Abstract
Background
The management and role of standard preventive treatments (SPTs) in patients co-treated with monoclonal antibodies (mAbs) directed towards calcitonin gene-related peptide (CGRP) has been poorly investigated. The present study aimed to prospectively compare the clinical profile of patients co-treated with SPTs and anti-CGRP mAbs with patients with anti-CGRP mAb monotherapy and to assess the possible SPT influence on their outcome. The SPT withdrawal or a new SPT prescription during the 12-month treatment period with anti-CGRP mAbs and their possible relation with comorbidities were also evaluated.
Methods
Our Italian multicentric, prospective observational cohort study enrolled patients with migraine receiving the first prescription of subcutaneous anti-CGRP mAbs. Only patients who completed the annual cycle of therapy were included in the analyses. At baseline, the population was divided into two groups: with (SPT+ patients) or without concomitant SPTs (SPT– patients). At baseline (T0), T6 (after six months of therapy) and T12 (at the end of the one-year treatment period), we collected migraine clinical data (monthly migraine days (MMDs) and/or the pain intensity, by a numerical rating scale (NRS)); disability (Migraine Disability Assessment (MIDAS) score); and the type and the presence of SPTs at baseline, the beginning of a new SPT or its withdrawal. The primary endpoint was to compare the clinical outcome (variation of MMDs at T6) of baseline SPT+ patients with that of baseline SPT– patients. Secondary endpoints were: (i) to describe the percentage of concomitant SPTs from T0 to T12 in the SPT+ group; (ii) to investigate the factors (i.e. comorbidities, demographics, migraine burden), if any, influencing the persistence of concomitant SPTs from T0 to T12; (iii) to evaluate whether baseline SPT presence influences pain intensity (NRS) and disability (MIDAS) at T0, T6 and T12.
Results
We enrolled 599 patients who started a new treatment with anti-CGRP mAbs. The analysis was conducted on 555 patients who started galcanezumab (260; 46.8%), erenumab 140 mg (167; 30.0%) or fremanezumab (128; 23.1%). Patients with baseline concomitant SPTs presented lower T0 MMDs than SPT+ patients (18.6 ± 7.8 vs. 20.3 ± 7.2; p = 0.007) and a lower MMD reduction from T0 to T6 (−10.4 ± 7.2 vs −12.4 ± 7.4, p = 0.007), reaching similar MMD numbers at T6 (p = 0.984). Baseline SPTs were not associated with MMD 50% response rate at T6 (odds ratio = 0.779, 95% confidence interval = 0.534–1.138; p = 0.205). Moreover, the changes in MIDAS score (p = 0.919) and NRS (p = 0.664) from T0 to T6 and T6 to T12 did not differ according to baseline concomitant SPTs. During the 12-month treatment period, anti-CGRP mAbs SPT+ patients progressively decreased from 35.0% at baseline to 28.8% at T6 and to 19.6% at T12. A comorbid condition, although neither MMD 50% response rate nor T12 MMDs, influenced the use of concomitant SPTs at T12 (odds ratio = 3.132, 95% confidence interval = 1.981–4.954; p < 0.001). The introduction of a new SPT during 12-month mAb therapy occurred only in seven subjects. Overall, 13% of patients reported at least one adverse event.
Conclusions
Our study confirms that concomitant SPTs at baseline do not influence the clinical outcome of CGRP mAbs after six months of treatment. A progressive withdrawal of SPTs during 12-month anti-CGRP therapy was observed, dissimilar among preventive classes, with persistence of SPTs at T12, mainly in patients with comorbid conditions.
This is a visual representation of the abstract.
Introduction
The impact of standard preventive treatments (SPTs) in patients co-treated with monoclonal antibodies (mAbs) targeting calcitonin gene-related peptide (CGRP) or its receptor has been poorly investigated so far (1,2).
In clinical practice, SPTs (including tricyclic antidepressants, beta-blockers, calcium antagonists, anticonvulsants and angiotensin-II receptor antagonists) are indicated for patients with frequent and/or disabling migraine attacks to reduce the frequency of monthly migraine days (MMDs) and/or the pain intensity, according to international guidelines (3,4). However, SPTs are frequently discontinued due to poor tolerability or lack of efficacy (5). Generally, the highest dropout rates occurred in patients treated with amitriptyline, valproate or topiramate, mainly due to adverse events, compared to placebo (6,7).
On the other hand, the mAbs directed towards the CGRP pathway demonstrated better tolerability and efficacy than SPTs (7,8). The Italian Medicines Agency (AIFA) requires that anti-CGRP therapy may be reimbursed only after ineffectiveness or poor tolerability, or contraindication to at least three classes of SPTs (i.e. tricyclic antidepressants, beta-blockers and/or anticonvulsants). The treatment continuation is contingent on achieving a ≥50% response rate (RR), measured by Migraine Disability Assessment Test (MIDAS) Score. In 2019, the European Headache Federation (EHF) guideline recommended anti-CGRP mAbs as a third-line treatment for migraine prevention in individuals with migraine and inadequate response, tolerability, or compliance to at least two classes of SPTs (9). During the last five years, several randomized controlled trials (RCTs) and real-world studies on anti-CGRP mAbs were published, leading to a revision of these criteria. The HER-MES study (10) and the APPRAISE study (11) comprised RCTs directly comparing an anti-CGRP mAb (erenumab) to SPTs, showing a superiority of erenumab with a more favourable tolerability and efficacy profile. A recent position statement of the American Headache Society (8) and an update of the EHF Guideline in 2022 (12) proposed anti-CGRP pathway mAbs as first-line treatment option, as they are effective and safe in the short and long-term.
