Abstract
Objective
To assess the presence and handicap due to vestibular symptoms in three subgroups of patients with migraine and controls.
Methods
Women between 18–55 years old were diagnosed by headache specialists and stratified as migraine with aura (n = 60), migraine without aura (n = 60), chronic migraine (n = 60) and controls (n = 60). Information regarding demographics, headache and vestibular symptoms were collected in this cross-sectional study. The self-perceived handicap related to vestibular symptoms was assessed through the Dizziness Handicap Inventory questionnaire.
Results
A total of 85% of women with migraine with aura and chronic migraine had vestibular symptoms contrasted to 70% of the migraine without aura group (
Conclusion
The prevalence of vestibular symptoms is increased in migraine during and between headache attacks, particularly in migraine with aura and chronic migraine along with an increased handicap due to those symptoms. Vestibular symptoms among subgroups of migraine should be considered when evaluating the functional impact of migraine.
Introduction
Headache and the presence of vestibular symptoms are commonly associated in the clinical practice and may coexist in different ways (1,2). The understanding of the relationship between migraine and vestibular symptoms is considered a challenging issue, since vestibular symptoms can manifest as an inherent migraine feature (3), or a vestibular disease can coexist with migraine (1,3–5). The vestibular migraine criteria have been included in the appendix of the International Classification of Headache Disorders (ICHD 3rd edition beta), indicating that further investigation should be done in order to consolidate it as a valid diagnosis (6).
Besides the variety of clinical presentations of vestibular symptoms in patients with migraine, including its occurrence in patients with brainstem aura, there is no confirmatory test for the diagnosis of vestibular migraine (5). Up to 58% of patients with migraine present some evidence of vestibular dysfunction during otoneurological tests, regardless of the complaint of clinical symptoms (2,7–9). For that reason, the diagnosis of vestibular migraine is made on the basis of the patients’ clinical history, and not by vestibular examination (2).
Moreover, the spectrum of symptoms in vestibular migraine is broad (10) and studies assessing the influence of migraine chronicity on vestibular symptoms are scarce (6). However, it is established that patients with chronic migraine present a greater number of comorbidities, including vertigo (11). Therefore, the investigation of vestibular symptoms and their impact on patients with migraine is very important to the understanding and management of this condition, considering the singularity of each migraine subtype.
The aim of this study was to investigate the association between migraine and vestibular symptoms in patients with aura, without aura and chronic migraine, including the self-perceived handicap due to dizziness among those patients. We hypothesized that migraine overall, as well as aura and migraine frequency, would correlate with the presence of and impact due to vestibular symptoms.
Methods
Consecutive patients diagnosed with migraine were screened from a tertiary headache clinic between February 2014 to March 2015 for this cross-sectional study. The diagnoses were made by headache specialists according to the International Classification of Headache Disorders ICHD – 3rd edition, and they were classified as: Migraine with aura group (MA), migraine without aura group (MoA) and chronic migraine group (CM) (6). Patients were diagnosed with chronic migraine if they had at least 15 days with headache within a month in the last three months (and at least eight of them fulfilled the migraine criteria). Patients with less than 15 days with headache in the last three months were diagnosed with migraine with or without aura. Vestibular migraine criteria were not considered to characterize the patients. Non-headache subjects were identified among patients’ family members and hospital employees in order to compose the control group (CG).
Female subjects were included in the sample when they were between the ages of 18–55 years old. Specifically for migraine patients, inclusion criteria comprised migraine diagnosis in the previous 1 year and presence of at least three attacks within a month during the last 3 months. The exclusion criteria adopted for all subjects were: Diagnosis of any concomitant headache (a); use of any medication prescribed for vertigo or dizziness treatment such as meclizine, flunarizine, cinnarizine, betahistine, and/or benzodiazepinics (b); self-report or history of vestibular disease (c); abnormal neurological exam (d); any musculoskeletal impairment that could affect balance (e); presence of a migraine attack during the interview (f); pregnancy (g) and any systemic disease with rheumatic, cardiovascular, neurologic or metabolic etiology (h).
