Abstract
Background
Migraine is a complex disabling neurological disease characterized by recurrent headache attacks lasting 4–72 h with moderate to severe intensity and other accompanying symptoms. While chronic migraine (CM) and episodic migraine (EM) are primarily differentiated by the frequency of headache and migraine days, underlying clinical and functional differences have been described. OnabotulinumtoxinA (onabotA) has been approved as a preventive treatment for CM with controlled clinical and real-world evidence suggesting potential benefits for treatment of EM. Given the lack of randomized controlled trial data, PRECLUDE, a prospective phase 3 trial was designed to evaluate the efficacy and safety of onabotA for the preventive treatment of EM.
Methods
The PRECLUDE trial was a phase 3 multicenter, randomized, double-blind, placebo-controlled, parallel-group trial with an open-label onabotA 195 U extension phase. In total, 775 patients aged 18–65 years with a history of migraine attacks were randomized (1:1:1) to receive placebo, onabotA 155 U, or onabotA 195 U. Patients recorded daily headache data and medication use via an electronic diary (eDiary) during a four-week screening phase, 24-week double-blind phase, followed by a 24-week open-label extension phase. The primary endpoint was the change in the frequency of monthly migraine days from baseline across months 5 and 6.
Results
All treatment groups showed a reduction in the frequency of monthly migraine days from baseline; however, neither the onabotA 155 U group nor the 195 U group demonstrated a statistically significant improvement compared to the placebo group (p >0 .05). Similarly, secondary endpoints, including changes in monthly headache days, 50% responder rates and monthly acute medication use days, did not reach statistical significance. Adverse events in this trial were consistent with previous findings for onabotA in CM and were generally mild to moderate in severity.
Conclusions
The PRECLUDE trial demonstrated that onabotA was well tolerated but did not show significant efficacy compared to placebo for the endpoint reducing migraine days from baseline in patients with EM as defined by the trial protocol. While onabotA is effective for CM, these findings highlight the need for further research to better understand the pathophysiological differences between EM and CM and to understand whether there is a potential subset of EM patients which respond to onabotA.
This is a visual representation of the abstract.
Keywords
Introduction
Migraine is a complex disabling neurological disease that significantly impairs quality of life (1). It is defined by the presence of recurrent headaches, often accompanied by symptoms such as nausea and a heightened sensitivity to light or sound (2–4). It is one of the most prevalent neurological diseases, affecting more than one billion people worldwide, and is ranked second among the cause of years lived with disability worldwide (5,6). The World Health Organization has identified migraine as a significant public health concern due to its high prevalence and impact on quality of life, a recognition which dates back to 2001 (7).
To guide clinical practice, the International Classification of Headache Disorders, 3rd edition (ICHD-3) defines chronic migraine (CM) based on the presence of attacks meeting migraine criteria or response to migraine specific acute treatment at least 8 days per month and at least 15 monthly headache days (MHDs) (2–4). By convention, people with migraine who fall short of the CM frequency threshold are said to have episodic migraine (EM) (8). CM and EM have been distinguished by differences in pathophysiology, comorbidities and disability (8–10). The International Headache Society (IHS) and guidance documents from global health authorities (e.g. Food and Drug Administration) also recognize the clinical difference between CM and EM, and therefore recommend that, when assessing the safety and efficacy of new treatments, these two patient populations are studied independently because the response to treatment may vary between them (2–4,11).
OnabotulinumtoxinA (onabotA) was established to be efficacious and safe for the treatment of CM in the PREEMPT 1 and 2 trials, which led to the approval by the US Food and Drug Administration (FDA) and European Medicines Agency in 2010 and other regulatory agencies around the world for the prevention of CM (12). PREEMPT 1 and 2 were randomized, double-blind, placebo-controlled clinical trials, each with an open-label extension phase. Both trial results demonstrated significant improvements compared to placebo in multiple headache outcomes including reduction in monthly migraine days, headache-related symptoms and headache-related disability; the trials also showed improved functioning, as well as overall health-related quality of life (13,14). Additionally, several open-label and real-world studies of onabotA treatment in CM further support these findings. A two-year open label long-term trial in 715 CM patients demonstrated consistent reduction of headache days (7–10 days reduction on average) with repeated treatments of onabotA every 12 weeks for up to nine treatment cycles (15). Improvements in health-related quality of life measures, patient satisfaction and continuous utilization of onabotA have been described in several prospective real-world studies. Additionally, a recently published meta-analysis of 10 years of real-world data evaluated the effectiveness of onabotA for the prevention of CM and concluded that real-world practice corroborated evidence from pivotal and long-term open-label studies of onabotA in CM preventive treatment (16).
Today, onabotA is listed as a first-line treatment option for CM by the American Headache Society and the American Academy of Neurology (17,18). Migraine treatment guidelines state onabotA should be offered as a treatment option to patients with CM to increase the number of headache-free days (Level A evidence) (17,18) and advisory bodies such as NICE UK and the National Health System in Spain recommend the use of onabotA as preventive treatment for CM before the use of anti-calcitonin gene-related peptide (CGRP) monoclonal antibodies (19,20).
While onabotA is approved for the preventive treatment of headache in adults with CM, patients who experience EM are not covered in the current indication because the efficacy and safety of onabotA use has not been established in randomized controlled trials. Earlier phase 2 randomized, double-blind, placebo-controlled studies evaluating safety and efficacy of onabotA in EM patients conducted from 1997–2004 did not demonstrate statistically significant improvement over placebo (21–24). However, these early phase 2 studies used different doses, injection paradigms, endpoints and evolving definitions for migraine. Subsequent emerging evidence from real-world studies involving high frequency EM patients and subgroup analyses of the previously conducted trials suggested that EM patients could benefit from treatment with onabotA. In two small open-label real-world studies and a subgroup analysis of phase 2 double-blind, placebo-controlled trials of onabotA, patients with high-frequency EM showed a reduction in monthly migraine days (3.68 days), a reduction in frequency of headache days (5.78 days) and a reduction in frequency of headache episodes (4) following onabotA treatment, respectively (see supplementary material, Table S2) (23,25,26).