In this scenario, the management of SPTs associated with anti-CGRP mAbs in patients with migraine is still controversial. Generally, no SPT withdrawal is required before initiating anti-CGRP mAbs therapy. In clinical practice, patients who start the treatment with anti-CGRP mAbs may encounter three options: (i) continuing the ongoing SPTs; (ii) being in monotherapy with anti-CGRP mAbs ab initio; and (iii) discontinuing SPTs before starting the anti-CGRP mAb. Less frequent is the case of patients who start SPTs during treatment with anti-CGRP mAb, aiming to optimize and/or maximize the mAb response.
The SPTs, unlike anti-CGRP mAbs, are not specific as they act on different brain targets not exclusively directed to the anti-CGRP network. They probably inhibit the cortical spreading depression by influencing ion channels and neurotransmission, modulating migraine attack genesis and pain processing (13). Accordingly, it is reasonable to hypothesize that a combined therapy (anti-CGRP mAbs plus SPTs) could enhance the clinical response more than either drug alone (multi-target polytherapy vs. target monotherapy). Furthermore, it is worthwhile investigating whether rapid and sustained improvement due to anti-CGRP mAbs can lead to a progressive withdrawal of SPTs. The role, if any, of SPTs as add-on therapy in patients treated with anti-CGRP mAbs remains uncertain.
The present study aimed to prospectively compare the clinical outcome of patients with SPTs at baseline versus that of those without, measured by the variation of MMDs at T6 (i.e. after six months of therapy) compared to baseline. The percentage of SPT withdrawal, maintenance or new prescription and the possible relation with comorbidities was also evaluated during the 12-month treatment with anti-CGRP mAbs.
Methods
Study design and population
We conducted a multicentric, prospective observational cohort study involving five headache centers across Italy (two in Rome, one each in Florence, Ancona and Milan) participating in the Italian Headache Registry (Registro Italiano delle Cefalee (RICe)) promoted by the Italian Society of the study of Headaches (SISC). This was a secondary analysis on previously collected data from the RICe study.
Inclusion and exclusion criteria
The present study included adult patients receiving the first prescription of anti-CGRP mAbs from January 2023 to October 2023 and with a follow-up until October 2024.
Patients with episodic (EM) or chronic migraine (CM), regardless of medication overuse headache (MOH), according to the International Classification of Headache Disorders criteria, 3rd edition (ICHD-3) (14), were consecutively enrolled.
Patients were excluded if:
they discontinued the anti-CGRP mAbs therapy (due to adverse events, ineffectiveness and loss to follow-up before six months of therapy); they had not filled out the headache diary three months before the enrollment; or during the observational period.
For the exclusion criteria, ineffectiveness was defined as the reduction of MIDAS scores of less than 50%, according to AIFA reimbursement criteria,
The three subcutaneous anti-CGRP mAbs (i.e. erenumab, galcanezumab and fremanezumab) were prescribed according to a headache specialist judgment considering AIFA reimbursement criteria, as described in the Introduction. Subjects received a galcanezumab loading dose of 240 mg in the first month and then 120 mg every month, fremanezumab 225 mg monthly or 675 mg quarterly, and erenumab 140 mg every 28 days. Eptinezumab was not reimbursed in Italy at the time of the study.
Variables collected
Patients were assessed at baseline by a headache expert with a face-to-face interview using a semi-structured questionnaire addressing sociodemographic characteristics, comorbidities and clinical migraine features. Patients were required to fill out a headache diary showing migraine details for the three months before enrolling and to fill it out during the observational period. Patients used an electronic or paper diary, based on patient preference, and maintained the same one throughout the period. Having been treated for a long time in the centers, patients had been previously instructed on how to use the diary.
The headache diary collected migraine-related data, such as the number of migraine days per month, the intensity of pain and the presence of aura.
At baseline, several data were collected: (a) demographics (sex, age, body mass index, BMI); (b) clinical data regarding migraine: migraine diagnosis, the presence of MOH and the MIDAS score; (c) the number and the type of past and concomitant oral SPTs, i.e. amitriptyline, anticonvulsants (topiramate, valproic acid, pregabalin and gabapentin), beta-blockers (propranolol, metoprolol and timolol), calcium antagonists (flunarizine and cinnarizine) and angiotensin-II receptor antagonists/sartans (candesartan, ramipril and lisinopril) and, in CM patients, also the use of onabotulinumtoxinA; (d) comorbidities: hypertension, psychiatric, cardio-cerebrovascular, endocrinological, gastrointestinal, autoimmune diseases and others (miscellaneous items); and (e) type of subcutaneous anti-CGRP mAbs.
According to the SPT use at the baseline, the population was divided into two groups: (i) patients starting anti-CGRP mAbs with concomitant SPTs (SPT+ patients) and (ii) patients starting anti-CGRP mAbs without concomitant SPTs (SPT– patients).
Given the observational nature of the study, during the observation period, headache specialists managed any prescription of SPTs according to their clinical practice. In the subset of CM patients co-treated with anti-CGRP mAbs and onabotulinumtoxinA, the latter was paid out of pocket by patients, according to Italian regulations. Adverse events were collected during the treatment.
At three time points, comprising baseline (T0), T6 (after six months of therapy) and T12 (at the end of the one-year treatment period), the following clinical evaluations were collected: the type and the presence of SPTs; the beginning of a SPT or its withdrawal; MMDs; pain intensity through numerical rating scale (NRS) values; and MIDAS score.
RR values were calculated as the percentage change in MMDs at evaluation times compared to baseline; a 50% RR indicates a reduction of at least 50% in MMDs.
Endpoints
The primary endpoint was to prospectively compare the clinical outcome of patients with SPTs at baseline with those without, measured by the variation of MMDs at T6 compared to baseline.
