Abstract
Background
The antimigraine drug sumatriptan causes adverse chest effects, namely dyspnea and chest tightness, through unclear mechanisms. Dyspnea is an unpleasant sensation evoked by peripheral sensory signals transmitted to the central nervous system. Capsaicin-sensitive lung vagal (CSLV) afferents are nociceptive-like fibers that provide sensory input from the airways and lungs, mediating airway defense reflexes and evoking unpleasant respiratory sensations. The present study was carried out to investigate the role of CSLV afferents in mediating these adverse chest effects.
Methods
Experiments were performed using male Brown-Norway rats. In an in vivo study, we investigated the effect of sumatriptan on CSLV-fiber activities and fiber-mediated airway reflexes using single fiber recordings and breathing pattern monitoring in anesthetized rats. In an in vitro study, the effect of sumatriptan on neuronal sensitivity was evaluated using Ca2+ imaging in rat primary cultured CSLV neurons.
Results
Our results showed that intravenous infusion of sumatriptan increased the excitability of CSLV afferents to chemical and mechanical stimuli in anesthetized rats; this sensitizing effect occurred 3–20 minutes after termination of the sumatriptan infusion and reversed by 80 minutes later. In isolated CSLV neurons, sumatriptan-induced enhancement of Ca2+ transients evoked by capsaicin was blocked by pretreatment with a 5-hydroxytryptamine 1B and 1D (5-HT1B/1D) receptor antagonist and a protein kinase A inhibitor, whereas an antagonist of the transient receptor potential ankyrin 1 failed to do so. Additionally, in anesthetized, spontaneously breathing rats, a sumatriptan infusion potentiated changes in CSLV afferent-mediated breathing patterns, suggesting that enhanced sensory signals were transmitted to the central nervous system. Similarly, this potentiating effect was also abolished by a 5-HT1B/1D receptor antagonist. Furthermore, immunofluorescence staining confirmed that 5-HT1B/1D receptors were expressed in isolated CSLV neurons.
Conclusions
We concluded that sumatriptan sensitizes CSLV afferents through a direct action on 5-HT1B/1D receptors expressing in nerve endings followed by protein kinase A activation in rats. These findings suggest that sensitization of CSLV afferents may contribute to the chest discomfort experienced by some migraineurs following sumatriptan administration.
Introduction
Migraine is a severe and recurrent headache disorder that greatly impairs patients’ quality of life (1). Sumatriptan, a selective agonist of 5-hydroxytryptamine 1B and 1D (5-HT1B/1D) receptors, is an effective drug for treating of migraines (2). In migraine patients, commonly reported side effects of sumatriptan include cutaneous hyperalgesia (3,4), transient worsening of headache (5) and transient chest discomfort (e.g. dyspnea and chest tightness) (6,7). The chest-related adverse effects, which occur in approximately 10% of sumatriptan users, substantially reduces medication adherence (6,7). Additionally, due to similar symptoms between chest discomfort and angina, a US cohort analysis revealed a 33.1% elevation in chest-related medical expenses after sumatriptan treatment compared to before treatment (8). Initially, adverse chest effects were suspected of heart attack due to angina-like symptoms and vasopressor properties of sumatriptan (9). However, subsequent studies showed that chest-related side effects in migraineurs who had no history of cardiovascular disease were not due to myocardial ischemia (6,10–13). This conclusion was based on no changes in electrocardiographic patterns (11,13–15) or myocardial perfusion (11) after sumatriptan administration, even when patients reported experiencing chest discomfort (11,13). Currently, the mechanism responsible for sumatriptan-induced chest symptoms remains elusive.
Chest discomfort is a sensation that involves activation of peripheral sensory nerves. Sensory nerves detect environmental stimuli and transmit sensory signals to the central nervous system for processing and integration, ultimately shaping expression of sensations. Capsaicin-sensitive lung vagal (CSLV) afferents are nociceptive-like free nerve endings that innervate the entire respiratory tract (16). These fibers are chemosensitive and can detect both exogenous substances (e.g. ozone (17) and cigarette smoke (18)) and endogenous substances (e.g. prostaglandin E2 (19), bradykinin (20) and reactive oxygen species (21)), thereby triggering multiple respiratory reflexes, including coughing, mucus secretion and bronchoconstriction, thus playing a crucial role in respiratory defense (22).
In addition to the respiratory reflexes described above, projections of sensory signals to the central nervous system also induce chest discomfort characterized by dyspnea and chest tightness in humans (23,24). These unpleasant sensations are similar to the chest discomfort caused by sumatriptan. Additionally, allodynia observed in migraine patients following repeated use of sumatriptan (25) may result from hypersensitivity of nociceptive somatic (4,26) and meningeal afferents (4,5). In the lungs, these sensory nerves correspond to CSLV afferents. Moreover, activation of CSLV afferents was reported to be responsible for drug-induced adverse chest effects, such as dyspneic sensations evoked by intravenous adenosine, a treatment for cardiac arrhythmias (27–29).
Based on the above information, sumatriptan-induced chest discomfort may be related to CSLV afferents; however, whether sumatriptan can affect sensory nerves remains unclear. The present study was carried out to investigate the effect of sumatriptan on CSLV afferents through multiple experimental approaches. We conducted this study to 1) determine whether sumatriptan alters the excitability of CSLV afferents in anesthetized, artificially ventilated Brown-Norway rats and the chemosensitivity of primary cultured CSLV neurons; 2) identify the contribution of 5-HT1B/1D receptors to sumatriptan-induced effects; 3) investigate the role of intracellular protein kinase A (PKA) signaling in these effects; and 4) determine whether sumatriptan enhances CSLV afferent-mediated respiratory reflex responses in anesthetized, spontaneously breathing rats.
Methods
Animals
Male Brown-Norway rats were purchased from the National Laboratory Animal Center (Taipei, Taiwan). Animals were handled in accordance with standards established by the Guide for the Care and Use of Laboratory Animals published by the National Institutes of Health, and approved by the IACUC (permits LAC-2018-0005 and LAC-2021-0538) of Taipei Medical University. Animals were housed two per cage under a 12:12 hour light/dark photocycle (lights on at 07.00 hours) with ad libitum water and food in the Laboratory Animal Center of Taipei Medical University. All efforts were made to minimize the number of animals used and their suffering. Anesthetics were administered before any surgical procedures, and supplemental doses were provided as needed to sustain the elimination of pain reflexes produced by pinching the rat's tail throughout the experiments. Rats were randomly divided into control and experimental groups. At the end of the experiment, the animals were euthanized using an intravenous injection of KCl during deep anesthesia. All experiments were carried out in accordance with ARRIVE (Animal Research: Reporting of In Vivo Experiments) guidelines (30).
In vivo study
General animal preparations
Adult Brown-Norway rats (250–350 g) were anesthetized with an intraperitoneal injection of α-chloralose (100 mg/kg) and urethane (500 mg/kg) dissolved in distilled water with 2% borax. A rat was placed in a supine position, and the body temperature was maintained at 36°C with a heating pad. Cannulation of the right femoral artery and left femoral vein respectively enabled arterial blood pressure recording and pharmacological agent administration. A catheter was inserted via the left jugular vein to the right atrium for intravenous injections of a CSLV-afferent stimulant. A tracheostomy allowed the insertion of a tracheal cannula for respiratory airflow and tracheal pressure (Ptr) measurements. Following the intraperitoneal injection of a fixed dose of anesthetics and completion of the cannulation surgical procedures, baseline cardiorespiratory parameters were measured before initiating the experimental protocols. If an anesthetized rat showed abnormal baseline values, such as an irregular breathing pattern, bradycardia or hypotension, subsequent experimental protocols were not conducted on that animal. Once the experimental protocols were initiated, all data were collected and included in the analysis.
