Abstract
Background
New daily persistent headache (NDPH) is a challenging and understudied primary headache disorder with no known effective treatment. Although the International Classification of Headache Disorders criteria require that the new onset continuous headache be present for at least three months before diagnosing NDPH, the biologic basis for when a new, continuous headache starts to behave as NDPH is unknown, and some pediatric headache experts consider that the minimum duration criterion could be shorter.
Methods
In this retrospective study, we reviewed the intake questionnaires and medical records of 5–17 year-olds seen in neurology clinic for headache at the Children's Hospital of Philadelphia. Those with a new onset continuous headache of at least one month in duration were eligible. The patient's self-report and clinician's description both had to indicate that the headache was new, of abrupt onset, and continuous to be included, although patients were allowed to have a prior history of infrequent headaches. We compared headache outcomes at last follow-up and at one year after continuous headache onset between those who had a continuous headache duration of 1 to <3 months (“new onset headache”, or NOH) at first visit vs. those with ≥3 months (NDPH). We used multivariate regression modeling to examine for predictors of headache outcomes.
Results
Of 472 patient records reviewed, 172 met the inclusion criteria for analysis. Of these, 84 had a headache duration of 1 to <3 months in duration and 88 had a duration of ≥3 months. Those with shorter duration continuous headache were younger (median (interquartile range) 13.5 (11.1–15.7) vs. 15.1 (12.3–16.5) years, and less likely to have previously received a prescription preventive for the continuous headache (n = 14 (17%) vs. 26 (30%), p = 0.046), but were otherwise similar to those with NDPH in terms of baseline clinical and demographic variables. Sixty-five (74%) of those with NDPH and 60 (71%) with NOH had follow-up data. At last clinic follow-up, 41/65 (63%) with NDPH and 43/60 (72%) with NOH had experienced any headache benefit (p = 0.307), although 39/65 (60%) with NDPH and 29/60 (48%) with NOH still had continuous headache (p = 0.191). Headache duration was not associated with outcomes in multivariate regression modeling.
Conclusions
Headache outcomes of children and adolescents with new onset continuous headache, whether of 1 to <3 months (NOH) or ≥3 months in duration (NDPH) are suboptimal. More research is needed to improve treatment outcomes for this patient population.
This is a visual representation of the abstract.
Introduction
New daily persistent headache (NDPH) is defined by the International Classification of Headache Disorders, 3rd edition (ICHD-3) criteria as a new headache that has a “distinct and clearly remembered onset,” that became continuous within 24 hours of onset, and has been persistent for at least three months (1). NDPH can be highly disabling, is often refractory to treatment, and may occur in children and adolescents more often than in adults (2-5). There are no treatments proven to be effective specifically for NDPH in either adults or youth, and we are aware of no randomized, controlled treatment trials for youth with NDPH. This leads to treatment being based on the clinical characteristics of the headache with migraine treatment often utilized.
There is a general sense that NDPH is a difficult to treat headache disorder (4,6). Yet, the question of when a new onset continuous headache becomes hard to treat is unanswered. Although diagnostic criteria require the new onset continuous headache to have been present for ≥3 months before an NDPH diagnosis is made, whatever biology it is that makes NDPH challenging to treat may be present well before the three-month mark. In a survey of pediatric headache specialists, 39% (24/63) thought that the minimum duration criterion for NDPH should be shorter than three months, and 13/24 suggested it should be one month (7).
In the present study, we set out to determine whether headache trajectories differ in youth who present to care after 1 to <3 months duration of new onset continuous headache vs. ≥ 3 months in duration. We hypothesized that youth with 1 to <3 months duration of new onset continuous headache would have similar headache trajectories to those with ≥3 months duration.
Methods
Design
This was a chart-review based cohort study of children and adolescents aged 5–17 years who were seen in neurology clinic at Children's Hospital of Philadelphia (CHOP). This is the primary analysis of these data on this question.
