Abstract
Abstract
Background
Clinical trials on anti-calcitonin gene-related peptide monoclonal antibodies poorly investigated their impact on migraine accompanying symptoms.
Objective
To evaluate the impact of basal accompanying symptoms on anti-CGRP monoclonal antibodies treatment response and their evolution after six months of treatment in migraine patients.
Methods
Patients with migraine diagnosis seen in the Headache Clinic and treated with erenumab, galcanezumab or fremanezumab were prospectively recruited. They completed a daily eDiary which provided data on headache frequency and the following accompanying symptoms of each day: photophobia, phonophobia, nausea, dizziness, and aura. Patients were classified as responders or non-responders based on 50% or greater reduction in headache days per month at month 6 (≥50% response rate). Accompanying symptoms ratios based on headache days per month were assessed per patient at baseline and after three and six months. Comparisons for basal characteristics, basal accompanying symptoms ratios and their evolution after six months between responders and non-responders were performed.
Results
One hundred and fifty-eight patients were included, 44% (69/158) showed ≥50% response rate after six months. A significant reduction in headache days per month in both groups was found at month 6 (−9.4 days/month in ≥50% response rate group; p < 0.001, −2.2 days/month in <50% response rate group; p = 0.004). Additionally, significant decreases in photophobia (−19.5%, p < 0.001), phonophobia (−12.1%, p = 0.010) and aura ratios (−25.1%, p = 0.008) were found in ≥50% response rate group. No statistically significant reductions were found in nausea and dizziness in any group since their reduction was correlated with the decrease in headache days per month. Higher photophobia ratios at baseline were predictive of an increased response between months 3 and 6 (Incidence Risk Ratio = 0.928, p = 0.040).
Conclusions
The days per month with photophobia, phonophobia and aura decreased at a higher rate than headache days per month after six months in the ≥50% response group. Higher photophobia ratios were associated with higher response rates between three and six months. It could indicate an involvement of peripheral CGRP in photophobia as well as a central modulation of migraine through these treatments which mainly act on the periphery.
Introduction
Migraine is a chronic neurological disease characterized by a predisposition to recurrent attacks of disabling headache and accompanying symptoms (1). The most characteristic symptoms including photophobia, phonophobia, nausea and vomiting, are included in the definition of migraine in the International Classification of Headache Disorders (ICHD-3) (1). Additionally, patients can experience a variety of other symptoms, such as dizziness, cutaneous allodynia, or even motor symptoms (2). Around a third of people with migraine can also experience aura, a symptomatology that originates within the central nervous system (3).
Monoclonal antibodies (mAbs) targeting calcitonin gene-related peptide (CGRP) or its receptor (anti-CGRP mAbs) have been the first target-driven treatment to be approved for migraine prevention. Their efficacy and safety have been demonstrated in randomized controlled trials (RCT) (4–12) as well as, in real-world evidence (RWE) studies (13,14). Both RCT and RWE studies have reported decreases in migraine and headache frequency, pain intensity and acute medication use in many patients. However, exploratory post-hoc analysis of anti-CGRP mAbs clinical trials have only recently started to assess these type of outcomes (15–18). Accompanying symptoms are usually better assessed and reported in RCT of acute treatment of migraine attacks (19,20). Specifically, Guidelines of the International Headache Society for controlled trials of acute treatment of migraine attacks in adults highly recommend the use of co-primary endpoints assessing the most bothersome symptom including nausea, vomiting, photophobia, and phonophobia (21).
Due to the large molecular weight of anti-CGRP mAbs, they poorly cross the blood-brain barrier (BBB), so their action is thought to be in the peripheral segment of the trigeminovascular system (22). As a result, many questions remain about how these treatments act in the pathophysiology of migraine since they do not act at a central level (23), where many of the structures related to the different phases of the migraine attack are located (24). However, a recent exploratory study showed that anti-CGRP mAbs could have an impact on aura and prodromal symptoms such as sleep or psychiatric comorbid symptoms in migraine patients (25).
In addition to reducing migraine frequency, we hypothesized whether anti-CGRP mAbs could reduce aura and certain accompanying symptoms such as phono- or photophobia, which are likely linked to a clearer migraine pathophysiology. Thus, the aim of this study was to evaluate the impact of baseline accompanying symptoms on anti-CGRP mAbs treatment response and their evolution after six months of treatment in migraine patients.
