Abstract
Background
Neck pain is reported in more than 50% of migraine patients during migraine attacks and may be an important source to migraine pain.
Objectives
To investigate phenotypical differences between migraine patients with and without ictal neck pain in the interictal phase. Additionally, to prospectively examine the association between pericranial muscle tenderness and the impending migraine attack.
Methods
Migraine patients (n = 100) and controls (n = 46) underwent a semi-structured interview and sensory testing interictally. Pericranial muscle tenderness was determined using total tenderness score and local tenderness score. The occurrence of migraine attacks was then prospectively recorded for the following seven days.
Results
Patients with ictal neck pain had increased tenderness of pericranial neck muscles compared to migraine patients without (p = 0.023). Ictal neck pain was not associated with migraine localization, tension-type headache, or markers of central sensitization. Prospective data of 84 patients showed that tenderness of trigeminal sensory innervated muscles increased the migraine attack rate (p = 0.035).
Conclusion
The distinction of migraine patients based on the occurrence of ictal neck pain could indicate migraine subtypes and possible involvement of peripheral tissue in the pathophysiology. Whether treatment responses differ among these groups would be fascinating. Additionally, we found that cephalic muscle tenderness is a risk factor for an impending migraine attack.
Introduction
The most commonly reported pain locations during migraine attacks are the orbital, frontal, and temporal areas (1,2). However, neck pain has also been reported both at onset and later during migraine attacks (3), thus it does not appear to be rare. In fact, 63% of migraine patients reported neck pain during attack using a prospective electronic diary collection (4). Neck pain, in general, is also more prevalent in migraine patients than in healthy volunteers, and it is associated with tender pericranial muscles (5). The anatomical framework for neck pain could originate from sensory nerve fibers innervating neck muscles and the occipital part of the dura (6–8). Interestingly, activation of sensory C1-C2 neurons causes pain in the frontal region (9,10). Peripheral nociception in neck muscles may therefore be a source and contributor to migraine pain.
More than half of migraine patients had tender neck muscles during migraine attacks (11), and a study of patients with unilateral migraine have shown increased muscle tenderness on the affected side (12). It has been suggested that sensory thresholds fluctuate outside of migraine attacks and descend towards the next attack (13) and that this cycling may originate from structures in the central nervous system (14). Possibly as a consequence of a shorter timespan to next attack (13), pericranial muscle tenderness measured outside of attacks is associated with headache frequency (15).
It is unknown whether migraine patients who experience neck pain associated with their attacks (ictal neck pain) are phenotypically different from patients without ictal neck pain, in terms of neck muscle tenderness in the interictal phase. If so, this could imply differences due to local alterations in neck muscles or central sensitization. To investigate these migraine phenotypes, we performed in-depth phenotyping with sensory testing in 100 patients with or without ictal neck pain investigated outside of attacks and a control group. Furthermore, we prospectively investigated whether pericranial muscle tenderness could be a risk marker of an impending migraine attack in the following seven days after the examination.
Methods
Study population
One hundred patients diagnosed with migraine with or without aura according to the International Classification of Headache Disorders 3rd edition (ICHD-3) (16) were recruited from the Danish Headache Center from July 2017 to August 2018. For comparison, we included 46 age and sex-matched controls without migraine, frequent episodic tension-type headache (TTH) or first-degree relatives with migraine (Figure 1). Exclusion criteria were migraine attacks within 48 hours of the examination, intake of simple analgesic within 24 hours, intake of opioids/triptans within 48 hours of the examination, medication-overuse headache, muscle exercise of the upper body within 48 hours of the examination, musculoskeletal injuries or other known pathologies (i.e. cervical disc herniation, neck trauma) in the neck or shoulder. Pregnant and lactating women were excluded due to possible hormonal effects. Patients could receive preventive treatment if there were no change in prophylactic dosage for at least 3 weeks. The study was approved by the Danish National Committee on Health Research Ethics with the protocol number H17010860. The study was registered to clinical-trials.org with the identifier NCT03626805.