Ethics and approval
Headache questionnaire data were collected clinically and extracted from the electronic health record into the CHOP Headache Registry with approval from the CHOP institutional review board (IRB), with a waiver of the requirement for informed consent in order to include patients seen by all neurologists across all locations.
Inclusion criteria
Patients aged 5–17 years old with possible NDPH, or shorter duration new onset headache (NOH), were identified based on patient responses on the CHOP Headache Questionnaire. This questionnaire is offered in the electronic health record to any new patient who self-reports that headache is the primary reason for the visit (8). Although the instructions request that the child answer the questions, it is assumed that family members assist with completing the headache questionnaire for younger children and possibly older adolescents as well. In the present study, the term “patient response” includes responses given by the patient and/or their family. Answers indicated abrupt onset of continuous headache of ≥1 month duration without escalating frequency of prior headache and without preceding head injury. We required that patients report either the date of onset or age of onset of continuous headache in the questionnaire. All charts were reviewed by one of the investigators (ME) to validate that the patients’ answers were concordant with the clinicians’ evaluation of headache pattern. Any chart that was thought to represent NDPH/NOH was subsequently reviewed by a fellowship-trained pediatric headache specialist (CLS). Only those where both patient and clinician history indicated NDPH/NOH were included in this analysis.
Data collection
Our team developed a standardized data abstraction form for this study in a secure REDCap (Research Electronic Data Capture 9) database. We piloted data collection by reviewing approximately 25 charts and iteratively refined the data collection form to ensure feasibility before abstracting all the charts. Data collected included:
Duration of continuous headache since onset – per patient questionnaire or clinical notes if not supplied in questionnaire. Age, legal sex, race, ethnicity – as reported in the electronic medical record. Self-reported depression and anxiety. Other chronic pain condition and/or hypermobility reported in the medical record. Family and personal history of headache prior to onset of new continuous headache. Headache severity, based on a 0–10 numerical scale as well as mild/moderate/severe. Migrainous headache phenotype vs. not. This determination was based on whether ≥3 of the four ICHD-3 criteria (A through D) for migraine were met. Perceived trigger to continuous headache (patient reported). Headache treatments tried prior to the visit (type and number). Headache-related disability – as measured by Pediatric Migraine Disability Assessment (PedMIDAS)10,11 score at initial visit (scoring: 0–10 = little or no disability, 11–30 = mild, 31–50 = moderate, > 50 =severe). Headache outcomes at: (i) last clinic follow-up and at (ii) one year after onset of continuous headache. Headache outcomes were abstracted from the clinical notes. For headache outcomes at one year, if the last follow-up visit took place at ≤1 year from continuous headache onset, we carried forward the outcome information from the last follow-up. Those who had already had continuous headache for ≥1 year at the time of their initial clinic visit were excluded in these analyses. “Significant improvement” in headache was defined as at least 4 weeks duration of at least 30% improvement in severity, frequency and/or disability. “Some improvement” was if some degree of improvement was recorded, but it did not meet criteria for “significant improvement”. Proportion with non-continuous headache – we extracted time to first headache-free moment from the chart.
For all patient reported variables, if the information was missing in the questionnaire, it was filled in from the medical record if possible.
Statistical analysis
There was no a priori power calculation. The sample size was determined based on the number of available patients who met inclusion criteria. Descriptive variables are described as means, medians, proportions, etc. as appropriate. Values are rounded to the degree of precision with which they were measured. If a continuous variable was not normally distributed, medians are reported. Normality was assessed using tests for skewness and kurtosis. The Wilcoxon rank sum test was used to compare medians of non-normal continuous data, and t-tests were used to compare means of normally distributed data. Chi-squared testing was used to assess distribution of categorical data, unless at least one cell had a count of <5 where Fisher's exact test was used. All hypothesis testing was two-tailed. p < 0.05 was considered statistically significant. No adjustments for multiple comparisons were made. Analyses were conducted in Stata, version 17 (StatCorp, College Station, TX, USA) and Excel 2016 (Microsoft Corp., Redmond, WA, USA).
