Abstract
Background
Our knowledge of the presence and type of premonitory symptoms in patients with cluster headache is limited.
Methods
Premonitory symptom presence and type in a clinical cohort of cluster headache and migraine was collated retrospectively from clinical notes, alongside a cluster headache nitroglycerin triggered experimental group and an age-matched migraine nitroglycerin experimental group. Demographic data and premonitory symptoms in cluster headache and migraine patients were analysed. The primary focus was on premonitory symptom presence and phenotype in cluster headache patients, with secondary analysis exploring associated factors and comparing spontaneous and nitroglycerin-triggered symptoms in cluster headache and migraine cohorts.
Results
Among 164 cluster headache patients, 122/164 (74%) males, aged 45.9 ± 13.8 years (mean ± SD), 66/164 (40%) had chronic cluster headache and 32/164 (20%) had also comorbid migraine. Among them, 85% exhibited premonitory symptoms for which the presence was associated with oxygen treatment. No significant differences were found in symptom frequency between cluster headache and migraine cohorts or within spontaneous and nitroglycerin-triggered symptoms.
Conclusions
This study highlights recognizable spontaneous and nitroglycerin-triggered premonitory symptoms in cluster headache and factors potentially impacting cluster headache management, which may aid in tailoring treatment strategies for both conditions through the use of treatment prediction and early intervention.
This is a visual representation of the abstract.
Introduction
Cluster headache (CH) is a primary headache disorder that affects around one in 1000 in the general population (1,2). Although CH is a separate headache disorder from migraine in several aspects, such as gender-specificity and headache specific characteristics, both share some clinical characteristics, trigger factors and response to certain treatments, as well as neurobiological substrates, such as shared trigeminovascular and trigeminal autonomic pathways and functional biomarkers, including the hypothalamic networks (3,4).
To date, several studies have identified premonitory symptoms of migraine (5), although fewer studies have evaluated the frequency and type of premonitory symptoms in patients with CH. Thirty-five signs and symptoms of CH pre-attack or premonitory phases have been previously reported and evaluated using semi-structured questionnaires (6–11). These symptoms were categorized into local painful symptoms, local painless symptoms and general symptoms (6). Premonitory symptomatology can occur before pain onset and continue into the pain phase (12). To date, neurobiological mechanisms of premonitory symptoms based on the functional imaging are few (13,14). However, factors associated with the presence of premonitory symptoms among patients with CH have not been fully explored. Moreover, understanding the premonitory symptoms in CH and their similarity with those present in migraine may offer insights into the underlying pathophysiological mechanisms, as well as structures involved in the early phases of a headache attack, and ultimately contribute to identifying a novel therapeutic approach or management that would be used to prevent the headache onset.
The present study aimed to explore the presence and prevalence of premonitory symptoms in CH patients and to determine any putative factors associated with CH. Moreover, we aimed to compare spontaneous and triggered CH premonitory symptoms in patients with CH and migraine. We hypothesized that spontaneous and triggered premonitory symptoms may be similar among CH and migraine patients, suggesting that they may be mediated via the same brain structures and neurochemical pathways.
Methods
Study population and study design
A retrospective service evaluation was conducted including patients with CH fulfilling criteria of the International Classification of Headache Disorders, 3rd edition (ICHD-3) (2) treated at a tertiary headache centre at King's College Hospital from 1 January 2014 until 29 September 2023, in which a review of records for clinical symptoms was conducted (‘clinical CH cohort’). Similarly, a service evaluation of patients with migraine from 1 January 2014 until 12 December 2018, was also included (‘clinical migraine cohort’). Additionally, both spontaneous and triggered data collected from subjects exposed to nitroglycerin (NTG) (‘experimental CH cohort’ and ‘experimental M cohort’) were secondarily evaluated.
Eligibility criteria
In the clinical CH and migraine cohorts, inclusion criteria were (i) patients aged over 18 years; (ii) diagnosis by a headache specialist according to the ICHD-3 criteria (2) of migraine, episodic CH or chronic CH; and (iii) able to report premonitory symptoms preceding CH or migraine headache phase of the attack. The criteria comprised significant psychiatric or somatic diseases that might influence their symptom report capacity.
