Abstract

Introduction
Consistent with the mission of the International Headache Society (IHS) to improve migraine management worldwide, this document focuses on providing practical recommendations on the preventive pharmacological management of migraine. Due to the inconsistent availability of medications across different regions of the globe, these recommendations are categorized into two levels:
In this second part of the IHS endeavor, we present the recommendations for the preventive pharmacological treatment of migraine together with the methodology and the evidence used to support them. Table 1 lists the drugs with evidence of efficacy for the preventive treatment of migraine listed in the WHO EML.
Drugs recommended for the preventive treatment of migraine by international guidelines, and their availability in the WHO model list of essential medicines, 23rd list (2023) (1).
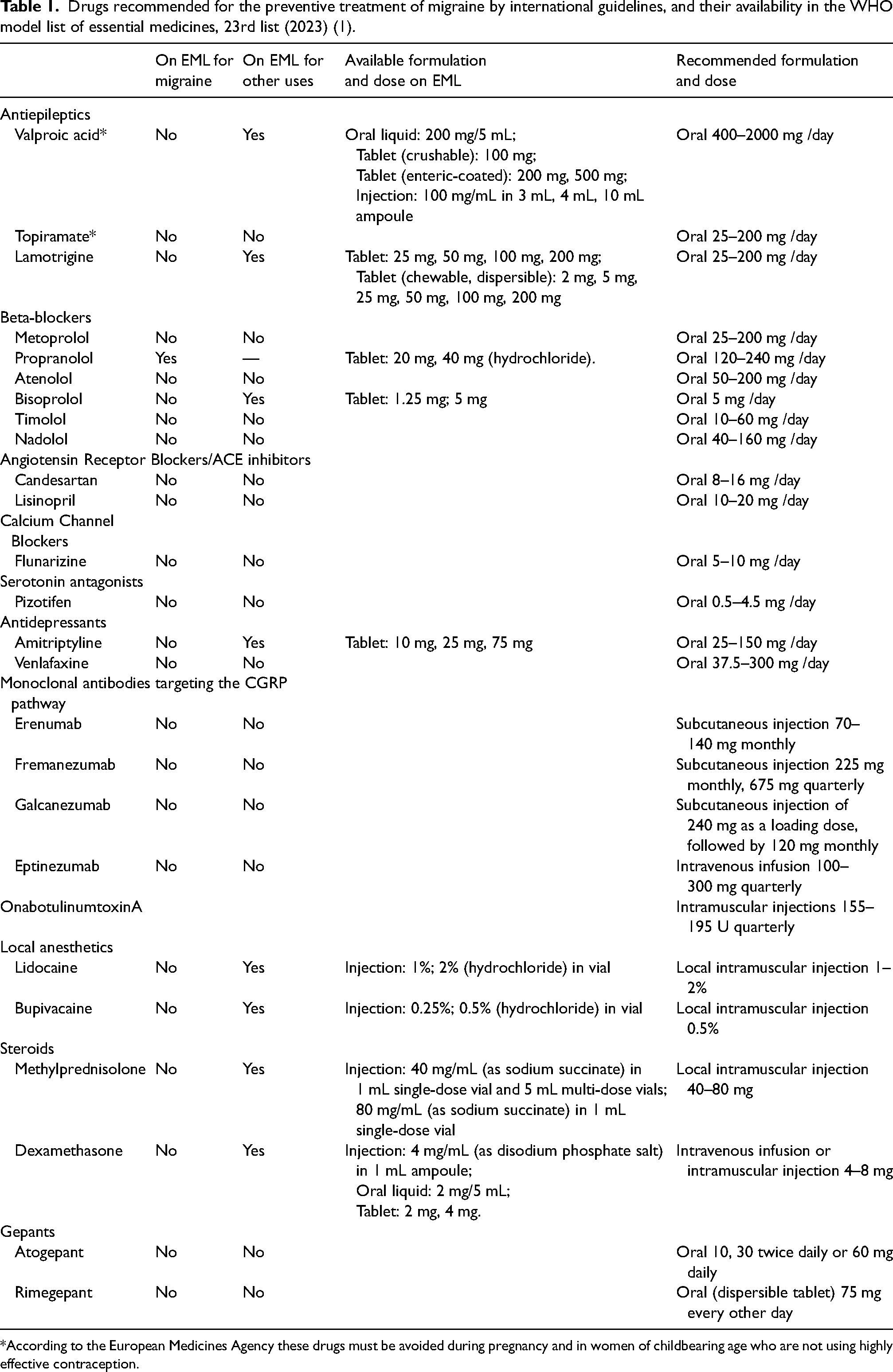
*According to the European Medicines Agency these drugs must be avoided during pregnancy and in women of childbearing age who are not using highly effective contraception.
The IHS practice recommendations are based on available treatment guidelines and expert consensus. They are intended to be a practical, quick reference, applicable in all countries across different care settings, including primary care. Given the global scope of these recommendations we have not customized the recommendations based on national registrations or specific labelling in individual countries. Nothing in these guidelines is designed to supersede local labelling and approvals.
These recommendations represent an instrument to motivate and facilitate policy changes. Our goal is to establish essential standards of migraine management in as many countries as possible. These standards will also serve as a reference document to drive local advances toward optimal care once essential standards of care are met.
Methodology used for the development of questions and recommendations
The methodology was similar to the one used for the IHS Practice Recommendations for the pharmacological acute treatment of migraine (2). The working group panel was nominated by the IHS board, selecting members based on their specific expertise in different areas of headache, previous experience developing guidelines or recommendations, and representation of different regions of the world. The group was gender-balanced and professional backgrounds included neurology, methodological expertise, evidence synthesis and statistics. Each senior member worked in collaboration with two junior headache experts from a different geographic origin for the analysis of the literature and the explanation of the recommendations. AC was specifically involved for his expertise in the methodology of evidence-based synthesis.
We used a consensus development panel approach, adapted from the methodology described and used by the US National Institutes of Health and WHO (3). The Steering Committee (HCD, MA, CT) developed an initial set of clinical questions in the Spring of 2022 based on the main issues that healthcare professionals may encounter in everyday practice when treating persons with migraine. The initial list of clinical questions was first shared and discussed with the coordinators (SS and FP) and, subsequently, with the entire working group (seniors and juniors) for interactive discussion and optimization. Following several iterations, the final set of clinical questions was agreed in the Fall of 2022.
AC devised and performed the search of the published literature to identify the National and International Guidelines for migraine treatment to be used for elaborating the recommendations. Considering the very specific questions and the relatively small amount of records overall for preventive treatment, AC ran and combined two searches: Search 1 (“Migraine Disorders” [MeSH Terms]) AND (“Secondary prevention” [MeSH Terms]) and Search 2 ((“Therapeutics” [Mesh]) AND (“Migraine Disorders” [Mesh]) AND (prevention OR prophylaxis)).
The following filters were applied to the searches: “Consensus Development Conference”, “Guideline”, “Guidelines”, “Meta-Analysis”, “NIH”, “Practice Guideline”, “Review”, “Systematic Review”.
FP and SS assessed the search output and selected a total of 15 national/international guidelines and guidance documents for elaborating the recommendations, based on: i) relevance of the paper; ii) publication date of less than 15 years prior; iii) availability in the English language. A further three guidelines (from German, Korean and Taiwanese societies) were added subsequently following either a translation to English being made available or internal suggestions coming directly from the working group (
The final list of clinical questions and the corresponding recommendations are summarized in Table 2.
Summary table illustrating the clinical questions for the pharmacological preventive treatment of migraine and the corresponding optimal and essential level recommendations.

CGRP: calcitonin gene-related peptide.
In the next sections we will illustrate in detail each clinical question, associated recommendations for the Optimal and Essential level, background for the question and evidence used for the elaboration.
Q1 – Which individuals with migraine are candidates for preventive pharmacologic treatment?
Recommendations
Optimal.
We suggest starting a migraine preventive treatment with drugs when one or more of the following conditions is present: - the person has 4 or more monthly headache days; - migraine has an impact on personal, social, and professional life according to personal patient perception; - optimized acute treatment is ineffective in providing migraine relief; - acute medications are used frequently to treat the attacks.
Essential.
As described in the optimal recommendations.
Comment: Due to the debilitating nature of migraine attacks and the potential risk of negative sequelae, preventive treatment can be considered with a lower threshold of monthly headache days in people with migraine with aura, especially hemiplegic migraine and migraine with prolonged aura.
Background
Migraine is a multifactorial disorder with a significant impact on patients' lives. The prevalence of migraine is the highest between 20 and 50 years of age, but people of all ages are affected (22). According to the latest assessment of the Global Burden of Disease, migraine is the first leading cause of disability due to a disorder of the nervous system worldwide in older children and adolescents aged 5–19 years and the second in people aged 20–59 years (23). Women are three times more affected than men (22). One of the pillars of treatment consists of preventive therapies to reduce the negative impact of migraine on everyday life by reducing the frequency of days with migraine. There are several different medication classes available for migraine prevention (e.g., anti-hypertensive, anti-epileptic, anti-depressant, treatments targeting the calcitonin gene-related peptide pathway). The decision to start a preventive migraine medication should take into consideration several factors, including frequency, severity, duration of migraine attacks, presence of other headaches (e.g. tension-type headache), magnitude of the impact that the migraine attacks have on daily functioning, effectiveness of acute medication, frequency of administration, and patient preference (24).