Included subjects and the examinations performed.
Semi-structured interview and terminology
Patients underwent a semi-structured interview identifying headache characteristics. The following parameters were defined: Ictal neck pain was either pain, soreness, or stiffness in the neck in more than 2/3 of the migraine attacks. Neck pain could occur either before the migraine attack (0–48 hours before), during the migraine attack or after (0–24 hours after). Patients were classified as having occipital migraine if they had migraine pain located in the occipital region and other parts of the head in at least one-third of their attacks. Isolated occipital migraine was defined as migraine pain exclusively at the occipital region. For this study, interictal neck pain was defined as neck pain without temporal relation to migraine attacks within the last year.
Total Tenderness Score (TTS)
All migraine patients and controls underwent sensory testing by the same examiner (JH). The examiner was trained to exert a pressure of 140 arbitrary units, corresponding to 80 kPa (17) during the testing. Training was performed with a palpometer (17). Eight paired pericranial muscles and tendons (Figure 2) were palpated in a standardized manner with small rotating movements. The patient was asked on a 4-point scale (0–3) how tender each location was. The score from each location was added to get a total tenderness score (TTS) ranging from 0 to 48. The method is reliable for testing pericranial tenderness (18). The TTS was divided into a cephalic tenderness score and a neck tenderness score, as previously described (19). This division was according to sensory innervation. Muscles and insertions innervated by the trigeminal nerve (frontal, temporal, coronoid process and masseter muscles) made up the cephalic tenderness score (cephalic-TS). The other muscles and insertions of the TTS (mastoid process, sternocleidomastoid, trapezius and neck muscle insertions) are innervated by the upper cervical roots and made up the neck tenderness score (neck-TS).
The total tenderness score consists of the cephalic tenderness score (blue markings) and the neck tenderness score (red markings).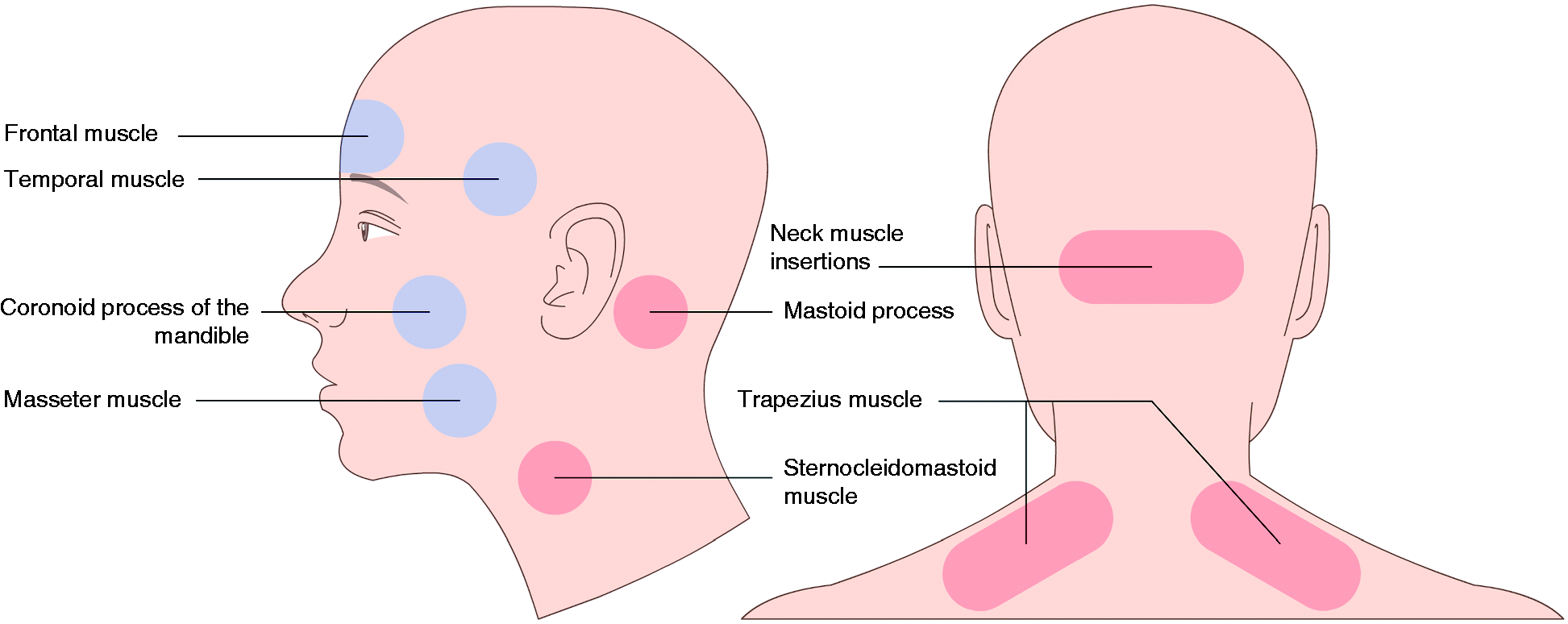
Local Tenderness Score (LTS)
Pressure controlled palpation was performed with the palpometer at a medial and lateral area of the trapezius muscle, defined as 1/3 and 2/3 of the length between the spinous process of C7 and acromion (Figure 3). The pressure used was 160 arbitrary units, corresponding to 105 kPa (17). The observer used small rotating movements with the second and third fingers and sustained the same pressure for 4–5 seconds. The subject specified the pain on a scale from 0 to 10, as described earlier (20). Measurements were repeated three times. Although similar in terms, the LTS is quite different from TTS. In comparison, the LTS measures at a more localized area, allowing us to compare anatomic locations close to each other.
