Abstract
Background
Monoclonal antibodies targeting calcitonin gene-related peptide (CGRP) or the CGRP-receptor have revolutionized the prevention of migraine. Despite their effectiveness, worries have surfaced regarding potential unwanted cardiovascular effects linked to the vasodilation function of CGRP, suggesting a potential influence on blood pressure (BP).
Methods
Studies were systematically retrieved from PubMed, Cochrane Database of Systematic Reviews, Web of Science, MEDLINE and EMBASE up to 1 May 2024. We focused on randomized controlled trials and observational cohort or case-control studies examining the impact of anti-CGRP(R)-monoclonal antibodies (mAbs) compared to control treatments on BP in patients with migraine. Two reviewers independently conducted study selection, data extraction and risk of bias assessment.
Results
The literature search yielded 693 articles. After removing duplicates and conducting screening, 22 full-text articles were evaluated, with only four studies meeting the inclusion criteria. Among these, only one study had a low risk of bias and reported elevated BP following initiation of anti-CGRP(R)-mAb treatment.
Conclusions
Although anti-CGRP(R)-mAbs offer substantial benefits for migraine prevention, the potential risk of increased BP requires attention. Despite the current limited evidence, clinicians are urged to monitor BP of migraine patients undergoing treatment with anti-CGRP(R)-mAbs and to remain aware of the increased risk of cardiovascular events in these patients.
This is a visual representation of the abstract.
Introduction
Monoclonal antibodies targeting calcitonin gene-related peptide (CGRP) (eptinezumab, fremanezumab, galcanezumab) or the CGRP-receptor (erenumab) are now accessible as preventive treatments for migraine. They can be administered once a month or once every three months. Erenumab, fremanezumab and galcanezumab can be self-administered subcutaneously, whereas eptinezumab requires intravenous administration. In clinical trials, approximately 40–60% of patients experienced ≥50% reduction in monthly migraine days, comparable to other non-specific migraine preventive medications (1–4). With a relatively low amount of treatment-emergent adverse events, anti-CGRP(R)-monoclonal antibodies (mAbs) are generally considered as a well tolerated treatment for migraine prevention (2).
The targeted peptide, CGRP, exerts potent systemic vasodilatory effects and influences blood pressure (BP), heart rate and myocardial contractility (5). Beyond its role in migraine pathophysiology, it is present in multiple organ systems, including the digestive tract, lungs, kidney, liver and adipose tissue (6,7). During myocardial infarction or stroke, CGRP plays a protective role by promoting vasodilation, thereby increasing blood flow and oxygen delivery to affected tissues (8,9). Consequently, it may mitigate the extent of tissue damage. Inhibiting CGRP, either through blocking its receptors or blocking the peptide, has the potential to exacerbate the deleterious effects of myocardial infarction or stroke (5). In rodents, genetic deletion of CGRP or receptor activity modifying protein 1 (RAMP1, one of the components of the canonical CGRP receptor) elevated baseline blood pressure in some studies (10–13). Contrastingly, a study with CGRP knockout mice did not show differences in baseline BP compared to wild-type mice. However, these mice did exhibit heightened susceptibility to angiotensin II-induced hypertension when compared to wild-type mice (14). Hence, inhibiting CGRP may potentially elevate the risk of hypertension, particularly in those subjects that have a higher cardiovascular risk (12,14).
Various network meta-analyses comparing anti-CGRP(R)-mAbs showed no difference in effectiveness or side effects (1,2). The guidelines from the European Headache Federation and the American Headache Society recommend using anti-CGRP(R)-mAbs as a first-line treatment option for migraine prevention (15,16). However, practical implementation of this recommendation is challenging because of the higher costs compared to other preventive medications.
Post-marketing case reports have raised concerns about elevated blood pressure associated with erenumab, with cases also reported to the Food and Drug Administration (FDA) (17). Because migraine is already associated with an elevated risk of myocardial infarction (odds ratio = 2.2, 95% confidence interval = 1.7–2.8) and stroke (odds ratio = 1.5, 95% confidence interval = 1.2–2.1) per se, these findings could have important clinical implications (18,19). Migraine with aura demonstrates an even stronger association with major cardiovasvcular disease (CVD) rates than obesity or unfavorable lipid levels, and a similar association as elevated systolic blood pressure or high total cholesterol (19,20). These findings emphasize the importance of monitoring cardio- and cerebrovascular risk in migraine patients. An important debate persists regarding the potential impact of anti-CGRP(R)-mAbs on blood pressure. Therefore, present study aimed to provide a comprehensive review of existing research on the impact of anti-CGRP(R)-mAbs on blood pressure and to provide advice on potential clinical implications.
