Abstract
Background
In an effort to improve migraine management around the world, the International Headache Society (IHS) has here developed a list of practical recommendations for the acute pharmacological treatment of migraine. The recommendations are categorized into optimal and essential, in order to provide treatment options for all possible settings, including those with limited access to migraine medications.
Methods
An IHS steering committee developed a list of clinical questions based on practical issues in the management of migraine. A selected group of international senior and junior headache experts developed the recommendations, following expert consensus and the review of available national and international headache guidelines and guidance documents. Following the initial search, a bibliography of twenty-one national and international guidelines was created and reviewed by the working group.
Results
A total of seventeen questions addressing different aspects of acute migraine treatment have been outlined. For each of them we provide an optimal recommendation, to be used whenever possible, and an essential recommendation to be used when the optimal level cannot be attained.
Conclusion
Adoption of these international recommendations will improve the quality of acute migraine treatment around the world, even where pharmacological options remain limited.
Introduction
Consistent with the mission of the International Headache Society (IHS) to improve migraine management worldwide, this document focuses on providing practical recommendations on the pharmacological management of migraine. Due to the inconsistent availability of medications across different regions of the globe, these recommendations are categorized into two levels:
In the first part of this IHS endeavor, we present the recommendations for the acute pharmacological treatment of migraine attacks, together with the methodology and the evidence used to support them. The practice recommendations for the pharmacological preventive treatment of migraine are presented in a companion paper. Table 1 lists the drugs with evidence of efficacy for the acute treatment of migraine listed in the WHO EML.
Drugs recommended for the acute treatment of migraine by regional and international guidelines, and their availability in the WHO Model List of Essential Medicines, 23rd List (2023) (1).

The IHS practice recommendations are based on available treatment guidelines and expert consensus. They are intended to be a practical, quick reference, applicable in all countries across different care settings, including primary care. Given the global scope of these recommendations we have not customized the recommendations based on national registrations or specific labelling in individual countries. Nothing in these guidelines is designed to supersede local labelling and approvals.
These recommendations represent an instrument to motivate and facilitate policy changes. Our goal is to establish essential standards of migraine management in as many countries as possible. These standards will also serve as a reference document to drive local advances toward optimal care once essential standards of care are met.
Methodology used for the development of questions and recommendations
The working group panel of the present practice recommendations was nominated by the IHS board. Members were selected based on their specific expertise in different areas of headache, previous experience developing guidelines or recommendations, and representation of different regions of the world. The group was gender balanced and professional backgrounds included neurology, methodological expertise, evidence synthesis and statistics. Each senior member worked in collaboration with two junior headache experts from a different geographic origin for the analysis of the literature and the explanation of the recommendations. A. Cipriani was specifically involved for his expertise in the methodology of evidence-based synthesis.
We used a consensus development panel approach, adapting the methodology described and used by the US National Institutes of Health and WHO (2). This method of consensus formulation was chosen as it allows the identification of questions, development of recommendations, and formulation of strategic plans. An initial set of clinical questions was elaborated in the spring of 2022 by the Steering Committee (HC-D, MA, CT) based on the main issues that healthcare professionals may encounter when treating a person suffering with migraine. This initial list was shared with the coordinators (SS and FP) and the entire working group (seniors and juniors) for interactive discussion and optimization. Following subsequent iterations, the final set of clinical questions was agreed in the fall of 2022.
A. Cipriani the search of the published literature to identify the National and International Guidelines and other guidance documents for migraine treatment to be used for elaborating the recommendations. The search terms for each clinical question are reported in Online Supplementary file 1. FP and SS assessed the search output and selected a total of 16 national/international guidelines and other guidance documents for elaborating the recommendations, based on: i) relevance of the paper; ii) publication date of less than 15 years prior; iii) availability in the English language. A further five guidelines (from German, Korean, Japanese, Taiwanese and Hungarian societies) were subsequently added following either an English translation being made available or internal suggestions coming directly from the working group. For reference please see Online Supplementary file 2. In the kick–off meeting held virtually on February 2023, each triad of experts, formed by a senior and two juniors, was given the task to elaborate a first draft of recommendations for 2–3 clinical questions. Once all the triads had elaborated the assigned recommendations, these were shared with the entire working group for discussion and refinement. Several runs of discussion via virtual meetings or e-mail exchanges led to the final version agreed by all the components in December 2023.
The final list of clinical questions and the corresponding recommendations are summarized in Table 2.
Summary table illustrating the 17 clinical questions and the corresponding Optimal and Essential level recommendations.

In the next sections we will illustrate in detail each clinical question, associated recommendations for the Optimal and Essential level, background for the question and evidence used for the elaboration.
Q1 – Should triptans be used when analgesics and non-steroidal anti-inflammatory drugs are ineffective?
Recommendations
Optimal
In people with migraine not responding* to analgesics or non-steroidal anti-inflammatory drugs taken at appropriate doses and early during the attack, we suggest switching to a triptan for the next attack.
Essential
In people with migraine not responding* to analgesics or non-steroidal anti-inflammatory drugs taken at appropriate doses and early during the attack, we suggest switching to any available triptan for the next attack.
*The individual is not pain-free two hours after the intake of the drug.
Comment: In people with migraine with severe attacks, triptans efficacy may be superior to non-steroidal anti-inflammatory drugs and therefore triptans can be used as the first line treatment.
Background
Pain control is important for people with migraine to reduce the burden and disability of migraine attacks. Multiple drug classes and individual drugs are available for the acute treatment of migraine attacks. These include: simple analgesics (e.g., paracetamol or acetaminophen); non-steroidal anti-inflammatory drugs (NSAIDs) such as ibuprofen, acetylsalicylic acid, diclofenac or naproxen; and migraine-specific drugs such as triptans. Triptans are a class that includes seven different molecules (almotriptan, eletriptan, frovatriptan, naratriptan, rizatriptan, sumatriptan and zolmitriptan) and act as 5-HT1B and 5-HT1D receptor agonists. NSAIDs and paracetamol are less expensive and generally more widely available than triptans. Ergots are migraine-specific medications, but no longer represent an option due to side-effects, risk of overuse and availability of better options. They can only be used in exceptional cases when all available acute treatments are not effective or contraindicated and ergots themselves are not contraindicated (see Q7 below).
People with migraine who do not respond to non-specific treatments for migraine such as NSAIDs should be assessed for specific treatments such as triptans. The efficacy of triptans for the treatment of migraine attacks has been shown in multiple randomized controlled trials (RCTs), systematic reviews and meta-analyses (3). Triptans are not effective to treat migraine aura (4,5) and have the potential to induce medication-overuse headache (MOH). The risk for MOH increases with the daily frequency of triptan use. In these recommendations we set the limit of triptan use to two days per week based on data indicating that use of triptans 10 days per month is considered overuse (6–8).
Acute treatment strategies include step or stratified care. Step care escalates treatment across or within attacks according to the treatment response, safety and costs. In stratified care, treatment selection is based on the assessment of disorder severity. Stratified care may lead to more effective acute treatment and is more cost-effective due to a decrease in physician office visits, emergency department visits and medical procedures (9,10).
Evidence on the comparative efficacy of triptans versus NSAIDs
For evidence on the comparative efficacy of triptans versus NSAIDs see Table 3.
Summary of studies investigating triptans versus analgesics and/or non-steroidal anti-inflammatory drugs for the treatment of migraine attacks with outcome of pain freedom at two hours, by triptan.

RCT: randomized controlled trial.
Eletriptan
In one observational study, poor responders to the combination of paracetamol/acetylsalicycic acid/caffeine responded well to eletriptan 40 mg. Forty-one percent of participants were pain-free at two hours with eletriptan (11).
Rizatriptan
In one RCT two-hour headache freedom was achieved by rizatriptan 10 mg in 20 (38%), ibuprofen in 16 (31%) and placebo in one (2%) participants (no statistical tests performed) (12).
Sumatriptan
In a trial comparing oral sumatriptan 100 mg, a combination of 1620 mg lysine acetylsalicylate (equivalent to 900 mg of aspirin) and 10 mg metoclopramide and placebo, the treatment strategies were equally effective in leading to resolution of migraine attacks within two hours, and were significantly more effective than placebo. No significant difference was observed between active treatments (13).
Another trial with sumatriptan 100 mg versus aspirin 900 mg plus metoclopramide 10 mg found a higher percentage of sumatriptan users were pain-free at two hours after treatment compared to the aspirin-metoclopramide combination (14).
Acetylsalicylic acid 1000 mg effervescent was tested against sumatriptan 50 mg and placebo. The efficacy of both drugs was comparable: 25% of people with migraine on acetylsalicylic acid and 24% of participants on sumatriptan 50 mg became pain-free (non-significant) (15).
In a placebo-controlled trial comparing acetylsalicylic acid 1000 mg effervescent, sumatriptan 50 mg and ibuprofen 400 mg, 27% of participants on acetylsalicylic acid 1000 mg effervescent, 33% of participants on ibuprofen 400 mg and 37% of participants on sumatriptan 50 mg became pain-free at two hours versus 13% for placebo (non-significant between active treatments, significant versus placebo) (16).
In a study comparing naproxen 500 mg with sumatriptan 50 mg, 45 of 248 participants (18%) using naproxen 500 mg were pain-free at two hours compared to 45 of 226 participants (20%) with sumatriptan 50 mg (17). Another study found that a numerically higher proportion of participants using sumatriptan 85 mg become pain-free at two hours compared to naproxen sodium 500 mg (study 1: 90 out of 361 participants (25%) on sumatriptan 50 mg vs 53 of 356 participants (15%) on naproxen; study 2: 82 of 362 participants (23%) on sumatriptan vs 57 of 364 participants (16%) on naproxen) (18).
Zolmitriptan
In a double-blind RCT comparing the efficacy of ketoprofen 75 mg or 150 mg with placebo or zolmitriptan 2.5 mg, freedom from headache at two hours was more frequent with the three active treatments than with placebo, with a significant difference between zolmitriptan and ketoprofen 75 mg, but not between zolmitriptan and ketoprofen 150 mg (19).