Local Tenderness score and stimulus-response curve. The areas of the local tenderness score (proximal and distal trapezius, purple markings) and the area used for the stimulus-response curve. The stimulus response-curve was performed at the non-dominant side (green markings).
Stimulus Response function (SR-function)
The examiner pressed with seven different pressure intensities in the neck. The examiner had the palpometer attached to the second finger, for validating the intensities. The point in the neck was over the C5-transverse process at the non-dominant side (Figure 3). The patient marked the pain on a visual analog scale, which represented the span between no pain and the worst imaginable pain. This area was chosen since earlier studies have shown a large difference in pressure pain threshold when comparing migraine patients to controls (21). An abnormal SR-function is believed to indicate a central sensitization, which has been found in patients with frequent headaches (22,23). As a quantitative measure, we used the area under the curve (AUC).
Quantitative sensory testing (QST) and allodynia
Subjects were tested in accordance with the standardized protocol from the German Research Network on Neuropathic Pain (24). We examined two parameters from the protocol: Cold pain threshold (CPT) and heat pain threshold (HPT). In accordance with the protocol, we used the TSA-II thermode (Medoc, Ramat Yishai, Israel). All tests were illustrated and practiced before the actual measurements at a non-test site. The thermal test sites were the dorsal side of the non-dominant hand, the right, and left part of the forehead (V1-innervated). The patients were defined as allodynic if they had a cold pain threshold at one of the test sites between 20–32℃ or a heat pain threshold between 32–40℃, as previously described (25).
Headache diary
All patients were asked to prospectively use a paper headache diary for 7 days after the examination and return either by post or electronically.
Statistics
Demographics and QST parameters are presented with median, 25 and 75 percentiles. Differences among groups were tested using the Mann-Whitney U-test and Chi-square test. The Shapiro-Wilk test was used to test for normal distribution. The sample size was based on a prediction that 50% of migraine patients had ictal neck pain. The TTS and the derived measures were considered the main variables. Additionally, we wanted to establish a reliable prevalence of neck pain and headache location in this population.
