Abstract
Background
There is a need for standardization of the definition of a migraine day for clinical and research purposes.
Methods
We prospectively compared different definitions of a migraine day with E-diary data of n = 1494 patients with migraine. We used a baseline definition based on migraine characteristics with a duration of ≥4 hours OR triptan intake (independently from its effect) OR (visual) aura lasting 5–60 minutes.
Results
Of all migraine days defined by triptan intake only, 66.2% had a duration <4 hours. Adjusting the headache duration criterion to ≥30 minutes led to a decrease in days defined by triptan intake only and resulted in a 5.4% increase in total migraine days (equals 0.45 migraine day increase in monthly migraine days). These additional migraine days had a median duration of 2.5 hours.
Conclusion
We propose to define a migraine day as follows: 1) (a) headache duration ≥30 minutes; (b) matching ≥2 of four characteristics: unilateral, pulsating, moderate to severe pain, aggravation by or causing avoidance of routine physical activity; and (c) during headache ≥1 of the following: nausea and/or vomiting, photophobia and phonophobia or 2) (visual) aura duration 5–60 minutes or 3) a day with headache for which acute migraine-specific medication is used irrespective of its effect.
Introduction
Determining the number of monthly migraine days (MMD) is essential for clinical practice and clinical trials or other research purposes. According to the current clinical trial guidelines, the number of MMD should be used as primary endpoint (1,2). However, no clear consensus for the definition of a migraine day (MD) exists in the current clinical trial guidelines for migraine, for example about the minimal headache duration or how to take acute migraine-specific treatment intake into account (1,2).
In general, the number of MMD is often determined in retrospect through clinical interviews, paper diaries or commercial applications (apps), mostly by patients self-reporting and deciding whether a day was a migraine day (3–5). However, recall bias substantially reduces the reliability of retrospective reports during clinical consultation (6). Some currently used diaries lack specificity, because patients are asked to self-report migraine days without further symptom specifications. Moreover, these diaries are not time-locked, making it possible to fill out for a longer period of time in retrospect. The Leiden Headache Center uses an electronic diary (E-diary) developed in collaboration with Interactive Studios (The Netherlands): the Patient Journey App (www.patientjourneyapp.com). With this E-diary reliable information on frequency of headache and associated characteristics and features can be obtained, and it has been showen to be a validated and useful tool in both clinical and research settings (6–9). With the emergence of more advanced headache E-diaries aforementioned issues are resolved, but in our opinion there is a need for consensus on the definition of a migraine day based on detailed criteria.
The International Classification of Headache Disorders (ICHD-3) was designed to diagnose migraine and its (sub)types on a patient level, but not on the level of every individual headache day. However, in practice, these criteria are often integrated in headache diaries to be able to define individual migraine days. This seems a very practical approach, but we make note of two important issues. Firstly, is it appropriate that days with use of migraine-specific acute medication are regarded as migraine days? It is likely that patients are instructed by their treating physicians to take acute medication before a complete migraine attack develops, and thus a migraine day may not develop into all required headache characteristics and associated symptoms. To avoid this problem, the ICHD-3 criteria currently state that headache attacks that meet the criteria for probable migraine should be counted as migraine. However, this remains a sub-optimal way to define migraine days as the ICHD-3 is primarily defined to classify patients and not individual days. Secondly, the required minimum headache duration used for defining a migraine day varies from at least 30 minutes to 4 hours in the current clinical trial guidelines, possibly also taking into account that effective non-migraine-specific acute medication may lead to a shorter duration (1,2).
The aim of the current study was to formulate recommendations to standardize the definition of a migraine day for clinical and research purposes by comparing different definitions of migraine days in a large cohort of patients with migraine.
Methods
Study population and headache E-diary
Headache E-diary data were collected between August 2018 and March 2021 from diagnosed patients with migraine at the Leiden Headache Center, aged ≥18 years (10,11). Ethical consideration for this study was obtained from the Medical Ethical committee of the Leiden University Medical Center (LUMC), who identified no ethical concerns. Therefore, participants did not have to provide additional informed consent. All data was analyzed in a fully anonymized setting.
