Abstract
Hydrosoluble diclofenac epolamine (DHEP) represents an interesting approach to acute migraine attacks, where gastrointestinal motility and drug absorption are often reduced. Its efficacy was investigated in a randomized, crossover, double-blind trial on 155 patients who treated four consecutive mild-to-moderate migraine attacks, either with DHEP (65-mg sachet) or placebo. If pain was not relieved within 1 h, a second dose was given. The total number of treated attacks was 481. A pain-free condition was achieved within 2h in 45.8% and 25.1% of attacks treated, respectively, with DHEP or placebo (P < 0.0001), with a therapeutic gain of 20.7%. Time to attack resolution, light and noise sensitivity and impact on working ability were significantly reduced by DHEP compared with placebo. Moreover, significantly fewer patients required a second drug dose or a rescue medication when treated with DHEP than with placebo. No adverse reaction was recorded. In conclusion, DHEP was effective and safe for pain relief in patients with an acute mild-to-moderate migraine attack.
Introduction
Several non-steroidal anti-inflammatory drugs (NSAIDs) have been reported to be effective in the management of acute migraine, such as aspirin (1–4), paracetamol (5, 6), naproxen (7–13), ibuprofen (14, 15), ketoprofen (16), ketorolac (17), tolfenamic acid (18), mefenamic acid (6) and appropriate combinations of these compounds (18–22).
Among them, diclofenac is one of the most effective compounds, when administered either orally (23, 24) or intramuscularly (25). Dahlöf and Björkman (26) have reported that diclofenac potas sium (50–100 mg) is significantly more active than placebo in reducing pain intensity and improving phonophobia, photophobia, working ability and the need for rescue medication in patients with acute migraine.
Moreover, single oral doses of 50-mg and 100-mg diclofenac potassium have been compared with a single 100-mg oral dose of sumatriptan and placebo in a double-blind, randomized, crossover trial in 156 adult patients suffering from migraine attacks, either with or without aura (27). Diclofenac potassium was more effective than placebo in reducing migraine headache pain from 60 min up to 8 h after dosing, and no difference was observed between the 50- and 100-mg doses. A similar effect was shown with sumatriptan; however, significant superiority to placebo was seen only from the 90-min time point. Diclofenac potassium was generally superior to placebo or sumatriptan in reducing accompanying symptoms, particularly nausea. No difference was observed between sumatriptan and diclofenac in the primary outcome (27).
Diclofenac epolamine (DHEP)1 is a salt of diclofenac endowed with higher solubility and stability in water than the parent compound (28–30), thus allowing the pharmaceutical formulation of ‘Granulate’ (sachet). (DHEP brandnames: Switzerland, ‘Flector EP® granulate’, IBSA Institut Biochimique SA.; Hungary, ‘Flector EP® Rapid’, IBSAPharma Kft; France, ‘Flector®’, Laboratoires Genévrier; Czech Republic, ‘Flector EP® Rapid’, IBSA Institut Biochimique (SA); Italy, at present not on the market.) Our aim was to investigate the efficacy and tolerability of DHEP sachet in the treatment of mild-to-moderate acute migraine attacks without aura (common migraine), according to a multicentre, randomized, crossover, double-blind, placebo-controlled design. The experimental protocol was developed in agreement with the ‘Guidelines for controlled trials of drugs in migraine’, 2nd edition (31) and implemented at a EU Regulatory level by the ‘Note for guidance on clinical investigation of medicinal products for the treatment of migraine’ (32).
Methods
The study was authorized by the Independent Ethics Committee of each involved clinical centre. Briefly, patients of both genders, aged between 18 and 65 years, who were consecutively seen at the headache centres because of common migraine without aura (32), were selected if <50 years old at the onset of the first migraine attack; with a migraine history of at least 1 year and five attacks of mild-to-moderate severity, and a frequency of one to six migraine attacks monthly. Patients were excluded in the case of migraine of severe intensity; known hypersensitivity to diclofenac; concomitant treatment with drugs interacting with diclofenac; serious systemic or psychiatric diseases; drug abuse for headache; or antipsychotic or antidepressant medications in the 3 months prior to the study.
Patients were informed about the study and, after they had signed the informed consent form, were required to treat the next four consecutive acute migraine attacks with DHEP or placebo for two migraine attacks each, according to a randomized, crossover sequence which was computer generated using the validated software RANCODE Version 3.6 (IDV, Gauting/Munich, Germany).