Methods
The reporting of this systematic review was guided by the standards of the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) Statement (21). We searched for randomized controlled trials, observational (cohort or case-control) studies investigating the effect of treatment with anti-CGRP(R)-mAbs on BP in patients with migraine. We combined different keywords, controlled vocabulary terms (MeSH) and free terms following a predefined framework:
Population: Individuals diagnosed with migraine Exposure: mAbs targeting CGRP such as fremanezumab, galcanezumab, eptinezumab, or those targeting the CGRP receptor such as erenumab Comparator: Placebo or alternative comparative treatment Outcome: Change in blood pressure
Studies entered up to 1 May 2024 in PubMed, Cochrane Database of Systematic Reviews (CDSR), Web of Science (WoS), MEDLINE and EMBASE were retrieved with assistance of information specialists of the Walaeus library of Leiden University Medical Center. Full search strategies for all databases concerned are available in the supplementary material, Table S1. Retrieved references were managed with EndNote, version 20.1.2 (https://endnote.com).
Study inclusion and data extraction
Firstly, the titles and abstracts of studies identified from the search were reviewed and duplicate records were removed. The eligibility criteria followed the PECO (i.e. Population, Exposure, Comparator, Outcome) question. We predefined the following exclusion criteria: (1) Not in English; (2) Animal or otherwise preclinical study; (3) Case reports, meeting abstracts, editorials, commentaries, or articles with incomplete information; (4) Review; (5) No migraine group included; (6) Observational design studies without a placebo or comparative group; (7) No anti-CGRP treatment, or other than monoclonal antibodies; and (8) No reported blood pressure measurements before and after start of treatment (Figure 1).

PRISMA flow diagram (21). Reasons for exclusion: (1) Not in English; (2) Animal or otherwise preclinical study; (3) Case reports, meeting abstracts, editorials, commentaries, or articles with incomplete information; (4) Review; (5) No migraine group included; (6) Observational design studies without a placebo or comparative group; (7) No anti-CGRP treatment, or other than monoclonal antibodies; (8) No reported blood pressure measurements before and after start of treatment
Two review authors (BWHvdA and GMT for first selection of abstracts; BWHvdA and FCvW for second selection of full articles) independently extracted data using data extraction forms. Information was collected on participants (age, sex, baseline BP and cardiovascular (CV) risk profile, follow-up time, inclusion/exclusion criteria, study design, allocation procedure, number of participants), interventions (type of anti-CGRP treatment, number and explanation for any drop-outs), outcome measures (description of measures used, continuous/dichotomous nature) and results (point estimates and measures of variability, frequency counts for dichotomous variables, adjusted confounders). Data were collated and entered into Table 1. There was 99.7% agreement on study selection for the selection of abstracts and 92.8% for the second selection of full articles. Any disagreements were resolved by discussion and consensus.