In the experimental CH and migraine cohorts, inclusion criteria were (i) patients aged over 18 years, age sex and age-matched; (ii) diagnosis by a headache specialist according to the ICHD-3 criteria (2) of episodic CH or chronic CH; (iii) development of headache after NTG-infusion and not after placebo infusion; and (iv) able to report premonitory symptoms preceding CH or migraine headache phase of the attack.
Ethical considerations
Clinic CH and M cohort data were acquired from headache semi-structured interview, at the first clinic consultation, as part of a specialized Headache clinic evaluation and reviewed as part of a service evaluation, which does not require Research Ethics Committee review in the UK (http://www.hra-decisiontools.org.uk/research). Informed consent was not required for use of patient data for this study because the data were acquired from notes and clinic letters for patients as part of a service evaluation process; therefore, only local hospital approval was needed. The experimental CH study was approved by the London, City & East Research Ethics Committee (16/LO/0693). The experimental M cohort study using NTG exposure in human subjects was approved by the Camden and King's Cross Research Ethics Committee (14/LO/2241). All subjects enrolled in the study provided their informed written consent for participation, according to the Declaration of Helsinki (15).
Sample size
The clinical cohorts included all patients with available information regarding the presence or absence of premonitory symptoms preceding the onset of the pain of individual attacks. The experimental cohorts included patients with available data regarding spontaneous and triggered premonitory symptoms included in previous studies (16–18). This was a convenience sample based on available data.
Variables and data collection
The work was conducted and reported according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines (19). Data were collected from clinical records in which in a semi-structured interview was conducted by a headache neurologist. A complete medical history was obtained from each patient during an in-person clinical interview, taken by a neurologist with experience in headache disorders. The first consultation letter, documenting the detailed headache history of selected patients during their initial visit to the Headache Clinic, was retrospectively reviewed. All patients had a normal brain imaging and pituitary function laboratory testing performed as part of the diagnosis workup in CH and were treated by the standard of care.
Clinical CH and M cohorts
Clinical and demographic variables as well as premonitory symptoms along with headache characteristics, were extracted from clinical notes from the patients included in the clinical CH and migraine cohorts. Demographic and clinical variables included are sex, age, age of onset, episodic or chronic CH, disease duration and premonitory symptoms. In patients with CH, attacks per day when in bout, duration of untreated and treated attacks, severity of untreated attacks based on a visual rating scale, current history of migraine, number of treatments, and concomitant preventive treatment were documented. Premonitory CH symptoms were evaluated. Assessments were based on routinely used data for clinical care. Note that each patient may have one or more premonitory symptoms.
Experimental CH and M cohorts
For triggering headache, all of the study visits were performed within the Clinical Research Facility at King's College Hospital. The visit involved written informed consent for study participation, followed by detailed phenotyping of spontaneous CH and migraine attacks as included in our previous studies (17,20) in which patients were exposed to 0.5 µg/kg per minutes NTG infusion. Premonitory symptoms were evaluated in both experimental cohorts using a questionnaire as described in our previous work (17).
Premonitory symptom definition
A premonitory symptom was defined as any symptom that the patient reliably experienced before the onset of a CH or a migraine headache (excluding typical aura).
Study endpoints
The main outcome, comprising the prevalence of spontaneous premonitory symptoms in patients with CH, was calculated and presented as a percentage. We conducted an exploratory analysis assessing the presence of variables associated with the presence of spontaneous premonitory symptoms in CH. Moreover, we evaluated the type of premonitory symptoms in patients with CH, CH and comorbid migraine, and migraine. Furthermore, we evaluated the similarities between spontaneous and triggered premonitory symptoms in patients with CH and migraine.
Statistical analysis
RStudio, version 3.6 (Posit, Boston, MA, USA) and SPSS, version 24 (IBM Corp., Armonk, NY, USA) were used to perform the statistical analyses. Demographic, clinical and headache characteristics, as well as the prevalence of premonitory symptoms, are presented as follows: categorical variables as frequencies and percentages and continuous variables as the mean ± SD or medians and interquartile ranges (IQR) if the distribution was not normal as determined by the Kolmogorov–Smirnov test and homogeneity of variance using Levene test.