Another study with long-term follow-up of patients with CM and high frequency EM treated with onabotA found similar proportions of reduction in headache day frequency between CM and high frequency EM at month 12 (CM 44.7% vs. high frequency EM 41.2%) (27). Additionally, patients with high frequency EM fluctuate in headache frequency and often move above the threshold for CM (28), suggesting that some may have a similar underlying biological or physiological characteristics (biotype) to CM. Preventive treatment may control fluctuation and reduce chronification. A longitudinal study over one year showed that 14.1% of patients with EM experienced worsening (increased frequency of headaches) and EM patients without adequate preventive treatment were more likely to demonstrate clinical signs of progression (29).
Considering that onabotA has a unique mechanism of action with established long-term safety in CM and is a non-systemic treatment given every 12 weeks, it has the potential to be a valuable treatment option for patients with EM. Encouraged by over 14 years of onabotA use in CM, the evidence in high frequency EM and the unmet medical need, the PRECLUDE trial intended to evaluate the efficacy of onabotA treatment in patients with lower headache frequency who are currently not covered in the approved indication. Therefore, the objective of this double-blind, placebo-controlled trial was to evaluate the efficacy and safety of two doses of onabotA (onabotA 155 U and onabotA 195 U) for preventive treatment in adults with EM.
Methods
Trial design
PRECLUDE was a multicenter, international, randomized, double-blind placebo-controlled, parallel-group trial with an open-label extension phase in adults with EM. The trial consisted of a four-week screening/baseline phase using an electronic diary (eDiary). Eligible patients were randomized 1:1:1 to receive placebo, onabotA 155 U or onabotA 195 U, and were administered two treatments at 12-week intervals during the 24-week double-blind placebo-controlled treatment phase. Those who completed the double-blind placebo-controlled phase continued into the 24-week open-label treatment phase and received up to two additional treatment cycles of onabotA 195 U given at 12-week intervals (Figure 1) (see detailed methods in the Supplementary material, Table S4).

PRECLUDE study design.
Trial patients
Patients were 18–65 years of age at visit 1 with a history of migraine attacks meeting ICHD-3 diagnostic criteria for migraine for ≥12 months, with onset before 50 years of age. Specifically, patients enrolled in the study had 6–14 migraine days and <15 headache days per month in the three months prior to screening and during the four-week screening period. Additionally, patients with a current diagnosis of chronic migraine were excluded from the study. Study patients were recruited from November 2021 to December 2023.
Randomization and trial treatment
In total, 775 patients were randomized 1:1:1 to receive onabotA 155 U, onabotA 195 U or placebo and were treated at 118 sites in the USA, Canada, UK, Spain, Germany, Sweden, Czech Republic, Poland and Israel. In total, 774 patients were treated and included in the safety population. Concurrent use of any migraine prophylactic treatment during screening or throughout the trial was not allowed. During the double-blind phase, to ensure the injector and patients were blinded to treatment, all patients received the same number of injections (i.e. 39 injections) of 0.1 ml of onabotA or placebo in the head, face and neck in a fixed-site-fixed-dose manner and an independent drug reconstitutor prepared the dosing syringes. Placebo patients received 39 injections of placebo, onabotA 155 U patients received 31 injections of onabotA following the PREEMPT protocol (30,31) and eight injections of placebo, and onabotA 195 U patients received 39 injections of onabotA (PREEMPT protocol plus eight additional sites). During the open-label, all patients received 39 injections of onabotA for a total dose of 195 U.
Outcome measures
Efficacy
Efficacy assessments were based on information recorded by participants. A handheld eDiary was provided to the participant. The primary efficacy endpoint was the change from baseline in the frequency of monthly migraine days across months 5 and 6 (weeks 17–24). The primary timepoint of months 5 and 6 refers to weeks 5–12 after the week 12 injection visit. A migraine day is defined as any calendar day on which a headache occurs which meets criteria A, B and C (migraine headache day) OR meets criteria D and E (probable migraine headache day), as listed in the Supplementary material (Table S1), operationalized using the eDiary.
Secondary endpoints included change from baseline in the frequency of monthly headache days across months 5 and 6, responder status of at least 50% reduction from baseline in the frequency of monthly migraine days across months 5 and 6, and change from baseline in the frequency of monthly acute headache medication days across months 5 and 6. Secondary patient-reported outcome endpoints include, change from baseline in Migraine-Specific Quality-of-Life Questionnaire, version 2.1 (MSQ v2.1) Role Function – Restrictive (RFR) (32) at month 6, Activity Impairment in Migraine – Diary (AIM-D) (33) across months 5 and 6, and 6-item Headache Impact Test (HIT-6) (34) across months 5 and 6.
Safety
The safety parameters included adverse event (AE), clinical laboratory test results (hematology, chemistry and urinalysis), vital signs and Columbia-Suicide Severity Rating Scale (C-SSRS) (35) scores. An AE was considered a treatment-emergent adverse event (TEAE) if the AE began or worsened (increased in severity or became serious) on or after the date (and time, if known) of the first trial intervention. An AE was considered a treatment-emergent serious adverse event (TESAE) if it was a TEAE that additionally met any serious adverse event (SAE) criterion.