Electrophysiological recording of CSLV afferents
The measurement of fiber activity (FA) of CSLV afferents was described in detail in our previous study (31). A schematic diagram of the experimental setup is shown in Figure 1(a). Briefly, the anesthetized rats were artificially ventilated with a respirator (model 683; Harvard Apparatus, Holliston, MA, USA); tidal volume and respiratory frequency were set at 8 ml/kg and 50 breaths/minute, respectively, to mimic those of anesthetized, spontaneously breathing rats. In our previous study, the CO2 levels can be constantly maintained in a physiological condition under this ventilation setting (32). Ptr was measured by a differential pressure transducer (model P300D; Validyne, Northridge, CA, USA) via a side-port of the trachea cannula, and arterial blood pressure was measured by a pressure transducer (Statham P23XL; Spectramed, Columbus, OH, USA) via a femoral catheter. A midline thoracotomy was then performed to expose the thoracic cavity. Both vagus nerves were ligated just above the diaphragm to eliminate afferent signals originating from visceral organs below the diaphragm. To maintain a near-normal functional residual capacity, the expiratory outlet of the respirator was placed under 3 cmH2O pressure. The neck was opened along the midline, and a segment (approximately 1 cm) of the right cervical vagus nerve was carefully separated from the adjacent common carotid artery. The nerve was then sectioned as far rostrally as possible, and the distal (caudal) end of the cut vagus nerve was gently placed on a small dissecting platform immersed in a pool of mineral oil. A fine filament was teased from the desheathed nerve trunk and positioned on a platinum–iridium hook electrode for recording. Action potentials were amplified by an AC/DC differential amplifier (model 3000; A-M Systems, Sequim, WA, USA), monitored by an audio monitor (AM8RS; Grass Technologies, West Warwick, RI, USA) and displayed on an oscilloscope (model 2211; Tektronix, Beaverton, OR, USA). The filament was progressively teased until a single-unit action potential was isolated. Signals were recorded and analyzed by a polygraph recorder (MP36; Biopac, Goleta, CA, USA). CSLV afferents were identified by 1) intense response to capsaicin (2 μg/kg) within two seconds after the right-atrial bolus injection via jugular catheter and 2) the locations of the fibers could be identified by their responses to pressing of the lungs with a cotton swab at the end of the experiment. FA responses were calculated as the difference between peak and baseline FA values (ΔFA). Peak responses to capsaicin, lung inflation, and adenosine were averaged over 3, 5 and 10 seconds, respectively, following stimulation.

Sumatriptan (Suma) increased the excitability of capsaicin-sensitive lung vagal (CSLV) afferents to a capsaicin injection in rats. (a) Schematic diagram illustrating the experimental setup of single-fiber recording of CSLV afferents in an anesthetized, artificially ventilated rat. The activity of capsaicin-sensitive afferents from pulmonary branches of the vagus nerve was measured from a thin filament of the caudal end of the right sectioned cervical vagus nerve placed on a platinum–iridium electrode. (b) Experimental timeline illustrating CSLV-afferent responses to right-atrial capsaicin injections (arrows) obtained from 10 minutes before and 3–80 minutes after termination of the infusion of Suma or its vehicle. (c) Experimental records illustrating CSLV-afferent responses to capsaicin injections (arrows) before (control), 3 minutes and 80 minutes after termination of the Suma (1.2 mg/kg, left panel) or vehicle infusion (isotonic saline, right panel) in two anesthetized rats. Notice that the fiber response was potentiated 3 minutes after termination of the Suma infusion and recovered by 80 minutes later. (d) Group data of the increase in fiber activities (ΔFA values) elicited by capsaicin injections collected at three time points: before the infusion (control), at peak response times of 3 or 20 minutes (sensitization), and at 80 minutes (recovery) after infusion termination. The open bars and two sets of green bars represent infusion of the vehicle, and low-dose (0.6 mg/kg) and high-dose Suma (1.2 mg/kg), respectively. ΔFA is the difference between the peak FA (averaged over a 3-second interval) and the baseline FA for each CSLV afferent. Data are the mean ± SEM (n = 8). *p < 0.05 and **p < 0.01 using a mixed-effects model followed by Tukey's HSD post-hoc test. AP, action potential; Ptr, tracheal pressure; ABP, arterial blood pressure; imp, impulses.
Measurement of airway reflexes
In spontaneously breathing, anesthetized rats, respiratory airflow was measured using a heated pneumotachograph (model 8420; Hans-Rudolph, Shawnee, KS, USA) with a differential pressure transducer (P300D, Validyne). The tidal volume was derived by integrating the airflow, and arterial blood pressure was measured with a pressure transducer (Statham P23XL; Spectramed) via a femoral catheter. Signals were recorded on a polygraph recorder (MP30; Biopac) and analyzed with an online system (Biocybernetics TS-100; Taipei, Taiwan). Before the capsaicin injection, lungs were hyperinflated (Ptr > 20 cmH2O) to standardize the volume history. The apneic ratio was calculated as the longest expiratory time (TE) within 20 seconds post-injection divided by the baseline TE (10-breath average).
In vitro study
Identification of vagal bronchopulmonary neurons
Cell bodies of bronchopulmonary neurons located in nodose and jugular (N/J) ganglia were identified by retrograde labeling using 1,1′-dioctadecyl-33,3′,3′-tetrameth-ylindocarbocyanine perchlorate (DiI; Invitrogen, Waltham, MA, USA), a fluorescent tracer, instilled into the tracheal lumen of anesthetized rats, following a previous protocol (33). Seven to 10 days before the experiments, young Brown-Norway rats (100–150 g) were anesthetized with 2% isoflurane aerosol. A midline neck incision was made, and DiI dissolved in isotonic saline containing 1% ethanol (0.2 mg/ml, 0.05 ml) was injected into the trachea with a 30-gauge needle. The incision was then closed.
Isolation and culture of CSLV neurons
A DiI-labeled rat was anesthetized with 5% isoflurane and decapitated. The head was immersed in Dulbecco’s modified Eagle’s medium (DMEM)/F12 for 30 minutes. N/J ganglia were harvested, cut into small pieces and incubated with collagenase (0.04%; MilliporeSigma, St. Louis, MO, USA) and dispase II (0.02%; Roche, Basel, Switzerland) in DMEM for 60 minutes at 37°C in 5% CO2. The suspension was centrifuged (150 g for 5 minutes), and the cell pellet was resuspended in modified DMEM and triturated. Dispersed cells were separated by centrifugation through 15% bovine serum albumin (MilliporeSigma), resuspended in modified DMEM, and placed on poly-
Measurement of intracellular Ca2+ transients in CSLV neurons
Ca2+ transients were measured using established methods (34). Neurons on coverslips were incubated with modified DMEM containing 5 μM Fura-2AM (Invitrogen) for 30 minutes at 37°C in 5% CO2. Coverslips were mounted into a perfusion chamber and perfused with extracellular solution (ECS) by a gravity-fed valve control system (VC-66CS; Harvard Apparatus) for 30 minutes for deesterification. Dual images (340 nm and 380 nm excitation, 510 nm emission) were captured using an inverted microscope (DM IL LED; Leica, Wetzlar, Germany) with a CCD camera (Zyla 4.2 sCMOS; Andor Technology, Belfast, UK). Pseudocolored ratiometric images were analyzed at 2-second intervals using MetaFluor software (Molecular Devices, San Jose, CA, USA). A Ca2+ transient evoked by a stimulant was calculated as the ΔRatio (ΔF340/F380, difference between the peak amplitude (4 seconds average) and the baseline (30 seconds average). DiI-labeled CSLV neurons that met the following criteria were selected from cultured cells to measure Ca2+ transients: 1) a spherical shape with no neurite outgrowths and 2) activated by capsaicin (0.1 μM for 30 seconds). To determine the sensitizing effect of sumatriptan on CSLV neurons, Ca2+ transients evoked by capsaicin (0.1 μM for 30 seconds) were compared between before (1st Ca2+ transient) and after (2nd Ca2+ transient) sumatriptan. An enhancing effect was calculated as a ratio of the 2nd Ca2+ transient to the 1st Ca2+ transient.
Immunofluorescence staining
The rat vagal neuron culture protocol was based on the above methods with adjustments of the DiI concentration (2 mg/ml; 0.2 ml × 2) and rat body weight (250–350 g). After 24 hours of plating, neurons were fixed with 4% paraformaldehyde for 10 minutes, followed by phosphate-buffered saline (PBS) washes. Fixed neurons were permeabilized and blocked with 0.1% Triton and 5% goat serum in PBS for 10 minutes at room temperature and incubated overnight at 4°C with guinea pig anti- transient receptor potential vanilloid 1 (TRPV1) (dilution 1:250; #ACC-030-GP; Alomone, Jerusalem, Israel), rabbit anti-5-HT1B (dilution 1:50; #ab13896; Abcam, Cambridge, UK) and rabbit anti-5-HT1D (dilution 1:50; #bs-12046R; Bioss, Woburn, MA, USA) antibodies. These primary antibodies were validated and successfully used for immunofluorescence staining in rat tissues (35–37). After PBS washes, neurons were incubated with a mixture of Alexa Fluor 488-conjugated goat anti-rabbit IgG (dilution 1:200; #ab150077; Abcam) and Alexa Fluor 405-conjugated goat anti-guinea pig IgG (dilution 1:200; #ab175678; Abcam) for one hour at room temperature. These stained neurons were covered with a coverslip and examined by fluorescence microscopy (Axio Observer Z1; Zeiss, Oberkochen, Germany) with a camera (CoolSNAP MYO CCD Camera; Teledyne, Waterloo, Ontario, Canada).