We used univariate logistic regression to test for association between all predictors and several outcomes. Predictors included duration of continuous headache (one month (meaning ≥28 days) to <3 months vs. ≥ 3 months as the primary predictor), age, age at first headache, legal sex, race, ethnicity, self-reported depression, self-reported anxiety, self-reported or chart documentation of other pain condition, self-reported or chart documentation of hypermobility, family history of headache/migraine, headache pattern prior to onset of continuous headache (none, only when sick, some prior headaches), headache severity (severe vs. mild/moderate and as a continuous variable on a 1–10 scale), headache phenotype (migrainous vs. not), disability measured by PedMIDAS (none, mild, moderate, severe), treatments prior to visit, and perceived trigger for continuous headache.
The following multivariate models were run based on our original hypothesis that there would be a difference in outcomes based on duration of <3 months vs. ≥ 3 months:
Any improvement in headache at last clinic follow-up. Any improvement at one year after onset of continuous headache (excluding those who presented to clinical care at ≥12 months after onset of continuous headache). No longer experiencing continuous headache at last clinic follow-up. No longer experiencing continuous headache at 6 months after initial clinic visit.
All predictors with p < 0.1 in the univariate analyses were included in the initial multivariate model, and backwards elimination was used. Odds ratios and 95% confidence intervals were calculated. The primary analysis excluded those with missing outcome data.
Sensitivity analysis
Sensitivity analyses were conducted assuming (i) all with missing outcome data had improvement (i.e. “best case scenario”) and (ii) all with missing outcome data did not improve (i.e. “worst case scenario”). In addition, when the above models did not show significant difference based on duration of continuous headache (<3 months vs. ≥ 3 months), we conducted post hoc analyses to examine for a more informative time point cutoff for duration of continuous headache (4 to <8 weeks, 8 weeks to <3 months, 3 months to <6 months, 6 months to <12 months, ≥ 12 months) with a similar approach to missing data as above.
Proportion with non-continuous headache over time
We generated time-to-event curves (Kaplan Meier curves) based on the duration of continuous headache from the first clinic visit to the first pain-free moment. For patients who had at least one follow-up visit but no documented pain-free moment by the time of the last visit we set days of continuous headache to 1500. We then repeated this analysis assuming that all patients without a follow-up visit had pain-free time one day after the initial visit.
Results
In total, 472 patients aged 5–17 years self-reported new continuous headache of at least one month in duration in their new patient headache questionnaire. After review of clinical charts, 172 met the inclusion criteria for analysis. Of these, 84 had a headache duration of 1 to <3 months (NOH) and 88 had a duration of ≥3 months (NDPH).
Baseline clinical and demographic characteristics for the n = 172 patients are shown in Table 1, broken down by headache duration. Those with NOH were younger than those with NDPH, with median (interquartile range (IQR)) age of 13.5 (11.1–15.7) years vs. 15.1 (12.3–16.5, p = 0.029), and were less likely to have previously received a prescription preventive treatment prior to their initial neurology clinic visit (14/84 (17%) vs. 26/88 (30%), p = 0.046). Demographic and clinical variables were otherwise similar.
Baseline clinical and demographic characteristics of n = 172 patients with new onset continuous headache.