Study characteristics of the included studies.
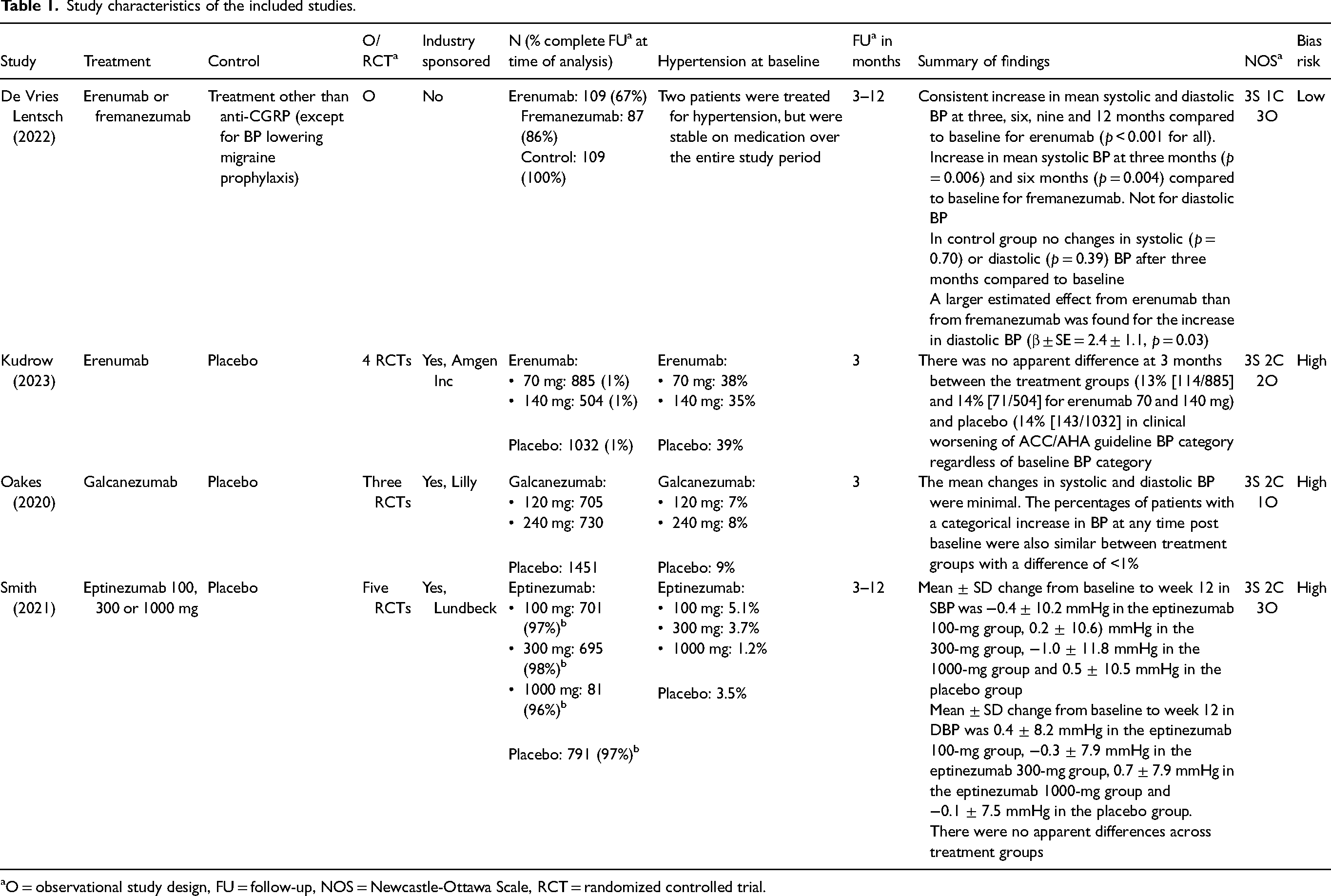
O = observational study design, FU = follow-up, NOS = Newcastle-Ottawa Scale, RCT = randomized controlled trial.
Quality assessment
In assessing the effect of anti-CGRP monoclonal antibodies on blood pressure, we selected the Newcastle-Ottawa Scale (NOS) for risk of bias evaluation (22). Although NOS is traditionally used for non-randomized studies and focuses on participant selection, comparability and outcome assessment, we applied it to both randomized controlled trials (RCTs) and observational studies in our review. The Cochrane Risk of Bias Tool standard for RCTs, emphasizes randomization and blinding, which are less relevant in the context where blood pressure was not the outcome of the RCTs. The NOS allowed for consistent assessment across study types. BWHvdA and FvW further customized the Risk of Bias assessment by incorporating additional fields that were not explicitly covered in the NOS scale, yet important for interpreting study findings. These added criteria encompassed of “sufficient sample size”, “distinction between anti-CGRP treatment”, “number of drop-outs specified”, “stable on anti-hypertensive medication” and “company funding”. The robvis tool was used to create risk-of-bias plots (23).
Results
Literature search results
The literature Search strategy retrieved 693 articles (Figure 1). After duplicate removal and screening on title and abstract of the retrieved studies, 24 full-text articles were assessed for inclusion. Three studies did not have a full-text available. Only four studies met the pre-specified inclusion criteria (24–27). Reasons for exclusion of the remaining 17 screened articles are reported in Figure 1 and the supplementary material, Table S2 (28–43).
Characteristics of included studies
Only four studies, which reported on blood pressure and treatment with anti-CGRP(R)-mAbs compared to a control treatment in patients with migraine, could be included (24–27). Author, year, study population, study design, anti-CGRP(R)-mAb treatment and follow-up time are presented in Table 1. The included studies were published between 2020 and 2023.
In 2022, De Vries Lentsch et al. (24) conducted a real world data (RWD) study using electronic patient records and daily e-headache diary data from the Leiden Headache Center. The study focused on patients treated with erenumab (n = 109), fremanezumab (n = 87) or standard of care (n = 109). BP measurements (mmHg) were collected before treatment initiation and every three months thereafter. The control group consisted of migraine patients who did not use any migraine prophylactic treatment or other medication that would possibly influence their BP. Patients with hypertension were excluded. A consistent increase was found in mean systolic and diastolic BP at three, six, nine and 12 months compared to baseline for patients on erenumab and an increase in mean systolic BP but not diastolic BP at three and six months compared to baseline for patients on fremanezumab. This contrasted with the control group, where no changes were observed in systolic or diastolic BP after three months compared to baseline. A larger estimated effect from erenumab than from fremanezumab was found for the increase in diastolic BP (24).