A minimum of 2 days should have separated the attacks; otherwise, the second attack was considered a relapse of the first one and the drug treatment scored as unsuccessful.
DHEP was administered orally at the single dose of a 65-mg sachet (equivalent to 50 mg of diclofenac sodium); the content of one sachet was solved in half a glass of water, care being taken to dissolve the drug completely with a spoon. The drug was taken at the earliest signs of migraine attack and, when the pain relief was not felt to be enough 1 h after dosing, the patient was allowed to take a second dose. In the case of a migraine attack recurring within 48 h, the patient was allowed to treat this attack with his ‘usually used attack medicine’ (‘rescue medication’).
The patients were given a diary to be filled with the following information: (i) time and dose of experimental drug and any ‘rescue medication’ taken; (ii) assessment of pain by means of a 100-mm visual analogue scale (VAS) (0 mm, no pain; 100 mm, unbearable pain) and a 4-point scale (0, none; 1, mild; 2, moderate; 3, severe pain); (iii) the impact of headache on working ability (0, normal; 1, slightly impaired; 2, impaired; 3, impossible; the improvement was defined as the passage: from 3 to 2, 1 and 0; from 2 to 1 and 0; from 1 to 0); (iv) presence of vomiting, nausea, phonophobia and photophobia; (v) any adverse event (AE).
The primary end-point of efficacy was the number of patients pain free within 2 h after drug administration (31). Since a complete resolution of migraine within 2 h is largely considered unrealistic (32), treatment was considered to be successful when the VAS-measured pain was <20 mm 2 h after treatment. Whenever an attack was resolved within 2 h and relapses within 24 h, treatment was considered to be unsuccessful (32).
Since one of the International Headache Society guidelines for clinical trials is a frequency of attacks ranging between once and six times monthly, the study lasted 4 months at maximum. Basically, after patients had treated four consecutive migraine attacks, they were required to return to the centre for the final visit; in any case, the patient was advised to return to the centre after a maximum of 4 months, independently on the number of treated migraine attacks.
Statistics
The primary end-point variable was evaluated by Fisher's exact test. Null and alternative hypotheses were: (i) H0: the treatment efficacies were equal; (b) HA: the treatment efficacy of DHEP was greater than that of placebo. A one-sided significance level of 5% was used for the evaluation. Another analysis was performed for independence of treatment outcomes in a contingency table using a χ2 test: the null hypothesis was that the outcome in one treatment was independent of the outcome in the other treatment, i.e. that there were no systematic responders to one or the other treatment.
Severity of headache (VAS) was analysed with repeated-measure
Data recorded as written scales were then transformed into discrete scores and analysed according to non-parametric tests, either to evaluate differences within (Friedman test) or between groups (Kruskal–Wallis test), both vs. basal values.
Finally, the number of patients taking the second dose and/or escape medication was analysed using a χ2 Fisher test with the Yates' correction.
Results
Enrolled in this study were 155 patients. Of these, 110 had a full cycle of four acute migraine attacks treated with the investigational drugs (i.e. 440 treated attacks); 23 had only one to three acute migraine attacks treated (i.e. other 41 treated attacks). These patients were included in the intention-to-treat (ITT) analysis, which was therefore performed on a total of 481 attacks in 133 patients; they were 14 males and 119 females with a mean age of 37.7 ± 13.4 and 45.3 ± 11.3 years, respecively. For different reasons, data regarding the migraine attacks were missing for 22 patients, who were excluded from the analysis.
Preliminary tests for carry-over effects were conducted using the data from consecutive treatments and analysing the outcome of either treatment (for attacks 2, 3 and 4) as a function of the preceding treatment; however, no carry-over effect was observed. The second part of the analysis investigated how the success treatment was influenced by period of treatment, i.e. the number of the attack when the DHEP or placebo was used. No significant ‘period effect’ was observed.
The ITT analysis of efficacy showed that DHEP was successful in 45.8% of the 238 acute migraine attacks treated with it, and ineffective in the remaining 54.2% of attacks. In contrast, placebo was effective in only 25.1% of 243 acute migraine attacks, and ineffective in the remaining 74.9% of attacks. The therapeutic gain was therefore of 20.7% [95% confidence interval (CI) 12.3, 29.1] (Tables 1 and 2). The result was highly significantly in favour of DHEP (P < 0.0001).