Methods
Study population
This is a prospective observational study conducted in a real-life clinical setting. Data was collected from January 2020 to January 2023. Patients were recruited from the outpatient Headache Clinic. They were adults who fulfilled the criteria for migraine and chronic migraine (CM) according to the ICHD-3 and were eligible to receive anti-CGRP mAbs based on the considerations of the National Regulatory Agency. These considerations included having eight or more migraine days per month and previous failure with three preventive treatments, one of which was onabotulinumtoxinA for CM patients (26).
Treatment response assessment
CGRP-mAbs were administered monthly (erenumab 140 mg and galcanezumab 120 mg with a loading dose of 240 mg) or quarterly (fremanezumab 675 mg). Response was based on 50% or greater reduction in headache days per month (HDM) after six months (≥50% Response Rate, RR). HDM was considered the sum of headache and migraine days per month, with 28 calendar days considered per month. Follow-up visits were conducted quarterly.
Clinical variables
Participants completed a daily electronic diary (eDiary), which provided data on headache and migraine frequency, pain intensity, acute medication intake and accompanying symptoms of every migraine attack including photophobia, phonophobia, nausea, dizziness and aura. The eDiary was completed for one month prior to baseline and during six months of treatment. Patient-Reported Outcome Measures (PROMs) were also completed at baseline and after three and six months: Migraine Disability Assessment (MIDAS), Headache Impact Test (HIT-6), Migraine specific quality of life questionnaire (MSQ) and Beck Anxiety Inventory (BAI). Ratios for accompanying symptoms were calculated for each by dividing the number of days per month with the specific symptom by the total headache frequency (HDM). These ratios were calculated at baseline as well as after three and six months of treatment.
Missing data
During the study period, a total of 849 patients were treated with mAbs. Of these, 319/849 were withdrawn before six months or had started treatment less than six months prior to January 2023. Out of the remaining 530 patients who reached six months of treatment, 146/530 achieved a minimum of 80% eDiary compliance during the study period. Additionally, 26/530 did not meet the 80% eDiary compliance threshold but had passed an eDiary double-check based on neurologist visit electronic records, while 358/530 did not properly complete the eDiary. The double-check between eDiary data and electronic records during visits was conducted to avoid removing a subgroup of episodic migraine (EM) patients who only filled their calendars in headache days, which would have introduced a sampling bias. Ultimately, after excluding these patients, 172 patients remained, of whom 14/172 had other primary headache, resulting in a total sample size of 158 patients. The percentage of missing data in the clinical and demographic data was <5.1% and in the scale data <15.4%. No data imputation was performed on missing values.
Statistical analysis
Statistical analysis was performed in three parts. First, we aimed to compare basal accompanying symptoms between 50% RR groups at month 6. Comparisons between groups for migraine characteristics and for basal accompanying symptoms ratios were performed. Descriptive and frequency statistics were obtained from demographics and clinical data. Counts (n) and percentages (%) were used as measures of frequencies for categorical variables and mean and standard deviation (SD) were used as measures of central tendency and dispersion for quantitative variables. Independent t-tests were performed for quantitative continuous data, Negative Binomial or Generalized Poisson GLM (generalized linear regression models) for count data, and Fisher’s exact tests for categorical variables. Central Limit Theorem was applied to verify the approximate normal distribution of the sampling distribution of the mean by group, as sample size per group was above 30 patients. False discovery rate was adjusted for multiple comparisons by Benjamini–Hochberg method.
In the second analysis, we investigated the evolution of accompanying symptoms between 50% RR groups at baseline (M0) and at months 3 (M3) and 6 (M6). Multiple two-factor Repeated Measures ANOVAs were carried out for continuous numerical data and Poisson and Generalized Poisson GLMM (generalized linear mixed-effects regression models) were performed for count data. Post-hoc comparative analysis between and within groups at baseline and after three and six months were performed on headache and migraine frequency, pain intensity, acute medication intake, accompanying symptoms ratios and PROMs scores. Square root transformation was applied to nausea, dizziness, and aura ratios, and estimates were back-transformed. False discovery rate was adjusted for multiple comparisons by Benjamini–Hochberg method.