Participants were followed using a headache E-diary over the course of at least three subsequent months (6). Subjects received a daily alert at 8 am and reported on headache, aura and associated symptoms of the previous day (from midnight to midnight). Questions included the presence or absence of headaches, (visual) aura symptoms, use of (migraine-specific and non-migraine-specific) acute medication, and, if applicable, additional questions on the severity (on a scale from 1–3), duration of the headache, and associated symptoms (unilateral location, pulsating quality, pain intensity, increasing with routine physical activities, nausea, vomiting, photophobia, and phonophobia). Also questions on (change of) preventive medication use and general wellbeing were asked for each day. Participants received a reminder at 6 pm if the E-diary was not yet completed that day. After completion or if the E-diary was not completed within 48 hours the response was locked and could not be altered. Patients were considered eligible for this study if they were followed with the E-diary for at least three subsequent months with at least 80% compliance per month.
Definitions of a migraine day
We evaluated four definitions of a migraine day, as listed in Table 1. The first two definitions of a migraine day were based on the typical migraine characteristics, where a distinguishment was made between (A) a headache duration of at least 4 hours and (B) a headache duration of at least 30 minutes. The other two definitions of a migraine day were based on acute medication use, where a distinguishment was made between (C) days with triptan intake and (D) days with effective triptan intake. A triptan was considered effective if the headache was reduced from moderate or severe to mild or no-pain within 2 hours post-dose. We chose triptans as no other acute migraine-specific medications were used at that time in the Netherlands (no ergotamine, no ditans nor gepants).
Definitions of a migraine day.

The frequency of headache and migraine days determined whether a patient was classified as either a patient with chronic migraine (CM) or episodic migraine (EM) according to the ICHD-3. To this end, we used the typical migraine characteristics with a duration of ≥4 hours, in combination with triptan intake (effective or not) as the baseline definition. Also, when a patient experienced (visual) aura symptoms with a duration of between five and 60 minutes the day was considered a migraine day (see Table 1). We defined a headache day as a day with headache of at least one hour. By definition, each migraine day in which a headache was present was also considered a headache day. An aura day without headache was considered a migraine day, but not a headache day.
Patients with migraine were classified as CM according to the ICHD-3 criteria of ≥15 headache days per month, with ≥8 migraine days per month during three subsequent months (12). Because of important differences in clinical impact a further distinction was made between high frequent episodic migraine (HFEM) and low frequent episodic migraine (LFEM) (13–15). Here, non-CM patients who had at least one month with ≥8 migraine days were included in the HFEM group. LFEM was determined as patients who had <8 MMD during all registered months. For each group, medication overuse headache (MOH) patients were defined according to the ICHD-3 criteria of ≥15 headache days per month with ≥15 analgesic days, or ≥10 triptan days, or ≥10 mixed acute medication days per month during three subsequent months.
The number of MMD, number of monthly non-migrainous headache days and number of monthly days with use of acute medication were determined based on the E-diary entries. Missing days were considered headache free and acute medication free. In the E-diary, a month was set to a patient specific fixed period of 28 days (6). Data from months with less than 80% compliance were eliminated from the dataset as the data quality was considered insufficient.
Data analysis and statistics
Baseline characteristics of the entire cohort and subgroups were determined. For each characteristic, the group mean and standard deviation (SD) was calculated from the patients’ means for normally distributed variables, otherwise the median and interquartile range (IQR) was determined. Differences between the groups CM, HFEM and LFEM regarding nominal variables including sex and MOH diagnosis were evaluated using Chi-squared tests. Differences between groups regarding continuous variables such as monthly acute medication days were tested using one-way analysis of variance (ANOVA) tests.