In 2023, Kudrow et al. (25) pooled from four randomized controlled trials (RCTs) involving patients treated with erenumab at doses of 70 (n = 885) or 140 mg (n = 504) compared to placebo (n = 1032). BP measurements were divided into categories according to the American College of Cardiology/American Heart Association guideline (Table 2). BP categories were defined as normal (systolic blood pressure (SBP) <120 mmHg and diastolic blood pressure (DBP) <80 mmHg), elevated (SBP 120–129 mmHg and DBP <80 mmHg), stage 1 hypertension (SBP 130–139 mmHg or DBP 80–89 mmHg) and stage 2 hypertension (SBP ≥140 mmHg or DBP ≥90 mmHg). Before entering in the placebo group, 13% of participants (n = 138) were classified with elevated BP, 31% (n = 316) with stage 1 hypertension and 8% (n = 82) with stage 2 hypertension. Before receiving erenumab 70 mg, 12% (n = 105) had elevated BP, 31% (n = 271) had stage 1 hypertension and 7.5% (n = 66) had stage 2 hypertension. In the erenumab 140 mg group, 17% (n = 84) of participants exhibited elevated BP, 29% (n = 148) had stage 1 hypertension and 6% (n = 29) were classified with stage 2 hypertension. Importantly, there was no data available on the use of or changes in antihypertensive medication during the study. Shifts in BP categories from baseline were summarized for all groups. The analysis was entirely descriptive with no formal statistical inferences. There seemed not to be a difference in the proportion of patients who moved to a higher BP category from baseline to months 1–3 of treatment treated with erenumab 70 or 140 mg (13% and 14%, respectively) compared to patients who received placebo (14%) (25).
Categories of blood pressure according to the American College of Cardiology/American Heart Association (ACC/AHA) guidelines.

In 2020, Oakes et al. (26) pooled data from three RCTs involving patients treated with galcanezumab 120 mg/month (n = 705), galcanezumab 240 mg/month (n = 730) or placebo (n = 1451) during at least three months. BP data were collected as continuous variables and used to evaluate the mean changes from baseline to the final follow-up visit. The exact timing of this final follow-up visit, however, varied among patients, and it was not further specified in the study, nor was the median follow-up duration mentioned. Data collection spanned up to a maximum of six months. At baseline, in the placebo group n = 135 patients (9%) had hypertension and 128 (9%) were on antihypertensive medication. In the galcanezumab 120 mg group, 47 patients (7%) had hypertension before starting the treatment, all of whom were using antihypertensives. In the galcanezumab 240 mg group, 56 patients (8%) had hypertension and 49 (7%) were on antihypertensives. During the follow-up period, the use of antihypertensive medication did not remain stable. In the placebo group, 27% (34/128) of patients who were treated required an increase in antihypertensive medication. For the galcanezumab 120 mg group, 23% (11/47) patients experienced an increase in antihypertensive medication usage. Similarly, in the galcanezumab 240 mg group, 25% (12/49) had an increase in antihypertensive medication. For the primary analysis changes from baseline to the final follow-up measurement during treatment were compared between treatment groups using a Poisson regression model, incorporating study, treatment, subgroup and the treatment-by-subgroup interaction as co-variables. An offset term of time at risk was used. No meaningful differences were observed in the mean change in BP between the groups. In addition to these continuous BP analyses, the proportion of patients with treatment-emergent hypertension was also compared between groups. Hypertension was defined as treatment-emergent high systolic BP of ≥140 mmHg with an increase of ≥20 mmHg or diastolic BP of ≥90 mmHg with an increase of ≥10 mmHg. No difference between the groups was found (26).
In 2021, Smith et al. (27) pooled data from five RCTs on eptinezumab 100 mg (n = 701), 300 mg (n = 695) and 1000 mg (n = 81) versus placebo (n = 791) over a one-year period. Resting systolic BP, diastolic BP and pulse rate was collected at each study visit, with mean ± SD values calculated. Regarding CV risk factors, 31.3% of patients were obese, and about half of the population was aged ≥40 years. Additionally, about 48% of the patient population had one or more CV risk factors and 13% had two or more. At baseline, the prevalence of hypertension was 5.1% in the eptinezumab 100 mg group, 3.7% in the eptinezumab 300 mg group, 1.2% in the eptinezumab 1000 mg group and 3.5% in the placebo group. Patients were allowed to increase or start new CV-related concomitant medication, such as antihypertensives. The available data on the number of patients who required adjustments to their antihypertensive medication are unclear, with varying terminology used to describe increases in blood pressure. In total, three patients in the eptinezumab 100 mg group, none in the eptinezumab 300 mg or 1000 mg groups, and two patients in the placebo group were reported as needing to modify their antihypertensive treatment. These patients were not excluded from the analysis. There were no clinically significant differences across treatment groups in mean changes in BP; however, there is no description of the statistical test used to assess these differences. Mean ± SD change from baseline to week 12 in systolic BP was −0.4 ± 10.2 mmHg in the eptinezumab 100 mg group, 0.2 ± 10.6 mmHg in the eptinezumab 300 mg group, −1.0 ± 11.8 mmHg in the eptinezumab 1000 mg group and 0.5 ± 10.5 mmHg in the placebo group. The findings were consistent at subsequent time points, including weeks 24 and 56, and also extended to diastolic BP (27).