In a randomized controlled trial comparing zolmitriptan 2.5 mg versus acetylsalicylic acid plus metoclopramide, 11% of participants in the zolmitriptan group became pain-free after two hours versus 5% of participants in the acetylsalicylic acid plus metoclopramide group (odds ratio 2.19, p-value <0.01) (20).
Other NSAIDs
We found no comparative data on the efficacy of diclofenac at two hours versus any triptan (21).
Based on the evidence, eletriptan, rizatriptan, sumatriptan and zolmitriptan are at least equally or more effective than simple analgesics and NSAIDs. There is no evidence that people with migraine who do not respond to simple analgesics or NSAIDs have a low probability of responding to triptans. Therefore, if triptans are available and no contraindications exist, triptans should be recommended for people with migraine not responding to simple analgesics or NSAIDs.
The relevant statements on triptan use when analgesics and NSAIDs drugs are ineffective in the guidelines reviewed and the guidance documents assessed are illustrated in Online Supplementary Table 1S.
Q2 – If a triptan is only partially effective, should the dose be increased?
Recommendations
Optimal
If a triptan taken early after migraine attack onset is only partially effective*, we suggest increasing the dose to the maximum recommended dose for that triptan for the next attack. If the response is still inadequate, we suggest switching to a different route of administration (see Q3) or to a different triptan for the next attack. If three triptans have been tried at the appropriate dose without a satisfactory response, we suggest switching to a different class of acute drugs (see Q5 and Q6).
Essential
If only sumatriptan 50 mg oral tablets (in the WHO EML) are available and they are only partially effective, we suggest increasing the dose to two tablets (100 mg) for the next attack.
If other triptans are available, follow above recommendations for Optimal level.
*Partial efficacy: the subject has not achieved pain relief two hours after the intake of the drug.
Comment: Two different triptans should not be taken in the same 24-h period. Similarly, a triptan and ergot should not be administered within 24 hours.
Background
Some triptans have been investigated and approved with multiple doses. A dose-response effect could justify the use of a higher triptan dose if the lower doses fail to show a response. However, adverse events (AE) could also present in a dose-dependent manner. Response to triptans can be complete, partial, or absent according to different definitions. Therefore, the advantages of triptan dose escalation when lower doses fail to provide an effect are a matter of debate.
Evidence
For the present recommendation, we only considered papers reporting a comparison among different doses of triptans currently authorised for use in clinical practice (Table 4). For each included paper, we considered a main efficacy outcome and a main safety outcome; main outcomes were selected as those with the highest number of included participants.
Reviews, systematic reviews, and meta-analyses reporting data on different doses of triptans.
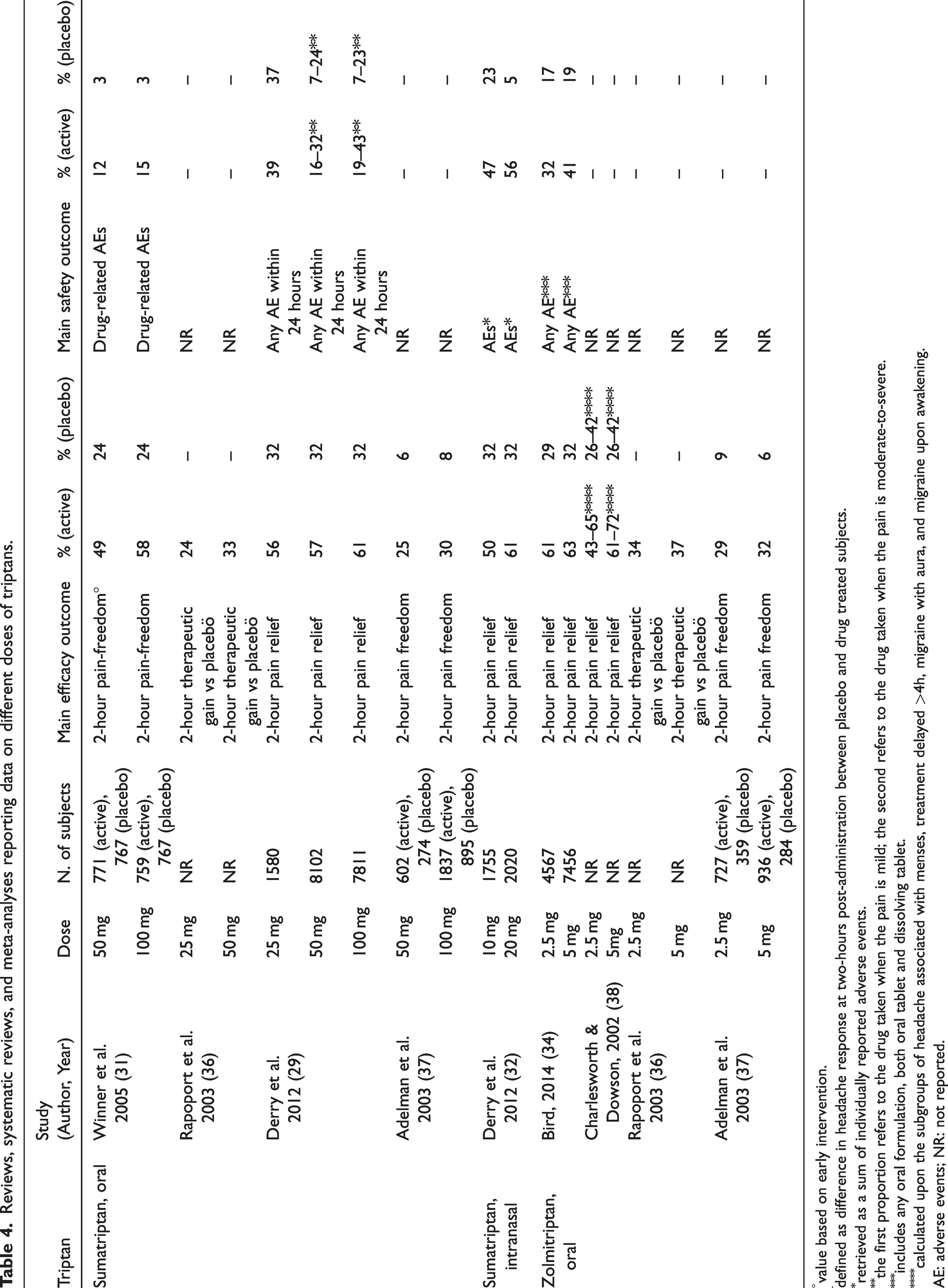
value based on early intervention.
defined as difference in headache response at two-hours post-administration between placebo and drug treated subjects.
retrieved as a sum of individually reported adverse events.
the first proportion refers to the drug taken when the pain is mild; the second refers to the drug taken when the pain is moderate-to-severe.
includes any oral formulation, both oral tablet and dissolving tablet.
calculated upon the subgroups of headache associated with menses, treatment delayed >4h, migraine with aura, and migraine upon awakening.
AE: adverse events; NR: not reported.
Triptans are currently commercialised in the following dosages (27):
Almotriptan: oral, 12.5 mg Eletriptan: oral, 20 mg or 40 mg Frovatriptan: oral, 2.5 mg Naratriptan: oral, 1 mg or 2.5 mg Rizatriptan: orally dissolving tablet, 5 mg or 10 mg Sumatriptan: oral, 25 mg, 50 mg, or 100 mg; subcutaneous, 6 mg; intranasal, 10 mg or 20 mg Zolmitriptan: orally dissolving tablet, 2.5 mg or 5 mg; intranasal, 5 mg
The availability of each specific triptan and of their formulations varies between countries.
Literature shows that the additional benefit of higher doses of triptans compared with lower doses within two hours from drug intake was 11% or less (Table 4). A dose-response effect could be hypothesized; however, the increased dose response was not significant. The therapeutic advantage of higher vs lower doses was modest or absent. The potentially higher efficacy of higher doses over lower doses of triptans is paralleled by an increase in AEs (28).
Overall, data suggests that lower doses of triptans should be preferred over higher doses, if effective. Tolerability issues should also be considered when recommending higher doses of triptans. If an individual does not experience sufficient relief with multiple doses of a triptan, other treatment options may be considered. For most people with migraine, switching acute treatment is more feasible than increasing the dose of a triptan. Therefore, increasing the dose of triptans may be a viable option for people who do not respond to an initial dose, but it is important to consider the potential risks and benefits of this approach, as well as other treatment options such as combination therapy (28).
Clinical practice considerations
Given the modest advantage demonstrated by increasing the dose of a triptan to obtain clinical benefit, clinical issues should be considered before considering a dose increase.
The importance of an adequate trial of triptans
Before assessing the efficacy of a triptan, its mode of consumption should be optimized. Triptans should be taken as early as possible after the onset of migraine to maximize efficacy (29). Additionally, the efficacy of triptans should be assessed over more than one migraine attack. The statement of the Consensus Panel of the European Headache Federation suggested that a triptan should be declared effective if able to treat at least three out of four consecutive migraine attacks (30). Vice versa, the ineffectiveness of a triptan in treating a single attack is not sufficient to consider it a failure.
Definition of partial response
Response to triptans can vary across different attacks. Response to triptans – as well as that to any drug for the acute treatment of migraine – can be defined according to different outcomes. Headache relief, cessation of associated symptoms, ability to prevent headache relapse and avoidance of rescue medication, tolerability, and patient satisfaction should all be considered. The Consensus Panel of the European Headache Federation considered headache relief, relief of associated symptoms, and absence of AEs within two hours from intake to define the response to triptans (30). According to this document, if headache relief is not complete, or headache recurs once relieved, or a rescue medication is needed despite the use of a triptan, the response is considered partial. In a partial triptan responder, the dose of the triptan can be increased, provided that the subject is not experiencing side effects. Viceversa, if there is no response at all at the tested dose, a dose increase is less reasonable, given that the expected benefit is likely modest.
