Abstract
Background
Migraine with aura (MWA) is a risk factor for stroke, but the mechanisms underlying this association remain unclear. Our aim was to assess the association between MWA and cerebral small-vessel disease (CSVD) ischemic stroke after adjustment for vascular risk factors in a population of young patients hospitalized for a first-ever ischemic stroke.
Methods
Patients aged 18–54 years consecutively hospitalized for a first-ever acute ischemic stroke at the neurovascular unit of our university hospital between January 2017 and July 2021 were included in this retrospective cohort study. CSVD lesions were assessed and classified according to ASCOD (Atherosclerosis, Small-Vessel Disease, Cardiac pathology, Others causes, Dissection) classification criteria.
Results
In total, 646 patients were included (median (SD) age, 44.03 (9.01) years; 61.8% male) including 115 patients with MWA and 110 patients with migraine without aura (MWoA). Grade S1, potentially causal, CSVD lesions were significantly less frequent in patients with MWA (odds ratio (OR) = 0.35, 95% cofdence interval (CI) = 0.13–0.95, p = 0.048) compared to non-migraine patients in univariate analysis. Logistic regression adjusting for vascular risk factors showed no significant association of CSVD of any grade (S1, S2 or S3 vs. S0) with migraine: OR = 0.78, 95% CI = 0.48–1.28, p = 0.34; MWoA: OR = 0.81, 95% CI = 0.42–1.47, p = 0.51; and MWA: OR = 0.84, 95% CI = 0.43–1.56, p = 0.60, as well as no association of grade S1 CSVD lesions with migraine: OR = 0.91, 95% CI = 0.40–1.92, p = 0.81; MWoA: OR = 1.11, 95% CI = 0.42–2.64, p = 0.81; and MWA: OR = 0.72, 95% CI = 0.20–1.98, p = 0.56.
Conclusions
In a retrospective study including almost 650 young adults hospitalized for a first ischemic stroke, MWA was not associated with CSVD cause of stroke after adjustment for vascular risk factors.
This is a visual representation of the abstract.
Introduction
The incidence of ischemic stroke among young adults, aged under 55 years, has been steadily increasing worldwide for over the past twenty years (1,2). Traditional vascular risk factors, such as hypertension, diabetes, overweight, low physical activity, hyperlipidemia, smoking or alcohol use increase the risk of stroke in young adults (3,4). Another leading risk factor for ischemic stroke in young people is migraine with aura (MWA). This risk is approximately doubled, regardless of traditional vascular risk factors (5–8). The risk is higher in women and increases with smoking and estrogen-progestin contraception use. The mechanisms underlying this association are still unclear and are probably multifactorial. Research has help to unveil part of the origin of this association by showing an increased risk of patent foramen ovale (PFO) and of atrial fibrillation (AF) in MWA (9–11). Other hypotheses are evoked such as focal hypoperfusion during the course of cortical spreading depression (12,13) and hypercoagulability (14,15). Based on the observations that MWA may be associated with an increased risk of cerebral small-vessel diseases (CSVD) markers (16), such as white matter (WM) hyperintensities (17–21) and silent infarctions (17,22–24), and that MWA is part of the phenotype of genetic small-vessel disease such as cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL) (25,26), it has been hypothesized that MWA was associated with CSVD cause of stroke. However, the association of MWA with stroke due to CSVD has been little investigated and remains unclear. Therefore, we aimed to evaluate the association of MWA with ischemic stroke due to CSVD, according to the ASCOD (Atherosclerosis, Small-Vessel Disease, Cardiac pathology, Other causes, Dissection) classification system, after adjustment for traditional vascular risk factors, in a large population of young adults with a first ever ischemic stroke.
Methods
Ethics
The study conformed to the principles outlined in the Declaration of Helsinki. All patients were informed that the clinical data collected during their hospitalization could be used for research purposes and gave their oral consent. The study was approved by our Institutional Review Board (internal reference RnIPH 2020-28). Because the study was part of routine clinical care, no written consent was required.