Association between categorical variables was checked with the chi-squared test or Fisher's exact test, with two tailed testing, depending on the counts in each cell of the cross-tabulation. The chi-squared test or Fisher's exact test was used to investigate homogeneity between symptom prevalence between clinical CH and M cohorts. Moreover, an association between categorical variables was examined using Fisher’s exact test to compare spontaneous and triggered premonitory symptoms within CH and M cohorts. Comparison of continuous variables between cases and controls were compared with the t-test for independent samples or the Mann–Whitney U-test depending on departure of normality assumption, with two tailed testing. For continuous variables, comparisons across three groups were performed using one-way analysis of variance or the Kruskal–Wallis test, depending on the normality assumption. p < 0.05 was considered statistically significant.
A forward and backward stepwise logistic regression model including variables achieving significant differences (p < 0.05) in the univariate model and clinically important demographic variables was estimated to evaluate differences between patients with and without premonitory symptoms. We present the odds ratios (OR) of association with 95% confidence interval (CI) of demographic and clinical variables in the clinical CH cohort with and without premonitory symptoms. No sample size calculation was conducted prior to the study. There were no missing data regarding the primary outcome. p < 0.05 was considered statistically significant. Spontaneous and NTG-triggered attack phenotypes and experimental and clinical cohort phenotypes were compared using agreement analysis (18). Percent of overall agreement was calculated by dividing the number of agreements between both triggered and premonitory symptoms by the total number of assessments, as follows: Percentage of overall agreement: yes spontaneous and yes triggered + No spontaneous and No triggered/total number × 100. Symptom reporting percentage agreement of 60% or more was considered moderate agreement and therefore significant and these results are highlighted in bold, in accordance with our previous work (18).
Results
Description of the clinical CH cohort
During the study period, a total of 164 CH patients were included in the clinical cohort. The sociodemographic characteristics of the patients included in the study are shown in Table 1. There were 122/164 (74%) males, aged 45.9 ± 13.8 years, of whom 66/164 (40%) had chronic CH and 32/164 (20%) had comorbid migraine. Patients had a mean ± SD of 4 ± 2 CH attacks and had previously used 4 ± 2 prior preventive treatments.
Sociodemographic characteristics of the clinical cluster headache cohort.

CH, cluster headache; VRS, visual rating scale.
Premonitory symptoms in the clinical ch cohort
The main characteristics of patients with CH regarding the presence or absence of premonitory symptoms are included in Table 2.
Baseline and demographic characteristics of the clinical CH cohort regarding the presence of spontaneous pre-attack symptoms.

CH, cluster headache; eCH, episodic cluster headache; VRS, visual rating scale.
*p < 0.05.
Premonitory symptoms were present in 139/164 (85%) of CH patients. The most common premonitory symptoms in CH patients were local and painful symptoms (57/139; 41%) followed by mood changes (45/139; 32%), cognitive impairment (34/139; 24%), neck stiffness (32/139; 23%), fatigue (30/139; 22%) and yawning (24/139; 17%). All premonitory symptoms evaluated in patients with CH are detailed in Table 3.
Main spontaneous premonitory symptoms reported by patients with CH.

CH, cluster headache.
*Local and painful symptoms: shooting pain or stabbing pain, dull or aching sensation and burning sensation preceding the onset of CH attack.
Predictors of premonitory symptoms in the clinical ch cohort
A higher number of attacks, use of oxygen and non-smoking were associated with the presence of premonitory symptoms in patients with CH. The logistic regression analysis showed that use of oxygen (B = 1.281; 95% CI = 0.84–0.176; p = 0.014) and smoking history (B = −0.965; 95% CI = –2.036 to 0.318; p = 0.041) were independently associated with the presence of premonitory symptoms in patients with CH (see supplemental material, Table S1).
Spontaneous premonitory symptoms in clinical ch and migraine cohorts
There were no statistical differences in the frequency of premonitory symptoms between patients with CH and migraine (139/164; 85% vs. 286/340; 84%; regression model adjusted p = 0.523). The clinical characteristics of patients with CH and migraine are included in the see supplemental material (Table S2). The type and frequency, including the number of spontaneous premonitory symptoms among patients with CH without comorbid migraine, patients with CH with comorbid migraine and patients with migraine are detailed in Figure 1 and Table 4 (see also supplemental material, Table S3). It is noteworthy that neck discomfort, mood changes and cognitive impairment, which are frequently reported in both cohorts, exhibit higher prevalence among patients with migraine or CH with comorbid migraine, compared to those with CH without comorbid migraine.