Chemical agents
In vivo study
Stock solutions of chemical agents were prepared as follows: sumatriptan succinate (10 mg/ml; United States Pharmacopeia, North Bethesda, MD, USA) was prepared in isotonic saline; capsaicin (250 μg/ml; MilliporeSigma) was prepared in 1% ethanol, 1% Tween 80 and 98% isotonic saline; adenosine (10 mg/ml; MilliporeSigma) was prepared in isotonic saline and GR127935 (2 mg/ml; MilliporeSigma) was prepared in 5% dimethyl sulfoxide (DMSO) and 95% isotonic saline. Solutions of these pharmacological agents at the desired concentrations were prepared daily by dilution with saline based on an animal's body weight.
In vitro study
Stock solutions of chemical agents were prepared as follows, Sumatriptan succinate (10 μM) was prepared in PBS; SB224289 (10 μM; Tocris, Bristol, UK) was prepared in 2% DMSO and 98% PBS; BRL15572 (10 μM; Tocris) was prepared in 2% DMSO and 98% PBS; GR127935 (10 μM; MilliporeSigma) was prepared in 0.01% DMSO and 99.99% PBS; Trans-cinnamaldehyde (100 mM; MilliporeSigma) was prepared in 10% ethanol, 10% Tween 80 and 80% PBS; and HC-030031 (85 mM; Cayman, Ann Arbor, MA, USA) was prepared in 100% DMSO. Solutions of these pharmacological agents at the desired concentrations were prepared daily by dilution with ECS.
Statistical analysis
The minimal sample size was based on the following criteria: 1) the sample size required to achieve statistical significance in comparable experiments conducted in our laboratory (33,34,38) and 2) the minimal sample sizes in each group are required to achieve a power of 0.8 and alpha = 0.05 calculated using G*Power software (http://www.gpower.hhu.de) based on preliminary data. The experimental protocol for each study is illustrated and described where appropriate. To ensure objectivity, data analysis and calculation were carried out in a blinded manner by two individual investigators. Statistical analyses were performed using Prism, version 10 (GraphPad Software Inc., San Diego, CA, USA). Prior to statistical analysis, Shapiro–Wilk tests were conducted to evaluate whether the data followed a normal distribution. For datasets that met the assumption of normality, two-way analysis of variance (ANOVA) followed by Tukey's honestly significant difference (HSD) post-hoc test was applied. Non-normally distributed variables were analyzed using a Mann–Whitney U-test, a Kruskal–Wallis test followed by Dunn's post-hoc test or a mixed-effects model (restricted maximum likelihood, REML) followed by Tukey's HSD post-hoc test. p < 0.05 was considered statistically significant. All data are reported as the mean ± SEM. Statistical analyses, numbers of animals used (n) and p-values are reported where appropriate. Detailed statistical results are provided in the supplementary material (Table S1).
Results
Sumatriptan enhanced the excitability of CSLV afferents to capsaicin in anesthetized, ventilated rats
To evaluate the potentiating effect of sumatriptan on capsaicin-evoked discharges (via activation of TRPV1), CSLV-afferent responses to a right-atrial bolus injection of capsaicin (0.5 μg/kg) were measured at control (10 minutes before infusion) and at 3, 20 and 80 minutes after the infusion of sumatriptan or its vehicle (Figure 1(b)). Under control conditions (before sumatriptan infusion), capsaicin injection elicited a mild discharge in CSLV afferents (Figure 1(c), upper left). This response was markedly enhanced 3 minutes after termination of the sumatriptan infusion (1.2 mg/kg; 0.1 ml/minute for 20 minutes) (Figure 1(c), middle left) and returned to baseline by 80 minutes post-infusion (Figure 1(c), lower left). In contrast, infusion of the sumatriptan vehicle (isotonic saline) did not induce such potentiation (Figure 1(c), right). Among CSLV afferents, peak capsaicin-induced activity typically occurred at 3 or 20 minutes after sumatriptan infusion. Sumatriptan potentiated these responses in a dose-dependent manner; relative to pre-infusion levels, the higher dose (1.2 mg/kg) increased the response by approximately 3.1-fold, whereas the lower dose (0.6 mg/kg) produced an approximately 1.4-fold increase (Figure 1(d)). Notably, Ptr and FA remained unchanged throughout the experimental period in both sumatriptan- and vehicle-treated groups (Table 1). These findings demonstrate that sumatriptan enhances the excitability of CSLV afferents to capsaicin without altering baseline Ptr and FA.
Effect of an infusion of sumatriptan or its vehicle on the baseline tracheal pressure (Ptr) and fiber activity (FA) in anesthetized, artificially ventilated rats.
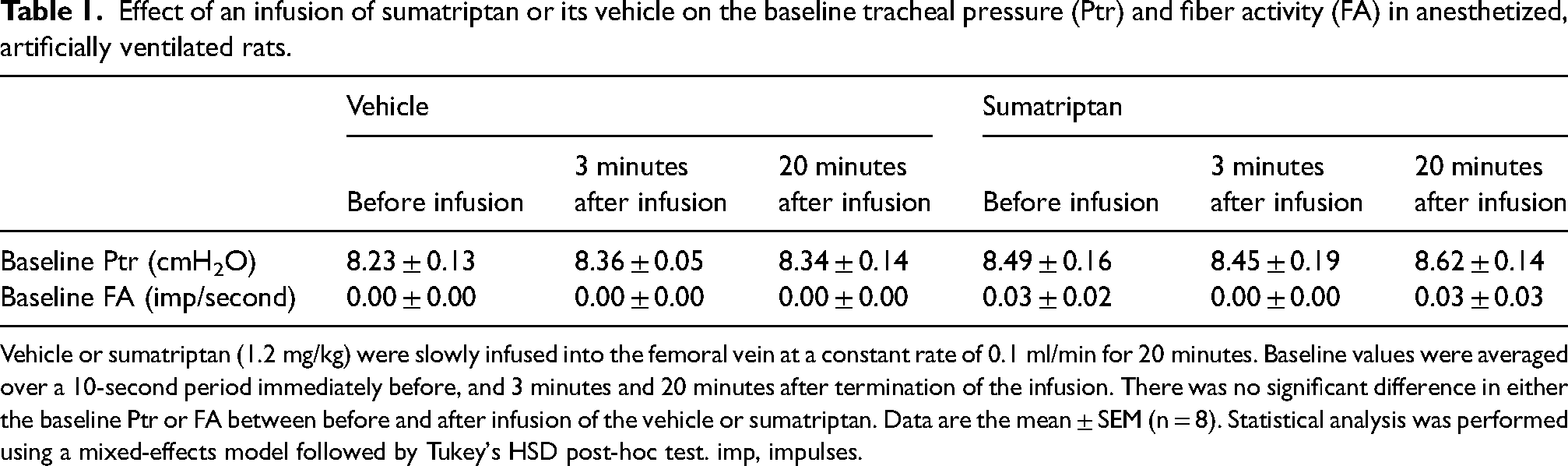
Vehicle or sumatriptan (1.2 mg/kg) were slowly infused into the femoral vein at a constant rate of 0.1 ml/min for 20 minutes. Baseline values were averaged over a 10-second period immediately before, and 3 minutes and 20 minutes after termination of the infusion. There was no significant difference in either the baseline Ptr or FA between before and after infusion of the vehicle or sumatriptan. Data are the mean ± SEM (n = 8). Statistical analysis was performed using a mixed-effects model followed by Tukey's HSD post-hoc test. imp, impulses.
Sumatriptan-induced sensitization of CSLV afferents was not restricted to capsaicin stimulation in anesthetized, ventilated rats
To determine whether the excitability increase induced by sumatriptan was specific to capsaicin stimulation, we evaluated CSLV afferent responses to two additional stimuli: adenosine injection (0.6 mg/kg), a chemical stimulant of CSLV fibers via adenosine A1 receptors (39), and constant-pressure lung inflation (Ptr = 30 cmH2O for 10 seconds), a mechanical stimulus. Under control conditions, both adenosine injection and lung inflation elicited only mild activation of CSLV afferents. In contrast, responses to both stimuli were significantly potentiated following sumatriptan infusion (Figure 2(a) and (b), left), whereas no such potentiation was observed in vehicle-treated animals (Figure 2(a) and (b), right). Group data confirmed that sumatriptan increased the adenosine-induced (Figure 2(c)) and lung inflation-induced (Figure 2(d)) changes in FA, suggesting that sumatriptan induces a non-selective sensitizing effect on CSLV afferents.