Results
Demographics and clinical characteristics
The intent-to-treat population included 775 patients randomized patients; 257 (33.2%) to placebo, 257 (33.2%) to onabotA 155 U and 261 (33.7%) to onabotA 195 U. The safety population included all treated subjects, giving a total of 774 patients grouped according to treatment received at first injection; 257 (33.2%) placebo, 255 (32.9%) onabotA 155 and 262 (33.9%) onabotA 195 U. There were no significant differences observed between the baseline demographics of the three treatment groups. The mean age was 40 years for placebo group, 41 years for onabotA 155 U and 41 years for the onabotA 195 U. Most patients were female, placebo, 231 (89.9%); onabotA 155 U, 227 (88.3%); and onabotA 195 U, 224 (85.8%) (Table 1).
Baseline demographics – ITT population.

BMI = body mass index; ITT = intention to treat; SD = standard deviation; U = unit.
Patient disposition
In the double-blind phase of the trial, a majority of patients (86.5%) successfully completed the 24-week treatment period, 86.1% of the patients entered the open-label phase and 74.5% of patients completed the 24-week open-label phase. Rates of discontinuation and the main reasons for discontinuation were similar across the different treatment groups. Primary reasons for discontinuation during the double-blind phase included withdrawal by the patient (8.9%), loss to follow-up (2.3%) and other reasons (1.9%) (Figure 2a). The main reasons for trial discontinuation during the open-label phase were withdrawal by subject (4.6%), trial terminated by sponsor (2.5%) and other reasons (2.3%) (Figure 2b). Twelve patients discontinued due to AEs at the end of double-blind phase and 6 patients discontinued due to AE at end of open-label phase.

Participants disposition. 2a. Participants disposition during double-blind phase – ITT population. 2b. participants disposition during open-label phase – ITT population. ITT = intention to treat.
Efficacy results
Primary endpoints
The primary endpoint of change from baseline in the frequency of monthly migraine days across months 5 and 6 (weeks 17–24) showed meaningful improvement from baseline for all treatment groups, but did not demonstrate statistically significant improvement of either dose of onabotA, 155 U or 195 U compared to placebo. The baseline mean frequency of monthly migraine days was similar across the trial groups: 8.9 days for placebo, 8.7 days for the onabotA 155 U group and 8.6 days for the onabotA 195 U group. The mean change from baseline was −3.6 days for placebo, −3.5 days for the onabotA 155 U group and −3.4 days for the onabotA 195 U groups at the primary timepoint. Least squares (LS) mean changes from the mixed model repeated measures (MMRM) model were −3.0 days for placebo, −3.1 days for the onabotA 155 U group and −3.0 days for the onabotA 195 U group. LS mean differences between either onabotA 155 U or onabotA 195 U, compared to placebo, were not statistically significant (p >0 .05) (Figure 3 and Table 2). The results from the supplemental analysis of covariance (ANCOVA) of ranks were consistent with the results from the primary analysis (p = 0.572 for the onabotA 155 U versus placebo group and p = 0.644 for the onabotA 195 U versus placebo group).

Primary endpoint.
Primary endpoint.

LS = Least squares; MMRM = mixed-effects model for repeated measures; SD = standard deviation; SE = standard error.
Secondary endpoints
No statistically significant differences were observed between the treatment and placebo groups for secondary efficacy endpoints. LS mean changes in monthly headache days at months 5 and 6 were similar across groups, with the placebo group experiencing a decrease of 3.2 days and the onabotA 155 U and onabotA 195 U groups showing decreases of 3.1 and 2.9 days, respectively. LS mean differences between either dose of onabotA, 155 U or 195 U, compared to placebo, were not statistically significant (p > 0.05). LS mean changes in monthly acute headache medication days at months 5 and 6 were similar across groups, with the placebo group experiencing a decrease of 2.0 days and the onabotA 155 U and onabotA 195 U groups showing decreases of 1.9 and 1.8 days, respectively. LS mean differences between either dose of onabotA, 155 U and 195 U, compared to placebo, were not statistically significant (p > 0.05). Additionally, the proportion of participants achieving at least 50% reduction in monthly migraine days was 46.2% for the onabotA 155 U dose and 47.1% for the onabotA 195 U dose compared to 44.9% in the placebo group, and these differences were not statistically significant (p > 0.05) (Figure 4 and Tables 3–6). The secondary endpoints of patient-reported outcomes showed no significant differences between the treatment groups and placebo. Specifically, the MSQ v2.1 RFR domain, which assesses migraine-related limitations in daily activities, did not show statistically significant improvements for either treatment group compared to placebo. LS mean differences were 0.8 for the onabotA 155 U dose and 0.4 for the 195 U dose (p > 0.05). The AIM-D Physical Impairment domain, which measures physical limitations due to migraine, also showed no statistically significant differences. The LS mean differences from placebo were 0.4 for the 155 U dose and 0.2 for the 195 U dose (p > 0.05). HIT-6 total score, reflecting the overall impact of headaches on daily life, was not significantly different between treatment groups and placebo (P > .05) (Tables 3–6). The results from the supplemental ANCOVA analysis of ranks for all secondary endpoints were consistent with the results from the primary MMRM analysis.

Secondary efficacy endpoint.
Secondary Endpoints.

SD, standard deviation; SE, standard error; LS, Least squares.
Secondary Endpoints.

SD, standard deviation; SE, standard error; LS, Least squares.
Secondary Endpoints.

SD, standard deviation; SE, standard error; LS, Least squares.
Secondary Endpoints.

