Abstract
Background
Migraine presents significant health and economic challenges. Despite the widespread use of triptans, some patients discontinue them because of insufficient relief or adverse effects. Using national registers, the present study investigates the excess costs and labour market disaffiliation of Danish patients discontinuing triptan treatment.
Methods
The study included all individuals ≥18 years (“patients”) who discontinued redemption of triptan prescriptions between 1998 and 2019. They were categorized by number of distinct triptans redeemed before discontinuation: one, two or three or more. A control group was established from the general population without triptan redemptions, three per patient, matched by year of birth, sex and region of residence. We estimated excess direct and indirect costs from 5 years prior (“year −5”) to 10 years post (“year 10”) the first triptan redemption.
Results
We identified 211,026 patients who discontinued triptan redemption, 82% after one, 14% after two and 4% after three or more distinct triptans. Over the period from year −5 to year 10, average excess healthcare costs per patient in these cohorts were EUR 9,554, EUR 10,942 and EUR 12,812 respectively. Over the same period, these patients earned EUR 27,964, EUR 35,920 and EUR 50,076 less than their respective controls, and received higher public transfer payments of EUR 20,181, EUR 23,264 and EUR 26,459.
Conclusions
Triptan discontinuers, who appear to have exhausted all current treatment avenues, face high direct and very high indirect excess costs attributable to migraine, and experience substantial increased labour market disaffiliation.
Introduction
Migraine is a prevalent and debilitating disease worldwide, ranked second in the world among the causes of years lost to disability (1,2). Furthermore, migraine is associated with substantial healthcare utilization and costs (2–7). In the Eurolight project, which surveyed eight countries, the estimated mean annual costs per person with migraine were EUR 1,222, with indirect costs constituting 93%, two-thirds of these attributable to reduced productivity at work and one third to absenteeism (7). Total costs attributed to migraine in Europe were estimated at EUR 50–111 billion in 2011, of which direct costs accounted for only 7% (7,8).
The current primary focus of treatment is to manage symptoms, either during an acute attack or by reducing the frequency of attacks. Both Danish and European guidelines recommend non-steroidal anti-inflammatory drugs and other simple analgesics as first-line acute treatment, and triptans as second-line when these are ineffective, poorly tolerated or contraindicated (9,10). In Denmark, seven distinct triptans are available, some in multiple formulations, differing in effectiveness and adverse-event profiles (9). It is evident that lack of response to one triptan does not preclude response to another, or, sometimes, the same in another formulation. Accordingly, the Danish guidelines recommend the trial of three distinct triptans before any conclusion that triptans as a class are ineffective in a patient (9). The Consensus Statement by the European Headache Federation and the European Academy of Neurology recommends trying several triptans in different formulations (10,11). Despite these guidelines, a recent Danish study revealed that, although 40–60% of triptan users entirely discontinued triptan treatment, only 10% had tried more than one triptan before discontinuation (12).
In recent years, new preventative and acute treatments for migraine have been introduced, and more are currently in development (13). The growing number of available treatment options highlights the need for research to identify, among these options, the most effective and appropriate in individualized care. To the best of our knowledge, no studies of this nature have been conducted using real-world nationwide register data. Similarly, no study has focused on patients who discontinue triptan treatment, apparently having exhausted current acute treatment options. Societal-level estimates of the burden and cost of migraine among these patients are crucial for understanding unmet healthcare needs, and the potential to mitigate them.
Using routinely collected individual-level national register data, the present population-based study aimed to analyse the excess costs (attributable to migraine) among patients who have discontinued triptan treatment. In addition, we sought to understand the labour market disaffiliation of these patients.
Methods
Data sources
We retrieved data relating to the period 1995 to 2021 from the comprehensive Danish national registers. Since 1968, all Danish residents have been assigned a unique personal identification number (Danish: Centrale Personregister, CPR) at birth or immigration. CPRs are registered in the Danish Civil Registration System, which contains information such as date of birth, sex assigned at birth, region of residence and family relationships for all individuals (14). The system enables identity-secure linkage of all information across all the Danish national registers at individual level. The Danish National Patient Register (NPR) has collected data on all inpatient hospital contacts from 1977 onwards, data on all outpatient contacts, emergency room contacts and contacts with psychiatric wards, including diagnoses and preformed procedures, from 1995 onwards, and information on unit costs (provided by Diagnosis Related Groups (DRG) tariffs) from 2002 onwards (15). The Danish National Health Service Register for Primary Care contains information on primary healthcare contacts and unit costs since 1990, but diagnoses and specific procedures are not available in this register (16). The Danish National Prescription Register holds data on prescription medicines purchased since 1995, providing information on medicine costs associated with every prescription redeemed at Danish community pharmacies (over-the-counter medicines are included only when prescribed by a physician) (17). The Danish Income Statistics Register contains data on salaries, taxes, public transfer payments, capital income and private pension contributions on a yearly basis from 1970 onwards (18). The DREAM (Danish: Den Registerbaserede Evaluering Af Marginaliseringsomfanget) Database holds information relating to labour market affiliation, in the form of weekly labour market public transfer payments (unemployment benefits and disability pension or disability payments) for all Danish residents since 1991 (18). Finally, the Cause of Death Register and the Migration Register hold information on deaths and migration.
