Abstract
Aim
Cortical spreading depression (CSD), the neural correlate of migraine aura, has been shown to cause activation of dural nociceptive neurons as well as immune cells, among which macrophages (MPs) are the most abundant and reactive. OnabotulinumtoxinA (onbotA) is used to treat chronic migraine but the mechanism of action is not fully understood. Here we investigate the role of meningeal MPs in a model of migraine activation and evaluate whether onabotA has an effect on their response.
Methods
We use our previously developed method to determine meningeal MP activation based on shape changes using time-lapse in vivo multiphoton microscopy.
Results
We found that a small subset (∼10%) of MPs contracted their processes in response to CSD induction, but only in female mice. A similar subset of MPs contracted with lipopolysaccharide injection, suggesting that this is an M1-like response. Together this may provide insight into the phenotypic differences of migraine across males and females. We also found a small subset of MPs (∼10%) that expanded their processes in response to IL-10 (presumably an M2-like response), but were not affected by CSD. In female mice, pre-treatment with onabotA (i) reduces overall MP number in the dura, (ii) reduces pro-inflammatory M1 MP response and (iii) increases anti-inflammatory M2 response post-CSD compared to pretreatment with saline.
Conclusion
This suggests that the mechanism of action of onabotA may not be simply due to its effects on nociceptors, but also due to an additional anti-inflammatory effect on the environment of the dura.

Introduction
Migraine is a debilitating disease that is commonly associated with headache, sensory aura, nausea, photophobia, phonophobia, and a variety of abnormal sensory, autonomic, affective (e.g., anxiety, depression) and cognitive dysfunctions (1). Migraine aura is likely due to cortical spreading depression (CSD), a slowly propagating wave of excitation followed by a lengthy inhibition of neurons and glia within the brain (2–4). While it is possible that CSD could directly affect sensory neurons within the central nervous system (5,6), specificity to migraine headache is almost certain to arise in activation of peripheral pain receptors in the dura originating in the trigeminal or spinal ganglia (7). However, it is still unknown how abnormal brain activity such as aura could lead to activation of nociceptors in the dura, which lies outside the blood-brain barrier.
The common use of non-steroidal anti-inflammatory drugs (NSAIDs) in migraine, especially with mild cases (8–10), suggests that inflammation is likely to play a role in migraine pathophysiology. Animal studies have found inflammatory pathology in the dura in migraine models (11–13), such as plasma protein extravasation (14), upregulation of pro-inflammatory cytokines (11,15), mast cell degranulation (16,17), and vasodilation (14). Inflammation is mediated by many cell types, however, macrophages are key players, as they are the most populous cells in the dura (over 60% (18–20)), are constituently monitoring their environment, and can activate sensory axons (21). Macrophages can polarize along several functional pathways. One pathway, sometimes called “M1” activation is induced by interferon-γ (IFNγ), tumor-necrosis factor alpha (TNF-α) and bacterial products such as lipopolysaccharide (LPS) (22–24), and promotes the production and release of pro-inflammatory cytokines such as IL6, IL12, TNF-α, and IL1β (25–27) – all capable of activating and sensitizing peripheral nociceptors (21), including those innervating the dura (28–30). Another pathway, one of the “M2” activations, is induced by IL-10, IL-4, IL-13, or TGF-β, and causes an anti-inflammatory response involving further IL-10 and TGF-β release, CCL1, CCL17, CCL18 release, and reduction of pro-inflammatory signalling (24,26,31).
In recent years, much progress has been made in unraveling the mechanism by which OnabotulinumtoxinA (onabotA) prevents migraine through its ability to attenuate activation of meningeal nociceptors at the origin of the trigeminovascular pathway (32–35). Seeking to widen the scope of scientific thinking about the mechanism of onabotA, we showed recently that extracranial injections of onabotA caused a decrease in the expression of multiple pro-inflammatory genes and a decrease in the abundance of 12 different immune cell classes (based on RNA expression specific to cell types), including macrophages, in the periosteum of chronic migraine patients (36).
The goal of the current study was to determine whether treatment with onabotA alters the responses of dural macrophages to CSD. Unlike our previous study (37), here we use a more direct approach to visualize macrophages and categorize them into contracting, expanding, or stable subsets. This distinction allows us to differentiate between pro-inflammatory M1-like and anti-inflammatory M2-like macrophage phenotypes. By gaining a deeper understanding of the role of MPs in migraine pathophysiology and how current treatment options impact these inflammatory cells, the data could stimulate interest in developing treatment approaches that deviate from the traditional focus on inhibiting neuronal activation.
Materials and methods
Animals
All procedures involving animals followed the experimental protocol approved by the Institutional Animal Care and Use Committee of Beth Israel Deaconess Medical Center and Harvard Medical School and adhered to the guidelines of the Committee for Research and Ethical Issues of the International Association for the Study of Pain (Protocols 005-2025 and 006-2022). Transgenic mice were obtained from Jackson Labs (Bar Harbor, ME), including C57BL/6J (wild-type), CX3CR1-GFP (Cx mice, green fluorescent protein inside of macrophages and microglia), and Thy1-ChR-YFP (ChR mice, channelrhodopsin and yellow fluorescent protein in astrocytes and many neurons in the brain), which was cross-bred with CX3CR1-GFP to generate Thy1-ChR-YFP/CX3CR1-GFP mice (CxChR). Transgenic mouse lines are all on the C57BL/6J background. Male and female mice at three to nine months old were used. Mice were kept in 12-h light/dark cycles and were housed two to five per cage.