Pain-free rates at 2 h postdose with diclofenac epolamine (DHEP) 65-mg sachet or placebo
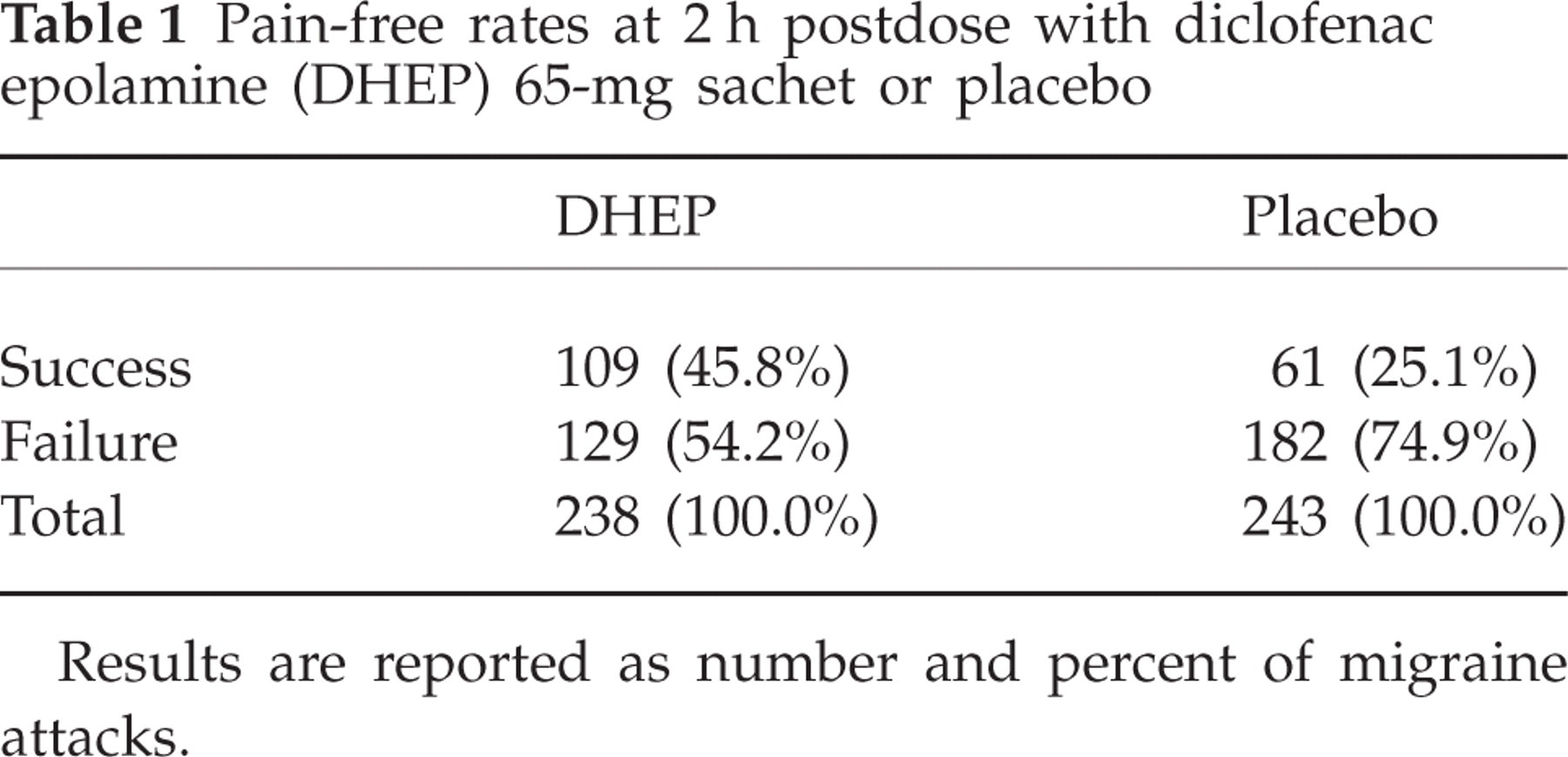
Results are reported as number and percent of migraine attacks.
Number of patients according to the number of attacks they successfully treated with diclofenac epolamine (DHEP) or placebo

In the DHEP group an improvement from severe/moderate to mild/no pain intensity was observed for 123 patients (54.4%); in the placebo group the same improvement was observed for 77 patients (33.8%). The therapeutic gain observed for this parameter was 20.65% (95% CI 11.8, 29.6).