Distribution of the premonitory symptoms reported by patients with CH without comorbid migraine, CH with comorbid migraine and migraine.
Comparison of the main spontaneous premonitory symptoms reported by patients with CH and migraine.
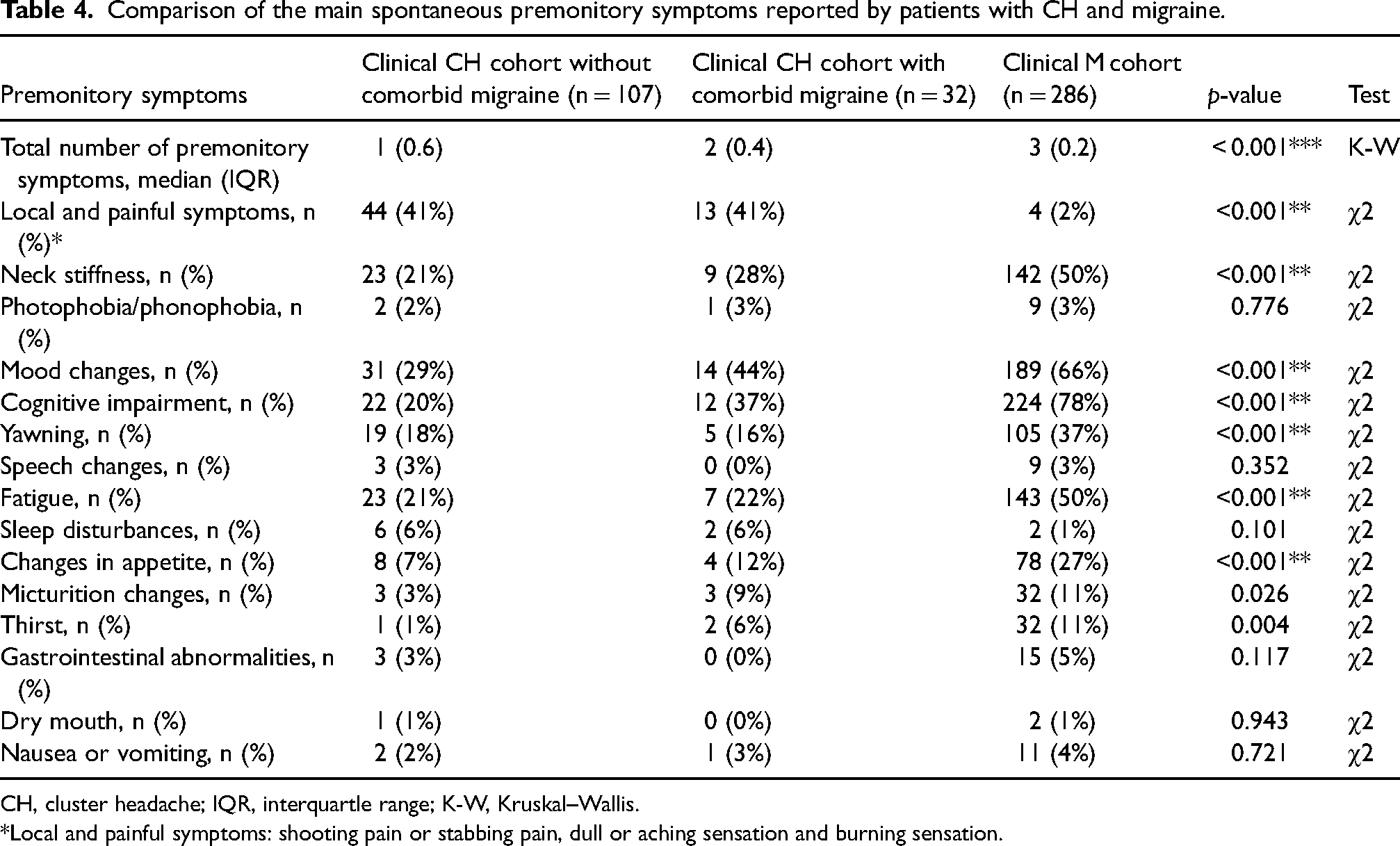
CH, cluster headache; IQR, interquartle range; K-W, Kruskal–Wallis.