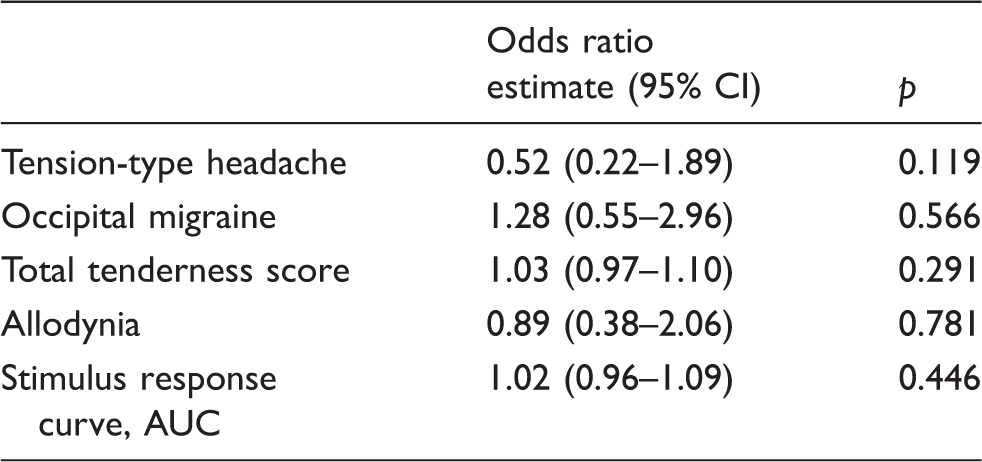
The primary outcomes were: a) If migraine patients with ictal neck pain had higher cephalic-TS, neck-TS, and LTS when correcting for headache frequency. This was tested using analysis of covariance (ANCOVA) analyses; b) if ictal neck pain is associated with occipital migraine, coexisting TTH and central sensitization parameters.
A multiple logistic regression model was performed with ictal neck pain as the dependent variable and the following covariates: TTH, occipital migraine, TTS, and central sensitization parameters of allodynia and SR-function (AUC).
The secondary outcomes were:
The relationship between pericranial tenderness (TTS, neck-TS, and cephalic-TS) and the next migraine attack, when correcting for headache frequency. This was analyzed using Cox proportional hazards analysis for each tenderness score and including the headache frequency as a covariate. TTS and associations with the following covariates: Years with migraine, headache frequency, interictal neck pain, occipital migraine, and allodynia, which were quantified with a multiple linear regression model. Interictal neck pain and the associations with the following covariates: Years with migraine, headache frequency, tension type headache, occipital migraine, and allodynia. This was quantified with a logistic regression model.
For descriptive statistics, Mann-Whitney U-test, and Cox survival analysis, we used IBM SPSS statistics, version 25. For the logistic regression models, and multiple linear regression model, we used SAS software version 9.4. The level of significance was set at 5%.
Results
The demographics of healthy controls and migraine patients with and without ictal neck pain included in the study.

BMI: Body mass index; AUC: Area under the curve.
Note: For numeric variables, median, 25 and 75 percentiles are shown. Comparisons of groups in numerical data were performed with Mann-Whitney U-test. Categorical data were tested with chi-square.
**Significant difference between the group of migraine patients and healthy controls, p < 0.001.
We found that 52 % of migraine patients reported ictal neck pain. Of those, 85% reported neck pain before, 90% during, and 37% after migraine attacks (only one had neck pain occurring exclusively after the migraine attack). Prodromes were reported by 70% of all migraine patients. In total, 77% of migraine patients with ictal neck pain reported prodromes compared to 63% of patients without ictal neck pain (p = 0.116). We did not find any relationship between ictal neck pain and any migraine characteristics or demographics such as occipital migraine or headache frequency (Table 1).