Anesthesia
Mice were deeply anesthetized using intraperitoneal injection of urethane (1.5 g/kg) and atropine (0.15 mg/kg). Using this method, we routinely kept mice at normal physiological levels (heart rate, 550–700; arterial O2 saturation, 90–100%; breathing rate, 120–200; monitored with a MouseOx; Starr Life Science Corp, Oakmont, PA) throughout the length of the experiment.
Skull thinning
Transcranial imaging was performed as described previously (37). Briefly, the scalp was shaved and sterilized, a midline scalp incision was made, and a metal plate was affixed to the skull using cyanoacrylate. An area of 1.5 mm in diameter was thinned on the right parietal skull plate one third of the distance from lambda to bregma posteriorly. We used a high-speed drill and a microsurgical blade to thin the skull to a thickness of 50 μm for the thin skull preparation. We found that this thickness still allows good resolution of superficial structures, but disturbs the dura minimally, as determined by intact dural blood vessels free from clotting or bleeding. Mice with dural bleeding after thinning were excluded from the study. Mice were maintained on anesthesia for 1 h post thinning, while providing oxygen, heating pad, and keeping the thinned skull submerged under glucose-free artificial cerebrospinal fluid (38).
In vivo imaging
Fully anesthetized mice were imaged in an Olympus FV1000MPE-E multiphoton Imaging System using a SpectraPhysics Maitai Deepsee laser and a PLAN 25X 1.05 NA objective. Three color channels were collected simultaneously (420–460 nm blue in a PMT; 495–540 green and 575–630 red in GaAsP detectors). A wavelength of 890 was used to excite all fluorophores and produce second harmonic generation at 445 nm (blue). After skull thinning and the 1-h post-thinning period, the mouse's head plate was then attached to an aluminum breadboard (ThorLabs) and placed under the objective, while still providing oxygen and a heating pad. Time-lapse three-dimensional image stacks were taken covering a space from the skull through the meninges to the brain. Images were 1024 × 1024 pixels x 23 slices covering an area of 509 × 509 μm wide, 60 to 70 μm deep, at a 3-min interval, for five timepoints (15 min) before CSD/sham/injection and 15 timepoints (45 min) after.
CSD initiation
CSD was initiated similar to previously described methods. For both CSD pinprick and pinprick sham surgery mice: A small (<1 mm2) area was thinned on the frontal skull plate 1 mm anterior to bregma and equidistant mediolaterally from the edges of the frontal plate immediately after the thin-skull image window was thinned. This area was chosen as it was far enough away from the imaging site that craniotomy did not affect imaged cells. The underlying dura was then exposed with a 31-gauge needle. For CSD initiation, a tungsten electrode approximately 100um in diameter was inserted through the dura to about 500 μm into the cortex for 20 s before removal (the “pinprick”). Sham did not receive any pinprick. Recently, THY1-ChR-YFP (ChR) mice were developed and shown to exhibit a similar CSD induced by exposure to high intensity light (39). We cross-bred these mice with CX3CR1-GFP (Cx) to produce mice in which we could both induce optogenetic CSD and quantify macrophage shape change (CxChR mice). However, the wide expression of YFP in the brain made pial macrophages much more difficult to quantify, so we did not quantify pial cells in these mice. At baseline, the macrophages in the dura of CxChR mice were significantly fewer in number compared to Cx mice (98 vs 124, p = 9.3 × 10−4; n = 21 Cx, 7 CxChR mice; Figure 1(a)), and their baseline circularity was not significantly different (0.22 vs 0.24; n = 1321 Cx cells, 280 CxChR cells, t = −1.4, p = 0.22 according to a linear mixed-effects model, accounting for genotype and within-mouse effects, Figure 1(b)). There was no significant difference in their circularity increase post-CSD using optogenetic induction compared to Cx mice with pinprick induction (Figure 1(c), linear mixed-effects model showed no interaction between time and CSD Method, t = 1.0, p = 0.3, and no time points were significantly different in a post-hoc comparison; n = 551 cells for Cx and 131 cells for CxChR). In addition, there was no difference in macrophage shape change between CxChR and Cx in sham mice who did not receive CSD induction (linear mixed model interaction t = 0.273, p = 0.785, not shown). This suggests that neither genotype, frontal craniotomy, nor method of CSD induction (pinprick vs optogenetic) was an important factor for the activation of dural macrophages post-CSD. Subsequently, we included both CxChR and Cx mice in the CSD group in later comparisons.

Optogenetically induced CSD causes similar macrophage rounding compared to pinprick. (a) Total number of macrophages in the imaged area at baseline in the dura and pia between Cx mice that underwent frontal craniotomy and were used for pinprick-induced CSD and CxChR mice that were used for LED-induced CSD (n = 21 Cx mice, 7 CxChR mice). (b) Baseline circularity of macrophages in the dura between Cx and CxChR mice (n = 1321 Cx cells, 280 CxChR cells). Pial cells could not be quantified because of Thy1-YFP fluorescence in the brain. (c) Plot of average circularity fold change compared to baseline for pinprick-induced or optogenetic LED-induced CSD in the dura. No time points are different according to a paired t-test (p > 0.05), and a linear mixed-effect model showed no effect of group (t = 0.22, p = 0.83) and no significant interaction of time and group (t = 1.0, p = 0.30). (n = 551 cells for Cx and 131 cells for CxChR). Significant comparisons indicated with asterisks in all figures.