Quality assessment
Overall, the studies demonstrated high risk of bias, except for De Vries Lentsch et al. (24) (Figure 2). De Vries Lentsch et al. (24) was rated as missing two stars in the NOS scale because of the utilization of an external control group and for not including additional covariates in the analysis (see supplementary material, Table S3). Moreover, the study included individuals with migraine from the same headache center, ensuring a comparable distribution in sex, age and migraine diagnosis, as well as consistency in BP measurements, with clinicians following the same protocol and cut-off values. Although this approach enhances internal consistency, it may decrease the generalizability of the findings compared to multicentre studies. Furthermore, the comparability of groups in the study missed a point on the NOS scale as a result of certain limitations in the mixed model used. Time and treatment were included as fixed effects and the patient as a random effect. Other covariates such as sex and age were not included because these were comparable between the groups and thus would not have changed the outcomes but might have led to overfitting of the model. De Vries Lentsch et al. (24) could have considered to provide a more detailed rationale for not including these covariates. Additionally, separate mixed models were used for the control and treatment groups. Thus, the magnitude of the effect should be interpreted with caution.
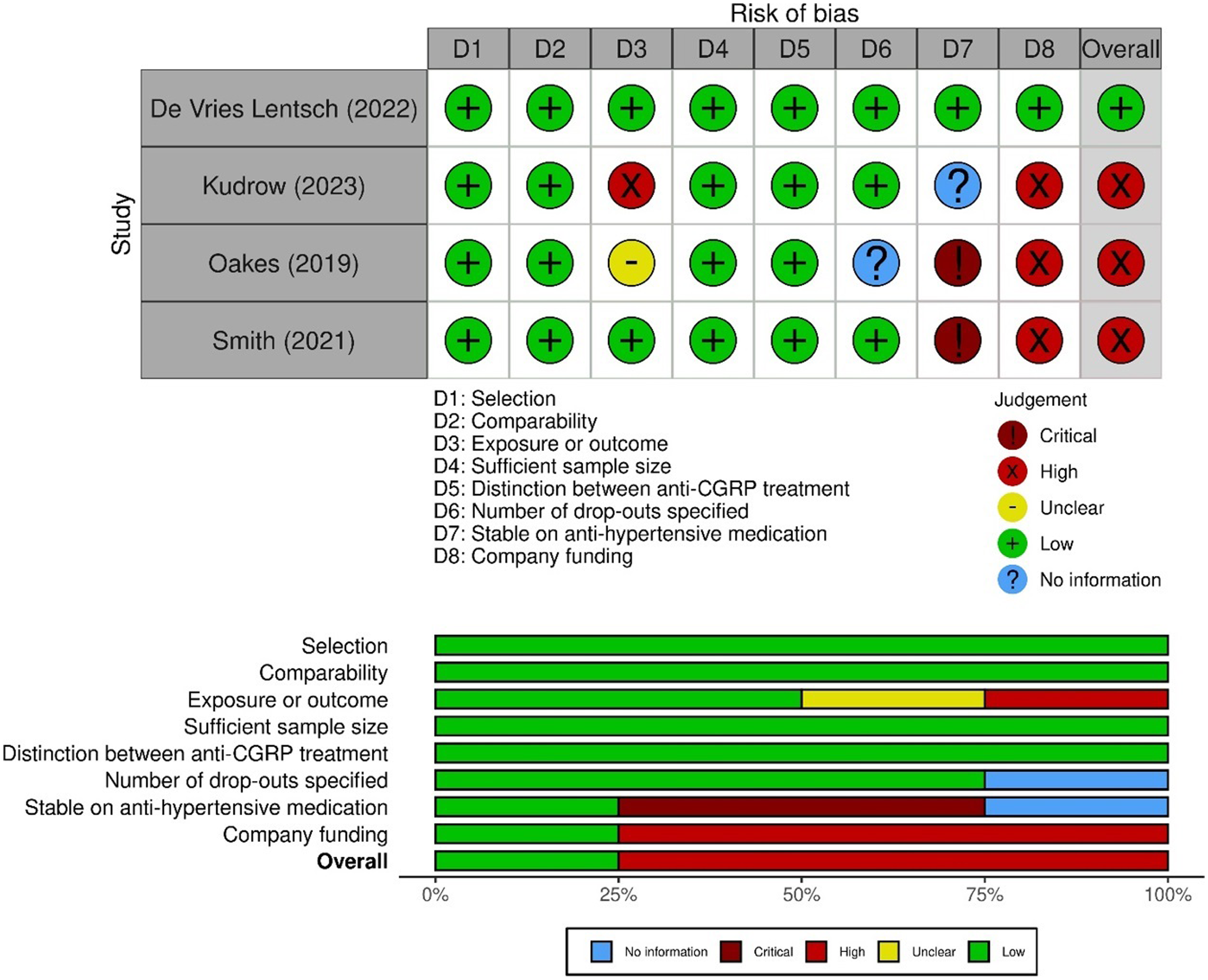
The risk of bias assessment related to the items “selection”, “comparability” and “exposure or outcome”, shown in the graph above, was performed on the basis of the Newcastle-Ottawa scale (NOS). In addition, the judgment for the risk of bias related to concerning “sufficient sample size”, “distinction between anti-CGRP treatment”, “no anti-CGRP treatment at baseline”, “number of drop-outs specified”, “stable on anti-hypertensive medication” and “company funding” is also included in the graphical representation. The robvis tool is used to create risk-of-bias plots. 23
A major strength is that these patients were not using any migraine prophylactic treatment or other medications that could potentially influence blood pressure. Given these efforts to establish comparability between the groups, the study's design appears robust, minimizing the risk of confounding bias. Another major strength is that this study excluded patients from the analysis after the start of anti-hypertensive treatment and only normotensive patients were included at the start of this study. By contrast, this is an important limitation observed in the other three included studies because they failed to report on or exclude BP measurements taken after the commencement of anti-hypertensive treatment, and also patients with higher BP at start were included. This may mask the true effect of anti-CGRP treatment on blood pressure. The study by De Vries Lentsch et al. (24) was also the only one to provide a statistical power analysis.