Study design
In this case–control study, patients aged 18–54 years hospitalized for a first-ever ischemic stroke in the Department of Neurology of our University Hospital were included from January 2017 to July 2021. Data were collected consecutively and were analyzed retrospectively from our institution's electronic database. Exclusion criteria included recurrent stroke, cerebral hemorrhage, cerebral venous thrombosis and transient ischemic attacks without evidence of infarction on brain imaging. Patients not assessable for migraine status at the time of stroke or at follow-up were not included.
Stroke work-up
Initial evaluation included cerebral magnetic resonance imaging (MRI) or computed tomography (CT) if MRI was contraindicated, non-invasive angiography of cerebral and cervical arteries by magnetic resonance or CT possibly supplemented by carotid duplex ultrasound and transcranial color coded sonography, electrocardiogram (ECG) and routine blood tests (complete blood count, prothrombin time, activated partial thromboplastin time, fibrinogen, D-dimer, C-reactive protein, serum creatinine, glucose, cholesterol and triglyceride concentrations). Axial T1-weighted MRI with fat saturation was performed to diagnose arterial dissection. ECG monitoring by telemetry for 72 hours was performed in patients with no definite cause of stroke on initial work-up. Transcranial Doppler testing for right-to-left shunt, as well as transthoracic and transesophageal cardiac echocardiography, was performed in patients who did not have a definite cause of stroke on initial assessment. The search for AF was completed by ambulatory 24-hour Holter ECG monitoring and, in selected cases, long-term recordings with Holter monitor or an implantable loop recorder. Depending on the clinical context, the work-up could include a search for arterial or venous thrombophilia, a homocysteine assay, cerebrospinal fluid analysis, a search for an active malignant tumor, a search for genetically determined small-vessel disease, digital substraction angiography and vessel wall MRI. The work-up was left to the discretion of the practitioner in charge of the patient.
CSVD was assessed on MRI or on CT. Four types of CSVD imaging markers were evaluated on MRI. Microbleeds were defined as round hypointensity of 5 mm or less in diameter on susceptibility weighted imaging or gradient echo images. WM hyperintensities were identified by fluid-attenuated inversion recovery (FLAIR) images. WM hypersignals were graded according to the Fazekas scale (27) from absence (0) to confluence of lesions (3). Enlargement of the perivascular spaces was defined as round lesions of the same intensity as the cerebrospinal fluid, with a diameter less than or equal to 3 mm on the T2 images. Lacunar infarctions were defined as hypersignals on T2 imaging and hyposignals with surrounding hyperintensity on FLAIR imaging between 3 mm and 15 mm in diameter in the territory of a perforating artery. All neuroimaging examinations were performed at our center and were routinely interpreted by senior neuroradiologists.
All individual data were reviewed by a senior vascular neurologist for stroke diagnosis and classification. Stroke causes were classified using the ASCOD (Atherosclerosis, Small-Vessel Disease, Cardiac pathology, Other causes, Dissection) classification system (28). Grade S0 was defined as the absence of CSVD on exhaustive work-up. A grade S1 (potentially causative) was defined by the combination of a lacunar infarction corresponding to the patient's symptoms associated with one or several small deep infarcts of lacunar type in other arterial territories and/or WM hypersignals graded Fazekas 3 or microbleeds or dilatations of the perivascular spaces realizing “état criblé” and/or repeated and recent, less than one month, transient ischemic attacks attributable to the same arterial territory as the index stroke. Grade S2 (uncertain causality) was defined by the presence of a recent lacunar infarction with no other imaging abnormalities or by a clinical presentation suggesting being related to ischemic stroke of a perforating artery. A grade S3 (unlikely causal link, but the disease is present) was defined by WM hypersignals graded Fazekas 3 and/or presence of microbleeds and/or dilatation of perivascular spaces and/or one or several old, small deep infarcts of lacunar type, when the symptomatic stroke was not lacunar. A grade S9 was defined when the work-up was insufficient to classify the disease.