Secondary endpoints were: (i) to describe the percentage of concomitant SPTs from T0 to T12 in the SPT+ group; (ii) to investigate the possible factors (i.e. comorbidities, demographics, migraine burden) influencing the maintainance of concomitant SPTs from T0 to T12; (iii) to evaluate how SPT presence at baseline influences pain intensity (evaluated with NRS) and disability (MIDAS score questionnaire) at T0, T6 and T12, (iv) to compare outcome (MMD variation) in patients on stable concomitant SPTs during the first six months with patients without any SPTs during the same period (i.e. receiving only mAbs) in the first six months of therapy.
Protocol approval, registrations and patient consent
All patients provided written informed consent within the RICe study. The RICe study, including its subsection on individuals treated with anti-CGRP mAbs, was approved by the Ethics Committee of the University of Florence on 20 March 2019 (CEAVC Studio RICe, 14591_oss and subsequent amendments 2022-609; 2023) and mutually recognized by the other local ethical committees. The study was reported according to the Guidelines for Strengthening the Reporting of Observational Studies in Epidemiology (STROBE).
Statistical analysis
The analysis was performed on the available data (convenience sampling) including all consecutive outpatients, and no formal statistical power calculation was conducted. In the final analysis, we included only subjects with complete information regarding the primary studied variable (MMDs) according to study aims and design. For the secondary variables, we reported the available sample if different from the whole cohort in Table 1.
Baseline demographic and clinical characteristics of the whole cohort and between SPT+ patients and SPT– patients.

Legend: BMI = body mass index; CGRP = calcitonin gene-related peptide; MMDs = monthly migraine days; mAbs = monoclonal antibodies; MIDAS = Migraine Disability Assessment Score; NRS = numerical rating scale; MOH = medication overuse headache; SD = standard deviation. Percentages are expressed on column total. Statistically significant values are reported in bold. *The available sample is reported if different from the whole cohort.
Miscellaneous items were: heart rhythm disorders, multiple disc protrusions of the spine, tumors, epilepsy, bronchial asthma, endometriosis, hypovitaminosis D, osteoporosis.
Demographic and baseline characteristics were summarized descriptively: mean ± SD for interval variables and number (percentage) for categorical data.
The interval variables between groups were compared with an independent t-test (expressed as the mean ± SD). Contingency tables (chi-squared tests) and unadjusted odds ratios (OR) with their 95% confidence intervals (CI) were run to compare frequencies between groups. The McNemar test for proportions of paired samples was applied to assess changes in SPTs over the evaluation times. Data distribution to normality was assessed with the Kolmogorov–Smirnov test. The Mann–Whitney test or t-test were run to compare groups according to data distribution. We applied the Wilcoxon signed-rank test to analyze the interval variable without normal distribution.
To address the primary endpoint, we applied logistic regression to assess the possible factors (i.e. independent variables) influencing the change in MMDs at T6 compared to baseline (dependent variable). The independent variables included in the model (FORCED ENTRY approach) were the presence of baseline SPTs, demographics (age and sex) and comorbidities. The same forced entry approach was applied for secondary endpoints when comparing the outcome (baseline-T6 MMD change, dependent variable) in patients with stable SPTs in the first six months with those only on mAbs, including also demographics and comorbidities as independent variables. Binary logistic regression (forced entry) was also run to evaluate whether T12 MMDs, 50% RR, comorbidities, age and sex as independent variables influenced the use of concomitant SPTs at T12 (dependent variable). The independent variables included in the above-described models were selected based on clinical relevance to our endpoints and available evidence (2). p < 0.05 (two-tailed) was considered statistical significant. Statistical analyses were performed using SPSS, version 27.0 (IBM Corp., Armonk, NY, USA).
Results
The population enrolled in the present study is reported in Figure 1 (flow chart). We enrolled 599 patients who started a new treatment with anti-CGRP mAbs. The analysis was conducted on 555 patients who started galcanezumab (259; 46.8%), erenumab 140 mg (168; 30%) or fremanezumab (128; 23.1%) and completed the 12-month treatment. Among the 44 subjects excluded from the analysis, 20 discontinued mAbs for ineffectiveness within the first six months of therapy. Of these, 13 patients were treated with concomitant SPTs at baseline and seven patients were in monotherapy (p > 0.1). No patient was lost at follow-up or discontinued anti-CGRP mAbs after six months of therapy.

Number of patients on standard preventive treatments (SPTs) from T0 (baseline) to T12 (at the end of the one-year treatment period). CGRP = calcitonin gene-related peptide; mAbs = monoclonal antibodies.
Table 1 summarizes the baseline demographic and clinical characteristics of the whole cohort and of the two study groups: SPT+ patients and SPT– patients. At the baseline of 555 patients, 195 (35.1%) were SPT+ patients.
In the overall cohort (n = 555), among prior SPT failures, amitriptyline had been the most frequently used (96.2%), followed by anticonvulsants (94.8%), beta-blockers (85.2%), calcium antagonists (63.2%) and angiotensin-II receptor antagonists (3.4%). OnabotulinumtoxinA had been previously administered without clinically relevant effectiveness in 258 (CM) patients (46.5%).
Differently, at baseline in SPT+ patients, anticonvulsants (16.2%) were the most frequently used therapy, followed by beta-blockers (11.2%), amitriptyline (9.4%), calcium antagonists (3.1%), angiotensin-II receptor antagonists (2.2%) and, only in CM patients, onabotulinumtoxinA (5.8%).
In the overall cohort, all migraine-related clinical variables improved significantly from baseline to T12 (consistently with p < 0.001).