The average time of resolution of the acute migraine attack (defined as the minimum of the times when the VAS value first fell below 20 and the duration of attack as recorded in the diary) was 4.48 ± 7.95 h for DHEP and 6.78 ± 10.94 h for placebo (P < 0.0001); the difference was statistically significantly in favour of DHEP (P < 0.0001, Wilcoxon test). Survival analysis of the time to resolution data yielded P < 0.0001 also in favour of DHEP (Fig. 1). The average pain vs. time was analysed by means of a repeated-measures
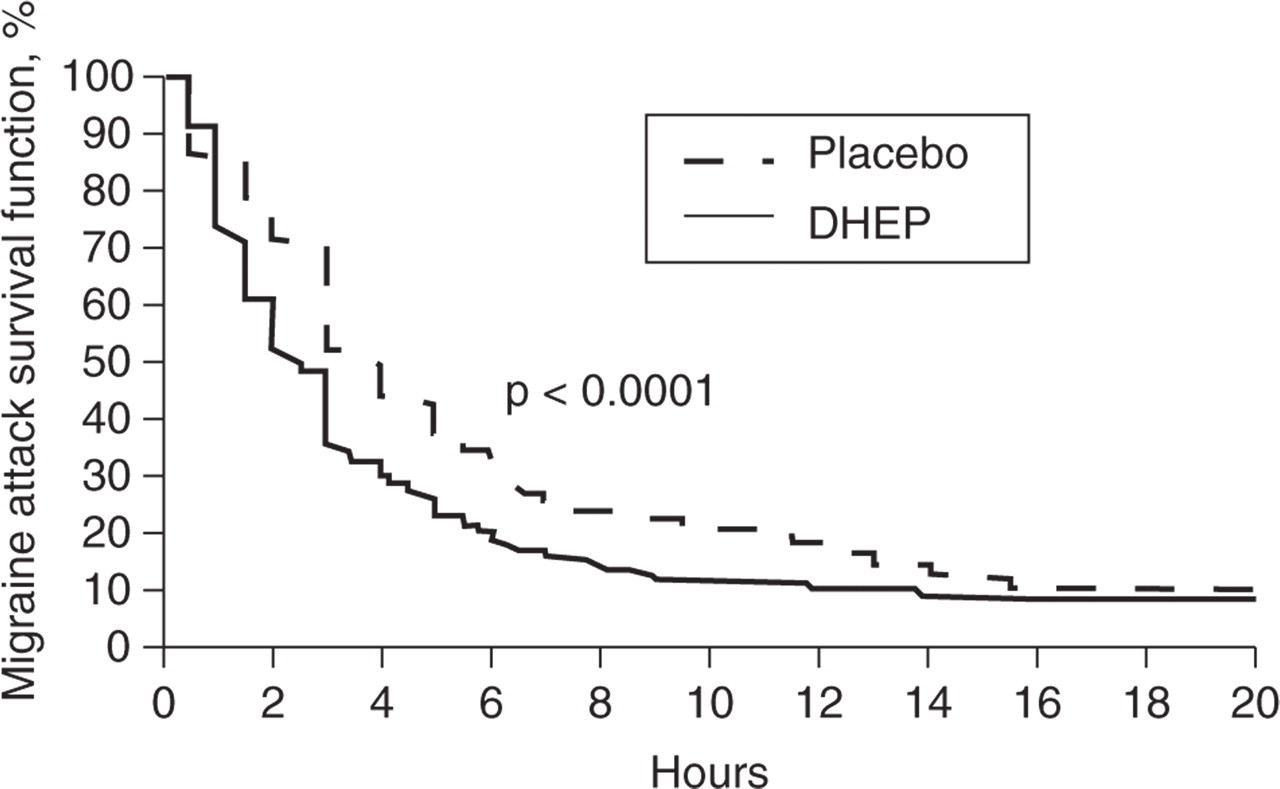
Kaplan–Meier plot of migraine attack survival functions during migraine attacks treated with diclofenac epolamine (DHEP) or placebo. Statistically significant difference between drugs: P < 0.0001, Wilcoxon test.