We used the migraine characteristics with a headache duration of ≥4 hours, in combination with triptan intake (effective or not) as the baseline definition for our analyses. We did not include other acute migraine-specific treatments as these were not used by our group of patients (e.g. ergotamines, ditans or gepants). We firstly evaluated the adjustment of the minimum headache duration by comparing the number of migraine days in our data defined by the baseline criteria (headache duration ≥4 hours) to the same criteria with a duration of ≥30 minutes. We investigated the use of analgesics in these additional migraine days and visualized the reported headache durations. Next, we further looked into days defined by triptan intake only (effective or not). We used Venn diagrams to visualize the proportions of migraine days for CM, HFEM and LFEM patients independently. Missing migraine characteristics in days defined by triptan intake solely (effective or not) were analyzed. Statistical differences between the groups were determined using Chi-squared tests. Linear regression analysis was used to determine the association between the number of baseline monthly migraine days and the relative number of migraine days matching the migraine criteria. The number of migraine days with effective triptan intake was compared to the total days with triptan intake. Finally, the effect of adjustment of the duration (≥4 hours or ≥30 minutes) and triptan intake (effective or not) on the number of MMD was evaluated for all four definitions.
All analyses were performed in Python version 3.10.7 using statistical methods implemented in SciPy version 1.9.3. Two-sided p-values ≤.05 were considered statistically significant.
Results
We identified a group of n = 2021 clinically diagnosed patients with migraine, of which 199 (9.8%) were excluded due to less than 80% compliance per month. In order to be able to distinguish EM and CM patients, three consecutive months of E-diary follow-up were needed. Therefore, an additional number of 328 (16.2%) patients were removed from the dataset. This resulted in a representative dataset of n = 1494 patients with migraine. Median follow-up time was 174 (IQR 84–390) days with a mean ± SD of 6.91 ± 4.9 migraine days/month (MMD). Based on the E-diary data, patients were classified as either CM (18.9%, n = 283), HFEM (48.9%, n = 731) or LFEM (32.1%, n = 480) (Table 2). From the total patient population 5.6% were classified as MOH patients (n = 83). Baseline characteristics for the groups are presented in Table 2. Patients with CM had a mean ± SD of 13.7 ± 5.5 MMD, for HFEM and LFEM this was 6.9 ± 2.7 and 2.9 ± 1.6 MMD respectively.
Baseline characteristics of study population.

a. Months are defined as periods of 28 days.
b. Headache days also include migraine days.
CM, Chronic migraine is defined as ≥3 consecutive months with ≥15 headache days per month with ≥8 monthly migraine days (MMD). HFEM, High frequent episodic migraine is defined as ≥1 month with ≥8 MMD. LFEM, Low frequent episodic migraine is defined as <8 MMD. MOH, Medication Overuse Headache is defined as ≥3 consecutive months with ≥15 headache days per month with either ≥15 days with use of analgesics/NSAIDS, or ≥10 triptan days, or ≥10 combined acute medication days (12).
There was a difference in monthly acute medication days with a mean ± SD of 6.6 ± 4.6 for CM patients vs 6.2 ± 3.2 and 4.7 ± 3.1 days/month for HFEM and LFEM patients respectively (p < 0.001). Triptans were least often used by LFEM patients with a mean ± SD of 1.5 ± 1.5 days/month compared to 3.8 ± 2.6 and 3.6 ± 3.4 days/month for HFEM and CM respectively (p < 0.001).
Baseline definition of migraine days
Using migraine characteristics with a duration of ≥4 hours led to a total sum of 91,407 migraine days, on which a triptan was used in 34.0% (n = 31,071 days) of these days. Adding days defined by triptan intake alone (n = 15,023 days) led to a total sum of baseline migraine days of n = 106,430 days (Figure 1). Patients with migraine with aura (MA) had a total of n = 10,171 migraine days with visual aura symptoms lasting five to 60 minutes. Of these migraine days with aura, 8.5% (n = 860) were days with aura without a headache present.