Study population
From the Danish National Prescription Register, we obtained data on all redemptions of triptan prescriptions by Danish residents (in Denmark, triptans may be obtained only on a physician's prescription). We identified all adults (≥18 years) who made at least one such redemption (Anatomical Therapeutic Chemical Classification System (ATC) codes: N02CC01–N02CC07) during the study period. We assumed triptans to be prescribed only for migraine (although some are indicated for cluster-headache also, this condition is rare and is not expected to affect the results of the present study). We excluded patients redeeming triptans in the period 1995–1997, aiming to ensure that only patients commencing triptan treatment within the relevant period were included. Among the latter, we defined triptan discontinuers as those making no further triptan redemptions for at least a two-year period and until the end of follow-up (between 1 January 2020 and 31 December 2021). These were included in the study population and divided into three cohorts. Cohort 1 comprised those who discontinued triptan treatment after redeeming only one distinct triptan; cohort 2 discontinued after redeeming prescriptions for two distinct triptans; and cohort 3 discontinued after redeeming prescriptions for three or more distinct triptans. We disregarded formulation in this study (with some triptans being available in multiple formulations).
Data from the National Prescription Register and the National Health Service Register do not include information that allows categorization by type of migraine (i.e. episodic vs. chronic migraine, or migraine with or without aura).
We identified comparison individuals from the general Danish population, who had not redeemed triptans throughout the study period (1995–2021), by applying data from the Danish National Prescription Register. Applying data from the Danish Civil Registration System, we exactly matched each triptan discontinuer with three controls by year of birth, sex and region of residence. Thus, discontinuers and their three matched controls were born the same year, had the same sex and lived in the same region.
Outcome variables
The outcome variables in the present study were excess direct and indirect costs along with labour market affiliation. Excess direct costs were defined as the difference in healthcare costs between triptan discontinuers and their matched controls. Healthcare costs included those in the primary healthcare sector as well as in hospital care (both hospital admissions and outpatient visits), estimated by fees and DRG tariffs. Costs from both private and public clinics and hospitals were included. Prescription medicine costs, both the public reimbursement and the patient co-payment portions, were included using pharmacy selling prices (including the Danish value-added tax of 25%). Excess indirect costs were defined as the difference in earnings (total annual salary) and public transfer payments (both retrieved from the Danish Income Statistics Register) between triptan discontinuers and matched controls. Labour market disaffiliation (only among those aged up to 65 years, the retirement age in Denmark) was assessed as in receipt of disability pension, being or becoming unemployed (defined as in receipt of social security or unemployment benefit for at least five weeks) or being on long-term sick leave (more than four weeks).
Statistical analysis
The study followed each patient from up to five years before the date of their first triptan redemption (the index date) until 11 years after this date, or until death, emigration, or the end of follow-up (31 December 2021) if any of these occurred first. Average direct and indirect costs were calculated annually from year −5 to year 10 with respect to the index date for triptan discontinuers and their matched controls. Observations were weighted with respect to n to balance the dataset in each year. Excess costs were calculated as the differences in costs between the three cohorts and their respective controls for each year before and after the index date.
Differences in the proportions unemployed or on long-term sick leave for any period of time within the year were similarly calculated annually from year −5 to year 10.
Receipt of disability pension was regarded as an irreversible event. Hazard ratios (HRs) were calculated using Cox proportional hazard regression models with the three cohorts as the reference groups, adjusted for age and sex. In the Cox regression analysis, individuals were censored when reaching 65 years of age because this was the retirement age for most of the study period. Furthermore, individuals were censored at death, emigration or end of follow-up (31 December 2021). All censoring and event dates were exact, and there were no dropout events or unexplained losses to follow-up. Because cases and controls were matched on birth year, censoring was assumed to be non-informative. Kaplan–Meier curves were derived. We controlled for reverse causality in sensitivity testing by requiring individuals to survive at least six months in the model without event. Models with 10-year follow-up were estimated to evaluate sensitivity to duration of follow-up. Proportional hazard assumption was tested by Schoenfeld residuals.
All costs were set to 2021 levels. Fees and tariffs in both the Danish National Health Service Register for Primary Care and the NPR, as well as drug prices in the Danish National Prescription Register, were applied as unit costs and inflated using the combined price and wage index specific for healthcare services, according to the Danish Regions (19). Data on healthcare costs were available only from 2002; therefore, for patients redeeming triptans before 2002, costs were not included for the first years. All costs are reported in euros at an assumed exchange rate of EUR 1.00 = DKK 7.50.
