Abstract
Aim
Long-term data for oral calcitonin gene-related peptide receptor antagonist, atogepant, in episodic migraine (EM) has been reported. This is the first report on one-year outcomes in participants with chronic migraine (CM) and in the EM population with prior preventive treatment failures. Here, we report the long-term safety, tolerability, efficacy and functional outcomes of one-year preventive treatment of EM or CM with atogepant.
Methods
This is an interim analysis of an ongoing, open-label, multicenter, 156-week, safety extension study that enrolled completers from phase 3 PROGRESS and ELEVATE trials. The participants completing week 52 or early termination were evaluated. Eligible adults with at least a one-year history of migraine, with either CM (PROGRESS) or EM who previously had inadequate response to two to four classes of conventional oral preventive treatments (ELEVATE). All participants received atogepant 60 mg once daily. The primary outcome was safety and tolerability of atogepant. Efficacy and functional outcomes were prespecified exploratory analyses.
Results
Of 596 participants, 595 (PROGRESS, n = 325; ELEVATE, n = 270) were treated and included in the safety population and 524 (PROGRESS, n = 284; ELEVATE, n = 240) were included in the modified intent-to-treat population. In this interim analysis, mean duration of atogepant exposure was 496.5 days. Treatment-emergent adverse events (TEAEs) occurred in 79.0% of participants; most were mild/moderate and not related to atogepant. Common TEAEs (≥5%) included COVID-19 (28.7%), nasopharyngitis (10.9%) and constipation (8.2%). TEAEs leading to discontinuation occurred in 5.9% of participants. One death attributed to asphyxia by housefire was observed. Other serious TEAEs occurred in 5.5% of participants and none were related to atogepant. Alanine aminotransferase and/or aspartate aminotransferase ≥3× upper limit of normal occurred in two participants; neither met Hy's law criteria. Improvements in efficacy and functional outcomes from lead-in study baseline were observed at weeks 13–16 in this open-label study and were consistent through 48 and 52 weeks, respectively.
Conclusions
Overall safety results were consistent with the known safety profile of atogepant and the drug was well-tolerated over the course of the study. No new safety signals were identified. Improvements in efficacy and functional outcomes were consistent during the study.
Trial Registration
ClinicalTrials.gov identifier: NCT04686136.
This is a visual representation of the abstract.
Introduction
Migraine is a highly prevalent, burdensome, chronic neurological disease (1,2) and is the second leading cause of global disability, impacting 1.16 billion individuals worldwide (1–3). People with migraine experience limitations in physical functioning, low health-related quality of life, stigma, and reduced emotional and social well-being (3–7). Due to the chronic nature of migraine, long-term efficacy and safety are important treatment attributes for the optimal management of migraine.
Migraine is categorized into episodic migraine (EM) (<15 monthly headache days, (MHD)) and chronic migraine (CM) (≥15 MHD, ≥8 monthly migraine days (MMD)) subtypes (8,9). Migraine management includes disease education, lifestyle management, acute treatments, aimed at halting the headache after onset, and/or preventive treatments, aimed at preventing migraine attack (9–11). According to the American Headache Society Consensus Guidelines, preventive treatments should be suggested for people with migraine with severe disability or with frequent attacks of four or more headache days per month (9). Both European Headache Federation and the American Headache Society recommend mechanism-based calcitonin gene-related peptide (CGRP)-targeting therapies as a first-line approach for the preventive treatment of migraine (9,10,12). Available preventive migraine treatments targeting CGRP include CGRP monoclonal antibodies (mAbs) and oral CGRP receptor antagonists (i.e. gepants). Pivotal phase 3, randomized trials evaluating the use of CGRP treatments for the preventive treatment of migraine are generally 12-week trials (13–16). Migraine, even in its episodic form, is a chronic disease, and most people require long-term preventive treatment (17). Despite reports of long-term outcomes mostly in the EM population (18,19), there remains a need for long-term trials evaluating safety, efficacy and functional outcomes of preventive treatments in CM and EM with prior preventive treatment failure.
Atogepant is an oral CGRP receptor antagonist developed for the preventive treatment of migraine in adults (20). The efficacy and safety of atogepant 60 mg once daily (QD) for the preventive treatment of CM was demonstrated in the phase 3 PROGRESS trial (21). The efficacy and safety of atogepant 60 mg QD for the preventive treatment of EM and for the preventive treatment of EM in participants who previously had an inadequate response to two to four classes of conventional oral preventive medications were demonstrated in the phase 3 ADVANCE and ELEVATE trials, respectively (16,22). The long-term safety and efficacy of atogepant for the preventive treatment of EM has been reported in a 52-week, open-label, phase 3 trial and the long-term safety was reported in a 40-week open-label extension (OLE) trial (19,23).
