 81 years.
Methods
Reykjavik Study briefly
The Reykjavik Study is a large, population-based cohort study, which started in 1967 (7). Men born 1907–1934 and women born 1908–1935, residing in the Reykjavik area and a few adjacent communities on 1 December 1966, were randomly selected for participation. A total of 19 390 persons agreed to take part, which was approximately 70% of all those invited. The study cohort, which was divided into six groups of men and women, was investigated at the Heart Preventive Clinic in Reykjavik during the period 1967–1996 [CRP was not measured in the sixth group (n = 478) and this group was therefore omitted from the analysis]. The first examination of each person occurred between 1967 and 1993 for men and between 1968 and 1996 for women. The age range at the time of CRP measurement was 33–81 years. The average year of examination used was 1974
In order to study subjects younger than the participants of the Reykjavik Study, a new sample was selected in 1972, the Reykjavik Study for the Young (8). This group comprised equal groups of men and women, 2781 in all, born 1940–1954. The subjects were invited to be examined three times in the years 1973–1974, 1983–1985 and 2001–2003. Those who participated in the third stage and had participated in either of the first two stages were sampled, and the first visit was used. The age range of the Reykjavik Study for the Young at the time of CRP measurement was 19–45 years. The present analysis is based on the CRP measured at the first examination. The number of subjects examined at least once was 1037 men and 1109 women. The average year of examination used was 1975
Combining the subjects from the Reykjavik Study and the Study for the Young, the total number of men was 10 171 and of women 10 878, and their ages at the time of measurement were 19–81 years.
Every participant received an invitation letter with standardized questions about health and social factors, including questions related to the presence of headache and headache features (see below).
The present analysis of the Reykjavik Study is based on a subset of participants who were selected for a nested case–control study of CRP and myocardial infarction (MI) (9). The cases were 2400 subjects without a history of MI at entry into the study who had a major coronary event during the mean (±
Every participant received an invitation letter with standardized questions about health and social factors, including questions related to the presence of headache and headache features (see below).
Examinations
Participants came in a fasting state to the clinic. After a 5-min rest, supine blood pressure was measured twice, between 08.30 and 10.30 h, by a nurse, and 10–14 days later between 11.00 and 13.30 h, by a physician. Subjects were not instructed to be fasting at the second blood pressure measurement. The instruments used were mercury sphygmomanometers of the type ‘Erkameter’ wall-model (Erka, Bad Tölz, Germany). The cuffs had a rubber bladder 15–32 cm, and the total length of the cuff was 66 cm. The same types of cuff and instrument were used throughout the study. Blood pressure was measured according to World Health Organization recommendations (10). Major coronary heart disease was defined as: death from coronary heart disease and non-fatal MI. Deaths from coronary heart disease were ascertained from central registers on the basis of a death certificate listing an International Classification of Diseases code of 410 to 414, and the diagnosis of non-fatal MI was based on the criteria of the Monitoring Trends and Determinants in Cardiovascular Disease study.
CRP measurements
Blood was drawn at the first visit, when subjects came in fasting. The concentration of CRP (high-sensitivity CRP, referred to as CRP in this study) was measured as described (9) by latex-enhanced immunoturbidimetry, with a lower limit of detection of 0.02 mg/l (Roche Diagnostics, Basel, Switzerland). The variation in CRP values within runs was < 1%, and the between-day variability was 1% at a concentration of 14 mg/l and 3.7% at a concentration of 3.8 mg/l (9). CRP measurements of samples in the Reykjavik Study and the Reykjavik Study for the Young were identical except for the recording of the CRP values; the former was recorded to two decimal points and the latter to one decimal point. We excluded subjects with CRP > 10 mg/l (n = 313) because values above this cut-point are usually associated with acute-phase stimulus (11), such as bacterial infection (12).
Definition of migraine
In this study a modified version (8) of the 1988 International Headache Society (IHS) criteria was used (13).
The questions on headache in the study questionnaire were the same for the original and younger Reykjavik cohorts. The questions were as follows.
Questions concerning symptoms during the last 12 months:
Is the pain usually on one side of the head?
Do you feel nauseated or vomit when you get the headache?
Do you get visual disturbances simultaneously or shortly before the pain starts?
Do you get photophobia during the headache attack?
Do you get numbness on one side of the face or numbness in either arm before the headache begins?
