Abstract
Background
New daily persistent headache (NDPH) is an often treatment-refractory primary headache disorder with a lack of evidence base for treatment.
Methods
We performed an observational study using prospectively collected data in consecutive patients with NDPH, chronic migraine with daily headache (daily-CM) and without daily headache (non-daily-CM). Patients were treated with two cycles of OnabotulinumtoxinA. Propensity score matching was used to control for imbalances between the groups. The primary outcome measure was the proportion who experienced a ≥ 30% improvement in monthly moderate-to-severe headache days at 24 weeks.
Results
We included 58 patients with NDPH, 148 with daily-CM, and 84 with non-daily-CM. In NDPH, 34.5% of patients experienced a ≥ 30% improvement in monthly moderate-to-severe headache days, compared to 43.2% in daily-CM and 51.2% in non-daily CM. In NDPH, 6.9% experienced an improvement in monthly headache days, 27.6% an improvement in headache severity, 25.9% a ≥ 6 point improvement in HIT-6 score, and 59% a patient reported improvement. There was no significant difference in response rates between the three groups. Adverse event rates were similar in all groups and there were no serious adverse events.
Conclusion
OnabotulinumtoxinA is effective in approximately 1/3 patients with NDPH and has a favourable safety profile.
This is a visual representation of the abstract.
Background
New daily persistent headache (NDPH) is a primary headache disorder defined by its sudden onset and persistence for at least three months (1). The phenotype of primary NDPH frequently resembles chronic migraine, although it is still unknown whether it represents a distinct disorder with different pathophysiology, and little is known about how its response to treatment compares to chronic migraine (2). Treatment studies of NDPH are limited to case reports and small case series, and due to lack of alternatives, NDPH is usually treated with the same treatments used in chronic migraine. Despite this, NDPH is recognised to frequently be treatment refractory (3–5).
OnabotulinumtoxinA is one of the few treatments proven in randomised control trials to be effective in the preventive treatment of chronic migraine (headache on at least 15 days per month, of which at least eight have features of migraine) according to the PREEMPT protocol (6). Other than case reports and one small retrospective case series, response of NDPH to onabotulinumtoxinA has not been studied (7–9). NDPH is rare with an estimated prevalence of only 0.03% (10), and a previous open-label clinical trial of onabotulinumtoxinA for NDPH was terminated due to difficult enrolment (11).
We performed an open label observational study using prospectively collected clinical data, of patients with NDPH treated with onabotulinumtoxinA, and compared outcomes in NDPH to chronic migraine.
Objectives
To assess the effectiveness and safety of onabotulinumtoxinA treatment for primary NDPH compared to daily and non-daily chronic migraine.
Methods
Population
This prospective observational study investigates consecutive patients with primary NDPH and chronic migraine treated with onabotulinumtoxinA between 2013 and 2022 in a single centre secondary and tertiary headache clinic at the National Hospital for Neurology and Neurosurgery, London, UK.
We included three groups of patients. Group 1 consisted of patients with NDPH who met International Classification of Headache Disorders 3rd Edition (ICHD-3) criteria (1), had the phenotype of CM, had an ongoing daily headache, had no symptoms to suggest a secondary cause for the headache, and had normal MRI brain imaging. Group 2 (daily-CM) consisted of patients with chronic migraine which had transformed from episodic migraine, who had a daily headache (and therefore had the same headache frequency as the NDPH group); and Group 3 (non-daily-CM) consisted of patients with chronic migraine which had transformed from episodic migraine, who had between 15 and 27 headache days per month. The decision to divide patients with CM into two groups depending on whether they had daily or non-daily headaches was made so that there was a group (Group 2) which more closely resembled NDPH at baseline in terms of headache frequency, severity, and disability levels. By definition, all patients with NDPH have a daily headache, whereas this is not true for CM, who may have anywhere between 15 and 28 days of headache per month. Therefore if we compared NDPH to a group including all patients with CM together, this would not be a fair comparison.