All analyses were performed using R statistical software, version 4.2.1 (R Foundation, Vienna, Austria) on Statistic Denmark's (https://www.dst.dk) research computers via a remote server.
Results
Study population
A flowchart of the population identification is presented in Figure 1. We identified 401,990 adults making at least one triptan redemption between 1995 and 2021, of whom 354,546 were incident triptan users (first redemption between 1998 and 2021). Among the latter, 211,026 (59.5%) discontinued triptan treatment between 1998 and 2019, with no further redemptions during the remainder of the study period (until 31 December 2021).
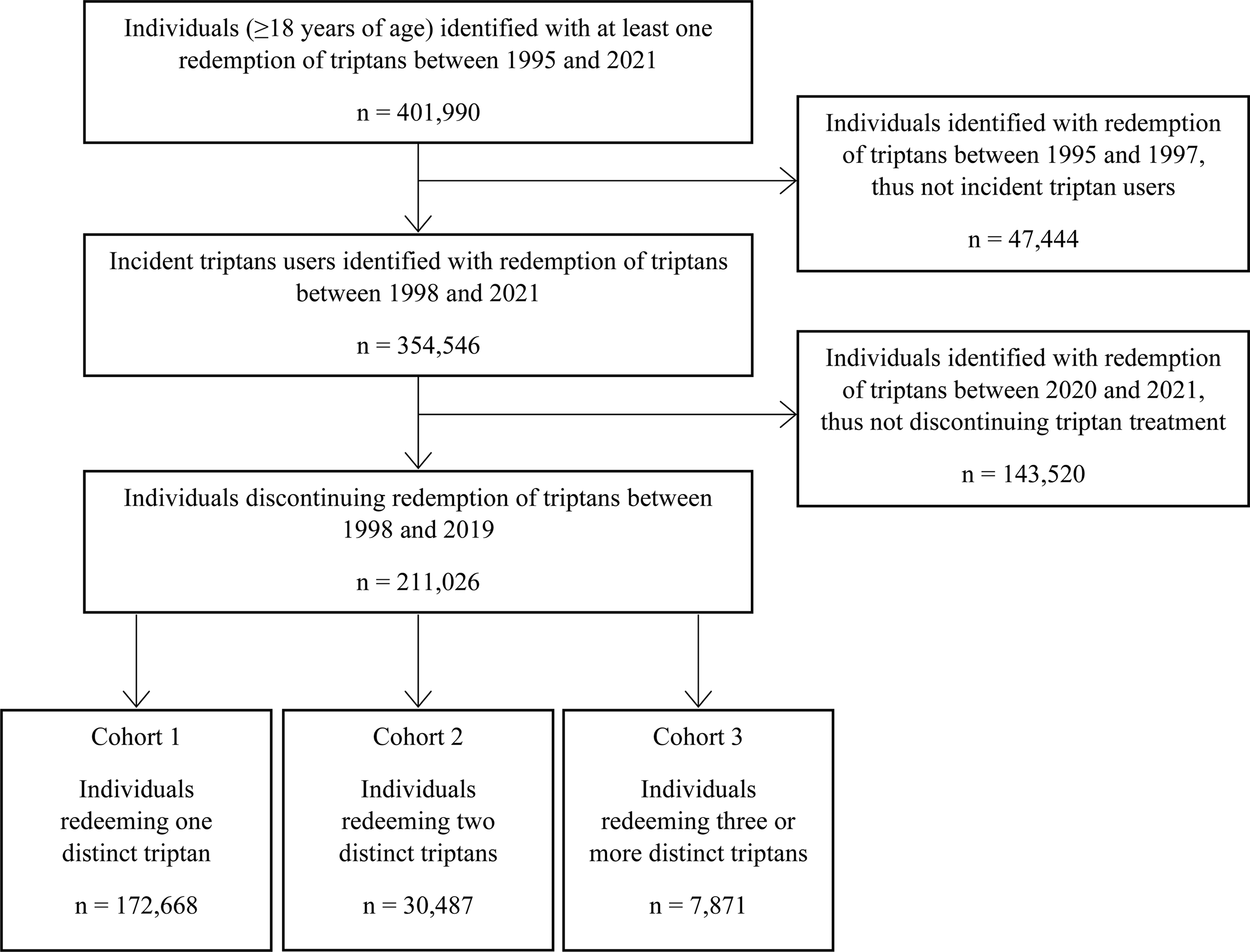
Flowchart of the study population.
We found no evidence of differences in mortality or migration between groups.
Within the three cohorts were 172,668 individuals (81.8%) redeeming one (cohort 1), 30,487 (14.4%) redeeming two (cohort 2) and 7871 (3.7%) redeeming three or more distinct triptans (cohort 3) before discontinuing (Table 1). Among cohort 3, the majority (77%) had redeemed three distinct triptans, 19% had redeemed four and 4% had redeemed five or more distinct triptans before discontinuing. The mean time on treatment increased from 1.5 years for cohort 1 to 9.6 years for cohort 3. Proportions of females exceeded those of males: 73% in cohort 1 (reflecting the sex distribution of migraine in the population), but somewhat higher in cohorts 2 (80%) and 3 (85%). Median age at first triptan redemption (36–39 years) was similar between the three cohorts. All three cohorts were similarly distributed across the five Danish regions.