Differences among triptans in dose-response effect
The type of triptan to which people with migraine respond might be relevant when considering a dose increase. Pooled analyses of RCTs showed that the 100 mg dose of oral sumatriptan was more effective than the 50 mg dose, even considering the increase in adverse events (29,31). Intranasal sumatriptan 20 mg was also more effective than the 10 mg dose (32). A dose-response effect has been attributed to eletriptan (33), while oral zolmitriptan 5 mg had similar efficacy compared with the 2.5 mg dose (34).
Different response to triptans across migraine attacks
A further element to consider when assessing the response to triptans is the possible difference among migraine attacks based on trigger factors, circadian rhythm, seasonality, or physiological states. An example of decreased response to triptans is menstrual migraine, in which attacks are longer and more debilitating compared with non-menstrual migraine (35). Migraine with onset during sleep is also associated with a reduced response to triptans, as it prevents early administration. Close monitoring of headache intensity, timing, and trigger factors through a headache diary could help in identifying different clinical situations where the adoption of different doses of the same triptan may be useful.
In summary, evidence indicates that increasing the dose of a triptan may be considered if the initial treatment is only partially effective. However, it is important to note that the use of these medications should be tailored to individual needs and medical history and should be done under the guidance of a healthcare professional. Therefore, the decision to consider a dose increase should be carefully assessed based on the pattern of migraine and its previous acute management. The expected gain of benefit from increasing the dose of a triptan is modest and more side effects can occur.
The relevant statements on triptan dose escalation in the guidelines reviewed and the guidance documents assessed are illustrated in Online Supplementary Table 2S.
Q3 – If people with migraine are not responding to the first triptan, should they switch to another triptan?
Recommendations
Optimal
If people with migraine are not responding* to the first triptan, used in adequate dosages, following the correct route of administration, and taken at the proper time** in two out of three attacks, we suggest switching to another triptan. This strategy can be repeated for up to a maximum of three triptans, after which another drug class is suggested.
Essential
As described in the recommendations for the Optimal level if at least two triptans are available.
If only one triptan is available, we suggest combining it with non-steroidal anti-inflammatory drugs or antiemetics.
*Not responding: the individual has not achieved pain-freedom two hours after the intake of the drug.
**Proper time: triptans are more effective when taken early during the attack. Patients should be educated to take them as early as the headache begins.
Background
Triptans are considered the first line therapy for the treatment of moderate to severe migraine attacks. An effectively treated attack can be defined by pain freedom being achieved within two hours and lasting for 24–48 hours (30,41). This should include the relief of migraine related non-pain symptoms and absence of AEs (3). Relapse is defined as the occurrence of migraine within 48 hours of obtaining pain freedom with an acute treatment (see Q10) (41). A meta-analysis demonstrated that triptans achieved headache relief in 42% to 76% of individuals within two hours, sustaining headache relief in 29% to 50% of subjects at 24 hours. Pain freedom at 24 hours was obtained in 18% to 33% of people with migraine (42).
One of the factors affecting the response to triptans is the timing of drug administration during the attack (see Q8). Triptans are not effective in relieving migraine if administered too early during the aura phase of migraine, as these drugs do not work on cortical spreading depolarization (43) and are less effective when taken too late, after central sensitization is fully developed (44). Route of administration also has an important effect on efficacy. Besides vomiting, delayed absorption in the gastroenteric system occurring during attacks may lead to slower or reduced absorption of oral triptans (3,45). Timing of attacks is also an issue. Many people with migraine awake with a full-blown attack. These people will likely benefit more from subcutaneous triptan (sumatriptan 3 mg or 6 mg) instead of the oral route.
Evidence
Head-to-head studies did not show clear superiority of one triptan over another with regards to pain relief (46). Four systematic reviews, however, including 111 individual studies, reported that sumatriptan, zolmitriptan and almotriptan showed similar efficacy, while eletriptan and rizatriptan were superior based on pain freedom at two hours, and eletriptan led to a lower recurrence rate (47). The findings of a meta-analysis of 133 RCTs suggested that the majority of triptans (except frovatriptan and naratriptan) provide similar efficacy in the acute management of migraine attacks. The results also suggested better pain relief with eletriptan and rizatriptan (42). Although all triptans have a similar molecular structure, differences in pharmacokinetic and pharmacodynamic profiles of these drugs are the underlying causes of their slightly variable efficacy and side effects, and the variability in individual responses (48). Naratriptan and frovatriptan have the longest duration of action due to their long half-lives (26). Almotriptan, naratriptan and frovatriptan caused fewer AEs in comparison to sumatriptan and other triptans (3,49).
Considering these individual differences between triptans, guidelines or guidance documents recommend offering a different triptan to a subject who does not benefit from one triptan (Online Supplementary Table 3S). Lack of efficacy in two attacks with a particular triptan is considered a failure in the British Guidelines (46), whereas other guidelines set this limit at three attacks (25,50). The Consensus Panel of the European Headache Federation recently provided a definition for triptan-responders that suggested considering a triptan effective if well-being is restored in at least three out of four migraine attacks. Failure of triptans is set as not meeting the condition of triptan-responder (30), with further specification of triptan resistance (failure of at least two triptans) and triptan refractoriness (failure of at least three triptans, including the subcutaneous formulation). In general, guidelines underline the importance of timing of drug intake as well as the route of administration.
Q4 – In people with migraine with nausea and/or vomiting, should antiemetics be combined with analgesics, non-steroidal anti-inflammatory drugs or triptans?
Recommendations
Optimal
In people with migraine with nausea and/or vomiting that is not manageable with timely intake of an acute attack drug, we suggest adding an antiemetic to analgesics, NSAIDs or triptans, if not contraindicated.
Where available, fixed combinations of analgesics, non-steroidal anti-inflammatory drugs or triptans may be considered.
Essential
In people with migraine with nausea and/or vomiting that is not manageable with timely intake of an acute attack drug, we suggest adding an antiemetic to analgesics, non-steroidal anti-inflammatory drugs or triptans, if not contraindicated.
Comment: Gastroprotection may be required in cases with multiple doses of NSAIDs.
Background
Nausea is one of the most common symptoms associated with a migraine attack. The addition of an antiemetic can improve the efficacy of migraine rescue treatment and alleviate nausea or vomiting related to a migraine attack.
Evidence
Combining analgesics, NSAIDs, or triptans with antiemetics may be considered to improve the efficacy of migraine treatment, especially in indivduals with nausea and vomiting. This approach is supported by several guidelines/guidance documents, although the available literature is limited (Online Supplemental Table 4S). According to the American Headache Society, antiemetics are recommended as first-line adjunctive therapy for individuals with moderate to severe nausea or vomiting associated with migraine, emphasizing the importance of using antiemetics in conjunction with acute migraine treatments (53).
One study compared the effectiveness and tolerability of a fixed combination of domperidone and paracetamol with sumatriptan in treating moderate to severe migraine attacks. The results showed that both treatments had comparable efficacy in relieving headache and reducing nausea and vomiting and were well-tolerated with no serious AEs (54). Domperidone and paracetamol may be a more cost-effective first-line treatment option for people with migraine seen in routine general practice compared to sumatriptan and other triptans. Other studies compared acetylsalicylic acid (ASA) alone versus ASA plus metoclopramide, suggesting a more marked effect of ASA when administered with the antiemetic (55).
Another study compared the efficacy of sumatriptan alone versus sumatriptan combined with the antiemetic metoclopramide and found that the combination therapy was significantly more effective in achieving pain relief and reducing nausea and vomiting (56). Further studies are needed to determine whether initiating therapy when pain is mild or using a higher dose of sumatriptan would provide additional benefits.
Combining analgesics, NSAIDs, or triptans with antiemetics may be a viable option to improve the efficacy of migraine treatment in individuals with nausea and vomiting. The use of antiemetics should be tailored to individual needs and medical history and should be conducted under the guidance of a healthcare professional. Evidence supporting the use of antiemetic drugs in combination with analgesics, NSAIDs, or triptans is illustrated in Table 5. Antiemetic drugs that can be combined with analgesics are reported in Table 6.
Summary of evidence on the combination of an anti-emetic with analgesic, non-steroidal anti-inflammatory drugs, or triptan in available literature.

AE: adverse event; ASA: acetylsalicylic acid; i.m.: intramuscular; i.v.: intravenous; SR: systematic review; RCT: randomized controlled trial; N/A: not available.
Antiemetic medications for migraine that can be used in addition to analgesics. Adapted from Marmura et al. (65)

i.m.: intramuscular; i.v.: intravenous; p.o.: by mouth; p.r.n.: as needed; RCT: randomized controlled trials.
Online Supplementary Table 4S summarizes the statements of the guidelines reviewed and the guidance documents assessed on the combination of an anti-emetic with analgesics, NSAIDs, or triptans.
Q5 – If triptans are only partially effective, should a combination of non-steroidal anti-inflammatory drugs and triptans be used?
Recommendations
Optimal
In people with migraine who only respond partially* to triptans as single agents, even after triptan treatment has been optimized (see Q2 and Q3), we suggest the combination of oral sumatriptan (50–100 mg) and oral naproxen sodium (550 mg) as first choice.
Alternatively, a triptan can be combined with any fast release oral formulation of a non-steroidal anti-inflammatory drug.
Essential
Combine any available triptan with available non-steroidal anti-inflammatory drugs.
*Partial efficacy: the individual has not achieved pain relief two hours after the intake of the drug.
Comment: Gastroprotection may be required in case of multiple dosing of NSAIDs.
Background
Between 20% and 40% of people with migraine who treat an acute migraine attack with a triptan do not achieve the treatment goal of pain freedom after two hours. In these people, a combination of NSAIDs and triptans could represent a valid alternative to improve efficacy of acute treatment.
Evidence
The fixed combination of sumatriptan and naproxen sodium was investigated in 11 randomized trials in adults (sumatriptan 85 mg, naproxen 500 mg). Importantly, these studies were not restricted to people with migraine who did not benefit from either sumatriptan or naproxen alone. The combination was superior to placebo, sumatriptan or naproxen sodium monotherapy for the endpoint of pain freedom after two hours. Several RCTs have also investigated the non-fixed combination, including two trials in adolescents (sumatriptan 85 mg, naproxen 500 mg) (70–73). Available guidelines and guidance documents on this topic are summarized in Online Supplementary Table 5S.