Patients were treated under the UK National Health Service in accordance with National Institute for Health and Care Excellence (NICE) guidelines (12). This meant that all patients were required to have failed to respond to at least three classes of oral preventive treatment (including tricyclic antidepressants, beta blockers, topiramate, angiotensin reception antagonists, sodium valproate, flunarizine, serotonin and norepinephrine reuptake inhibitors, pizotifen), and that patients with medication overuse, as per ICHD-3 criteria (1), were excluded from receiving treatment with onabotulinumtoxinA. These guidelines do not specifically mention NDPH, however as there are no guidelines for treatment of NDPH, and NDPH (when it has chronic migraine phenotype) is considered by many to be a form of chronic migraine, it is common practice to apply guidelines for chronic migraine to NDPH which has a chronic migraine phenotype. Patients in all three groups were required to meet the same criteria.
Intervention
OnabotulinumtoxinA was administered in two cycles, 12 weeks apart, according to the Phase III REsearch Evaluating Migraine Prophylaxis Therapy (PREEMPT) protocol (13).
Study assessment
Patients were instructed to complete a headache diary for a period of four weeks prior to starting treatment to act as a baseline, and then continue the diary up until the 24 week follow up date. The diary captured the presence, severity (on 0–10 verbal rating scale - VRS) and duration (in hours) of headache, and whether abortive medications were taken for each day for a 28-day period. Patients were also asked to complete the Headache Impact Test-6 (HIT-6) questionnaire of headache related disability and the Hospital Anxiety and Depression Scale (HADS) at baseline and again prior to the 24 week follow up appointment (14,15).
Safety was monitored throughout the study, with patients being able to report adverse events to the clinical team. At the 24-week appointment, patients were also asked to report any side effects experienced at any time during the 24-week period.
Endpoints
The primary outcome measure was the proportion of patients with a ≥ 30% improvement in monthly moderate-to-severe headache days (defined as days with VRS scale of 4 or above) in the four-week period preceding the 24 week follow up date, compared to baseline.
Secondary outcome measures were improvement in monthly headache days, mean headache severity (on 0–10 verbal rating scale), monthly headache load (a composite measure calculated as the monthly sum of headache VRS multiplied by headache hours for each day), patient's overall estimate of improvement (on 0–100% scale), the Headache Impact Test-6 (HIT-6) measure of headache-related disability (14), and adverse event rate. To categorise patients as responders or non-responders we used a 30% improvement cut-off for headache diary metrics, as this is what is specified by the National Institute for Health and Care Excellence (NICE) guidelines (12). For HIT-6 score an improvement of 6 or more points was used to define responders, as proposed by other authors (16).
Sample size
The sample size was primarily driven by practical consideration by including data from all consecutive patients who met eligibility criteria during the period of the study. A sample size calculation using two-sided alpha of 0.05 and power of 80% and enrolment ratio of 1:2.5 between NDPH (Group 1) and daily-CM (Group 2) showed that to detect a difference in response rates of 25% in NDPH and 50% in daily-CM would require sample sizes of 47 in Group 1 and 118 in Group 2. To detect a difference in response rates of 30% in NDPH and 50% in daily-CM would require samples sizes of 73 and 183 respectively.
Statistical analysis methods
We included all consecutive patients who met eligibility criteria during the period of the study. All data were collected prospectively within the clinical service using headache diaries and validated questionnaires. Missing data were not imputed.
Descriptive data were used to summarise the characteristics of the two groups as means with standard deviation (SD) or medians with interquartile ranges (IQR) depending on the distribution of data. Normality assumptions were assessed based on visual inspection of histograms. Statistical tests for comparisons of baseline data between the different diagnostic groups were obtained using an appropriate statistical test for the data type (categorical vs continuous). The following tests were used: Chi Squared (χ2) test and ANOVA (F) with Tukey post-hoc test. Where the assumptions required for a statistical test are not met, an alternative equivalent non-parametric test was used.