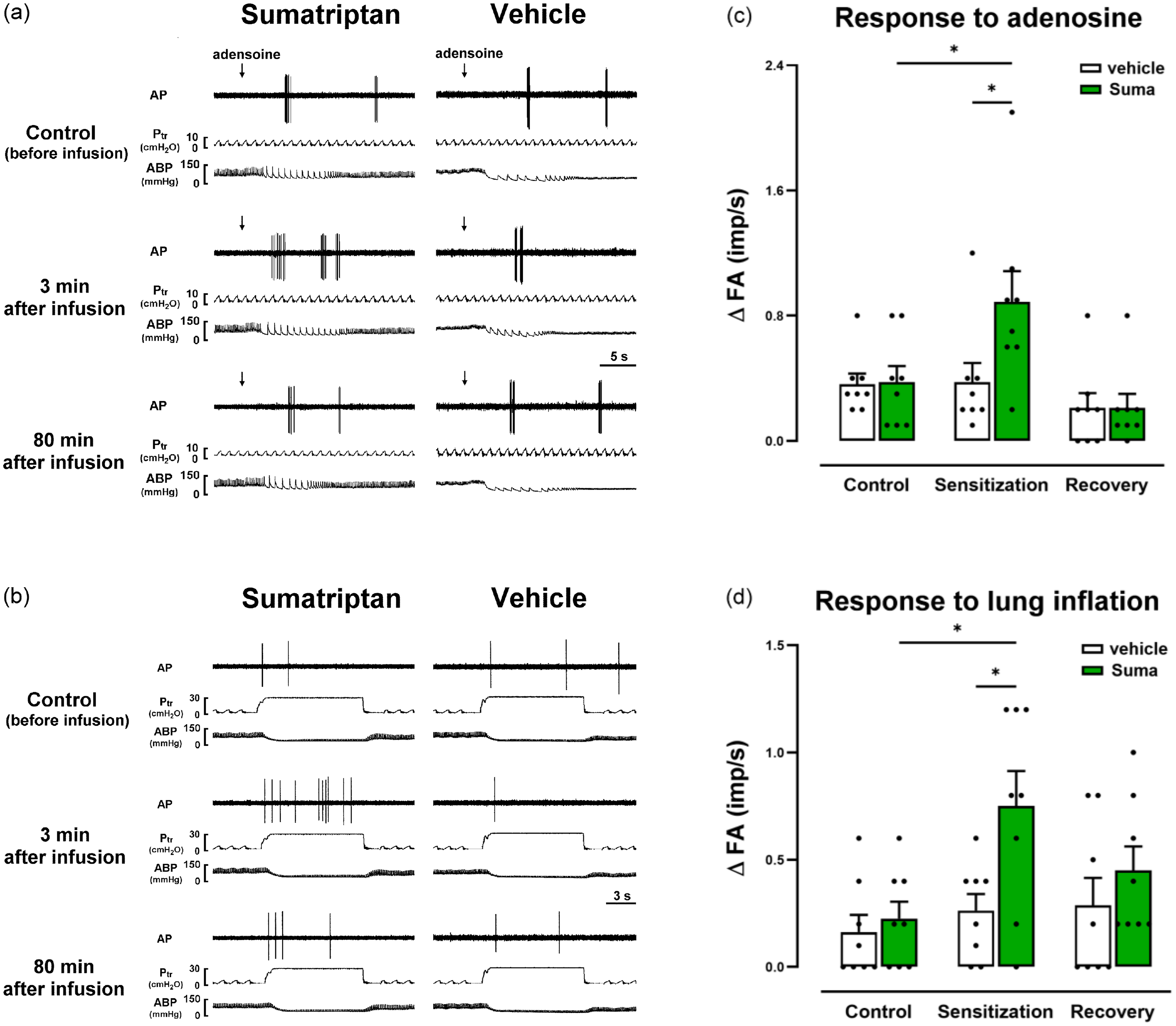
Sumatriptan (Suma) increased the excitability of rat capsaicin-sensitive lung vagal (CSLV) afferents to an adenosine injection and to lung inflation. (a, b) Experimental records illustrating CSLV-afferent responses to right-atrial adenosine injections (arrows, a) and lung inflation (b) before and after termination of a Suma (1.2 mg/kg) or vehicle infusion in four anesthetized, artificially ventilated breathing rats. (c, d) Group data of fiber activities (ΔFA values) elicited by adenosine injections (c) or by lung inflation (d) in rats treated with Suma or its vehicle. For further explanation of the ΔFA calculation, see Figure 1. Data are the mean ± SEM (n = 8). *p < 0.05 using a mixed-effects model followed by Tukey's HSD post-hoc tests. AP, action potential; Ptr, tracheal pressure; ABP, arterial blood pressure.
Sensitizing effect of sumatriptan was observed in isolated rat CSLV neurons
Because sumatriptan was administrated by systemic infusion route in the in vivo study, whether the sensitization resulted from a direct action on CSLV neurons or an indirect effect via non-CSLV pathways remained unclear. To clarify this, we examined the effects of sumatriptan on isolated CSLV neurons. Neuronal responses were assessed by monitoring changes in intracellular calcium concentration ([Ca2+]i), using the F340/F380 ratio. Under control conditions (before sumatriptan perfusion), capsaicin (0.1 μM for 30 seconds) elicited a mild, transient increase in [Ca2+]i. However, 10 minutes after perfusion of high-concentration sumatriptan (1 nM for 60 seconds, n = 23), reapplication of capsaicin evoked an enhanced Ca2+ transient (Figure 3(c) and (d)). This enhancement was not observed in neurons perfused with the vehicle (ECS, n = 21) or with low-concentration sumatriptan (0.5 nM, n = 26) (Figure 3(d)). These findings indicate that sumatriptan can directly sensitize CSLV neurons.

Sumatriptan (Suma) enhanced capsaicin (Cap)-evoked Ca2+ transients in isolated capsaicin-sensitive lung vagal (CSLV) neurons. (a–c) Experimental records illustrating Ca2+ transients evoked by Cap perfusion (arrows) before and 10 minutes after termination of perfusion. Treatment with the vehicle (Veh, extracellular solution) (a), low-concentration Suma (0.5 nM) (b) or high-concentration Suma (1.0 nM) (c) is indicated by white, and light- and dark-green bars, respectively. Cell viability was tested with a KCl solution (arrows, 60 mM). Notice that only high-concentration Suma treatment induced a significantly enhanced response. (d) Concentration effects on Suma-enhanced Ca2+ transients. The sensitizing effect was determined by the ratio of Ca2+ transient evoked by Cap after perfusion (2nd Cap) to those before perfusion (1st Cap). The horizontal dashed line depicts a ratio of 1 (indicating no sensitizing effect). Respective n values for the vehicle, and low- and high-Suma groups were 21 (from two rats), 26 (from two rats), and 23 (from two rats). Data are the mean ± SEM. *p < 0.05 and ***p < 0.005 using a Kruskal–Wallis test followed by Dunn's post-hoc test.
5-HT1B and 5-HT1D receptors were expressed in isolated CSLV neurons
Immunofluorescence staining was performed in cultured vagal neurons isolated from rat nodose and jugular (N/J) ganglia. Bronchopulmonary neurons were identified by retrograde labeling with DiI, which marked 25.7% of the total N/J neuronal population. Among the DiI-labeled neurons, 76.3% were immunopositive for TRPV1, indicating that these cells represent CSLV neurons. Notably, both 5-HT1B and 5-HT1D receptors were expressed in TRPV1-positive CSLV neurons (Figure 4). Specifically, 84.3% and 88.9% of these neurons expressed 5-HT1B and 5-HT1D receptors, respectively. The selectivity of the antibodies was confirmed through two standard control conditions in which either the primary or secondary antibody was omitted (see supplementary material, Figure S1). The expression of 5-HT1B and 5-HT1D receptors in CSLV neurons provides a rationale for further pharmacological investigation into their functional roles.