Injections
Lipopolysacharride (LPS; O26:B6 from Sigma-Aldrich, St Louis, MO) at 1 mg/kg (1 mg/ml in saline), an amount used in the activation of macrophages in a model of peritonitis (40,41), or IL-10 (Sigma-Aldrich, St Louis, MO) at 250 μg/kg (200 μg/ml in saline), an amount meant to provide at least a concentration of 20 ng/ml in the dura, which can polarize macrophages to an M2 phenotype (42), was administered by tail vein injection. Both LPS and IL-10 injections were performed under anesthesia. For onabotA or vehicle control experiments, one unit of OnabotA (Abbvie, North Chicago, IL) in 2 μl saline (∼30 U/kg) or 2ul of saline alone was injected into the periosteum above the sagittal suture at approximately halfway between bregma and lambda, using a Hamilton syringe (Hamilton Company, Reno, NV).
Image analysis
Images were analyzed using Fiji (www.fiji.sc), a version of ImageJ (NIH, Bethesda, MD). The arrival of the CSD wave under the imaging window was confirmed by changes in blood vessel width, and this always occurred in the timepoint immediately following CSD initiation (within 3 min). Five baseline time points were taken over 15 min before CSD induction, with the CSD confirmed at timepoint 6. Timepoint 5 was labeled as time 0 for “time post-CSD”, which represents the time point immediately previous to the CSD arrival confirmation. In sham mice, an arbitrary time point 0 was set at the fifth timepoint to match the duration of baseline imaging in the respective CSD mice. Macrophage shape was quantified using a custom written, automated, threshold and segmentation script that provided shape data over multiple time points. Images are registered for xy- and z- shift, then run through a 3 × 3 median filter, and normalized for brightness over time. Each z-slice is combined with the next two slices in a maximum intensity projection. Each MP is selected via wand region of interest (ROI) based on a threshold chosen by the user, which triggers the software to find the equivalent MP in each subsequent time point. MP ROI selection is confirmed by the user at each time point. At each time point, area, perimeter, and circularity were measured. Circularity is defined as (4*pi*area)/ perimeter2), which is a ratio of the perimeter of a circle with the shape's area and the shape's actual perimeter. In this ratio, the highest possible value is 1.0 (for a perfect circle). Researchers were blind to sex and animal group but could not be blinded to the presence of CSD due to the blood vessel changes present in the images, or to genotype based on presence of YFP in the brain. However, the automated script-based segmentation helps to remove bias from the quantification.
Statistical analysis
Multiple two-tailed paired Student t-tests with Bonferroni correction were used to compare individual time points post-CSD (or the first five timepoints in sham) to baseline for macrophage circularity change. A linear mixed-effect model was used to compare how circularity changed as a trend from time point 0 to 30 m between treatment groups. To determine cell activation, a Student's t-test was used to compare the normalized baseline timepoints with time points 3 m through 45 m, with Bonferroni correction for multiple cells per mouse. Student's t-test was used to compare between two groups of mice in bar plots. For comparisons of more than two groups (shape change comparisons in Figure 3), Welsh's one-way ANOVA was performed unless the data was not normal, in which case a Kruskal Wallis was performed. Normality was tested with the Shapiro-Wilk test. Post-hoc analysis was performed using a linear mixed-effects model with Bonferroni correction applied. Linear mixed-effects models were also used to compare the effects of groups on individual macrophage cells while accounting for within-mouse correlations (effect of group, like genotype, sex, or treatment, on output variable like baseline area, baseline circularity, or onset, with consideration of which mouse each cell came from as a random effect). In figures, * indicates p < 0.05, ** p < 0.01, *** p < 0.001 and **** p < 0.0001, and bars represent mean with error bars plus or minus the standard error of the mean. Animal numbers were based on a power calculation from preliminary studies. Statistics were calculated using the R Statistics program.
Results
Baseline macrophage characteristics are similar in male and female mice
We found no significant difference between male and female mice in the baseline characteristics of macrophages. There was no significant difference in the number of macrophage cells per imaging field in the dura (117 ± 5.5 cells for M; n = 19 mice vs 116 ± 5.3 for F; n = 17 mice, p = 0.88 according to t-test) or in the pia (71 ± 3.7 cells for M vs 73 ± 4.1 for F, p = 0.71, Figure 2(a)) at baseline (n = 17 F and 19 M mice), and no significant difference in their baseline circularity (linear mixed-effects model t = 0.12, p = 0.91; n = 986 cells F and 1061 cells M for dura; and t = 1.4, p = 0.17; n = 336 F and 593 M for pia, Figure 2(b)). Of note, dural macrophages in both sexes combined are significantly more numerous (117 ± 5 dural cells vs 74.7 ± 4 pial cells, p = 5 × 10−8) and significantly less circular than pial macrophages (0.229 ± 0.003 dural circularity vs 0.373 ± 0.009 pial circularity, t = 24, p < 1 × 10−16).