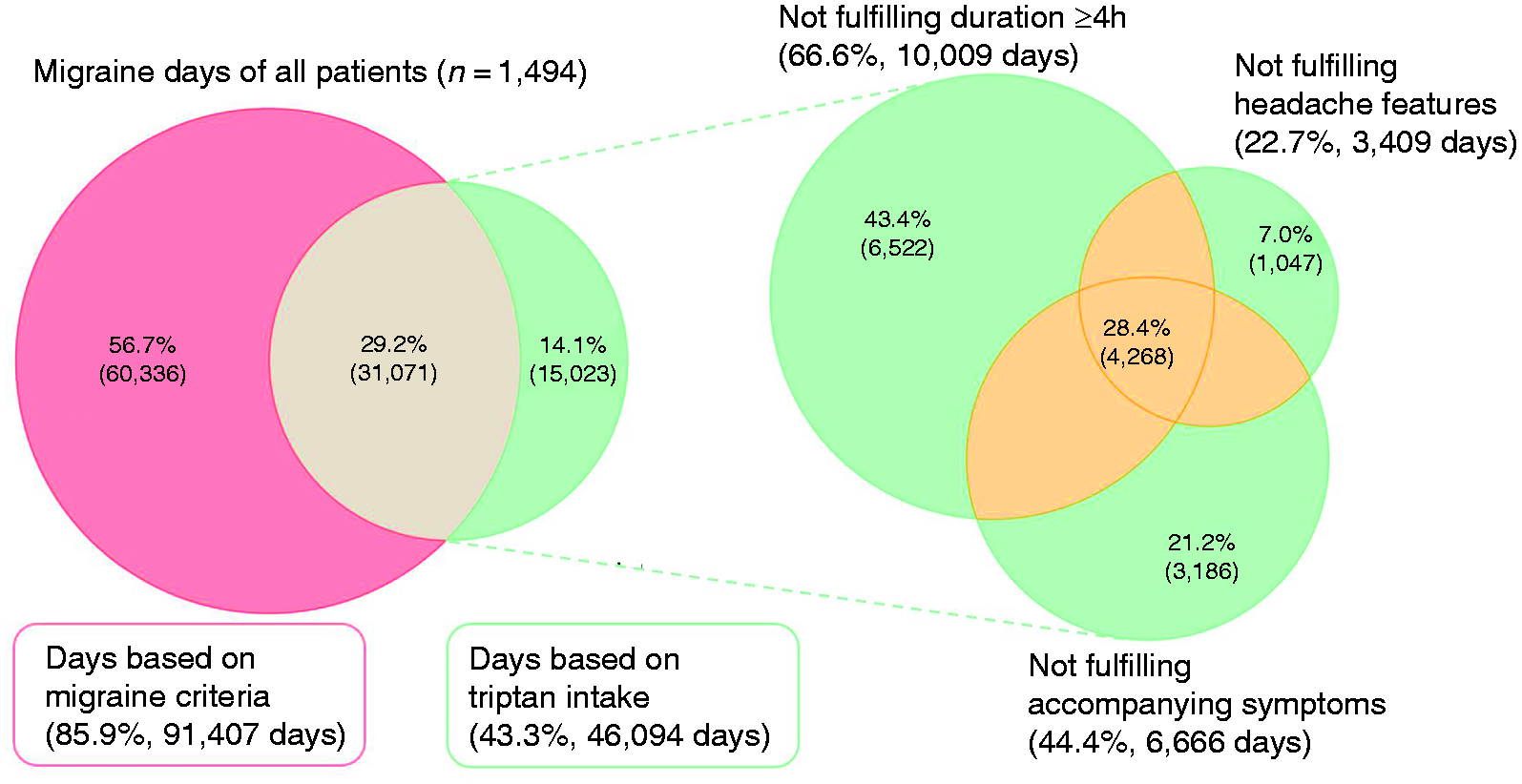
Proportion of days based on migraine characteristics with a duration of ≥4 hours and/or triptan intake (irrespective of its effect). Percentages are calculated based on the total number of migraine days as defined by the baseline criteria for all patients in the dataset (n = 106,430 days). The righthand-side Venn diagrams show the missing migraine characteristics on days defined by triptan intake only.