Pericranial muscle tenderness in patients with ictal neck pain
Tenderness scores for healthy controls (uncorrected) and the two subdivisions of migraine patients corrected for headache frequency.

Note: p-values in bold are statistically significant. Comparison with healthy controls was performed on uncorrected data using Mann-Whitney U-test. Significance is indicated by *p < 0.05, **p < 0.001.
Clinical characteristics and the odds-ratio for ictal neck pain.
Pericranial muscle tenderness associated with days to next migraine attack
Eighty-four (84%) patients returned their diary. Sixty (71%) patients experienced a migraine attack during the 7 days (Figure 4). Corrected for headache frequency, we found that cephalic-TS, but not neck-TS or TTS, is a risk factor for developing a migraine attack (Table 4). An increase of one cephalic-TS point increased migraine rate by 7% (95% CI: 1–13%, p = 0.035). An estimated difference of 10 cephalic-TS points caused almost twice as large a migraine rate, 92% (95% CI: 5–353%). Predicted estimates for patients with cephalic-TS of 3 and 13 and a headache frequency of 14 per month (the mean of our migraine population) is illustrated in Figure 5. The range in cephalic-TS among migraine patients was 0–17.
Descriptive data showing when the 84 examined migraine patients had their first migraine attack following the examination. Day 0 is on the day of examination. Patients are divided in a high/low group according to the depicted median tenderness score. Red: Patients with tenderness score ≥ median. Blue: Patients with tenderness score < median. The Cox proportional hazard model estimates the migraine incidence when correcting for the headache frequency. The impact of cephalic tenderness score is illustrated by showing the estimated incidence for cephalic tenderness scores of 3 (blue) and 13 (red). Muscle tenderness scores and the hazard ratio estimates for getting a migraine attack, corrected for headache frequency. Note: p-values in bold are statistically significant.


Total tenderness score and headache frequency
Clinical characteristics and the associations with total tenderness score in migraine patients.

Note: p-values in bold are statistically significant.
Associations of interictal neck pain
Interictal neck pain was equally distributed among migraine patients with (46%) and without (48%) neck pain during migraine attacks. We found that 30% of controls have had neck pain during the last year.
Clinical characteristics and the odds ratio for interictal neck pain in migraine patients.
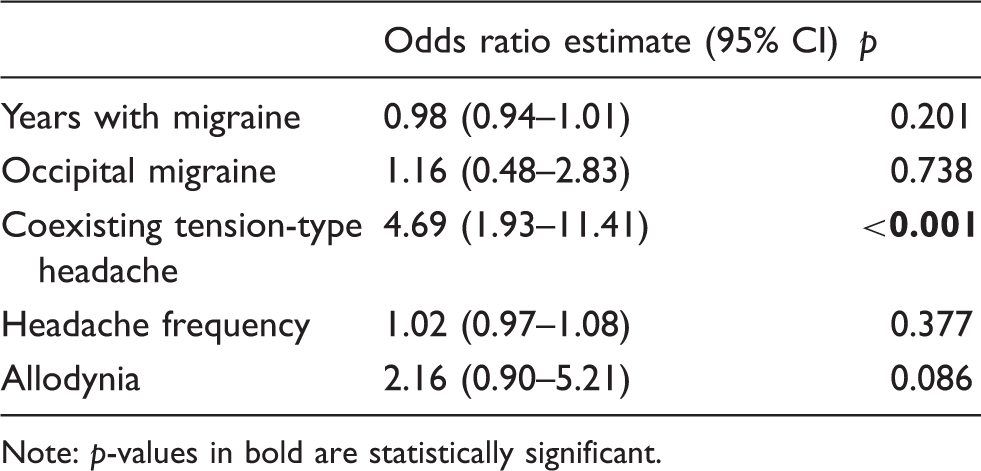
Note: p-values in bold are statistically significant.