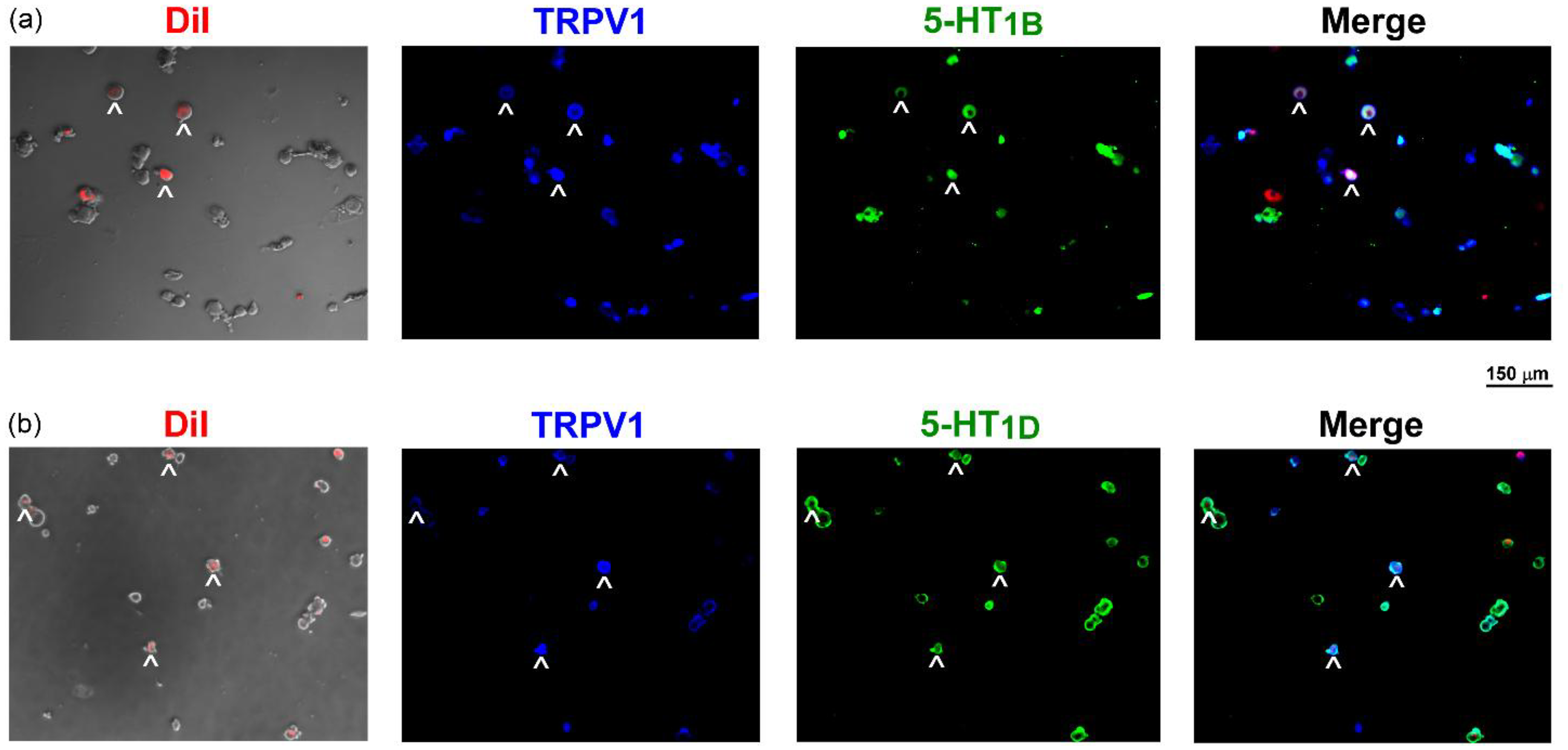
5-Hydroxytryptamine 1B and 1D (5-HT1B/1D) receptors expressed in rat capsaicin-sensitive lung vagal (CSLV) neurons. Double-labeling immunofluorescence staining was performed on CSLV neurons following intratracheal injection of 1,1′-dioctadecyl-33,3′,3′-tetrameth-ylindocarbocyanine perchlorate (DiI) (a retrograde tracer), showing (a) DiI (lung labeling, red)/transient receptor potential vanilloid 1 (TRPV1) (a marker of capsaicin-sensitive neuron, blue)/5-HT1B receptors (green). (b) As in (a), green staining targeted 5-HT1D receptors instead of 5-HT1B receptors. Arrows indicate neurons with triple labeling. Scale bar = 150 μm.
Sumatriptan-induced sensitization of CSLV neurons required 5-HT1B , 5-HT1D , and PKA activation, but was independent of transient receptor potential ankyrin 1 (TRPA1)
To determine whether 5-HT1B and 5-HT1D receptors are critical for sumatriptan-induced sensitization, selective antagonists targeting the 5-HT1B receptor (SB224289), the 5-HT1D receptor (BRL15572), or both 5-HT1B/1D receptors (GR127935) were administered 15 minutes before sumatriptan perfusion (1 nM for 60 seconds). Pretreatment with any of these antagonists (1 nM each) did not affect baseline F340/F380 (see supplementary material, Table S2) or the initial capsaicin-evoked Ca2+ transients (see supplementary material,, Table S3), but completely abolished the sumatriptan-induced enhancement of these responses (Figure 5(b) to (d)). In contrast, vehicle pretreatment (ECS) did not block the enhancing effect (Figure 5(a)), and group analysis confirmed that the sensitization was fully suppressed by receptor antagonism but remained intact in the vehicle group (Figure 5(e)).

Pretreatment with an antagonist of 5-hydroxytryptamine 1B and 1D (5-HT1B/1D) receptors abolished the enhancing effect of sumatriptan (Suma) on Ca2+ transients evoked by capsaicin (Cap) in isolated capsaicin-sensitive lung vagal (CSLV) neurons. (a-d) Experimental records illustrating the effect of a Suma perfusion (1 nM, black bar) on Ca2+ transients evoked by Cap (arrows) after pretreatment with the vehicle (Veh) (a), SB224289 (SB, a 5-HT1B receptor antagonist) (b), BRL15572 (BRL, a 5-HT1D receptor antagonist) (c) or GR127935 (GR, a 5-HT1B/1D receptors antagonist) (d). (e) Group data showing enhancement of Ca2+ transients after Suma was pretreated with Veh (n = 22 from two rats), SB (n = 26 from three rats), BRL (n = 20 from two rats) or GR (n = 25 from two rats). Data are the mean ± SEM. ***p < 0.005 using a Kruskal–Wallis test followed by Dunn's post-hoc test. (f, g) Experimental records illustrating the effect of a Suma perfusion (1 nM) on Ca2+ transients evoked by Cap after pretreatment with the Veh (f) or HC-030031 (HC, a TRPA1 antagonist) (g). (h) Group data showing enhancement of Ca2+ transients after Suma was pretreated with the Veh (n = 25 from two rats) or HC (n = 24 from two rats). Data are the mean ± SEM. Statistical analysis was performed using a two tailed unpaired Mann–Whitney test. For further explanations of the horizontal dashed line and calculation of the sensitized ratio, see Figure 3.
Given that sumatriptan has been shown to activate TRPA1 in HEK293 cells (40), we next investigated whether TRPA1 contributes to this sensitization. However, pretreatment with an TRPA1 antagonist (HC-030031; 10 μM) or its vehicle (0.01% DMSO) did not alter the sumatriptan-induced enhancement of capsaicin-evoked Ca2+ transients (Figure 5(f) to (h)). The efficacy and selectivity of HC-030031 were validated by use of cinnamaldehyde (a TRPA1 agonist, 0.3 mM for 40 seconds) and capsaicin (0.1 μM for 30 seconds): HC-030031 suppressed the cinnamaldehyde-evoked Ca2+ transients (ΔF340/F380: 0.51 ± 0.12 vs. 0.04 ± 0.02; p < 0.005 using two tailed unpaired Mann–Whitney test), but had no effect on capsaicin responses (ΔF340/F380: 0.60 ± 0.14 vs. 0.69 ± 0.13; p = 0.2406 using two tailed unpaired Mann–Whitney test) (see supplementary material, Figure S2). These findings indicate that TRPA1 activation is not involved in the sensitizing effect of sumatriptan on CSLV neurons.
Finally, to assess whether PKA activation is essential for the sensitizing effect of sumatriptan, we pretreated cells with H89, a selective PKA inhibitor, before sumatriptan perfusion. Pretreatment with H89 (10 μM), but not its vehicle (0.02% DMSO), completely abolished the sumatriptan-induced enhancement of the capsaicin-evoked Ca2+ transient (Figure 6), indicating that PKA activation is required for this sensitizing effect.

Pretreatment with a protein kinase A (PKA) inhibitor abolished the enhancing effect of sumatriptan (Suma) on Ca2+ transients evoked by capsaicin (Cap) in isolated capsaicin-sensitive lung vagal (CSLV) neurons. (a, b) Experimental records illustrating the effect of Suma perfusion (1 nM, black bar) on Ca2+ transients evoked by Cap (arrows) after pretreatment with vehicle (Veh) (a) or H89 (a PKA inhibitor) (b). (c) Group data showing enhancement of Ca2+ transients after Suma pretreated with the Veh (n = 22 from two rats) or H89 (n = 20 from two rats). For further explanations of the horizontal dashed line and calculation of the sensitized ratio, see Figure 3. Data are the mean ± SEM. ***p < 0.005 using a two tailed unpaired Mann–Whitney test.