Here we report the first long-term safety, efficacy and functional outcomes of preventive atogepant 60 mg QD treatment in the CM population and EM in participants with inadequate response to two to four classes of conventional oral preventive treatments, from an interim analysis at one year of the ongoing, 156-week, safety extension study.
Methods
Study design
This is an ongoing, phase 3, open-label, multicenter, long-term, safety extension study. The trial includes a 156-week open-label treatment period and a four-week follow-up period (see supplementary material, Figure S1). An interim analysis (cutoff date 14 September 2023) was conducted to evaluate long-term safety, efficacy and functional outcomes of atogepant 60 mg QD in participants who completed the week-52 efficacy data collection portion or those who terminated earlier in the trial. Early termination was defined as a participant who signed informed consent form then ceased participation in the study, regardless of circumstances (e.g. voluntary withdrawal, adverse event (AE), lack of efficacy, lost to follow-up, non-compliance with atogepant, pregnancy, protocol deviation, site terminated by sponsor, study terminated by sponsor), before completion of the study. For all AEs, the investigator provided an assessment of the severity, mild, moderate and severe. Mild AE is the one usually transient and may require only minimal treatment or therapeutic intervention, and the event does not generally interfere with the usual activities of daily living. Moderate AE is described as the AE usually alleviated with additional specific therapeutic intervention. The event interferes with the usual activities of daily living, causing discomfort but poses no significant or permanent risk of harm to the research participant. The AE that interrupts usual activities of daily living, or significantly affects clinical status, or may require intensive therapeutic intervention, is described as severe AE. In this trial, strong/moderate CYP3A4 inducers (e.g. barbiturates, systemic (oral/intravenous) glucocorticoids, nevirapine, efavirenz, pioglitazone) and strong CYP3A4 inhibitors (e.g. systemic (oral/intravenous) itraconazole, ketoconazole, clarithromycin, telithromycin, nefazodone and HIV protease inhibitors) were prohibited 30 days before visit 1 and throughout the study for all participants. All eligible participants received the study intervention, oral atogepant 60 mg QD. Some participants had a gap between the lead-in study and OLE, during which they were off atogepant treatment.
Participants
Enrolled participants included lead-in trial completers from either ELEVATE (NCT04740827) or PROGRESS (NCT03855137) trials (21,22). Lead-in trial completers were those who completed the double-blind treatment period (visit 7) or the follow-up period (visit 8), if applicable, without significant protocol deviations (e.g. non-compliance to protocol-required procedures) and who did not experience an AE that may indicate an unacceptable safety risk. In ELEVATE, participants had EM with 4–14 MMDs and <15 MHDs at baseline and had a history of inadequate response to two to four classes of conventional oral preventive treatment. In PROGRESS, participants had CM with ≥15 MHDs and ≥8 MMDs at baseline. PROGRESS allowed enrollment of participants with acute medication overuse at baseline, whereas ELEVATE did not. Participants were excluded if they had a clinically abnormal electrocardiogram (ECG), had any clinically significant diseases (e.g. hypertension, hematologic, endocrine, cardiovascular, pulmonary, renal, hepatic, gastrointestinal, neurologic), were pregnant/planning to become pregnant during the trial, or currently lactating. Details can be found in the supplementary material (Methods S1).
The safety analyses included all participants who were treated in the OLE at the time of cutoff, regardless of their duration of participation. Safety assessments included clinical laboratory evaluations, vital signs, ECG and Columbia-suicide severity rating scale (C-SSRS) scores, as well as incidence of AEs, serious AEs (SAEs) and AEs leading to discontinuation. Treatment-emergent AEs (TEAEs) were defined as AEs with a recorded onset date on or after the date of the first dose of open-label study treatment. AEs occurring more than 30 days after the last dose of open-label study treatment were not counted as a TEAE.
Assessments
Efficacy assessments were recorded daily by participants using an eDiary. The efficacy outcomes included change from lead-in study baseline in MMDs, MHDs and monthly acute medication use days, and the proportion of participants who achieved either a ≥50%, ≥75% or 100% reduction in MMDs from lead-in study baseline. Efficacy assessments were collected at weeks 13–16, 29–32 and 45–48.
Detailed methods on Activity Impairment in Migraine-Diary (AIM-D) and Migraine-Specific Quality of Life Questionnaire v2.1 (MSQ v2.1) have been previously published (24). Analysis of change from lead-in study baseline in mean monthly performance of daily activities (PDA) and physical impairment (PI) domain scores of AIM-D were evaluated at weeks 13–16, 29–32 and 45–48. Details of additional functional measures (MSQv2.1 role function-restrictive (RFR), role function-preventive (RFP) and emotional function (EF) domain scores) can be found in the supplementary material (Methods S1).
