Abstract
Background: Whether migraine is associated with a higher prevalence of hypercoagulable states (HS) in ischemic stroke patients is unknown.
Methods: This was a prospective study of patients under 55 years of age with brain ischemia. A systematic questionnaire addressed the antecedent of migraine with aura (MA) or without aura (MO). We investigated the presence of HS by an extensive battery of haematological tests. The presence of patent foramen ovale (PFO) was assessed by trans-oesophageal echocardiography.
Results: A total of 154 patients (95 men; mean ± SD age, 44.12 ± 8.4 years) were included; 44 had migraine, 15 had MA. HS were more frequent in the migraine than non-migraine group (38.6% vs. 16.4%, p < 0.01). The multivariate analysis showed that MO was associated with a 2.88-fold (95% CI, 1.14 to 7.28) increased risk of HS diagnosis. However, in the group of patients with brain infarction under 50 years old, MA, but not MO, was independently associated with HS (OR 6.81; 95% CI, 1.01 to 45.79).
Conclusion: In young patients with ischemic stroke, migraine may be associated with a higher frequency of HS.
Introduction
Migraine, especially with aura (MA), is considered to be a cerebrovascular risk factor associated with a two-fold greater probability of ischemic stroke, particularly in young women without other well-established vascular risk factors (1). The relationship between migraine without aura (MO) and ischemic stroke is not clear, although a meta-analysis of observational studies suggests that there is one (2). The possible aetiopathogenic mechanisms of migraine as the cause of ischemic stroke are also not fully clear (3).
Thrombophilia could play a role in ischemic stroke, particularly in young people (4). Curiously, many studies suggest that a prothrombotic condition characterizes migraine attacks, with a predisposition to cerebral ischemia, particularly in MA (5). Although the results of these studies are contradictory, findings suggest a higher frequency of some genetic abnormalities in MA patients. An increased frequency of factor V G1691A mutation and protein S deficiency has been found in MA patients and ischemic stroke patients compared to controls (6). Thus, persistent hypercoagulability may explain the tendency for migraine patients to develop thromboembolic cerebrovascular events, especially when they are exposed to additional procoagulant stresses. However, whether migraine increases the probability of having HS in patients who have suffered an ischemic stroke is not known, and the studies analysing this issue are anecdotal (7).
Our goal was to determine whether HS are associated with migraine in a cohort of adult ischemic stroke patients under the age of 55.
Methods
We prospectively analysed consecutive patients with symptoms of acute cerebral ischemia, admitted within 72 hours of symptom onset to our Stroke Unit between October 2006 and March 2009. Patients who met inclusion criteria were adults under 55 years of age who had experienced their first ever ischemic stroke or transient ischemic attack (TIA). The exclusion criteria were being on anticoagulants during the period of the study.
Informed consent was obtained, and the study was approved by the Ethics Committee of the hospital.
Migraine assessment
A systematic questionnaire upon hospital admission was administered by a physician (P. MS.) blinded to patients’ clinical details in order to assess the antecedent of MO and MA according to the International Classification of Headache Disorders-II (codes 1.1. and 1.2) (8). The questionnaire included the age of onset and the frequency of migraine attacks (episodes/month).
Hypercoagulable states
An extensive coagulation study to investigate prothrombotic states was performed during hospitalization and, in the case of abnormal values (especially in protein S or C), repeated 3 months later. This coagulation study included Factor V Leiden and prothrombin G20210A gene mutations; deficiencies in protein S, protein C and antithrombin levels; lupus anticoagulant; anticardiolipin antibodies; antinuclear antibodies, elevated factor VIII levels; resistance to activated protein C and abnormal platelet function. The collagen/adrenaline and the collagen/adenosine diphosphate tests were used to screen for abnormal platelet function. Elevated anticardiolipin antibody was defined as IgG and/or IgM isotype in serum or plasma, present in medium or high titre (> 20 MPL), on two or more occasions, at least 12 weeks apart, measured by a standardized ELISA. Thrombocytosis was defined as serum platelet count > 450 × 1010/L.
Other laboratory analyses
A lipid profile including total cholesterol, low-density lipoprotein cholesterol (LDL-c), high-density lipoprotein cholesterol (HDL-c) and triglycerides was determined in the first 48 hours after brain ischemia.