Evidence
Multiple headache expert groups and organizations have developed guidelines that provide recommendations on when preventive treatments should be considered for persons with migraine (summarized in Online Supplemental Table 1).
These guidelines consistently suggest that migraine frequency and migraine-associated disability are important factors when deciding to offer migraine preventive treatment with drugs. Most guidelines propose that migraine preventive medications should be offered if there is a minimum of two to four days with migraine per month. This is especially true if the migraine attacks cause disability and are not quickly responsive to acute migraine medications. People with migraine with attacks associated with prolonged and/or severe auras may be offered migraine preventive medications, regardless of attack frequency. If acute migraine treatment is not effective or the subject has contraindication to their use, preventive treatment should be offered. All people with migraine overusing acute medications, as well as those who are at risk of medication overuse should be offered preventive treatment. Other indicators for initiating preventive treatment are absences from work or school due to migraine and substantial negative impact on daily routines.
The decision to initiate preventive treatment must be made in agreement with the patient, adopting a personalized and shared decision-making process. Potential benefits, side effects, and risks associated with the use of preventive medications should be discussed and carefully considered. This discussion and decision-making process should include patient-specific factors such as age, child-bearing potential, presence of other health conditions, potential for medication interactions, and ability to adhere to the medication dosing schedule, amongst others.
Q2 – When should the effectiveness of a migraine preventive treatment be assessed?
Recommendations
Optimal.
In individuals initiating a new migraine oral preventive treatment, we suggest evaluating effectiveness* after three months at the target dose. We recommend a minimum of three months for injectable drugs taken on a monthly basis, and a minimum of six months for injectable drugs administered quarterly.
Essential.
In individuals initiating a new oral preventive treatment, we suggest evaluating effectiveness* after three months of treatment at target dose.
*See Q6 for the criteria for assessing the effectiveness of a preventive therapy.
Comment: Some evidence suggests that a small percentage of persons with chronic migraine treated with onabotulinumtoxinA may become responders at later times (after the second or third cycle). In non-responders to onabotulinumtoxinA after the first cycle, the dose should be increased from 155 U to 195 U in the subsequent cycle (25,26).
Background
The number of weeks needed for a preventive treatment to show maximum efficacy varies between drugs and subjects and may require several weeks. In contrast, adverse effects typically occur early. In the case of oral treatments, adverse effects may be minimized with a gradual titration.
Evidence
Preventive medication may show a rapid onset of benefit. This can occur within a week in the case of onabotulinumtoxinA, erenumab, galcanezumab, fremanezumab, eptinezumab and atogepant; or even within a few hours as described for eptinezumab and for rimegepant that has proven effective in the acute treatment of migraine also (27–35). However, most drugs require a minimum of four to six weeks after reaching the therapeutic dose to show their maximum therapeutic benefit (29–37). In addition, efficacy might be dose dependent, especially for traditional oral preventive drugs. In case of insufficient efficacy but good tolerability, the dose of these can be increased until the maximum tolerated or recommended dose (36–39).
Oral preventive treatment should be started and titrated progressively, to minimize adverse effects. Progressive titration of the selected drug may decrease the probability of adverse effects, and some adverse symptoms may dissipate after a few days of use (34–37). Many treatment-emergent adverse effects are drug-specific, and typically present early in treatment (40–43).
The response to preventive medications is highly heterogeneous among the migraine population. In addition, migraine is a cyclic and fluctuating disease, and the frequency of migraine attacks changes depending on several different factors (44). There is no consensus on the optimal time point to judge treatment efficacy, however, most randomized controlled trials (RCTs) and real-world studies assess the efficacy after three months of use, with some exceptions. Most recent guidelines recommend a minimum treatment duration of 4–12 weeks (Online Supplemental Table 2). A delayed response has been reported in some cases, however, and needs to be considered (45,46).
Q3 – If an initial migraine preventive drug is ineffective or not well tolerated, should alternative drug options be considered?
Recommendations
Optimal.
If an initial migraine preventive drug is ineffective* or not well tolerated, we suggest switching to a different class of medication. In individuals with multiple drug failures, a further option may be switching to a different preventive treatment in the same therapeutic class or to drugs such as onabotulinumtoxinA, monoclonal antibodies targeting calcitonin gene-related peptide (CGRP) and gepants, which have demonstrated efficacy in individuals who did not benefit from multiple previous preventive treatments.
Essential.
If an initial migraine preventive drug is ineffective* or not well tolerated, we suggest switching to a different class. In individuals with multiple drug failures, a further option may be switching to a different preventive treatment in the same class.
*Less than 30% reduction in monthly migraine days or moderate-to-severe headache days or rated as such by the person with migraine after an appropriate period of time (see Q2) at adequate doses.
Background
Migraine preventive drugs have a broad range of efficacy and side effects. In some cases, an initial preventive drug is ineffective or not tolerated, and therefore must be terminated. When it is determined that a migraine preventive medication is ineffective, or if it is not tolerated, switching to another migraine preventive medication is a reasonable next step (15,18,19).
Evidence
There is strong evidence that compliance with oral migraine preventive medications is low, mostly due to lack of long-term efficacy and side effects. A 2017 retrospective analysis in the US revealed that only 25% of people with chronic migraine who started a preventive drug persisted with the treatment after six months (45). Among participants who discontinued, 23% switched to another preventive drug. Pooled persistence from 19 RCTs on propranolol, amitriptyline, and topiramate showed rates of 77%, 55%, and 57%, respectively, at 16–26 weeks (47). Adverse events were the most common reason for discontinuation.
There is a lack of studies that have directly investigated the methods for switching migraine preventive medications and the related outcomes. There are several recently completed clinical trials, however, demonstrating that people with migraine who have not responded to or not tolerated prior migraine preventive medications can respond to other pharmacologic options. The monoclonal antibodies targeting the calcitonin gene-related pathway (anti-CGRP mAbs), for example, have been studied for the treatment of both episodic and chronic migraine in participants who had failed previous preventive treatment (48–50). OnabotulinumtoxinA has demonstrated efficacy in people with chronic migraine and prior preventive treatment failures (51–53).
Most guidelines suggest switching preventive treatments in case of ineffectiveness after an adequate trial, although the length of ‘adequate’ may vary, and periodic re-evaluation is recommended. Online Supplemental Table 3 summarizes guidance on the timing for assessing outcomes with migraine preventive medications and switching such medications reported in the guidelines and guidance documents assessed.
Q4 – If a migraine preventive drug is ineffective, is it appropriate to use a combination therapy with two migraine preventive drugs?
Recommendations
Optimal.
We suggest combination therapy with two migraine preventive agents in individuals who do not have enough benefit from any single migraine preventive treatment, or if the combination of two agents represents an advantage on the management of comorbidities.
Drugs targeting the CGRP pathway and onabotulinumtoxinA have very low/absent drug-to-drug interactions and they can therefore be easily combined with oral preventive treatment.
Essential.
We suggest a combination therapy with two migraine preventive agents in individuals who do not have enough benefit from any single migraine preventive treatment, or if the combination of two agents represents an advantage on the management of comorbidities.
Oral preventive treatments have several drug-to-drug interactions, therefore caution is suggested when combining them. On the other hand, some combinations may improve the tolerability of individual drugs.
Background
Preventive migraine options consist of several evidence-based pharmaceutical interventions as well as nonpharmacologic treatments including lifestyle changes and behavioral approaches.
Recommended migraine preventives include beta-blockers, topiramate, valproate, flunarizine, amitriptyline and angiotensin receptor blockers, as well as newer migraine specific anti-CGRP mAbs and gepants. Injections with onabotulinumtoxinA are also approved in several countries, albeit for chronic migraine only. Effectiveness of drug prophylaxis is generally defined as a 50% reduction in monthly migraine days in episodic migraine and 30% for chronic migraine. Drug prophylaxis is sometimes not (or only partially) effective, and adherence may be limited by tolerability.
Evidence
No formal evidence exists, in the form of controlled studies, for combination therapy of two preventives (54). In one study, the specific combination of topiramate and propranolol did not provide additional benefits compared to the use of topiramate alone or placebo (55). Similarly, the use of combined tricyclic antidepressant and propranolol was no better than propranolol monotherapy (54). The combination of propranolol with nortriptyline did not provide better results than propranolol alone (56). Anecdotal evidence exists that the addition of anti-CGRP mAbs to onabotulinumtoxinA may lead to a further reduction in headache frequency in some persons with chronic migraine (57–59). An open trial showed that combining a beta-blocker and sodium valproate could lead to an advantage in efficacy in participants with migraine previously resistant to the two medications in monotherapy (60). Similar benefits were found for combining beta-blocker and topiramate (59) as well as topiramate and nortriptyline (61) in individuals resistant to both respective monotherapies. Examples of viable combinations with preventive drugs for migraine are provided in Table 3.
Possible combinations of preventive drugs for migraine based on mechanisms of action of the drugs or their drug-to-drug interaction profile.

CGRP: calcitonin gene-related peptide.
We suggest monotherapy as a goal in addition to lifestyle modification and other non-medical therapies when possible. In cases of partial (or no) effectiveness, the adherence to treatment, adequate trial of at least eight weeks at the target dose, and any potential medication overuse should be reviewed. In people with migraine with unmet needs despite monotherapy plus lifestyle changes and behavioral support, combinations can be considered. To select the drugs to be combined it is necessary to consider the individual patient, their medical and other co-morbidities, personal preferences, side effect profiles, and route of administration.