Characteristics of the study population.

Abbreviations: IQR = interquartile range; NA = not applicable.
Direct healthcare costs
Figure 2 presents the average annual individual excess healthcare costs per cohort, relative to their respective controls, from year −5 to year 10 for each triptan discontinuer. The median follow-up times for cohorts 1–3 were 11.6 years (interquartile range (IQR) = 10–14), 12.3 years (IQR = 11–15) and 12.0 years (IQR = 12–14), respectively.

Average annual individual excess healthcare costs before and after the index. date in patients in the three cohorts. (a) Hospital admission costs, (b) Outpatient visit costs, (c) Primary healthcare sector costs and (d) Prescription medicine costs, EUR, 2021 price level. Note: Cohort 1 = patients who discontinued triptans after one distinct triptan; Cohort 2 = patients who discontinued triptans after two distinct triptans; Cohort 3 = patients who discontinued triptans after three or more distinct triptans. Excess costs calculated as the difference in costs between each of the three cohorts and their respective controls for each year before and after the index date.
Excess annual hospital admission (EUR 92–491) (Figure 2a) and outpatient-visit costs (EUR 138–305) (Figure 2b) varied between years −5 and 10, with little difference between the three cohorts, but an increase in excess outpatient-visit costs in the index year (year 0) for all three cohorts. Even prior to the first triptan redemption, the excess hospital admission and outpatient costs were high. With regard to admissions, the International Statistical Classification of Diseases and Related Health Problems (ICD)-10 chapters for the registered primary diagnoses explaining the majority of the differences throughout the study period were consistent across all cohorts, with chapters 21 (“Factors influencing health status and contact with health services”), 19 (“Injury, poisoning and certain other consequences of external causes”), 18 (“Symptoms, signs and abnormal clinical and laboratory findings not finding elsewhere classified”), 13 (“Diseases of the musculoskeletal system and connective tissue”) and 11 (“Diseases of the digestive system”), in that order, between them explaining 78–80% of the differences (see Supporting information, Tables S1 to S3). With regard to outpatient visits, the ICD-10 chapters explaining the majority of the differences throughout the study period were similarly consistent across all cohorts, with chapters 21, 13, 18, 11 and 6 (“Diseases of the nervous system”), in that order, between them explaining 72–83% of the differences (see Supporting information, Tables S4 to S6).
The excess primary healthcare sector costs were highest in cohort 3 for the entire study period (Figure 2c). Up to the index year, the annual excess primary healthcare sector costs increased in all three cohorts, reaching EUR 169, EUR 214 and EUR 261 for cohorts 1, 2 and 3. The excess cost of prescription medicines increased considerably in the year of first triptan redemption, more so in cohort 3 (EUR 378) than cohort 2 (EUR 268) or cohort 1 (EUR 162) (Figure 2d), and remained high for the rest of the study period. After the index year, patients in all three cohorts made significantly more redemptions of prescription medicines in all ATC 1st-level classification groups than their respective controls, not only in ATC group N, which include triptans (data not shown).
The total individual excess healthcare costs from year −5 to year 10 were EUR 9554 in cohort 1, EUR 10,942 in cohort 2 and EUR 12,812 in cohort 3 (Figure 3). Prescription medicines constituted 31% of the total costs in cohort 3, 22% in cohort 2 and 17% in cohort 1.

Total individual excess healthcare costs from year −5 to year 10 in patients in the three cohorts, EUR, 2021 price level. Note: Cohort 1 = patients who discontinued triptans after one distinct triptan; Cohort 2 = patients who discontinued triptans after two distinct triptans; Cohort 3 = patients who discontinued triptans after three or more distinct triptans. Excess costs calculated as the difference in costs between each of the three cohorts and their respective controls for the entire study period (year −5 to year 10).
Indirect costs
For all three cohorts, the average annual individual difference in gross earnings between triptan discontinuers and matched controls increased in the years following the index date, in cohort 3 > cohort 2 > cohort 1 (Figure 4). At year 10, the difference reached EUR −2907, EUR −3975 and EUR −4762 in cohorts 1, 2 and 3. Over the entire study period, all three cohorts had significantly lower earnings than their respective controls, both before and after the index year.

Average annual individual difference in gross earnings in patients in the three cohorts, EUR, 2021 price level. Note: Cohort 1 = patients who discontinued triptans after one distinct triptan; Cohort 2 = patients who discontinued triptans after two distinct triptans; Cohort 3 = patients who discontinued triptans after three or more distinct triptans. Differences in each cohort are with respect to their respective controls.