Change from lead-in study baseline in RFR, RFP and EF domain scores were evaluated at weeks 12, 20, 28, 36, 44 and 52.
Statistical analysis
This study enrolled completers from PROGRESS and ELEVATE trials (21,22). No separate sample size calculation was performed for this extension study. The safety population included all participants who received one or more dose of study intervention in the OLE period. The modified intent-to-treat (mITT) population included participants who received one or more dose of the study intervention and had one or more evaluable post-baseline four-week period of eDiary data in the OLE period. The four-week diary efficacy endpoints are defined in a prorated fashion (i.e. months with 14 days or more of completed eDiary data were prorated to 28-day equivalents). Baseline MMDs were calculated during the 28-day baseline period in lead-in studies with ≥20 days of completed eDiary data required to be evaluable. If <28 days of completed eDiary days were reported, baseline MMDs and similar counting variables were prorated to standardize the count to a 28-day equivalent assuming that the rate of migraine was the same on days with eDiary data and on days with missing data. If any post-baseline four-week interval had ≥14 days but <28 days of completed eDiary data, a similar proration method was used. If <14 days of eDiary data were available, the observed count for that four-week interval was set to missing.
Safety analyses were reported as descriptive statistics using the safety population. Continuous variables were summarized by the number of participants, mean ± SD, and median, minimum and maximum values. Categorical variables were summarized by the number and percentage of participants.
Both efficacy and functional outcome analyses were reported using the mITT population. The baseline values for efficacy assessment defined for the lead-in study (ELEVATE or PROGRESS trials) were used as the baseline in this extension study. MMDs, MHDs, acute medication use days and functional outcomes were evaluated as continuous variables, whereas the responder rates are evaluated as binary variables. The efficacy outcomes (MMDs, MHDs, acute medication use days) were evaluated using a mixed-effects model for repeated measures for the change from baseline. The model included derived visit as a categorical fixed effect, and the baseline score and baseline-by-visit interaction as covariates. The restricted maximum likelihood method was used. An unstructured covariance matrix was used to model the covariance of within-participant repeated measurements. If the model did not converge, simplified covariance structures were used to fit the model in the order: (1) Toeplitz and (2) compound symmetry. The mean change from baseline was estimated by least square mean along with the corresponding standard error and 95% confidence interval (CI). The analysis was performed based on all post-baseline values using only the observed cases without imputation of missing values. Descriptive statistics were reported by visits for the functional outcomes (AIM-D and MSQ v2.1 domain scores). The ≥50%, ≥75% and 100% responder rates during each four-week interval were reported. Sensitivity analysis was conducted to evaluate 50% or greater responder rate in visit 7 rollovers. The efficacy and functional outcomes were not powered for statistical comparisons in this interim analysis. Analyses were performed using SAS, version 9.4 or newer (SAS Institute, Cary, NC, USA).
Results
Participants
Of 596 participants (ELEVATE, n = 271; PROGRESS, n = 325), 595 (ELEVATE, n = 270; PROGRESS, n = 325) were treated with atogepant 60 mg QD and included in the safety population; 524 (ELEVATE, n = 240; PROGRESS, n = 284) were included in the mITT population (Figure 1). Baseline demographics are summarized in Table 1.

Study flow diagram. Total study population includes all lead-in completers. CM = chronic migraine; EM = episodic migraine; OL = open-label; mITT = modified intent-to-treat.
Baseline demographics (safety population).