The statements on the combination of two preventives reported in the guidelines reviewed and the guidance documents assessed are summarized in Online Supplemental Table 4.
Q5 – How long should effective migraine prevention be continued?
Recommendations
Optimal.
An effective preventive treatment should be continued for at least six months for oral drugs and 12 months for non-oral treatments before considering discontinuation. For people with chronic migraine, longer treatment periods should be considered.
The decision to stop a migraine preventive drug should be based on having less than four monthly migraine days over a period of three consecutive months or based on the patient's satisfaction with the reduction of disease burden achieved.
Essential.
An effective preventive treatment with available oral drugs should be continued for at least six months. For chronic migraine longer treatment periods should be considered.
The decision to stop a migraine preventive drug should be based on having less than four monthly migraine days over a period of three consecutive months or based on the patient's satisfaction with the reduction of disease burden achieved.
Background
Several drugs have shown efficacy in the preventive treatment of migraine. Most randomized clinical trials evaluate the efficacy of preventive medications after three to six months. The IHS Guidelines for controlled trials of preventive treatment of chronic and episodic migraine suggest the use of a double-blind treatment phase lasting at least 12 weeks (62,63). These Guidelines suggest that trials of 24 weeks or longer may be useful in evaluating cumulative benefit, persistence of efficacy, adherence to treatment, safety, and tolerability.
Evidence
Most RCTs and real-world evidence studies (RWE) evaluated the efficacy of treatment after three or six months (64–66). Some open-label studies and a few RCTs reported the persistent effectiveness of some treatments over longer treatment periods (51,67–70). Based on this evidence, most guidelines recommend the use of preventive treatments for 6–12 months (Online Supplemental Table 5) (71,72).
The vast majority of RCTs and RWE studies focused on the effect of the treatment on migraine during the treatment phase, however, and information regarding the discontinuation phase is limited. This post-treatment period observation would be highly beneficial in order to identify possible rebound phenomena, but this is not always evaluated or reported in the clinical trials (62,63).
To date, one double-blind RCT assessed outcome in participants continuing topiramate for six additional months open-label, after a six-month treatment period compared to placebo. Placebo-treated subjects showed a higher increase in the number of migraine days and reported lower quality-of-life compared to participants who continued the intake of topiramate (73). Open-label studies have reported sustained benefit after discontinuation of other oral preventive drugs, onabotulinumtoxinA and CGRP mAbs (74–79). The sustained response is not, however, universal. In some subjects, the therapeutic benefit is short-lasting, and they return to their pre-treatment baseline (80). In these subjects, treatment should be restarted (81). Most international guidelines recommend the use of preventive treatments for six or 12-month periods (Online Supplemental Table 5).
Q6 – What are the success criteria to rate preventive therapy as effective?
Recommendations
Optimal.
The success criteria that determine whether preventive therapy is effective and should be continued according to the recommendations provided in Q5 are any one or more of the following: - a ≥ 50% decrease in monthly migraine days or moderate-to-severe headache days, optimally based on the use of a headache diary; - a clinically meaningful subjective improvement as reported by the person with migraine or evaluated by Patient Global Impression scales - a clinically meaningful improvement in MIDAS or HIT-6 questionnaire scores. For patients with chronic migraine who have not achieved a ≥ 50% decrease in monthly migraine days or moderate-to-severe headache days with multiple preventive options (including oral drugs, onabotulinumtoxinA and drugs targeting the CGRP pathway), a ≥ 30% decrease in monthly migraine days or moderate-to-severe headache days is acceptable for treatment continuation beyond three months.
Essential.
Given the limited number of options available, it is recommended to continue treatment in all persons with migraine that report a meaningful subjective improvement and who do not have tolerability issues or unwanted side effects.
Background
Expert recommendations suggest that a two- to three-month evaluation period (starting once the maximally tolerated dose has been reached) represents the ideal time interval to evaluate whether a given preventive medication is effective (82). Based on clinical trials and real-world data, most guidelines and recommendations indicate a 50% reduction in the frequency of migraine attacks or monthly migraine days (MMD) or monthly headache days (MHD) as an acceptable measure of success (Online Supplemental Table 6) (5,7,8,18). A 30% reduction is acceptable for chronic migraine. Clinical trials, however, are designed to evaluate treatment efficacy in ‘controlled’ conditions, and thus the 50% reduction in monthly migraine frequency or MMD may not accurately depict treatment effectiveness in everyday clinical practice. Therefore, in addition to headache frequency and intensity responses, patient-centered outcomes (such as disability, quality of life, medication use for acute migraine attacks, and effect) should be considered (9,14,16).
Evidence
Although used in multiple clinical trials, a 50% response in the reduction of migraine days per month might be too aggressive in clinical practice, and beyond the satisfaction threshold of the person with migraine (14,18). Reductions in headache intensity appear to be as significant for people with migraine as reductions in headache frequency (83,84). Patient-Reported Outcomes (PRO) evaluation tools such as the Migraine-Specific Quality of Life (MSQ) questionnaire and Patient Global Impression of Change (PGIC) can also be considered (85) and have been associated with treatment continuation in a real-world evidence study (86).
The clinically meaningful improvement in MIDAS score is defined by the AHS as i) reduction of ≥5 points for baseline score 11–20 or ii) reduction of ≥30% for baseline score ≥20. The clinically meaningful improvement in HIT-6 score is a reduction of ≥ 5 points (9).
Q7 – In individuals who have discontinued a migraine preventive after a successful period, what are the criteria for restarting preventive treatment?
Recommendations
Optimal.
In individuals who have discontinued migraine prevention after a successful period of treatment, we suggest waiting at least one month before considering restarting treatment, provided they still satisfy the criteria for prevention (refer to Q1).
Monitoring of headache frequency with a headache diary or a monthly calendar is recommended after stopping a migraine preventive treatment.
Essential.
As described in the optimal recommendations.
Background
Preventive treatment holidays may be recommended in subjects experiencing a meaningful reduction in migraine days per month who are not severely disabled, in order to determine whether a given preventive is still required and to avoid unnecessary drug exposure (72,82,87). Research suggests that there might be a sustained effect after cessation of certain migraine preventive treatment (73,77,79), however, evidence-based monitoring algorithms for these subjects are lacking.
Evidence
Only one-quarter of persons with migraine adequately responding to prevention with flunarizine or beta-blockers exhibited a sustained response 18 months after terminating treatment. Most (75%) subjects experienced an increase in migraine frequency after a variable period following the discontinuation ranging between one to 28 months (with a mean of 7.2 and 4.4 months in the flunarizine and beta-blockers groups, respectively) (79). Higher attack frequency at baseline, prior history of medication overuse, and poor response to previous preventives were predictors of headache recurrence, decreased effectiveness of subsequent prophylaxis, and lower willingness to receive a new drug (79). A similar six-month study comparing flunarizine with nimodipine reported significant reductions in attack frequency, severity, and duration after discontinuation lasting 8.4 months with the former and 4.9 months with the latter (75). Prevention with topiramate for an additional six-month period after a six-month open label treatment showed sustained improvements with respect to baseline and significantly improved efficacy compared to placebo (73). In contrast, sodium valproate induced sustained relief in only 40% of subjects, although this study included only participants with what is now known as medication overuse headache, and the treatment phase lasted no more than three months (88).
Current data is no better for injectable treatments. One real-life study found that 31% of participants treated with erenumab restarted treatment due to disease rebound to baseline levels at week 4 after treatment completion (87). Furthermore, a pooled analysis of two phase III trials of the anti-CGRP mAb galcanezumab showed that quality of life at the end of a four-month post-treatment period was no different between galcanezumab-treated patients and those who received placebo (89). In a cohort of subjects with migraine treated with erenumab or galcanezumab, ≥ 50% responders-rate dropped from 73.3% to 27.6% in participants with high-frequency episodic migraine, and from 60.6% to 35.5% in participants with chronic migraine at the end of the third month of suspension (90). In another study, almost 90% of participants who had interrupted preventive therapy with CGRP mAbs restarted treatment (91). Additional evidence suggests that, in comparison, migraine worsening might be faster following erenumab discontinuation compared to galcanezumab or fremanezumab suspension (78). This difference is only temporary, however, and all treatment groups return to baseline values in the end (78). Regarding onabotulinumtoxinA, a sustained response at six months has been reported in people with chronic migraine who attained a reduction in headache days to less than five per month, and of migraine-related disability to mild or less, representing around 40% of the total number of treated subjects (74).
Online Supplemental Table 7 reports the statements regarding monitoring of people with migraine after discontinuation of the preventive treatment in the guidelines reviewed and the guidance documents assessed.
Q8 – Should the choice of migraine preventive drugs be determined by the presence of comorbidities?
Recommendations
Optimal.
The first treatment option should always be a migraine-specific drug.
In individuals with migraine and an ongoing comorbid condition, it is possible to use agents that can provide benefit on both the migraine and the existing comorbidity. This applies specifically to oral preventive treatments (see Table 4).
Medications preferred or avoided for preventive treatment of migraine depending on comorbidities.

ACEIs: angiotensin-converting enzyme (ACE) inhibitors; ARBs: Angiotensin II receptor blockers; SNRIs: Serotonin–norepinephrine reuptake inhibitors.
Essential.
When migraine-specific treatments are not available, the choice of oral preventive treatment should be guided by the presence of comorbidities. We therefore suggest using agents that can provide benefit on both migraine and the existing comorbidity as first line preventive therapy.