DHEP was generally more effective than placebo in the relief of the other migraine attack-related symptoms. Light hypersensitivity was reduced in 31.4% and 20.1% of patients treated, respectively, with DHEP and placebo, at 2 h postdose (P = 0.01). At the same time, DHEP was also significantly more effective than placebo in the relief of noise sensitivity: 31.1% of DHEP-treated patients improved compared with only 14.8% of patients receiving placebo (P = 0.0001). Nausea and vomiting also improved more frequently in the DHEP- (20.4% and 4.9% of patients, respectively) than in the placebo-treated group (18.9% and 2.6% of attacks, respectively) but, in both cases, the difference between groups only approached statistical significance (P = 0.051).
Another statistically significant difference between treatments was observed in the detrimental effects of acute migraine attacks on the working ability of patients. In total, 54.4% of patients improved with DHEP compared with only 39.8% with placebo (P = 0.0001 between treatments). The therapeutic gain observed for this parameter was 14.6% (95% CI 5.6, 23.7).
Finally, a second dose of investigational drug was taken during 78.4% and 63.6%, respectively, of the placebo- and DHEP-treated attacks: the difference between treatments was highly significant (P = 0.0005). Moreover, the patients resorted to their preferred ‘rescue medication’ in 56.7% and 30.5%, respectively, of the placebo- and DHEP-treated attacks: again, there was a highly significant difference between DHEP and placebo (P < 0.0001).
No serious AE possibly related to the active treatment with DHEP granulate was observed. One patient reported abdominal cramps after placebo and one reported stomach pain with both with DHEP and placebo.
Discussion
Migraine is a frequent, painful condition, which in many sufferers can be severely debilitating (33). Besides simple analgesics such as aspirin, paracetamol and ergotamine derivatives, NSAIDs are an effective therapeutic class in the acute treatment of migraine (34). In particular, diclofenac has been found to be very effective in the acute treatment of migraine (23).
Our study was performed according to a randomized, double-blind, placebo-controlled design, in agreement with the European Medicines Agency recommendations for clinical investigation of medicinal products for treatment of migraine (31). The results show a clinically evident greater efficacy of DHEP over placebo in the management of acute migraine attacks, as documented by a significantly higher number of migraine attacks which had relief from DHEP compared with placebo (45.8% vs. 25.1%). The high pain-free rates observed in our study for both verum and placebo were probably due to the fact that patients were treated early and had mild-to-moderate attacks.
Most of the secondary criteria for efficacy confirmed the trend in favour of DHEP, such as the shortest time to attack resolution, the lower scores of pain and a lower symptomatology observed in DHEP- than in placebo-treated patients. Moreover, the need for a second drug dose or rescue medication was significantly lower with DHEP than with placebo. There was not any single efficacy parameter where placebo proved superior, not even as a trend.
DHEP granulate was administered at the single dose of 65 mg (corresponding to diclofenac sodium 50 mg); a second dose was eventually administered within 1 h after the beginning of the acute migraine attack, whenever the pain was still felt as unbearable by the patient. These doses correspond to the recommended therapeutic doses of DHEP for other therapeutic indications (35).
A recent clinical study has shown that doses of 50 and 100 mg of diclofenac potassium were similarly effective in the management of acute migraine attack, and no better response was observed by increasing the dose of diclofenac. Thus, the 50-mg dose appears the most appropriate for pain relief in acute migraine attacks (27) and confirms the choice of the DHEP dose to be administered in an acute migraine attack (i.e. 65 mg, corresponding to 50 mg of diclofenac sodium).
Although the methods of efficacy evaluation were somewhat different between our trial and that with diclofenac potassium in similar patients with acute migraine attack (27), it is interesting to note that an average pain VAS score of 20 mm was obtained within 4.5 h with DHEP 65 mg (equivalent to 50 mg diclofenac) in our study, and with a double dose of diclofenac potassium (100 mg) in the Diclofenac-K/Sumatriptan Migraine Study Group, which also failed to achieve the same mean VAS score with the lower dose of diclofenac potassium (50 mg), even at later times (27).
Our results are especially noteworthy when one considers the large number of both primary and secondary criteria of efficacy that have been significantly modified by DHEP granulate, but not by placebo, as well as the well-known ‘placebo effect’ in migraine attacks (34).
In conclusion, the results of our clinical trial confirm that diclofenac epolamine is an effective and well-tolerated treatment for mild-to-moderate migraine attacks.