Likewise, all three cohorts received significantly more public transfer payments than their respective controls over the entire study period (Figure 5), increasing in the years following the index date. In year 10, differences reached EUR 1363, EUR 1807 and EUR 2085 in cohorts 1, 2 and 3.
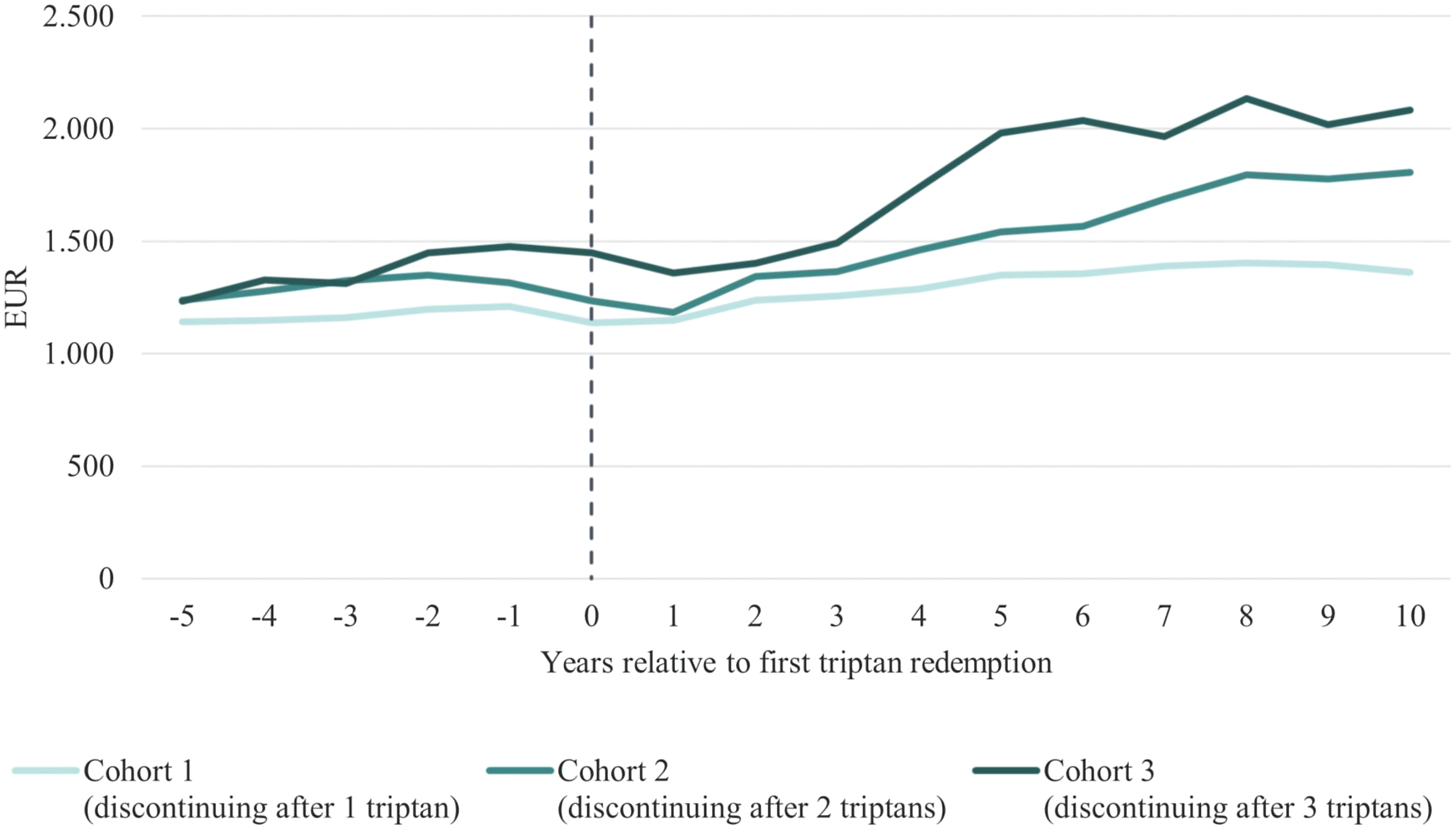
Average annual individual difference in public transfer payments in patients in the three cohorts, EUR, 2021 price level. Note: Cohort 1 = patients who discontinued triptans after one distinct triptan; Cohort 2 = patients who discontinued triptans after two distinct triptans; Cohort 3 = patients who discontinued triptans after three or more distinct triptans. Differences in each cohort are with respect to their respective controls.
Labour-market affiliation
Over the entire study period, the proportions unemployed were 3–7% higher in the three cohorts; in the index year, the proportions unemployed were 4–6% higher than in their respective matched controls (Figure 6a). The proportions on long-term sick leave were 2–7% higher in the three cohorts over the entire study period; in the index year, the proportions on long-term sick leave were 6–7% higher than in their respective matched controls (Figure 6b).