Evaluating missing criteria in days defined by triptan intake alone
Days defined by triptan intake only missed ≥1 of the migraine characteristics, which is set out on the right side of Figure 1. In 66.6% (n = 10,009) of these days the headache did not fulfill the duration of ≥4 hours. In 22.7% (n = 3409) the headache did not meet at least two of the four following headache features: unilateral; pulsating; moderate to severe pain; aggravation by activity. In 44.4% (n = 6666) no accompanying symptoms such as nausea/vomiting or photophobia and phonophobia were present. Furthermore, 28.4% (n = 4268) of all days defined by triptan intake alone matched less than two migraine characteristics, and 5.3% (n = 793) missed all three characteristics (Figure 1, overlapping part).
Headache duration was the only missing criterion in 43.4% of days (Figure 1). Of these days, 97.7% still had a headache duration of ≥30 minutes. Adjusting the minimum headache duration to 30 minutes increased the total number of migraine days by 5.4% (n = 5720 days) to n = 112,150 days compared to all baseline migraine days (Figure 2), which equaled a 0.45 day increase in MMD. In these additional migraine days introduced by a shortened duration criterion, an analgesic was used in 22.7% (data not shown). This percentage is comparable to the number of analgesic days in days defined by the baseline criteria (with a duration of ≥4 hours) (23.3%). Furthermore, these additional days had a median duration of 2.5 (IQR 2.0–3.0) hours, which is shown in Figure 3.

Migraine days based on typical migraine characteristics with a duration of ≥

Reported headache durations (<4 hours, ≥30 minutes) for migraine days matching the migraine characteristics (with a shortened duration of ≥30 minutes) without triptan use. The median headache duration in these days was 2.5 (IQR 2.0–3.0) hours.
Differences between CM, HFEM and LFEM
The ratio between migraine days matching migraine characteristics and those defined by triptan intake alone differed between CM, HFEM and LFEM patients (p < 0.001) (Table 3). For CM patients in particular, the majority of days (95.2%) matched the typical migraine characteristics. For HFEM and LFEM this was 78.7% and 74.0% respectively. Headache duration was the most common missing factor for CM (58.5%) and HFEM (71.3%). For LFEM patients, nausea/vomiting or photophobia/phonophobia was most often missing (82.8%), whereas headache duration of ≥4 hours was missing in 26.3% of days (online Supplementary Figure 1).
Number of MMD (mean ± SD) using the migraine characteristics with a duration of ≥4 hours is compared to using an adjusted minimum headache duration criterion of 30 minutes, in combination with migraine days defined by triptan intake or days with effective triptan response. In bold the number of MMD following our proposed definition.

CM, Chronic migraine is defined as ≥3 consecutive months with ≥15 headache days per month with ≥8 MMD. HFEM, High frequent episodic migraine is defined as ≥1 month with ≥8 MMD. LFEM, Low frequent episodic migraine is defined as <8 MMD.
There was a strong, positive linear correlation (r = 0.94, p < .001) between the number of MMD (baseline definition) and the relative number of migraine days matching all migraine criteria (without days only defined by triptan intake). Figure 4 shows the linear regression line with a slope of 3.64 ± 0.034.

Percentage of migraine days matching the typical characteristics for migraine (with a duration of ≥4 hours) shows a positive correlation with the total number of migraine days per patient using our baseline definition (≥4 hours and all triptan intake). In red the linear regression line fitted on the data.
Effective triptan response
Considering only migraine days with an effective triptan response instead of all days with triptan intake independent of whether there was an effective response reduced the number of total migraine days with 5.2% (n = 100,905 days vs n = 106,430 days). Figure 5 shows the number of migraine days where a triptan was used (43.4%, n = 46,094 days) compared to the subset of these days (27.1%, n = 28,804 days) with an effective response.

Migraine days based on typical migraine characteristics with a duration of ≥4 hours (85.9%) vs days defined by triptan intake, divided in effective triptan response (27.1%) or all triptan intake (43.3%) for all patients (n = 1494). A triptan was considered effective if the headache was reduced from moderate or severe to mild or no-pain within 2 hours post-dose.