For the selected outcome measures, comparisons between the diagnostic groups were carried out using propensity score matching (17) to control for imbalances between the groups by ensuring similarity between the different groups on their propensity score weighted distributions for the following baseline covariates: gender, age, duration of chronic daily headache, number of failed preventative therapies, and baseline HAD-D. The propensity score method has proven to be an effective tool to reduce bias in nonrandomised studies, especially when the number of (potential) confounders is large and dimensionality problems arise (18).
Propensity scores were estimated using generalised boosted models (GBMs) (19), which has been implemented in the twang package in R (20). This approach is useful for implementing propensity score weighting when examining multiple groups. The propensity score analysis employed the average “treatment” effect (ATE). We used the multinomial propensity scores (mnps) function in the twang package in R to estimate the ATE, using absolute standardised mean difference as measure of balance. The ATE of a particular diagnostic group relative to an alternative diagnostic group is the comparison of the mean outcomes had the entire population been observed under the particular diagnostic group versus had the entire population been observed under the alternative diagnostic group. We used a combination of the overlap plot and the balance table to assess whether the groups were sufficiently similar.
The statistical comparison of outcome measures between the three diagnostic groups was performed using negative binomial regression for monthly moderate-to-severe headache days and headache days (as these variables were negatively skewed); linear regression for other continuous outcomes (which were approximately normally distributed); and logistic regression for responder analysis; with the derived propensity score weights as weights in all analyses.
Analyses were performed in R (R Studio version 4.3.3), IBM SPSS (Version 29.0), and Stata (Version 18).
Ethics and consent
Ethics board approval was obtained from Northwick Park Hospital Research Ethics Committee, London, UK (reference number 11/LO/1709). All patients gave written informed consent to be included.
Results
Population
A total of 58 patients with NDPH with CM phenotype and 232 patients with CM (of whom 148 had a daily headache) were treated with onabotulinumtoxinA during the study period and met the study inclusion criteria. All patients completed follow up.
