Abstract
Through December 1998, sumatriptan had been used to treat more than 236 million migraine attacks world-wide. In clinical trials alone, more than 88 000 migraine patients had treated more than 300 000 migraine attacks with sumatriptan, and 2000 normal healthy volunteers had been exposed to the drug. This paper describes the safety and tolerability profile of sumatriptan in three sections: adverse events reported in clinical trials, special issues, and spontaneous post-marketing reports of adverse reactions. Data from the extensive clinical trials programme coupled with information from nearly 10 years of experience in clinical practice demonstrate that sumatriptan is generally well-tolerated, with an acceptable benefit–risk ratio when used properly. Significant cardiovascular and cerebrovascular events are rare but have been observed. This fact highlights the need for careful patient selection and vigilant adherence to the prescribing recommendations for sumatriptan. The wealth of clinical trials and post-marketing information for sumatriptan may be useful in guiding prescribing decisions for members of this class of drugs.
Keywords
Introduction
No migraine therapy has been studied as extensively and rigorously as sumatriptan. In clinical trials alone, through December 1998, more than 88 000 migraine patients had treated more than 300 000 migraine attacks with sumatriptan, and 2000 normal healthy volunteers had been exposed to the drug. In addition, post-marketing and clinical practice data on sumatriptan, available in more than 80 countries, now extends to millions of migraineurs.
This paper assesses the safety and tolerability profile of sumatriptan. Since the last review in 1994 (1), other selective 5HT1 agonists have become available for the acute treatment of migraine. The wealth of clinical trials and post-marketing information for sumatriptan may be useful in guiding prescribing decisions for members of this class of drugs. The tolerability of sumatriptan is considered in sections on adverse events reported in clinical trials, special issues and spontaneous post-marketing reports of adverse reactions.
Adverse events
Clinical trials, patient population and methods
This section of the paper describes adverse event data from clinical trials conducted by the sumatriptan manufacturer (Glaxo Wellcome). Obtained from consistently designed trials meeting rigorous regulatory standards, these data were considered to provide a comprehensive and accurate picture of side-effects arising during use of sumatriptan in the clinical trials setting. A review of the literature to identify case reports of side-effects arising during normal clinical practice was not undertaken; any adverse event reported as a case report is recorded in the post-marketing surveillance safety database; these data are addressed in the section ‘Data from post-marketing spontaneous reports’ below.
In all clinical trials, adverse events were collected using the prospective definition of any medical or clinical change (occurring or worsening post-dose) noted by the patient on diary cards or reported to or observed by the clinician during a clinical trial. In evaluating the safety of a product, it is often difficult to separate treatment-related events from coincidental events. The strategy, then, is to record all adverse events regardless of their suspected origin or cause—including features of the migraine itself (e.g. nausea, headache or postdromal symptoms). In the controlled trials database, these adverse event frequencies can be compared between active treatment and placebo groups. The placebo-associated adverse events can be used as an estimate of the spontaneous or coincidental adverse events. The clinical significance of any excess frequency of adverse events associated with sumatriptan can be determined by comparison of these two groups.
The majority of patients (> 75%) enrolled in the sumatriptan clinical trials were women, a demographic consistent with that of the general migraine population. More than 85% of patients were Caucasian. Most patients suffered migraine without aura, although patients suffering from migraine with aura and from both migraine types were included.
Adverse events during short-term clinical trials
Adverse events reported more frequently for sumatriptan than placebo and by ≥ 3% of patients in the active treatment group (highest dose administered) in short-term (usually single-attack), controlled clinical trials are listed in Table 1. Most (> 80%) adverse events were mild or moderate, and most (∼70% to 85% depending on formulation) lasted 1–3 h or less.
Adverse events∗ in sumatriptan injection, tablets and nasal spray clinical trials

Events that occurred at least 1% more often in the active treatment group (highest dose administered) than the placebo group and that occurred in > 3% of patients in the active treatment group (highest dose administered).