Background
Migraine can be associated with several comorbid conditions. Some comorbidities, such as anxiety, depression, acute medication overuse, obesity or insomnia, may also represent a risk factor for the development of chronic migraine (92). Comorbidity is defined as an illness that occurs more frequently in association with a specific disorder than that would be found as a coincidental association in the general population.
Given the numerous drug options available for migraine prevention and the range of existing comorbidities, it can be difficult to predict which drug will a) be safe, b) be tolerable, and c) achieve the highest efficacy. In subjects with comorbidities, oral drugs can offer the opportunity to improve both migraine and the comorbid condition. Examples of this include the use of topiramate for migraine with comorbid obesity or essential tremor, amitriptyline for comorbid insomnia or depression, valproate for bipolar mood disorder or venlafaxine for comorbid depression or generalized anxiety. In certain comorbidities, however, there are drugs that should be avoided, such as valproate in obesity or propranolol in asthma or Raynaud's syndrome. Issues regarding dosing and selection of agents should always be considered. Doses of amitriptyline for depression, for example, are much higher than doses needed for migraine prevention. These higher doses of amitriptyline typically cause more adverse events than lower doses.
The option to treat two conditions with one drug is not typically possible with migraine-specific drugs, although their reduction of migraine symptoms may have an impact on other comorbidities; examples include improving low mood or insomnia when these are caused by migraine pain itself (93). It has also been suggested that the efficacy of anti-CGRP mAbs appears to be independent from comorbidities such as depression.
Evidence
Migraine is associated with both comorbid and concomitant illnesses that influence treatment strategy (92). Various drugs not only have a preventive effect on migraine but also target frequently occurring comorbid conditions (93,94).
A comprehensive list of medications preferred or avoided in the preventive treatment of migraine depending on comorbidities can be found in Table 4.
The approach of using one drug to treat two conditions may, at the same time, also have limitations. One single medication may not treat two different conditions optimally, sometimes two or more medications are required in optimized dosages. Close monitoring after starting one (or multiple) drugs in migraine prevention and comorbidity treatment is usually necessary and referral to an appropriate specialist may be required. Available guidelines on this topic are summarized in Online Supplemental Table 8. It is worth mentioning that some of the guidelines were developed before the advent of migraine-specific treatments.
Q9 – Which preventive medication is suggested for people with chronic migraine?
Recommendations
Optimal.
We suggest atogepant, erenumab, eptinezumab, fremanezumab, galcanezumab, onabotulinumtoxinA and topiramate for people with chronic migraine.
Of note, topiramate should be avoided during pregnancy and in women of childbearing age who are not using highly effective contraception methods.
Essential.
We suggest topiramate, which however must be avoided during pregnancy and in women of childbearing age who are not using highly effective contraception methods. If topiramate is not effective, not tolerated or not indicated, amitriptyline, beta-blockers, valproate can be used.
Combination of two drugs may be necessary, following recommendations provided in Q4.
Background
The current concept of chronic migraine is relatively recent (95). Several drugs approved for preventive migraine treatment were investigated before chronic migraine was officially included in the ICHD and have not been specifically tested in this subgroup of subjects. When compared to episodic migraine, fewer drugs have been formally tested and approved specifically for chronic migraine patients. Nevertheless, most guidelines consider that preventive drugs with good evidence for the treatment of episodic migraine might also be used for the treatment of chronic migraine, without presenting evidence for the recommendation (Online Supplemental Table 9).
Evidence
Two randomized double-blinded studies support the use of topiramate for the treatment of chronic migraine (36,96). OnabotulinumtoxinA is approved in several countries for the treatment of chronic migraine. The evidence is based primarily on one positive study (PREEMPT 2) and on the pooled analysis of two studies (PREEMPT 1 and PREEMPT 2) (97,98). CGRP mAbs were studied for the treatment of migraine and consistently demonstrated efficacy (49,50,99–102). Among the class of small molecules antagonists of the CGRP receptor, atogepant presented efficacy for the treatment of chronic migraine (103). This trial was recently published and is not yet included in available guidelines. Other drugs with some evidence of efficacy for chronic migraine treatment include valproate and propranolol (104–106). Flunarizine is used in several European countries, with a recommended dose of 5–10 mg, however published trials showing evidence for its efficacy are mostly outdated and of limited quality (107).
Q10 – If a monoclonal antibody targeting the CGRP pathway is not effective, is it beneficial to switch to another anti-CGRP monoclonal antibody?
Recommendations
Optimal.
If a monoclonal antibody targeting the CGRP pathway (anti-CGRP mAb) is ineffective, we suggest switching to another anti-CGRP mAb in individuals who have no other viable therapeutic options among other migraine preventive drug classes due to ineffectiveness, contraindications or tolerability issues.
Essential.
Not applicable.
Background
CGRP-targeted therapies are effective for migraine prevention. The 50% responder rates range from 30% to 60%. Therefore 40–70% of people with migraine do not respond adequately to the CGRP mAbs (108).
Currently, there are up to four CGRP mAbs available in different countries. They have different characteristics in terms of target (CGRP peptide vs. receptor), route of administration (subcutaneous vs. intravenous), flexibility of dose escalation (70 to 140 mg for erenumab and 100 to 300 mg for eptinezumab) and injection schedule (monthly vs. quarterly for fremanezumab) (108).
Evidence
No controlled head-to-head studies have examined the comparative efficacy of monoclonal antibodies targeting the CGRP pathway. Meta-analyses have shown comparable efficacy, safety and tolerability across the four antibodies (42,66).
Some real-world studies reported the outcome of switching from one to another monoclonal antibody in participants who had insufficient response to the first treatment. One multicenter retrospective study from Germany assessed the ≥30% responder rate at month three after switching from erenumab to another antibody (galcanezumab or fremanezumab). The switch from erenumab led to a ≥ 30% response in one-third (32%) of the participants after three months of treatment. A ≥ 50% response was achieved in 12% of the participants (109). Another study from Spain reported a 30% response rate in 50% participants and a 50% response rate in 27% of them when galcanezumab was administered to 15 erenumab non-responders (110).
Despite limited available evidence at this time regarding the efficacy of antibody switching, several guidelines suggest switching as a potential option (Online Supplemental Table 10). The guidelines of the European Headache Federation state that there is insufficient scientific evidence on the potential benefits of antibody switch, but switching may be an option (19). The guidance of the American Headache Society emphasizes that the efficacy and tolerability should be individually evaluated and determined, and that clinical decision making regarding switching to an alternative treatment should be made in a patient-centric fashion (16).
Q11 – What are the possible preventive options for people with migraine who have a positive response to a monoclonal antibody targeting the CGRP pathway, but still have a clinically meaningful residual migraine burden?
Recommendations
Optimal.
In people with migraine who have a positive response to one monoclonal antibody targeting CGRP but still have a clinically meaningful residual migraine burden, we suggest adding a traditional oral preventive drug or, if the person has chronic migraine, onabotulinumtoxinA.
Essential.
Not applicable.
Background
CGRP-targeted therapies induce a 50% reduction of MMDs in a percentage of persons with migraine ranging from 30 to 60% (111). In people with high frequency of chronic migraine, the remaining disease burden may therefore still be high.
Evidence
Preclinical studies suggest that onabotulinumtoxinA and monoclonal antibodies targeting CGRP exert a synergistic effect within the trigeminovascular system, thus favoring this combination (112). RCTs, however, on the efficacy and safety of this dual therapy for migraine prevention are lacking. To date, a limited number of real-world studies have reported that partial responders to onabotulinumtoxinA may benefit from dual therapy with anti CGRP antibodies as an add-on. There is also no evidence that this combination would be harmful. A meta-analysis investigating real-world evidence, showed a reduction of migraine headache days in subjects with migraine who received the combined therapy in comparison to onabotulinumtoxinA alone (113). Those studies suffer from limitations due to the observational nature of the evidence. Additionally, it is unclear if the same benefit could have been obtained with the anti CGRP antibody alone.
Regarding dual therapy with CGRP targeted therapies, the guidelines of the European Headache Federation stated that there is insufficient evidence to make suggestions that the combination of CGRP mAbs with other preventives improves migraine clinical outcomes (19). The guidance document of the American Headache Society suggests adding CGRP mAbs to the existing preventive drugs, without introducing other changes until the effectiveness of the anti-CGRP therapy is observed (16). Considering the favorable tolerability profile of CGRP mAbs and CGRP receptor antagonists, it is possible that their combination may improve the efficacy, without affecting tolerability, but evidence-based data is needed for a clearer view of this option.
The relevant statements of the European Headache Federation and of the American Headache Society are reported in Online Supplemental Table 11.
Q12 – Do greater occipital nerve blocks have a place in migraine prevention?
Recommendations
Optimal.
Greater occipital nerve blocks with local anesthetics, with or without corticosteroids, have limited evidence of efficacy, but may represent an option for migraine prevention. They can also be used in pregnant women.
Essential.
As described in the optimal recommendations.
Comment: Videos on how to perform peripheral nerve anesthetic/steroid blocks are available from the IHS education platform in multiple languages. The links to two videos are provided below:
- Peripheral nerve blocks in headache patients: indications and practical guide; https://www.youtube.com/watch?v=frlojGkX7C4 - GON blocks– general aspects and indications;
Background
Peripheral nerve blocks have emerged over the past decades as a potential treatment option for migraine. The detailed mechanisms of migraine alleviation by peripheral nerve blocks remain uncertain. Modulation of central sensitization is a potential candidate (114). Whether short-acting local anesthetics with/without corticosteroids have a place in migraine prevention is under debate.