Subjects were considered to have migraine by ‘relaxed criteria’ if they answered yes to any two or more of questions 1–5 (8). Subjects were considered to have migraine by ‘strict criteria’ if they answered yes to any three or more of questions 1–5 (8). Migraineurs ‘relaxed criteria’ with visual or sensory symptoms (questions 3 and 5) accompanied by other symptoms were further defined as having migraine with aura (MA), those without visual or sensory symptoms were defined as migraineurs without aura (MO).
The questionnaire of the present study was composed in 1967. No questions were asked about the duration, intensity, phonophobia or pulsating quality of the headache, which are part of the IHS criteria from 1988 (13). Also missing were questions about the less common symptoms of unilateral weakness and speech difficulty.
Analysis
We compared the average CRP between subjects with migraine (with or without aura) and those without migraine, using linear regression. As the distribution of CRP was log-normal, we transformed CRP in all linear regression models.
All regression analyses were performed separately for men and women and performed separately within each age category (four age categories, 19–34, 35–49, 50–59 and 60–81 years). Adjustments were made for case–control status, age, body mass index (calculated as weight in kg divided by height in m2), cholesterol, smoking status (never smoked, former smoker, current smoker), education (elementary school or less, high school education, junior college or university education), current hormone use, current diabetes mellitus, systolic blood pressure (SBP) and antihypertensive therapy. SBP and antihypertensive use were in three categories: (i) SBP < 130 mmHg; (ii) SBP 130–160 mmHg; and (iii) SBP ≥ 160 mmHg and/or antihypertensive use. For women adjustment was made for oral contraceptive use, which has been shown to be associated with CRP levels (14). Adjustment for physical exercise (defined as 0, 0–5 and ≥ 6 h per week) was made but the variables listed above were stronger predictors of CRP levels in the regression model, and physical exercise was therefore left out of the final model. In addition, we tested for cohort difference in the association of CRP with migraine. The age group represented both in the Reykjavik Study and in the Reykjavik Study for the Young was used to estimate a possible cohort effect. A linear regression model with log-transformed CRP as a function of migraine status was used. In a multivariable-adjusted model for men, aged 30–39 years (n = 139) in the Reykjavik Study and (n = 186) in the Reykjavik Study for the Young, cohort was not a significant variable (P = 0.30). Similarly for women, (n = 84) for the Reykjavik Study and (n = 62) for the Reykjavik Study for the Young, cohort was not a significant variable (P = 0.76) in the model.
The above analysis was also performed for subjects who were not diagnosed with a major coronary event during follow-up.
Significance testing was two-sided and based on a 5% probability level. Thus, results are presented with 95% CI. The software package used was
Results
Migraine prevalence
Table 1 shows characteristics of the study population, consisting of the subset of the Reykjavik Study and the Reykjavik Study for the Young participants with available CRP measurements. The crude 1-year prevalence of migraine (relaxed criteria) was 5.0% among men and 14.3% among women. This prevalence was similar to that of migraine in the whole cohort (5.2% men, 14.1% women, Fig. 1) (8). The prevalence of MA was 3.6% for men and 8.8% for women (see Table 1).

Comparing the age- and sex-specific 1-year prevalence of migraine between those with C-reactive protein (CRP) measurements (n = 7251) used in the current study and the entire cohort (n = 21049) of the Reykjavik Study and the Reykjavik Study for the Young.
Characteristics of subjects with C-reactive protein (CRP) measurements in the Reykjavik Study (n = 5906) and the Reykjavik Study for the Young (n = 1345) according to migraine∗ status

Values are percentages unless otherwise indicated.
Migraine, relaxed criteria, defined as answering yes to two or more out of five questions on migraine.
Subjects with CRP ≥ 10 mg/l were excluded.
MA, migraine with aura; MO, migraine without aura; BMI, body mass index.
[Correction added after online publication 20 May 2009: ‘—’ was removed from columns 2–7 for the row ‘Contraceptive use’]
CRP values
After excluding subjects with CRP > 10 mg/l (n = 313), the median CRP value for all other subjects in the Reykjavik Study (n = 5906) was 1.31 mg/l (25th and 75th percentile 0.63 and 2.68 mg/l), and the corresponding median CRP value for subjects (n = 1345) in the Reykjavik Study for the Young was 0.6 mg/l (25th and 75th percentile 0.3 and 1.4 mg/l).