Demographics and baseline headache diary results are shown in Table 1. Age distribution was similar between the three groups, but there was a greater female preponderance in both chronic migraine groups than in NDPH. The non-daily-CM group differed from the other two groups in several headache diary metrics due to having fewer headache days by definition. The NDPH and daily-CM groups exhibited similar characteristics. HIT-6 scores were similar in all three groups and HADS-D score was highest in the daily-CM group.
Baseline demographic and headache characteristics.
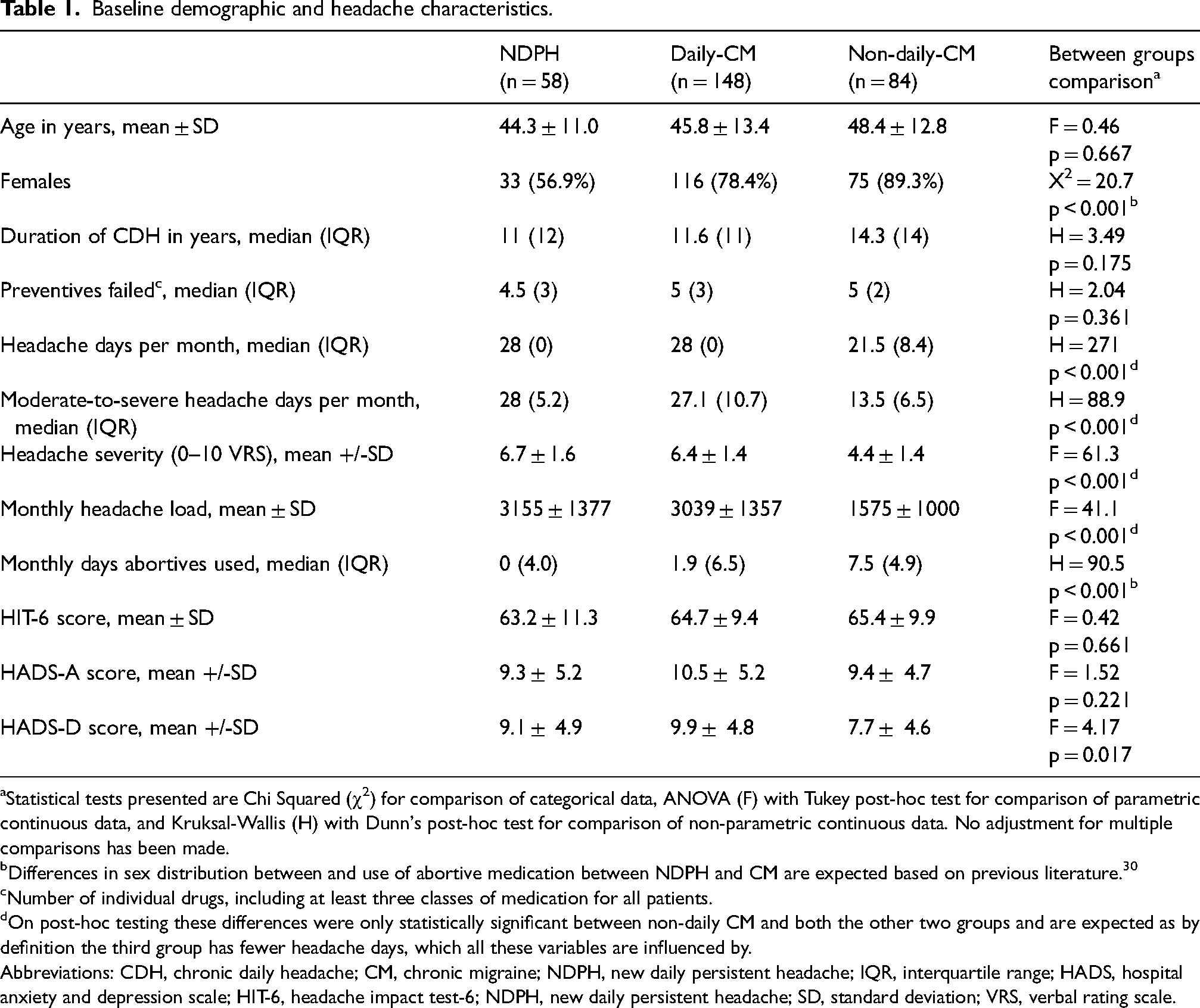
Statistical tests presented are Chi Squared (χ2) for comparison of categorical data, ANOVA (F) with Tukey post-hoc test for comparison of parametric continuous data, and Kruksal-Wallis (H) with Dunn's post-hoc test for comparison of non-parametric continuous data. No adjustment for multiple comparisons has been made.
Differences in sex distribution between and use of abortive medication between NDPH and CM are expected based on previous literature. 30
Number of individual drugs, including at least three classes of medication for all patients.
On post-hoc testing these differences were only statistically significant between non-daily CM and both the other two groups and are expected as by definition the third group has fewer headache days, which all these variables are influenced by.
Abbreviations: CDH, chronic daily headache; CM, chronic migraine; NDPH, new daily persistent headache; IQR, interquartile range; HADS, hospital anxiety and depression scale; HIT-6, headache impact test-6; NDPH, new daily persistent headache; SD, standard deviation; VRS, verbal rating scale.
Effectiveness
Headache diary data and patient estimate of improvement were collected in all patients with no missing data. HIT-6 data was missing in 10.3% patients with NDPH, 8.8% with daily-CM, and 8.3% with non-daily CM.
In the NDPH group 34.5% (20/58) of patients experienced a ≥ 30% improvement in moderate-to-severe headache days between baseline and 24 weeks. The mean (SD) reduction in moderate-to-severe headache days was 6.7 (9.7) days. Fewer patients with NDPH experienced a reduction in total headache days - only 6.9% (4/58) had a ≥ 30% improvement. Improvement in headache severity of ≥30% and HIT-6 of ≥6 points was reported in 27.6% (16/58) and 25.9% (15/52) respectively. More than half of patients in the NDPH group reported an overall subjective improvement of their headaches by ≥30% (see Table 2).