Change in number of monthly migraine days
Table 3 shows the number of MMD as result of the use of different migraine day definitions. The use of our baseline definition resulted in a mean ± SD of 6.9 ± 4.9 MMD in all patients. By adjustment of the headache duration criterion to ≥30 minutes and including all days with triptan intake as migraine days resulted in a mean ± SD of 7.4 ± 5.1 MMD. The numbers of mean MMD with the different definitions for the subgroups of CM, HFEM and LFEM patients are shown in Table 3.
Discussion
There is a need for consensus on the definition of a migraine day for clinical and research practices. In order to formulate a standardized definition, we compared four different definitions. We initially defined a migraine day based on the typical migraine headache and associated characteristics with a duration of ≥4 hours, or a day with triptan intake. We found that 14.1% of all identified migraine days were defined by triptan intake alone, and did not match all migraine characteristics. Headache duration was the most frequent missing criterion (66.6%) in these days defined by triptan intake alone, which is likely explained by the effectiveness of the triptan. Using a headache duration of ≥30 minutes resolves this issue, and results in only 5.4% increase in monthly migraine days (MDD) in which a triptan was not used. This 5.4% increase in migraine days equals on average a 0.45 day increase in MMD. Although shorter, the majority of migraine days identified by the adjusted duration criterion still have a headache duration of ≥2 hours. We propose to use the ≥30 minutes instead of ≥4 hour duration criterion, since all other migraine characteristics are still met, and the frequency of acute medication use seems similar as in migraine days defined by the ≥4 hour duration criterion. Since clinicians advise patients to take their acute medication at the onset of the migraine headache phase, it would be incorrect not to define an aborted attack as a migraine day. Defining too few migraine days should be avoided, since it can lead to an underestimation of the actual burden of patients, for example due to patients following the advice not to delay taking acute medication.
While we propose to use a uniform definition for all patients, it should be noted that the use of different definitions may have different implications for CM, HFEM and LFEM patients. For instance, defining days with triptan intake as a migraine day influences the number of MMD for EM patients in particular. There are several possible explanations for this finding. For example, in our cohort in HFEM and CM patients the number of medication days may be relatively low in relation to the total MMD as compared to EM patients as our patients receive the instruction to prevent MOH (16). Correspondingly, patients with LFEM may have more latitude to take a triptan, since they are less prone for MOH. We hypothesize LFEM patients take their triptan in time, before the attack becomes complete, which results in relatively more days with triptan intake and missing migraine characteristics.
Headache duration was the most common missing factor for CM and HFEM, whereas nausea/vomiting or photophobia/phonophobia was most often missing for LFEM. Patients with CM or HFEM might take their medication earlier in an attack, preventing the headache from lasting ≥4 hours, presumably driven by a higher anxiety for upcoming attacks (17–19).
We showed that including only days where the headache successfully responds to triptan intake as migraine day, as stated in the clinical guidelines (1,2), reduces the number of migraine days with 5.2%, equaling a 0.38 average decrease in MMD. However, we argue that the definition for triptan effectiveness has limitations. For example, migraine days with “mild” severity would never be marked as having an effective triptan response, because a triptan is only considered effective according to the clinical guidelines when it reduced a moderate or severe headache to mild or no pain two hours post-dose. In clinical practice, patients may already take a triptan when the headache is still mild. Moreover, in most other E-diaries the information on triptan effectiveness is not asked or asked without specifying the severity before and after triptan intake. We therefore propose to include all days with triptan intake independent of effectivity in the definition of a migraine day to avoid heterogeneous outcomes.