Discussion
The most important findings in this study are that migraine patients with ictal neck pain had more tender neck muscles compared to migraine patients without ictal neck pain investigated in the interictal state. Ictal neck pain was not associated with occipital migraine location, coexisting TTH, TTS, or signs of central sensitization outside of attacks. The study also revealed that high cephalic-TS is a risk factor for an impending migraine attack, even when correcting for headache frequency. Allodynia was detected in 54% of the controls and was not more frequent in either migraine patients with or without ictal neck pain.
Pericranial muscle tenderness and the association to migraine
Sensory neurons in C2 and C3 dorsal root ganglions innervate both suboccipital muscles and the dura overlying the cerebellum (8). Several studies have shown referral of pain to cephalic trigeminal innervated areas in humans following stimulation of neck muscles (9), cervical roots (10,26), the infratentorial dura (27), and subcutaneous tissue innervated by C2 (28,29). Furthermore, stimulation of the greater occipital nerve induces increased excitability of dural afferent input (30), and greater occipital nerve block is followed by an increase of pressure pain thresholds in the trigeminal area (31). The intertwined relationship between trigeminal and cervical extracranial pain signaling is also evident from the findings in the present study. Only 3% of migraine patients reported isolated occipital migraine pain location, which is in line with a previous large cohort of 1283 migraine patients, where 0.8% reported isolated occipital headache (1). Thus, almost all our patients with occipital migraine pain also experienced pain in trigeminal innervated areas, which suggests either that both trigeminal and cervical sensory nerves are activated or the presence of a functional connection at the second-order neuronal level in the trigeminocervical complex or higher central levels. The interictally increased neck muscle tenderness in patients with ictal neck pain suggests a latent peripheral sensitization, which could contribute to the development of migraine attacks. Local inflammation or other processes could cause this peripheral sensitization via collateral dural or muscle nociceptors. In support, a recent study reported that chronic migraine patients with muscle tenderness had upregulation of inflammatory genes in the calvarial periosteum (32). Pro-inflammatory substances have also been found in increased concentrations in the trapezius muscle in subjects with active trigger points (33), which are more prevalent in patients with migraine compared to healthy controls (34–36). CGRP mRNA was upregulated in temporal relation to inflammation of cranial muscles in experimental models, suggesting an important role for CGRP in muscle nociceptive processing (37). Anti-CGRP monoclonal antibodies are effective in episodic and chronic migraine (38) and do not cross the blood-brain barrier in effective doses (39). CGRP-positive nociceptive neurons are found in muscles in animal models (40). Thus, further investigation of local pathologies in neck muscles in migraine patients is highly warranted based on our findings.
Pericranial muscle tenderness and days to next migraine attack
The present study demonstrated that increased cephalic-TS, but not neck-TS or TTS, is associated with a higher risk of an impending migraine attack despite correcting for headache frequency. This is the first study to prospectively examine such an association. A study examining migraine patients during and outside of migraine attacks showed that pericranial muscles are more tender during attacks (12). Whether this means that a gradual increase in pericranial tenderness prior to a migraine attack is unknown, and needs to be examined in longitudinal studies. It is interesting that we only found local cephalic-TS as a risk factor for the development of a migraine attack, which could point to sensory changes in the cephalic muscles, the trigeminal nerve, or centrally before migraine attacks. However, we cannot rule out that the TTS and neck-TS are insignificant due to the variability caused by interictal neck pain and neck muscle tenderness without context to migraine. Our results show a rather large confidence interval, reflecting the inter-patient variation in cephalic-TS, and thus these results should be interpreted with caution. More precision could probably be achieved with more personalized scores or additional measurements over time.