Migraine assessment
All patients were systematically evaluated by two headache specialists to establish the diagnosis of MWA and migraine without aura (MWoA) according to the International Headache Classification, 3rd edition (29), using a semi-structured questionnaire. Migraine assessment was performed during the initial hospitalization or remotely at the follow-up visit for patients with initial aphasia who had recovered secondarily. Patients who could not be asked about their migraine status were not included. To avoid information bias, we did not retain the diagnosis of migraine based on medical history or family questioning.
Vascular risk factors
Overweight was defined as a body mass index ≥25 kg/m2. Hypertension was defined by persistent systolic blood pressure >130 mmHg or diastolic blood pressure >80 mmHg documented prior to stroke, or treatment with antihypertensive medication prior to stroke, or diagnosed at the time of stroke. Diabetes was defined as a previous or new diagnosis of type 1 or type 2 diabetes according to the 2019 recommendations of the American Diabetes Association (30). Smoking was considered in case of current smoking over the past year. Alcohol misuse was defined as alcohol consumption of more than 40 g/day in men and 20 g/day in women according to World Health Organization recommendations on alcohol consumption (31). Hyperlipidemia was defined as previously treated hyperlipidemia, or newly diagnosed elevated low-density lipoprotein cholesterol >1.6 g/L or hypertriglyceridemia >2.0 g/L.
Data analysis
No statistical power calculations were performed prior to the study and the sample size was based on available data. Continuous variables were described by their mean ± SD if the distribution was normal, or their median and interquartile range. Categorical variables were described by the absolute number of events observed and their percentage. The distribution of continuous variables was visually assessed using histograms. Univariate analysis was performed using the Wilcoxon test, the chi-squared test or Fisher's exact test. After univariate analysis, the association of migraine, MWA or MWoA (independent variable) and CSVD (dependent variable), compared to no migraine, was tested using logistic regression. Cofactors included were age, sex, current smoking, overweight, hyperlipidemia, arterial hypertension, alcohol misuse and diabetes. CSVD was tested as a binary dependent variable including only the S1 potentially causative grade (S1 vs. S0) as primary outcome or any grades (S1 + S2 + S3 vs. S0) as secondary outcome. All regression analyses were reported as unadjusted OR and adjusted OR. The analysis represents the OR on the outcomes for patients with migraine (or MWoA or MWA) vs. non-migraine, with non-migraine as the reference category. All tests were two-tailed. p < 0.05 was considered statistically significant. Statistics were produced using Prism, version 9.5.1 (GraphPad Software Inc., San Diego, CA, USA).
Results
Patient characteristics
In total, 751 patients aged 18–54 years were hospitalized for a first ever ischemic stroke at the neurovascular unit of the Toulouse University Hospital during the study period. One hundred and five patients were not included. Reasons for non-inclusion were stroke leading to death (n = 18), persistent severe aphasia (n = 21), language barrier (n = 20), cognitive or psychiatric disorders (n = 17) and missing data (n = 29). Of the 646 patients included (median age (SD), 44.03 (9.01) years; 61.8% male), 110 (17%) had MWoA and 115 (17.8%) MWA (Figure 1). Patient characteristics are summarized in Table 1. Of the included patients, 10/646 (1.5%) were explored using CT (MRI was contraindicated), non-invasive angiography of cerebral and cervical arteries by magnetic resonance or CT (240/646 (37.2%)) possibly supplemented by carotid duplex ultrasound and transcranial color-coded sonography (388/646 (53.9%)).