Outcome measures at 24 weeks follow up.

Abbreviations: CM, chronic migraine; NDPH, new daily persistent headache; IQR, interquartile range; HIT-6, headache impact test-6; NDPH, new daily persistent headache; SD, standard deviation; VRS, verbal rating scale.
The response rate for improvement in moderate-to-severe headache days of 34.5% in NDPH compared to 43.2% in daily-CM (odds ratio 1.23, 95% CI 0.61-2.47, p = 0.565) and 51.2% in non-daily-CM (odds ratio 1.19, 95% CI 0.49-2.90, p = 0.701). Mean (SD) improvement in moderate-to-severe headache days was also similar: 6.1 (8.6) days in daily-CM and 4.5 (6.4) days in non-daily-CM.
A greater than 30% improvement in total headache days was most common in non-daily CM with 37/84 (44.1%) compared to 22/148 (14.8%) in daily-CM and 4/58 (6.9%) in NDPH. There were no statistically significant difference in any of the secondary measures between the three groups (see Figure 1 and Table 2).
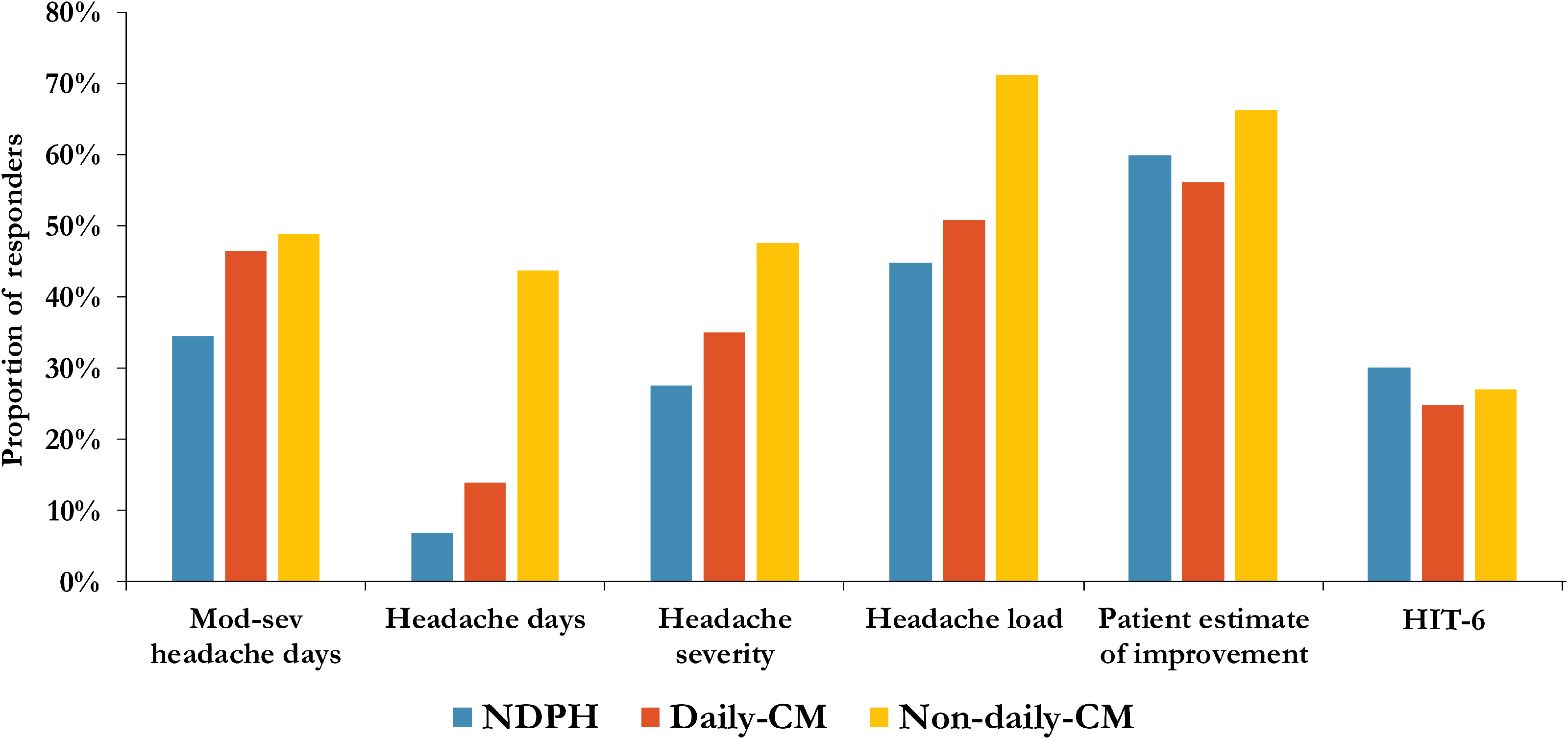
Proportion of responders for each outcome measure. Definition of responder for all variables except HIT-6 is a ≥ 30% improvement between baseline and 24 weeks. Definition of responder for HIT-6 score is a ≥ 6 point increase between baseline and 24 weeks. Abbreviations: CM, chronic migraine; HIT-6, headache impact test-6; mod-sev, moderate-to-severe; NDPH, new daily persistent headache.
Safety
Adverse event rates were similar in each group and are listed in Table 3. The most common adverse event was ptosis, which occurred in 17 (6.7%) of patients There were no serious adverse events.
Adverse events experienced at any time during the first 24 weeks of treatment.

Discussion
This study demonstrates that approximately a third of patients with NDPH with CM phenotypeexperience a ≥ 30% reduction in monthly moderate-to-severe headache days, and almost 60% of patients perceived an improvement in their condition. Response rates (i.e., proportion experiencing a ≥ 30% improvement) in NDPH were not statistically significantly different to patients with CM, who were similar in other ways and matched using propensity scores (derived using age, gender, previously failed treatments, and depression scores). Overall, onabotulinumtoxinA was similarly well tolerated in both NDPH and CM. This suggests that onabotulinumtoxinA could be used at a similar point in the management pathway for patients with NDPH as for CM.