Age- and multivariable-adjusted CRP values for non-migraineurs, migraineurs, MO and MA subjects are shown in Table 2. Age-adjusted values among men were borderline lower for MO subjects compared with non-migraineurs, and the values for MA subjects were borderline higher than those of MO subjects. These differences were attenuated with multivariable adjustment. For women there were no statistically significant differences between the groups. Multivariate-adjusted values were similar among groups, ranging from 0.70 to 0.83 mg/l for men and 0.85 to 0.87 mg/l for women.
Age- and multivariable-adjusted C-reactive protein (CRP) values (mg/l) with respect to migraine status, relaxed criteria∗ and gender

Migraine, relaxed criteria, defined as answering yes to two or more out of five questions on migraine.
Subjects in the Reykjavik Study and the Reykjavik Study for the Young with available CRP measurements. CRP values ≥ 10 mg/l excluded.
CRP and migraine status in a linear regression model adjusted for case–control status, age, body mass index, cholesterol, smoking, education, hormone use, diabetes mellitus, systolic blood pressure (SBP) and antihypertensive therapy. For women adjustment was also made for birth control use. The profile used in the multivariable-adjusted model was: control with average values for continuous variables, non-smoker, high school education, SBP between 130 and 160 mmHg, without: diabetes, hormone use and antihypertensive therapy. Women without birth control use.
All comparisons between MA and No migraine gave P-values > 0.8.
MA, migraine with aura; MO, migraine without aura.
[Correction added after online publication 20 May 2009: The table head of Table 2 has been corrected]
Table 3 shows a comparison similar to that in Table 2, but with the stricter migraine criteria. The CRP values are almost identical for migraine defined with strict and relaxed criteria.
Age- and multivariable-adjusted C-reactive protein (CRP) values (mg/l) with respect to migraine status and gender, using strict migraine criteria∗
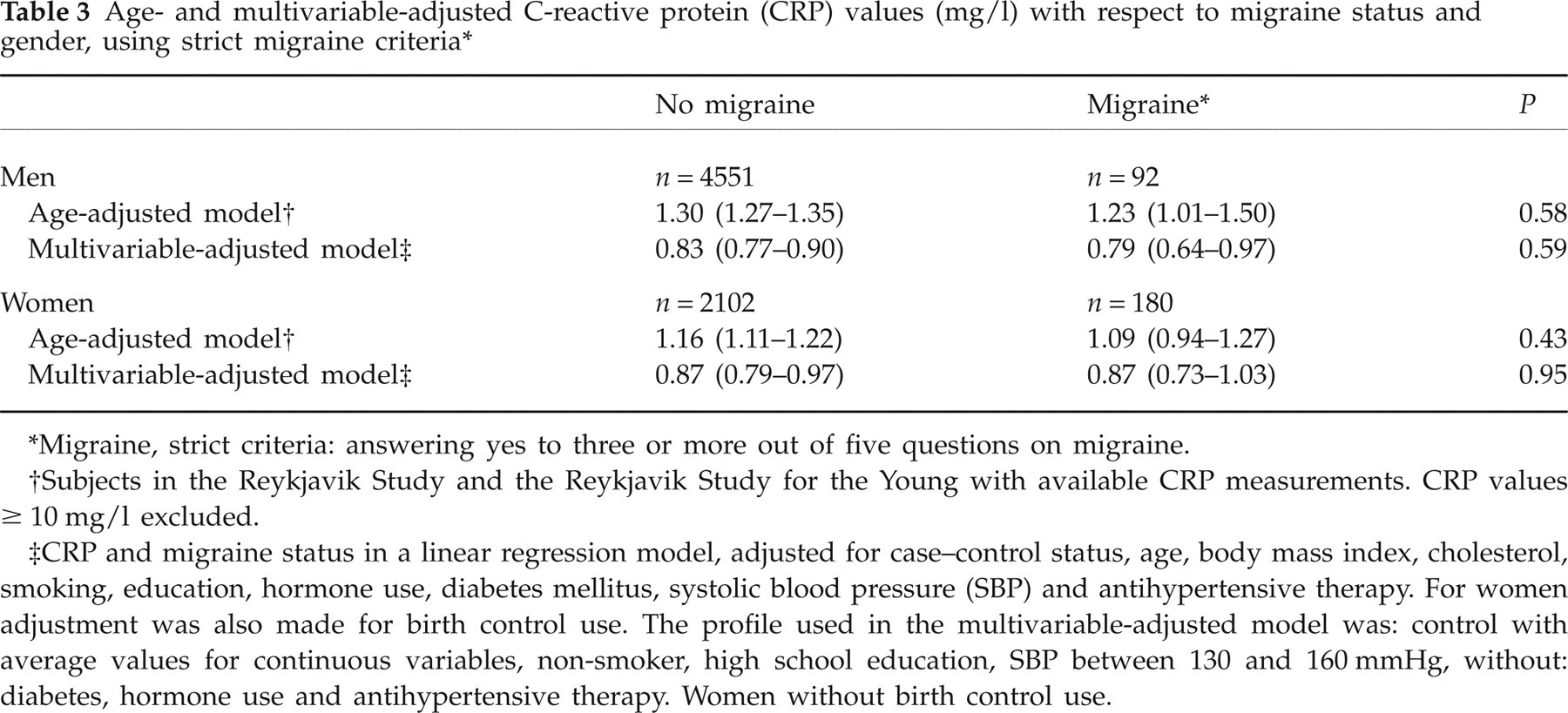
Migraine, strict criteria: answering yes to three or more out of five questions on migraine.
Subjects in the Reykjavik Study and the Reykjavik Study for the Young with available CRP measurements. CRP values ≥ 10 mg/l excluded.