Demographic data and vascular risk factors
The following parameters were recorded in a specific data bank: (a) demographic characteristics: age, sex; (b) previous treatments: antiplatelet, statins, antihypertensive drugs, oral contraceptive use and hormone replacement therapy; (c) vascular risk factors and comorbid conditions: obesity (body mass index ≥ 30), arterial hypertension (defined as the existence of a previous clinical diagnosis of arterial hypertension, regular treatment with antihypertensive drugs, or the existence of two or more pre-stroke readings > 140 and/or 90 mmHg), current smoking, alcohol abuse (history of alcohol intake > 80 g/day), other drug use (cocaine, cannabis, heroin, ecstasy, etc.) reported by the patient/family or detected in a blood/urine analysis when suspected by the physician in charge of the patient; diabetes mellitus (previous diagnosis and/or current treatment with insulin or oral hypoglycaemic medications), hyperlipidaemia (previous diagnosis and/or current treatment with lipid-lowering drugs), coronary arterial disease (angina pectoris or myocardial infarction), peripheral arterial disease, chronic or paroxysmal atrial fibrillation, valvular heart disease, and deep vein thrombosis.
Right to left shunt and atrial septal abnormalities
The presence of a right-to-left shunt (RLS) was investigated by transcranial Doppler sonography (TCD) (TC4040 Pioneer/Nicolet) with intravenous injection of agitated saline following a standardized protocol (9).
All patients underwent a transthoracic echocardiography study as part of the routine aetiological work-up for stroke. A trans-oesophageal echocardiography was required when RLS was detected by TCD or the patient was under 50 years of age to establish the presence of patent foramen ovale (PFO) and/or atrial septal aneurysm (ASA).
In-hospital management and stroke classification
During the first 24 hours of hospitalization each patient was assessed according to a standard neurovascular protocol including urgent brain CT scan, chest X-ray, ECG, routine laboratory blood analyses and carotid plus transcranial ultrasound examination. Further cranial MRI and angio-MRI were performed at between 24 and 72 hours after admittance to examine the magnitude of the brain lesion and to detect arterial pathology.
A brain infarction was diagnosed when the CT or MRI showed an ischemic brain lesion corresponding to the patient’s symptoms. A TIA was defined as a transient episode of neurological dysfunction caused by focal brain ischemia without acute infarction.
Stroke aetiological subtype was classified, following published criteria, as large vessel disease, cardioembolic infarction, small vessel disease, cerebral infarction of uncommon aetiology, or cerebral infarction of undetermined aetiology (10,11). Brain ischemia caused by PFO or HS was classified as uncommon aetiology. Stroke severity on admission was assessed according to the National Institute of Health Stroke Scale (NIHSS).
Statistical analyses
Comparisons between groups were analysed with the χ2 or Fisher’s exact test for dichotomous variables. Continuous variables were expressed as mean ± SD or median (interquartile range [IQR]) and compared with Student’s t-test or the Mann–Whitney test, as appropriate. The relationship between HS and migraine was assessed using a multivariate logistic regression model. The following variables were included in the maximum model: age (years), sex (male/female), PFO (yes/no) and migraine (coded as no, without aura or with aura, according to a dummy scheme using ‘no’ as reference). A backward procedure was followed as the modelling strategy, using the log likelihood ratio test to assess the goodness of fit and compare nested models. Those variables that when eliminated produced a change of ≥ 15% of the OR were considered confounding variables. An interaction term between migraine and PFO was analysed but was not included in the final model because it was not found to be significant. Ninety-five per cent confidence intervals (CIs) are presented. A separate analysis was performed with patients <50 years old with brain infarction but not TIA in order to minimize the number of postmenopausal women and also avoid a potential confusion between TIA and aura in this group. All tests were two-sided and p-values of 0.05 or less were considered statistically significant.
Statistical analysis was performed using SPSS 19.0 (Statistical Package for Social Science, SPSS Inc., version 19.0 for Windows).
Results
One hundred and sixty-two patients under 55 years of age presented with brain ischemia during the study period. Eight patients were excluded, seven for being on anticoagulant treatment and one for not being able to communicate due to severe aphasia. One hundred and fifty-four patients (95 men; mean ± SD age, 44.12 ± 8.4 years) met the inclusion criteria and were included in the study; 153 patients had a brain infarction and 51 a TIA. Forty-four had migraine, 15 MA; 56.8% of the migraine cases fulfilling IHS criteria for migraine diagnosis had not been previously evaluated by a physician. The group of brain infarction patients under 50 years of age was composed of 79 cases, 18 with migraine, 5 MA.