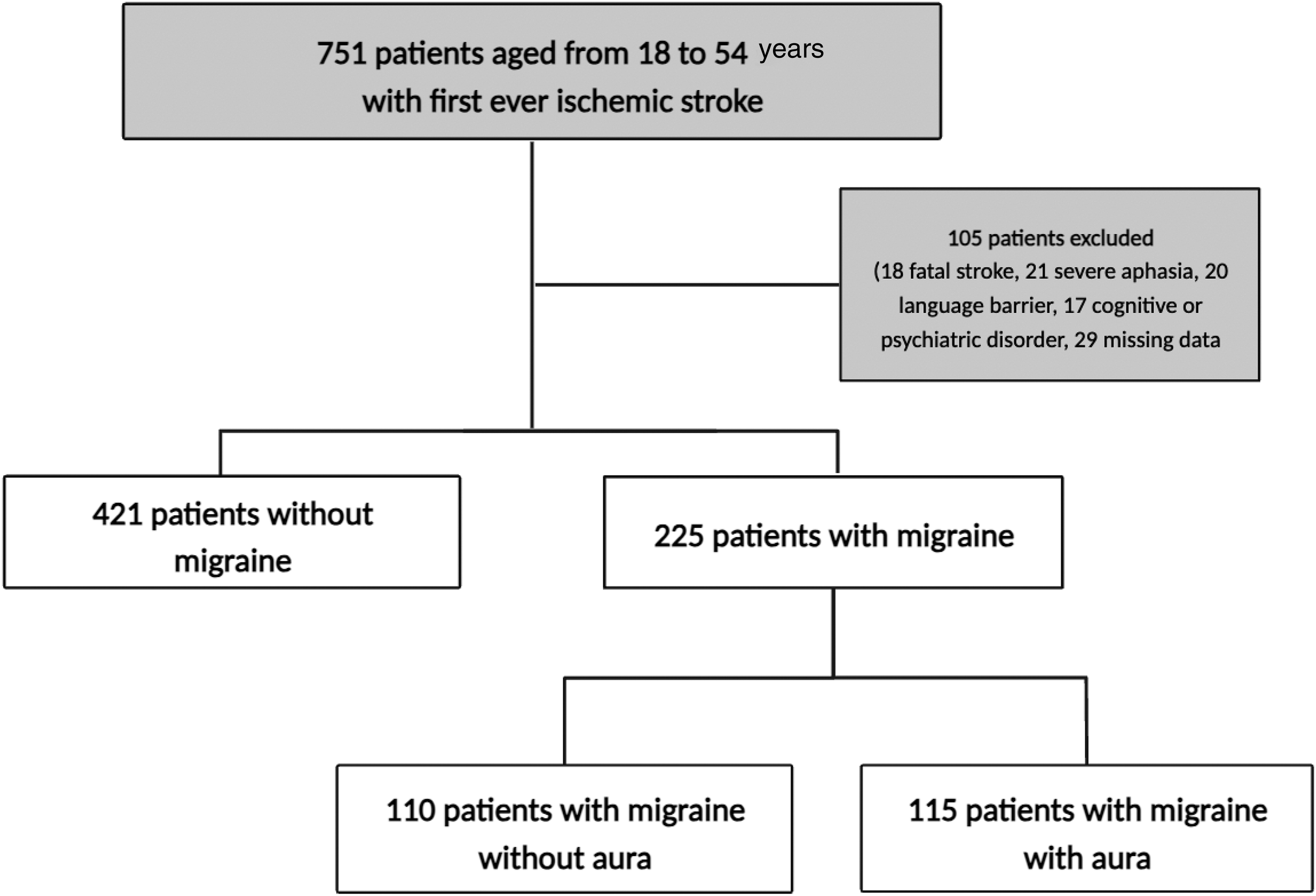
Study flow chart.
Patient characteristics.