In the overall cohort (n = 555), MMDs significantly decreased from 19.7 ± 7.4 at T0 to 7.99 ± 6.3 at T6 and to 8.0 ± 6.6 at T12 (p < 0.001). At the same intervals, a reduction of pain intensity (NRS) from 8.3 ± 1.3 to 5.92 ± 1.8 at T6 and to 5.9 ± 1.7 at T12 (n = 369; p < 0.001) and a decrease of MIDAS questionnaire score from 92.3 ± 53.5 at T0 to 21.47 ± 19.9 at T6 and to 20.4 ± 20.3 at T12 (n = 541; p < 0.001) occurred.
As detailed in Table 1, SPT+ patients were more frequently women (odd ratio (OR) = 2.316, 95% confidence interval (CI) =1.351–3.971: p = 0.002), less frequently with CM (OR = 0.529, 95% CI = 0.354–0.798; p = 0.003) and with MOH (OR = 0.597, 95% CI = 0.419–0.959; p = 0.005). Baseline MMDs were lower in SPTs+ patients than in the SPT– group (p = 0.007). Those with SPT had more frequently comorbid conditions (OR = 1.980, 95% CI = 1.321–2.967; p = 0.001) than SPT– patients. Specifically, they were more frequently affected by psychiatric disorders (OR = 3.872, 95% CI = 2.337–6.415), gastrointestinal (OR = 3.765, 95% CI = 2.269–6.248), autoimmune (OR = 3.527, 95% CI = 1.284–9.689) and endocrinological (OR = 2.421, 95% CI = 1.403–4.177) diseases. In detail, the most common psychiatric disorders were anxiety, depression and bipolar disorders; irritable bowel syndrome and Crohn's disease as gastrointestinal disorders; psoriasis, Raynaud's phenomena and rheumatoid arthritis as autoimmune disorders; and thyroid alterations, dyslipidemia and pituitary adenoma as endocrinological disorders. Other comorbidities (i.e. miscellaneous items) included: heart rhythm disorders, multiple disc protrusions of the spine, tumors, epilepsy, bronchial asthma, endometriosis, hypovitaminosis D and osteoporosis.
Clinical outcomes of SPT+ patients vs. SPT– patients at baseline and T6
The presence of baseline concomitant SPTs was associated with a lower reduction in MMDs from T0 to T6 (primary endpoint; −10.4 ± 7.2 in SPTs+ vs. −12.4 ± 7.4 in SPT–; p = 0.007) (Figure 2). However, when adjusting for sex and the presence of comorbidities, this difference was no longer significant (p = 0.166).

Monthly migraine days ( MMDs) variations from baseline to T12 (at the end of the one-year treatment period) in patients with and without concomitant standard preventive treatments (SPTs). *p = 0.013.
In the subgroup analysis, patients with stable SPTs during the first six months (n = 160) presented a lower reduction in MMDs from T0 to T6 (−10.4 ± 7.2 in SPT+ vs. −12.4 ± 7.3 in SPT–; p = 0.004) compared to patients without concomitant SPTs up to T6 (receiving only mAbs). As above, when adjusting for sex and the presence of comorbidities, this difference was no longer significant (p = 0.214). Of note, patients with stable concomitant SPTs presented lower baseline MMDs (18.9 ± 7.8) compared to the T6 SPT– group (20.3 ± 7.8; p = 0.047).
For secondary endpoints, MMDs at T6 and T12 did not differ consistently between baseline SPT+ vs SPT– groups (p > 0.6). Moreover, the presence of SPTs at baseline did not influence the MMD 50% RR at T6 (OR - 0.779, 95% CI = 0.534–1.138; p = 0.205). Finally, the changes in MIDAS score (p = 0.919) and NRS (p = 0.664) from T0 to T6 and from T6 to T12 did not differ according to baseline concomitant SPTs. No difference was observed when considering the different types of anti-CGRP Mabs. Table 2 summarizes these findings.
Clinical outcomes (SPT+ patients vs. SPT– patients).

Legend: T0 = baseline (T0), T6 (after 6 months of therapy) and T12 (at the end of the treatment year); MMDs = monthly migraine days; MIDAS = Migraine Disability Assessment Score; NRS = numerical rating scale; SD = standard deviation; SPT = standard preventive treatments. Percentages are expressed on column total. Statistically significant values are reported in bold.
SPT longitudinal changes
Details on SPTs were fully available for the entire cohort from T0 to T12. The number of SPT+ patients decreased from 195 (35.1% of the whole cohort) at baseline to 160 (28.8%) at T6 (T6 STP+) and to 109 (19.6%) at T12 (T12 SPT+) (Figure 3). Anticonvulsants (from 90 patients at T0 to 79 patients at T6, p = 0.029), beta-blockers (from 62 patients at T0 to 50 patients at T6, p = 0.01) and amitriptyline (from 52 patients at T0 to 40 patients at T6, p = 0.003) significantly decreased from T0 to T6. From T6 to T12, anticonvulsants further decreased (from 79 patients at T6 to 68 patients at T12, p = 0.002); only amitriptyline use increased from T6 to T12 (from 40 patients at T6 to 56 patients at T12, p = 0.003). Of note, at T0, 65 patients (33.3% of SPT+ group) were on more than one concomitant SPT: 51 at T6 (26.2%) and 67 (34.3%) at T12.