Adverse events during long-term clinical trials
Sumatriptan injection and tablets have been used for migraine attacks for up to 2 years (2, 3) and sumatriptan nasal spray for up to 1 year (3) in four open-label studies. Two subcutaneous 6 mg doses (n = 412 patients in two studies), three 100 mg oral doses (n = 275 patients), or two 20 mg intranasal doses (n = 182 patients) could be used to treat each migraine attack experienced during the study periods. (Sumatriptan prescribing information indicates that the daily dose of sumatriptan tablets should not exceed 200 mg) The results suggest that the type, incidence and frequency of adverse events with each of the sumatriptan formulations in long-term studies are consistent with data from single-attack studies. The most frequently reported adverse events in three representative long-term trials with sumatriptan injection, tablets and nasal spray are listed in Table 2.
Adverse events most frequently reported in long-term trials∗

Adverse events reported in ≥ 2% of attacks are listed.
One indication of adverse event severity is whether the adverse event precipitates the patient's withdrawal from the study, either at the patient's own wish or by the investigator. Few patients (12% with injection, 7% with tablet, 4% with nasal spray) withdrew from the studies because of adverse events.
Special issues
Triptan sensations
Although the most common adverse events varied according to sumatriptan formulation, some events were frequently reported with multiple formulations. Warm/hot sensation, tightness, tingling, flushing, and feelings of heaviness or pressure, were experienced in areas such as the face, limbs and, in 1% to 5% of patients, the chest (1), with all sumatriptan formulations (although less frequently with the tablets and the nasal spray). Approximately half the patients reporting symptoms such as tightness or feelings of heaviness in the chest in sumatriptan clinical trials also experienced these sensations in other body areas. These events have been designated ‘triptan sensations’ because they appear to occur with all 5HT1‘triptan’ agonists developed to date (4).
Chest pain associated with the use of sumatriptan has been observed in clinical use (5–12) as well as in clinical trials, and there are isolated reports from clinical practice of myocardial infarction associated with the use of sumatriptan. With rare exceptions (5), these incidents of chest pain lack temporal, electrocardiographic, angiographic and enzymatic evidence of myocardial infarction. Therefore, there is not sufficient evidence to indicate whether or not the incidents of chest pain signify myocardial ischaemia. In post-marketing use involving more than 9 million patient exposures to, and more than 236 million attacks treated with, sumatriptan, events such as myocardial infarction are extremely rare. However, the fact that such events have been observed highlights the importance of careful adherence to standard medical practice and the sumatriptan prescribing information, which prohibits use of the medication in patients with history, symptoms or signs of ischaemic cardiac, cerebrovascular or peripheral vascular syndromes and recommends careful screening of patients for cardiovascular risk factors such that satisfactory clinical evidence of freedom from cardiovascular disease is confirmed.
The mechanism of chest symptoms and other triptan-associated sensations has not been determined to date. It has been hypothesized that migraine patients may suffer from a generalized vasospastic disorder, the coronary manifestation of which is variant angina and the cerebral vascular manifestation of which is migraine headache (13). Oesophageal or pulmonary mechanisms have also been hypothesized to underlie the chest symptoms sometimes reported with sumatriptan and other 5HT1 agonists (14, 15).
Cardiovascular safety of sumatriptan
In order to evaluate the possibility that a vascular mechanism underlies sumatriptan-associated chest symptoms, the cardiovascular safety of sumatriptan was investigated using a variety of approaches and study methods.
In vitro effects on human coronary arteries
The contractile effects of serotonin on large coronary arteries appears to be mediated predominantly by 5HT2 receptors and to a lesser extent by 5HT1 or 5HT1-like receptors (16–19). Sumatriptan, even at relatively high doses, has little or no effect on 5HT2 receptors. Results from in vitro studies on human coronary arteries reveal that sumatriptan causes small contractions via activation of a small population of 5HT1B/1D receptors in these vessels (16). The maximum sumatriptan-induced arterial contraction is equivalent to only 21% of the maximum contraction caused by serotonin, with only very small effects produced at therapeutically relevant concentrations of sumatriptan. Serotonin-induced contractions were blocked by the 5HT2 antagonist ketanserin, but sumatriptan-induced contractions were not.