No sex differences in dural and pial macrophage number or circularity at baseline. (a) Total number of macrophages visible in the imaged area at baseline in the dura and pia (before CSD or other stimulus) showing no significant differences between sexes (p = 0.88 dura, p = 0.71 pia according to t-test). (n = 17 female mice and 19 male mice) (b) Baseline circularity of macrophages showing no significant differences between sexes (p = 0.91 dura, p = 0.17 pia according to a linear mixed-effects model). (n = 986 cells for F, 1061 for M in the dura, 336 for F, and 593 for M in the pia).
Macrophage cells respond differently to CSD, and their proportions differ based on sex
We sought to look at the distribution of circularity changes in macrophages and determine the range of changes that occur in individual cells post CSD. To achieve this, we divided macrophage shape change into subgroups. We classified cells as “contracting” if their circularity was significantly increased post-CSD or post-Sham compared to their circularity at baseline as judged by a Student's t-test, “stable” if there was no significant change, and “expanding” if significantly decreased (Figure 3(a)). These were so named because increased circularity likely represented the contraction of cellular processes, and decreased circularity likely represented the expansion of cellular processes (Figure 3(b)).
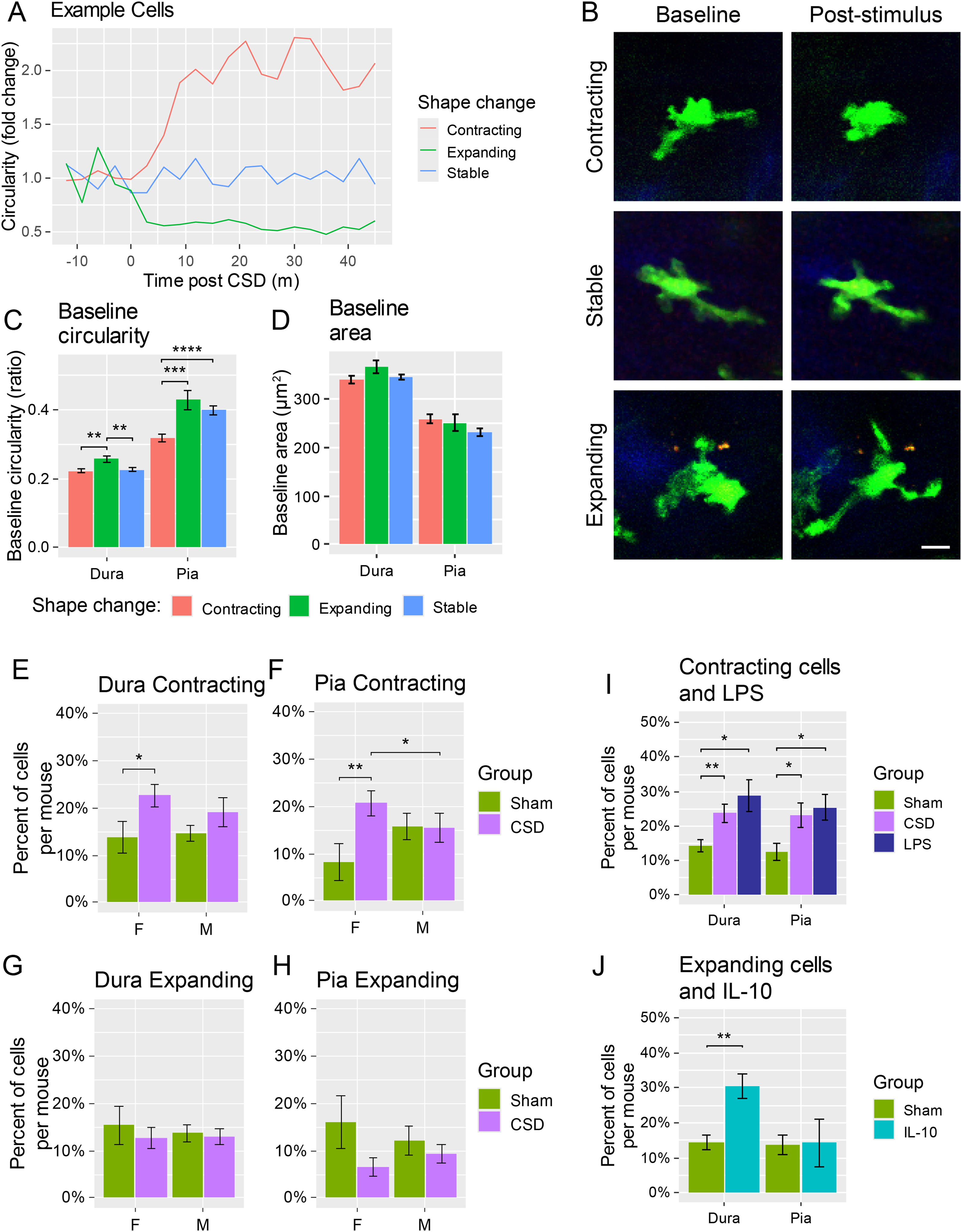
Contracting and expanding macrophage cells and effect of mouse sex. (a) Plot of fold change in circularity for three example cells pre- and post-CSD induction, color coded by whether the macrophage was determined to have contracted (significantly more circular by t-test, red), stable (no significant change, blue), or expanded (significantly less circular by t-test, green). (b) Images of example cells at baseline (left) and after stimulus (right, CSD or IL10-injection) that were determined to be contracted (top) stable (middle) or expanded (bottom). Scale bar 10 μm. (c) Baseline circularity (pre-CSD) of cells in the dura and pia in cells grouped by their post-CSD shape change. (d) Baseline area in the dura and pia based on post-CSD shape change. (For C and D, n = 316 cells contracting, 135 expanding, and 565 stable in the dura, 140 contracting, 46 expanding, and 226 stable in the pia) (e–f) Percentage of cells that became significantly more circular (“contracted”) in male and female sham and CSD-induced mice in the dura (e) and pia (f). (g–h) Percentage of cells that became significantly less circular (“expanded”) in male and female sham and CSD-induced mice in the dura (g) and pia (h). (For e–h, n = 6 F sham, 8 M sham, 11 F CSD, and 11 M CSD mice) (i) Percentage of contracted cells in LPS-injected mice compared to CSD-induced or sham (no CSD or LPS) in the dura and pia with both sexes combined. (j) Percentage of expanded cells in IL-10-injected mice compared to sham mice (no CSD or IL-10) in the dura and pia with both sexes combined. LPS and IL-10 had no significant differences between sexes, so they were combined and compared to the combined-sex CSD and Sham groups. (For i–j, n = 14 sham, 22 CSD, 6 LPS, and 5 IL-10 mice).