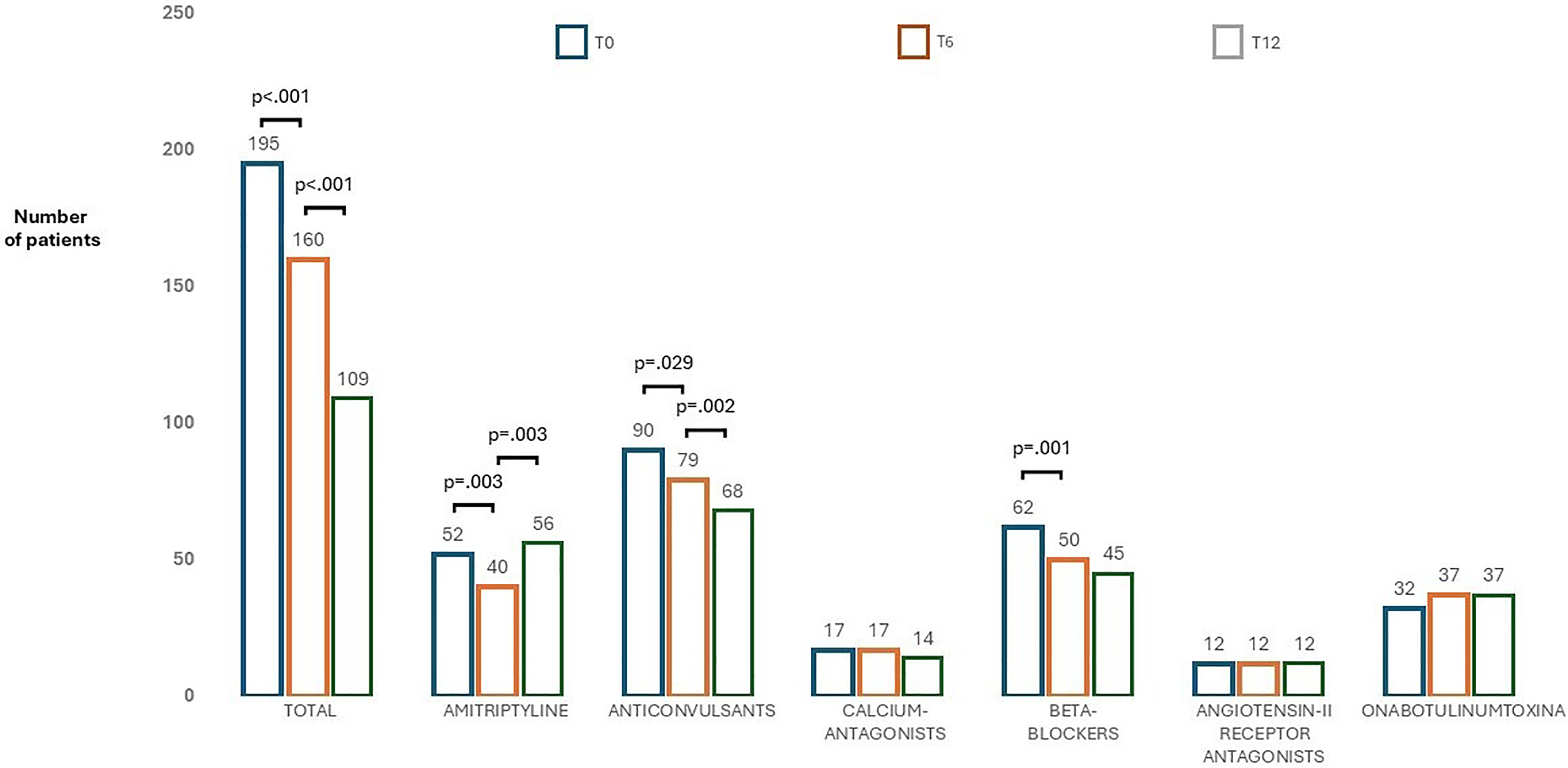
Number of patients treated with different standard preventive treatments (SPTs) classes from T0 (baseline) to T12 (at the end of the one-year treatment period).
Concomitant SPT+ at T12
In the regression analysis (Table 3 3), only the presence of a comorbid condition, differently from MMD 50% RR and T12 MMDs, influenced the use of concomitant SPTs at the end of the one-year treatment with anti-CGRP mAbs (OR = 3.132, 95% CI = 1.981–4.954; p < 0.001). This association remained significant also after correction for multiple comparisons (p < 0.001). Collinearity was observed between MMD 50% RR and T12 MMDs (correlation matrix 0.566). No effect was observed by the interaction among independent variables (p > 0.05, consistently). We analyzed the association between hypertension and psychiatric disorders with the presence of SPTs at T12 (T12 SPT+), more specifically (Table 4). We found that only psychiatric disorders were significantly associated with T12 SPT+ (p = 0.003, after correction for multiple comparisons). No collinearity or effect by the interaction among independent variables (p > 0.05, consistently) was observed.
Multivariate logistic binary forced entry analysis to assess factors associated with the use of concomitant STP at T12 in SPT+ patients (n = 195).
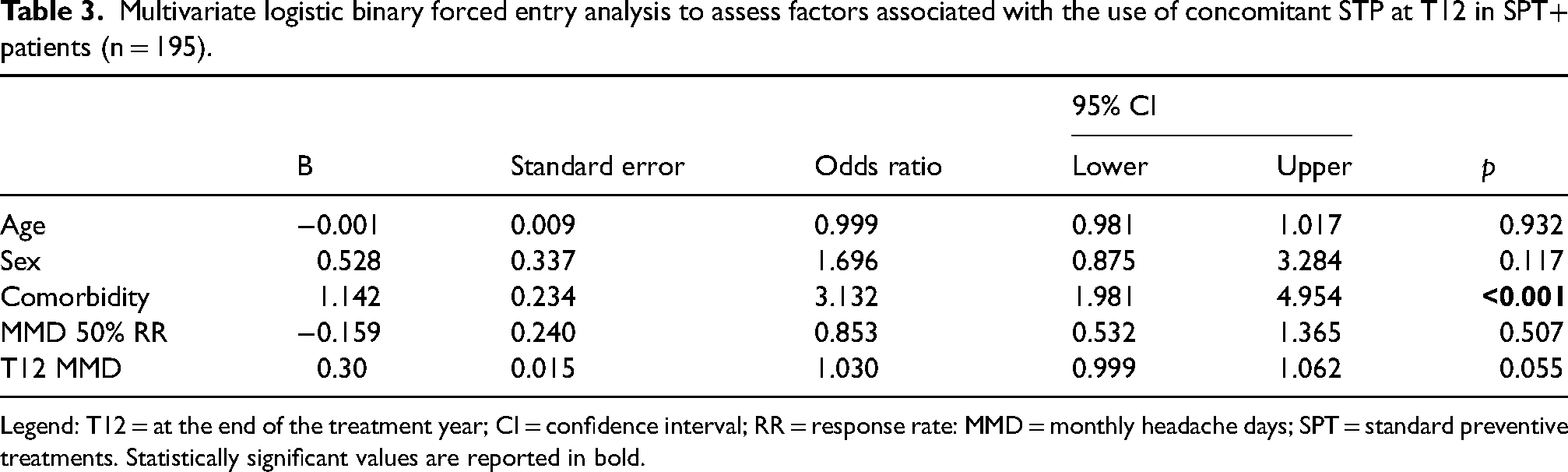
Legend: T12 = at the end of the treatment year; CI = confidence interval; RR = response rate: MMD = monthly headache days; SPT = standard preventive treatments. Statistically significant values are reported in bold.