This is one of few treatment studies of NDPH in which data has been collected prospectively, thereby increasing quality of the data compared to retrospective reporting of treatment effictiveness. It is also one of few observational studies in NDPH where response rates have been compared to CM. Previous reports of onabotulinumtoxinA response in NDPH consist only of case reports and one small case series. Neither of the two case reports appeared to use the PREEMPT protocol (7,8). The case series describes retrospective data in 19 patients treated according to the PREEMPT protocol, of whom eight out of 16 (50%) had an improvement in headache frequency after six months (9). This is higher than the response rate in the current study. However, that study relied on patient self-reports of headache frequency instead of headache diaries, used any improvement in frequency (whereas we used a 30% improvement threshold), and omitted data from three patients who dropped out, potentially inflating its reported effectiveness. In our study all patients completed 24 week follow up and the prospective completion of headache diaries was ensured by the headache nursing team, with the only missing data being a minority of approximately 10% of patients who did not complete HIT-6 questionnaires either at baseline or 24-week follow up (data shown in Table 2).
The improvement in headache days was greatest in the non-daily-CM group. The evidence for onabotulinumtoxinA in CM comes primarily from the PREEMPT randomised placebo-controlled clinical trials (6). Patients with continuous headache were excluded from these trials, therefore the population included was similar to the non-daily-CM group in the current study. A previous study assessing association of clinical features with response to onabotulinumtoxinA in CM found that responders had a median monthly headache days of 25/30, compared with non-responders who had a median of 30/30 monthly headache days (i.e., daily-CM) (21). The presence of a daily headache has also been associated with lack of response to other migraine treatments including candesartan and erenumab (22–24). A reason for this may be that the biology of a continuous headache is different to that of headache which still occurs in episodes – for example it may involve more cortical mechanisms involved in central sensitisation, rather than peripheral trigeminovascular factors, which are targeted by onabotulinumtoxinA. Pathophysiological studies comparing daily and non-daily CM have rarely been performed, but studies comparing CM to episodic migraine have shown neuroimaging and neurophysiological differences suggestive of central sensitisation in CM (25,26). Conditions where the headache frequency normally fluctuates from month to month (as is common in non-daily-CM) will also be more likely to have a random improvement over a treatment response threshold (such as the 30% cut off used in this study), than conditions where the headache is continuous and daily (as in NDPH and daily-CM).