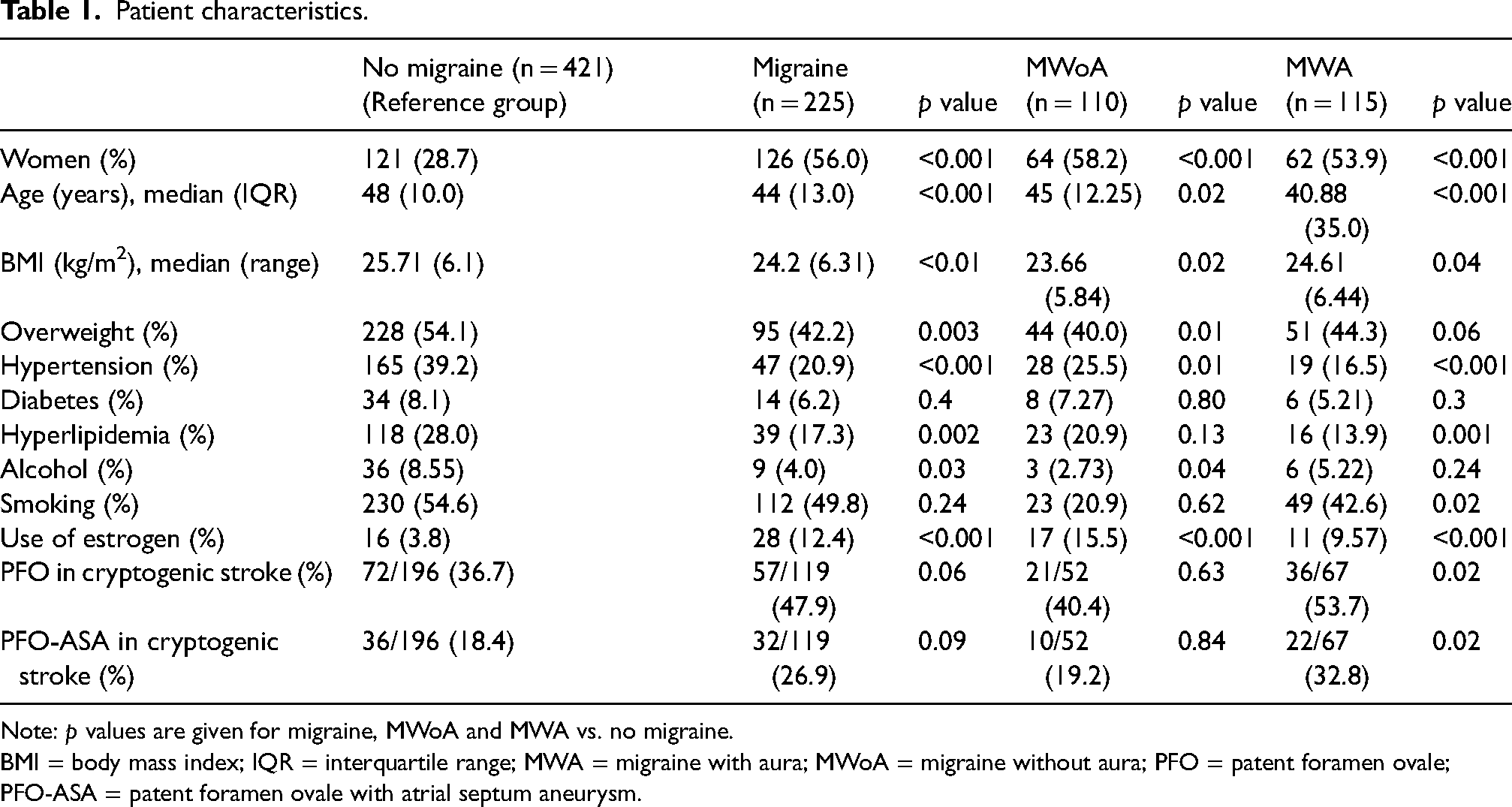
Note: p values are given for migraine, MWoA and MWA vs. no migraine.
BMI = body mass index; IQR = interquartile range; MWA = migraine with aura; MWoA = migraine without aura; PFO = patent foramen ovale; PFO-ASA = patent foramen ovale with atrial septum aneurysm.
Patients with migraine were significantly younger than patients without migraine (mean age (SD) = 44 (13) years vs. 48 (10) years, p < 0.001) and more often female (126/225 (56.0%) vs. 121/421 (28.7%), p < 0.001) (Table 1). Overweight (95/225 (42.2%) vs. 228/421 (54.1%), p = 0.003), hypertension (47/225 (20.9%) vs. 165/421 (39.2%), p < 0.001), hyperlipidemia (39/225 (17.3%) vs. 118/421 (28%), p = 0.002) and alcohol misuse (9/225 (4%) vs. 36/421 (8.55%), p = 0.03) were less frequent in patients with migraine than in patients without migraine.