Kudrow et al. (25) took a different approach by examining changes in blood pressure categories rather than focusing on absolute changes. Although this method may offer some insights, it introduces a potential loss of statistical power and may compromise the study's ability to accurately capture changes in BP. By categorizing data, the study may overlook “mild” changes in continuous BP values, which could lead to less precise conclusions. Furthermore, the analysis conducted by Kudrow et al. (25) was entirely descriptive, lacking any adjustments for possible confounding variables. Without accounting for potential confounders such as age, gender, comorbidities, or medication use, the validity of the study's conclusions may be limited. Finally, the insufficient or lack of data on antihypertensive medication use could have significant effects on measured outcomes.
Oakes et al. (26) lacked reporting on the numbers of patients that were included in the analysis. No drop-outs or missing data was reported with regards to BP readings. The apparent discrepancy in participant numbers between the reported total sample sizes for galcanezumab 120 mg/month, 240 mg/month and placebo in the study (n = 705, n = 730 and n = 1451, respectively) and the numbers stated in the supplemental material (Table S3) (n = 705, n = 709 and n = 1410) may add to this concern. Furthermore, exact data (mean ± SD) on the mean changes in BP were not provided and only the outcome of the regression model was presented. The most important methodological concern is that patients were allowed to increase or initiate new cardiovascular-related concomitant medications, such as antihypertensives, during the treatment period. This factor influences the outcome (BP) and should have been considered a confounding variable. Alternatively, BP values obtained after the initiation of antihypertensive medication should have been excluded from the analysis. Oakes et al. (26) used a Poisson regression model, in which the last follow-up BP measurement was taken during treatment, but this follow-up period varied between patients. A more nuanced statistical method, such as a mixed model with the patient included as a random effect, could provide a more refined analysis of the data by accounting for individual patient variability and the varying follow-up periods.
Smith et al. (27) appear to have several methodological shortcomings that warrant attention. First, the study lacks a clear description of the statistical methods employed, as only descriptive BP values (mean ± SD) are provided in the methods section. The study's conclusion that there were no clinically relevant differences in BP between eptinezumab and placebo seems based solely on descriptive data. As for the other above described studies of Kudrow et al. (25) and Oakes et al. (26), an important concern is the lack of adjustment for potential confounders, particularly anti-hypertensive medication. The study did not account for the concomitant use of anti-hypertensive medication, which was initiated by 2.6% of participants in the eptinezumab 100 mg group, 3.2% in the eptinezumab 300 mg group, 6.2% in the eptinezumab 1000 mg group and 2.3% in the placebo group, as indicated in the supplementary meaterial to the manuscript. By including BP values of these patients in the analysis without adjusting for the influence of anti-hypertensive medication, the authors may have underestimated the true effect of eptinezumab on BP.
Discussion
This systematic review serves as a comprehensive overview of the existing evidence, presenting findings from studies that specifically investigated the influence of anti-CGRP(R)-mAbs on blood pressure changes in individuals diagnosed with migraine. It helps to address the ongoing debate and emphasizes the importance of evidence-based answers to clinical questions, particularly given the increased cardiovascular risk profile of migraine patients.