AIM-D, Activity Impairment in Migraine Diary; HIT-6, 6-Item Headache Impact Test; LS, Least squares; MSQ, Migraine-Specific Quality of Life Questionnaire; PI, Physical Impairment; RFR, Role Function Restrictive; SD, Standard Deviation; SE, Standard Error; U, Unit.
Safety endpoints
In the double-blind phase of the trial, adverse events profiles were generally comparable across treatment groups. TEAEs were reported in 342 of 774 participants (44.2%) across all treatment groups. Of these, 106 (41.2%) were in the placebo group, 117 (45.9%) in the onabotA 155 U group and 119 (45.4%) in the onabotA 195 U group. The incidence rate of treatment-related TEAEs (TR-TEAEs) was greater in onabotA 155 U and onabotA 195 U than for placebo. The incidence rate of TESAEs was generally low among the treatment groups (<3%). None of the TESAEs were considered to be related to trial treatment by the investigator. None of the TEAEs suggested a distant spread of toxin. The incidence rate of TEAEs leading to withdrawal of trial treatment was also generally low (<3%) (Table 7). Patients with TR-TEAEs ≥1% included eyelid ptosis (2.2%), headache (1.8%), injection site pain (1.4%), migraine (1.4%) and injection site reaction (1.0%). Brow ptosis and neck pain were less common, being observed in 0.8% and 0.6% of participants, respectively (Table 8).
Summary of adverse events double-blind phase – safety population.
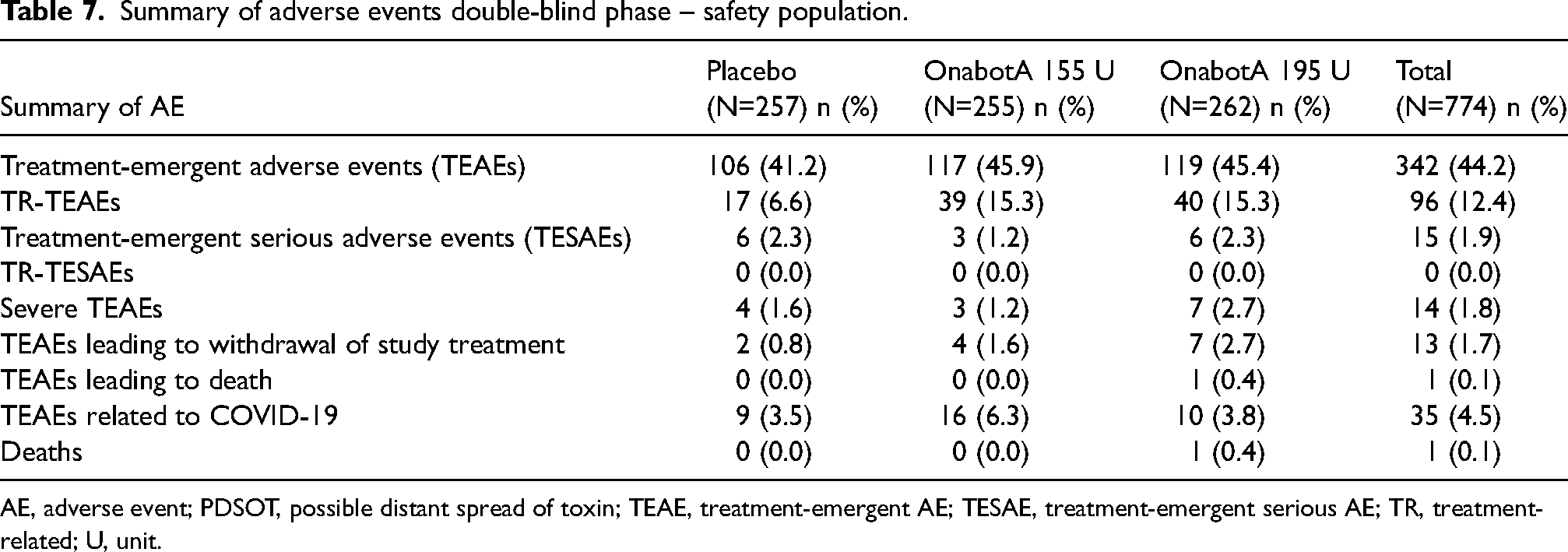
AE, adverse event; PDSOT, possible distant spread of toxin; TEAE, treatment-emergent AE; TESAE, treatment-emergent serious AE; TR, treatment-related; U, unit.
Most frequent (≥1%) TR-TEAEs in double-blind phase – safety population.

TR-TEAE, treatment-related treatment-emergent adverse event; U, unit.
In the open-label phase of the trial, the overall incidence of TEAEs was comparable across groups, with 35.0% in the placebo group, 33.3% in the onabotA 155 U group and 35.9% in the onabotA 195 U group (Table 9). TR-TEAEs were reported in 9.7% of placebo subjects but were notably lower in the onabotA groups, at 5.3% and 5.8% for the onabotA 155 U and onabotA 195 U doses, respectively. The incidence of TESAEs was low, at 0.9% in the placebo group and 0.4% in both onabotA groups, with no serious TR-TESAEs reported. Severe TEAEs appeared more frequently in the onabotA 155 U group at 1.3% compared to 0.5% in the placebo and 0.4% in onabotA 195 U groups (Table 9). None of the TEAEs suggested a distant spread of toxin. TR-TEAEs ≥1% included eyelid ptosis (1.7%), injection site pain (1.1%). Migraine and the Mephisto sign were less common, at 0.9% and 0.6%, respectively (Table 10).
Summary of treatment-emergent serious adverse events open-label phase – safety population.

All patients received 195U during open-label phase.
TESAE, treatment-emergent serious adverse events; U, unit.