In all cases, the first reported migraine attack had occurred at least one year before the brain ischemia, with a median (IQR) of 21 (22) years for the whole sample and 12 (25) years for the group of brain infarction patients under 50 years old.
Demographic data, cerebrovascular risk factors, previous treatment, subtype of brain ischemia and echocardiographic findings in migraine and non-migraine patients
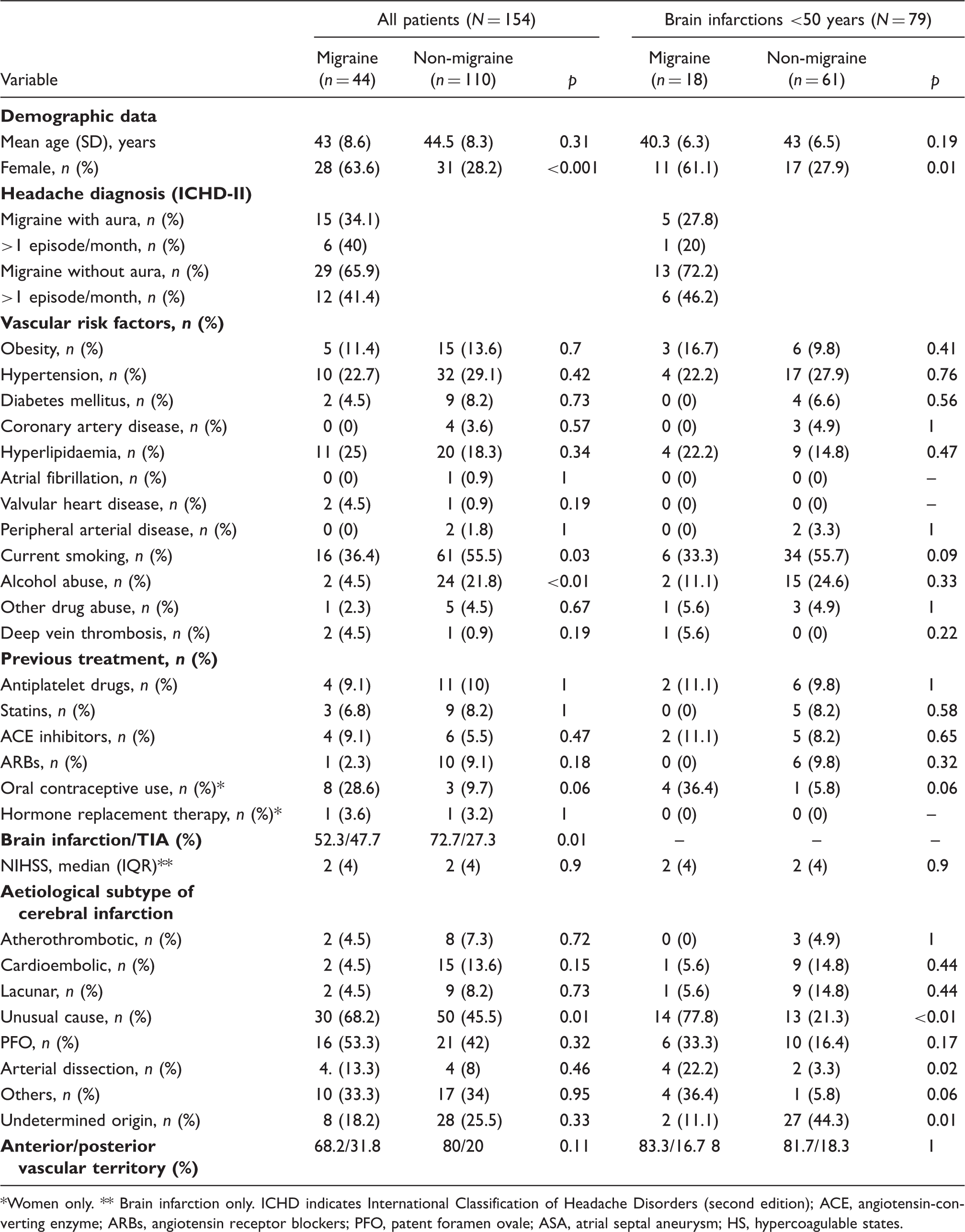
Women only. ** Brain infarction only. ICHD indicates International Classification of Headache Disorders (second edition); ACE, angiotensin-converting enzyme; ARBs, angiotensin receptor blockers; PFO, patent foramen ovale; ASA, atrial septal aneurysm; HS, hypercoagulable states.