In the absence of controlled trials investigating the combination of other triptans and NSAIDs, we recommend the combination of sumatriptan and naproxen sodium as first choice. Combinations of other triptans with other NSAIDs are also reasonable despite not having been specifically investigated.
Q6 – Do gepants and lasmiditan have a role in treating migraine attacks?
Recommendations
Optimal
Gepants and lasmiditan are an option for treating the acute attack in people with migraine for whom triptan monotherapy or combination therapy (see Q2, Q3 and Q5) are not effective, only partially effective or not tolerated, or in subjects with contraindications to triptans.
Essential
Not applicable.
Background
In recent years, the armamentarium for acute migraine management has increased significantly, with a new generations of oral small molecule calcitonin gene-related peptide antagonists (gepants) (74), and lasmiditan, a potent and selective agonist of the 5-HT1F receptor. These drugs have been developed and designed specifically for migraine and do not present the vasoconstriction issues that reduce the bandwidth of triptan use (75).
Evidence
Lasmiditan has bene tested in three doses (50, 100 and 200 mg) for the acute treatment of migraine in three large phase-3 RCTs: SAMURAI (76), SPARTAN (77) and CENTURION (78), as well as in the open label continuations of these same studies (79–81). Overall, lasmiditan has shown superiority to placebo in primary efficacy outcomes, including two-hour pain freedom and freedom from the most bothersome migraine-associated symptom at two hours, as well as secondary endpoints such as freedom from photo-phonophobia and sustained pain freedom at 24 hours. Side effects associated with its activity on the central nervous system may limit tolerability (82), causing nausea, dizziness and fatigue (75). In some countries, individuals are not allowed to drive or use machinery for several hours after lasmiditan intake.
Ubrogepant was studied at the doses of 25, 50 and 100 mg in two RCTs (83,84). Only the 50 and 100 mg formulations been approved commercially. Both RCTs showed higher efficacy than placebo, with the most common AEs being nausea, somnolence, and dry mouth. A one-year open label extension study also showed good safety and tolerability profiles for long-term use of the 50 mg and 100 mg doses (85).
Rimegepant is available as a 75 mg orally disintegrating tablet and was tested in two RCTs (86,87), where it has demonstrated efficacy and tolerability. The drug has also induced a reduction in monthly migraine days in an open label extension trial (88), with nausea being the most commonly reported AE.
Zavegepant 10 mg is the only gepant available in an intranasal formulation, and was recently approved for use by the FDA following one large RCT (89). This study showed efficacy in the main outcome measures of pain freedom and freedom from the most bothersome symptom at two hours, with side effects mostly characterized by dysgeusia, nausea and nasal discomfort.
Importantly, gepants have not been shown to cause medication overuse and seem to be useful if taken during the prodrome, the phase of migraine occurring prior to the onset of pain (see Q8).
Given the recent availability of these compounds, only a few guidelines guidance documents discuss their use in the acute treatment of migraine attacks. These are summarized in Online Supplementary Table 6S.
Q7 – Are ergot derivatives an option for treating migraine attacks?
Recommendations
Optimal
The use of ergot derivatives for treating acute migraine attacks can be considered if all recommended acute treatments with better safety profiles have failed.
Essential
As described in the recommendations for the Optimal level.
Comment: The individual should be advised about the potentially serious side effects, the possibility of headache relapse and the risk of developing medication overuse headache. The use of ergots should be limited to no more than one day per week.
Background
Ergot derivatives are primarily used for the acute treatment of migraine and their efficacy has been demonstrated in several studies (57,90,91). Ergotamine tartrate is available as an oral compound and, in some countries, in a rectal formulation, while dihydroergotamine (DHE) can be given via an intranasal, sublingual, intravenous or intramuscular route (92). Drawbacks of ergot derivatives are their low oral bioavailability, the risk of inducing medication overuse headache and the possibility of causing serious drug interactions (7,93). Frequent side effects include nausea and vomiting. Less frequent and more severe AEs with frequent or regular administration include ergotism, limb ischemia, arterial stenosis, myocardial infarction, cardiac valve lesions, ano-rectal ulcers, rectal stenosis and fibrosis (94). Ergots are contraindicated in arterial vascular diseases. A relative advantage of ergot derivatives may be the reduced frequency of headache recurrence, when compared to triptans (91).
Evidence
Studies regarding efficacy of ergot derivatives have varying methodological quality (90,91,95–109). Studies investigating inhaled DHE 0.5–1 mg, DHE nasal spray 0.9 mg or oral ergotamine tartrate 1 mg have shown significantly more pain freedom after two hours, as well as reduced headache duration and intensity compared to placebo (95,96,100). In other studies no efficacy difference between ergot derivatives and placebo was found when investigating ergotamine suppositories 2 mg, DHE nasal spray 0.5–1 mg and ergotamine 1 mg combined with caffeine 100 mg (102,103,105,107).
Ergot derivatives were also compared to other migraine abortive medications including ketoprofen, naproxen or tolfenamic acid (99,100,102). Ergotamine tartrate 1–2 mg (oral or suppository) was not superior to ketoprofen 100 mg (suppository) (102), acetylsalicylic acid 500 mg (100), naproxen 750 mg or tolfenamic acid 200 mg (100,105). One study showed better efficacy for ergotamine tartrate 1 mg compared to acetylsalicylic acid 500 mg (99).
Four RCTs investigated the efficacy of ergot derivatives vs triptans (97,101,104,109), and all studies favored acute treatment with a triptan. Two studies investigated the efficacy of nasal or subcutaneous DHE 1 mg compared to subcutaneous sumatriptan 6 mg (104,109). Significantly more participants treated with subcutaneous sumatriptan 6 mg reported complete headache relief. Two further studies evaluated the efficacy of the combination of 2 mg ergotamine tartrate and caffeine 200 mg versus oral sumatriptan 100 mg or eletriptan 40 mg or 80 mg (97,101). The treatment with either triptan showed significantly better headache relief at two hours compared to oral ergotamine.
An appraisal of guidelines found that the majority of them have cautioned against routine use of ergot derivatives and only a minority have recognized their utility as a last resource in refractory individuals (110). Some of the guidelines suggesting ergotamine were developed before new options (e.g. gepants and lasmiditan) were available. The summary of statements about ergot derivatives used in the guidelines and guidance documents considered in this manuscript are listed in Online Supplementary Table 7S.
Q8 – What is the recommended timing of administration of acute treatment?
Recommendations
Optimal
Patients with migraine without aura should take their treatment while the pain intensity is still mild, preferably as early as possible in the headache phase.
Patients with migraine with aura should take their treatment as soon as the headache phase starts.
Essential
As described in the recommendations for the Optimal level.
Comment: To avoid the risk of medication overuse, people with migraine should be advised that frequent use of most acute medications is considered to increase the risk of developing such condition. Gepants have not been associated with the risk of medication overuse headache to date.
Background
Around 30% of people with migraine who treat their migraine attacks with oral triptans do not achieve pain freedom after two hours (112). It has been suggested that taking the medication too late, when sensitization of central trigeminovascular neurons has developed, may be a reason for the lack of efficacy of triptans (113). On the other hand, treating headache pain too early might result in increased risk of medication overuse headache if attacks are frequent (114,115), therefore careful evaluation of individual cases is required.
Evidence
The effectiveness of treatment taken early and/or during mild pain compared to treatment taken during moderate to severe pain has been examined in 11 studies (116–126). All studies used pain freedom at two hours as the primary endpoint.
The efficacy of oral sumatriptan 50 mg or 100 mg taken when pain was mild was superior to placebo in four studies (116,117,119,120). Other studies have shown that oral rizatriptan 10 mg, eletriptan 20 mg or 40 mg, almotriptan 12.5 mg, zolmitriptan 2.5 mg and frovatriptan 2.5 mg are superior to placebo when taken while the pain is mild (121–125). A post-hoc analysis of a RCT reported that the therapeutic gain for pain-freedom at two hours was statistically higher when treating attacks with oral sumatriptan 50 mg while pain was mild compared to treating when pain was moderate-to-severe in the same participants (116). This finding has been confirmed in an open-label study with sumatriptan 100 mg (126). Similar findings have been reported with oral rizatriptan 10 mg and oral almotriptan 12.5 mg (118,121,123).
The efficacy of treating a migraine attack during the aura phase has been investigated, with contradictory findings. Three studies found that subcutaneous sumatriptan 6 mg, oral eletriptan 80 mg and oral zolmitriptan 20 mg were not effective at treating migraine headache when the medication was taken during the aura phase (127–129). In another trial, people with migraine were given oral sumatriptan 200 mg (non-approved dose) or placebo at the onset of migraine aura for three attacks. During the first attack, the reduction of migraine severity was superior with sumatriptan compared to placebo, however no difference was found for the subsequent two attacks (130). The statements on this topic from the guidelines and guidance documents reviewed are summarized in Online Supplementary Table 8S.
A recent study has shown that ubrogepant was effective in preventing headache when administered during the prodrome, the phase of migraine occurring prior to the onset of pain (131). This could represent a significant advance for the treatment of migraine attacks for individuals with reliable prodromal symptoms, particularly since gepants do not appear associated with medication overuse (74).
Q9 – Which treatment options are available for individuals who experience early vomiting during a migraine attack?
Recommendations
Optimal
In individuals with early vomiting, we suggest non-oral formulations of acute medications, such as subcutaneous injections, intranasal sprays or suppositories, based on availability, subjective preference, and medical history. Orally disintegrating tablets may also be considered.
Alternatively, we suggest a combination of simple analgesics, non-steroidal anti-inflammatory drugs or triptans with antiemetics.
Essential
As described in the recommendations for the Optimal level, for available treatments and formulations.
Comment: Gastroprotection may be required in cases with multiple doses of NSAIDs.
Background
More than 60% of people with migraine experience nausea and vomiting during their attacks. These symptoms may be more disabling than the headache itself. Vomiting early in an attack impairs the effectiveness of abortive oral treatment – leading to poor management of the attack.