Most frequent (≥1%) TR-TEAEs in open-label phase – safety population.
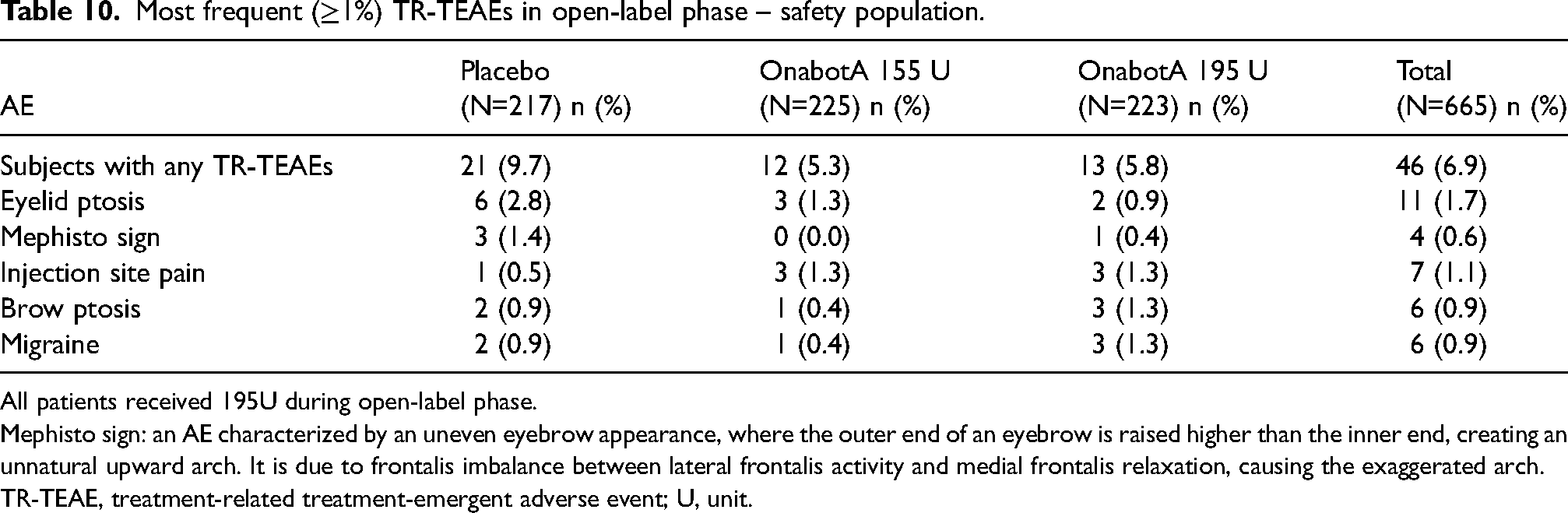
All patients received 195U during open-label phase.
Mephisto sign: an AE characterized by an uneven eyebrow appearance, where the outer end of an eyebrow is raised higher than the inner end, creating an unnatural upward arch. It is due to frontalis imbalance between lateral frontalis activity and medial frontalis relaxation, causing the exaggerated arch.
TR-TEAE, treatment-related treatment-emergent adverse event; U, unit.
Discussion
The purpose of the PRECLUDE trial was to evaluate the efficacy and safety of onabotA as a preventive treatment for EM, which is currently not covered as an approved indication for onabotA. The results of this trial do not support the efficacy of onabotA in EM given that neither onabotA 195 U nor 155 U achieved statistically significant improvements over placebo in the primary or secondary endpoints in the EM population as defined by the trial's enrollment criteria. The trial findings indicate that onabotA was well tolerated, with types and rates of adverse events consistent with previous studies on onabotA in migraine (30,31). The incidences and nature of adverse events aligned with the expected results and were generally mild to moderate in severity, with no new safety signals identified.
In comparison, the results of the PRECLUDE trial in EM differ from those observed in the PREEMPT trials, in which onabotA demonstrated greater and statistically significant efficacy as replicated in two large randomized controlled trials for CM. In both PREEMPT 1 and 2, onabotA demonstrated significant and clinically meaningful reductions in the number of headache days, as well as migraine days, compared to placebo, underscoring the efficacy of onabotA in a CM population where headaches are more frequent (31). The discrepancy in results between the PREEMPT CM trials and the PRECLUDE EM trial highlights the complexity and variability in migraine pathophysiology between CM and EM and the challenges in managing the different subtypes of migraine. Below, we explore these complexities and their potential implications for the trial findings.
Disease state: Differences between EM and CM biotypes
The diagnostic criteria define CM primarily based on headache day frequency: CM is defined by having ≥15 headache days per month, with at least eight of those being migraine days. In contrast, EM is not defined in ICHD-3 but is generally viewed as the migraine group that involves fewer than 15 headache days per month. Patients with EM may progress to CM at a rate of about 2.5% each year in the population (36) and at higher rates in clinic-based samples (37). Clinically, there are several differences in EM and CM phenotypes evaluated by epidemiological studies. CM patients experience two-fold longer headache attack duration than those with EM (24.1 vs. 12.8 hours untreated, p < .0001) and greater pain severity during attacks (8). CM patients also report higher rates of comorbidities such as depression, anxiety, chronic pain and cardiovascular events (38,39).