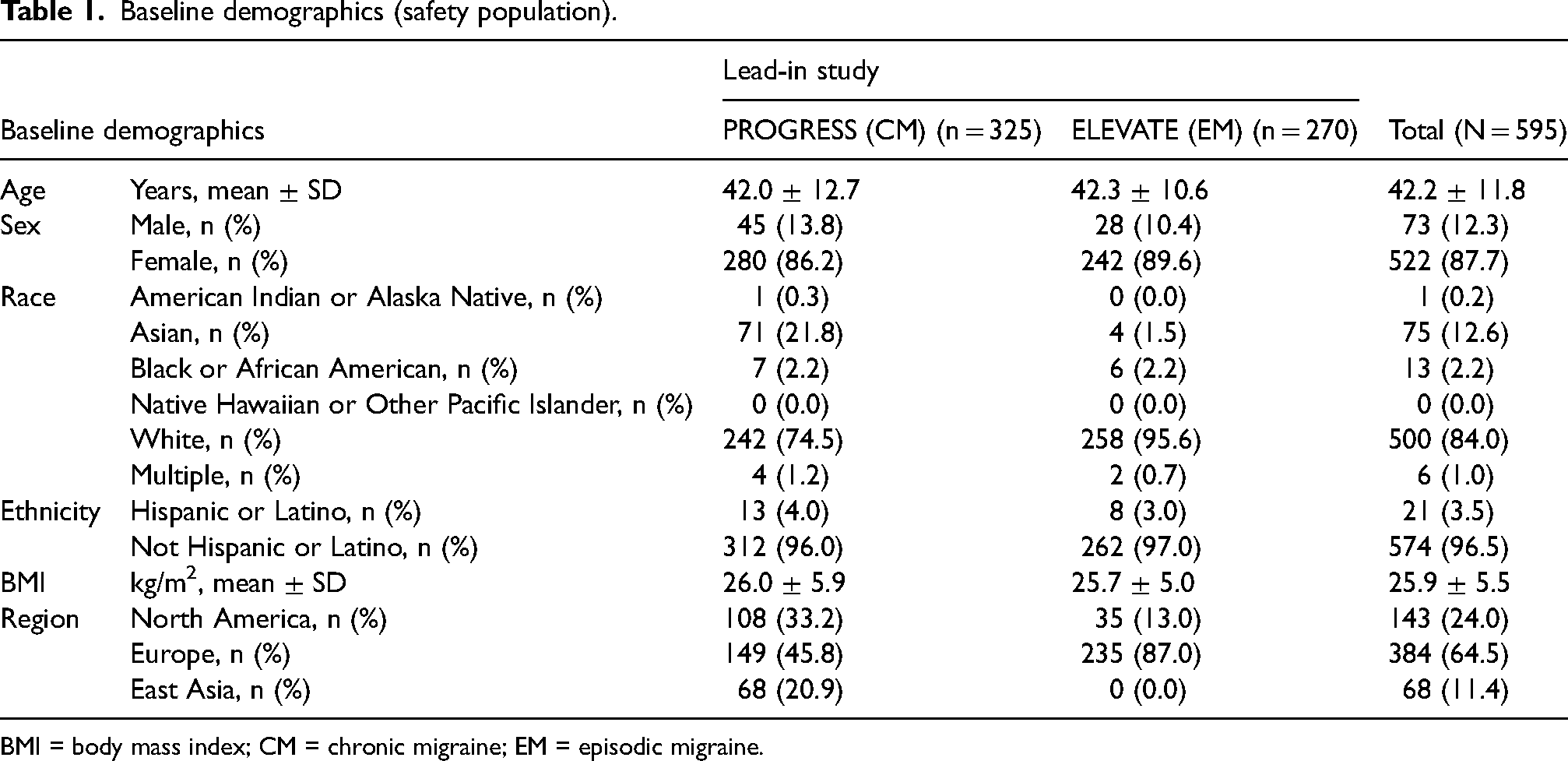
BMI = body mass index; CM = chronic migraine; EM = episodic migraine.
In total, 26.4% (157/595) participants receiving study intervention discontinued at the time of this interim analysis. Study treatment discontinuation rates were 19% (51/270) and 33% (106/325) in subjects originating from lead-in studies ELEVATE and PROGRESS, respectively. Most common reasons for discontinuation were withdrawal by subjects (9.7%, 58/595) followed by AEs (7.1%, 42/595). Discontinuations due to lack of efficacy occurred in 2% (12/595) of participants (Figure 1). Mean ± SD duration of atogepant exposure was 496.5 ± 224.7 days.
Safety and tolerability outcomes
An overview of TEAEs and TEAEs reported in ≥2% of the participants (i.e. common TEAEs) in lead-in PROGRESS, lead-in ELEVATE and total population are listed in Table 2.
Treatment-emergent adverse events (TEAEs) overview and common TEAEs (≥2%) (safety population).

CM = chronic migraine; EM = episodic migraine; SAEs = serious adverse events; TEAEs = treatment-emergent adverse events.
One death attributed to asphyxia by housefire was observed.
In this interim analysis, TEAEs occurred in 79.0% (470/595) of the total population treated with atogepant 60 mg QD; of these, 32.6% (194/595) were mild and 41.8% (249/595) were moderate and 4.5% (27/495) were severe. Common TEAEs occurred in total participants were COVID-19 (28.7%, 171/595), nasopharyngitis (10.9%, 65/595), and constipation (8.2%, 49/595), upper respiratory tract infection (4.5%, 27/595), urinary tract infection (4.5%, 27/595), back pain (4.0%, 24/595) and insomnia (3.9%, 23/595). One death attributed to asphyxia by housefire was reported. Other serious TEAEs occurred in 5.5% (33/595) of participants. The only serious TEAE reported by more than one participant was ovarian cyst, with one participant each from the ELEVATE and PROGRESS lead-in studies. Treatment-related constipation occurred in 7.4% (44/595) of the total study participants (see supplementary material, Table S1). None of the serious TEAEs were reported as treatment-related as assessed by the investigator. TEAEs leading to study discontinuation occurred in 5.9% (35/595) of the safety population (Table 2).