Sumatriptan enhanced CSLV afferent–mediated airway reflexes in anesthetized, spontaneously breathing rats
Given that sumatriptan increases the excitability of CSLV afferents, we next investigated whether this peripheral sensitization also potentiates central processing, leading to enhanced airway reflexes. To address this, we returned to the in vivo model to assess CSLV afferent–mediated airway reflexes after sumatriptan treatment. Under control conditions (before sumatriptan infusion), a right-atrial bolus injection of capsaicin (1.0 μg/kg) elicited a mild, transient apneic response, comprising a hallmark reflex mediated by activation of CSLV afferents (Figure 7(a), left). Following sumatriptan infusion (1.2 mg/kg; 0.1 ml/minute for 20 minutes), this apneic response was potentiated 3 minutes post-infusion (Figure 7(a), middle) and returned to baseline by 80 minutes later (Figure 7(a), right), paralleling the time course of afferent sensitization observed in Figure 1 and Figure 2. In contrast, no potentiation of the apneic response was observed in animals receiving vehicle infusion (isotonic saline) (Figure 7(b)).
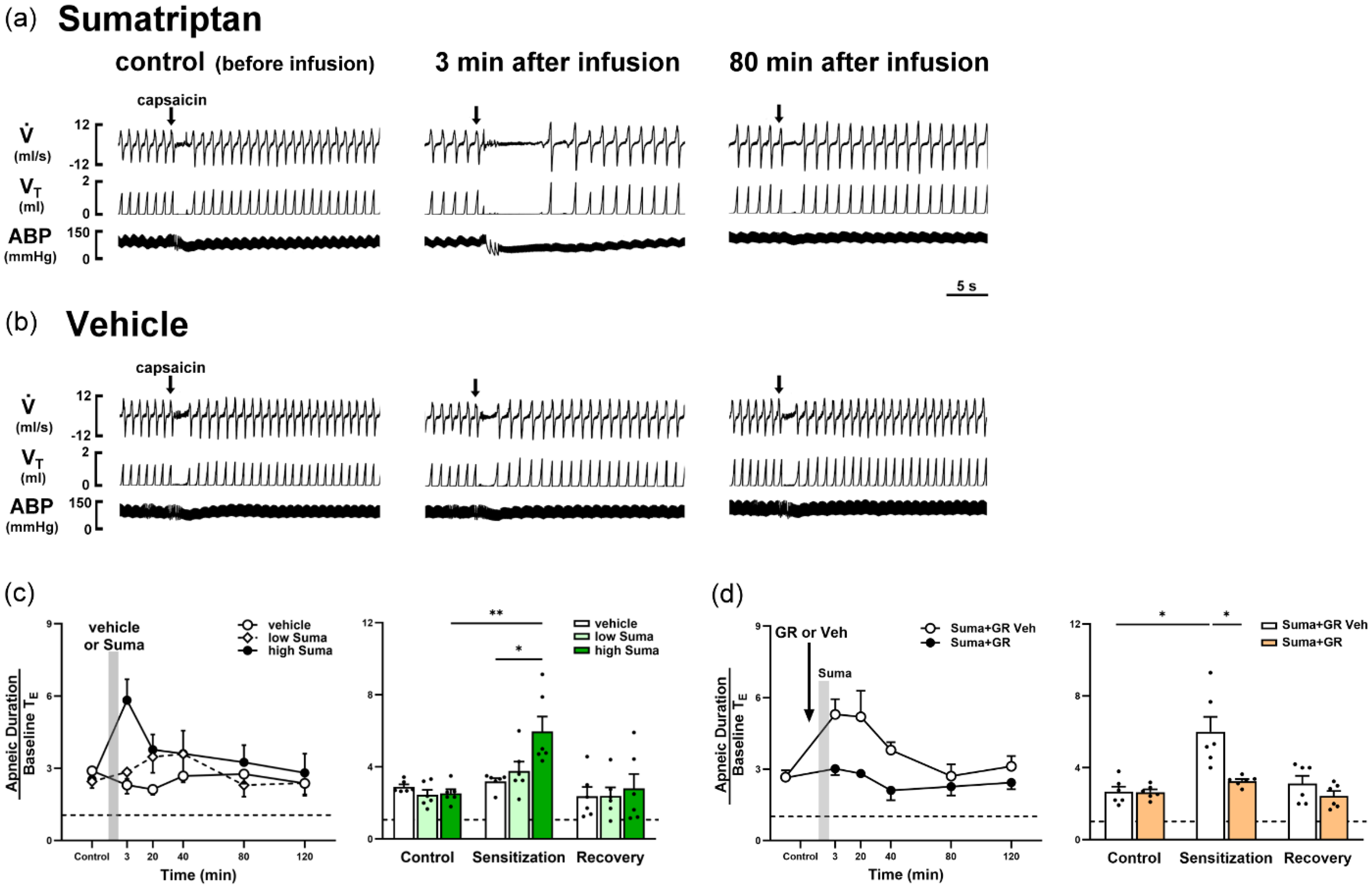
Infusion of sumatriptan (Suma) potentiated apneic responses to a capsaicin injection in rats. (a, b) Experimental records illustrating apneic responses to right-atrial capsaicin injections (arrows) before and after termination of a Suma (1.2 mg/kg) (a) or vehicle (Veh) (b) infusion in two anesthetized, spontaneously breathing rats. Notice that the apneic response was potentiated 3 minutes after termination of the Suma infusion and recovered by 80 minutes later. (c) Temporal response (left) and peak response (right) showing the dose effect on Suma-potentiated apneic responses to capsaicin. In the histogram, apneic responses were collected before, 3–120 minutes after termination of the infusion (gray vertical bar) of the Veh, or low-dose (0.6 mg/kg) or high-dose (1.2 mg/kg) Suma. In the bar diagram, apneic responses are compared between three time points: before infusion (control), at peak response times of 3 or 20 minutes (sensitization), and at 80 minutes (recovery) after infusion termination. Data are the mean ± SEM (n = 6). The horizontal dashed line depicts an apneic ratio of 1 (indicating no apnea). *p < 0.05 and **p < 0.01 using a mixed-effects model followed by Tukey's HSD post-hoc tests. (d) Role of 5-hydroxytryptamine 1B and 1D (5-HT1B/1D) receptors in Suma (1.2 mg/kg)-induced potentiation of the apneic response to capsaicin. The arrow on the left represents pretreatment with GR (300 μg/kg) or its Veh (0.75% DMSO). Collections of apneic responses for comparison were the same as those in (c). The horizontal dashed line depicts an apneic ratio of 1 (indicating no apnea). Data are the mean ± SEM (n = 6). *p < 0.05 using a two-way ANOVA followed by Tukey's HSD post-hoc test.
As summarized in Figure 7(c), apneic responses were measured before and after infusion of either vehicle or sumatriptan at low (0.6 mg/kg) or high (1.2 mg/kg) doses. Only the high-dose sumatriptan group showed significant potentiation at 3 and 20 minutes post-infusion, with responses returning to baseline by 80 minutes. Importantly, baseline cardiovascular parameters recorded at the corresponding time points did not change across groups (Table 2), indicating that potentiation of the airway reflex was independent of hemodynamic changes. These findings suggest that sumatriptan augments CSLV afferent-mediated signaling to the central nervous system, thereby potentiating airway reflex responses in a dose-dependent and reversible manner.
Effect of infusion of sumatriptan or its vehicle on baseline heart rate (HR) and mean arterial blood pressure (MABP) in anesthetized, spontaneously breathing rats.

The vehicle or sumatriptan (1.2 mg/kg) was slowly infused into the femoral vein. There was no significant difference in either baseline HR or MABP between before and after infusion of the vehicle or sumatriptan. Data are the mean ± SEM (n = 6). Statistical analysis was performed using a two-way analysis of variance followed by Tukey's HSD post-hoc test.