Sumatriptan and the newer triptans, naratriptan, zolmitriptan and rizatriptan were similarly effective and potent at contracting human isolated coronary arteries in one study (20). The plasma EC50/Cmax ratios for each triptan after a therapeutic dose were similar to one another. The Cmax for all was less than 40% of that required to elicit 50% of the maximum contractile response of the human isolated coronary artery. The clinical significance of these findings is unknown.
Electrocardiograms (ECGs)
More than 8000 patients in placebo-controlled studies with sumatriptan injection, tablets and nasal spray provided baseline and post-treatment 12-lead ECGs (used as a screening tool to exclude patients with ischaemic heart disease or clinically relevant ECG abnormalities). Holter monitoring was not routinely performed in clinical trials. The post-treatment incidence of possible angina (with or without ECG confirmation) or of ECG changes possibly indicative of ischaemia among these patients was consistently low for all of the sumatriptan formulations. Eleven patients exhibited potentially ischaemic ECG changes after sumatriptan injection (six women aged 21–45 years), sumatriptan tablets (one man aged 45 years), and sumatriptan nasal spray (one man aged 39 and three women aged 37–45 years). Of these 11 patients, nine (82%) did not experience other signs or symptoms consistent with ischaemia. Because 12-lead ECGs are an insensitive measure of myocardial perfusion, these data should not be interpreted to suggest that coronary constriction occurred only in patients with potentially ischaemic ECG changes.
ECGs were also evaluated in a re-challenge study conducted among 20 patients who had previously reported chest symptoms after administration of oral, intravenous or subcutaneous sumatriptan (3). (Intravenous use of sumatriptan is contraindicated.) These patients were challenged outside a migraine attack with placebo injection followed 1 h later by 6 mg sumatriptan injection. Chest pressure was reported by two patients after both placebo and sumatriptan and by one patient after sumatriptan only. No ECG abnormalities were noted in these patients. Considered together, the data from these studies suggest that the chest symptoms after sumatriptan rarely are linked to ischaemic ECG changes.
Angiography studies
Studies in patients undergoing diagnostic coronary angiography were also conducted to evaluate the cardiovascular safety of sumatriptan. Because these studies were not conducted in migraine patients during a migraine attack, the results of these studies should be extrapolated to migraine patients cautiously. Two studies (21, 22) in patients without migraine (n = 20) undergoing diagnostic coronary angiography showed clinically insignificant reductions in mean coronary artery diameter unaccompanied by ECG changes or anginal symptoms after intravenous or subcutaneous doses of sumatriptan (Table 3). No significant changes in ECG recordings and no chest symptoms were reported in either study. The authors concluded that although a vasoconstrictor effect was observed in the coronary arteries after administration of sumatriptan, the ‘changes were brief and not associated with clinical symptoms nor objective evidence of myocardial ischaemia’ (21).
Sumatriptan effects on coronary artery diameter in patients undergoing coronary angiography

Dose producing peak plasma levels more than twice as high as those associated with administration of sumatriptan injection 6 mg in clinical use in migraine. Note that intravenous administration of sumatriptan is contraindicated.
Both of these studies excluded patients found to have coronary artery stenosis ≥ 50%. There is evidence to suggest that coronary arteries of patients with ≥ 50% stenosis in one or more arteries respond atypically to serotonin receptor stimulation (23, 24). To evaluate the coronary artery effects of sumatriptan among patients with known coronary artery disease, sumatriptan injection (6 mg; n = 11) or placebo (n = 5) was administered subcutaneously to patients with > 50% stenosis in at least one vessel segment in a parallel-group study (3). The results show no statistically significant differences between the sumatriptan and placebo groups with respect to per cent change from baseline at 15 min or 30 min post-dose in mean, minimum or maximum diameters in either stenosed or non-stenosed segments (Table 4).