Among cryptogenic stroke, PFO was significantly more frequent in MWA compared to no migraine (36/67 (53.7%) vs. 72/196 (36.7%), p = 0.02). PFO with atrial septum aneurysm (ASA) was also significantly more frequent in MWA compared to no migraine in cryptogenic stroke (22/67 (32.8%) vs. 36/196 (18.4%), p = 0.02).
Among patients with migraine, 8/225 (3.5%) patients had chronic migraine and 13/225 (5.8%) patients had inactive migraine (no migraine attack over the past year). Eight patients were taking preventive treatment for migraine and 23 patients were taking triptans as for acute treatment. Auras were visual in 110/115 (95.6%) patients, sensory in 17/115 (15%), dysphasic in 4/115 (3%) and motor in 1/115 (1%) of patients with MWA.
Association between migraine and probably causative CSVD (S1)
In univariate analysis, grade S1 CSVD lesions were significantly less frequent in patients with MWA (odds ratio (OR) = 0.35, 95% CI = 0.130–0.95, p = 0.048) compared to patients without migraine. By contrast, no significant difference was observed in migraine (OR = 0.49, 95% CI = 0.246–1.00, p = 0.06) and MWoA (OR = 0.64, 95% CI = 0.268–1.45, p = 0.44) compared to no migraine.
Multivariate analysis showed no significant association of grade S1 CSVD with migraine (OR = 0.91, 95% CI = 0.40–1.92, p = 0.81), MwoA (OR = 1.11, 95% CI = 0.42–2.64, p = 0.81) and MWA (OR = 0.72, 95% CI = 0.20–1.98, p = 0.56) (Table 2).
Associations of migraine, migraine without aura and migraine with aura with small-vessel disease of grade S1.
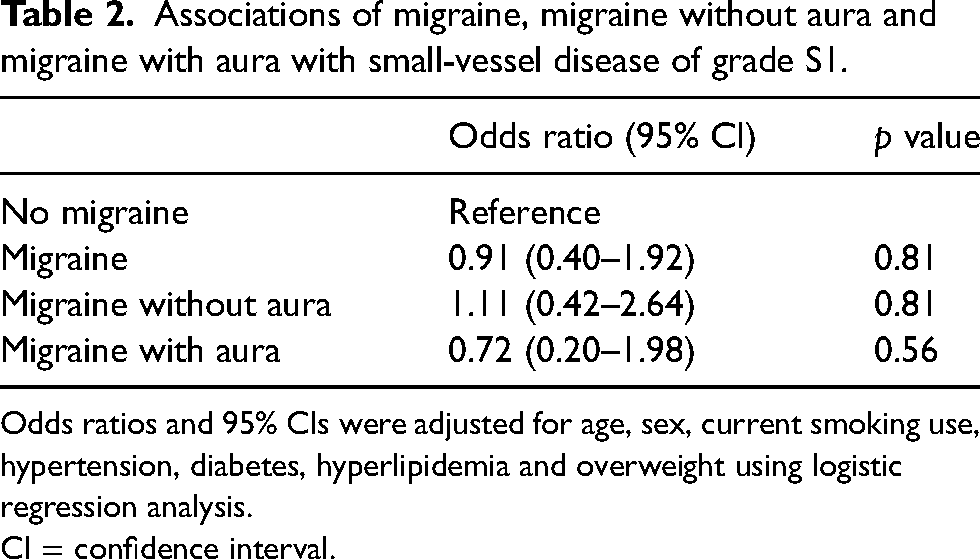
Odds ratios and 95% CIs were adjusted for age, sex, current smoking use, hypertension, diabetes, hyperlipidemia and overweight using logistic regression analysis.
CI = confidence interval.