Improvement in monthly headache days was the primary outcome measure used in the PREEMPT trials of onabotulinumtoxinA in CM (6), and is recommended for clinical use by National Institute for Health and Care Excellence guidelines for the use of onabotulinumtoxinA in CM in the United Kingdom (12). We used moderate-to-severe headache days as the primary outcome measure as it is one of the two recommended primary endpoints by International Headache Society guidelines for controlled trials of preventive treatment of CM in adults (27). The other recommended outcome measure, “migraine days”, was not possible to calculate using the headache diary utilised in this study which did not include all the migraine features required by ICHD-3 criteria, and is a limitation of this study. The 24 week follow up point was chosen as this is what has been used in previous studies of onabotulinumtoxinA in chronic migraine, including the PREEMPT studies (6). Interim observations prior to 24 weeks could have been useful to know whether the time to response differs between the groups. Longer follow up also would have been useful, as studies have shown that some patients can respond after 6 months of treatment (28).
Patient reported outcome measures are increasingly recognised as important in clinical trials. We found that response rates using patient estimate of improvement were higher than response rates based on any of the headache diary metrics (see Figure 1 and Table 2). This could be due to an improvement in non-headache symptoms such as nausea or photophobia, or that patients find improvements less than the 30% cut off to be clinically meaningful. Another consideration may be the quarterly interaction the patients have with a headache nurse, which may significantly enhance patients’ quality of life and their ability to cope with headaches, even if their headache disorder remains unchanged. Further work needs to investigate the disparity identified between headache diaries and patient's perception of treatment benefit. In future studies it would also be helpful to collect data on non-headache symptoms to identify whether these may have contributed to patient-reported improvements.
The lack of marked different response to treatment between NDPH with CM phenotype and transformed CM shown in this study may support the argument that NDPH is a subform of CM with a different mode of onset. The headache phenotype in NDPH often resembles CM in all other ways that the tempo of onset, and onset (other than post-traumatic) is not used as a classifier for other headache disorders (1). Other arguments for this are the similarities in demographics and comorbidities between NDPH and CM, and that many patients with NDPH have personal or family history of headache prior to onset of NDPH suggesting a shared predisposition (2,29). However, in a large study, patients with NDPH were found to be less likely to be female, have less migrainous, cranial autonomic symptoms, and aura, than patients with transformed chronic daily headache, and be less vulnerable to medication overuse, and these differences require investigation (30). In addition, NDPH is usually considered more difficult to treat than CM (3–5), and even invasive surgical treatment such as occipital nerve stimulation appears to rarely be effective in NDPH, in only 11% of patients compared to 45% of patients with CM (31,32). Further studies investigating differences in biomarkers between the two syndromes, and response to newer migraine treatments such as those targeting the calcitonin gene-related peptide pathway would be helpful in answering this question. There are several limitations to this study. Randomised controlled trials are the gold standard for establishing efficacy of a treatment. The current study was a non-randomised, uncontrolled, open label study. The study design has inherent limitations due to its inability to control for bias. There is known to be a large placebo response with injectable treatments for pain, and the placebo response was approximately 35% in the PREEMPT trials for onabotulinumtoxinA in non-daily-CM (6), meaning that the response in the NDPH group could potentially be largely explained by placebo effect. However, this study included a much more refractory patient population than was included in those trials and placebo rates in migraine are known to reduce with the more previous treatments that have previously failed (33). Patients in this study had a long duration of chronic daily headache prior to treatment (averaging more than 10 years in all three groups), and all had failed at least three (often many more) preventive medications already.