In the group of brain infarction patients under 50 years old, migraineurs were also more frequently female. Smoking was also more frequent among non-migraine patients (Table 1).
On the other hand, women with migraine tended to have used oral contraceptives more frequently than non-migraineurs in the whole sample (28.6% vs. 9.7%, p = 0.06) and in the under 50-year-old infarction group (36.4% vs. 5.8%, P = 0.02) (Table 1).
Brain infarction was more common than TIA in the non-migraine group than in the migraine group (72.7% vs. 52.3%, p = 0.015) (Table 1). However migraineurs had a brain ischemia of unusual cause more frequently than non-migraineurs. In addition, migraineurs tended to suffer brain ischemia in the posterior vascular territory more frequently than non-migraineurs, except in the group of brain infarction patients under 50 years old (Table 1).
In the whole sample, the frequency of arterial dissection was similar in the patients with and without migraine. However, in the patients under 50 years old, it was significantly higher in patients with migraine than in patients without (22.2% vs. 3.3%, p = 0.02) (Table 1). Arterial dissections were not associated with HS (data not shown).
Laboratory analyses in migraine and non-migraine patients

Three migraineurs and two non-migraineurs presented more than one hypercoagulable state.
All heterozygous.
Platelet count >450 × 1010/L
Patients with activated protein C resistance and not FV.
Factor V Leiden indicates Factor V Leiden G1691A mutation; APC, activated protein C resistance.
PFO was present in 38.6% (17 cases) of migraineurs and 30% (30 cases) of the non-migraine group (p = 0.34). The association between PFO and ASA was similar in both groups. However, the combination of PFO and HS was more frequent in the migraine group (13.6 vs. 2.7, p = 0.01).
In the group of brain infarction patients under 50 years old, PFO was present in 33.3% (6 cases) of migraineurs and 16.4% (10 cases) of non-migraine patients (p = 0.17). The association of PFO and ASA was non-significantly higher in migraineurs (11.1% vs. 3.3%, p = 0.22). The combination of PFO and HS was also more frequent in the migraine group (22.2% vs. 1.6%, p < 0.01).