In the third analysis, we evaluated the potential effect of basal accompanying symptoms on treatment response in terms of HDM evolution over time. We did not establish an HDM reduction percentage limit to divide patients into groups. Instead, we analyzed the pooled patient sample considering baseline HDM and HDM time evolution per patient at month 3 and month 6. A Generalized Poisson (GP) mixed-effects regression model (GLMM) with random intercepts was estimated due to the existence of underdispersion. HDM was considered as the count data outcome variable. HDM at baseline, basal accompanying symptoms ratios, mAbs type and medication overuse (MO) were considered as independent variables in the model, which was adjusted by age and sex. Interactions between time and basal symptom ratios were considered. Erenumab was used as the reference level for the mAbs type predictor. GLMM parameter estimation was performed using Restricted Maximum Likelihood Estimation (27). The best fitting model was obtained by stepwise forward elimination applying Likelihood Ratio Test criterion (28).
No statistical power calculation was conducted prior to the study, as the sample size was based on available data. Statistical analyses were performed using R software version 4.2.2 for Windows and p-values <0.05 were considered as statistically significant and reported for a two-tailed test.
Ethics approval and patients’ consent
The study was approved by the Vall d’Hebron Ethics Committee (PR(AG)53/2017/2022 amendment). All patients gave an informed consent for the analysis of their data. All patients consented to publication of anonymous individual data.
Results
Descriptive
The study included 158 patients with a mean age of 46.4 ± 10.3 years, of which 86.1% were women. Of the patients, 43.7% (69/158) showed ≥50% RR, while 56.3% (89/158) showed <50% RR. At baseline, 50% of patients reported photophobia or phonophobia on more than 72.2% of HDM, while 25% of patients reported nausea on more than 37.5% of HDM. 50% of patients reported at least one of these three symptoms on more than 94.4% of HDM. Dizziness was the least prevalent symptom, reported by 25% of patients on more than 21.4% of their HDM. A 45.6% (72/158) of patients had aura at baseline, with 50% of them reporting aura on more than 30.8% of their HDM. Frequencies, means and SD values are provided in Table 1.
Basal characteristics and comparison between ≥50% RR group and <50% RR group after 6 months of treatment.

Significance assessed with Fisher’s exact test.
Significance assessed with independent 2-sample t-test. Central Limit Theorem was considered for sample sizes per group above 30.
Significance assessed with Negative Binomial or Generalized Poisson GLM.
False Discovery Rate was adjusted by Benjamini–Hochberg method.
*Data available only for CM patients (82/158)
**Data only for patients with aura (72/158)
BAI: Beck anxiety inventory; BDI-II: Beck depression inventory-second edition; CM: chronic migraine; d/mo: days per month; EM: episodic migraine; GLM: Generalized linear regression model; HDM: headache days/month; HIT-6: headache impact test; MDM: migraine days/month; MIDAS: migraine disability assessment; MSQT: migraine-specific quality of life questionnaire (total score); p/mo: pills per month; RR: response rate; SD: standard deviation.
Basal accompanying symptoms comparison
None of the clinical variables describing basal accompanying symptoms ratios were found to be statistically significantly different between ≥50% RR and <50% RR groups (photophobia mean ratio: 66.6% ≥50% RR group, 58.3% <50% RR group, p = 0.985; Phonophobia mean ratio: 62.2% ≥50% RR group, 55.6% <50% RR group, p = 0.985; Nausea mean ratio: 23.1% ≥50% RR group, 21.6% <50% RR group, p = 0.985; Dizziness mean ratio: 19.0% ≥50% RR group, 15.4% <50% RR group, p = 0.985; Aura mean ratio: 48.0% ≥50% RR group, 36.4% <50% RR group, p = 0.985). We could neither find any significant difference between groups for any other demographic nor clinical baseline variable (Table 1).
Evolution of accompanying symptoms comparison
Both groups showed a statistically significant within-group reduction in the number of HDM at M3 and M6 (in particular, M0-M6: −9.4 days/month for ≥50% RR group, 95% CI −10.9 to −7.9, p < 0.001; −2.2 days/month for <50% RR group, 95% CI −3.6 to −0.9, p = 0.004), migraine days/month (MDM) (M0-M6: −6.8 days/month for ≥50% RR group, 95% CI −8.1 to −5.4, p < 0.001; −2.5 days/month for <50% RR group, 95% CI −3.6 to −1.5, p < 0.001), pain intensity (M0-M6: −0.68 for ≥50% RR group for a 0–3 score, 95% CI −0.77 to −0.60, p < 0.001; −0.23 for <50% RR group, 95% CI −0.31 to −0.14, p < 0.001) and acute medication intake (M0-M6: −6.0 days/month for ≥50% RR group, 95% CI −7.2 to −4.7, p < 0.001; −1.3 days/month for <50% RR group, 95% CI −2.4 to −0.3, p = 0.032). Statistically significant differences were also found between groups at months 3 and 6 (for ≥50% RR group, M6 HDM: −8.4 days/month, 95% CI −10.1 to −6.7, p < 0.001; M6 MDM: −4.8 days/month, 95% CI −6.1 to −3.6, p < 0.001; M6 pain intensity: −0.53, 95% CI −0.68 to −0.38, p < 0.001; M6 medication intake: −6.9 days/month, 95% CI −8.7 to −5.1, p < 0.001). Full contrasts details are shown in online supplementary material (Table A).