Evidence
Several RCTs evaluated the effectiveness of greater occipital nerve blocks (GONB) in migraine prevention. Nine RCTs in particular have assessed GONB outcomes at least four weeks after the first injection.
Six studies (115–120) were conducted in participants with chronic migraine. A meta-analysis (121) included four of them. In the active group, participants received GONB with bupivacaine 0.5% in three studies (pooled n = 66) and lidocaine 2% in the remaining one study (n = 17). The meta-analysis revealed that headache frequency decreased significantly in the active group receiving during the first (−4.45 days, 95%CI: −6.56 to −2.34 days) and second (−5.49 days, 95% CI: −8.94 to −2.03 days) month as compared to the placebo group. Of note, none of the RCTs reported the preferred primary endpoint suggested by the IHS guidelines for controlled trials of preventive treatment (62,63). Instead, all four studies cross-sectionally compared the pooled headache frequency between active and placebo groups at checkpoints, or longitudinally compared the pooled headache frequency between baseline and treatment phases within each group.
One RCT (116) reported that after receiving three monthly GONB with 2% lidocaine 2 mL for each injection, the reduction in monthly headache days and monthly migraine days from baseline, as well as the 50% responder rate in headache days, were significantly greater in the active group by the end of the blinded phase. The authors ensured blinding by preparing the injection site with lidocaine gel before the actual injection. The same team also conducted an RCT evaluating the combination effect of GONB and topiramate (115). The participants were randomized and allocated to three groups: topiramate monotherapy, topiramate and GONB with lidocaine + methylprednisolone, and topiramate and GONB with lidocaine only. The results showed that as compared with topiramate monotherapy, groups with add-on GONB (with or without methylprednisolone) had greater reductions in monthly migraine days by the end of the study. Groups with add-on GONB (with or without methylprednisolone) had a higher 50% responder rate in monthly headache days. There were no significant differences between GONB with or without methylprednisolone. To sum up, despite the high heterogeneity between different trials, GONB may be beneficial in the prevention of chronic migraine.
Two placebo-controlled RCTs focused on participants with episodic migraine. In the first (119), weekly injections of GONB plus supraorbital nerve block were administered for three weeks, and reported that the active group had lower headache frequency at the second month. The other study (122) randomly assigned participants into four groups: placebo (saline), lidocaine, triamcinolone, and lidocaine plus triamcinolone. As compared to baseline, the groups receiving GONB with lidocaine and lidocaine plus triamcinolone had lower headache frequency four weeks after a single GONB. However, there were no significant differences between the four groups. Of note, these two studies were also weakened by the abovementioned issue of applying inappropriate primary endpoints.
The remaining two RCTs did not specify whether the participants were diagnosed with episodic or chronic migraine. One study (117) reported negative results with no significant differences in the 50% responder rate between the active group (single GONB with 0.5% bupivacaine + methylprednisolone) and placebo group. Interestingly, to ensure adequate blinding, the authors mimicked the numbness over the injection site by adding 0.25 mL 1% lidocaine into 2.75 mL saline. The possible therapeutic effect of lidocaine, despite its small amount, may have interfered with the results. Another RCT (121) explored triamcinolone in GONB. The participants randomly received a single GONB with 2% lidocaine or 2% lidocaine plus triamcinolone. The preventive effect of lidocaine alone and lidocaine plus triamcinolone was comparable at the 2nd, 6th, and 8th week, as indicated by a similar reduction in headache frequency.
Three RCTs (115,117,122) did not demonstrate the additional benefits of injected corticosteroids. Taking the potential adverse effects of corticosteroid injection (e.g., subcutaneous fat atrophy, hypopigmentation, alopecia) into consideration, use of corticosteroids should be carefully evaluated.
Online Supplemental Table 12 summarizes the statements regarding greater occipital nerve blocks in migraine preventive treatment in the guidelines reviewed and the guidance documents assessed.
Q13 – What are the suggested drugs for migraine prevention in children and adolescents?
Recommendations
Optimal.
In children and adolescents with migraine who need pharmacological migraine prevention, beta-blockers or flunarizine, at doses adapted to body weight, can be used, although evidence of efficacy is very limited.
Essential.
In children and adolescents with migraine who need pharmacologic migraine prevention, beta-blockers, at doses adapted to body weight, can be used, although evidence of efficacy is very limited.
In case of ineffectiveness, low dose topiramate or amitriptyline represent possible alternatives.
Comment: Topiramate should be avoided in female adolescents of childbearing age who are not using highly effective contraception methods.
Background
Migraine is frequently reported among children and adolescents, with an estimated prevalence of ∼8%, and its prevalence further increases throughout adolescence (123,124). In school-age children, migraine is associated with more frequent school absenteeism and reduced performance, compared to classmates with no migraine (125). Preventive therapy can be an option when bed rest and acute medication provide insufficient pain relief or when attacks are frequent (71).
Evidence
Evidence is largely lacking for migraine prevention in the pediatric population. The Childhood and Adolescent Migraine Prevention (CHAMP) study, a National Institutes of Health–funded multicenter RCT, is the largest study to evaluate the efficacy of pharmacological therapy for the prevention of migraine in children and adolescents aged 8–17 years (126). This study aimed to compare the preventive effect of topiramate, amitriptyline, and placebo in children with episodic and chronic migraine. The study was stopped early for futility after a planned interim analysis. In all three groups, 50–60% of participants achieved the primary outcomes, defined as ≥50% reduction in monthly headache days in the last 28 days of the 24-week treatment compared to baseline. The study also reported more side effects among the topiramate and amitriptyline groups, compared to the placebo group. The authors concluded that the risk:benefit profile of topiramate and amitriptyline is unfavorable. The study, however, showed a placebo response which was higher than in adults.
The effect of flunarizine in children has been documented in one large meta-analysis and seems comparable to that of propranolol (127).
Antiepileptics
Based on data from a pilot study (126), two small RCTs (128,129), and a post hoc subgroup analysis (130), topiramate has been suggested to be more effective than placebo at reducing the frequency of monthly headache and migraine days. The Food and Drug Administration (FDA) has also approved topiramate for migraine prevention in children aged 12 and older. In contrast, the CHAMP study did not support these results (126). Nor did a meta-analysis on the efficacy of topiramate, which found no differences between topiramate and placebo for achieving the ≥50% reduction in headache frequency during treatment (131). Thus, the available data is inconclusive regarding whether pediatric subjects with migraine treated with topiramate are more prone than those who receive placebo to experience ≥50% reduction in monthly headache or migraine days (10). However, potential side effects following topiramate treatment, such as cognitive disorder, paresthesia and fatigue are well-known (126). Therefore, we recommend that physicians discuss the available evidence and side effects of topiramate in the prevention of pediatric migraine.
Sodium valproate has not been shown to be better than placebo and is associated with adverse events, suggesting it is not an ideal choice in children and adolescents.
Tricyclic antidepressants
One prospective, open-label, single center study demonstrated that amitriptyline can reduce the incidence and intensity of different types of headaches in children (132). Among the different headache disorders included, migraine was the most prevalent diagnosis. The study did not use a ≥ 50% reduction in headache frequency as the primary outcome. We therefore do not believe that there is sufficient data to determine whether pediatric individuals with migraine treated with amitriptyline are more prone than those who receive placebo to experience ≥50% reduction in monthly headache or migraine days (132).
Beta-blockers
One network meta-analysis published in 2020 reviewed whether preventive pharmacologic treatments are more effective than placebo in pediatric migraine (133). The authors revealed a significant effect of propranolol 60–120 mg daily compared with placebo. There were no significant differences in adverse events between propranolol and placebo in the short term, while in the long-term analysis, propranolol was not more acceptable than placebo. Therefore, it is possible that pediatric individuals with migraine treated with propranolol are more prone than those who receive placebo to experience ≥50% reduction in monthly headache or migraine days (10). However, we recommend weighing the benefits of propranolol against its potential harms.
Online Supplemental Table 13 reports the statements on pharmacological migraine prevention for children and adolescents in the guidelines reviewed and the guidance documents assessed.
Q14 – Which migraine preventive drugs can be used during pregnancy and lactation?
Recommendations
Optimal.
Pregnancy
We suggest non-pharmacologic treatments or peripheral nerve blocks (see Q12) as first line options in pregnant or breastfeeding women. If non-pharmacological treatment is not possible or effective, we suggest propranolol or amitriptyline, after balancing risks and benefits and informing the subject on potential secondary effects and associated risks. Propranolol should be discontinued in the last part of the third trimester to avoid the risk of adverse events to the fetus and neonate.
For persons with chronic migraine, onabotulinumtoxinA may represent an option after balancing risks and benefits given the limited systemic effects.
We strongly recommend avoiding valproate and topiramate during pregnancy due to their teratogenic effects. Similarly, candesartan and lisinopril should be avoided during pregnancy because of the risk of harm and malformation to the fetus.
Breastfeeding
During breastfeeding, non-pharmacologic treatment or peripheral nerve blocks should be considered first line. If non-pharmacologic treatment is not possible or effective, we suggest propranolol or amitriptyline, after informing the subjects of potential secondary effects and associated risks, or onabotulinumtoxinA for persons with chronic migraine. Candesartan can be used with caution. Lisinopril should be avoided.