Clinical and epidemiological differences reported in EM and CM are supported by structural and functional brain alteration differences between the two patient populations. Structural changes between EM and CM patients were noted in a functional magnetic resonance imaging study where brain cortical thickness, cortical surface area and regional volumes were evaluated (40). Neurophysiological and functional imaging studies have reported persistent excitability during migraine attacks in CM compared to intermittent excitability in EM, increased activity in pons and cortical areas during interictal period in CM compared to EM and excessive cortical hyperexcitability in CM (41,42). Furthermore, there are greater CGRP, vasoactive intestinal peptide and amylin levels in CM patients compared to EM indicating differences in molecular activity involved in migraine pain signaling (43). These observations are consistent with not only anatomical and morphological differences across the spectrum of EM and CM, but also migraine-subtype specific variable interconnectivity across the multimodal network of the peripheral and central nervous system.
Regarding differences in treatment response between CM and EM, previous studies with other migraine treatments have shown variations in efficacy between CM and EM populations. Although topiramate is approved broadly for migraine prevention, encompassing both EM and CM, the literature reports suggest that response to treatment may be variable in the EM population compared to CM. Of four studies in EM, two did not show significance for a reduction in migraine frequency compared to placebo for topiramate 200 mg dose, whereas two studies did show significance (44).
These epidemiological, pathophysiological and treatment response findings highlight the clinical and biological differences between EM and CM, emphasizing the importance of differentiating between CM and EM biotypes when evaluating and developing therapeutic interventions.
Disease state – frequency is an insufficient marker
Although there are notable differences between EM and CM biotypes, more precise clinical measures and biomarkers to identify the disease state changes have yet to be established. Currently, monthly headache and migraine day frequencies are used to classify CM. In the PREEMPT CM trials, the subgroup of patients with 8–14 migraine days and ≥15 headache days per month at baseline showed significant improvement in reduction of headache days for onabotA treated patients compared to placebo (−2.0 days difference from placebo, p < 0.001). In contrast, the subgroup of patients with 8–14 migraine days and <15 headache days at baseline in the PRECLUDE EM trial did not show significant improvement from placebo (see Supplementary material, Table S3). This finding was replicated across multiple endpoints (50% responder, triptan days, MSQ v2.1 and HIT-6). The differing responses to onabotA between these subgroups which overlap in migraine days but not headache days, suggest that neurophysiological and potentially morphological changes may be occurring around the threshold of 15 headache days that may impact the efficacy of onabotA in these two headache day frequency categories. These differences in treatment response may reflect differences in underlying biology (i.e. the biotype). We also take note of the clinical observation that many patients with a diagnosis of CM who respond to onabotA when their headache day frequency is ≥15 also continue to respond and maintain their headache frequency improvement, even when retreated at a point in time that their HA day frequency is <15 days (45). This is consistent with the above-cited evidence for neurophysiologic changes at play in the process of migraine progression, suggesting that, once established, the CM biotype generally persists. Monthly headache frequency is an imperfect biomarker for differentiating EM from CM and additional markers are needed to better characterize the CM and EM population. The 15 headache day clinically practical definition used to delineate EM and CM requires further refinement in the setting of pathophysiology and treatment response. Consequently, a key practical research question is a fundamental understanding of the mechanisms of differentiated response to pharmacologic interventions, one of the 25 research priorities identified by the migraine headache experts (46).
Disease state: Allodynia, peripheral sensitization and Mechanism of Action of onabotA in migraine
Beyond headache day frequency, one candidate of biological difference is cutaneous allodynia, a clinical marker of central sensitization. Allodynia is a predictor of migraine chronification and is associated with higher frequency of migraine attacks (47,48). The pathophysiology of allodynia and migraine is related to the activation of and sensitization of the trigeminovascular neurons resulting in sensitization of second and third order neurons, which, once established, becomes active, independent and maintains itself in absence of peripheral sensory input (48,49). Epidemiological studies have reported greater severity and higher frequency of cutaneous allodynia in patients with CM compared to those with EM (49–51). This suggests CM patients who have frequent activation of the trigeminovascular pathway due to repeated migraine attacks of long duration experience greater central sensitization than EM patients (48). Although there is evidence for central sensitization and allodynia as clinical markers of central sensitization, peripheral sensitization has not been well characterized and currently there are no specific markers to measure peripheral sensitization. One theoretically plausible marker is the presence of allodynia in the area of spontaneous pain (52), however more understanding is needed. This lack of ability to measure peripheral and to some degree central sensitization makes it difficult to assess if there is a biological link to migraine chronification and/or if differences in levels of sensitization between migraine biotypes impact treatment responsiveness.
However, the PRECLUDE trial findings provide some evidence to support the hypothesis that peripheral sensitization is present in CM, either ictally or interictally, and might be less prominent in EM and this could impact the treatment response to onabotA. OnabotA, when injected near nerve endings in the periphery, inhibits sensory nerve activation, reducing pain signals and preventing sensitization of neurons in the migraine cascade (53,54). A recent preclinical study evaluating onabotA activity in trigeminal ganglion neurons showed that SNAP25 (i.e. synaptosome-associated protein 25) cleavage was enhanced when neurons were treated with an inflammatory soup prior to administration of onabotA compared to a non-inflammatory soup. This is consistent with greater onabotA activity under sensitized conditions (53). Another preclinical study with dorsal root ganglion neurons sensitized with prostaglandin and bradykinin showed greater internalization of onabotA compared to control neurons (55). Considering the mechanism of action of onabotA, it might be hypothesized that, in migraine, onabotA efficacy is dependent on a higher rate of toxin uptake by sensitized peripheral sensory neurons, either ictally or interictally. This sufficient uptake of onabotA in sensory neurons is more likely in the setting of persistent sensitization, which might be greater in CM, where there is a greater frequency of migraine attacks resulting in more time spent in a sensitized state with its molecular associated consequences. Much more research is needed to understand if greater rates of peripheral sensitization occur in CM and how this influences response to onabotA treatment.