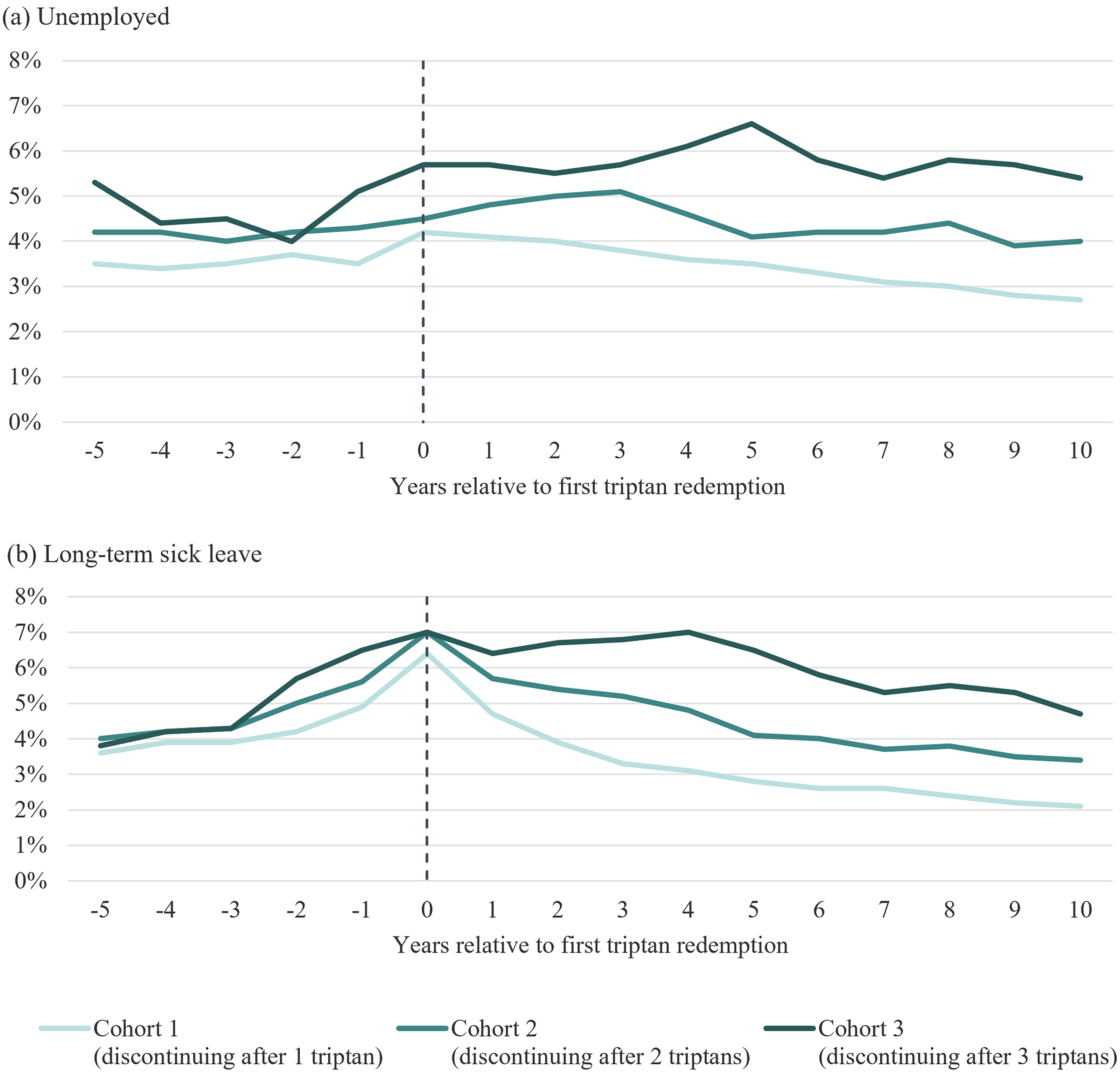
Annual differences in proportions who are (a) unemployed and (b) on long-term sick leave (more than four weeks) between the three cohorts and their respective controls. Note: Cohort 1 = patients who discontinued triptans after one distinct triptan; Cohort 2 = patients who discontinued triptans after two distinct triptans; Cohort 3 = patients who discontinued triptans after three or more distinct triptans.
Patients in all three cohorts had significantly higher risks of being in receipt of disability pension than their respective matched controls (HR = 1.59; 95% confidence interval= 1.55–1.62) for cohort 1; 1.73 (1.66–1.86) for cohort 2; and 1.84 (1.75–2.04) for cohort 3). Figure 7 shows the Kaplan–Meier probability curves for not being in receipt of disability pension. Schoenfeld residuals demonstrated time-varying covariance for cohort 1 and cohort 2. Therefore, HRs for five-year intervals were estimated (see Supporting information, Table S7). None of the sensitivity tests influenced our inferences from the results.