Notably, only the RWD study stands out because it explicitly investigated changes in blood pressure as the primary outcome and had a low risk of bias (24). This study, with data from the Leiden Headache Center, was also co-authored by some of our own authors (BWHvdA, AMvdB and GMT). Despite this, we maintained a rigorous and systematic review approach to minimize potential bias. Our review process included strict adherence to predefined inclusion and exclusion criteria, and a thorough assessment of the methodological quality of each study, ensuring objectivity. Additionally, our review was independently verified by external experts to confirm the robustness of our results. The study by De Vries Lentsch et al. (24) found a mean increase of 5.2 mmHg in systolic BP and 3.5 mmHg in diastolic BP associated with the use of erenumab and fremanezumab, even in normotensive patients without CVD. This may seem rather mild, but it is crucial to recognize the clinical consequences of even small changes in BP. Although the consequences of a diagnosis of hypertension are well-documented, even modest increases in BP resulting from treatment could have implications, particularly given the continuous relationship between BP and vascular mortality (44). For example, a mere 5 mmHg reduction in systolic BP has been shown to lower the risk of non-fatal cardiovascular events by approximately 10%, irrespective of previous diagnoses of CVD, and even at normal or high–normal blood pressure values (45,46). This reduction of CV risk is particularly notable in participants aged 55 years or younger, who are most representative of patients with migraine (45,46). Additionally, patients with migraine are known to have an increased CV risk, especially women diagnosed with migraine with aura; however, this specific risk factor was not delineated in the studies reviewed (47,48). Because BP and vascular mortality have a continuous relationship, and CGRP provides a key compensatory mechanism against hypertension, we speculate that blocking CGRP may be particularly problematic for at-risk subgroups (12,44). These include individuals with other cardiovascular risk factors or a family history of cardio- or cerebrovascular disease. Hence, seemingly modest elevations in BP associated with erenumab and fremanezumab should be considered as clinically relevant in patients with migraine and warrant careful consideration and monitoring to ensure optimal cardiovascular health outcomes.
The other studies did not find an increase in blood pressure (25–27). As indicated in the risk of bias table, these studies varied in their methodological quality. Methodological differences could explain the divergence in results; for instance, categorized blood pressure variables might have less power to detect differences (25). Additionally, the other studies did not account for the initiation of antihypertensive treatment and involved patient populations with a high percentage of hypertension, potentially masking the true effects of anti-CGRP(R)-mAbs on blood pressure (25–27). In conclusion, these studies should be interpreted with caution because of their high risk of bias. Several excluded studies focused on hypertension AEs or the incidence rates of high systolic BP, comparing these to placebo. However, these studies were excluded from this review because they did not investigate the absolute change in BP and often did not report the applied definition of hypertension (29,33,36–38,43,49). We also note that multicentre studies involving multiple local principal investigators may be more susceptible to variability in labelling BP increases as clinically relevant. This interpersonal variability can affect the consistency and reliability of BP data. Additionally, industry-driven RCTs, although strictly regulated, may tend to overlook side effects because of the administrative burden associated with identifying and reporting adverse events. For example, constipation was underreported in multicentre erenumab trials compared to post marketing real world data studies, suggestive for a risk of overlooking other side effects (50–54). Among 222 novel therapeutics approved by the FDA from 2001 through 2010, 32% were affected by a post market safety event (55). The case of levetiracetam, where significant neuropsychiatric side effects were detected only post-marketing, highlights this issue (56). These findings underscore the importance of thorough and ongoing safety monitoring for newly approved therapeutics.
An excluded RWD study worth mentioning is that by Wang et al. (40), which examined changes over a period ranging from six months to four years of treatment with a broad range of anti-CGRP treatments (mAbs and gepants analysed as one group) compared to topiramate. The results showed an increase in SBP over 0.5–4 years for the anti-CGRP group, although this was not different when compared to topiramate. They observed an annual 3.7% rise in antihypertensive medication use in hypertensive patients on anti-CGRP treatment compared to topiramate, suggesting potential BP changes. However, the absence of a comparison between follow-up blood pressure and baseline measurements, as well as the lack of distinction between gepants and mAbs, in this study appears to represent a missed opportunity for directly assessing the impact of anti-CGRP initiation on BP. Another RWD study by Chhabra et al. (42) was excluded by lack of a control group. This study examined the change in BP before and after erenumab treatment and reported an increase in BP after initiation of erenumab. Even with categorization of BP values according to American College of Cardiology/American Heart Association (ACC/AHA) guideline, instead of using continuous outcome variables, significance was observed. There was no association between pre-existing hypertension and increase in BP and they reported an increase in anti-hypertensive drugs (increase in dose or start of medication). However, because of the lack of a control group, it is uncertain whether this increase is a result of the erenumab treatment alone. Similarly, another recent RWD study by Guerzoni et al. (41) lacked a control group and had methodological shortcomings. It was reported found that 5.7% of patients receiving anti-CGRP(R)-mAbs experienced a “meaningful” increase in BP for which anti-hypertensive treatment was started during follow-up, although the precise definition of “meaningful” remained unspecified. They also examined continuous BP data over a treatment period of three, six, nine and 12 months, but did not exclude these patients who started anti-hypertensive treatment. No notable changes were observed in continuous BP data over time, but Guerzoni et al. (41) did not specify the statistical tests employed, and it appears that they did not adjust for potential confounders, such as the initiation of anti-hypertensive medication. In summary, although these RWD studies provide some insights into the effects of anti-CGRP(R)-mAbs on BP, the absence of control groups and methodological limitations, including the lack of statistical adjustments and unclear definitions, necessitate cautious interpretation of their findings.