Within the migraine group, the same percentage of MA and MO were diagnosed with PFO and ASA. The distribution of HS was different when the presence of migraine and PFO was taken into account. As shown in Figure 1, HS were more frequent in the group of migraine + PFO (43.8%), less frequent in migraineurs without PFO (35.7%), and even less frequent in non-migraineurs (17.3%) and these differences were statistically significant (p = 0.01). Similar results were found in brain infarction patients under 50 years old (Figure 2).
Distribution of hypercoagulable states according to the presence of migraine and patent foramen ovale (PFO); p = 0.01 for comparison between three groups, p = 0.04 between the migraine + PFO and the non-migraine group, and p = 0.03 between the migraine without PFO and the non-migraine group. Distribution of hypercoagulable states according to the presence of migraine and patent foramen ovale (PFO) in brain infarction patients <50 years old (N = 79); p = 0.02 for comparison between three groups, p = 0.02 between the migraine + PFO and the non-migraine group, and p = 0.25 between the migraine without PFO and the non-migraine group.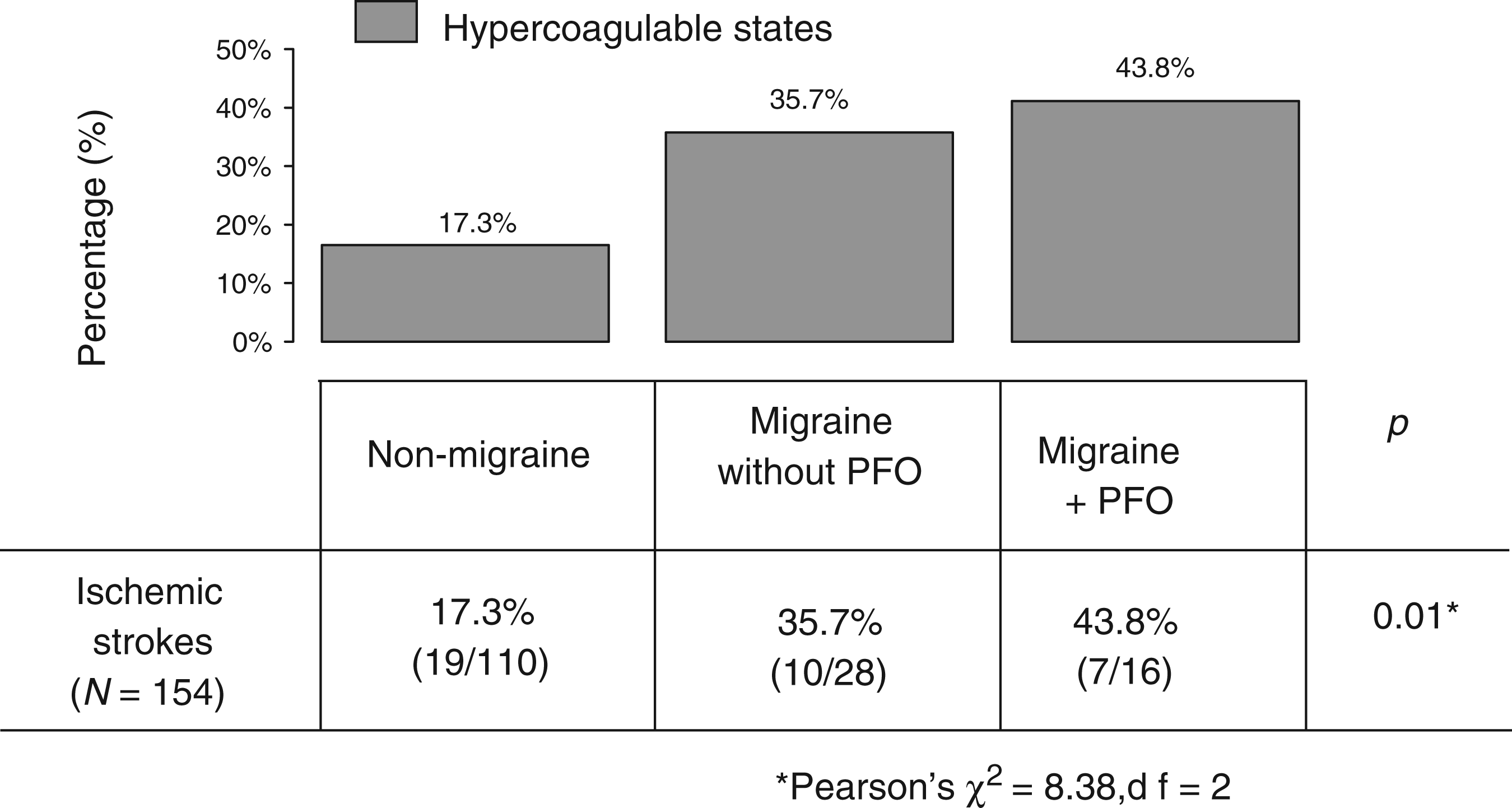

Bi- and multivariate logistic regression analysis for factors associated with hypercoagulable states

Multivariate logistic analysis for association of migraine, age, female sex, patent foramen ovale and migraine × PFO with hypercoagulable states.
PFO, patent foramen ovale; MA, migraine with aura; TIA, transient ischemic attack.
Discussion
In this study, young stroke patients with migraine had an increased risk of presenting with some HS, above all protein C or S deficiencies and prothrombin G2021A mutation.
Previous studies have addressed the possible relationship between migraine, stroke and genetic thrombophilias, reporting similar results to the present study. D’Amico et al. showed an increased frequency of protein S deficiency and of activated protein C resistance due to factor V mutation in MA and stroke patients compared with controls (6). In the present study protein C or S deficiency were also more frequent among migraineurs. Although the frequency of Factor V Leiden was high (6.8%) among migraineurs, and double the frequency in non-migraineurs, this was not statistically significant, as in a previous study (12). Moreover, a recent study has found that stroke patients who are carriers of Factor V Leiden mutation, prothrombin G2021A mutation, or both prothrombotic genotypes had a greater than two-fold increased risk for MA (13). In our study, carriers of the prothrombin mutation and Factor V Leiden mutation also doubled the frequency of migraine, compared with the whole sample.