CRP and migraine status in a linear regression model, adjusted for case–control status, age, body mass index, cholesterol, smoking, education, hormone use, diabetes mellitus, systolic blood pressure (SBP) and antihypertensive therapy. For women adjustment was also made for birth control use. The profile used in the multivariable-adjusted model was: control with average values for continuous variables, non-smoker, high school education, SBP between 130 and 160 mmHg, without: diabetes, hormone use and antihypertensive therapy. Women without birth control use.
Table 4 shows age- and multivariate-adjusted CRP levels by age. Adjusted CRP levels increased gradually with age. In men CRP levels were consistently lower for MO subjects, compared with non-migraineurs and MA, but the difference was not significant. In women non-migraineurs and women with migraine and aura, there was a gradual increase in CRP levels with age. Women with MO had higher multivariable-adjusted CRP values in the young cohort, age group 19–34 years (1.01 mg/l for MO vs. 0.81 mg/l for non-migraineurs, P = 0.08), but with increasing age the CRP levels for women migraineurs with no aura gradually decreased, and by age 60–81 years CRP levels were significantly lower compared with migraineurs with aura and non-migraineurs (0.52 mg/l MO vs. 1.01 mg/l MA, P = 0.029; 0.52 mg/l MO vs. 1.07 mg/l control, P = 0.007).
Age- and multivariable-adjusted C-reactive protein (CRP) values (mg/l) and migraine status relaxed criteria for men and women the Reykjavik Study and Reykjavik Study for the young

Migraine relaxed criteria defined as answering yes to two or more out of five questions on migraine. MA, migraine with aura; Migr., migraine; MO, migraine without aura. CRP, migraine status, age and gender in the Reykjavik Study and the Reykjavik Study for the Young using linear regression adjusting for case–control status, age, body mass index, cholesterol, smoking, education, hormone use, diabetes mellitus, systolic blood pressure (SBP) and antihypertensive use. Each age category was analysed separately and subjects with CRP values ≥ 10 mg/l were excluded. Profile used in multivariable adjusted model was: control with average values for continuous variables, non-smoker, high school education, SBP between 130 and 160 mmHg, without: diabetes, hormone use and antihypertensive therapy. Women without birth control use.
Results with and without subjects who developed a coronary event during follow-up were similar. The difference in multivariable-adjusted CRP values after excluding those who were later diagnosed with MI ranged from zero to 0.15 mg/l lower (0–19% decrease) for men and from being 0.14 mg/l higher to 0.05 mg/l lower (23% increase to 8% decrease) for women (data not shown).