*Local and painful symptoms: shooting pain or stabbing pain, dull or aching sensation and burning sensation.
Spontaneous and triggered premonitory symptoms in experimental CH and M cohorts
Among patients with CH that were triggered using NTG (n = 24), 18 subjects had completed both the nitroglycerin and placebo visits, and 14 developed headache after NTG infusion and did not develop headache after placebo infusion in a double-blind randomized design, and were therefore included in the present study. In the experimental group, all patients who reported spontaneous premonitory symptoms (12/14; 86%) developed typical premonitory symptoms and headache symptomatology when triggered. The clinical characteristics of the experimental CH cohort are included in the supplemental material (Table S4).
In the experimental CH cohort, the proportion of premonitory symptoms regarding spontaneous and NTG-triggered symptoms are included in Table 5. The clinical characteristics of the experimental cohorts are included in the supplemental material (Table S5).
Comparison of spontaneous and NTG-triggered premonitory symptoms in CH patients.

CH, cluster headache; NTG, nitroglycerin.
POA: Percentage of overall agreement: Yes spontaneous and yes triggered + No spontaneous and No triggered)/total number)×100. There were no missing data.
Among NTG-triggered patients, 18/24 (75%) patients with CH and 47/49 (96%) patients with migraine developed premonitory symptoms and 12/14 (86%) developed premonitory symptoms followed by a headache. The characteristics of the 12 CH patients and 12 age and sex-matched migraine patients are included in the supplemental material (Table S5). A comparison between spontaneous and NTG-triggered premonitory symptoms among CH and migraine is also included in the supplemental material (Tables S6 and S7).
In the experimental group, there was a similar proportion of spontaneous and NTG-triggered-premonitory neck stiffness, mood changes and yawning in the same individual and among experimental CH and migraine groups. Among them, seven of 12 (58%) patients had comorbid migraine.
Discussion
In the present study, spontaneous and NTG-triggered premonitory symptoms in CH and migraine were evaluated. Regarding the clinical CH cohort, an association between smoking and oxygen treatment with premonitory symptoms was found in patients with CH, which may impact CH premonitory symptoms management. Moreover, a similar frequency of spontaneous and NTG-triggered premonitory symptoms phenotype was identified in both CH and migraine patients. Interestingly, the number of premonitory symptoms was higher in patients with migraine compared to patients with CH with comorbid migraine and also compared to patients with CH without comorbid migraine. Moreover, certain premonitory symptoms were more prevalent in migraine, suggesting a common link in the pathophysiology of premonitory symptoms and a possible shared premonitory phenotype in patients with migraine and CH. These symptoms occurring in migraine and CH with comorbid migraine might allude to more widespread limbic involvement in migraine on functional imaging relative to cluster- as per our recent imaging study (21).
To date, the literature regarding the evaluation of premonitory symptoms in CH is less than that reported in migraine. Here, we report over 80% of patients with CH report premonitory symptoms, a similar percentage compared to previous reports (6,7,9,11). Among them, local and painful symptoms were very common, raising the question of whether this might be a premonitory symptom or a primary manifestation of the attack. Other general premonitory symptoms such as mood changes, cognitive impairment, neck discomfort, fatigue and yawning were present in 20–30% of patients, and this is consistent with what has been reported in previous premonitory CH studies (6).
Furthermore, our study revealed an association between oxygen treatment and smoking with the presence of identifiable spontaneous premonitory symptoms in patients with CH. Oxygen is strongly recommended as acute treatment for CH attacks in the European Academy of Neurology guidelines (22). Our findings indicate that patients experiencing premonitory symptoms may more frequently utilize oxygen as a treatment, a strategy that holds potential for efficacy even during the premonitory phase, which might explain this association. Moreover, from a mechanistic point of view, oxygen therapy may alleviate premonitory symptoms by modulating hypothalamic activity and the trigeminocervical complex, key components in CH pathophysiology, through its effects on autonomic and nociceptive pathways (23,24). Moreover, the warning would require an effective therapy with a quick route of administration. In this sense, the recent study involving ubrogepant during the premonitory (prodromal) migraine phase has shown that it is effective and well-tolerated in this phase of the migraine attack (25). Future studies using a gepant in the premonitory stage of CH attacks may worth consideration, such as using a nasal formulation (26).Moreover, while the link between smoking and CH is well-established, with smokers exhibiting a higher incidence and increased severity of CH attacks (27), the relationship between smoking status and premonitory symptoms remains less understood. A recent investigation suggested that pre-attack symptoms, primarily head and facial discomfort, occurring minutes before headache onset are linked to smoking or exposure history surprisingly, we did not find this association (28). We found that smoking status correlated with the absence of premonitory symptoms in the multivariate analysis. However, there is scarce evidence of the relationship between smoking in the presence of premonitory symptoms and further research is warranted to elucidate the underlying mechanisms driving this association.