Trial population not representative of EM patients in clinic
Several trial constraints may have resulted in a trial population that is less representative or translatable to the complex EM patients typically seen in headache clinics. The trial aimed to evaluate efficacy in an EM population defined as patients experiencing 6–14 migraine days per month and <15 headache days per month. However, migraine patients spontaneously fluctuate in their frequency of headache days per month throughout their course of disease (28,29). This may have excluded some high frequency EM patients who fluctuate between the cut-off of 15 headache days per month and ≥15 headache days per month who may be responsive to onabotA (25).
Patients with complex comorbidities such as temporomandibular joint/temporomandibular disorder, fibromyalgia, craniofacial pain, moderate to severe depression, and other significant psychiatric disorders were excluded from the trial; notably, several of these patient groups are predisposed to allodynia, a marker of sensitization (36,56–58). As noted above, the existence of sensitization associated with these comorbidities may enhance uptake of toxin into peripheral neurons.
Patients were also not permitted the concurrent use of any migraine preventive treatment during both screening and trial period, and those who have failed more than four preventive treatments (two of which have different mechanisms of action) in the past were excluded. Use of oral CGRP antagonists such as rimegepant and atogepant was prohibited during the trial, with ubrogepant specifically prohibited during the double-blind phase but allowed with limited use (no more than eight days per month) during the open-label phase.
Increasing placebo response in migraine trials
Finally, controlling patient expectations in clinical trials remains a challenge, particularly in pain outcomes evaluated for migraine indications. Over the past 30 years, the placebo response has increased in migraine preventive trials and is thus an important factor when interpreting findings (59). Although comparison of results across different trials has limitations, we note that the proportion of patients achieving at least 50% reduction in monthly migraine days for active treatment arms in Phase 3 EM trials for approved anti-CGRP agents (monoclonal antibodies and gepants) was 39.7–62.3% (60–62). The 50% response rate in the PRECLUDE trial was 46.2% for the onabotA 155 U dose and 47.1% for the onabotA 195 U dose, placing it in the range of the anti-CGRP agent active arm outcomes. The corresponding 50% response rate for placebo arms in Phase 3 EM trials was 26.6%-41.5%, all of which were lower than the 50% response rate of 44.9% for the placebo arm in the PRECLUDE trial (60–62).
Placebo response varies by route of administration (oral, injectable and intravenous), with more invasive treatments yielding higher placebo responses (59). OnabotA treatment administration involves multiple injections in the head, face and neck. In PRECLUDE, all patients received 39 injections, which may have limited the ability to show improvement between treatment groups (63). Although speculative, the reputation of onabotA as a well-known treatment for CM globally, coupled with direct-to-consumer marketing across major geographies, may have heightened patients’ overall expectations for improvement, potentially contributing to a higher placebo response.
Future research
Overall, our findings stress the importance of exploring tailored treatment strategies for different migraine biotypes. Characterization of peripheral and central sensitization across the spectrum of migraine is warranted to further understand the migraine disease state and better define the CM biotype and chronification process, focusing on identifying clinical, functional and molecular biomarkers that correlate with CM and complement the current headache day frequency definition. This aligns with the key headache research priorities outlined by international headache experts; specifically, understanding the evolution of migraine, risk factors for chronification and factors that predict improvement are key research priorities for diagnosis and management of migraine. The concept of different endophenotypes and pathophysiologies within migraine is recognized by the headache experts who noted the need to connect biomarkers to clinical phenotypes and treatment response (46,64). Future studies on the mechanism of action of onabotA in migraine would further expand upon the current understanding of migraine pathophysiology and treatment pathways. As patient expectations remain a challenge, novel approaches to trial design and identification of patient populations representative of real-world migraine patients are needed to ensure the applicability of the results in clinic. While onabotA has been established as an effective preventive treatment for CM (30,31), the current trial underscores the necessity for continued research to identify and develop effective preventive treatments specifically for EM and high frequency EM. Given the substantial burden of EM and its impact on quality of life, ongoing efforts to understand and mitigate EM remain a critical area of interest.
Conclusions
In conclusion, the PRECLUDE trial confirmed the safety profile of onabotA in EM but did not demonstrate significant efficacy of onabotA with respect to reducing migraine days in participants with EM compared to placebo in the trial population enrolled. This does not necessarily rule out the potential benefit of onabotA for patients with EM characteristics, such as patients who experience 8–14 migraine days per month who fluctuate between <15 and ≥15 headache days. Future research to explore the biological differences between EM and CM may help refine treatment approaches and guide the development of new therapies.
Article highlights
The PRECLUDE trial confirmed the safety profile of onabotA in EM but did not demonstrate significant efficacy of onabotA in reducing migraine days in participants with EM compared with placebo in the trial population enrolled.
The discrepancy in results between the PREEMPT CM trials and the PRECLUDE EM trial highlights the complexity and variability in migraine pathophysiology between CM and EM and the challenges in managing the different subtypes of migraine.