There were no clinically relevant lab parameters or vital sign changes observed in the interim analysis. In people with EM or CM, atogepant was associated with consistent weight loss when used as a preventive treatment (25). In this interim analysis, post-baseline potentially clinically significant (PCS) (≥7%) weight loss at any time was reported in 32.6% (193/592) of participants, whereas 10.6% (63/592) reported ≥7% anytime weight gain (see supplementary material, Table S2). Two participants had alanine aminotransferase (ALT)/aspartate aminotransferase (AST) ≥3× upper limit of normal. An external Hepatic Event Adjudication Committee determined one case as possibly related to atogepant (i.e. the evidence for atogepant causing the injury was equivocal but present) and one case as unlikely to be related to atogepant (i.e. there is evidence that an etiological factor other than the drug caused the injury); confounding factors were noted in both cases. No participant met criteria for Hy's law (see supplementary material, Table S3).
Per the C-SSRS assessment, no participant showed any suicidal ideation with intent, with or without a plan, or any suicidal behaviors.
Efficacy outcomes
Lead-in study baseline MMDs were 14.5 days (PROGRESS, 19.2; ELEVATE, 9.1). The least squares mean change from lead-in study baseline in MMDs at weeks 13–16 was −8.5 (95% CI = −9.0 to −8.0) (PROGRESS, −10.9 (95% CI = −11.7 to −10.1); ELEVATE −5.5 (95% CI = −6.0 to −5.0)) and this reduction was consistent through 48 weeks (Figure 2a).

Change from lead-in study baseline in mean (a) monthly migraine days, (b) monthly headache days and (c) monthly acute medication use days at weeks 13–16, 29–32 and 45–48 (modified intention-to-treat population). Error bars represent the standard error (SE).
Lead-in study baseline MHDs were 16.1 days (PROGRESS, 21.4; ELEVATE, 9.9). The least squares mean change from lead-in study baseline in MHDs at weeks 13–16 was −8.5 (95% CI = −9.0 to −8.0) (PROGRESS, −11.2 (95% CI = −12.0 to −10.3); ELEVATE −5.4 (95% CI = −5.9 to −4.8)) and this reduction was consistent through 48 weeks (Figure 2b).
Lead-in study baseline monthly acute medication use days were 11.9 days (PROGRESS, 15.6; ELEVATE, 7.5). The least squares mean change from lead-in study baseline in monthly acute medication use days at weeks 13–16 was −7.0 (95% CI = −7.4 to −6.5) (PROGRESS, −9.3 (95% CI = −9.9 to −8.5); ELEVATE −4.3 (95% CI = −4.8 to −3.9)) and this reduction was consistent through 48 weeks (Figure 2c).
At weeks 13–16, 70.0% of participants (359/513) (PROGRESS 67.0% (187/279); ELEVATE, 73.5% (172/234)) achieved a ≥50% reduction in MMDs from lead-in study baseline and similar response rates were observed up to 48 weeks (Figure 3). At weeks 13–16, 43.9% (225/513) (PROGRESS 39.4% (110/279); ELEVATE, 49.1% (115/234)) and 18.1% (93/513) (PROGRESS 12.9% (36/279); ELEVATE, 24.4% (57/234)) of participants achieved a ≥75% and 100% reduction in MMDs from lead-in baseline, respectively, which was maintained through week 48. Evaluable total population was 513 at weeks 13–16, 432 at weeks 29–32, and 419 at weeks 45–48 due to expected attrition rates over the course of a 52-week study.

Responder rate: proportions of participants achieving ≥50%, ≥75% and 100% reduction in monthly migraine days (MMDs) at weeks 13–16, 29–32 and 45–48 (modified intention-to-treat population). CM = chronic migraine; EM = episodic migraine, n, number of responders who responded within a specific category; N1, observed data at baseline and at the timepoint.
Similar improvements in efficacy outcomes during the long-term OLE study were observed in study participants on active and placebo treatment in the lead-in studies.
A reduction in all efficacy outcomes in the OLE study were similar for lead-in participants, irrespective if they were originally assigned to the placebo or atogepant treatment group or if they had a gap in atogepant treatment between studies or not. The results from the sensitivity analysis were consistent with the primary results (see supplementary material, Tables S4 and S5).
Functional outcomes
Lead-in study baseline PDA domain score of the AIM-D was 25.6 (PROGRESS, 31.6; ELEVATE, 18.9). The mean ± SD change from lead-in study baseline in PDA domain score at weeks 13–16 was −16.0 (PROGRESS, −19.7 ± 16.0; ELEVATE −12.0 ± 10.3) and this reduction (improvement) was consistent through 48 weeks (Figure 4a).