Mean per cent change from baseline in mean, minimum and maximum diameters in non-stenosed and stenosed segments
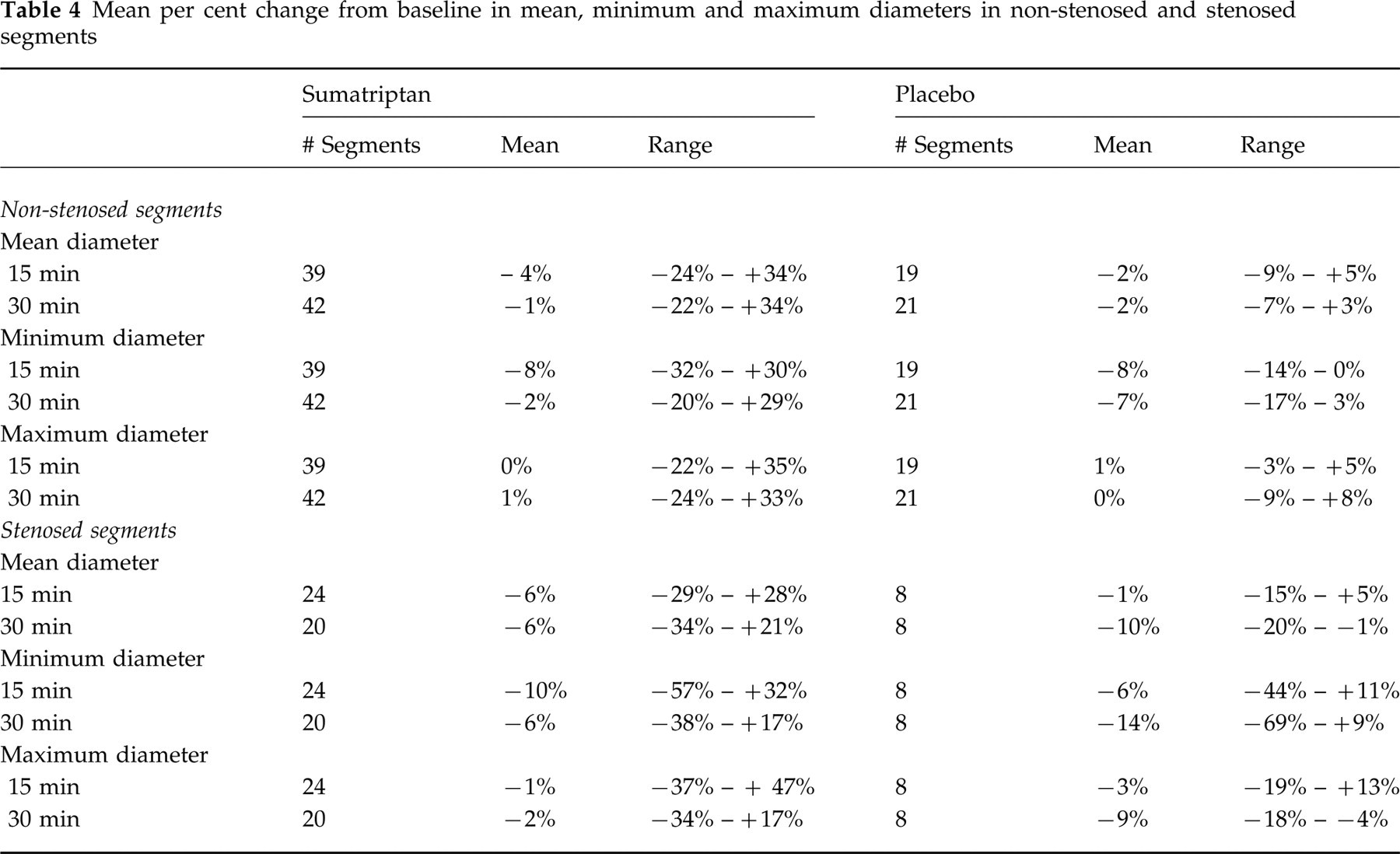
The mean of the maximum reductions in minimum lumen diameter was 38% (
Myocardial perfusion assessed using positron emission tomography (PET)
The effects of sumatriptan injection 6 mg compared with placebo on cardiovascular functioning have been assessed using PET in a double-blind, placebo-controlled, crossover study (25) in 19 female migraineurs (median age 48 years) whose baseline PET scans were normal with no evidence of coronary artery disease. The results demonstrate no statistically or clinically significant effect of sumatriptan injection 6 mg compared with placebo on global or regional myocardial perfusion. The mean percentage change from baseline (±
Summary: cardiovascular safety of sumatriptan
These data from studies evaluating the cardiovascular safety of sumatriptan do not support the contention that chest symptoms among patients with no underlying coronary artery disease were routinely due to ischaemic mechanisms. Nonetheless, serious cardiac events including acute myocardial infarction, life-threatening disturbances of cardiac rhythm, and death, have been reported within a few hours following the administration of sumatriptan. Considering the extent of use of sumatriptan in patients with migraine, the incidence of these events is extremely low. The fact that sumatriptan can cause coronary vasospasm, that some of these events have occurred in patients with no prior cardiac disease history and with documented absence of coronary artery disease, and the close proximity of the events to sumatriptan use, support the conclusion that some of these cases were caused by the drug. In many cases, however, where there has been known underlying coronary artery disease, the relationship is uncertain. The prescribing information for sumatriptan indicates that the drug should not be given to patients with history, symptoms or signs of ischaemic cardiac, cerebrovascular or peripheral vascular syndromes. It is strongly recommended that sumatriptan not be given to patients in whom unrecognized coronary artery disease is predicted by the presence of risk factors (e.g. hypertension, hypercholesterolemia, smoker, diabetes, strong family history) unless a cardiovascular evaluation provides satisfactory clinical evidence that the patient is reasonably free of coronary artery and ischaemic myocardial disease or other significant underlying cardiovascular disease.
Neurological safety of sumatriptan