Monoclonal antibodies targeting CGRP can be used with caution after at least two weeks postpartum.
Essential.
Pregnancy
We suggest non-pharmacologic treatments or peripheral nerve blocks (see Q12) as first line options in pregnant or breastfeeding women. If non-pharmacological treatment is not possible or effective, we suggest propranolol or amitriptyline, after balancing risks and benefits and informing the person with migraine on potential secondary effects and associated risks. Propranolol should be discontinued in the last part of the third trimester to avoid the risk of adverse events to the fetus and neonate.
We strongly recommend avoiding valproate and topiramate during pregnancy due to their teratogenic effects.
Breastfeeding
Non pharmacologic treatment or peripheral nerve blocks should be considered first line. If non-pharmacological treatment is not possible or effective, we suggest propranolol or amitriptyline, after informing the subjects of the potential secondary effects and associated risks.
Background
Up to 90% of females with migraine experience improvement in migraine during pregnancy, with a significant reduction in the frequency and intensity of their attacks during the second and third trimester of pregnancy (17,134–136). Women who have migraines with aura are at greater risk for having more frequent attacks during pregnancy (137).
Most of the recommended drugs for migraine prevention are FDA pregnancy category C, D or even X as there are no specific clinical trials evaluating drug treatment of migraine during pregnancy and breastfeeding.
Evidence
Based on existing data, although evidence for non-pharmacologic treatment in pregnancy is sparse, this should be the first line considering the potential risk to the fetus of pharmacologic therapy. For this reason, although these recommendations focus specifically on pharmacologic treatment, we have included information on ‘Non-pharmacologic’ therapies below.
Non-pharmacologic therapies
Awareness and avoidance of triggering factors has been considered helpful, even though there is no definitive evidence in migraine treatment (138). Physical fitness has been found to reduce stress and provide a healthy balance between strain and relaxation (139–142).
Behavioral interventions such as relaxation training, thermal biofeedback, electromyographic biofeedback, cognitive behavioral therapy, mindfulness-based therapy and acceptance and commitment therapy may be used alone or in combination with drug therapy to achieve improvement in migraine symptoms (143–147).
Non-invasive neuromodulation including supraorbital electrical nerve stimulation, vagal nerve stimulation and transcranial magnetic stimulation are effective for migraine prevention (148–153). The safety of their use during pregnancy has not yet been established, but animal studies and limited open-label studies have shown no reproducible adverse effects on fetal development (154).
Riboflavin is a vitamin recommended for migraine prevention in persons averse to taking traditional medications (155). Certain vitamins, minerals and herbal preparations have been proposed for preventive therapy in pregnancy and lactation, but they lack the evidence to support their recommendation (145).
Pharmacological therapy
Although beta-blockers have been considered relatively safe in pregnancy, they may be associated with intrauterine growth restriction, hence caution is recommended (156–158). Use of beta-blockers in the third trimester may cause neonatal pharmacologic effects such as bradycardia, hypotension and hypoglycemia, so the drugs should be tapered or discontinued two to three days before delivery (159,160) to decrease the risk of adverse events to the fetus/neonate and potential reductions in uterine contractions (161). Tricyclic antidepressants are considered the safest second-line option when beta-blockers are contraindicated or ineffective. Amitriptyline is preferred. Some studies suggest a possible teratogenic effect of tricyclic antidepressants (e.g., cardiovascular or limb abnormalities), but a clear causal relationship cannot be proven (135,160,162).
CGRP mAbs are not recommended for use in pregnancy due to lack of studies (19). Other preventive drugs contraindicated include flunarizine, topiramate, sodium valproate and zonisamide (141,160,163,164).
During lactation, valproate and topiramate are classified under Hale Lactation Risk Categories L2 (safer) and L3 (moderately safe), respectively (165,166). According to the Drug and Lactation database (167), propranolol and onabotulinumtoxinA reach low levels in the milk and are considered safe. Amitriptyline may occasionally induce sedation in the infant. Candesartan levels are low in the milk, but caution is advised. No evidence is available on lisinopril, therefore an alternate drug is preferable.
No information is available on the use of CGRP mAbs during breastfeeding. As the four monoclonal antibodies are large protein molecules with a molecular weight of about 143,000 Daltons, the amount in milk is likely to be very low and is probably partially destroyed in the infant's gastrointestinal tract, with low or minimal absorption by the infant. Waiting for at least two weeks postpartum to resume therapy may minimize transfer to the infant. No information is available so far on the clinical use of gepants during breastfeeding.
Peripheral nerve blocks are considered safe in pregnancy and breastfeeding as systemic effects are lower than with use of oral medications. The preferred agent to inject is lidocaine. Bupivacaine or betamethasone may be used as alternatives. Bupivacaine may be associated with fetal cardiotoxicity (168).
Online Supplemental Table 14 reports the statements regarding migraine preventive therapy in pregnancy and lactation in the guidelines reviewed and the guidance documents assessed.
Q15 – Which migraine preventive drugs can be used in people with migraine over 65 years of age?
Recommendations
Optimal.
We suggest selecting the drug for migraine prevention in people with migraine over 65 years after considering possible comorbidities and the needs of dose adjustments, for all treatments.
We suggest careful clinical monitoring to allow for early detection of adverse effects, as well as the potential need to modify the course of treatment.
Some drugs targeting the CGRP pathway have been tested in populations up to 80 years old without safety issues and can therefore represent an option.
For people with chronic migraine, onabotulinumtoxinA may represent an option given the limited, if any, systemic effect.
Essential.
We suggest selecting the drug for migraine prevention in people with migraine over 65 years after considering possible comorbidities and the needs of dose adjustments, for all treatments.
We suggest careful clinical monitoring to allow for early detection of adverse effects, as well as the potential need to modify the course of treatment.
Background
The rapid growth of the aging population gives rise to the issue of appropriate drug selection for this population. Older adults, aged 65 and older, despite the decreased incidence of headache with age (169), may still require migraine prevention. The selection of an appropriate migraine preventive medication for elderly individuals is complex. These people are more likely to experience other pain syndromes in general, as well as polypharmacy, multiple comorbid medical conditions, and intolerance to pain medication (170,171). Elderly individuals are also typically underrepresented in clinical trials, leading to a significant lack of evidence on drug use (171). These factors account for the complex prescription process for migraine preventive treatment in this population.
Evidence
No guidelines or meta-analyses have been published that offer high-quality data or recommendations regarding migraine prevention in older adults. Only the Danish Guidelines (15) mention ‘Elderly patients should only receive half the dose of flunarizine’ in passing.
Trials usually exclude individuals older than 65 years old, although recently some trials have started to expand their evaluation to people up to 80 years old. For these reasons, the available information is based on the existing guidelines.
Overall, standard treatment options may be considered for older adults. It is important to use these medications with caution because the elderly have a higher risk of side effects (24,172). When selecting a preventive medication for migraine, clinicians should take into account any concurrent comorbidity (e.g., cardiovascular disease, asthma, diabetes mellitus) (173). Drugs contraindicated by any comorbid conditions (e.g., β-adrenoceptor antagonists in subjects with asthma) and drugs that could worsen migraine (e.g., nifedipine in individuals with hypertension) should be avoided whenever possible. Particular attention should be also given to drug-drug or drug-food interactions (174).
The use of tricyclic antidepressants in older individuals may be associated with higher plasma concentrations and metabolites compared to younger people, leading to an increased incidence of adverse events (175). Amitriptyline can cause cardiac conduction abnormalities, orthostatic hypotension, seizures, cognitive impairment and confusion, thus strongly limiting its indication in older adults (176). Further, tricyclic antidepressants are effective, especially in individuals with comorbid insomnia, mood and anxiety disorders but contraindicated in people with glaucoma or prostatic hypertrophy. Additionally, tricyclic antidepressants should be avoided in people with recent myocardial infarction or with prolonged QTc intervals, since a mean prolongation of 10–20 msec has been reported in treated elderly individuals (171). Finally, they are contraindicated in subjects with heart failure or co-administration with monoaminoxidase inhibitors. We suggest the administration of small doses between 10 and 30 mg with slow titration (177).
Among the antidepressants, excluding venlafaxine, the use of selective serotonin reuptake inhibitors (SSRIs) and selective norepinephrine reuptake inhibitors (SNRIs) for the preventive treatment of migraine is not supported by evidence (178). In older individuals, studies have shown that there is a strong association between developing hyponatremia after treatment with SSRIs and venlafaxine. Therefore, monitoring of sodium levels is recommended (179). Overall, venlafaxine has moderate evidence for migraine prevention with fewer side effects than amitriptyline, which makes it a favorable option to consider in older adults (180), with a recommended dose of 75–150 mg/daily (181).
The use of beta-blockers, a first-line drug for migraine prevention, needs to be restricted in the elderly population since these drugs may worsen congestive heart failure, promote conduction disorders or exacerbate asthma, diabetes, glaucoma or depressive symptoms (177). Beta-blockers might be a drug of choice for migraine prevention in elderly adults if they have concomitant hypertension or coronary artery disease (174,181). Lisinopril and candesartan are drugs with low levels of recommendation for preventing migraines but have not been shown to increase side effects in subjects over 65 (182).