A structured questionnaire was used to interview the participants regarding the following aspects: Demographics, migraine onset, pain intensity (numeric pain rating scale (NPRS)), attack frequency (number of days in the prior month) and duration (hours), and presence and description of vestibular symptoms, during and between migraine attacks. Patients that reported vestibular symptoms prior to migraine onset, which were present only between migraine attacks, or with chronic dizziness, were also excluded. Primary vestibular symptoms including vertigo (sensation of self-motion), dizziness (disturbing spatial orientation), vestibulovisual (false sensation of motion or tilting) or postural symptoms (symptoms related to balance and to postural stability were classified according to the Bárány Society’s Classification of Vestibular Symptoms (12) and patients who referred multiple symptoms were included in more than one classification).
Furthermore, a senior headache specialist administered the Dizziness Handicap Inventory (DHI) questionnaire in order to assess the self-perceived impairment related to the vestibular symptoms. This questionnaire was applied just for subjects with any report of vestibular symptoms. The DHI has 25 questions encompassing physical (seven questions, 0–28 points), functional (nine questions, 0–36 points) and emotional aspects (nine questions, 0–36 points), with total score ranging from 0 to 100 points (0 = no, 2 = sometimes, 4 = yes). Total scores are classified as a mild handicap (0–30 points), moderate handicap (31–60 points) or severe handicap (61–100 points) (13). This tool provides a useful, reliable and valid measure of self-perceived handicap associated with dizziness (14,15), considering the total score as well as the physical, functional and emotional domains (16,17). This questionnaire is correlated with functional measures of gait and balance (18–20).
The study was performed in accordance with the Helsinki Declaration and with the approval of the Local Ethics Committee (protocol number 16693/2012). Written informed consent was obtained from all participants.
Based on a pilot study, the sample size was calculated to provide a power of 95% to the selected α level of 5% to detect 13 DHI points of difference between any migraine group and controls with a standard deviation (SD) of 10.6. The sample was calculated as being 55 subjects for each group, therefore 60 were included to account for imprecision in the pilot study.
Demographic data of the four groups were normally distributed (Kolmogorov-Smirnov test NS) and then were compared using a two-way ANOVA with Bonferroni’s
Results
Among 320 potential patients, 28 were excluded due to the presence of systemic diseases such as fibromyalgia, diabetes, non-controlled hypertension, stroke and idiopathic intracranial hypertension. Up to 17 subjects were excluded due to concomitant headaches such as post-traumatic headache, tension-type headache or medication-overuse headache. Up to 15 subjects presented musculoskeletal impairments such as knee osteoarthritis, injury of knee or ankle ligaments, and prior knee surgery. Up to 11 subjects had fewer than three migraine attacks per month and nine reported histories of vestibular diseases including labyrinthitis and benign paroxymal positional vertigo (BPPV). Therefore, 240 subjects were included in the study, equally distributed among the four groups (MoA, n = 60; MA, n = 60; CM, n = 60; CG, n = 60).
Average and standard deviation (SD) of demographic data among subjects with migraine with aura (MA) and without aura (MoA), chronic migraine (CM) and controls (CG).

Presence and classification of vestibular symptoms among subjects with migraine with aura (MA) and without aura (MoA), chronic migraine (CM), and controls (CG).

For cells with n < 5, Fisher’s exact test was performed.
Average (95% CI) of Dizziness Handicap Inventory (DHI) scores and handicap frequency among subjects with migraine with (MA) and without aura (MoA), chronic migraine (CM) and controls (CG).