Using 3 mg/l as a cut-off for elevated CRP levels, and excluding subjects with a history of MI, oral contraceptives and other medical hormone use, the proportion of men and women with elevated CRP was similar among subjects with and without migraine (19.3 and 20.0% for men and 14.1 and 16.6% for women). When stratifying by migraine aura, the proportions of non-migraineurs, MO and MA with elevated CRP were 20.0, 13.8 and 21.4% for men (P = 0.43, logistic regression with age adjustment) and 16.6, 9.7 and 16.8% for women (P = 0.43).
Discussion
In this nested case–control study, age-specific analysis of serum CRP levels was performed in subjects from two large cohort studies. Differences in CRP values between migraineurs and those without migraine were not significant, regardless of using linear regression with log-transformed CRP levels as a continuous variable or logistic regression with CRP as a binary variable. There was a moderate negative association between CRP levels and age among women with MO.
Inflammatory mechanisms have been involved in recent years and conceptualized in the neurogenic inflammation theory postulated by Waeber and Moskowitz (15). This theory accounts for the clinical efficacy of non-steroidal anti-inflammatory drugs and other anti-inflammatory drugs in aborting migraine attacks. Furthermore, inflammation being an important factor in atherogenesis and atherothrombosis, the association of migraine, especially MA, with stroke could be based on vascular inflammation as a link. The inference that inflammation is an important component in subsets of migraineurs has been further supported by the recent findings that the inflammatory marker, CRP, may be elevated in migraineurs (2, 5, 6). However, in all of those studies the elevation of CRP was modest at most and restricted to migraineurs without aura, a group that has not been generally found to be at increased risk of stroke (3, 16, 17). The principal finding of the present study is that CRP levels were not elevated among migraine sufferers compared with non-migraineurs. Nevertheless, certain subtleties require further consideration.
The multivariable-adjusted values in the Reykjavik Study were fairly low because they reflected subjects with a low cardiovascular disease risk profile. Age-adjusted CRP levels among men with MO were borderline lower than those of non-migraineurs and also borderline lower than those of migraineurs with aura, but after multivariate adjustment the differences between these groups were smaller and not statistically significant. CRP levels among migraineurs with aura and non-migraineurs were similar in all age categories for both men and women.
The association between migraine status, CRP and age seemed to be more complex in women than in men. Women with MO had slightly higher multivariable-adjusted CRP values in the age group 19–34 years. With increasing age the CRP level for women without aura fell and became significantly lower than that of non-migraineurs in the age group 60–81 years. Results for women in the age group 19–34 years are similar to those published by Vanmolkot and de Hoon (5). In their study 78% of participants were women, and the mean age was 25 years. The median CRP of those with MO was 2.11 mg/l, compared with 0.90 mg/l in controls (P = 0.0002). In a cohort study of women (mean age 55 years), the multivariable-adjusted prevalence odds ratio of having CRP > 4.2 mg/l was 1.14 (95% CI 1.02, 1.27) for active MO and 1.10 (95% CI 0.97, 1.26) for active MA vs. 1.00 for no history of migraine as control (6). This cohort study also showed a slight positive association between the attack frequency and the odds of having elevated CRP (6). Although it is clearly established that the prevalence of migraine is lower in older adults compared with middle-aged adults, it is not clear whether average attack frequency decreases with age. For example, Bigal et al. showed that the proportion of migraineurs with 10–14 headache days per month increased with age, and the attacks were less typical in elderly individuals (18), and Prencipe et al. have shown a similar attack frequency with increasing age among elderly migraineurs (19). In light of current knowledge on changes in attack frequency with age, it is not evident what effect age could have on CRP values among different age groups of migraineurs. Headache frequency was not estimated in the Reykjavik Study, so this question cannot be addressed.
In the present study, although not statistically significant, multivariable-adjusted CRP levels were generally higher in migraineurs with aura compared with migraineurs without aura, and this contradicts the results in the retrospective study of Welch and co-workers (2), the case–control study of Vanmolkot and de Hoon (5) and the cohort study of Kurth et al. (6).
The data in the current analysis from the Reykjavik Study were selected on the basis of availability of CRP measurements. The CRP measurements were part of a nested case–control study, thus raising the possibility of selection bias. However, we note that the age-specific prevalence of migraine in the sub-cohort used was almost identical to the prevalence within the entire cohort, which was randomly selected. This suggests that selection bias with regard to the presence of migraine is not more prevalent in the sub-cohort than in the entire cohort of > 21 000 subjects (8). Due to the selection criteria, there is probably an overrepresentation of cases with pre-existing coronary heart disease in the current study. When those who did not have a major coronary event during follow-up were analysed separately, CRP values were similar to those based on all cases. This indicates that the association between CRP values and migraine status is not different among subjects with coronary heart disease.