The unfavorable adverse effects profile usually limits the use of antiepileptics in the elderly (171). Among antiepileptic drugs, topiramate has strong evidence for migraine prevention and can be used in the elderly. It is, however, contraindicated in individual suffering from nephrolithiasis and glaucoma (174,181). Topiramate use is also linked with paresthesia, taste disturbances, reduction of cognitive performance – particularly presenting word-finding difficulty – and weight loss (183). Valproate is contraindicated in persons with liver disease and thrombocytopenia. Moreover, its use in older adults should be cautious because this drug can cause liver dysfunction, hyperammonemia, decreased bone marrow density, delirium, tremor and ataxia (177,184).
The use of calcium channel blockers in the elderly should only be considered after a careful evaluation of the associated risks (174). Flunarizine should be avoided in individuals with hypotension, heart failure, atrioventricular block, Parkinson's disease, familial risk of Parkinson's disease or depression (185). For these reasons, the Danish guidelines suggest that older adults should only receive half the dose of flunarizine (15).
OnabotulinumtoxinA is a relatively safe and useful option in people with drug-resistant chronic migraine as it has few side effects and has already been largely used in other neurological conditions such as spasticity in older adults (177,186,187).
CGRP-targeting agents can be administered in older adults, but their use is not yet recommended by guidelines (170,172). Although most of the data from clinical trials involving CGRP mAbs are derived from individuals with migraine younger than 65 years, emerging data support the possibility of safely using them in the elderly. The latest data from clinical trials now available include adults as old as 70/75 years of age, suggesting an acceptable safety profile. This is the case of fremanezumab (188) that was assessed in individuals up to 70 years old and eptinezumab that was assessed in individuals up to age 75 (33,189). The age of a person does not affect the efficacy of galcanezumab in preventing migraine attacks, and there is no clinically meaningful influence of age on its pharmacokinetics (190). Galcanezumab was tested up to age 75 and pooled data from available trials suggest that there is no need to adjust the dose for older people (191). Erenumab has not been tested in persons with migraine older than 65 years, but it has a similar safety profile to placebo across younger age groups in individuals with episodic or chronic migraine and it was well tolerated in older participants with multiple comorbidities, polypharmacy, and age-related physiological changes (192). While these studies provide preliminary evidence that using CGRP mAbs is safe in individuals beyond 65 years, more research is needed in larger populations of older people.
Emerging data support the possibility to use gepants as well in older adults, but most clinical trials included few participants older than 65 years (193), although recent trials on atogepant included participants up to 80 years of age (103,194,195). Rimegepant appeared effective in trials also involving elderly participants (196,197) and it was reported as safe and well tolerated in adults older than 65 years old, following a single oral 75 mg dose (198). Overall, despite lacking specific trials addressing the elderly population, gepants are likely to be well tolerated by the older population, although their hepatic metabolism warrants a careful evaluation during polypharmacy (170).
A group of experts has recently proposed the use of candesartan or beta-blockers without partial agonist activity (e.g., bisoprolol, propranolol, metoprolol, nadolol, or atenolol) as first-line treatments for migraine prevention, while antidepressants (e.g., amitriptyline and venlafaxine) and anticonvulsants (e.g., sodium valproate and topiramate) are suggested as second-line preventive medications, requiring particular caution in older adults (199). The third line is represented by onabotulinumtoxinA, the CGRP mAbs and CGRP receptor antagonists.
Q16 – What is the recommended approach to people with migraine and medication overuse?
Recommendations
Optimal.
In people with migraine and medication overuse the following approaches are recommended: - reduced intake of overused drug(s) simultaneous with the initiation of preventive treatment; - reduced intake of overused drug(s) followed by initiation of preventive treatment; - interruption of overused drug(s) followed by initiation of preventive treatment.
The selection of the preventive treatment must be based on evidence of therapeutic efficacy, personal history and comorbidities.
Monoclonal antibodies targeting the CGRP pathway, topiramate and onabotulinumtoxinA have proven effective regardless of the presence of medication overuse, therefore the immediate withdrawal or reduction of the overused drug might not be necessary in subjects who are initiating such a treatment.
Individuals overusing opioids or barbiturate containing drugs may require hospitalization to manage drug discontinuation safely and successfully.
Essential.
In people with migraine and medication overuse the following approaches are recommended: - reduced intake of overused drug(s) simultaneous with the initiation of preventive treatment; - reduced intake of overused drug(s) followed by initiation of preventive treatment; - abrupt interruption of overused drug(s) followed by initiation of preventive treatment. The selection of the preventive treatment must be based on evidence of therapeutic efficacy, personal history and comorbidities.
Topiramate has proven effective regardless of the presence of medication overuse, therefore the immediate withdrawal or reduction of the overused drug might not be necessary in subjects who are initiating such a treatment.
Individuals overusing opioids or barbiturate containing drugs may require careful monitoring to manage drug discontinuation safely and successfully.
Background
Medication overuse headache (MOH) is defined as headache occurring on 15 or more days/month in an individual with a pre-existing primary headache disorder, which develops due to regular overuse of acute or symptomatic headache medication. Medication overuse is defined as regular intake for more than three months of: > 15 days/month of paracetamol or NSAIDs; > 10 days/month of triptans, ergotamine, opioids or combination-analgesics (200).
MOH represents a significant issue within headache management, as it is associated with substantial disability and reductions in quality of life, and very often goes underrecognized in clinical practice. It presents in more than half the people who have headache on >15 days/month, and is estimated to affect around 59 million people worldwide (201,202).
Evidence
MOH is most commonly seen in people with a pre-existing diagnosis of either migraine or tension-type headache (203) and results from an interaction between frequently used acute headache medication and individual susceptibility. Certain risk factors are associated with developing MOH, such as female gender, low socio-economic status, presence of a concomitant painful condition and psychiatric comorbidities (204).
For the management of MOH, a patient-centered approach and education on the condition are crucial (205). Evidence from RCTs has in fact shown that advice alone can determine similar treatment outcomes to pharmacological interventions in individuals with uncomplicated MOH (206).
In general, the majority of people with MOH improve on withdrawal of the overused medication, and conversely MOH is less likely to resolve unless the overused medication is stopped (207). Withdrawal itself has shown to be more effective when the drug is eliminated completely rather than restricted (208), particularly when associated with early start of preventive medication (209). For prevention, specific drugs that have shown efficacy in MOH include topiramate (37), valproic acid (210) and onabotulinumtoxinA (211). As regards this latter, one study showed that that the improvement is related to the withdrawal from overused drugs rather than to the administration of the drug (212).
Although drug withdrawal can certainly be effective, the timing and even absolute necessity of it is still debated. One study used a ‘pragmatic approach’ to MOH and showed that, as long as appropriate preventive medication was started, switching or limiting symptomatic medication was just as effective as not switching at all (213). Recent subgroup analyses of studies investigating CGRP mAbs have shown promising results in MOH even without strict discontinuation (214–216), indicating that novel treatments might change the approach to the condition in the future. These studies did not, however, include individuals with opioid overuse. Overall, the data suggests that even the use of acute analgesics two to three days a week can potentially have an impact on efficacy of concomitant preventive treatment (217).
Online Supplemental Table 15 reports the statements on the approach to medication overuse in the guidelines reviewed and the guidance documents assessed.