Evidence
Non-oral treatments, including intranasal, subcutaneous or rectal administration, is preferred in individuals who vomit early in an attack. Antiemetics, in combination with simple analgesics or triptans, may also be efficient in some cases.
Intranasally administered sumatriptan, zolmitriptan, zavegepant or DHE are good alternatives as they mostly by-pass abdominal absorption (132). Intranasal sumatriptan and zolmitriptan have been shown to be as effective or superior to oral triptans and superior to placebo at achieving pain freedom two hours postdose (133–136). Studies show that oral sumatriptan and zolmitriptan are as good as the combination of an antiemetic (metoclopramide) and acetylsalicylic acid at reducing the incidence of nausea and vomiting (137,138). Furthermore, intranasal DHE is significantly better than placebo at relieving migraine pain (139–141). In a clinical trial comparing the efficacy of intranasal sumatriptan 20 mg and intranasal DHE 1 mg (with optional second dose), the treatment with sumatriptan resulted in significantly greater pain relief than treatment with DHE (142). Intranasal zavegepant was developed for the acute treatment of migraine attacks. Two phase 3 trials have shown that the rate of pain freedom at two hours postdose was higher following treatment with intranasal zavegepant 10 mg or 20 mg compared to placebo (143,144). As an alternative, oral disintegrating tablets (e.g., rizatriptan, zolmitriptan and rimegepant) can also be recommended (145). These options provide faster onset of action than traditional tablets and are superior to placebo at treating migraine attacks (87,146–148).
Subcutaneous sumatriptan is a fast-acting option due to its route of administration and therefore an ideal formulation for treating the migraine headache associated with early vomiting. Multiple clinical trials have shown that subcutaneous sumatriptan 6 mg is superior to placebo for pain freedom at two hours (149). Subcutaneous sumatriptan may also be effective at the dose of 3 mg (150). One study reported that subcutaneous sumatriptan 6 mg was superior to placebo at reducing the incidence of nausea, vomiting and/or photo-/phonophobia (151).
Alternatively, antiemetics in combination with either a simple analgesic or a triptan may be used. In a double-blind, randomized, crossover study, the combination of sumatriptan 50 mg and metoclopramide 10 mg was more effective than sumatriptan 50 mg and placebo at reducing pain, nausea and vomiting (56). A Cochrane subgroup analysis concluded that metoclopramide combined with aspirin was superior to aspirin alone in relieving nausea and vomiting (55).
Even though available guidelines and guidance documents on the topic (summarized in Online Supplementary Table 9S) are consistent, current evidence is limited due to the small number of participants in clinical studies reporting vomiting. In the absence of head-to-head studies, we recommend either intranasal sumatriptan, zolmitriptan, zavegepant, DHE or subcutaneous sumatriptan as first line treatment based on availability, cost and patient preference. Alternatively, oral disintegrating tablets (e.g., rizatriptan, zolmitriptan and rimegepant) can be recommended. If the optimal-mentioned treatments are not available, antiemetics in combination with a triptan or NSAID can be considered based on availability and patient preference.
Q10 – How can headache relapse be treated following the initial successful treatment of a migraine attack?
Recommendations
Optimal
In people with migraine with headache relapse* after the initial successful treatment of a migraine attack, we suggest taking a second dose of the same medication within the recommended dose limit. If this approach is not effective, we suggest switching to another drug, possibly belonging to a different class.
If early headache relapse occurs in most of the attacks, we suggest switching to a different treatment option.
Combining a triptan with a non-steroidal anti-inflammatory drug may also be a viable option. It is important to wait at least two hours from the first dose before repeating a combination treatment.
Essential
In people with migraine with headache relapse* after the initial successful treatment of a migraine attack, we suggest taking a second dose of the same medication within the recommended dose limit.
If this approach is not effective, we suggest switching to another drug belonging to a different class or, if not available, to use a combination of a triptan with a non-steroidal anti-inflammatory. It is important to wait at least two hours from the first dose before repeating a combination treatment.
*Relapse is defined as the recurrence of migraine of any intensity within 48 hours of obtaining pain freedom with an acute treatment.
Comment: Gastroprotection may be required in cases requiring multiple dosing of NSAIDs.
Background
Relapse is a phenomenon that has been well described and investigated after the availability of triptans. About 15–40% of the people with migraine taking an oral triptan experience relapse.
Evidence
One RCT showed that about one-fourth of participants with migraine experience a headache relapse within 16 h after successful treatment of a migraine attack with sumatriptan. In the same study, sumatriptan 100 mg was superior to placebo when treating headache recurrence: 70–74% vs 30–49% (152).
If relapse with triptans occurs frequently, one option is to switch to a triptan with a longer half-life, such as naratriptan and frovatriptan (153). It should be noted, however, that these drugs may have a slower onset of effect and lower efficacy than other triptans (154). An alternative option, especially if single drug options do not work or are not available, is the combination of a triptan with an extended release NSAID. An appraisal of the statements provided in the guidelines and guidance documents assessed in these practice recommendations is reported in Online Supplementary Table 10S.
Q11 – How should migraine attacks that persist for more than 72 hours (status migrainosus) be treated?
Recommendations
Optimal
Although there is a lack of reliable evidence, in subjects with attacks lasting more than 72 hours (status migrainosus), we suggest intramuscular or other forms of administration of non-steroidal anti-inflammatory drugs or subcutaneous sumatriptan, or oral/intranasal dihydroergotamine (in combination with antiemetics).
In the emergency room setting, we suggest considering the following medications, preferably using intravenous formulations: non-steroidal anti-inflammatory drugs or acetylsalicylic acid, with or without an antidopaminergic agent (e.g. prochlorperazine, metoclopramide, and chlorpromazine).
Steroids, peripheral nerve blocks, intravenous magnesium, sodium valproate or dihydroergotamine can be offered to individuals not responding to the previous options.
Opioids should be avoided at all times.
Essential
As described in the recommendations for the Optimal level, utilizing available treatments and formulations.
Intravenous dexamethasone (on the WHO list of available medications) can be considered.
Comment: Gastroprotection may be required in cases requiring multiple dosing of NSAIDs.
Background
Status migrainosus is defined by ICHD-3 criteria as a debilitating migraine attack lasting for more than 72 hours in an individual with migraine (with or without aura) (6). It is consistent with previous attacks except for its duration and severity. Status migrainosus presents with pain and/or associated symptoms that are debilitating and unremitting for more than 72 hours (remissions of up to 12 hours due to medication or sleep are accepted).
Evidence
Specific clinical trials focusing on status migrainosus are lacking (61). Several studies and recommendations, however, analyze the acute treatment of migraine attack in the emergency department and/or using parenteral medications and we therefore refer to these studies to guide our recommendations.
Oral medications, including triptans are usually not helpful when standard acute medications have failed to abort an attack (155). Evidence supports the use of subcutaneous sumatriptan and parenteral prochlorperazine or metoclopramide for the treatment of acute migraine attacks in an emergency room setting. A total of 14 trials of sumatriptan in the emergency department setting were found, and a meta-analysis showed consistent evidence favoring sumatriptan over placebo (60). It should be noted, however, that not all participants included in those studies had status migrainosus and that sumatriptan is frequently not available in the emergency room in many regions. A meta-analysis of 11 trials on prochlorperazine versus placebo showed the superiority of prochlorperazine (156). Six trials have compared parenteral metoclopramide to placebo and yielded positive results, although the studies could not be fully meta-analyzed due to significant heterogeneity (157–163). Efficacy of chlorpromazine i.v. was tested in comparison with placebo in a small randomized controlled trial and recommended by the Canadian and TOP guidelines (Online Supplementary Table 11S). Parenteral NSAIDs such as ketorolac 30 mg intravenously and lysine acetylsalicylic acid are reasonable first-line choice and recommended by several guidelines. DHE can be used as alternative to parenteral triptans. Opioids are not recommended due to the absence of placebo-controlled trials and risk of long-term overuse, relapse or abuse.
Recommendations regarding parenteral steroids are conflicting across guidelines. A meta-analysis of seven randomized controlled studies of dexamethasone added to the standard abortive therapy reported a significant benefit favoring dexamethasone to prevent recurrence of migraine headache (164). The guidelines of the American Headache Society report a meta-analysis of three class I RCTs that showed a small benefit of dexamethasone over placebo in preventing relapse (68). While controversy exists, experts generally recommend steroids, including dexamethasone or prednisolone, as adjunctive therapy in cases where all first-line therapies have failed as the side effect profile of single-use steroids is favorable, unless contraindicated (22).
Evidence for the use of nerve blocks, intravenous magnesium or sodium valproate is scarce. These treatments can be evaluated if all other options are unavailable, contraindicated, or ineffective.
Q12 – What is the maximum number of days that acute medications can be administered without increased risk of developing medication overuse headache?
Recommendations
Optimal
We suggest limiting the use of analgesics, non-steroidal anti-inflammatory drugs or lasmiditan to 2–3 days per week and to less than 10 days per month.
For combined analgesics and triptans we suggest limiting the intake to 2 days per week and to less than 8 days per month.
Essential
We suggest limiting the intake of analgesics and non-steroidal anti-inflammatory drugs to 2–3 days per week and to a maximum of 10 days per month.
For combined analgesics and triptans we suggest limiting the intake to 2 days per week and to less than 8 days per month.
Comment: Gepants have not been associated to medication overuse headache and may therefore become the preferred choice in people with migraine that require frequent use of acute drugs.
Background
Excessive acute medication use is associated with an increase in headache pain and associated symptoms and medication overuse headache. One of the major concerns for clinicians is whether the frequent use of acute medications can accelerate the development of high-frequency/chronic headache. Unfortunately, there is no definitive evidence on the exact amount of acute medications that can be used without causing MOH, defined by ICHD-3 as a headache on 15 or more days per month and intake of acute medication 10 or 15 or more days per month depending on the drug class (6).
It is also difficult to separate frequent intake of acute medications due to an increase in migraine frequency from the increase of migraine frequency due to frequent intake of acute medication. The present recommendations have been developed based on evidence describing acute medication involvement in migraine chronification, and not necessarily on MOH-specific studies.