Sumatriptan has been hypothesized to relieve the symptoms of migraine by stimulating 5HT1 receptors on cranial blood vessels. In an in vitro study (26), sumatriptan dose-dependently increased the perfusion pressure in the perfused human dura mater — presumably by activating the cranial vascular 5HT1 receptor. This vasoconstrictor effect of sumatriptan occurs at large conducting cranial arteries; cerebral blood flow studies reveal that sumatriptan exerts little or no effect on resistance vessels. Sumatriptan-associated effects (if any) on regional cerebral blood flow (rCBF) were small and inconsistent. Two studies (27, 28) demonstrated no effect of sumatriptan on rCBF in healthy subjects or migraineurs. One study (29) demonstrated both small increases and small decreases in rCBF associated with sumatriptan administration. (Migraine itself provokes both decreases and increases in rCBF (30, 31).)
Although sumatriptan is active at cranial vessels, it is unlikely to exert significant central nervous system side-effects due to its minimal penetration of the blood–brain barrier (though during migraine attacks, blood–brain barrier permeability may be increased and sumatriptan may penetrate more easily) (26). In fact, sumatriptan (100 mg to 200 mg) administered orally did not alter psychomotor performance on tests for saccadic eye movements, alertness and attention measured with visual analogue scales and Tiplady continuous attention testing, or cognitive function assessed with choice recognition tasks (15). In another study (32), sumatriptan injection 8 mg and 16 mg were discriminated from placebo in 12 male subjects with a history of substance abuse. Sumatriptan injection decreased euphoria scores and increased scores on measures of disliking in these subjects, but it did not produce feelings or responses characteristic of drugs of abuse. Morphine, in contrast, was dose-dependently reinforcing. (Note that 6 mg is the marketed dose of sumatriptan injection.)
Adverse events that could be of neurological origin (e.g. tingling, numbness, weakness and dizziness) have been reported after administration of sumatriptan in clinical trials. Many of these events may be related to the migraine attack rather than to administration of sumatriptan. Some of the neurological events may be manifestations of aura.
The frequent occurrence of neurological symptoms with migraine highlights the need for careful differential diagnosis of headache. Some of the neurological features of migraine with or without aura may occur with other migraine subtypes such as hemiplegic migraine, for which sumatriptan is contraindicated. Furthermore, headache and other neurological symptoms are typical in cerebral infarction or subarachnoid haemorrhage.