Multivariate logistic binary; forced entry analysis to assess if hypertension and psychiatric disorders were associated with SPT presence at T12 in SPT+ patients (n = 195).

Legend: T12 = at the end of the treatment year; CI = confidence interval; MMD = monthly headache days; RR = response rate; SPT = standard preventive treatments. Statistically significant values are reported in bold.
Along the same line, we also assessed which type of T12 SPT was more often prescribed in patients with psychiatric disorders. They were amitriptyline (OR = 3.251, 95% CI = 1.729–6.110; p < 0.001), anticonvulsants (OR = 2.146, 95% CI = 1.133–4.063; p = 0.031) and beta-blockers (OR = 3.680, 95% CI = 1.874–7.229; p < 0.001).
SPT new introduction
The introduction of new SPTs during 12-month mAb therapy was rare; therefore, a separate analysis was not conducted. After six months of anti-CGRP treatments, a novel SPT was introduced in only seven subjects (four patients with amitriptyline, two anticonvulsants and one beta-blocker). Among them, three patients were not on SPTs at baseline.
Adverse events
In the overall cohort, 13.0% of patients reported at least one adverse event. The most commonly observed adverse events were fatigue (10 patients, 0.18%), constipation (33 patients, 5.9%), swelling and redness at the injection site (28 patients, 5.04%), alopecia/effluvium (two patients, 0.36%) and Raynaud phenomenon (two patients, 0.36%).
Three patients discontinued the therapy during the first six months due to serious adverse events (one for pulmonary thromboembolism and two for retinal thrombosis), which required hospitalization.
Discussion
Before the introduction of anti-CGRP drugs, migraine non-specific SPTs were the only available options to improve migraine clinical outcomes and disability. The RCTs on oral SPTs are limited and did not show strong and consistent efficacy for these drugs (15–17). Patients treated with SPTs often have poor therapeutic adherence and complain of a high incidence of adverse events (6).
By contrast, the monoclonal antibodies directed toward the CGRP pathway were demonstrated to have better tolerability and efficacy than SPTs (7,8), with strong and consistent evidence provided by both RCTs and real-world studies.
The correct integration between SPTs and mAbs is still debated, and its management is often left to the clinician's judgment. The recent International Headache Society guidelines (18) suggested that adding an oral SPT or onabotulinumtoxinA to anti-CGRP mAbs could exert a synergistic action (due to different mechanisms of action and indications) to fully manage migraine. Moreover, this combination could be an advantage in those patients who had a positive response to anti-CGRP mAbs, but still have a clinically meaningful residual migraine burden.
In the real-life setting, when starting an anti-CGRP treatment, the concurrent SPT treatment is usually retained (1). The decision is probably because most patients have ongoing long-term treatment, and clinicians may prefer to observe the effect of the new therapy before stopping SPTs. Similarly, patients may choose to continue a partially effective SPT before experiencing the potential clinical benefit of the new treatment. Previous studies (1,2) reported that half of patients in concomitant treatment continue the polytreatment before discontinuing SPTs. These data are confirmed by our results, in which at least one-third of patients started anti-CGRP treatment, maintaining an ongoing SPTs. Our primary endpoint, however, was to assess whether concomitant SPTs actually influence the outcome of migraine patients starting anti-CGRP mAbs.
In our study, concomitant SPTs at baseline did not influence the clinical outcomes at six months. Assessing the outcome in the short term (T6 instead of T12) minimized the bias from multifactorial conditions that may have influenced the outcome in the long term (1,2). Indeed, clinical evidence shows that most of the effectiveness of anti-CGRP mAbs is evident within six months (19).
At baseline, SPT+ patients were less frequently affected by CM and MOH, with lower MMDs but with more comorbidities than SPT– patients. Taken together, these findings suggest that SPTs could have some efficacy in reducing migraine burden before starting anti-CGRP mAbs. Nevertheless, SPT+ patients had a less pronounced decrease in MMDs from baseline to six months of therapy when the two groups had comparable MMDs. This suggests that the reduction of T0-T6 MMDs induced by anti-CGRP mAbs could represent the residual burden of migraine not effectively treated by SPTs, as indicated by other studies (18). On the other hand, the introduction of anti-CGRP mAbs in both groups reduced MMDs to a comparable level. It has been described that the action of anti-CGRP mAbs is primarily exerted downstream (i.e. peripherally outside the blood-brain barrier), while SPTs act mainly centrally (also within the blood–brain barrier) (13,20). In this view, we can hypothesize that the pretreatment with SPTs might have acted centrally, reducing migraine burden, whereas anti-CGRP mAbs further decreased MMDs acting peripherally. Differently, in SPT patients, anti-CGRP mAbs alleviated migraine, directly inhibiting the trigemino-vascular system and thus were able to decrease MMDs to levels as low as in the SPT+ group, overcoming the initial difference in MMDs at T0. If so, to further address the residual burden of migraine, other strategies need to be implemented, such as acting even more downstream to the peripheral action of anti-CGRP mAbs, addressing the second messenger signaling (e.g. cAMP and ATP-sensitive potassium channels) (21,22).