Statistically significant decreases in photophobia (M0-M6: −19.5%, 95% CI −29.8% to −9.3%, p = < 0.001) and phonophobia ratios (M0-M6: −12.1%, 95% CI −20.3 to −3.8%, p = 0.010) were found exclusively in ≥50% RR group between baseline and month 6 (in <50% RR group, photophobia M0-M6: −0.7%, 95% CI −5.9% to +4.4%, p = 0.870; phonophobia M0-M6: −0.4%, 95% CI −6.9% to +6.2%, p = 0.944). Aura ratios decreased significantly in ≥50% RR group at 3 and 6 months (M0-M3: −26.0%, 95%CI −42.4% to −9.7%, p = 0.006; M0-M6: −25.1%, 95%CI −41.4% to −8.8%, p = 0.008), but did not in <50% RR group (M0-M3: −10.3%, 95%CI −21.0% to 0.0%, p = 0.107; M0-M6: −10.7%, 95%CI −21.5% to 0.0%, p = 0.092). Nausea did not reach statistically significant differences within any group (M0-M6: −5.3%, 95% CI −11.7% to +1.1%, p = 0.180 in ≥50% RR group; M0-M6: −4.8%, 95% CI −10.2% to +0.5%, p = 0.124 in <50% RR group) and dizziness did not improve over time in ≥50% RR group (M0-M6: −1.0%, 95% CI −4.7% to +6.7%, p = 0.82) and even reached a statistically significant increase between month 3 and 6 in <50% RR group (M3-M6: +6.4%, 95% CI +1.3% to +11.4%, p = 0.029). No statistically significant differences were found at month 3 or 6 between groups. We did notice that both photophobia and phonophobia ratios showed bimodal frequency distributions, showing two peaks of incidence, and this division was not explained by any of the clinical or demographical variables collected during this study. Bar plots showing evolution at M0, M3 and M6 of accompanying symptoms ratios in ≥50% RR and <50% RR groups are shown in Figure 1. Figure 2 provides the results of post-hoc comparison tests of clinical outcomes and accompanying symptoms ratios based on ANOVA Repeated Measures analysis and Poisson/Generalised Poisson GLMM.