Kaplan–Meier curves for not being in receipt of disability pension among patients in the three cohorts and their corresponding matched controls: (a) Cohort 1 = patients who discontinued triptans after one distinct triptan; (b) Cohort 2 = patients who discontinued triptans after two distinct triptans; (c) Cohort 3 = patients who discontinued triptans after three or more distinct triptans.
Discussion
Using individual-level national register (real-world) data, the present study reveals substantial excess costs of illness among patients with migraine who initiate and then, for any reason, discontinue triptan treatment. Particularly high were the costs as a result of lower earnings combined with higher public transfer payments. The average per-patient costs over the period from year −5 to year 10 relative to the first triptan redemption were, for cohorts 1–3, EUR 9554, EUR 10,942 and EUR 12,812 in excess healthcare costs, EUR 27,964, EUR 35,920 and EUR 50,076 in lower earnings, and EUR 20,181, EUR 23,264 and EUR 26,459 in excess transfer payments. Thus, triptan discontinuers experienced lower gross earnings combined with more public transfer payments than their respective matched controls, reflecting their overall disaffiliation from the labour market, with cohort 3 > cohort 2 > cohort 1. These findings, also reflecting increased health-seeking behaviour among patients discontinuing triptan treatment, demonstrate high burden of disease at individual level that inevitably has impact at societal level. The excess costs are reasonably attributed to migraine because the only difference between triptan discontinuers and their corresponding matched controls was that the former had migraine (evidenced by use of triptans). The clear differences among the three cohorts suggest that the number of triptans tried, each presumably an attempt to improve treatment, is indicative of disease severity.
Our findings correlate well with those of other studies showing that migraine is associated with comorbidities, reduced productivity, lower income, more unemployment and greater use of healthcare resources (20–23). The mean annual cost per person in our study is higher than the findings from the Eurolight project (7), but with a lower ratio of indirect to direct costs (approximately 6:1 compared with approximately 10:1 in Eurolight 7 ). However, comparisons with Eurolight should be made cautiously. Eurolight, conducted over a decade ago, was survey-based among population samples, not all of which represented the general population adequately. 7 It considered all individuals with migraine, whereas the present study focused on those initiating and then, for any reason, discontinuing triptan treatment. It may be that such patients were not only at the higher end of the disease severity spectrum, but also among the group who were more challenging to treat. If so, higher overall costs might be expected. However, among the indirect costs, Eurolight assessed costs due both to reduced productivity at work and absenteeism from work, rather than lost income, which probably relates only to the latter. This difference may explain the high ratio of indirect to direct costs in Eurolight.
These are important findings, although subject to the study limitations discussed below. Patients who have discontinued triptans appear to have exhausted current treatment options, at least regarding acute treatment. In Denmark, they constitute a majority (59.5%) of those ever commencing triptans. The implications are two-fold: triptan discontinuers are numerous, and they have substantial unmet treatment needs. This situation exists in a country where healthcare for headache is as good as anywhere else in the world.
The reasons for triptan discontinuation are at issue, and several possibilities exist. Ineffectiveness and/or poor tolerability are the most likely, although there is clear evidence in this study that the great majority (81.8%) of those who discontinued triptans did so after trial only of one. This, despite both Danish and European guidelines recommending trial of at least three different triptans (9–11), suggests a healthcare failure in Denmark on an unexpectedly large scale. Other possible explanations for triptan discontinuation include improvement, such that triptans are no longer indicated. This improvement might be brought about by successful introduction of preventative medications, or it might represent spontaneous remission, which becomes increasingly probable with age, particularly in women passing the menopause (8,9). Alternatively, some patients, after trying one or more triptans, may decide that they prefer first-line therapy. These explanations are less likely given the observed continuing burdens.
Strengths and limitations
This study has several strengths. The real-world data from the Danish national registers are widely acknowledged to be some of the most comprehensive of their kind (24). The registers’ unique content of detailed and exhaustive individual-level records, covering the entire population over a long period of time, provide high-quality data with few missing observations (24,25). Because the registers collect data from every resident, and prospectively, these data are associated with minimal selection bias and no recall bias. The study population was large, reducing the risk of random variation influencing the findings. It included all incident triptan users aged 18 years or over who discontinued triptan treatment over a 22-year period. The controls, matched with patients for potential confounders, were randomly selected from the general population and thus consisted of both healthy and non-healthy persons, including people with migraine (albeit not prescribed triptans). Although the HRs estimated for the disability pension hazard for cohorts 1 and 2 breached the proportional hazards assumption, the time-stratified estimates presented in the supplementary analysis demonstrate that the estimates were nonetheless a valid representation of the actual hazards.
Social reforms, changes in treatment recommendations, an increase in the number (and formulations) of triptans on the market and marked reductions in the selling prices of some triptans with introduction of generic equivalents may have influenced our findings. However, because triptan commencements and discontinuations were distributed over 22 years, these changes were more likely to increase the uncertainty of estimates rather than introduce bias.