Sumatriptan potentiated CSLV afferent–mediated airway reflexes through 5-HT1B/1D receptors in anesthetized rats
To determine whether the sumatriptan-induced potentiation of CSLV-mediated airway reflexes depends on activation of 5-HT1B/1D receptors, GR127935 (300 μg/kg) or its vehicle (0.75% DMSO) was administered 10 minutes prior to sumatriptan infusion (1.2 mg/kg) in anesthetized, spontaneously breathing rats. GR127935, but not the vehicle, markedly suppressed the sumatriptan-induced potentiation of apneic responses to capsaicin (Figure 7(d)). To determine whether GR127935 alone affected the apneic response to capsaicin, capsaicin was injected before and after sumatriptan vehicle infusion in rats pretreated with GR127935. No significant difference was observed in the apneic responses (2.61 ± 0.37 vs. 2.40 ± 0.31; n = 6; p = 0.24 using two-tailed paired t-test), indicating that GR127935 did not attenuate the capsaicin-evoked reflex per se but specifically inhibited the potentiating effect of sumatriptan.
Consistent with this, electrophysiological recordings in anesthetized, artificially ventilated rats further demonstrated that GR127935 pretreatment abolished the sumatriptan-induced potentiation of CSLV fiber responses to both capsaicin injection (see supplementary material, Figure 3(a)) and lung inflation (see supplementary material, Figure 3(b)). These findings demonstrate that the potentiation of both reflex and afferent responses induced by sumatriptan requires activation of 5-HT1B and 5-HT1D receptors.
Discussion
Our results demonstrated that sumatriptan induced a non-specific increase in the excitability of CSLV afferents to chemical and mechanical stimuli in rats (Figures 1 and 2). In addition, this sensitizing effect was also found in isolated rat CSLV neurons (Figure 3), suggesting direct action by sumatriptan on CSLV afferents. After identifying expressions of 5-HT1B/1D receptors in CSLV neurons (Figure 4), we investigated their role in sumatriptan-induced sensitization. Inhibition of 5-HT1B/1D receptors and PKA by a pharmacological intervention abolished the sensitization in CSLV neurons (Figures 5 and 6). Based on an intriguing previous finding, we additionally investigated the potential involvement of TRPA1 in sensitization. While 10 μM sumatriptan activated TRPA1-expressing HEK293 cells (40), antagonism of TRPA1 did not affect sumatriptan-induced sensitization at 1 nM in our study (Figure 5). After validation of TRPA1 antagonist efficacy (see supplementary material, Figure S2), we determined that the sensitization in CSLV neurons was independent of TRPA1 signaling. In addition, sumatriptan potentiated CSLV afferent-mediated airway reflexes in rats (Figure 7), suggesting that enhancing sensory signals were sufficient for transmission to the central nervous system and triggering of respiratory responses such as chest discomfort. Furthermore, this potentiating effect on airway reflexes was also abolished by inhibiting activations of 5-HT1B/1D receptors (Figure 7). Based on all of these observations, we concluded that sumatriptan sensitizes CSLV afferents by a direct action on 5-HT1B/1D receptors expressed in nerve endings of CSLV afferents followed by PKA activation, which in turn induced airway hypersensitivity in rats.
A limited role for myocardial ischemia in sumatriptan-induced chest discomfort
Sumatriptan-induced chest discomfort was reported shortly after its marketing began in 1991 and was systematically documented in The Lancet in 1993 (10). Initially, researchers speculated that the mechanism was of cardiac origin since the chest discomfort resembled angina symptoms and sumatriptan had vasopressor properties. Indeed, a vasoconstrictive effect was observed in ex-vivo experiments which showed that sumatriptan caused a significant contraction in isolated human coronary arteries (41,42). However, the maximum concentration/median effective concentration (Cmax/EC50) ratio of sumatriptan was only about 0.4, which meant that the maximum plasma concentration in humans after an effective dose of sumatriptan reached just 40% of the level needed for a half-maximal effect in isolated coronary arteries, suggesting that sumatriptan at a therapeutic plasma concentration could not cause significant coronary vasoconstriction (41). Numerous human studies supported this finding, such as that by angiographic examinations, where only 12–16% decreases in coronary artery diameter occurred after intravenous or subcutaneous sumatriptan in migraineurs without cardiovascular diseases (14,15). Notably, even when these patients experienced chest-related adverse effects, sumatriptan did not alter myocardial perfusion (11). Throughout those human studies, sumatriptan did not affect the heart rate or electrocardiographic readings (11,14,15). Current investigations indicated that therapeutic doses are unlikely to cause myocardial ischemia in healthy migraineurs.
Involvement of CSLV afferents in sumatriptan-induced chest discomfort
For many decades, CSLV afferents were postulated to be the source of chest unpleasant sensations (e.g. dyspnea, chest tightness, breathlessness, etc.) (23). Studies in humans showed that projections of CSLV afferent signals to the central nervous system play a vital role in triggering chest discomfort symptoms, such as shortness of breath, chest tightness and breathlessness (23,24). In healthy human subjects, dyspneic sensations can be induced by inhalations or injections of various CSLV-afferent stimulants (e.g. adenosine (28,29), histamine (43) and nicotine (44)) or sensitizers (e.g. prostaglandin E2 (45)). Furthermore, when intravenous adenosine (known to stimulate CSLV afferents (27,39)) is used to treat tachycardia, the dyspneic adverse reaction can be reduced by inhaling lidocaine, a local anesthetic. This indicates a primary role of lung sensory nerves in the sensation of dyspnea (28,29). In addition, studies showed that patients with certain lung diseases experience significant relief from chest discomfort after the vagal nerve pathway is blocked, either through local anesthetic injections or surgical vagal nerve cutting (a vagotomy) (46–49). Our experimental results showed that intravenous infusion of sumatriptan sensitized rat CSLV afferents, thus enhancing airway reflexes through projections of sensory signals to the central nervous system. Furthermore, we observed that the timing of sumatriptan-induced airway reflex enhancement in rats (3–20 minutes) matched the period in which patients reported chest discomfort after taking sumatriptan, implying that enhancement of signal inputs of CSLV afferents may contribute to the development of chest discomfort after sumatriptan treatment in migraineurs.
Determination of the dose and administrated route of sumatriptan in this study
Although sumatriptan is mostly administered orally (50), we gave it by systemic infusion route in our study for two main reasons. First, oral administration exhibits higher variability in absorption and lower bioavailability patterns than systemic infusion routes among experimental subjects (51). Second, oral users experience chest discomfort less frequently than subcutaneous and intravenous users (6,7), which is highly associated with bioavailability patterns. Based upon these observations, oral administration may have constrained our capacity to observe and analyze the sensitizing effect of sumatriptan on CSLV afferents. In humans, intravenous or subcutaneous sumatriptan doses range 0.05–0.2 mg/kg (52,53). Applying the human-to-rat conversion factor (Km ≈ 6.2) (54), the translation from a human dose (0.2 mg/kg) to a rat dose is approximately 1.2 mg/kg. In addition, we administered sumatriptan through a slow infusion over 20 minutes (2 ml administered over 20 minutes) to prevent a rapid surge in the plasma concentration, simulating the time required to reach peak concentration following subcutaneous administration. Overall, the pharmacokinetic profile of sumatriptan in our rat model may have closely resembled that observed with clinical use in humans.
Sumatriptan-induced sensitization of CSLV neurons: Role of 5-HT1B/1D receptors
This study revealed that pretreatment with either an antagonist of 5-HT1B receptor or 5-HT1D receptor fully abolished sumatriptan-induced sensitization of CSLV neurons, suggesting that both receptors play essential roles in this process. However, if these receptors act through independent pathways, partial residual sensitization would typically be expected when only one is inhibited. A similar all-or-none blocking pattern was observed in other models, such as in sumatriptan-inhibited hyperalgesia induced by formalin in rats, where antagonism of either receptor alone eliminated the effect (55). Although the underlying mechanism remains to be fully elucidated, it seems to be unlike the involvement of insufficient antagonist selectivity in our study. The high-selectivity antagonists we used (BRL15572 and SB224289) were reported in other studies examining the effects of sumatriptan on the sensitivity of sensory neuron (56,57) and the contractility of vascular smooth muscle (58,59). Moreover, concentrations of antagonists we used were lower than those reported in the referenced studies, minimizing potential non-selective binding effects. Given prior reports of functional interaction between 5-HT1B and 5-HT1D receptors (60), we speculate that receptor crosstalk, such as heterodimerization or shared intracellular signaling, may allow inhibition of one receptor to impair the activity of the other. Confirming this hypothesis requires further experimental investigations.