Association between migraine and any grade CSVD (S1 + S2 + S3)
Using univariate analysis, CSVD lesions of any grade (S1, S2 or S3 vs. S0) were significantly less frequent in migraine patients (p = 0.003), patients with MWoA (p = 0.04) and patients with MWA (p = 0.02) compared to patients without migraine (Table 3).
Prevalence of small-vessel disease in patients with migraine, MWoa and MWA compared to patients without migraine.

Comparison with chi-squared test.
MWA = migraine with aura; MWoA = migraine without aura.
Multivariate analysis showed no significant association of CSVD of any grade (S1, S2 or S3 vs. S0), with migraine (OR = 0.78, 95% CI = 0.48–1.28, p = 0.34), MwoA (OR = 0.81, 95% CI = 0.42–1.47, p = 0.51) and MWA (OR = 0.84, 95% CI = 0.43–1.56, p = 0.60) (Table 4).
Associations of migraine, migraine without aura and migraine with aura with small-vessel disease of any grade.
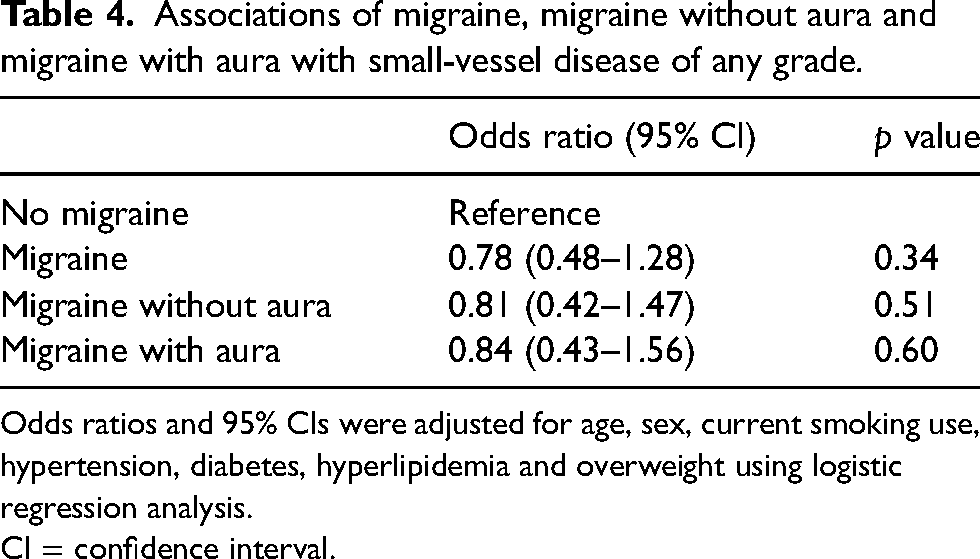
Odds ratios and 95% CIs were adjusted for age, sex, current smoking use, hypertension, diabetes, hyperlipidemia and overweight using logistic regression analysis.
CI = confidence interval.
Discussion
In this observational study of almost 650 young adults hospitalized for a first-ever ischemic stroke, MWA was not associated with stroke due to CSVD.
Four studies have previously reported the small-vessel disease phenotype of stroke in migraine, two of which involved young subjects. Pezzini et al. (32) showed that MWA was less often associated with CSVD. However, this association was not adjusted for traditional vascular risk factors and MWA patients had a better vascular profile than no-migraine patients. In a previous publication of a subset of the present data, Gollion et al. (11) showed that MWA was negatively associated with CSVD cause of stroke, but the small sample size did not allow to adjust for confounding factors. Two other studies have been carried out in populations of patients with stroke not limited to young subjects: a prospective study by Androulakis et al. (33) and a population-based study by Li et al. (34) did not find any association between MWA and CSVD stroke. However, the inclusion of elderly patients may have attenuated this association.
The results of the present study invalidate our hypothesis of an increased risk of stroke due to CSVD in MWA. This hypothesis was partly based on previous observations of an increased risk of T2 cerebral WM hyperintensity in MWA. However, this association is still a matter of debate because of the conflicting results. Seminal studies found that MWA was associated with T2 WM hyperintensities in a subset of patients with the most severe deep WM lesions (19), or in a subset of patients with frequent migraine attacks (23), mitigating the generalizability of these results in MWA. Conversely, a large study conducted among female twins did not find any association of T2 WM hyperintensities and MWA (35).