This study has several major strengths. Firstly, data was collected in a large, well-defined patient cohort over a long follow-up period. Moreover, we used our validated headache E-diary which has been shown to provide reliable migraine data (6). Our study also presents some limitations. Firstly, we did not look in detail at the definition of a migraine day based on aura symptoms, but we do propose to include all days with (visual) aura symptoms lasting 5–60 minutes as migraine days (6). We solely took visual aura symptoms into account, since 98–99% of all migraine attacks with aura are accompanied by visual symptoms (20–22). Moreover, it is impossible to ask all possible aura symptomatology for each day as this is too time consuming, thus simplification of aura symptomatology for headache E-diaries is needed to lower the data entry burden for patients and thereby improve diary compliance (23). Secondly, when defining days of acute medication intake for migraine, only days with triptan intake were taken into account. Ergotamines, ditans, and gepants are currently not prescribed in clinical practice in the Netherlands. We expect no change in results when these acute migraine medications are included. Thirdly, although prodromal symptoms are part of migraine attacks, these symptoms are difficult to quantify and not part of migraine definitions in the ICHD-3 and were therefore not included in this study. Finally, further research might be needed for larger numbers of patients with MOH, although those patients were also included in our study, as well as non-white populations. Our study was performed in a European country, whereas in those from the Western Pacific with Asian ethnicity osmophobia is more often reported besides photo- and phonophobia. However, osmophobia was not added in the ICHD-3 and therefore not studied in this study (24).
In this study we showed that headache duration is the main factor causing migraine days with triptan intake not to match the typical migraine characteristics. In addition, adjustment of the minimum required headache duration to ≥30 minutes led to a minor 5.4% increase in the total number of migraine days and the majority still had a duration of multiple hours. We propose to define a MD as follows: 1) (a) headache duration ≥30 minutes; (b) matching ≥2 out of 4 characteristics: unilateral, pulsating, moderate to severe pain, aggravation by or causing avoidance of routine physical activity; and (c) during headache ≥1 of the following: nausea and/or vomiting, photophobia and phonophobia or 2) (visual) aura duration 5–60 minutes or 3) a day with headache for which acute migraine-specific medication is used irrespective of its effect.
For future releases of the ICHD criteria, we would recommend including consensus on the definition of migraine days to prevent the use of different definitions hindering the comparability of different studies. However, initial individual assessment will remain essential for accurate diagnosis and classification of patients.
Article highlights
A migraine day should have a duration of ≥30 minutes instead of ≥4 hours. All days with headache for which acute migraine-specific medication is used should be counted as migraine days, irrespective of its effect.
Supplemental Material
sj-jpg-1-cep-10.1177_03331024231166625 - Supplemental material for Defining migraine days, based on longitudinal E-diary data
Supplemental material, sj-jpg-1-cep-10.1177_03331024231166625 for Defining migraine days, based on longitudinal E-diary data by Britt W. H. van der Arend, Iris E. Verhagen, Matthijs van Leeuwen, Marnick Q. T. P. van der Arend, Daphne S. van Casteren and Gisela M. Terwindt in Cephalalgia
Supplemental Material
sj-pdf-2-cep-10.1177_03331024231166625 - Supplemental material for Defining migraine days, based on longitudinal E-diary data
Supplemental material, sj-pdf-2-cep-10.1177_03331024231166625 for Defining migraine days, based on longitudinal E-diary data by Britt W. H. van der Arend, Iris E. Verhagen, Matthijs van Leeuwen, Marnick Q. T. P. van der Arend, Daphne S. van Casteren and Gisela M. Terwindt in Cephalalgia
Footnotes
Acknowledgements
We thank our patients with migraine and Hermes Spaink for their contribution.
Author Contributions
BWHvdA, IEV, DSvC and GMT contributed to conception and design of the study. BWHvdA, IEV and MQTPvdA contributed to acquisition and analysis of data. BWHvdA, IEV, DSvC, GMT contributed to drafting a significant portion of the manuscript. All authors contributed to the critical review of the manuscript.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: IEV, BWHvdA and GMT report independent support from the Dutch Research Council (849200007) and the Dutch Brain Foundation (HA2017.01.05). GMT reports consultancy or industry support from Abbvie/Allergan, Lilly, Lundbeck, Novartis, and Teva, and independent support from the European Community, Dutch Research Council, Dutch Heart and Brain Foundations and Dioraphte.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article: This project was not funded by any of the grants indicated in the COI section.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