Cerebrovascular disease and migraine may exist in some patients. Migraineurs compared with individuals who do not suffer from migraine have three to six times the risk of spontaneous stroke (33, 34). For example, Tzourio and colleagues (34) found that ischaemic stroke among women under 45 was strongly associated with both migraine without aura (odds ratio 3.0, 95% confidence interval 1.5–5.8) and migraine with aura (odds ratio 6.2, 95% confidence interval 2.1–18.0). The basis of the increased risk of stroke among migraineurs remains to be determined.
Data from post-marketing spontaneous reports
Sumatriptan is unique among the 5HT1 agonist migraine medications in the extent to which it has been used in clinical practice. Through December 1998, sumatriptan had been used to treat more than 236 million migraine attacks in world-wide clinical practice involving more than 9 million patient exposures. (Note that these numbers represent best estimates based on prescription data. Because patients may have been prescribed sumatriptan but not taken it, the numbers may overestimate actual use of sumatriptan.) Spontaneous reports of adverse events obtained during the extensive post-marketing use of sumatriptan provide a rich source of data from clinical practice to complement the clinical trials data and allow continuing assessment of sumatriptan's safety profile. This section reviews spontaneous reports through December 1998 of adverse events occurring after administration of sumatriptan injection, tablets and nasal spray.
Spontaneous reports of adverse events are voluntary and come to the pharmaceutical manufacturer from multiple sources (e.g. medical literature, clinical practice, post-marketing studies). Events are reported spontaneously from world-wide post-marketing experience, and the role of sumatriptan in their causation cannot always be reliably determined. Nearly all types of adverse events occur at a background frequency regardless of whether a medical treatment is given. Other disadvantages of using post-marketing experience compared with clinical trials include incomplete reporting of adverse events, underreporting of adverse events, the lack of a control group, and the inability to follow up on the short- and long-term outcomes of adverse events. Attempts to determine the incidence of post-marketing events (numerator = number of events occurring; denominator = number of exposures to a medicine) are severely limited due to both underreporting, which results in the numerator's representing only a subset of the events that have actually occurred, and to absence of information about population exposure to a product, which results in an inaccurate denominator.
Serious cardiac events
The occurrence of serious cardiac events in post-marketing experience is of particular interest. Serious cardiovascular events were defined for the purposes of this summary as myocardial ischaemia, angina/cardiac pain, increased enzymes, any ST segment change, any serious dysrhythmia, cardiovascular test abnormalities, cardiac arrest or asystole, ventricular tachycardia or fibrillation, and vasospasm. The approximate numbers of serious cardiac events within 24 h of administration of sumatriptan injection or tablets between 1992 and 1998 and sumatriptan nasal spray between 1996 (the year it was introduced) and 1998 are shown in Fig. 1. These numbers can only be approximated because the review of spontaneous adverse events is a subjective process. Between 1992 and December 1998, approximately 451 serious cardiac adverse events within 24 h were spontaneously reported after administration of sumatriptan injection, tablets or nasal spray for more than 236 million migraine attacks and more than 9 million patient exposures.