This was a single centre study with limited sample size, and multi-centre studies would be ideal both in addressing the issue of enrolment as well as the generalisability of the study results to the broader population with NDPH. The study compared outcomes between three diagnostic groups. There were differences in the demographics between the groups.
The greater female preponderance in both CM groups than in NDPH is expected based on previous literature (2,30). The differences in headache diary metrics between the non-daily-CM group and the other two groups in several headache diary metrics was also expected due to having fewer headache days by definition, and this was the reason that CM was divided into two groups for analysis. We attempted to account for differences in baseline characteristics using propensity score matching. However, unlike randomisation, propensity score matching can only balance the groups with respect to known confounders and not unknown confounders (unmeasured and unobservable confounders).
Given the known poor response of NDPH to oral preventive medications, the fact that a third of patients with refractory NDPH did respond to onabotulinumtoxinA, and that it is safe and well tolerated we recommend trialling treatment with onabotulinumtoxinA in patients with treatment refractory NDPH at a similar stage in the treatment pathway to that of CM. Further work is also required to identify treatments for the subgroup of patients who do not respond to either preventive medications or onabotulinumtoxinA.
Conclusions
In patients who were highly treatment refractory, onabotulinumtoxinA led to an improvement in moderate-severe headache days in 34.5% of patients with NDPH with CM phenotype, a rate which was comparable to daily CM. OnabotulinumtoxinA was safe and well tolerated, and therefore until other evidence for treatment of NDPH becomes available, we suggest that treatment with onabotulinumtoxinA may be trialled in patients with treatment-refractory NDPH with CM phenotype, and continuing to collect relevant outcomes in real-world setting to support its safety and efficacy. Other treatment options are required for the sizeable proportion of patients in whom neither oral preventive medications nor onabotulinumtoxinA are effective.
Article highlights
Approximately 1/3 of patients with NDPH responded to treatment with onabotulinumtoxinA
Response rates in NDPH were similar to daily chronic migraine
OnabotulinumtoxinA was safe and well tolerated with no serious adverse events experienced
Footnotes
Acknowledgements
We would like to acknowledge all the patients who agreed for their data to be included anonymously in this research study.
We would like to thank the headache nursing team for valuable help in collecting data: Susie Lagrata, Maha Ahmed, Rachel Pickering, Lyka Pacana, Dominique Penlaflor.
We would like to acknowledge Christian Pipper for his technical support using the twang package in R.
Availability of data and materials
Anonymised data used in this study are available upon reasonable request to the corresponding author by any qualified investigator.
Authors’ contributions
SC: conception and design of study, acquisition of data, analysis of data, interpretation of data, drafting of manuscript, revision of manuscript. SL: design of study, acquisition of data, interpretation of data, revision of manuscript. KRR: analysis of data, interpretation of data, revision of manuscript. MA: acquisition of data, revision of manuscript. SK: acquisition of data, interpretation of data, revision of manuscript. MSM: conception and design of study, acquisition of data, interpretation of data, revision of manuscript. All authors have approved the submitted version.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SC, KRR, and MA have nothing to declare. SL has received payment for the development and delivery of educational presentations from AbbVie and has received payment for the development and delivery of educational presentations from AbbVie, Eli Lilly, TEVA, Lundbeck and Tillots Pharma.
SK has received payment for the development and delivery of educational material from AbbVie and sat on an advisory board for Lundbeck.
MSM is chair of the medical advisory board of the CSF Leak Association; serves on the advisory board for AbbVie, Eli Lilly, Lundbeck, Pfizer Salvia, and TEVA; has received payment for the development of educational presentations from AbbVie, Eli Lilly, Lundbeck, Organon, Pfizer and TEVA; has received grants from Abbott, Medtronic and Ehlers Danlos Society; and has a patent on system and method for diagnosing and treating headaches (WO2018051103A1, issued).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