Accompanying symptoms ratios based on HDM at baseline and after three and six months of treatment in ≥50% RR group and <50% RR group.
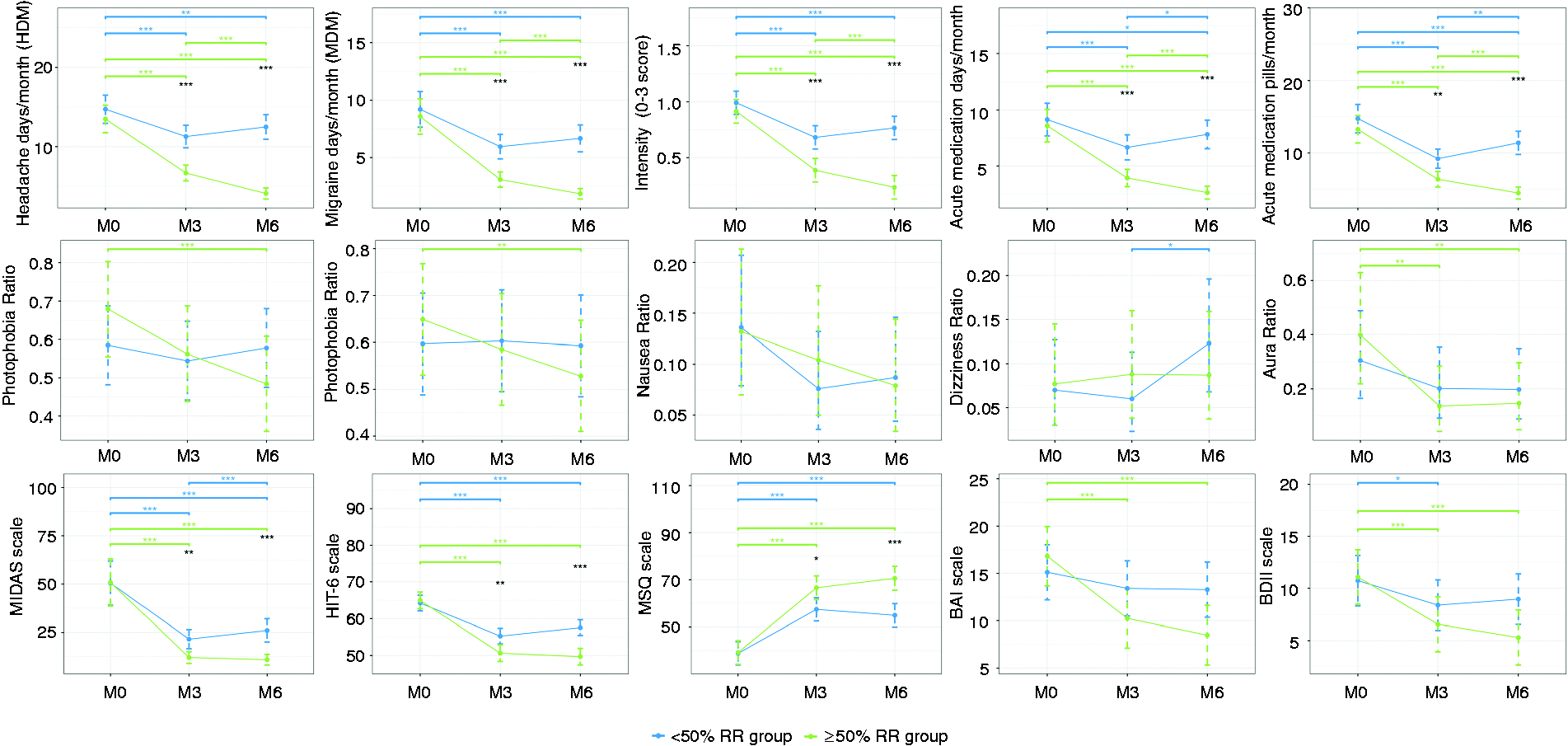
Comparison of clinical outcomes and accompanying symptoms ratios between M0, M3 and M6 in ≥50% RR group and <50% RR group based on ANOVA Repeated Measures analysis and Poisson/Generalised Poisson GLMM.
Statistically significant reductions were found within groups in MIDAS score after three and six months (in particular, M0-M6: −39.8 in ≥50% RR group, 95% CI −49.5 to −30.2, p < 0.001; −24.4 in <50% RR group, 95% CI −30.2 to −18.5, p < 0.001), HIT-6 score (M0–M6: −15.3 in ≥50% RR group, 95% CI −17.7 to −12.9, p < 0.001; −6.7 in <50% RR group, 95% CI −8.9 to −4.4, p < 0.001) and increases in MSQ score (M0-M6: +31.4 in ≥50% RR group, 95% CI +26.5 to +36.3, p < 0.001; +16.1 in <50% RR group, 95% CI +11.1 to +21.1, p < 0.001) as well. Additionally, statistically significant differences were found between groups at months 3 and 6 (M6 MIDAS: −15.1 in ≥50% RR group, 95% CI −21.7 to −8.6, p < 0.001; M6 HIT-6: −7.9 in ≥50% RR group, 95% CI −11.0 to −4.8, p < 0.001; M6 MSQ: +15.7 in ≥50% RR group, 95% CI +8.6 to +22.7, p < 0.001). BAI and BDI-II scores significantly improved within in ≥50% RR group at both three and six months (M0-M6 BAI: −8.3, 95% CI −10.8 to −5.8, p < 0.001; M0-M6 BDI-II: −5.8, 95% CI −7.7 to −3.9, p < 0.001). In <50% RR group, a slight improvement was found only in BDI-II at month 3, however, it was not maintained at month 6 (M0-M6 BAI: −1.8, 95% CI −4.1 to +0.4, p = 0.180; M0-M3 BDI-II: −2.4, 95% CI −4.0 to −0.7, p = 0.011; M0-M6 BDI-II: −1.8, 95% CI −3.4 to +0.1, p = 0.064). No statistically significant differences were found at month 3 or 6 between groups for neither BAI nor BDI-II.