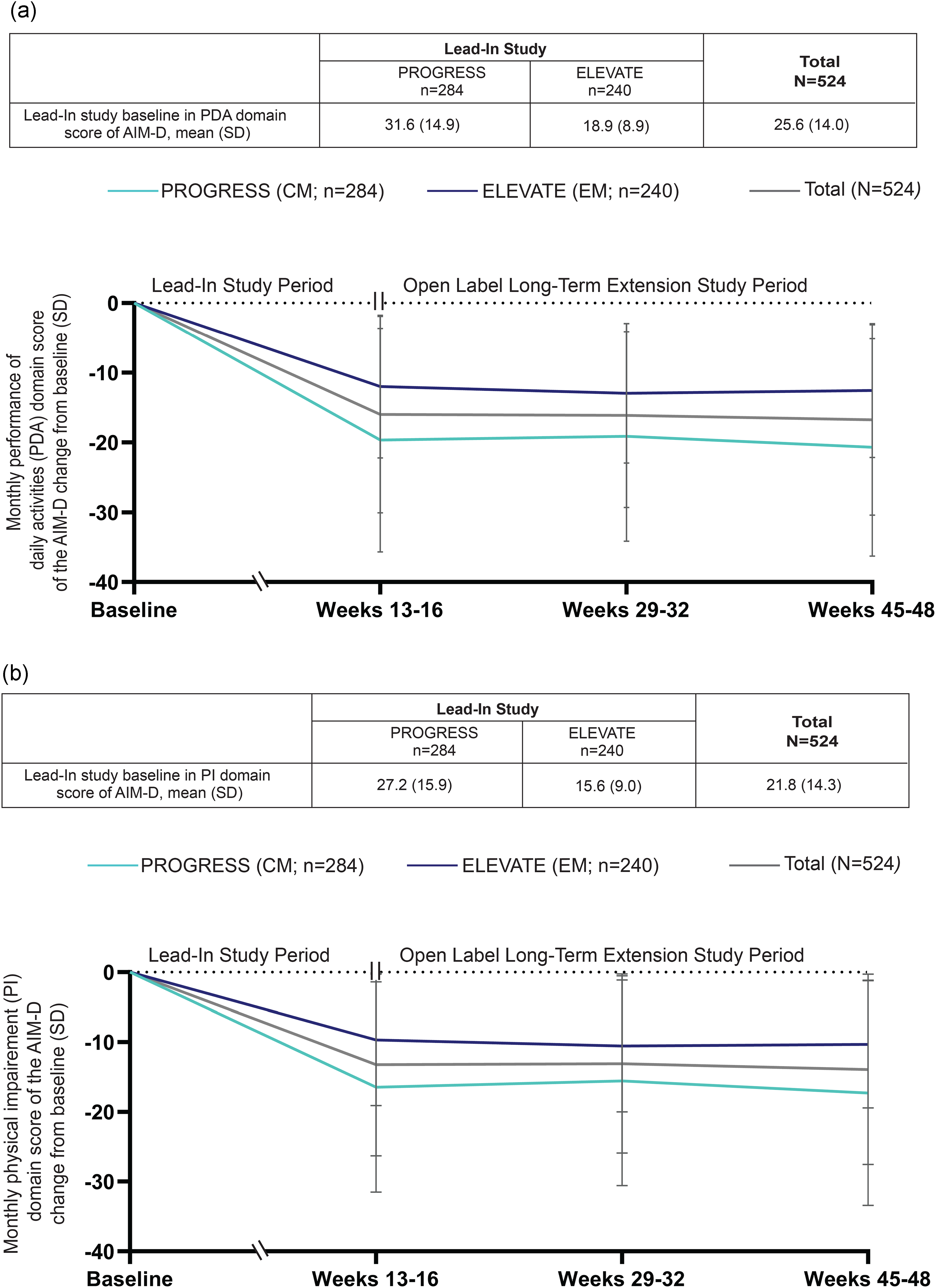
Changes from lead-in study baseline in Activity Impairment in Migraine-Diary (AIM-D) domains: (a) performance of daily activities (PDA) and (b) physical impairment (PI) at weeks 13–16, 29–32 and 45–48 (in modified intention-to-treat population). Error bars represent the SD. CM = chronic migraine; EM = episodic migraine.
Lead-in study baseline PI domain score of the AIM-D was 21.8 (PROGRESS, 27.2; ELEVATE, 15.6). The mean ± SD change from lead-in study baseline in PI domain score at weeks 13–16 was −13.2 (PROGRESS, −9.7 ± 9.4; ELEVATE −16.5 ± 15.1) and this reduction was consistent through 48 weeks (Figure 4b). An improvement in RFR, RFP and EF domain scores of MSQv2.1 was demonstrated at week 12 and was consistent through week 52 (see supplementary material, Figure S2).