Supplemental Material
sj-docx-1-cep-10.1177_03331024241269735 - Supplemental material for International Headache Society Global Practice Recommendations for Preventive Pharmacological Treatment of Migraine
Supplemental material, sj-docx-1-cep-10.1177_03331024241269735 for International Headache Society Global Practice Recommendations for Preventive Pharmacological Treatment of Migraine by Francesca Puledda, Simona Sacco, Hans-Christoph Diener, Messoud Ashina, Haidar M. Al-Khazali, Sait Ashina, Rami Burstein, Eric Liebler, Andrea Cipriani, Min Kyung Chu, Alexandra Cocores, Freda Dodd-Glover, Esme Ekizoğlu, David Garcia-Azorin, Carl H. Göbel and Maria Teresa Goicochea, Amr Hassan, Koichi Hirata, Jan Hoffmann, Bronwyn Jenkins, Katharina Kamm, Mi Ji Lee, Yu-Hsiang Ling, Marco Lisicki, Daniele Martinelli, Teshamae S. Monteith, Raffaele Ornello, Aynur Özge, Mario Fernando Prieto Peres, Patricia Pozo-Rosich, Volodymyr Romanenko, Todd J. Schwedt, Marcio Nattan P Souza, Tsubasa Takizawa, Gisela M. Terwindt, Janu Thuraiaiyah, Mansoureh Togha, Nicolas Vandenbussche, Shuu-Jiun Wang, Shenguan Yu, Cristina Tassorelli in Cephalalgia
Supplemental Material
sj-docx-2-cep-10.1177_03331024241269735 - Supplemental material for International Headache Society Global Practice Recommendations for Preventive Pharmacological Treatment of Migraine
Supplemental material, sj-docx-2-cep-10.1177_03331024241269735 for International Headache Society Global Practice Recommendations for Preventive Pharmacological Treatment of Migraine by Francesca Puledda, Simona Sacco, Hans-Christoph Diener, Messoud Ashina, Haidar M. Al-Khazali, Sait Ashina, Rami Burstein, Eric Liebler, Andrea Cipriani, Min Kyung Chu, Alexandra Cocores, Freda Dodd-Glover, Esme Ekizoğlu, David Garcia-Azorin, Carl H. Göbel and Maria Teresa Goicochea, Amr Hassan, Koichi Hirata, Jan Hoffmann, Bronwyn Jenkins, Katharina Kamm, Mi Ji Lee, Yu-Hsiang Ling, Marco Lisicki, Daniele Martinelli, Teshamae S. Monteith, Raffaele Ornello, Aynur Özge, Mario Fernando Prieto Peres, Patricia Pozo-Rosich, Volodymyr Romanenko, Todd J. Schwedt, Marcio Nattan P Souza, Tsubasa Takizawa, Gisela M. Terwindt, Janu Thuraiaiyah, Mansoureh Togha, Nicolas Vandenbussche, Shuu-Jiun Wang, Shenguan Yu, Cristina Tassorelli in Cephalalgia
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:
Francesca Puledda declares speaker and consultancy fees from Abbvie and Teva. Simona Sacco declares grants/contracts, consulting fees or honoraria from Novartis, Uriach, Allergan-AbbVie, Teva, Lilly, Lundbeck, Pfizer, NovoNordisk, Abbott, AstraZeneca, Pfizer. Hans-Christoph Diener received honoraria for participation in clinical trials, contribution to advisory boards or oral presentations from: AbbVie, Lilly, Lundbeck, Novartis, Pfizer, Teva and WebMD. The German Research Council (DFG) supports headache research by HCD. HCD serves on the editorial boards of Cephalalgia, Lancet Neurology and Drugs. Messoud Ashina is a consultant, speaker, or scientific advisor for AbbVie, Amgen, Astra Zeneca, Eli Lilly, GlaxoSmithKline, Lundbeck, Novartis, Pfizer, and Teva; a primary investigator for ongoing AbbVie and Pfizer trials; and is the past president of the International Headache Society. MA is supported through the Lundbeck Foundation Professor Grant (R310-2018-3711) and received institutional research grant from Lundbeck and Novartis. MA serves as associate editor of Journal of Headache Pain, and associate editor of Brain. Sait Ashina has received honoraria for consulting from Allergan/AbbVie, Eli Lilly, Impel NeuroPharma, Linpharma, Lundbeck, Pfizer, Satsuma, Teva, Theranica. SA is an associate editor for Neurology Reviews, BMC Neurology, and Frontiers in Neurology, and serves on the editorial board for Journal of Headache Pain. SA serves as a Trustee on the Board of the International Headache Society and is a member of the Education Committee of the International Headache Society. Eric Liebler declares consulting fees from ElecroCore, Inc. Andrea Cipriani is supported by the National Institute for Health Research (NIHR) Oxford Cognitive Health Clinical Research Facility, by an NIHR Research Professorship (grant RP-2017-08-ST2-006), by the NIHR Oxford and Thames Valley Applied Research Collaboration and by the NIHR Oxford Health Biomedical Research Centre (grant NIHR203316). The views expressed are those of the authors and not necessarily those of the UK National Health Service, the NIHR, or the UK Department of Health. Min Kyung Chu was a site investigator for a multicenter trial sponsored by Biohaven Pharmaceuticals, Allergan Korea, and Ildong Pharmaceutical Company. He received lecture honoraria from Eli Lilly and Company, Handok-Teva, and Ildong Pharmaceutical Company over the past 24 months. He received grants from Yonsei University College of Medicine (6-2021-0229), the Korea Health Industry Development Institute (KHIDI) (Grant No.: HV22C0106), and a National Research Foundation of Korea (NRF) grant. Alexandra Cocores declares grants/contracts from AbbVie, Eli Lilly, Rehaler. Esme Ekizoğlu declares honoraria from Allergan-AbbVie. David Garcia-Azorin declares grants/contracts from Carlos II Research and Castilla and Leon Regional Health, consulting fees from Eli-Lilly, WHO, Lundbeck or honoraria from AbbVie, Teva, Novartis, Pfizer. Carl Göbel declares honoraria from Fielmann AG and German Society for the Study of Pain. Maria Teresa Goicochea, personal fees as speaker or advisory board and/or non-financial support from Pfizer, AbbVie and Teva. Amr Hassan declares consulting fees or honoraria from Novartis, Sanofi Genzyme, Biologix, Merck, Hikma Pharma, Janssen, Inspire Pharma, Future Pharma, Elixir pharma, Allergan, Merck, Biologix, Roche, Bayer, Chemipharm, Al Andalus, and Clavita Pharm. Koichi Hirata declares consulting fees from Otsuka Pharma, and honoraria from Amgen Astellas BioPharma KK, Daiichi Sankyo, Eisai, Eli Lilly Japan KK, MSD, Otsuka Pharma, Pfizer Japan, and Sawai Pharma. Jan Hoffmann declares grants/contracts from Bristol Myers Squibb, International Headache Society (IHS), National Institute for Health and Care Research (NIHR), Medical Research Council (MRC) and Migraine Trust, consulting fees or honoraria from AbbVie, Cannovex, Eli Lilly, Novartis, Teva, Allergan, Chordate Medical AB, Lundbeck, Sanofi, MD Horizonte, Pfizer, Sage, NEJM Journal Watch Neurology, Oxford University Press, Quintessence Publishing, Springer Healthcare. He holds stock options from Chordate Medical AB. Bronwyn Jenkins declares honoraria from Allergan/AbbVie, Eli-Lilly, Novartis, Pfizer, Teva, Viatris. Katharina Kamm declares grants/contracts from the German Headache and Migraine Society, Novartis, consulting fees from Hormosan, and honoraria from Lundbeck, TEVA, derCampus, Novartis. Mi Ji Lee declares consulting fees or honoraria from Eli-Lilly, Lundbeck, Pfizer, NuEyne Co, Teva AbbVie, SK Chemical, CKD Pharma, and YuYu. Marco Lisicki declares honoraria from Teva, AbbVie, Novartis, Pfizer and Allergan. Daniele Martinelli declares honoraria from AbbVie and Lundbeck. Teshamae S. Monteith has the following disclosures over the past three years: clinical trial site principal investigator for studies sponsored by Abbvie, Eli Lilly, Ipsen and Rehaler, participation in advisory board/consultancy for AbbVie, Teva, Linpharma, e-Neura, Novartis, Merz, and Pfizer, Educational grant from Amgen and AbbVie, personal fees from Medscape, Massachusetts Medical Society, American Headache Society, American Academy of Neurology, Neurodiem, Academic CME, AbbVie, Novartis, unpaid co-author for research funded by AbbVie, Pfizer/Biohaven, and Theranica. She is an associate editor for Cephalalgia and Continuum Audio, and is on the editorial board for Neurology, American Migraine Foundation, and Brain and Life Magazine. Dr Monteith has provided unpaid service on the board of directors for the International Headache Society (2021-2023) and currently on the executive board for the Florida Society of Neurology. Raffaele Ornello declares grants/contracts from Novartis, consulting fees/honoraria from Eli Lilly, AbbVie, Pfizer, and Teva. Aynur Özge declares consulting fees or honoraria from AbbVie Lilly, Teva, Abdi Ibrahim, İlko and Drogsan. Mario Peres accepted honoraria for lectures, presentations, speakers’ bureaus, manuscript writing and educational events from Pfizer, Teva, AbbVie-Allergan, Lundbeck. Accepted travel support from Teva. Participated in advisory boards, AbbVie-Allergan, Eli Lilly, Pfizer, Kenvue, Eurofarma, Sanofi-Aventis. MP is president of ABRACES. Patricia Pozo-Rosich declares grants/contracts from ERANet Neuron, Instituto Salud Carlos III, Novartis, Teva and AbbVie and consulting fees or honoraria from AbbVie, Almirall, Eli Lilly, Lundbeck Medscape, Novartis, Pfizer, Teva. Todd J. Schwedt declares grants/contracts with American Heart Association, Amgen, Henry Jackson Foundation, National Headache Foundation, National Institutes of Health, Patient Centered Outcomes Research Institute, Pfizer, Spark Neuro, United States Department f Defense, American Migraine Foundation and Mayo Clinic. Consulting fees from AbbVie, Allergan, Amgen, Axsome, Biodelivery Science, Biohaven, Click Therapeutics, Collegium, Eli Lilly, Equinox, Ipsen, Unpharma, Lundbeck, Novartis, Satsuma, Scilex, Theranica, and Tonix. He declares royalties from UpToDate and stock options from Aural. Marcio Nattan P. Souza declares consulting fees or honoraria from Teva, Libbs, Pfizer, Sandoz, Lundbeck, Allergan/AbbVie, Lilly. Tsubasa Takizawa declares grants/research funds from Eli Lilly, Pfizer and Tsumura and advisory/consulting fees and/or honoraria from Eli Lilly, Daiichi Sankyo, Otsuka, Amgen, Teijin, Pfizer, Kyowa Kirin, Elsai, Kowa, UCB Japan, Takeda, and Santen Pharma. Gisela M. Terwindt declares grants/contracts from European Community, Dutch Heart Foundation, IRRF, Dioraphte and Dutch Brain Council with consulting fees or honoraria from Novartis, Lilly, Teva, AbbVie/Allergan, Lundbeck, Pfizer, Spring Media, Ashfield MedComms, Remedica Cygnea. Nicolas Vandenbussche declares consulting fees or honoraria Pfizer, AbbVie, Novartis, Teva, Babinski VZW, ACREHAB UGent. Shuu-Jiun Wang declares honoraria from AbbVie, Pfizer and Biogen. Cristina Tassorelli declares grants/contracts from AbbVie with consulting fees or honoraria from AbbVie, Eli Lilly, Dompé, Ipsen, Lundbeck, Pfizer, Medscape, Teva. All remaining authors report no disclosures or conflicts of interest
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