The questions in our study covered the most common migraine symptoms but did not include all of those identified in the 1988 IHS criteria (13). This is a cross-sectional analysis and cannot account for within-individual changes in CRP values. Furthermore, cross-sectional analysis cannot be used to determine any temporal relationship between CRP levels and the onset of migraine. Subjects entered the study from 1967 to 1991, resulting in a long period of sampling and analysis. However, a study of CRP and cardiovascular disease in subjects from the Reykjavik Study showed that the decade-to-decade consistency of CRP values was good (9). Alcohol use was not measured in the current study. Migraineurs have been reported to be less likely to consume alcohol than non-migraineurs (20), and alcohol is associated with a decrease in CRP levels (21, 22). Therefore, alcohol use is a potential confounder that cannot be adjusted for in the current study.
In the present study, contraceptive use among women resulted in a two- to threefold increase in CRP values (data not shown). Fröhlich et al. found that CRP levels among 844 women, in the MONICA Augsburg survey, aged 25–44 years, were 0.81 mg/l among non-users of oral contraceptives and 2.59 mg/l among oral contraceptive users (14). Our results for women without migraine are similar when multivariable adjustment (including oral contraceptive use) is applied (Tables 2 and 4).
In the present study migraine aura symptoms were more prevalent than expected, relative to MO symptoms (Table 4). Previous results suggest the proportion of migraineurs with aura symptoms to be 31% (20). A population-based study in the Netherlands (GEM Study) of 6491 adults aged 20–65 years reported the 1-year prevalence of migraine to be 25% for women and 7.5% for men (23). By combining MA and migraineurs both with and without aura in the GEM Study, the prevalence of MA, MO and unspecified migraine would be 7.5, 16 and 1.3%, respectively, for women and 2.3, 4.8 and 0.4% for men. In the present study the prevalence of MA and MO for women was 8.8 and 5.5%, and the prevalence for men was 3.6 and 1.3%. Comparing the prevalence in the Reykjavik Study and the GEM studies suggests that the prevalence of MA in the Reykjavik study is consistent with prior results, but the prevalence of MO may be underrepresented. One possible explanation could be recall bias when asked about headache symptoms, especially among milder migraine cases. This has been postulated by Liew et al., who found a higher lifetime prevalence of MA than MO among migraineurs in a population-based study of older men and women (24).
There is also potential misclassification of MO subjects as MA subjects, as some MO subjects experience non-specific visual disturbances during headaches, and the questions used do not distinguish between visual disturbances during the pain and before it starts. The effect of this misclassification might have been to attenuate or obscure possible differences in CRP levels between migraineurs with and without aura.
The questions on migraine symptoms in the Reykjavik Study did not allow for identifying subjects with cluster headache and chronic paroxysmal hemicranias, and these subjects were probably included with the migraineurs. Two studies by Remahl et al. did not show a difference in CRP values between subjects with cluster headache and subjects without headache, but these studies had few subjects (27 and 21 cases of cluster headache) and thus lacked the power to detect small differences (25, 26). The estimated prevalence of cluster headache and chronic paroxysmal hemicranias combined is < 0.5% (27, 28) and is therefore unlikely to affect our results to a great extent.
Information on the use of aspirin and statins was not recorded in this study, and both drugs can alter CRP values. However, we note that the use of these drugs was uncommon in the general population of Reykjavik between 1967 and 1991.
In conclusion, CRP levels were not increased among migraine sufferers compared with non-migraineurs. Migraineurs without aura tended to have lower CRP values than non-migraineurs and migraineurs with aura, except for young women migraineurs without aura, who had borderline higher CRP levels compared with migraineurs with aura and non-migraineurs. The association between CRP and migraine status was similar among those developing coronary heart disease during follow-up and those who did not.
Competing interests
None to declare.
Acknowledgements
We thank all the employees of the Icelandic Heart Preventive Clinic (Hjartavernd) for their skilful contribution to data collection. We thank Dr Jon Hersir Eliasson for helpful comments. This study was supported by the Icelandic Research Council and the University of Iceland Research Fund.