Discussion
This is the first report evaluating one-year safety, efficacy and functional outcomes for atogepant 60 mg QD for the preventive treatment of migraine in participants with CM and extends previously observed one-year atogepant data in EM to include a population with multiple oral conventional preventive treatment failures. The results from this interim analysis of the ongoing phase 3 open-label trial demonstrated that atogepant 60 mg QD was safe and well tolerated and the overall safety results were consistent with the known safety profile of atogepant for the preventive treatment of CM and EM (16,22,26,27). No new safety signal was identified.
Long-term evaluation of preventive treatments including novel mechanism-based treatments is critical for the management of migraine. The long-term efficacy and safety of onabotulinumtoxinA for the preventive treatment of CM has been reported in a 108-week open-label long-term prospective study (28). The long-term safety and efficacy for the preventive treatment of CM has been reported for CGRP mAbs such as erenumab (29), eptinezumab (30), galcanezumab (31,32) and fremanezumab (33). Additionally, the long-term safety and efficacy for the preventive treatment of EM with prior preventive treatment failure has been reported for erenumab (34–37), eptinezumab (38), galcanezumab (39) and fremanezumab (40).
Long-term functional outcomes have been reported for all four CGRP mAbs (30,31,33,34,38–40). The results of this interim analysis of a 156-week, long-term, open-label study demonstrating the long-term safety and tolerability of oral atogepant were consistent with these prior studies. Atogepant is the only gepant approved for the preventive treatment of both CM and EM and it is the only gepant that has shown the long-term maintenance of efficacy and functional outcomes in the CM and EM with prior preventive treatment failure populations (21,22,24).
Atogepant effectively reduced acute medication use days in participants with CM with acute medication overuse over 12 weeks (27). Here, atogepant treatment reduced monthly acute medication use days at weeks 13–16, and this reduction remained consistent through 48 weeks. Medication overuse headache is a common complication of CM, with the overuse of acute headache medications being a significant risk factor for its development (41). The observed reduction in acute medication use is an important outcome measure in that it may be associated with a lower risk of developing medication overuse headache in subjects with high frequency EM or CM.
Overall, TEAEs leading to discontinuation of atogepant by two or more participants from any lead-in study were hepatitis E antibody positive, constipation, nausea, increase in ALT or AST, hypertransaminasaemia, insomnia and fatigue. CGRP is reported to accelerate intestinal transit via propulsion and secretion, and there is a potential for related AEs due to the physiologic effects of the CGRP neuropeptide. Constipation is one of the TEAEs that occurred in at least 5% of participants in the OLE study. However, the rates of constipation observed in this study were consistent with rates seen in previous atogepant studies (16,22).
Data from five large clinical trials showed that atogepant was associated with consistent, modest, dose- and duration-dependent weight loss when used as a preventive migraine treatment in the EM and CM populations (25). Post-baseline PCS ≥7% weight loss was experienced by one-third of the participants in the safety population when weight change was analyzed for safety monitoring. The long-term safety and tolerability of atogepant as well as the efficacy and functional outcomes in the EM population have been previously reported (19,38,42).
Together with the results presented in this interim analysis, atogepant has demonstrated long-term safety and tolerability and long-term improvements in efficacy and functional outcomes across the spectrum of migraine (CM, EM and EM with prior preventive treatment failure). The findings in this study demonstrated reductions of MMDs and MHDs and improvement in responder rates. Reductions in PI (based on AIM-D) and improvement in PDA (based on AIM-D) and migraine-specific quality of life (based on MSQ v2.1) were also noted. The full analyses of this study will report three-year safety and efficacy outcomes for atogepant, which will further add to the scientific literature supporting the long-term use of atogepant for the preventive treatment of migraine.
Our study has several limitations. The study populations of the lead-in studies included mostly female and mostly white participants. The trials excluded participants with clinically significant diseases and those using certain medications or other non-pharmacological treatments, and so the results may not be generalized to the full patient population. This is a long-term, open-label study; thus, it was not blinded and did not include a placebo control, and so comparative analyses could not be performed.
Conclusions
In summary, this interim analysis at week 52 of the ongoing, phase 3, 156-week, open-label, long-term safety extension study indicates that atogepant was safe and well tolerated over 52 weeks. The overall safety results were consistent with the known safety profile of atogepant, and no new safety signals were identified. Improvements in efficacy and functional outcomes at weeks 13–16 with atogepant were consistent over the first year of open-label treatment. The results from this interim analysis support the use of once daily oral atogepant as a long-term preventive treatment across the migraine episodic to chronic spectrum. This open-label data may be supported by future real-world studies. The full three-year results of this study will continue to add to the available data on the long-term use of atogepant for the preventive treatment of migraine.