Looking retrospectively, these cells had some differing baseline characteristics. Expanding cells (that decreased their circularity post CSD) in the dura started with a significantly higher baseline circularity than contracting or stable cells (Figure 3(c), p < 0.001 compared to Contracting or Stable, Welsh's one-way ANOVA: F(2364) = 7.2, p = 8.4 × 10−4), while contracting and stable cells’ baseline circularities were not different from each other (p = 0.18 according to linear mixed-effects model; n = 316 cells contracting, 135 expanding, and 565 stable). In the pia, expanding cells were significantly more circular at baseline than contracting cells (p = 8.0 × 10−3) but not different than stable cells (p = 0.83), which were also significantly rounder at baseline than contracting cells (p = 4.8 × 10−3; Welsh's one-way ANOVA: F(2135) = 9.8, p = 1.0 × 10−4; n = 140 contracting, 46 expanding, and 226 stable). There were no significant size differences in the dura (Kruskal Wallis test χ²(2) = 4.9, p = 0.09) or pia (Kruskal Wallis test χ²(2) = 1.8, p = 0.41). Together, this data may suggest that cells that contract or expand may represent different initial populations of cells.
In sham mice that did not receive CSD, there was a distribution of about 14% of cells that contracted post-baseline, 15% of cells that expanded post-baseline, and 71% of cells that had no change, suggesting a baseline level of circularity changes that occurs in these mice under our experimental conditions.
Comparing CSD vs sham, we found that CSD significantly increased the percentage of contracting cells per mouse in the dura compared to sham in females (p = 0.03 according to t-test; n = 6 sham, 11 CSD mice), but not males (p = 0.11; n = 8 sham, 11 CSD mice; Figure 3(e)). There was also a corresponding increase of contracting cells in the pia of CSD-induced female mice compared to sham (p = 0.005, Figure 3(f)) but no apparent change in the percentage for males (p = 0.96). The percentage of contracting cells per mouse post-CSD was also significantly less in males than females in the pia (p = 0.03), but not the dura (p = 0.60). The percentage of cells that contract in response to CSD was surprisingly small, only around 10%–15%. This suggests that the significant increases in average circularity we see post CSD can be explained by a small subset of macrophages. There were no significant changes in the percentages of expanding macrophages in CSD vs sham, in the dura or pia for either sex (Figure 3(g) and (h)), suggesting that CSD does not affect the macrophages that expand their processes.
We next compared the proportions of macrophage shape changes in mice that received intravenous lipopolysaccharide (LPS), which is known to activate macrophages along an M1 pro-inflammatory pathway, or interleukin-10 (IL-10), which is known to polarize macrophages along an M2 anti-inflammatory pathway. There were no significant differences between sexes in LPS or IL-10 injected mice, so sexes were combined and compared to combined-sex CSD and sham groups. Interestingly, LPS caused a similar percentage of cells to contract as CSD in both the dura (23.7% ± 2.6 CSD, 28.8% ± 4.7 LPS; p = 0.37) and pia (23.1% ± 3.5 CSD, 25.5% ± 3.8 LPS; p = 0.66), all of which were significantly more than sham (p < 0.05, Figure 3(i)) (n = 14 sham, 22 CSD, 6 LPS mice). This may suggest that there is only a small subset of CX3CR1-positive macrophages that are capable of becoming activated within 45 min of an inflammatory stimulus. The percentage of expanding cells in LPS-injected mice was not different than in CSD or sham (not shown). In contrast, IL-10 injection (n = 5 mice) caused a significant increase in expanding cells compared to sham (or CSD) in the dura (p < 0.005) but not the pia (p > 0.25) (Figure 3(j)). The lack of response in the pia could be due to a failure of IL-10 to cross the blood brain barrier (43).