Impact of basal accompanying symptoms on HDM evolution over time
The resulting best fitting model concluded that the following variables were significantly affecting HDM evolution over time (Table 2, Figure 3): HDM at baseline (Incidence Risk Ratio IRR = 1.060, 95% CI 1.047 to 1.072, p < 0.001), MO (IRR = 1.194, 95% CI 1.107 to 1.287, p < 0.001) and the interaction between basal photophobia ratio and months under treatment (basal photophobia ratio: IRR = 1.195, 95% CI 0.791 to 1.805, p = 0.389; months: IRR = 1.014, 95% CI 0.964 to 1.067, p = 0.584; interaction term: IRR =0.928, 95% CI 0.863 to 0.997, p = 0.040). Each unit increase in basal HDM increased a 6% the predicted HDM at M3 and M6. Presence of basal MO increased HDM at M3 and M6 a 19.4%. The effect of interaction between basal photophobia ratio and months under treatment on HDM evolution is showed in Figure 4. Higher photophobia ratio at baseline was associated with higher RR between M3 and M6.
Impact of basal accompanying symptoms on HDM evolution over time.

Statistics obtained from fitted model were 1826.84 (AIC); 1886.93 (BIC); −897.42 (-2LL); 1.25 (dispersion parameter) and 0.124 (RE variance component). §p-value significance codes: 0 to 0.001: ‘***’; 0.001 to 0.01: ‘**’; 0.01 to 0.05: ‘*’; non-significant: ‘ns’
Months variable was considered as quantitative (three and six months timepoints).
AIC: Akaike information criterion; BIC: Bayesian information criterion; CI: confidence interval; GLMM: Generalized linear mixed-effects regression model; IRR: Incidence Risk Ratio; RE: random effects; -2LL: log-likelihood.

Incidence Risk Ratios (IRR) for Generalized Poisson mixed-effects regression model analysing the impact of basal accompanying symptoms on HDM evolution over time.

Interaction effects of photophobia ratio at baseline on HDM evolution over time.
The model provided no statistically significant evidence that other basal accompanying symptoms ratios had an effect on HDM reduction during six months of treatment (basal phonophobia ratio: IRR = 1.037, 95% CI 0.819 to 1.312, p = 0.765; basal nausea ratio: IRR = 1.051, 95% CI 0.790 to 1.397, p = 0.734; basal dizziness ratio: IRR = 1.013, 95% CI 0.772 to 1.329, p = 0.925; basal aura ratio: IRR = 0.976, 95% CI 0.745 to 1.280, p = 0.863). No significant effect was found on either mAbs type (IRR-galcanezumab = 0.995, 95% CI 0.889 to 1.114, p = 0.930; IRR-fremanezumab = 1.034, 95% CI 0.875 to 1.221, p = 0.693), age (IRR = 0.998, 95% CI 0.990 to 1.005, p = 0.529) or sex (IRR-Woman = 1.071, 95% CI 0.964 to 1.189, p = 0.203).
Discussion
The aim of this prospective study was to assess the relationship between the response to anti-CGRP mAbs treatment and the most common centrally-driven accompanying symptoms of migraine attacks. Specifically, we aimed to investigate the evolution of these symptoms after anti-CGRP mAbs treatment and whether the presence of these symptoms at baseline could affect or predict treatment response.
Participants in this study completed a daily headache eDiary providing information on every attack for 28 days before starting treatment and during six months under anti-CGRP mAbs. They were effective reducing traditional outcomes such as headache and migraine frequency, intensity and acute medication intake, as well as improving PROMs scores in all patients except for BAI and BDI-II in <50% RR group. Our results showed that anti-CGRP mAbs were able to reduce accompanying symptoms due to the reduction in headache frequency. Interestingly, in ≥50% RR group, there were statistically significant reductions in the ratios of photophobia, phonophobia, and aura after six months of treatment. This means that the number of days per month with these symptoms decreased at a higher rate than the reduction in HDM after six months. This was not observed for other symptoms such as nausea and dizziness, where the number of days per month with these symptoms decreased proportionally to the reduction in HDM Therefore, the symptomatology of nausea and dizziness on the remaining headache days after six months did not change remarkably compared to baseline.