Evidence
The majority of acute medications have the potential to cause MOH. Some products appear to be more hazardous than others. The retrospective study of Limmroth et al. provided the correlation between MOH and each acute medication (166). With the shortest mean critical duration until the onset of MOH and lowest mean critical monthly intake frequency, people with migraine overusing triptans demonstrated the highest tendency to develop MOH, followed by ergot derivatives, then analgesics (including simple and combination analgesics).
NSAIDs are unique amongst acute treatments as they can play a positive role in inhibiting the progression of MOH. In some of the studies, overuse of NSAIDs showed protective effects against developing MOH (167,168). However, this does not mean NSAIDs can be used on an unlimited basis. Based on a nationwide, large sample size, longitudinal study by Lipton et al. (169), the relationship between NSAID consumption and headache chronification interfered with the headache frequency of an individual. In people with <10 headache days per month, the use of NSAID was protective against migraine chronification. In contrast, in people with headache on
As for ergots, paracetamol/acetaminophen, and combined analgesics (e.g., consisting of paracetamol/acetaminophen, aspirin, and caffeine), there is no direct evidence providing the critical level of use for developing MOH. The study by Bigal et al. (170), which investigated the association between the acute medication use and migraine chronification, provides hints at reasonable limitations on acute medication use. Two hundred and nine (2.5%) out of the 8219 people with episodic migraine (EM) had transformed to chronic migraine (CM) during the one-year follow-up. Those who remained episodic reported fewer monthly days of exposure to acute medications. The average monthly days of exposure to ergots, acetaminophen, and combination analgesics (acetaminophen + aspirin + caffeine) in EM were 4.3, 6.4, and 5.4 days, whereas the CM counterpart reported 8.6, 12.8, and 10.0 days. It is difficult to generate a definite upper limit for acute medications use from these data. This well-designed and large-scale observational study does suggest that it is safe to confine the use of ergots, and combined analgesics to no more than eight days per month.
The current evidence regarding the association between MOH and gepants or lasmiditan is limited. Preclinical studies in rodents (171–173) reveal that persistent exposure to lasmiditan, but not gepants, may cause cutaneous allodynia, a possible indicator of migraine chronification and MOH (168,174). In the case of gepants, a long-term, open label study has suggested that rimegepant may not be associated with MOH (175). This observation, when combined with the efficacy of rimegepant in the preventive treatment of migraine, suggests that frequent rimegepant use may not be associated to an increased risk of developing MOH.
The recommended limits for acute medication use to prevent MOH vary across guidelines and consensus statements. Most of them suggest that acute medication use should not exceed 2–3 days per week, i.e., 8–10 days per month (Online Supplementary Table 12S). The Danish Headache Society recommends the limit of simple analgesics use is up to 14 days per month while combined analgesics should be limited to no more than nine days per month. A national awareness campaign to prevent MOH conducted in Denmark recommended a maximum intake of analgesics with two days per week (176). These suggestions are based on experts’ consensus. Individual heterogeneity should be considered in clinical practice and the thresholds should be modified individually (8).
We recommend that the use of triptans, ergots or combination analgesics be kept to less than 2–3 days per week and no more than 10 days per month, to minimize the risk for developing MOH. If more frequent use is required, it should be limited to less than three consecutive months. Gepants may be preferable in individuals with higher risk for MOH.
Q13 – Which treatment options are preferable during pregnancy and breastfeeding?
Recommendations
Optimal
In pregnant women whose attacks cannot be adequately managed with non-pharmacologic approaches, paracetamol/acetaminophen and triptans can be used with caution across the three trimesters of pregnancy.
Metoclopramide may be added if needed for nausea or vomiting, or in women with inadequate pain relief.
During breastfeeding, paracetamol/acetaminophen is the preferred choice. Diclofenac, naproxen, triptans and gepants can be used with caution, such as withholding breastfeeding for 8–12 hours.
Essential
As described in the recommendations for the Optimal level, utilizing available treatments and formulations.
Background
In up to 90% of cases, migraine progressively improves or remits during pregnancy (178). Approximately 8% of women, however, experience worse attacks or an increase in their frequency during pregnancy (179). Migraine also typically recurs shortly after delivery or cessation of breastfeeding (180). Acute management options are therefore needed for this population.
Evidence.
Paracetamol/acetaminophen
The European Medicines Agency (EMA) investigated whether paracetamol use during pregnancy is associated with neurodevelopmental problems in children (181). The authors concluded that acetaminophen exposure during pregnancy has no negative effects on neurodevelopment (181). More recently, a large cohort analysis showed that paracetamol, when used alone, is not associated with adverse neonatal outcomes (182). Low amounts of paracetamol are excreted in breast milk. Newborns and adults have roughly the same capacity for acetaminophen metabolism (183,184). Recent evidence, however, suggests that paracetamol/acetaminophen may be associated with urogenital/reproductive disorders in the offspring, possibly due to the endocrine effect of the drug, which suggests avoiding uncontrolled use in pregnancy, until more reliable evidence is made available (185–187). Paracetamol/acetaminophen is considered relatively safe during breastfeeding (188).
NSAIDs
AEs following NSAIDs use differ according to the trimester of exposure. NSAIDs use during the first trimester has been associated with miscarriage (189–191) and congenital malformations (192–194) in some studies, while other studies did not find increased risk of miscarriage after NSAIDs use (195–197). Similarly, some population-based studies suggest that NSAIDs use during the third trimester and prenatal period is associated with congenital malformations (192–194), while other population-based studies do not confirm these results (198–200).
NSAIDs are considered compatible with breastfeeding, and no AEs have been documented in breastfed infants (184). Ibuprofen is therefore recommended as the drug of choice, due to its low amount of excretion in breast milk and short half-life (23,201). Newer evidence also supports the use of diclofenac and naproxen when breastfeeding (202). In regard to the potential risks of miscarriage and congenital malformations, we recommend limiting use of NSAIDs to the second trimester, as well as after birth. We emphasize that the use of NSAIDs is only justifiable when non-pharmacological therapy provides no benefits in the acute treatment of a migraine attack.
Triptans
Of the seven available triptans, most available data relate to the use of sumatriptan during pregnancy. Cohort studies have shown no risks of congenital malformations following triptans use (203–208). In support, one meta-analysis investigated pregnancy outcomes following prenatal exposure to triptans from 1991–2013 (209). The meta-analysis included six studies and a population of 4208 infants of mothers with migraine who used triptans (sumatriptan was the mostly used triptan), compared to 1,466,994 infants of mothers with migraine who did not use triptans during pregnancy. The authors found no significant increase in the incidence of congenital malformations ([OR] = 0.84 [0.61–1.16]), prematurity ([OR] = 0.90 [0.35–2.30]), or spontaneous abortions ([OR] = 1.27 [0.58–2.79]), when comparing the triptan-exposed infants to the control group. The data suggests that use of sumatriptan is safe during pregnancy, while knowledge regarding other triptans’ safety remains limited. If necessary, we recommend sumatriptan as the first choice of treatment with triptans, however this must be under strict supervision of a headache specialist. Recommendations for triptan use during breastfeeding is hindered by the lack of data.
Antiemetics
For nausea and vomiting associated with migraine during pregnancy, metoclopramide can be used (23,210). Safety data on the use of domperidone during pregnancy is scarce, but pediatric use of domperidone has shown QT prolongation in newborns (211).
Metoclopramide use during lactation is not associated with any adverse effects in breastfed infants (212).
Combination formulations with butalbital or other opioids should be avoided during pregnancy and breastfeeding.
As reliable evidence is lacking, there is some disparity across published guidelines and guidance documents regarding the preferred options of acute medication that can be used during pregnancy (Online Supplementary Table 13S). A reliable source for reference is represented by the Drug and Lactation Database ((LactMed®) https://www.ncbi.nlm.nih.gov/books/NBK501922/, which is regularly updated.
Q14 – What drugs can be used in children and adolescents with a migraine attack?
Recommendations
Optimal
We suggest paracetamol/acetaminophen (15 mg/kg; maximum 60 mg/kg per day) or ibuprofen (10 mg/kg; maximum 30 mg/kg per day) to treat acute migraine attacks in children and adolescents.
If those drugs are not effective, triptans can be used as second line therapy for adolescents. Among triptans, rizatriptan (5 mg for a body weight <40 Kg, 10 mg for body weight ≥40 Kg) or sumatriptan nasal spray 10 mg are preferable as these are the most studied triptans in adolescents.
Metoclopramide might be added in cases with nausea or vomiting or in very disabling attacks.
Essential
As described in the recommendations for the Optimal level, utilizing available treatments and formulations.
Comment: Gastroprotection may be required in cases requiring multiple dosing of NSAIDs.
Background
The characteristics of migraine in the pediatric age may be different from those in adulthood. Attacks tend to be shorter, bilateral, and with prominent gastrointestinal symptoms (213,214). Different drugs for the acute treatment of migraine have been used in children and adolescents, including non-specific analgesics, triptans, ergotamine derivatives, and antiemetics. The present practice recommendations only refer to drugs and exclude non-pharmacologic treatments for the acute management of migraine.
Another difference between pediatric and adult populations is the placebo response, which is much higher in children and adolescents, especially in parallel trials (215). Placebo response is so high in children and adolescents with headache that it might influence the results of placebo-controlled trials, even when the response to the active drug is high. For this reason, the effectiveness of drugs in real-world settings could be better than shown in RCTs.
Evidence
Table 7 presents a summary of available randomized placebo-controlled trials of acute treatments of migraine in childhood and adolescence. Paracetamol and ibuprofen were more effective than placebo in treating migraine in an age range including infancy (216,217). Figure 1 illustrates the drugs recommended or suggested for the acute treatment of migraine in children and adolescents.
Summary of evidence from randomized placebo-controlled trials on oral drugs for the acute treatments of migraine in children and adolescents.