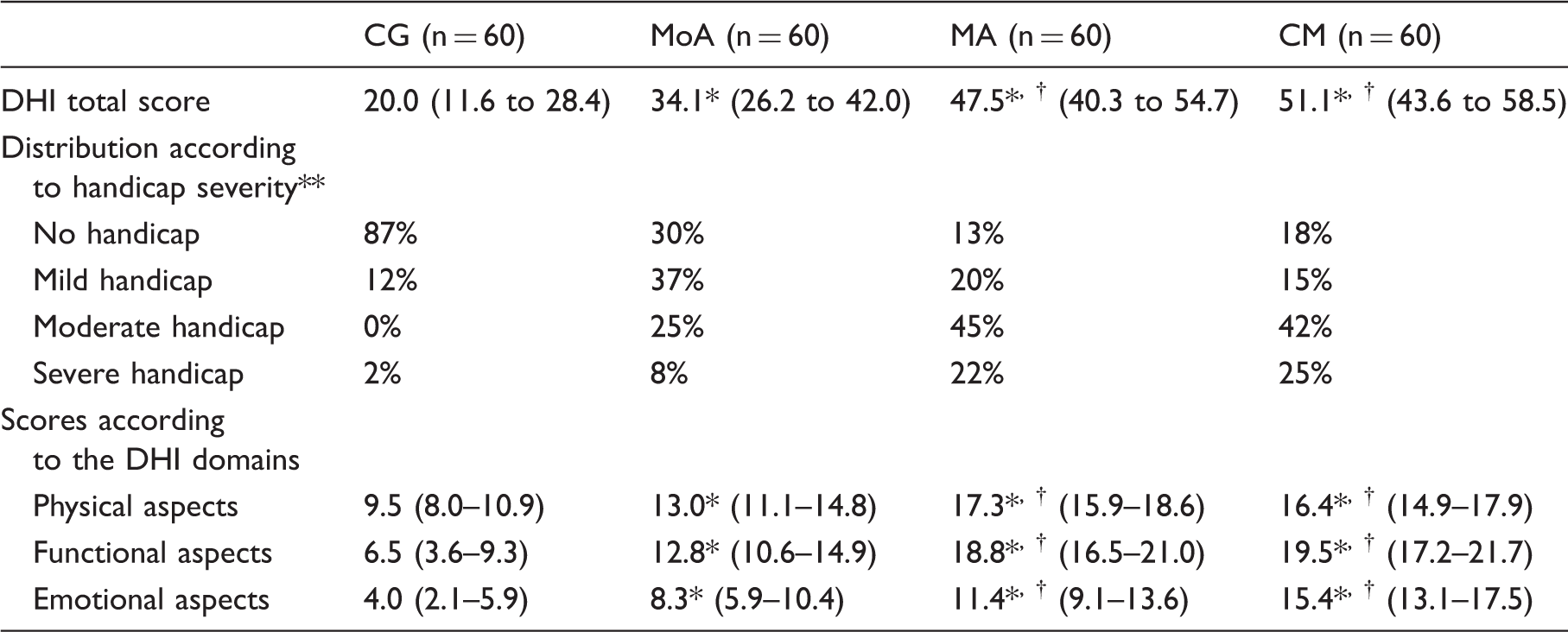
Regarding the DHI domains, patients with MA and CM reached higher scores than MoA and CG considering the physical, functional and emotional scores ( Physical, functional and emotional domains of the Dizziness Handicap Inventory (DHI) among patients with migraine without aura (MoA), with aura (MA), chronic migraine (CM) and controls (CG). *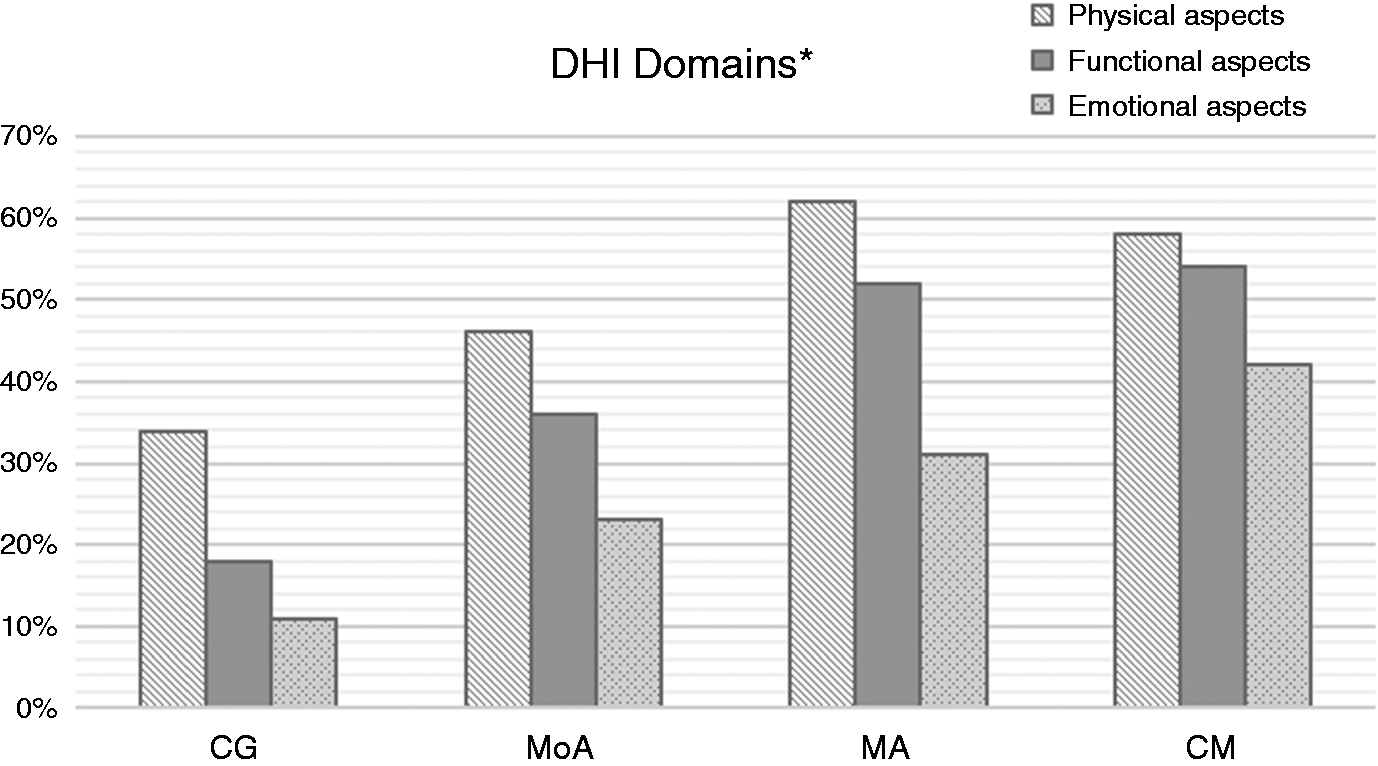
Prevalence ratios of the presence of any kind of vestibular symptoms and Dizziness Handicap (DHI) among subjects with migraine using controls and migraine without aura groups as references (ref.).

Ref.: Prevalence ratio reference. *
GC: control group; MoA: migraine without aura group; MA: migraine with aura group; CM: chronic migraine group.
The presence of migraine is associated with a greater risk of any handicap due to dizziness (MoA: 5.25, MA: 6.50 and CM: 6.12,
Linear regression models explaining the variability of the DHI scores by migraine and demographic variables*.

Dependent variable: Dizziness Handicap Inventory scores (DHI).
Independent variables: Age, migraine aura, frequency, intensity and onset.
Discussion
Our findings support the possibility of a link between migraine and vestibular symptoms, with a greater frequency when patients exhibit aura or chronic migraine. The handicap severity is higher in both chronic and aura groups, despite the presence of some level of handicap in all migraineurs compared to controls. The presence of migraine, especially with aura and chronic, is associated with the risk of vestibular symptoms, mainly vertigo, and of any handicap or moderate-to-severe handicap related to vestibular symptoms. The dizziness handicap level is influenced by the presence of aura, by migraine frequency and intensity.