Article highlights
This interim analysis of an ongoing, phase 3, open-label, 156-week, safety extension study evaluated participants with chronic or episodic migraine who completed PROGRESS or ELEVATE trials, respectively.
Safety results were consistent with the known safety profile of atogepant. No new safety signals were identified.
Efficacy and functional outcomes showed improvement from lead-in study baseline at weeks 13–16 and were consistent over 48 weeks and 52 weeks, respectively.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251365206 - Supplemental material for Long-term safety, efficacy and functional outcomes of atogepant for the preventive treatment of migraine
Supplemental material, sj-docx-1-cep-10.1177_03331024251365206 for Long-term safety, efficacy and functional outcomes of atogepant for the preventive treatment of migraine by Sait Ashina, Messoud Ashina, Dagny Holle-Lee, Cristina Tassorelli, Soo-Jin Cho, Molly Yizeng He, Rosa De Abreu Ferreira, Pranav Gandhi, Jonathan H. Smith, Kimberly Pfleeger and Joel M. Trugman in Cephalalgia
Footnotes
Acknowledgments
Statistical analysis of functional outcomes by the lead-in study was performed by Yingyi Liu, PhD, of AbbVie (North Chicago, IL, USA). Medical writing support was provided by Ishita Chatterjee, PhD, and editorial assistance was provided by Angela T. Hadsell, BA, of AbbVie. AbbVie and the authors thank all trial investigators and patients who participated in this clinical trial. We also thank our independent data safety monitoring board members, Benjamin M. Scirica, Paul G. Mathew and Vernon M. Chinchilli, for their contributions.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SA is an associate editor of Cephalalgia, Sage Publications, and has received honoraria for consulting from AbbVie/Allergan, Amgen, Biohaven, Eli Lilly, Impel NeuroPharma, Linpharma, Lundbeck, Novartis, Pfizer, Percept, Satsuma, Supernus, Teva, and Theranica. MA received honoraria for consulting from AbbVie, Astra Zeneca, Eli Lilly, GlaxoSmithKline, Lundbeck, Novartis, Pfizer and Teva Pharmaceuticals; and research grants from Lundbeck Foundation, Novo Nordisk Foundation, Lundbeck and Novartis. DH-L received honoraria for consulting from AbbVie/Allergan, Amgen, Eli Lilly, Novartis, Teva, Lundbeck, Hormosan and Zuellig Pharma. CT is an associate editor of Cephalalgia, Sage Publications. S-JC has received honoraria for speaking or consulting from AbbVie, Chong Kun Dang Holdings Corp, GC Biopharma, Handok-Teva, Lundbeck, Organon Korea, Pfizer, WhanIn Pharm Co., Ltd and Yuyu Pharma, Inc. in the past 24 months. She is the principal investigator in clinical trials sponsored by AbbVie, Ildong Pharmaceutical Co., JW Pharmaceutical, Lundbeck, Pfizer and Teva-Handok in the past 24 months. MYH, RDF, PG, JHS and KP are employees of AbbVie and may hold AbbVie stock or options. JMT is a former employee of AbbVie and may hold AbbVie stock.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: AbbVie funded this study and participated in the study design and conduct of the study, research, analysis, data collection, management, interpretation of data, preparation, reviewing and approval of the publication, as well as the decision to submit the manuscript for publication. All authors had access to relevant data and participated in the drafting, review and approval of this publication. No honoraria or payments were made for authorship.
Data availability
AbbVie is committed to responsible data sharing regarding sponsored clinical trials. This includes access to anonymized, individual and trial-level data (analysis data sets), as well as other information (e.g. protocols, clinical study reports or analysis plans), as long as the trials are not part of an ongoing or planned regulatory submission. This includes requests for clinical trial data for unlicensed products and indications. These clinical trial data can be requested by qualified researchers who engage in rigorous, independent, scientific research and will be provided following review and approval of a research proposal, statistical analysis plan, and execution of a data sharing agreement. Data requests can be submitted at any time after approval in the US and Europe and after acceptance of this manuscript for publication. The data will be accessible for 12 months, with possible extensions considered. For more information on the process or to submit a request, visit: ![]() then select “Home”.
then select “Home”.
Ethical statement
The trial was approved by a local or central Institutional Review Board at each participating institution and conducted in accordance with the International Conference for Harmonisation guidelines, applicable regulations, and the Declaration of Helsinki. Participants provided written informed consent before screening. The study is registered with ClinicalTrials.gov (NCT04686136). The study protocol and statistical analysis plan have been published for the lead-in studies.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