Sumatriptan-induced sensitization of CSLV neurons: The role of PKA
Pretreatment with GR127935 or H89 effectively prevented sumatriptan-induced sensitization of CSLV neurons, demonstrating activation of 5-HT1B/1D receptors and PKA plays a key role. 5-HT1B/1D receptors known to be coupled with Gi/Go proteins (61) suggests that activation of 5-HT1B/1D receptors triggers Gi/Go protein-mediated signaling, further leading to elevation of PKA levels. This postulation was supported by a study which demonstrated that hyperalgesia elicited by an intradermal injection of sumatriptan in rat hind paws was reversed by inhibiting activation of either 5-HT1B/1D receptors, Gi/Go protein, or PKA (26). However, this pathway of PKA activation mediated through the Gi/Go protein contrasts with the classical signaling pathway. Typically, Gi/Go activation decreases PKA levels through inhibition of cAMP activity (61). Consistent with this classical pathway, sumatriptan administration was found to suppress forskolin-induced cAMP activation in isolated dog saphenous veins (62) and rabbit carotid arteries (63). Interestingly, some studies using other species of cells showed inconsistent results with the above findings. For example, sumatriptan did not alter intracellular cAMP levels in rat trigeminal sensory neurons (64,65), astrocyte cells (66), or anococcygeus muscle (67). We assumed that sumatriptan-mediated PKA activation may be independent of cAMP and only exhibited in specific species and cell types. However, further studies are required to investigate this mechanism.
Dual effects of sumatriptan on hyperalgesia and analgesia
Sumatriptan demonstrates efficacy in migraine treatment; nevertheless, our findings revealed its capacity to increase the sensitivity of CSLV neurons, presenting an apparent paradox in the modulation of pain perception. This dual effect can be attributed to distinct mechanisms of sumatriptan's action on sensory neurons. The therapeutic benefits primarily arise from its ability to suppress the releases of inflammatory neuropeptides, particularly calcitonin gene-related peptides, from sensitized trigeminal nerve terminals to the dura mater (64,68). These neuropeptides typically trigger a cascade of pro-inflammatory mediator release, further resulting in the self-sustaining inflammatory cycle and hypersensitivity of sensory nerves (69,70). Sumatriptan effectively disrupts this vicious cycle by inhibiting neuropeptide synthesis and release (61,64). After excessive inflammatory mediators in the dura matter are degraded, neural sensitivity can return to the baseline. However, substantial studies revealed that before the onset of analgesic effects, sumatriptan increases the sensitivity of peripheral sensory nerves (5,26,71,72). For example, transient hyperalgesia, a common adverse effect of sumatriptan, results from sensitization of somatic nociceptors (26); this comparable effect of transient sensitization was documented in both cultured rat sensory trigeminal neurons (71) and CSLV neurons examined in our investigation. Taken together, these findings indicate that sumatriptan administration elicits a biphasic response in sensory neurons, characterized by an initial sensitization phase followed by therapeutic analgesic effects.
Limitations of this study
A major limitation of this study is the exclusive use of male rats. Clinically, both the prevalence of migraine (73) and the incidence of chest symptoms after sumatriptan use (6,7) are higher in women. In our preliminary experiments using female rats, sumatriptan did not enhance airway sensitivity, with responses showing considerable individual variability. We speculate that this sex difference may be due to fluctuations in estrogen levels during the estrous cycle. Estrogen modulates airway sensitivity in a concentration-dependent, biphasic manner. Huang et al. (74) showed that intermittent hypoxia-induced airway hypersensitivity was markedly potentiated in ovariectomized female rats, and attenuated by low-dose estrogen replacement. In contrast, replacement of high-dose estrogen, which raised plasma estrogen to levels comparable to the proestrus phase, re-enhanced airway sensitivity. In the same study, a similar biphasic effect of estrogen on the excitability of CSLV afferents was also observed (74). Therefore, a comprehensive assessment of the sensitizing effect of sumatriptan in female rats would require the use of ovariectomized rats with controlled estrogen replacement to mimic hormonal cycling. However, conducting such investigations would require a significantly larger number of animals, which is beyond the scope of the present study. Despite these limitations, whether sumatriptan alters the sensitivity of CSLV afferents in female rats remains an important and unanswered question that requires further investigation.
Clinical implications of this study
In this study, we have proposed a novel perspective based on sensory neural mechanisms to elucidate the longstanding phenomenon of sumatriptan-induced chest discomfort. However, because dyspnea is a subjective sensation that can only be verbally expressed and evaluated in conscious humans, further clinical investigations are required to validate the translational relevance of our findings from rodents to humans. Nevertheless, our findings indicated that sumatriptan sensitized CSLV afferents, vagal afferents primarily responsible for dyspnea, through its direct action on nerve endings. Furthermore, 5-HT1B/1D receptor-mediated signaling is critical for sensitizing CSLV afferents. We suggest that inhibiting activation of these receptors in the lungs could alleviate chest discomfort caused by sumatriptan. Given that the lungs can receive targeted drug delivery, airway exposure to an antagonist of 5-HT1B/1D receptors might serve as a therapeutic strategy. Localized pulmonary administration minimally interferes with oral sumatriptan for headache relief and maintains a therapeutic effect on chest side effects.
Conclusions
Sumatriptan sensitized CSLV afferents through a direct action on 5-HT1B/1D receptors expressed on nerve endings followed by PKA activation, which in turn induces airway hypersensitivity in rats. We propose that sumatriptan, an antimigraine drug, increases the sensitivity of CSLV afferents, which is potentially responsible for sumatriptan-induced chest discomfort, such as dyspnea, in migraineurs (Figure 8).

Schematic diagram illustrating the proposed mechanism underlying the involvement of sensitization of capsaicin-sensitive lung vagal (CSLV) afferents in dyspneic sensations evoked by sumatriptan. (a) Administration of the antimigraine drug sumatriptan is absorbed into the pulmonary circulation. (b) Sumatriptan acts on ① 5-hydroxytryptamine 1B and 1D (5-HT1B/1D) receptors expressed in CSLV neurons followed by ② activation of intracellular protein kinase A (PKA), which in turn causes ③ sensitization of CSLV neurons. (c) The sensitizing effect of sumatriptan on CSLV afferents further causes a non-specific increase in the excitability to various stimuli. (d) The increases in fiber discharges elicited by stimuli enhance sensory signals transmitted to the brain. (e) The brain processes and integrates these projections of sensory signals, ultimately evoking a dyspneic sensation. This diagram was created using BioRender.com.
Article highlights
Sumatriptan induces a non-specific increase in the excitability of CSLV afferents to chemical and mechanical stimuli in rats. Sumatriptan enhances capsaicin-evoked Ca2+ transients via 5-HT1B/1D receptors and PKA in CSLV neurons. 5-HT1B and 5-HT1D receptors are expressed in rat CSLV neurons. Sumatriptan sensitizes CSLV afferents, which potentially contributes to sumatriptan-induced chest discomfort, such as dyspnea, in migraineurs.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251355949 - Supplemental material for Sensitizing effect of antimigraine drug sumatriptan on capsaicin-sensitive lung vagal neurons via 5-HT1B/1D receptors and PKA: Relevance to adverse chest effects
Supplemental material, sj-docx-1-cep-10.1177_03331024251355949 for Sensitizing effect of antimigraine drug sumatriptan on capsaicin-sensitive lung vagal neurons via 5-HT1B/1D receptors and PKA: Relevance to adverse chest effects by Nai-Ju Chan, Yueh-Yin Chen, Chun-Chun Hsu and You Shuei Lin in Cephalalgia
Supplemental Material
sj-docx-2-cep-10.1177_03331024251355949 - Supplemental material for Sensitizing effect of antimigraine drug sumatriptan on capsaicin-sensitive lung vagal neurons via 5-HT1B/1D receptors and PKA: Relevance to adverse chest effects
Supplemental material, sj-docx-2-cep-10.1177_03331024251355949 for Sensitizing effect of antimigraine drug sumatriptan on capsaicin-sensitive lung vagal neurons via 5-HT1B/1D receptors and PKA: Relevance to adverse chest effects by Nai-Ju Chan, Yueh-Yin Chen, Chun-Chun Hsu and You Shuei Lin in Cephalalgia
Footnotes
Acknowledgements
We are grateful to Mr. Chamberlin Daniel Pickren for help with language editing.
Author contributions
N-JC, C-CH and YSL designed this research study. N-JC, Y-YC, C-CH and YSL conducted the experiments. N-JC and Y-YC acquired and analyzed the data. C-CH and YSL interpreted results of the experiments. N-JC, C-CH and YSL wrote the manuscript.
Funding
This study was supported by grants MOST 107-2320-B-038-049 (to YSL) and MOST 111-2628-B-038-018-MY3 (to C-CH) from the National Science and Technology Council, Taiwan.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Data availability
Data are available upon reasonable request.
Supplemental material
Supplemental materials for this article are available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