On the other hand, acquired prothrombotic conditions such as antiphospholipid syndrome have also been associated with migraine, with contradictory results. In this study no differences were found between migraine and non-migraine patients, in agreement with the most recent evidence (14). In brain infarction patients under 50 years old, migraine was significantly associated with anticardiolipin antibodies. However, the present sample is too small and this observation needs to be confirmed in a larger study.
The majority of the studies analysing the association of migraine and HS have included patients previously diagnosed with migraine, but have not employed a specific questionnaire to confirm this diagnosis (6,7,12,13). This is an important issue since we have observed that 15 of the 44 migraine patients (56.8%) met IHS criteria for migraine but had not been previously evaluated by any physician. Similar results were reported in the CAMERA study, where 54% of migraineurs had not been previously diagnosed (15). Consequently, the under-diagnosis of migraine could be a significant limitation of many studies that wish to establish a relationship between migraine and other pathologies.
Migraine is found in some patients with ischemic stroke, suggesting a complex bidirectional relationship between migraine and ischemic stroke, which would include migraine as a stroke cause, as a vascular risk factor and as a consequence of cerebral ischemia, and this suggests a common aetiology for migraine and cerebral ischemia (16). Moreover, migraine is a known risk factor for arterial dissection, suggesting that the possible underlying arterial wall disease leading to dissection could be a predisposing factor for migraine (17–19). Our data show that arterial dissections in the younger stroke group are more strongly associated with migraine than strokes not caused by arterial dissection, a finding in agreement with previous reports (17). Furthermore, it has been suggested that thrombophilia plays a role in ischemic stroke, particularly in young people (4), and migraine could be associated with a higher frequency of prothrombotic states.
On the other hand, PFO has also been associated with acquired and/or inherited HS, and, in turn, migraine has been related to atrial septal abnormalities (20,21). In the present study migraine patients had a higher frequency of thrombophilia, and PFO appears to enhance this association in the unadjusted analysis. However, in the multivariate analysis the presence of FOP did not enhance the association between migraine and HS.
MO appears to be associated with HS in the whole sample and in the TIA group, whereas MA is related to HS in brain infarction patients under 50 years old. One possible explanation is that some TIAs could actually be auras, and this may be acting as a confounding factor. Indeed, MA has been more strongly related to HS than MO (13,17) and is considered a cerebrovascular risk factor in young people (1).
The pathophysiology of migraine is not completely understood and continues to be investigated. Endothelial dysfunction and hypercoagulability could be responsible for some types of migraine. A recent study in young women found a strong relation between biomarkers of endothelial activation and hypercoagulability such as von Willebrand factor (vWF) activity, high-sensitivity C-reactive protein (hs-CRP), tissue-type plasminogen activator (t-PA) antigen, and total nitrite/nitrate concentration, and migraine. Furthermore, this association was stronger for MA, the subtype most closely linked to cardiovascular disease. The authors suggest that endothelial dysfunction may be causally related to migraine, rather than a consequence, although the exact mechanism is not well known (22). The present study suggests that migraine patients who suffer brain ischemia could have a higher frequency of thrombophilic disorders than non-migraine patients. The accidental or genetic association with atrial septal abnormalities could predispose these patients to an ischemic stroke.
This study has some limitations. One is the limited number of migraine patients, especially MA patients. However, we have found a statistically significant association between migraine and HS, one which could be even greater if the number of patients increases. Another limitation is the lack of a group of migraine patients without brain ischemia, which would allow us to establish whether migraine was associated with HS in non-stroke patients. However, our objective here was to determine if the presence of migraine is related to an increase of HS in stroke patients, to enable identification of groups with a higher risk of recurrence. Finally, eight patients were excluded for being on anticoagulant treatment or could not consent due to severe aphasia. The absence of information from these patients is a potential drawback of the study.
In conclusion, migraine may be associated with a higher frequency of HS in young patients with ischemic stroke. Screening for prothrombotic conditions should be recommended in young patients with migraine and TIA or ischemic stroke.
Footnotes
Acknowledgements
We are grateful to Elena Díaz for her assistance with statistical analyses.
Funding
This project is part of the Spanish collaborative research network RENEVAS (Instituto de Salud Carlos III, Ministerio de Ciencia e Innovación, RD06/0026/008, RD07/0026/2003). B. Fuentes was provided with a grant by the Instituto de Salud Carlos III and the Agencia Laín Entralgo – Comunidad de Madrid.