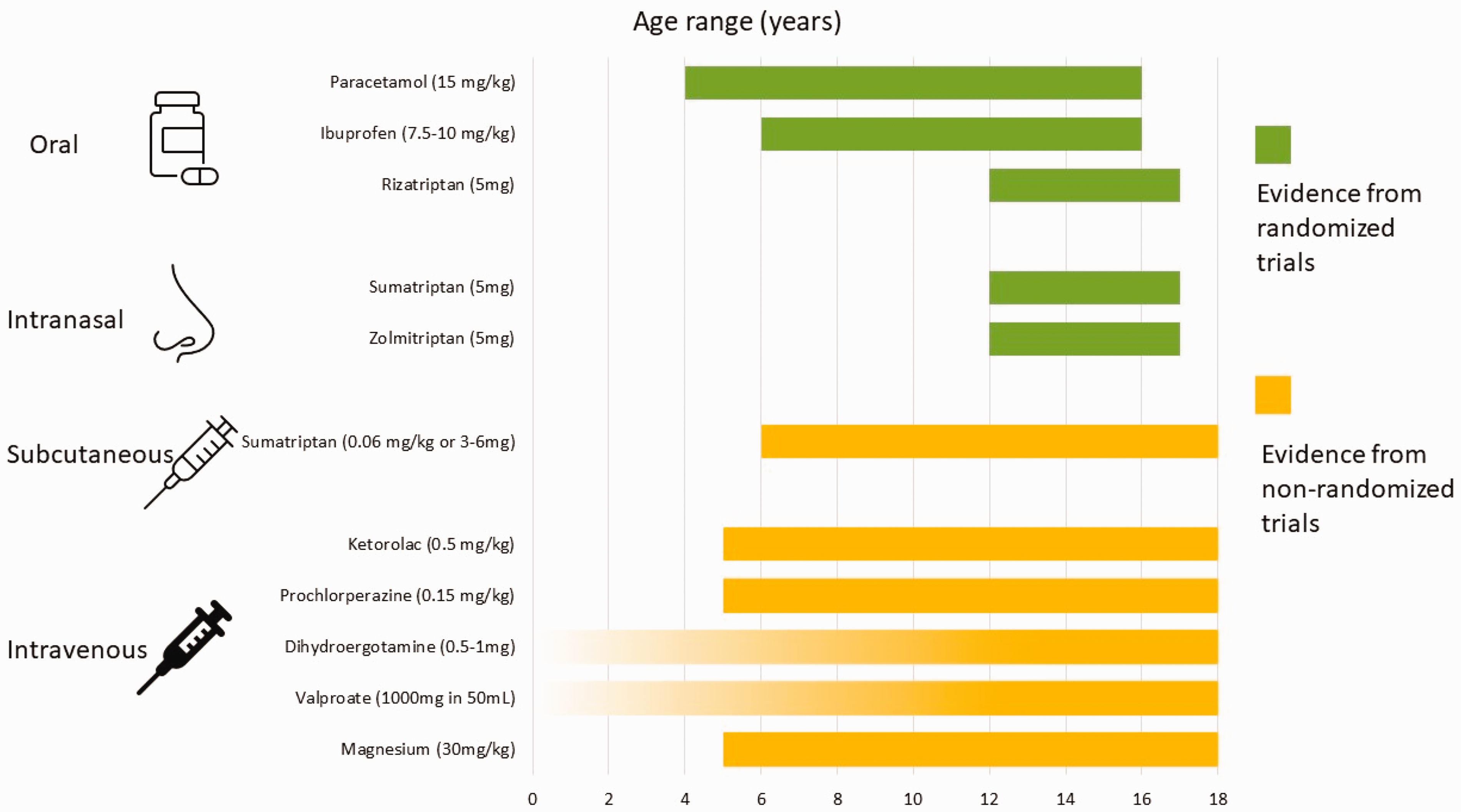
Summary of drugs recommended or suggested for the acute treatment of migraine in children and adolescents. For dihydroergotamine and valproate, the available studies did not specify a lower age limit; thus, the corresponding bars were represented as fading.
Triptans studies have shown conflicting results likely due to the high placebo response. Most triptans were not formally tested or approved in children. Rizatriptan was effective in adolescents (12–17 years) according to one study, but not in children (age <12 years) (218,219). Sumatriptan and zolmitriptan nasal sprays can be considered safe and effective options for adolescents. Combined treatment with sumatriptan plus naproxen has been evaluated in one placebo-controlled trial and was effective (220). This combination could be considered in adolescents (age 12–18 years) not responding to analgesics or to triptans alone.
Several acute pharmacologic treatments for migraine have been studied in non-randomised trials or in trials that did not have a placebo-controlled design and mostly refer to parenteral drugs used in an emergency care or inpatient setting to treat status migrainosus or attacks that are resistant to common medications. Although the quality of these studies is lower than that of randomized placebo-controlled trials, they are relevant for clinical practice.
A single-blind, parallel group randomized trial including 49 participants aged 5–17 years assessed the efficacy of the administration of intravenous fluids as an adjunct to medication. The trial did not find a significant effect of intravenous fluids on migraine (221). In an emergency setting, the administration of intravenous fluids can rapidly rehydrate subjects without provoking any relevant AE and can therefore be considered as an adjuvant to acute migraine treatment.
Parenteral administration of antiemetics can also provide relief especially in individuals with severe nausea and/or vomiting who did not respond to analgesics. Retrospective single-center studies with a limited number of participants showed the potential effectiveness of intravenous prochlorperazine for the acute treatment of migraine (222,223). A subsequent prospective, randomized, double-blind trial compared intravenous prochlorperazine (0.15 mg/kg; maximum 10 mg) with intravenous ketorolac (0.5 mg/kg; maximum 30 mg) for migraine treatment in people aged 5–18 years. The trial included 62 children and adolescents and found 50% pain relief after one hour in 84.2% of subjects treated with prochlorperazine and 55.2% of those treated with ketorolac (224). This study also showed the potential efficacy of intravenous ketorolac for migraine treatment. It lacked a placebo arm, however and therefore cannot be considered conclusive. The efficacy of intravenous metoclopramide in the acute treatment of migraine in children and adolescents has not been proven. A trial in adults showed that prochlorperazine is more effective than metoclopramide, even if both drugs were superior to placebo (160). For this reason, intravenous prochlorperazine should be considered the antiemetic of choice for pediatric migraine treatment in an emergency setting. Treatment with antiemetics in children and adolescents should be reserved to migraine attacks resistant to commonly used medications, as the use of antiemetics could be associated with extrapyramidal AEs such as dystonic reactions. Those events could be more frequent with prochlorperazine than with metoclopramide (225). Non-randomised studies suggest the potential effectiveness of other intravenous compounds including DHE (226), valproic acid (off-label) (227,228), and magnesium (229). As well as antiemetics, those compounds could be useful in the emergency setting and to manage status migrainosus (230,231).
Subcutaneous sumatriptan is a very effective treatment for migraine in adults, but in children this has been tested only in non-randomised, observational studies (232,233).
Among the guidelines and guidance documents reviewed, three explicitly address the topic of the acute treatment of migraine in children and adolescents (Online Supplementary Table 14S). Overall, the guidelines and recommendations agree that pharmacologic treatment should be reserved for children and adolescents who do not respond to non-pharmacologic measures and need rapid resolution of the most disabling migraine attacks. Paracetamol and ibuprofen are preferred by all guidelines and recommendations. Triptans could be considered with preference for nasal spray formulations.
Q15 – What drugs are preferred in people over 65 years of age with a migraine attack?
Recommendations
Optimal
In people over 65 years of age with normal liver function, we suggest paracetamol/acetaminophen as first line therapy. Combinations of paracetamol with caffeine can also be used, but caution is advised to avoid risks related to excessive caffeine use, including medication overuse headache and caffeine withdrawal headache.
As a second line option, we suggest acetylsalicylic acid and non-steroidal anti-inflammatory drugs with monitoring of potential adverse events related to gastrointestinal bleeding and renal and hepatic insufficiency.
In individuals without uncontrolled hypertension or serious cardiovascular or cerebrovascular disease, we suggest the use of triptans as a third line treatment option. Lasmiditan and gepants are alternative options for subjects with contraindications, or not responding, to triptans. When using lasmiditan, the subjects should be advised about the potential central side effects and risk of falls due to dizziness.
Adjunctive therapy with antiemetics can be helpful, with a preference for non-centrally acting options due to the increased risk for sedation and extrapyramidal side effects of centrally acting antiemetics.
Essential
In people with migraine over 65 years of age with normal liver function we suggest paracetamol/acetaminophen as first line therapy. Combinations of paracetamol with caffeine can also be used, but caution is advised to avoid risks related to excessive caffeine use, including medication overuse headache and caffeine withdrawal headache.
As second line option, we suggest acetylsalicylic acid or non-steroidal anti-inflammatory drugs with monitoring of potential adverse events related to gastrointestinal bleeding and renal and hepatic insufficiency.
In people without uncontrolled hypertension or serious cardiovascular or cerebrovascular disease, we suggest the use of triptans as third line treatment option.
If triptans alone are not effective, a combination of a triptan with a non-steroidal anti-inflammatory drug can be used.
Adjunctive therapy with antiemetics can be helpful, but caution should be applied due to the increased risk for sedation and extrapyramidal side effects.
Comment: In otherwise healthy people with migraine >65 years of age who have no known comorbidities that could specifically alter drug pharmacokinetics, recommendations can follow those of younger age groups, with closer monitoring of side effects.
Background
Even though migraine tends to remit with older age (188), it continues to have a one-year prevalence of between 3.0–10% in the elderly population (>65 years) (242,243). Given the worldwide increase in life expectancy, migraine in the elderly is destined to become a more prevalent public health issue. In general, advancing age occurs with an increased susceptibility to additional diseases. Therefore, management of migraine in the elderly is likely to be influenced by other health problems and consequent association with polypharmacy (244). In addition, general physiological changes such as slowing of gastric emptying, changes in rates of liver metabolism of drugs, and reduced renal mass and glomerular filtration rate which occur with age can directly impact pharmacokinetics and pharmacodynamics (245).