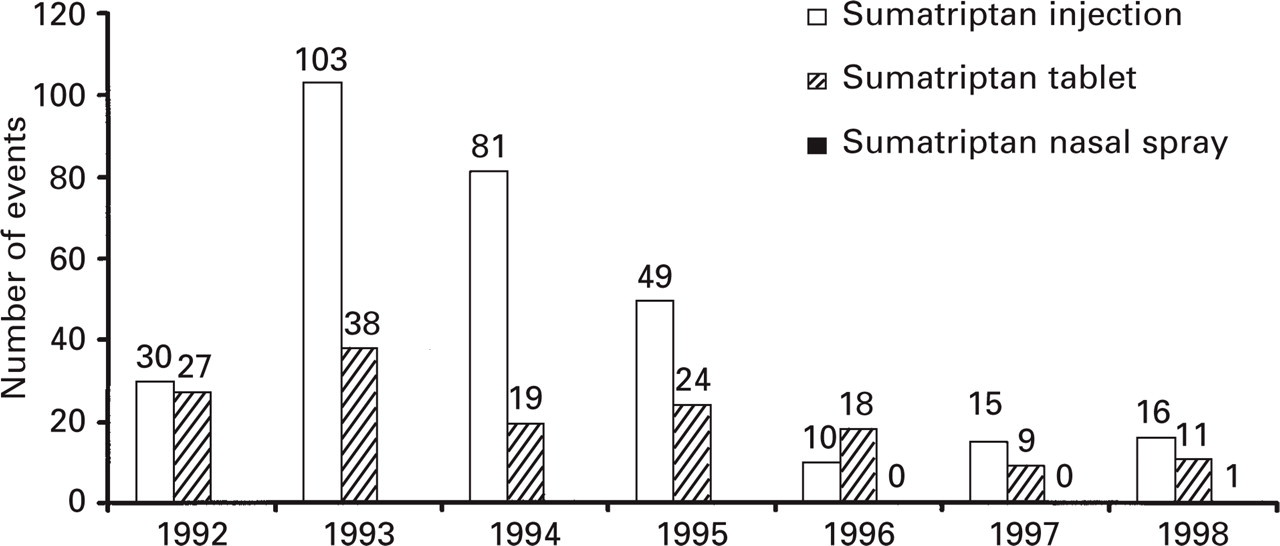
Serious cardiovascular adverse events occurring within 24 h of administration of sumatriptan injection, tablet or nasal spray. Reported through December 1998.
Sumatriptan has a half-life (t1/2) of approximately 90 min. Almost invariably, patients with serious cardiac events within 1–3 h of sumatriptan administration had risk factors predictive of coronary artery disease, and the presence of significant underlying coronary artery disease was established in most cases.
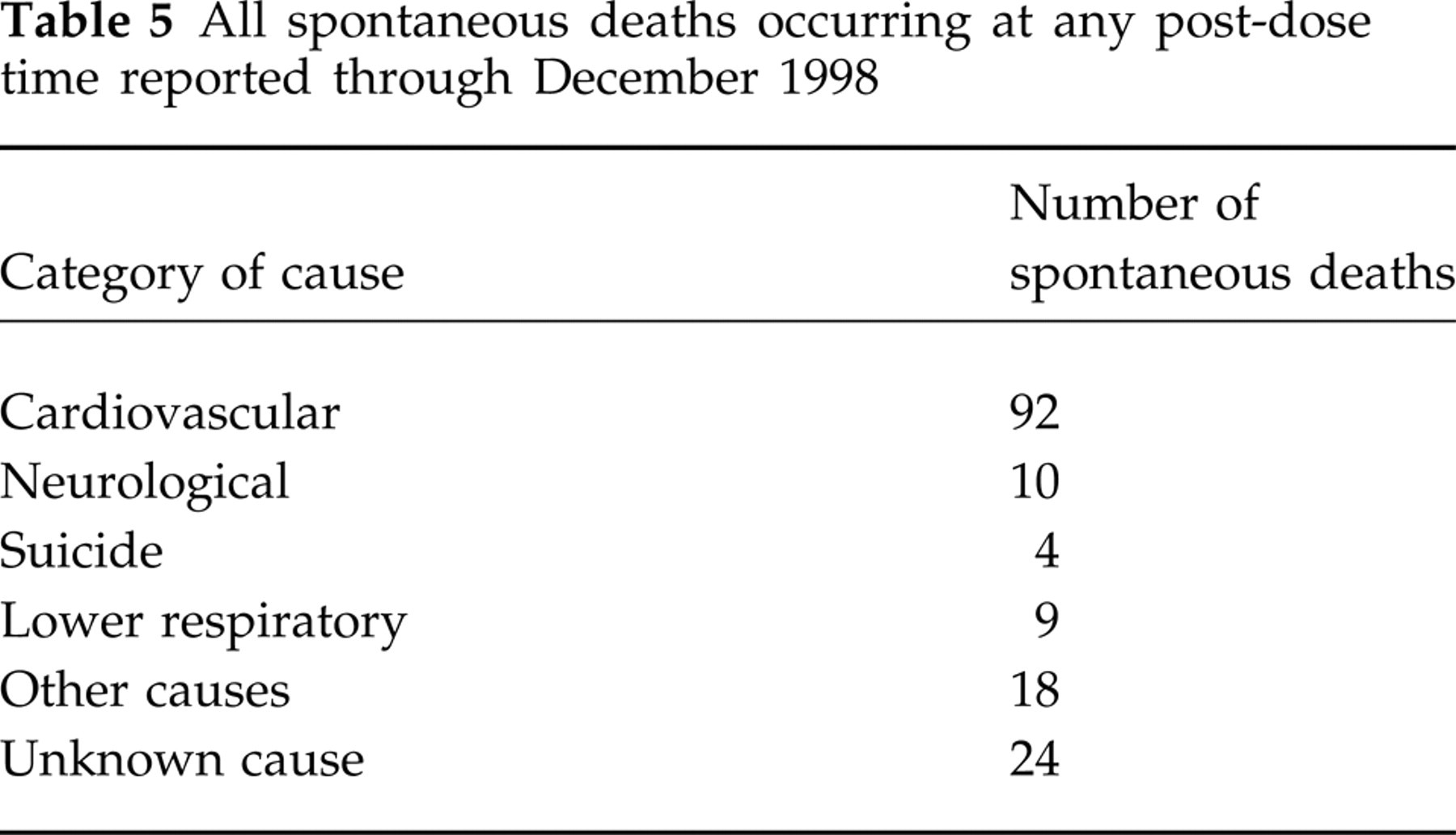
Spontaneous deaths
All spontaneous deaths reported at any time after sumatriptan use through December 1998 are listed in Table 5. Approximately 92 cardiac fatalities (Fig. 2) and 10 neurological fatalities (Fig. 3) were reported at any post-dose time after administration of sumatriptan injection, tablets or nasal spray in 9 million patient exposures for more than 236 million attacks.
All spontaneous deaths occurring at any post-dose time reported through December 1998

Spontaneous cardiac deaths occurring at any post-dose time after sumatriptan injection, tablet or nasal spray. Reported through December 1998.

Spontaneous neurological deaths occurring at any post-dose time after sumatriptan injection, tablet or nasal spray. Reported through December 1998.
Although the incidence of cerebrovascular fatalities after sumatriptan administration is not high, cerebral haemorrhage, subarachnoid haemorrhage, stroke and other cerebrovascular events have been reported in patients treated with sumatriptan injection or tablets. The relationship of sumatriptan to these events is uncertain. In a number of cases it appears possible that the cerebrovascular events were primary, sumatriptan having been administered in the incorrect belief that the symptoms experienced were a consequence of migraine when they were not. As mentioned, the risk of stroke is higher in individuals suffering from migraine compared with individuals who do not suffer from migraine (33, 34).
Conclusions
Data from the extensive clinical trials programme coupled with information from nearly 10 years of experience in clinical practice demonstrate that sumatriptan is generally well-tolerated with an acceptable benefit–risk ratio when used properly. Significant cardiovascular and neurological events are rare but have been observed. This fact highlights the need for careful patient selection and vigilant adherence to the prescribing recommendations for sumatriptan. The prescribing information for sumatriptan states that because of rare reports of coronary vasospasm, sumatriptan should not be given to patients with history, symptoms or signs of ischaemic cardiac, cerebrovascular, or peripheral vascular syndromes. In addition, patients with other significant underlying cardiovascular diseases should not receive sumatriptan. Sumatriptan should also not be administered if the headache is atypical for the patient. Prescribing behaviour with the newer 5HT1 agonists, which as a class have the potential to share many of the effects of sumatriptan, should be influenced by these sumatriptan data gleaned from years of clinical trials and clinical practice experience.
Acknowlegement
Data described in this manuscript were previously presented at the American Association for the Study of Headache meeting (New York, New York, USA, June 1997), the World Congress of Neurology meeting (Buenos Aires, Argentina, September 1997), and the European Federation of Neurological Societies meeting (Lisbon, Portugal, September 1999).