Noteworthy, WM lesions and MWA are part of the phenotype of genetic small-vessel diseases such as CADASIL (25,26). These rare genetic diseases provide a model associating MWA and CSVD. Skin and skeletal muscle biopsy in people with migraine and WM hyperintensities revealed abnormalities in the microvessels reminiscent of CADASIL (36) suggesting a continuum between these conditions. Nevertheless, our results did not support CSVD being responsible for the increased risk of stroke in MWA. Genome-wide association studies showed that migraine predisposition rely on genetic susceptibility pattern involving over 180 genetic variants, half of which are expressed in vascular tissue (37–39). However, the vascular phenotypic expression resulting from these associations has yet to be unraveled and is probably polymorph. Thus, a variant of the PHACTR1 gene has been found to be positively associated with migraine, arterial dissection and fibromuscular dysplasia, but negatively with atherosclerosis (40,41).
Recent clinical research on the causes of ischemic stroke in young migraine patients showed significant associations of MWA with PFO (10,13,42,43) and AF (11,33,44,45) and of MWoa with dissection (46,47). By contrast, migraine was found to be negatively associated with large vessel atherosclerosis (48). The present study showing no association of migraine with CSVD adds to our knowledge of the etiological spectrum of ischemic stroke in migraine. It also does not suggest a common genetic ground between migraine and CSVD. More clinical studies that investigate underlying mechanisms and etiologies of MWA-related stroke are strongly needed.
Considering these observations, a key message for clinicians is that cardiac causes of stroke should be thoroughly explored in young patients with migraine. The association of migraine, notably with aura and PFO is stronger when PFO is at risk (i.e. large), with ASA and a high risk of paradoxal embolism score (9). This implies a search for PFO using the most sensitive methods, such as transcranial Doppler with the Valsalva maneuver, and the routine performance of transesophageal cardiac echocardiography (49). If the cause of stroke remains unexplained in a patient with migraine with aura, a thorough investigation of AF should be conducted, including using an insertable cardiac monitor (11).
The strengths of the present study include the size of the population and the use of the ASCOD classification, which enables several causes of stroke to be recognized and ranked according to their level of likelihood. Each patient's migraine diagnosis was assessed by a headache specialist. Limitations must be acknowledged such as the retrospective design, although data were collected consecutively by semi-structured interview to avoid possible recall bias. Migraine status was not collected blind to the cause of the stroke, even though the assessment was often not complete at the time the patient was interviewed. Migraine status could not be collected in 105 patients (13.9%), due to cognitive or psychiatric disorders, language barriers or fatal stroke, which may have introduced a selection bias. We acknowledge that certain risk factors have not been taken into account such as low physical activity and hypercoagulable states. The etiological classification of stroke was carried out by a neurologist, without comparison with assessment by a second neurologist or calculation of the kappa index. Patients who only had a CT scan at initial assessment, without MRI, were included, which could lead to a bias in the assessment of small-vessel disease, but only concerned 1.5% of patients.
Conclusions
In a retrospective study including almost 650 young adults hospitalized for a first ischemic stroke, MWA was not associated with CSVD cause of stroke after adjustment for vascular risk factors. Further studies are needed to better understand the link between MWA and ischemic stroke.
Migraine is not associated with cerebral small-vessel disease in young adults with ischemic stroke. Cardiac causes source of stroke (such as high-risk patent foramen ovale or atrial fibrillation) should be thoroughly explored in young patients with migraine.
Footnotes
Declaration of conflicting interests
CG reports speaker fees from Novartis, Teva, Lilly, Pfizer, Abbvie, Lundbeck and Orkyn.
FC, GG, FL, MM and VL declare that they have no conflicts of interest.
Funding
No financial support was received for the research, authorship and/or publication of this article.