A strength of our systematic review is the thorough literature search through multiple databases, ensuring a comprehensive inclusion of all relevant papers pertaining to the impact of anti-CGRP(R)-mAbs on BP. We were mindful of potential publication bias and therefore took measures to minimize its impact by applying well-defined criteria for study inclusion and conducting a thorough evaluation of the methodological quality of the included studies, contributing to the quality of our review. A limitation of our review is that we included only papers on human studies. However, our choice was driven by the intention to directly translate our findings to clinical practice. Second, it is important to acknowledge the limitations of the current evidence based method because many studies did not primarily focus on the absolute effects of BP, but were more focused on the efficacy of the drug and the reporting of adverse events in general (29,33,36–38,43,49). Furthermore, the limited number of included studies and the absence of reported point estimates by Kudrow et al. (25) and Oakes et al. (26) precluded the possibility of pooling data into a meta-analysis.
Given the context of the increased CV risk in patients with migraine, healthcare providers should exercise caution when managing BP in patients undergoing anti-CGRP(R)-mAb treatment. Regardless of anti-CGRP(R)-mAb treatment, adhering to the new guidelines of maintaining BP targets ≤130/80 mmHg for patients younger than 65 years and ≤140/90 mmHg for patients aged 65 years or older is important to decrease potential CV risks (57,58). It is equally important not to be overly anxious or dismissive regarding the use of anti-CGRP(R)-mAbs, considering both the advantages and disadvantages in the context of individual patient profiles. In cases of low-risk grade 1 hypertension, characterized by BP levels of 140–160/90–100 mmHg, initiating single antihypertensive medication is typically advised (57). However, the decision to start antihypertensive treatment in young migraine patients with office BP between 130–140/80–90 mmHg may be challenging because it has only been studied in patients with known CVD. Although some general practitioners may hesitate to initiate antihypertensive treatment in this low-risk group, meta-analyses from the Blood Pressure Lowering Treatment Trialists’ Collaboration focusing on low-moderate risk groups provide support for considering treatment in such cases (45,46). Therefore, these patients should be offered the opportunity to concomitantly initiate antihypertensive treatment, such as candesartan or beta blocking agents, which both have blood pressure-lowering effects and demonstrated effectiveness as a preventative measure for migraine (59). Furthermore, despite potential practical challenges associated with providing remote care, the increasing availability of reimbursement options for telemonitoring including BP-measurements may resolve practical difficulties with BP-monitoring. By considering these factors, neurologists can optimize the treatment of blood pressure and improve the overall care for migraine patients.
Conclusions
Despite the limited amount of evidence, one low bias study indicates an increase in BP during treatment with anti-CGRP(R)-mAbs. Monitoring and managing BP, along with considering individual patient profiles, is of importance to ensure optimal outcomes in patients with migraine. Young females with migraine (especially those with aura) should be informed about possible increased relative risks when initiating anti-CGRP(R)-mAbs. More post marketing studies are necessary to investigate the effect of anti-CGRP(R)-mAb on blood pressure and to strengthen the current body of evidence.
Clinical implications
Monoclonal antibodies targeting CGRP or the CGRP-receptor may increase BP, but evidence is still limited to one study that indicates a mean increase of 5.2 mmHg in systolic BP during treatment with anit-CGRP(R)-mAbs. The decision to initiate antihypertensive therapy, particularly in (young) patients with mild elevations in BP, should be individualized. Although some may argue against starting treatment in low-risk scenarios, evidence from meta-analyses supports considering antihypertensive therapy, such as candesartan or beta-blockers, which also offer migraine prophylactic benefits. Given the limited current data, additional post-marketing studies are needed to assess the long-term effects of anti-CGRP(R)-mAbs on BP and overall cardiovascular safety. Until more evidence is available, it seems prudent to monitor blood pressure during treatment with anti-CGRP(R)-mAbs.
Supplemental Material
sj-docx-1-cep-10.1177_03331024241297673 - Supplemental material for Impact of CGRP monoclonal antibody treatment on blood pressure in patients with migraine: A systematic review and potential clinical implications
Supplemental material, sj-docx-1-cep-10.1177_03331024241297673 for Impact of CGRP monoclonal antibody treatment on blood pressure in patients with migraine: A systematic review and potential clinical implications by Britt W.H. van der Arend, Floor C. van Welie, Michael H. Olsen, Jan Versijpt, Antoinette Maassen Van Den Brink and Gisela M. Terwindt in Cephalalgia
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Author contributions
BvdA, FCvW and GMT made the draft of the manuscript, performed the literature search and selected the literature that fitted the main research question. All authors contributed to the critical review of the manuscript.
Data availability
All data are available from the corresponding author upon reasonable request.
Registration
This protocol/review was not registered.
Supplemental material
Supplemental material for this article is available online.