There were three study limitations. First, although triptan prescriptions for cluster headache were assumed not to influence the results, given the rarity of cluster headache, we also assumed that diagnoses of migraine leading to triptan prescription were accurate. There is no way of verifying this in the available registers. In addition, we cannot rule out that some individuals in the control populations had migraine.
Second, we assumed that that triptans were actually taken after prescription redemption, an assumption particularly precarious for those who made only a single redemption of triptans (75% of those in cohort 1). Strictly, those who did not try at least one dose (an unknown but probably small proportion) could not be considered triptan discontinuers. Nevertheless, cohort 1 generated findings of excess costs in line with (on a gradient) those of cohorts 2 and 3.
Third, and perhaps more significantly, none of the registers captures reduced productivity at work or short-term sick leave, with both more likely being consequences of migraine than long-term sick leave. Our findings regarding reduced labour-market affiliation among triptan discontinuers compared with the general population are therefore conservative, which strengthens rather than weakens the principal messages.
Finally, the attributable healthcare costs derived in the study were not migraine-specific. Because of the nature of the data, we do not claim a causal relationship between migraine and increased healthcare costs, but we observe a significant association between the two factors. In extension of this notion, we highlight that the control population was identified independently of other comorbidities than migraine. The mean non-migraine healthcare cost per individual in the two groups should therefore be comparable, and the attributable costs derived should reflect the added burden of disease associated with migraine.
Conclusions
Excess costs, both direct and indirect, among patients who have discontinued triptan treatment are high at an individual level and, by presumption, at a societal level. These patients comprise a sizable group, who appear to have exhausted current treatment options but still have substantial unmet treatment needs. Although emerging treatments for migraine offer a partial solution, there is evidence of healthcare failures in Denmark that will hinder their introduction. This is unlikely to be unique to Denmark, a high-income country with healthcare services among the best in the world: a similar if not greater challenge to the implementation of new treatments probably exists worldwide. If this is not addressed, the high costs and very considerable ill-health burden of inadequately treated migraine will persist undiminished.
Our findings are a potent encouragement for policymakers to revisit migraine management policies and systems of care. They clearly demonstrate unrecognized but heavy economic penalties on people and society that are associated with ineffectively treated migraine. They emphasize both the importance of adherence to clinical guidelines on use of triptans and the unmet need for alternative treatments for triptan non-responders.
Supplemental Material
sj-docx-1-cep-10.1177_03331024241269758 - Supplemental material for Cost of illness and labour market disaffiliation among patients with migraine discontinuing triptan treatment: A Danish nationwide register study from 1995 to 2021
Supplemental material, sj-docx-1-cep-10.1177_03331024241269758 for Cost of illness and labour market disaffiliation among patients with migraine discontinuing triptan treatment: A Danish nationwide register study from 1995 to 2021 by Messoud Ashina, Timothy J. Steiner, Jakob Møller Hansen, Daniel Sloth Hauberg, Ulla Sofie Lønberg, Maria Spanggaard, Jens Olsen, Sandra Elkjær Stallknecht and Thomas Folkmann Hansen in Cephalalgia
Footnotes
Author contributions
DSH and USL conceptualized the study. MS, JO and SES performed the literature search, ensured data collection, performed the analyses, created figures, and wrote the original draft of the manuscript. MA, JMH, TFH, TJS, DSH and USL contributed to data interpretation. All authors contributed to reviewing, revising and editing, and approved the final version of the manuscript submitted for publication. All authors consent to publication of this article.
Data availability
The data that support the findings of this study are available from Statistics Denmark's Research Service. However, restrictions apply to the availability of these data, which were used under licence/authorization for the current study and are not publicly available. Additional data analyses are available from the authors upon reasonable request and with permission from Statistics Denmark's Research Service.
Declaration of conflicting interests
MA reports receiving personal fees from AbbVie, Amgen, Astra Zeneca, Eli Lilly, GlaxoSmithKline, Lundbeck, Novartis, Pfizer and Teva Pharmaceuticals, and also reports serving as associate editor of Cephalalgia, associate editor of The Journal of Headache and Pain and associate editor of Brain. TJS is co-editor of the Journal of Headache and Pain, a director and trustee of Lifting The Burden, and reports receiving personal fees from Eli Lilly, Teva and Pfizer. JMH reports receiving personal fees from Pfizer. DSH and USL are current employees of Pfizer Denmark and may own shares in Pfizer Inc. MS, JO and SES are current employees of EY, a paid vendor to Pfizer. TFH declares no conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical statement
The study was register-based and complied with the regulations and instructions set up by Statistics Denmark. We used only anonymized data and present data only in aggregate and anonymous form. We did not contact or require any active participation from study participants. Ethics committee approval and written informed consent are not required for register-based research, according to Danish law.
Funding
This study was sponsored by Pfizer.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