Onset of contracting but not expanding cells is later in the dura than in the pia
To determine when these shape changes for individual cells occur post-CSD, we asked which was the first time point post-CSD where circularity increased or decreased by at least twice the standard deviation of baseline for contracting or expanding cells, respectively (Figure 4(a)). We then compared the average time of onset of these changes in the dura and pia in female mice (that had a significant increase in contracting cells post-CSD) and found that the onset of shape change was significantly later in the dura than in the pia for contracting cells (11.2 ± 0.7 vs 7.0 ± 0.7, t = -2.5, p = 0.013 according to a linear mixed-effects model that accounts for within-mouse effects, n = 168 dura, 63 pia), but not for expanding cells (t = 0.39, p = 0.70, n = 67 dura, 10 pia) (Figure 4(b)). In addition, there was no difference between contracting and expanding cells’ time of onset in the dura (t = 0.34, p = 0.73), but contracting cells’ onset was earlier than expanding cells in the pia (t = 0.39, p = 0.04).
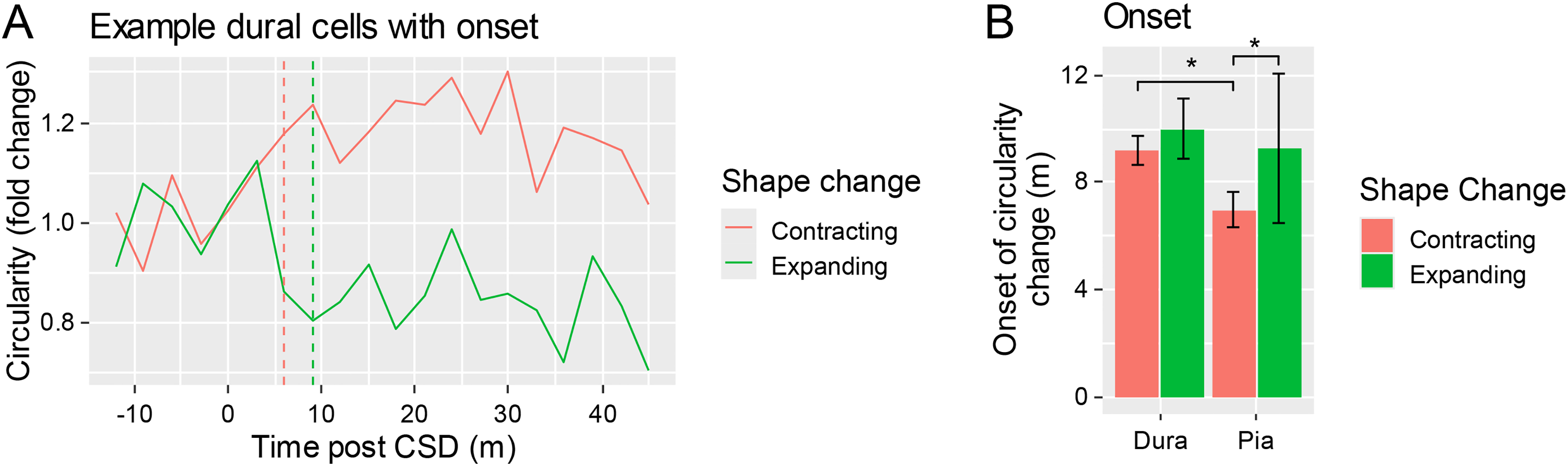
Onset of contraction or expansion in female mice. (a) Example cell circularity fold change in a contracted cell (red) and expanded cell (green), with their onset (time point at which the change from baseline exceeded at least 2x the baseline standard deviation) indicated in dashed lines. (b) Onsets of shape change of contracting or expanding macrophages in female mice in the dura and pia of CSD-induced mice (asterisks indicate significance according to linear mixed-effects model). (n = 168 contracting and 67 expanding cells in the dura, and 63 contracting and 10 expanding cells in the pia).
OnabotA significantly reduces macrophage activation compared to vehicle injection
We next sought to test the effects of onabotA (1 unit injected in 2 ul of saline) pretreatment or vehicle (2 ul saline), injected onto the skull suture just beneath the periosteum seven days before CSD induction, on macrophage activation by CSD in female mice. Pretreatment with onabotA (n = 9 mice) compared to saline (n = 6 mice) significantly decreased the number of macrophages at baseline in the dura (p = 0.01 according to t-test, Figure 5(a)), but not the pia (p = 0.19). Pretreatment with saline or onabotA had no significant effect on macrophage size or baseline circularity for either sex (p > 0.05 for all comparisons, using linear mixed-effects models, Figure 5(b)).

OnabotA reduces CSD-induced macrophage activation in females. (a) Number of macrophages per imaged area at baseline in the dura or pia for females in mice that received saline or onabotA injection one week previously. (b) Baseline circularity in the dura and pia in female mice that received saline or onabotA injection. (c) Percentage of contracted macrophages per mouse in female mice that received saline or onabotA in the dura and pia. (d) Percentage of expanded macrophages per mouse in female mice that received saline or onabotA in the dura and pia. (For a–d, n = 6 saline, 9 onabotA mice).