Spontaneous premonitory symptoms in clinical ch and migraine cohorts
In the comparison between spontaneous premonitory symptoms in the clinical CH and migraine cohorts we found a similar frequency of premonitory symptoms with approximately 85% in both groups. Although the types of premonitory symptoms were largely overlapping between the two cohorts, patients with migraine tended to report a higher overall number of symptoms compared to those with CH and also compared to patients with CH and comorbid migraine. This observation suggests the existence of variable premonitory symptom manifestations in the patients with both pathologies, which may underly an intermediate premonitory symptoms phenotype of CH and comorbid migraine. There is good functional correlation between the brain areas involved in the premonitory phase including the hypothalamic and periaqueductal grey networks, which are shared by migraine and CH patients, suggesting their involvement in the development of premonitory symptoms (3,29). Moreover, brain functional neuroimaging has shown distinct and also shared neurobiological substrates in the two conditions, which could cause overlap of symptoms by early brainstem involvement, as well as alluded to oscillatory brainstem, hypothalamic and limbic networks prior to the onset of pain, with perhaps more prominent limbic symptoms in migraine (13).
Experimental CH and migraine cohorts
In the experimental CH cohort, we found that all patients who reported spontaneous premonitory symptoms also developed premonitory symptoms after NTG infusion. Moreover, the fact that NTG may trigger premonitory symptoms in the absence of a headache support our current understanding and theory that vasodilatation is not driving the headache; therefore a pain attack might not be exclusively initiated or modulated by a peripheral input (17).
In addition, our results suggest that premonitory symptoms are similar in the same individual in triggered and non-triggered CH attacks and among patients with migraine and CH. These results suggest that non-painful premonitory symptoms in patients with CH and migraine may be influenced by common changes in areas of the brain controlling arousal, cognition and homeostasis (3). There is considerable support for the involvement of the hypothalamus in premonitory symptoms, as evidenced by imaging studies using H215O positron emission tomography, which show increased hypothalamic blood flow during the premonitory phase of migraine (30), Importantly, the hypothalamic regions implicated in CH and migraine are not identical (4,31). Further studies evaluating neuroimaging in spontaneous vs. triggered premonitory symptoms would allow to understand better the role played by the hypothalamus in early stages of an attack (13). Regarding the phenotype and number of premonitory symptoms among the two groups, a similar number was found between the two NTG-triggered groups, aligned with previous triggering studies in which the provocation leads to a higher number of symptoms, which might be related to direct questioning and reporting bias.
Regarding putative differences in premonitory symptom phenotype between CH and migraine, whereas some symptoms are shared, in CH local and painful symptoms alone, such as cranial autonomic symptoms, are prominent. This would suggest superior salivatory nucleus activation before headache. Recognizing and acknowledging premonitory symptoms is crucial for physicians during history-taking, allowing them to understand the prevalence and characteristics of the premonitory phase. Moreover, it will allow exploration of the potential effects of abortive medications on premonitory manifestations, as well as the depth and duration of disability experienced by the patient not only during an attack, but also beforehand. Gaining a deeper understanding of this significant aspect of the CH and migraine attack is pivotal for appreciating the neurobiological mechanisms involved in attack generation as well as in the therapeutic processes for pain management.