At baseline, our cohort showed similar proportions of accompanying symptoms as those reported in the literature, making it comparable to any previously described migraine population (29). Photophobia and phonophobia were the most prevalent symptoms, followed by nausea, aura, and dizziness in our cohort. In the third analysis, we could not identify any demographic predictor of response to mAbs treatment. However, higher photophobia ratios at baseline were associated with an increase in response between three and six months of treatment. MO significantly affected HDM evolution over time, increasing in a 19.4% the predicted HDM for a given basal headache frequency and treatment month. In this regard, different predictors of response have been found in migraine patients under anti-CGRP mAbs treatment in RWE studies. Previous studies have focused on predictors of negative or incomplete response to treatment, such as MO, higher acute medication intake, a higher number of prior preventive treatment failures (30–32), the presence of allodynia (33) psychiatric comorbidities (14) or daily headache (34). Other studies have identified predictors of positive response to treatment, such as triptans (35), unilateral localization of pain (36), allodynia, osmophobia, unilateral autonomic symptoms, and dopaminergic symptoms (34).
According to our results, the absolute number of days with nausea and dizziness significantly decreased after six months, in parallel with a decrease in HDM for both groups, except for a rebound in dizziness in <50% between month 3 and 6. However, anti-CGRP mAbs significantly reduced the percentage of days per month with photophobia, phonophobia or aura based on the remaining HDM in the group of ≥50% RR after six months, but no significant differences were found between groups over time. It should be noted that in both photophobia and phonophobia ratios, the frequency distributions were clearly bimodal, showing two peaks of incidence. This could be the reason for not finding differences between groups due to data distribution dispersion. Further research should evaluate additional clinical or biological variables that could explain these two different groups of patients with different symptomatology incidences.
These results prompt the question of how and where this class of drugs act, as the mechanism of action of these treatments still needs to be fully understood. Theoretically, due to their large molecular weight they are not expected to cross the BBB quickly, and therefore, a rapid central effect is not anticipated (37,38). So, how can these treatments potentially affect central migraine symptoms? There is accumulating evidence on how modulation the periphery in migraine can have an impact on central mechanisms. In fact, other studies have demonstrated changes in central neuroimaging (39,40), and neurophysiological parameters (41–43) or have even shown an impact on migraine aura (44,45), as in our case. Interestingly, a previous exploratory study found that treatment with anti-CGRP mAbs could prevent prodromal symptoms such as aura, stress or anxiety and could modulate sleep and weight (25).
Another possible explanation for our findings could be the direct regulatory role of CGRP in migraine symptoms such as photophobia, phonophobia and aura, which may also be partially triggered in peripheral receptors. Biochemically, CGRP seems to have a role in photophobia. Animal model studies have shown that genetically engineered mice with elevated expression in nervous tissue of the human receptor activity-modifying protein 1 (RAMP1) spend less time in light environments (46) and that intracerebroventricular administration of CGRP causes a significant increase in light aversion prevented by olcegepant (47). In addition, certain accompanying symptoms such as photophobia or phonophobia appear to be related to higher levels of CGRP during migraine attacks (48). Despite the lack of evidence about how these treatments act at a molecular level, it seems that they may be able to reduce or regulate CGRP levels (49). Thus, the reduction of photophobia days per month at a higher rate than monthly headache frequency could be explained by the reduction or regulation of CGRP levels. Accordingly, the rest of the accompanying symptoms not directly linked to CGRP are reduced in both groups due to the direct reduction in headache frequency, except for dizziness in <50% RR group. Henceforth, our study would support the fact that photo or phonophobia might be at least partially regulated from the periphery and by CGRP (37,38).
In a previous study, we found that low frequency EM patients who experienced photophobia and phonophobia during migraine attacks showed a greater increase in salivary CGRP levels (ictally and interictally) than patients with nausea and dizziness as accompanying symptoms (47). Our current study found that higher basal photophobia ratios were associated with an increased response to treatment between months 3 and 6. Therefore, it could be speculated that patients with higher ratios of photophobia and phonophobia could have better response to anti-CGRP treatment due to their higher baseline CGRP levels. The presence of photophobia and phonophobia could also indicate that EM patients, for whom CGRP levels are an important predictor of response to mAb treatment (48), have a higher probability of having better response. This may not be the case for CM patients, in whom a clear differentiation of symptoms is more difficult due to their constant “migrainous” state (49).
In migraine, it is becoming important to assess other clinical outcomes in addition to the traditional ones in order to better reflect real improvement in the patient's quality of life (29,50) and to understand the very target-driven pathophysiological mechanisms. Post-hoc analysis of anti-CGRP mAbs studies have already shown a reduction of migraine accompanying symptoms, such as photophobia and phonophobia, nausea and/or vomiting in the case of eptinezumab and fremanezumab and even in prodromal symptoms in the case of galcanezumab (15–18). Our study also shows a reduction in some of these parameters, finding that the presence of photophobia could predict an increased response to treatment at month 6, and that photophobia, phonophobia and aura days per month decrease even more than HDM.