Similarly, despite the lower MMDs at baseline, patients with concomitant SPT at T12 had higher MMDs than those in anti-CGRP monotherapy at both T6 and T12. Furthermore, the presence of concomitant SPT at T12 was not associated with the decrease from baseline in MMDs and NRS score and, although patients with SPTs displayed a higher reduction in MIDAS from T0 to T12, their MIDAS scores at T12 were higher than in SPT– group (26.5 ± 24.4 vs. 18.9 ± 18.9).
Altogether, these observations demonstrate that patients with SPTs at baseline showed similar treatment responses at six months compared to those without SPTs at baseline. By contrast, SPTs appear to be continued as the consequence of an unsatisfactory response or in specific comorbid conditions.
To support this interpretation, the percentage of SPTs+ patients gradually decreased from T0 to T12. Moreover, only a very small percentage of patients needed to start a new SPT during the one-year treatment with anti-CGRP mAb. These data are in line with two previous studyies (1,2). In the first study (1), in a sample of 99 patients treated with galcanezumab, a progressive decrease of SPTs from 51% to 35% after one year was found. A second, more recent retrospective study reported that 43.8% of patients achieved a SPTs withdrawal: 20.8% a total withdrawal and 23.0% a partial withdrawal (i.e. a reduction of doses or the discontinuation of at least one of the SPTs).
The lower percentage of concomitant SPTs at baseline in our study (35.1%) compared to the above-mentioned studies (between 40 and 50%) could be explained by higher clinicians’ confidence in the efficacy of anti-CGRP drugs, which led to the withdrawal of SPTs even before starting them. It is also possible that there is variability between centers in therapeutic choices in the absence of specific guidelines.
Interestingly, in the overall cohort, the distribution of previous SPTs and ongoing SPTs was different: amitriptyline was the most frequently prior prescribed therapy (96.2% of patients), followed by anticonvulsants (94.8%), beta-blockers (85.2%), calcium antagonists (63.2%), angiotensin-II receptor antagonists (3.4%) and onabotulinumtoxinA (46.5%) in patients with CM. Among SPT classes ongoing at baseline, anticonvulsants (46.2%) were the most frequent, followed by beta-blockers (31.8%), amitriptyline (26.7%), onabotulinumtoxinA (16.4%), calcium-antagonists (8.7%) and angiotensin-II receptor antagonists (6.1%).
The SPT withdrawal was dissimilar among classes. Anticonvulsants and beta-blockers significantly decreased from T0 to T12, whereas the percentage of patients treated with angiotensin-II receptor antagonists, calcium antagonists and onabotulinumtoxinA remained approximately stable. Amitriptyline decreased from baseline to T6 but slightly increased between T6 and T12. This could be explained by three factors: (i) the intention to add amitriptyline to improve anti-CGRP mAbs response in those patients who did not reach an optimal response; (ii) the presence of concomitant conditions (such as insomnia, anxiety or depression, which are common comorbidities with migraine), which may benefit to be treated with amitriptyline; and (iii) the clinician decision to re-introduce a SPT in the last trimester of anti-CGRP treatment, before the anti-CGRP annual pause (that was mandatory, at the moment of the beginning of this study in Italy, now it is not required), aiming to avoid a potential rapid worsening of migraine frequency.
As shown in the PREVENAC study (2), a decrease in all SPTs (including amitriptyline) during the anti-CGRP treatment was observed. Anticonvulsants (valproic acid (66.6%) and lamotrigine (40.0%)) had the highest rates of total withdrawal. The same study evidenced that the percentage of withdrawal was significantly higher in responders to anti-CGRP compared to non-responders and that the main reasons for maintaining SPTs during anti-CGRP therapy were the partial effectiveness (28.6%) and the presence of comorbidities (27.4%). In detail, the presence of insomnia, hypertension, obesity, chronic migraine and medication overuse were the main causes of continuing co-treatment. Around 20% of patients in our population had at least one comorbidity, with a high rate of hypertension and psychiatric, endocrinological, gastrointestinal and autoimmune disorders. Even if the causal sequence of relationships (direct or reverse causality) or shared underlying predisposition between migraine and the comorbid condition is still debated, headache pain intensity and migraine frequency are associated with an increased risk for many conditions (23,24). Particularly, migraine frequency was associated with an increased risk for many conditions related to inflammation (psoriasis and allergy), psychiatric disorders (depression and anxiety), sleep alterations (insomnia), comorbid gastric ulcers/gastrointestinal bleeding, diabetes, asthma and allergies/hay fever (23). In our population, there was a high rate of these different comorbidities, probably because most patients were chronic with a heavy clinical burden. In our population, comorbid conditions were the only factor associated with concomitant SPTs that were ongoing at the end of the treatment (T12). More specifically, while hypertension explained only the use of beta-blockers and sartans, psychiatric disorders were related to different classes of SPTs (amitriptyline, anticonvulsants, and beta-blockers), highlighting the more difficult-to-treat profile of these patients. Of note, 30.0% (17/56) of those who were still on therapy with amitriptyline at T12 had a concomitant psychiatric comorbidity, and most of them (94.0%) were taking another concomitant antidepressant. In the same line, at T0, 65 patients (33.3% of SPT+ group; 11.7% of the entire cohort) were on more than one concomitant SPT, and concomitant polytherapy was still present at T6 in 51 patients, and at T12 in 67 subjects.