OnabotA pretreatment significantly reduced the percentage of macrophages that contracted post-CSD in compared to saline-treated female mice in the dura (p = 0.008 using t-test), but not in the pia (p = 0.19). OnabotA pre-treatment also increased the number of expanding macrophages in comparison to saline-treated female mice in the dura (p = 0.04), while having no effect in the pia (p = 0.48).
Discussion
Using in vivo multiphoton microscopy in mice with GFP expression under the CX3CR1 promoter to study dural macrophage behavior in a mouse model of migraine, we found that 20% of dural macrophages significantly change their cellular shape regardless of the occurrence of CSD, and that a small but significant 10% of macrophages contract their processes (i.e., become activated) in response to CSD. Surprisingly, we also found that 70% of macrophages do not change their cellular shape characteristics under any conditions (i.e., unresponsive/unaffected). We interpret these findings as suggesting that only 10% of all dural macrophages are responsive to CSD. We also found that while the number and shape characteristics of dural macrophages were similar in female and male mice, the percentage of cells that responded by contracting their processes (i.e., became active) increased in females but not males. This sex-specific difference calls attention to the possibility that the M1/M2-like polarity of dural macrophages may contribute to the difference in migraine prevalence among women and men. We also found that pre-treatment with onabotA (i) reduces the overall number of macrophages in the dura, (ii) reduces the percentage of M1-activated macrophages (i.e., the contracting cells) in response to CSD, and (iii) increases the percentage of M2-activated macrophages (i.e., the expanding cells) within the dura. Based on prior evidence (37,44) and their respective responses to LPS and IL-10, we conclude that the contracting cells are likely to be the pro-inflammatory M1-activated macrophages, whereas the expanding cells are likely to be the anti-inflammatory M2-activated macrophages. Along this line, we also conclude that onabotA is able to inhibit activation of pro-inflammatory macrophages, facilitate the activation of anti-inflammatory macrophages, and reduce the overall number of macrophages in the dura in females. These findings provide new insight into mechanism of action of this neurotoxin in prevention of migraine.
We found that the majority (70%) of macrophages remain stable (do not change their overall circularity) after CSD, that the morphology of 15%-20% changes regardless of CSD, and that only 10%-15% of the macrophages show a distinct response to CSD by contracting their processes. This is consistent with the single cell RNA sequencing studies that show the presence of multiple populations of macrophages in the dura (45,46), which likely map onto the multiple phenotypic subpopulations (47,48) of the M1 and M2 pathways. Macrophages in their non-activated state (M0) can be polarized into the pro-inflammatory “classically” activated M1 pathway, which can be associated with a contracting shape change (49–51), and secretion of multiple pro-nociceptive molecules such as IL6, IL12, TNF-α, and IL1β (21,25–27). They can alternately be polarized along an anti-inflammatory “regulatory”, or “wound-healing” (M2) pathway associated with a spindle-forming, expanding shape change (44), and reduction of nociception. Based on our observations that LPS caused dural macrophages to contract and IL-10 to expand, we concluded that the contracting cells are pro-inflammatory macrophages whereas the expanding cells are anti-inflammatory macrophages.
Only 10% of all dural macrophages were activated by CSD. However, if one takes into consideration the fact that less than half of dural nociceptors are activated by CSD or that far less than 1% of all trigeminovascular neurons in the spinal trigeminal nucleus project to the thalamus (along the classical sensory trigemino-thalamic pathway), it is not surprising that only a small number of macrophages are activated by CSD. But given macrophages’ ability to secrete a large quantity of inflammatory molecules that can impact neighboring cells and neurons (21,52), it is possible that even a small number of activated macrophages can increase the inflammatory environment in which meningeal macrophages and nociceptors co-exist.
Regarding the 20% of dural macrophages whose morphological changes (both contracting and expanding) were seen at baseline and in sham experiments, our hypothesis is that the thin skull surgery might induce these wound-healing M2 cells to expand, along with inducing some reacting M1 cells to contract, or that a certain subset of cells might be changing over time at baseline. Regarding the 70% of dural macrophages that did not seem to respond with circularity change post CSD or LPS, we speculate that these macrophages represent some of the macrophage phenotypes that may fall in between the classical M1/M2 categories (53), which may have more regulatory functions than reactionary phagocytic or destructive functions. This speculation is consistent with a broad expression pattern of macrophages that highly express CD206, but have low expression of MHCII, which is a substantial portion of the macrophages (∼50%) in the dura (45). The high presence of macrophages that do not respond to CSD, along with the high variability of circularity of macrophages at baseline, underscores the importance of in vivo quantification in the use of circularity to determine macrophage activation.
Using our method, we found that dural macrophages become activated only minutes after pial macrophages. Pial macrophages are on the brain side of the blood-brain barrier (37), and are thus directly exposed to all inflammatory signals emitted by the over-stressed neurons and astrocytes during CSD (54–56). Dural macrophages, on the other hand, are outside the blood brain barrier (37) and thus, are less likely to be affected directly by the brain signaling. It is possible that dural macrophage activation is triggered by the release of cytokines from the group of unmyelinated dural nociceptors that respond to CSD within minutes (57,58). Other possibilities include plasma protein extravasation and the resultant release of pro-inflammatory molecules in the dura, or the release of inflammatory molecules from dural lymphatics following the massive release of molecular waste by cortical neurons and astrocytes during CSD (55,59), and their clearance by the glymphatic vessels (60–62).