Previous studies have found a prevalence of dizziness or vertigo ranging from 12% to 52% among patients with migraine (2,21,22) reaching up to 62% in women (21). Lower prevalence rates are usually related to the diagnosis of migraine without aura, while higher prevalence is verified when migraine aura is present (2,22). Furthermore, the prevalence of headache among patients reporting dizziness and vertigo is around 35% (23).
The prevalence found in the present study of 70–85% refers to all categories of vestibular symptoms described by the Bárány Society’s Classification of Vestibular Symptoms, including the postural symptoms (12), which was not accounted for in the above-mentioned studies. Since the patients may refer to more than one category of vestibular symptom, we verified a point-prevalence of dizziness ranging from 3–11% and of vertigo from 26–54% among patients with migraine without aura, with aura and chronic migraine. These results are according to the previous studies, and also highlight the greater prevalence in patients with aura.
Considering the broad variability of the confidence intervals found, we can highlight that the presence of migraine is 5.20 to 6.60-fold associated to the risk of presenting vestibular symptoms, and patients with aura and greater migraine frequency are more likely to present vertigo than controls. On the other hand, patients with migraine without aura are more likely to report postural symptoms.
An interesting aspect verified in our sample was the similar prevalence of vestibular symptoms among patients with aura and chronic migraine. The chronicity of migraine frequency is not accounted for in the proposed vestibular migraine criteria (6) and it is poorly explored by the studies on the topic (5,10,21,22,24–28). This could be due to the challenge of establishing a temporal relationship between migraine and other conditions, such as vestibular symptoms (21), or to the modification of migraine features along with the increment of attacks (>15 within a month) (29). However, Calhoun et al. (2) reported the additional presence of dizziness and vertigo with increasing age, suggesting an association with a greater lifetime burden of illness or with migraine chronicity.
Cho et al. (30) reported a concomitant diagnosis of chronic migraine among 29% of patients who fulfilled the vestibular migraine criteria. We can speculate that a part of our sample would meet the vestibular migraine criteria, since 26% of MoA, and 54% of MA and CM, reported vertigo during migraine attacks. However, according to the proposed criteria for vestibular migraine, at least 50% of the vestibular symptoms should be associated with migrainous features (6). If patients with chronic migraine exhibit vestibular symptoms just on the headache days that do not fulfill the migraine features, they would not meet the proposed criteria.
The etiology of vestibular symptoms in patients with migraine could be related to the overlap between the trigeminal and vestibular pathways (26), suggesting a concomitant activation of vestibular and cranial nociceptive afferents (25). The vascular, neurogenic inflammation and neural mechanisms involved in migraine pathophysiology are also verified in the inner ear and vestibular pathways (26). Moreover, several migraine neurotransmitters play a role in the modulation of vestibular neurons activity (3). Furthermore, other theories involving cortical spreading depression can also trigger vestibular symptoms due the activation of multisensory cortical areas (3,26), and genetic defects of ion channels may also be involved in the MV pathogenesis (3).
The presence of abnormalities in the vestibular function (7,9,28,31,32) and balance changes (31–37) are commonly verified among patients with migraine. Interestingly, some authors demonstrated those alterations regardless of the presence of vestibular symptoms or diagnosis of vestibular migraine (7,9,26,33,34). It highlights the plausible hypothesis of a continuum between migraine and vestibular migraine, suggesting that both conditions should not be considered separate entities with distinct pathophysiologies (7,9).
Furthermore, despite the greater reporting of vestibular symptoms during a migraine attack, we observed that 25% of the MoA group and 40% of the MA and CM groups presented symptoms in the headache-free interval. This was already verified in previous studies (3,9,26), and suggests that those symptoms can be considered inherent to the migraine condition, such as other interictal manifestations including cutaneous allodynia (38) and photo and phonophobia (39).