The presence of concomitant other types of mAbs for other diseases (adalinumab for Chron disease in one patient, denosumab for osteoporosis in one patient) was rare, and did not influence the continuation of anti-CGRP therapy or the change of the concomitant SPT for migraine.
The correct timing of the reduction and/or withdrawal of SPTs is not known. Previous studies had reported a withdrawal time more frequently at three to five months after the beginning of anti-CGRP therapy, followed by another interval of reduction between six and eight months (1,2). We are not able to evaluate the timing of withdrawal in our population because the monthly SPT variation was not the aim of our study, evaluating only two follow-up intervals (T6 and T12) with a final reduction from 35% to 19%.
Our study limitations are: (i) we did not consider the evaluation of SPTs presence at monthly time points but only at six and 12 months; (ii) we did not collected clinical data at three months of mAb therapy because in our opinion this period appears to be early to allow an assessment of the outcome of concomitant SPTs (19); (iii) due to the observational nature of the study, we could not standardize the method of stopping SPTs, which might have lead to heterogeneous results; (iv) we did not collect data about the development of new comorbidities during 12-month mAb therapy, focusing only on migraine SPT adjustments, and, finally (v) being an open-label study, we cannot rule out the cyclical exacerbations of migraine over time.
The strength of our study is that it is a prospective real-world study specifically designed to evaluate the contribution of SPTs on the clinical outcome of a large cohort of patients treated with anti-CGRP mAbs. Moreover, we have observed a very low rate of anti-CGRP mAbs discontinuation in the first six months (rarely due to ineffectiveness but mainly due to loss at follow-up and adverse events) and a very low rate of missing data.
In conclusion, our study has some clinical implications; it confirms and strengthens the evidence of a progressive reduction of SPTs during anti-CGRP mAbs treatment. However, SPTs still seem to have a role as a combination therapy in migraine prevention, mainly in those patients with comorbidities. The increasing use of anti-CGRP mAbs and the recent introduction of gepants as preventive treatment have changed the rules of migraine management, and the increasing overlap of different standard preventive therapies will need to be carefully studied. Further studies are needed to specifically address whether there are possible benefits of combining the central effect of standard preventives with the peripheral inhibition of CGRP, or whether, to gain an enhanced response, it is necessary to act even more downstream. To evaluate the role of SPTs in the long term, interventional randomized trials must be designed in which SPTs are added or removed in a standardized way.
Clinical implications
This prospective real-world study conducted on a large cohort of patients with migraine starting de novo anti-CGRP mAbs treatment, evidenced that the concomitant SPTs at baseline did not influence the clinical outcomes at six months.
Anti-CGRP mAbs treatment led to a progressive withdrawal of SPTs during the 12-month interval, different among preventive classes.
By contrast, SPTs seem to be continued as the consequence of an unsatisfactory response or in specific comorbid conditions.
Footnotes
Acknowledgments
The ‘Società Italiana per lo Studio delle Cefalee’ (SISC) is acknowledged for the ‘Registro Italiano delle Cefalee (RICe)’.
Author contributions
Luisa Fofi, Claudia Altamura and Fabrizio Vernieri had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis; designed the study; and drafted and revised the manuscript. Claudia Altamura performed statistical analysis. Luisa Fofi, Luigi Francesco Iannone and Fabrizio Vernieri performed administrative and technical support. All authors critically reviewed the manuscript, agreed to be fully accountable for ensuring the integrity and accuracy of the work, and read and approved the final manuscript submitted for publication.
Data availability statement
Data supporting the findings in the present study are reported in the article. The data collected and analyzed for the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: Luisa Fofi received honoraria for scientific presentations and travel fee from Novartis, Eli Lilly, TEVA, Pfizer, Organon, Abbvie and Lundbeck. Claudia Altamura is PI for a research collaborative study with TEVA; received honoraria for scientific presentations and/or advisory borad from TEVA, Eli Lilly, Pfizer, Abbvie and Lundbeck, and was supported with travel fee from Eli Lilly and Abbvie. Mariena Marcosano received honoraria for travel fees from Abbvie and TEVA Lundbeck. Nicoletta Brunelli received honoraria for scientific presentations and travel fees from Abbvie and Lundbeck. Luigi Francesco Iannone received honoraria for scientific presentations advisory board and travel fees from TEVA, Eli Lilly, Pfizer, Organon, Abbvie and Lundbeck. International Headache Society junior grant, unrelated to study results. Alberto Doretti received compensation for scientific consulting services and/or speaking activities from ABBVIE, Eli Lilly, Teva, Lundbeck, Novartis, Zambon, Neopharmed Gentili, Piam, Exeltis, Organon, Pfizer, Ipsen and Merz. Giovanna Vitichi has no conflict of interest. Francesco DeCesaris received honoraria for scientific presentations and travel fees from TEVA, Eli Lilly, Organon, Abbvie, Lundbeck, Geopharma and Aesculapius. Alessandro Alesina has no potential conflict of interest. Mauro Silvestrini has no conflict of interest. Marco Peresson has no conflict of interest. Fabrizio Vernieri received travel grants, honoraria for advisory boards, speaker panels or clinical investigation studies from Allergan/Abbvie, Angelini, Lilly, Lundbeck, Novartis, Organon, Pfizer and Teva.
Ethical statement
All patients provided written informed consent within the RICe study. The RICe study, including its subsection on individuals treated with anti-CGRP mAbs, was approved by the Ethics Committee of the University of Florence on March 20, 2019.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