One study has suggested that the number of dural macrophages is larger in female than male rats (20). We found that CSD increases the number of contracting dural macrophages in female but not male mice. An increased pro-inflammatory tendency of macrophages in females could contribute to women's greater prevalence of migraine, as well as asthma, inflammatory bowel and Crohn's disease, eczema, and other autoimmune disorders (63).
The ability of onabotA to prevent the pro-inflammatory M1 macrophage response, facilitate the anti-inflammatory M2 macrophage response, and reduce the overall number of macrophages in the dura are novel findings and hold the potential to expand our understanding of onabotA's mechanism of action in migraine prevention, shifting focus from neuronal inhibition alone to its ability to attenuate responses and recruitment of inflammatory cells to the dura during migraine. This ability to reduce the overall number of macrophages is also consistent with our findings using onabotA in patients (36). We propose two potential scenarios to explain these data: 1) The effects could be secondary to onabotA's effects on nociceptors. If the activation of dural macrophages is at least partially mediated through the release of neuronal CGRP, substance P, and subsequent cytokines, and glutamate (64), onabotA's ability to reduce the release of these molecules from the nociceptors’ nerve endings (32,34) might be sufficient for attenuating the activation of macrophages. More specifically, we propose that the reduced macrophage population of cells in the dura may be mediated by inhibition of neuronally produced chemoattractants such as CCL2 and CCL7 (65) which can directly recruit blood-derived monocytes (66), or substance P, which induces production of chemoattractants MIP2 and MCP1 from already present macrophages (67). Recent evidence of CCL2 – CCR2 signalling in the development of sensitization and headache chronification through a signalling cascade involving expression of CCL2 in TG neurons and cells associated with dura blood vessels, and expression of CCR2 in subsets of macrophages and T cells in the TG and dura (68) further support this possibility. Additionally, unpublished data from our previous study (36) supports the idea that onabotA reduces macrophage numbers through a CCL2/7 – CCR2 axis as onabotA reduced expression of CCL2, CCL7, and CCR2 in the periosteum of migraine patients who responded to onabotA treatment. 2) OnabotA may act directly on macrophages mediated through Toll-like receptor 2 (TLR2). Evidence that onabotA alters expression of pro-inflammatory genes and presence of immune cells such as dendritic cells, T-cells, neutrophils and NK-cells in injected sites in the cranium, or in slice culture (36,69,70), and that it alters gene expression and reduced inflammatory responses in a murine alveolar macrophage cell line in vitro (71) through macrophage Toll-like receptor 2 (TLR2), are supported by the presence of TLR2 in trigeminal ganglia (72), its upregulation in the periosteum of migraine patients (73), and a dramatic reduction in its expression in the periosteum of migraine patients following onabotA injections (36).
In summary, the ability of onabotA to reduce the number of activated proinflammatory dural macrophages and increase the number of anti-inflammatory dural macrophages following the occurrence of CSD in our established animal model of migraine raises the possibility that onabotA can ‘calm’ meningeal nociceptors indirectly, by diminishing the number of inflammatory molecules secreted from macrophages in the environment where they exist. Equally important is the possibility that for their dominance in the dura, macrophage inhibition may represent a novel target for the development of anti-migraine drugs, and specifically for cases in which hyperactive inflammatory cells may be involved in the pathophysiology of the headache. Mechanistically, the data suggests that the mechanism of action of onabotA may not be simply due to its effects on nociceptors, but also due to an additional anti-inflammatory effect on the environment of the dura.
Article highlights
Percentage of pro-inflammatory contracting dural macrophages is increased in female but not male mice post-CSD
Pro-inflammatory contracting macrophage increase post-CSD is reversed by onabotA
Percentage of anti-inflammatory expanding macrophages in the dura post-CSD is increased by onabotA
Fewer total dural macrophages post onabotA
OnabotA may have a non-neuronal mechanism of action in migraine: reducing pro-inflammatory and increasing anti-inflammatory behavior
Footnotes
Author contributions
A.J.S., A.M.S, and R.B. were responsible for study concept and design. A.J.S. was responsible for acquisition of data. A.J.S. and D.D.F. analyzed and quantified the data. A.J.S., A.M.S., and R.B. drafted the manuscript and figures. R.S.B., A.D.B.-A., A.M.A., S.K., and M.F.B. provided editorial support for the manuscript.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: R.S.B., A.D.B.-A., A.M.A., and M.F.B. are full-time employees of Allergan, an AbbVie company. R.B. received funding for this study from Abbvie and is a consultant for Abbvie. A.J.S., D.D.F., A.M.S., and S.K. have no conflicts to declare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a research grant from Abbvie, and by NIH grants R01 NS094198 and R01 NS128045.
Ethical considerations
All procedures involving animals followed the experimental protocol approved by the Institutional Animal Care and Use Committee of Beth Israel Deaconess Medical Center and Harvard Medical School and adhered to the guidelines of the Committee for Research and Ethical Issues of the International Association for the Study of Pain (Protocols 005-2025 and 006-2022).
Data availability statement
Original data is available upon request.