It is known that overall migraine disability increases with the presence of vestibular symptoms (21,27). Patients diagnosed with vestibular migraine had lower quality of life scores compared to dizziness-free controls (27). Also, the presence of dizziness is independently associated with migraine-related disability (Migraine Disability Assessment [MIDAS]), depression, and disability (World Health Organization Disability Assessment [WHODAS]) (21).
Data regarding the DHI questionnaire in patients with migraine is scarce, but a baseline of a clinical trial with patients with vestibular migraine demonstrated an overall score of around 50 points (40). Another study found similar scores among patients with vestibular migraine and Menière’s disease (38 and 36.6, respectively) (41). The lack of information regarding the presence of aura and chronicity in those studies may interfere with the comparison to our results, but in general, all scores were classified as mild-to-moderate handicap due to dizziness.
The verified handicap is mainly in the physical and functional aspects of the DHI, along with its correlation to functional measures of gait and balance (18–20), and the verified balance changes (31–37) demonstrate the impact of migraine and vestibular symptoms on patients’ daily living. Migraine aura, attacks frequency and attacks intensity can explain 36% of the DHI scores. Moreover, the presence of migraine and the presence of aura or greater frequency are associated to the risk of presenting moderate-to-severe levels of dizziness handicap at least 2.8-fold, according to the lower bound of the prevalence ratio’s CI 95%. Its clinical significance can not be neglected during migraine treatment. Along with pain management, physical therapy strategies including vestibular rehabilitation and balance retraining should be encouraged (42–44).
We can list the following limitations of the present study. The data collection in a tertiary clinical setting and inclusion of a sample of women may restrict the generalizability and overestimate our results. Moreover, due to the cross-sectional design, no statement regarding causality can be made. This study had no intention to establish populational prevalence values, since methodological strategies for this kind of research were not performed. We concluded the data collection when the pre-established number of individuals in each group was reached, in a random and consecutive sampling. Despite the similarities of our results to population-based samples, the data cannot be extrapolated as prevalence for the disease.
Another important limitation is the lack of individual application of the vestibular migraine criteria or the assessment of specific psychological conditions such as anxiety in our sample. The diagnosis of vestibular migraine would provide the possibility of association between the different categories described by the IHS and the Bárány Society’s Classification of Vestibular Symptoms, even though it is not a validated diagnosis. However, the description of the vestibular symptoms and their relation to the migraine attack in different subgroups of migraine adds to the current knowledge of this emerging area.
This is the first study that has included the investigation of vestibular symptoms and measured their specific handicap in patients with chronic migraine. Along with exclusion of any case with history of a vestibular disease and a robust sample size, conclusions regarding the association and risk between migraine and vestibular symptoms can be drawn. The present results are of clinical importance and may contribute to the understanding of the vestibular migraine entity.
Conclusion
Vestibular symptoms are prevalent in patients with migraine, especially vertigo in individuals with aura and chronic migraine. These symptoms are related to a greater perceived handicap due to dizziness, especially in both migraine subgroups, impacting mainly the physical and functional domains. The presence of migraine, especially with aura and chronic, is associated with the risk of vestibular symptoms, of any handicap and of moderate-to-severe handicap related to vestibular symptoms. Migraine aura, intensity and frequency can be considered predictors of the handicap related dizziness.
Article highlights
Prevalence of vestibular symptoms and handicap due dizziness are greater in patients with migraine than controls. Vertigo is the main presentation among the vestibular symptoms in patients with aura and chronic migraine. Patients with aura and chronic migraine have an additional impairment due to vestibular symptoms, especially in the physical and functional domains. The presence of migraine is associated to the risk of vestibular symptoms 5.2 to 6.6-fold, and to moderate-to-severe handicap due dizziness 20.0 to 40.0-fold. Migraine aura, intensity and frequency can predict 36% of the handicap due dizziness.
Footnotes
Acknowledgements
We would like to thank the Clinic of Craniofacial Pain of the Ribeirão Preto Clinics Hospital.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the FAPESP Foundation (grant number: 2012/20046-2).