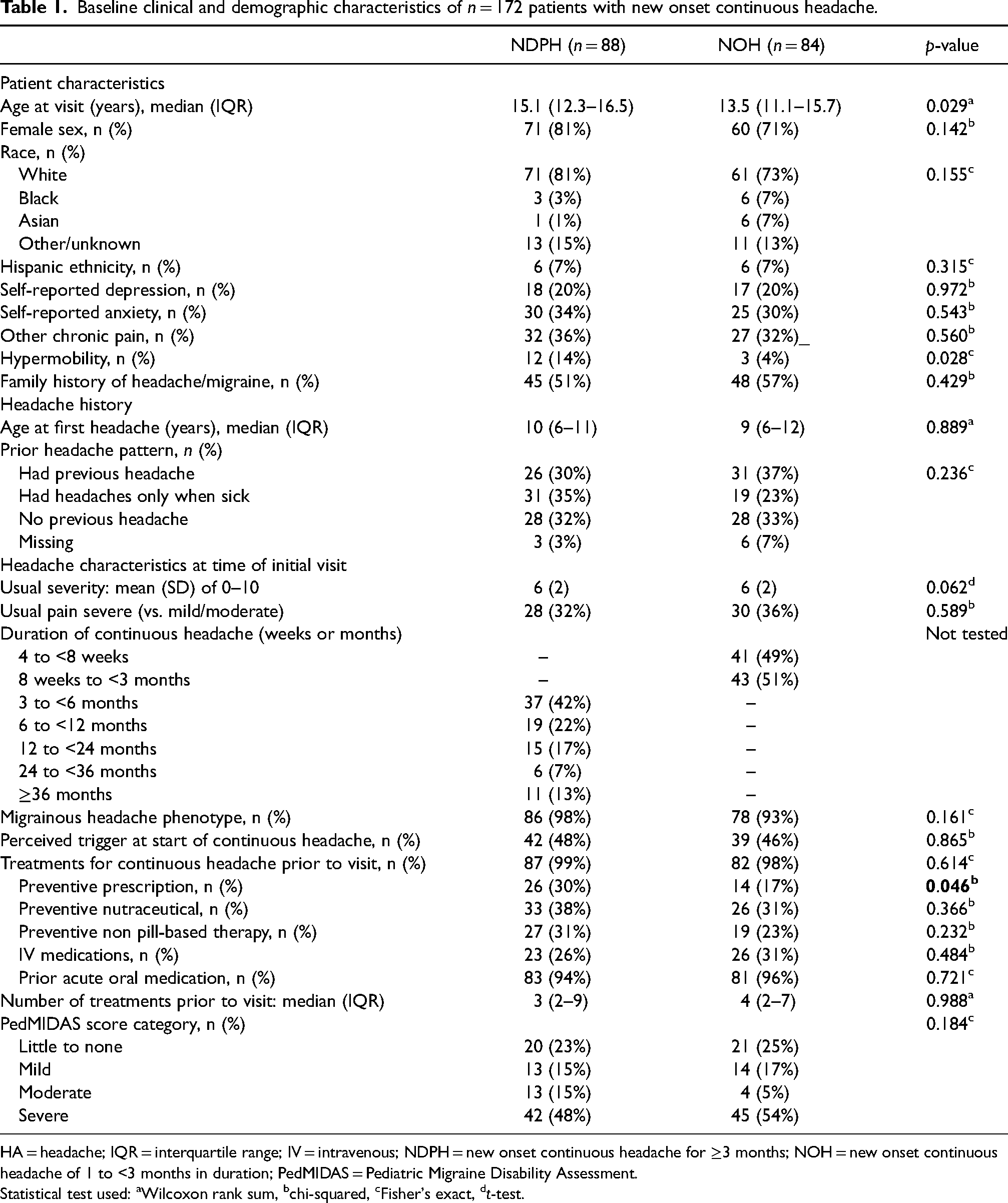
HA = headache; IQR = interquartile range; IV = intravenous; NDPH = new onset continuous headache for ≥3 months; NOH = new onset continuous headache of 1 to <3 months in duration; PedMIDAS = Pediatric Migraine Disability Assessment.
Statistical test used: aWilcoxon rank sum, bchi-squared, cFisher's exact, dt-test.