Our study has used in a real-world setting, using tools such as eDiaries, patient reported outcome measures (PROMs) and a very homogenous cohort of patients, giving strength to the findings. A limitation is the lack of a control group, which makes it impossible to quantify the placebo effect. However, the efficacy results in our cohort are similar to other clinical trial and real-world series (30). Future studies should integrate clinical and molecular parameters in large cohorts in order to shed light on the relationship between clinical symptoms, CGRP and treatment response.
To the best of our knowledge, this is the first study to prospectively evaluate the accompanying symptoms of migraine patients treated with anti-CGRP mAbs in a deeply studied cohort. This supports the fact that these specific treatments are not only effective in reducing traditional endpoints of clinical trials but also they improve patients quality of life by reducing bothersome accompanying symptoms in a real-life setting. Additionally, it elucidates how the peripheral action of these treatments could finally modulate central mechanisms, or the fact that photophobia, phonophobia and aura are partially, at least, modulated by reducing CGRP in the periphery, which in turn, might be helpful in increasing migraine-related quality of life.
Conclusion
In conclusion, the ratios of photophobia, phonophobia and aura were significantly reduced in migraine patients who showed 50% or greater response to anti-CGRP mAbs preventive treatment at six months. Higher basal photophobia ratios predicted an increased response between months 3 and 6, suggesting peripheral CGRP involvement in photophobia and central modulation of migraine through treatments that primarily act on the periphery.
Article highlights
The number of days per month with photophobia, phonophobia, and aura decreased further compared to the reduction in headache frequency after six months in the group of migraine patients who had a response rate of 50% or greater to anti-CGRP mAbs treatment. The rest of the accompanying symptoms decreased in parallel with the reduction in headache frequency, except for dizziness, which rebounded in the group of patients who had a response rate of less than 50% between months 3 and 6. A higher baseline photophobia ratio was associated with an increased response rate between months 3 and 6.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024231177636 - Supplemental material for Impact of anti-CGRP monoclonal antibodies on migraine attack accompanying symptoms: A real-world evidence study
Supplemental material, sj-pdf-1-cep-10.1177_03331024231177636 for Impact of anti-CGRP monoclonal antibodies on migraine attack accompanying symptoms: A real-world evidence study by Alicia Alpuente, Anna Torre-Sune, Edoardo Caronna, Eulalia Gine-Cipres, Marta Torres-Ferrús and Patricia Pozo-Rosich in Cephalalgia
Footnotes
Authors' contributions
AA collected data, interpreted the data and drafted the manuscript; ATS analyzed the data and performed statistical analysis; EC and MT-F collected and interpreted the data and revised the manuscript for intellectual content; EG-C collected clinical data; PPR designed and conceptualized study and revised the manuscript for intellectual content.
Availability of data and materials
Data are available upon request from the corresponding author.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: In relation with this paper the authors have nothing to disclose. AA has received honoraria as speaker from AbbVie, Novartis and Eli Lilly. ATS has nothing to disclose. EC has received honoraria as speaker from Novartis. MT-F has received honoraria as a speaker from AbbVie, Chiesi, Eli Lilly and Novartis. In the last 18 months, PP-R has received honoraria as a consultant and speaker from AbbVie, Biohaven, Chiesi, Eli Lilly, Lundbeck, Medscape, Novartis, Pfizer and Teva Pharmaceuticals. Her research group has received research grants from AbbVie, AGAUR, EraNet Neuron, FEDER RIS3CAT, Instituto Investigación Carlos III, MICINN, Novartis, and Teva; and has received funding for clinical trials from AbbVie, Biohaven, Eli Lilly, Lundbeck, Novartis and Teva. She is the Honorary Secretary of the International Headache Society. She is in the editorial board of Revista de Neurologia. She is an associate editor for Cephalalgia, Headache, Neurologia, Frontiers of Neurology and an advisor of the Scientific Committee of the Editorial Board of The Journal of Headache and Pain. She is a member of the Clinical Trials Guidelines Committee and Scientific Committee of the International Headache Society. She has edited the Guidelines for the Diagnosis and Treatment of Headache of the Spanish Neurological Society. She is the founder of ![]() . She does not own stocks from any pharmaceutical company.
. She does not own stocks from any pharmaceutical company.
Funding
The authors received no financial support for the research, authorship, and/or publication.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