Limitations
The retrospective design introduces the possibility of recall bias. Although semi-structured interview and systematic standardized patient questioning may introduce a potential reporting bias, such an approach can result in improved and more detailed phenotypes. Additionally, direct observation of CH and migraine attacks contributes to this enhanced understanding. Although the present study allows us to identify possible premonitory symptoms that are present in both CH and migraine, a prospective study should be performed to address the predictability. Our effect size is very difficult to account for and sample size calculation would hold value for future studies. A strength of this study is that, to our knowledge, it is one of the largest series published to date of patients with CH regarding the study of premonitory symptoms. Moreover, there is a high proportion of CCH in our cohort probably due the fact that the headache clinic is a tertiary centre.
Conclusions
Our data demonstrate that premonitory symptoms are reported by approximately 80% of CH patients. Among them, general symptoms suggestive of hypothalamic involvement are present in approximately 20–30% of CH patients prior to an attack. Moreover, we found that the use of oxygen is independently associated with the occurrence of premonitory symptoms. Overall, the similarity between spontaneous and triggered CH premonitory symptoms and with migraine supports the link in the pathophysiology of premonitory symptoms in these two entities. It is also worth noting that CH patients are largely male and migraine patients are female. There is a similarity between premonitory symptoms that might be influenced by comorbid migraine in patients with CH. Therefore, studying men alone would therefore be useful in this regard to determine whether there are hormonal influences on premonitory symptoms phenotype and occurrence, and if this accounts for any differences between CH and migraine. Further studies focusing on CH spontaneous and triggered premonitory symptoms and its relationship with migraine premonitory symptoms can aid in the development of more tailored treatment strategies for these patients.
Clinical implications
Premonitory symptoms are reported by approximately 80% of CH patients. Symptoms suggestive of hypothalamic involvement are present in approximately 30% of patients prior to an attack, underscoring the need of neuroimaging research. The use of oxygen treatment is independently associated with the occurrence of premonitory symptoms. The similarity between spontaneous and triggered CH premonitory symptoms and with migraine supports the shared biology of premonitory symptoms in these two entities. Studying potential differences between premonitory symptoms within patients with cluster headache and comorbid migraine would be a useful next step to furthering understanding in this area.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251331554 - Supplemental material for Evaluation of premonitory spontaneous and nitroglycerin triggered symptoms among patients with cluster headache and migraine
Supplemental material, sj-docx-1-cep-10.1177_03331024251331554 for Evaluation of premonitory spontaneous and nitroglycerin triggered symptoms among patients with cluster headache and migraine by Alicia Gonzalez-Martinez, Diana Y. Wei, Nazia Karsan, Karthik Nagaraj, Helin Gosalia and Peter J. Goadsby in Cephalalgia
Footnotes
Acknowledgments
We thank all of the patients and healthcare workers. Acknowledgements are also due to Instituto de Salud Carlos III (ISCIII) and the European Union (ESF+) through a Río Hortega Fellowship (CM21/00178), Juan Rodés Fellowship (JR23/00005) according to O.A., M.P. de 13 de diciembre de 2023 through a project (PI24/01085) to AGM, and to the European Academy of Neurology (EAN) and Sociedad Española de Neurología (SEN) for a clinical fellowship to AGM.
Declaration of conflicting interests
PJG reports, over the last 36 months, grants from Kallyope, and personal fees from Aeon Biopharma, Abbvie, Amgen, Aurene, CoolTech LLC, Eli-Lilly and Company, Linpharma, Lundbeck, Pfizer, PureTech Health LLC, Satsuma, Shiratronics, Teva Pharmaceuticals, Tremeau, and Vial; personal fees for advice through Gerson Lehrman Group, Guidepoint, SAI Med Partners and Vector Metric; fees for educational materials from CME Outfitters and WebMD; publishing royalties or fees from Massachusetts Medical Society, Oxford University Press, UptoDate and Wolters Kluwer; and a patent magnetic stimulation for headache (No. WO2016090333 A1) assigned to eNeura without fee. A.G.M has received speaker honoraria from TEVA, Lilly and Altermedica.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the Instituto de Salud Carlos III, (grant number CM21/00178, JR23/00005, PI24/01085).
Data availability
Anonymised data that underlies the results of this article can be shared on reasonable request to qualified investigators.
Ethical statement
Clinic CH and M cohort data were acquired from headache semi-structured interview. Informed consent was not required for use of patient data for this study.
The experimental CH study was approved by the London, City & East Research Ethics Committee (16/LO/0693). All subjects enrolled in the study provided their informed written consent for participation, according to the Declaration of Helsinki.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