Headache outcomes at the time of the last clinic follow-up, and at one year after onset of continuous headache (for those who had not already passed this milestone by the time of their initial clinic visit), are shown in Table 2. Median duration of continuous headache was longer in the NDPH group at the time of last follow-up (30.4 (19.3–51.1) vs. 13.1 (4.9–28.9) months, p < 0.001). Outcomes at last clinic follow-up and at one year did not differ between groups, including in sensitivity analyses assuming that all those with missing follow-up data had improved (i.e. “best case scenario”) and when assuming that those with missing follow-up data had not improved (i.e. “worst case scenario”).
Headache outcomes at time of last clinic follow-up visit and at one-year after onset of continuous headache

*Those who already had ≥1 year of continuous headache at the time of their first clinic visit were excluded. IQR = interquartile range NDPH = new onset continuous headache for ≥3 months; NOH = new onset continuous headache of 1 to <3 months in duration.
Statistical test used: aWilcoxon ranksum, bchi-squared, cFisher's exact, dt-test.
Table 3 shows multivariate regression modeling results for outcomes of: (i) any headache improvement at last clinic follow-up; (ii) any improvement at one year after onset of continuous headache (excluding those who presented to clinical care at ≥12 months); (iii) no longer having continuous headache at last clinic follow-up; and (iv) no longer having continuous headache within 6 months of initial clinic visit, with NOH vs. NDPH as the primary predictor variable. None of these headache outcomes differed by duration group. Multiple models suggested that history of another chronic pain disorder and more treatments tried before the first visit were associated with decreased odds of positive outcome. By contrast, family history of headache and/or migraine was associated with increased odds of favorable outcome. The presence of anxiety was associated with increased odds of headache improvement by one year, whereas depression was not associated with outcomes. The findings were unchanged when headache duration was further broken down into five categories in post-hoc analysis (data not shown).
Multivariate model results examining predictors of headache improvement
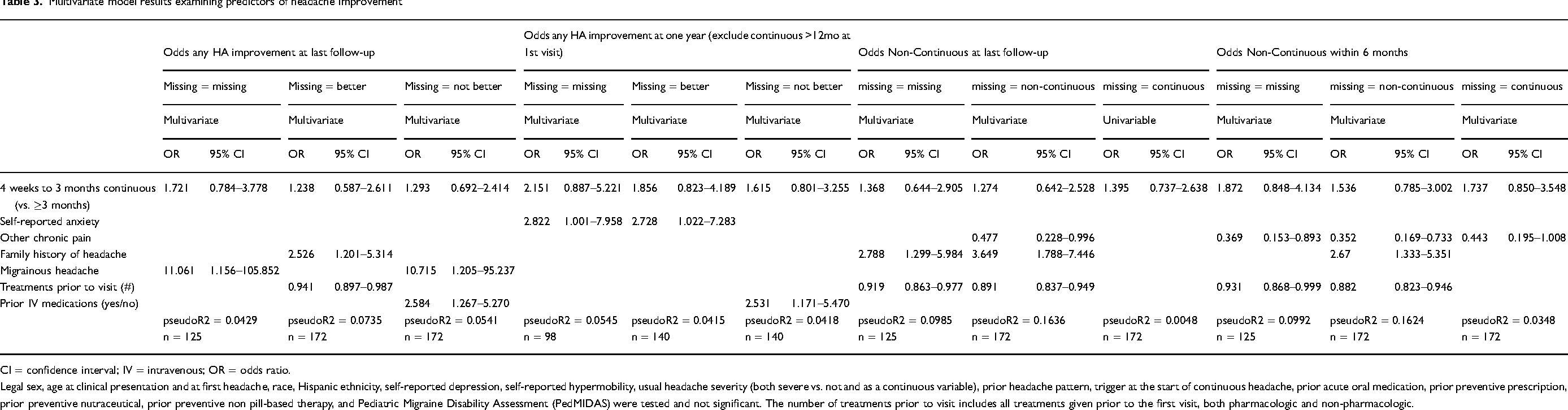
CI = confidence interval; IV = intravenous; OR = odds ratio.