Total tenderness score and interictal neck pain
We showed that episodic migraine patients had a higher TTS than controls, and chronic migraine patients had a higher TTS than episodic migraine patients, in line with a previous study (19), whereas other studies did not find a difference among migraine patients and controls (23,41). We also showed that TTS is associated with headache frequency, as previously shown in both TTH patients (42) and migraine patients (12). The association between TTS and allodynia implies that TTS also reflects sensitization of skin nociceptors at a peripheral or central level. Interestingly, interictal neck pain was associated with the coexistence of TTH, but not headache frequency or allodynia. Neck pain prevalence over the last year has been reported to occur in 76% of patients with pure migraine, and 89% in patients with both TTH and migraine in a population-based study (5). In the present clinic-based study, we found that 46% of migraine patients with ictal neck pain also had interictal neck pain over the last year, while only 30% of controls reported neck pain. The 1-year prevalence of neck pain in the general population varies quite a lot depending on the study design, showing ranges from 5–80% (mean: 26%) in 28 studies with low or moderate risk of bias (43), but our findings support a higher occurrence of interictal neck pain in episodic migraine patients.
In summary, this study demonstrates increased local muscle tenderness in migraine patients with ictal neck pain compared to patients without. We suggest this is a result of peripheral changes because of the very localized findings. We did not find any association with ictal neck pain and either clinical migraine characteristics or neck pain experienced interictally. Our study also demonstrated that patients with a high cephalic tenderness score have an increased migraine risk, when correcting for headache frequency. This could indicate the general decrease in sensory thresholds toward a migraine attack (13).
Limitations
Data were collected by an examiner who was not blinded for headache diagnosis or severity. The diagnosis and headache characteristics of each patient were not prospectively collected using a headache diary, which may have flawed the results due to recall bias. Some patients were worried that the tenderness testing could cause an attack. This, in combination with asking patients to keep a headache diary and the actual testing, could cause more attacks and this might be the reason that within 48 hours after the examination 42% of respondents reported migraine attacks. To skew our prospective results, an interaction between increased muscle tenderness and the higher attack rate effect had to be present. Our migraine cohort consisted of migraine patients with a median duration of 19 years with migraine, which might cause changes to have plateaued out and could explain why we do not see any association with the duration of migraine. Patients were from a highly specialized tertiary headache center and we had very few male participants, which makes a generalization to migraine patients in the general population difficult.
Conclusions
The present study demonstrates that migraine patients with ictal neck pain have increased tenderness of neck muscles interictally; this localized neck tenderness suggest that there are peripheral alterations in a subgroup of migraine patients, although central sensitization may also be involved. Future studies should investigate the underlying cause of this localized neck tenderness. Cephalic tenderness increases the risk of an impending migraine attack, which might be useful in some patients for predicting and ultimately starting treatment earlier.
Footnotes
Key findings
Migraine patients with ictal neck pain have increased neck muscle tenderness outside of attacks.
The localized findings in patients with ictal neck pain indicate a peripheral contribution to migraine in this subtype.
Increased tenderness in trigeminally innervated muscles is a risk factor for an impending migraine attack.
Acknowledgements
We thank our colleague Thien Phu Do for creating the figures and statistician Lene Theil Skovgaard for guidance with the statistical analysis. We greatly appreciate the discussions with Professor Jes Olesen during preparation of the manuscript.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: HWS has received speaking fees from Novartis and Teva, and has received grant from Novartis. MA has received personal fees from Alder BioPharmaceuticals, Allergan, Amgen, Eli Lilly, Novartis, and Teva, and also participated in clinical trials as the principal investigator for Alder, Amgen, electroCore, Novartis, and Teva. This author also serves as an associate editor of Cephalalgia, associate editor of Headache and co-editor of the Journal of Headache and Pain, and is President of the International Headache Society and General Secretary of the European Headache Federation. MA also reports research grants from Lundbeck Foundation, Research Foundation of the Capital Region of Copenhagen, and Novo Nordisk Foundation. The other authors declared no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present work was funded by Candys Foundation, Torben & Alice Frimodts Fond and The A.P. Møller Foundation for the Advancement of Medical Science.
ORCID iDs
Jeppe Hvedstrup https://orcid.org/0000-0002-4342-6226 Samaira Younis ![]()