Future research to explore the biological differences between EM and CM may help refine treatment approaches and guide the development of new therapies.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251370769 - Supplemental material for OnabotulinumtoxinA for the preventive treatment of episodic migraine: Results from the phase 3, multicenter randomized, double-blind, placebo-controlled phase of the PRECLUDE trial
Supplemental material, sj-docx-1-cep-10.1177_03331024251370769 for OnabotulinumtoxinA for the preventive treatment of episodic migraine: Results from the phase 3, multicenter randomized, double-blind, placebo-controlled phase of the PRECLUDE trial by Patricia Pozo-Rosich, Andrew M. Blumenfeld, Richard B. Lipton, Ronald E. DeGryse, Beibei Li, Aubrey M. Adams, Thu Nguyen, Lynn James and Mitchell F. Brin in Cephalalgia
Supplemental Material
sj-docx-2-cep-10.1177_03331024251370769 - Supplemental material for OnabotulinumtoxinA for the preventive treatment of episodic migraine: Results from the phase 3, multicenter randomized, double-blind, placebo-controlled phase of the PRECLUDE trial
Supplemental material, sj-docx-2-cep-10.1177_03331024251370769 for OnabotulinumtoxinA for the preventive treatment of episodic migraine: Results from the phase 3, multicenter randomized, double-blind, placebo-controlled phase of the PRECLUDE trial by Patricia Pozo-Rosich, Andrew M. Blumenfeld, Richard B. Lipton, Ronald E. DeGryse, Beibei Li, Aubrey M. Adams, Thu Nguyen, Lynn James and Mitchell F. Brin in Cephalalgia
Supplemental Material
sj-docx-3-cep-10.1177_03331024251370769 - Supplemental material for OnabotulinumtoxinA for the preventive treatment of episodic migraine: Results from the phase 3, multicenter randomized, double-blind, placebo-controlled phase of the PRECLUDE trial
Supplemental material, sj-docx-3-cep-10.1177_03331024251370769 for OnabotulinumtoxinA for the preventive treatment of episodic migraine: Results from the phase 3, multicenter randomized, double-blind, placebo-controlled phase of the PRECLUDE trial by Patricia Pozo-Rosich, Andrew M. Blumenfeld, Richard B. Lipton, Ronald E. DeGryse, Beibei Li, Aubrey M. Adams, Thu Nguyen, Lynn James and Mitchell F. Brin in Cephalalgia
Footnotes
Acknowledgements
AbbVie and the authors thank all of the trial investigators and the patients who participated in this clinical trial. Amutha Selvamani, PhD, of AbbVie Inc. provided medical writing assistance for the development of this publication.
Author contributions
All authors met the ICMJE authorship criteria. Neither honoraria nor payments were made for authorship.
Data availability
AbbVie is committed to responsible data sharing regarding the clinical trials we sponsor. This includes access to anonymized, individual and trial-level data (analysis data sets), as well as other information (e.g. protocols, clinical trial reports or analysis plans), as long as the trials are not part of an ongoing or planned regulatory submission. This includes requests for clinical trial data for unlicensed products and indications. These clinical trial data can be requested by any qualified researchers who engage in rigorous, independent, scientific research, and will be provided following review and approval of a research proposal, Statistical Analysis Plan (SAP) and execution of a Data Sharing Agreement (DSA). Data requests can be submitted at any time after approval in the USA and Europe and/or after acceptance of this manuscript for publication. The data will be accessible for 12 months, with possible extensions considered. For more information on the process or to submit a request, visit the following link: ![]() then select “Home”.
then select “Home”.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Financial arrangements of the authors with companies whose products may be related to the present report are listed below, as declared by the authors.
Patricia Pozo-Rosich, MD, PhD, reports personal fees for consulting from AbbVie, Amgen, Dr Reddy's, Eli Lilly, Lundbeck, Medscape, Novartis, Organon, Pfizer, and Teva in the last three years. She has received personal fees for speaking from AbbVie, Amgen, Dr Reddy's, Eli Lilly, Lundbeck, Medscape, Novartis, Organon, Pfizer and Teva, and has had grants paid to her research group from AbbVie, AGAUR, EraNet Neuron, FEDER RIS3CAT, Instituto Investigación Carlos III, Novartis and Teva. She is a member of the Scientific Advisory Board for Lilly Foundation Spain.
Andrew M. Blumenfeld, MD, within the past 12 months, has served on advisory boards for, consulted for, and/or been a speaker or contributing author for Aeon, Allergan/AbbVie, Alder, Amgen, Axsome, BDSI, Biohaven, Eli Lilly, Impel, Lundbeck, Novartis, Revance, Teva, Theranica and Zosano. He has received grant support from AbbVie and Amgen.
Richard B. Lipton, MD, has received research support from the National Institutes of Health, the FDA, and the National Headache Foundation. He serves as consultant for, advisory board member of, or has received honoraria or research support from AbbVie/Allergan, Amgen, Biohaven, Dr Reddy's Laboratories (Promius), electroCore, Eli Lilly, GlaxoSmithKline, Lundbeck, Merck, Novartis, Teva, Vector and Vedanta Research. He receives royalties from Wolff's Headache, 8th edition (Oxford University Press, 2009) and Informa. He holds stock/options in Axon, Biohaven, and Manistee.
Ronald E. DeGryse, MS, MA, C.Phil, is an employee of AbbVie and may own AbbVie stock.
Beibei Li, PhD, is an employee of AbbVie and may own AbbVie stock.
Aubrey M. Adams, PhD, is an employee of AbbVie and may own AbbVie stock.
Thu Nguyen, PharmD, is an employee of AbbVie and may own AbbVie stock.
Lynn James, MPH, is an employee of AbbVie and may own AbbVie stock.
Mitchell F. Brin, MD, is an employee of AbbVie and may own AbbVie stock.
Ethical statement
The studies were conducted in accordance with the most recent ISPOR (International Society for Pharmacoeconomics and Outcomes Research) Good Practices for Outcomes Research for guidance in the development of the protocol. This is a retrospective study with anonymized data (IBM MarketScan and Optum databases are HIPAA compliant de-identified claims data)
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: AbbVie (Allergan prior to the acquisition) funded this study and participated in the study design, research, analysis, data collection, interpretation of data, reviewing and approval of the publication. All authors had access to relevant data and participated in the drafting, review, and approval of this publication. No honoraria or payments were made for authorship.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