Legal sex, age at clinical presentation and at first headache, race, Hispanic ethnicity, self-reported depression, self-reported hypermobility, usual headache severity (both severe vs. not and as a continuous variable), prior headache pattern, trigger at the start of continuous headache, prior acute oral medication, prior preventive prescription, prior preventive nutraceutical, prior preventive non pill-based therapy, and Pediatric Migraine Disability Assessment (PedMIDAS) were tested and not significant. The number of treatments prior to visit includes all treatments given prior to the first visit, both pharmacologic and non-pharmacologic.
Figure 1 shows the proportion of patients who had a break in continuous headache over time, grouped by duration of continuous headache at the time of first clinic visit. Time zero represents the day of the first neurology clinic visit. Most patients who developed non-continuous headache did so within six months of their first clinic visit.

Time to event curves showing proportion with non-continuous headache based on time elapsed from first clinic visit (days). (A) Comparison of those with NOH vs. NDPH at first clinic visit. NOH = new onset headache; NDPH = new daily persistent headache. n = 125 (those without follow-up were excluded). (B) Comparison of NOH vs. NDPH, those without follow-up were assumed to have non-continuous headache starting one day after first clinic visit (n = 172).
Discussion
This study examined the headache outcomes of 5–17 year-olds with new onset continuous headache of 1 to <3 months in duration (i.e. “new onset headache”, or NOH) vs. ≥ 3 months (i.e. NDPH) at the time of their initial neurology clinic visit. Our hypothesis was that headache trajectories would be similar between groups, and indeed they were both at one year after headache onset and at last clinic follow-up. There was also no difference in terms of proportion who had a break in continuous headache at six months and at last clinic follow-up. In multivariate regression modeling, there were no strong, consistent predictors of headache improvement at one year or at last clinic follow-up.
In keeping with a common clinical perception about NDPH,2-4 headache outcomes were relatively poor overall. In both groups, many patients were still experiencing continuous headache at last clinical follow-up, though the majority had experienced some degree of headache improvement. Based on the time to event curves (Figure 1), most who had a break in continuous headache had this occur within the first six months after their initial neurology clinic visit, regardless of their duration of continuous headache before the visit. However, there was a long tail of improvement.
As clinical follow-up duration was variable, examining outcomes at one year after continuous headache onset is also informative. Those who had already had ≥1 year of continuous headache at the time of their first clinic visit needed to be excluded from such analyses; thus, the results may not generalize to this group. However, of the subset who had not yet reached the one year milestone at the time of first neurology assessment, accounting for “best case” to “worst case” scenario for missing data, 39–71% in the NDPH group and 52–81% in the NOH group had any degree of headache improvement at one year, with no significant difference between groups.
Previous studies have reported on demographic and clinical characteristics of adolescents with NDPH who present to neurology clinic care, however, these studies have typically been cross-sectional (12-14). As in our study, in the literature the majority of youth with NDPH have been female and have reported a migrainous phenotype to their headache (12-14). In terms of NDPH outcomes, Reidy et al. reported on headache trajectories in a sample of 782 children and adolescents with continuous headache for at least one month in duration at the time of their first clinic visit. Of the 782, 116 (14.8%) had NDPH, whereas the majority had chronic migraine. Follow-up data (at 4–16 weeks) were available for 526 (67.3%). Of these, 280/526 (53.2%) still had continuous headache at follow-up. Having NDPH was a negative prognostic indicator. Broken down by diagnosis, 59/79 (75%) with NDPH still had continuous headache at follow-up vs. 220/439 (50%) with chronic migraine (15). Adult cohorts with NDPH have also been predominantly female and with a migrainous phenotype headache. Outcomes in adults with NDPH appear to be generally suboptimal, with many experiencing a treatment refractory course (4,16). In a retrospective chart review study of 46 children and adolescents with NDPH seen at a headache clinic, predictors
Putting the current study findings into clinical practice, child neurologists could counsel patients and families whose child's continuous headache duration has been less than one year at the time of initial visit that six or seven out of 10 patients in this situation will see some degree of headache improvement by one year. When looking at all comers, headache outcomes at last clinic follow-up were similar to the above; however, because the time to last clinical follow-up varied in this study, it is more challenging to translate this into concrete prognostic counseling. However, and perhaps most importantly from a patient perspective, many of the patients in this study still had continuous headache at last clinic follow-up, (68/125; 54% of those who had follow-up) despite being cared for by child neurologists at a tertiary care children's hospital.
Our study findings suggest that whatever biology it is that makes NDPH hard to treat is likely already present before the one month mark
There are several limitations inherent in retrospective chart review studies such as this one. There were substantial amounts of missing follow-up data, which we attempted to mitigate by performing sensitivity analyses
Another limitation is that some clinical information was missing from clinical notes, even among those with did come for follow-up. Additionally, our analysis focused predominantly on headache outcomes, and improvement in other meaningful symptoms such as photophobia, nausea, etc. was not captured. A prospective study of youth with NDPH is needed to better understand the outcomes of this disease in children and adolescents.
To optimize the likelihood that we included only those with new onset continuous headache, we limited our analysis to those where both the patient and the clinician's description of headache onset matched and indicated it was a new onset continuous headache. It is possible that, in some cases, clinicians may not have ascertained the pattern of headache onset correctly or documented it clearly enough, leading to the unnecessary exclusion of some patients. We may also have been underpowered to detect some differences in outcomes based on headache duration.
In conclusion,
Clinical implications
In this retrospective cohort study of headache outcomes in children and adolescents with new onset continuous headache, outcomes did not differ between those with headache duration of 1 to <3 months vs. ≥ 3 months at initial neurology visit. Clinicians can counsel patients and families that approximately six or seven out of 10 can expect to experience at least some degree of headache improvement by one year.
Footnotes
Declaration of conflicting interests
CS: Dr Szperka or her institution have received compensation for serving as a consultant for AbbVie and Teva. She has received personal compensation for serving on a data safety monitoring board for Eli Lilly and Upsher-Smith. She has also received research support from the NIH NINDS (K23NS102521), PCORI and the CHOP CARES/ Doris Duke COVID-19 Fund to Retain Clinical Scientists. ME: None. CPG: Dr Patterson Gentile has received salary support from grants from the American Academy of Neurology Clinical Research Training Scholarship in the past 24 months, and is currently funded by the National Institutes of Health/National Institute of Neurological Disorders and Stroke (K23 NS124986). PP: None. NR: None. BMP: None. ADH: Dr Hershey or his institution have received compensation for serving as a consultant for AbbVie, Amgen, Biohaven, Eli Lilly, Lundbeck, Supernus, Teva, Theranica and Upsher-Smith. His institution has also received research support from Amgen, Biohaven, Eli Lilly, Theranica, Upsher-Smith, and the NIH NINDS/NICHDS. AAG: In the last 24 months, Dr Gelfand has received honoraria from UpToDate (for authorship) and from the Headache Cooperative of the Pacific and the Taiwan Headache Society for speaking. She receives payment to her institution from the American Headache Society for her role as Editor of Headache. She receives grant support from PCORI as a member of the Steering Committee for the REACH study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Grant from the International Headache Society for seed funding in Child & Adolescent headache research, supplemented by funding from the CHOP CARES/Doris Duke COVID-19 Fund to Retain Clinical Scientists. Dr Szperka & Dr Patterson Gentile received research support from NIH NINDS (K23NS102521; K23 NS124986). Ms Evans was a participant in the Children's Hospital Research Institute Summer Scholars Program (CRISSP), funded by R25HD101365 from National Institute of Child Health and Human Development, as well as the CHOP Research Institute.
Ethical statement
The CHOP Headache Registry, approved by the CHOP institutional review board (IRB), with a waiver of informed consent, includes data from all patients seen by neurologists across all locations.