Evidence
In the majority of migraine treatment trials, subjects over 60–65 years old have been excluded (246), and thus little rigorous evidence exists in this population. More recent studies on new acute treatments (gepants, ditans) did not exclude older individuals (247). According to general expert consensus, however, in the absence of contraindications, triptans and other acute treatments should not be withheld simply due to age (246). The statements regarding this population in the guidelines and guidance documents reviewed are summarized in Online Supplementary Table 15S.
Paracetamol/acetaminophen is generally considered the first-line option: liver function monitoring is recommended in elderly individuals, especially at higher doses, given the risk for hepatic insufficiency (248–252). The suggested daily dose is <3000 mg per day. In subjects with renal or hepatic dysfunction, it is advisable to reduce the dose by 50–75% (246,249).
Acetylsalicylic acid and NSAIDs should be used with caution in this population given the increased risk for NSAID-induced hypertension, renal impairment, upper gastrointestinal bleeding and ulceration, particularly among more elderly individuals and those with a history of peptic ulcer disease or gastrointestinal bleeding (245,249). Concurrent proton pump inhibitor use (251) and monitoring of renal and hepatic function can help minimize these AEs (248), noting that the regular intake of proton pump inhibitors has been linked to headache worsening (253). Frequent intake of NSAIDs requires intermittent monitoring of kidney function, as well as following any harmful effects on the gastro-intestinal and cardiovascular systems (248,252).
Antiemetic dopamine receptor antagonists may be helpful adjunctive treatments and should be used with caution in the elderly, given the increased risk of anticholinergic effects, extrapyramidal effects, drowsiness, dizziness, and additive sedation with other depressants of the central nervous system (252,254). They should be avoided in people with Parkinson’s disease (250). Domperidone does not cross the blood-brain barrier and therefore is not associated with extrapyramidal AEs or other central effects (252).
The use of ergotamine derivatives and triptans can be limited by their vasoconstrictive effects (254,255). Triptans can be used in elderly people without medical contraindications (uncontrolled hypertension, cardiovascular, cerebrovascular, or peripheral vascular disease) (248,256). The recent Guidelines of the European Society of Hypertension, endorsed by the International Society of Hypertension, provide a clear definition of optimal and normal blood pressure, as well as of different grades of hypertension (257). In the absence of specific evidence, and considering the increase in blood pressure associated with the intake of triptans (0–10 mmHg), a reasonable approach is to use triptans in elderly subjects falling in the optimal (systolic blood pressure <120 mmHg and diastolic blood pressure <80 mmHg) or normal (systolic blood pressure <129 mmHg and diastolic blood pressure <85 mmHg) blood pressure category, with/without anti-hypertensive treatment. Age itself is not a contraindication to triptan use, however, age and vascular risk factors should be considered before prescribing triptans in people older than 65 years. There is no evidence from clinical experience that triptans are less safe after age 65 when prescribed appropriately (256).
Lasmiditan does not cause vasoconstriction and therefore can be used in elderly people with vascular disease or risk factors (250). It should, however, be used with caution as there is potential for side effects related to its central effects, including drowsiness, dizziness and fatigue (75). Clinical studies of lasmiditan did not exclude people with migraine based on the upper limit of age: about 4% of subjects were ≥65 years. There was an increase in dizziness and paresthesia in older adults. Side effects were similar regardless of uncontrolled hypertension, coronary artery disease, or arrhythmia (76,77). Driving a motor vehicle or operating machinery is discouraged until eight hours after taking lasmiditan (258).
Gepants do not have vasoconstrictive activity and could be a safer option in the elderly including people with cardiovascular disease or risk (250), although they may impair vasodilation or have effects on blood pressure. They can be used in older adults, when appropriate (254). A clinical trial examining rimegepant for the acute treatment of migraine did not exclude older subjects (259). A post hoc analysis comparing the safety and tolerability of rimegepant between younger (aged ≥18 and ≤45 years) and older participants (aged ≥65 years) found that rimegepant was equally well tolerated in both cohorts and that the pharmacokinetics did not differ significantly between older and younger individuals (260). Clinical trials examining ubrogepant for the acute treatment of migraine included participants up to age 75 years and reported no clinically significant pharmacokinetic differences between older and younger subjects (84); these clinical studies did not include sufficient numbers of individuals ≥65 years of age to determine if they respond differently (247). Ubrogepant should be avoided in people with migraine with end-stage renal disease and the dose should be adapted in individuals with severe hepatic or renal impairment. Ubrogepant and rimegepant interact with strong CYP34A inducers and concomitant use should be avoided (247).
Patients should be encouraged to identify and avoid triggers (252) which can include irregular lifestyle (e.g. poor sleep patterns or irregular food intake) and the intake of triggering food items, although there is no clear evidence for this (25).
Q16 – What is the recommended approach to the acute treatment of migraine in people with a history of stroke, other vascular diseases or uncontrolled hypertension?
Recommendations
Optimal
In people with an acute migraine attack who have a history of stroke, cardiovascular diseases or uncontrolled hypertension, we suggest paracetamol as first line treatment, with lasmiditan or gepants as second line options.
Non-steroidal anti-inflammatory drugs can be used, but administration should be limited considering the concomitant use of antithrombotic therapy.
Adjunctive therapy with antiemetics can be helpful.
Triptans can be used with caution if the above conditions are under control in people with migraine who did not benefit from paracetamol, lasmiditan or gepants.
Dihydroergotamine and ergotamine should be avoided at all times.
Essential
In people with an acute migraine attack who have a history of stroke, cardiovascular diseases or uncontrolled hypertension, we suggest paracetamol as first line treatment, and non-steroidal anti-inflammatory drugs as a second line option. The administration of non-steroidal anti-inflammatory drugs should be limited, however, considering the concomitant use of antithrombotic therapy.
Adjunctive therapy with antiemetics can be helpful.
Available triptans can be used with caution if the above conditions are under control in individuals who did not benefit from paracetamol.
Dihydroergotamine and ergotamine should be avoided at all times.
Background
There are several types of cardiovascular diseases (CVDs) including atherosclerosis, peripheral artery disease, coronary artery disease, cerebrovascular disease, aneurysms, venous thromboembolism, and systemic hypertension. Over the last four decades, numerous studies have demonstrated a connection between primary headaches and conditions such as stroke or ischemic heart disease, with a pooled prevalence among the several studies available of 5% (95% confidence interval (CI) 3–7%) for stroke and 7% (95%CI 5–10%) for ischemic heart disease, which are higher in people suffering from primary headache than the general population. Hypertension is also highly prevalent in poeple living with headache (24% [95%CI 22–26%]), atrial fibrillation and flutter (2% [95%CI 1–3%]) as well as other CVDs (9% [95%CI 7–11%]) (261). It is important to consider that certain drugs commonly used to treat migraine attacks may have potential impacts on vascular functions, as they can induce vasoconstriction or increase blood pressure (262), which should be taken into account when managing individuals with significant vascular diseases.
Evidence
Oral acetaminophen/paracetamol represents a safe option in CVD people living with migraine even though intravenous paracetamol is well-documented to cause hypotension and special attention is warranted (263).
Frequent intake of NSAIDs is associated with an increased risk of bleeding, mostly gastrointestinal, because they can inhibit platelet aggregation and secretion by inhibiting cyclo-oxygenase 1 (COX-1) (264). Although NSAIDs use is generally discouraged in individuals with cardiovascular diseases (265), their use may be considered when no other pharmacologic options are available. This should be done only after a thorough evaluation of the potential risks involved. Several special conditions should be considered, in particular:
Long-term antithrombotic therapy is commonly prescribed in subjects with CVDs. In this setting, NSAID therapy further increases the risk of gastrointestinal bleeding and therefore the use of proton pump inhibitors is recommended (266). It is important to counsel all people who are prescribed NSAIDs along with one or more antithrombotic agents about the potential negative interaction. In people receiving antithrombotic drugs, such as clopidogrel, ticagrelor, and cilostazol, the intake of NSAIDs increases the risk of bleeding;
Concurrent use of NSAIDs (mainly ibuprofen but also naproxen) may, however, inhibit the antiplatelet effect of aspirin (267), which is commonly prescribed at low doses for primary and secondary prevention of CVDs and therefore increase the risk of ischemic events;
Among people receiving antithrombotic therapy, the frequent use of NSAIDs is associated with an increased risk of bleeding and increased thrombotic events, even after short-term treatment. Physicians should therefore exercise appropriate caution when prescribing NSAIDs, preferably avoiding them in subjects who have recently experienced myocardial infarction or stroke (268);
Finally, NSAIDs can increase blood pressure (BP) or interfere with BP control (269,270). There are differences between nonselective NSAIDs with regard to their effect on BP. Indomethacin, naproxen, and ibuprofen have been associated with clinically significant changes in BP and a higher incidence of new-onset hypertension, albeit only if taken on a daily basis (271). We therefore advise against their use in people with hypertension or severe CVDs.
Among the migraine-specific abortive medication available, triptans have significantly changed the treatment landscape for migraines over the past 30 years (57). These drugs are potent agonists of the 5-HT1B/1D receptor and, due to their activity on the 5-HT1B subtype, can be considered vasoconstrictive agents (272). Widespread concern regarding their potential cardiovascular side effects has limited their use in subjects with cardiovascular or cerebrovascular disease, despite decades of real-life experience showing their safety and effectiveness in people with vascular diseases (273). Moreover, a systematic review of the most relevant observational studies published in 2015 (42) found no association between triptan use and risk of cardiovascular events. This is due to the pathophysiology of vascular diseases, in which the vasoconstrictive mechanism plays a minor role (274). Additionally, a cohort study conducted by Hall et al. in 2004, which included 63,575 subjects with migraine, 13,664 of whom treated with a triptan, did not find any connection between triptan prescription and stroke, other cardiovascular incidents, or mortality (275). It should be noted, however, that the subjects who received triptans were classified as having a low cardio or cerebrovascular risk, which limits the certainty of these findings.
As summarized in Online Supplementary Table 16S, existing guidelines and guidance documents recommend against triptan use in subjects with hemiplegic migraine, basilar migraine, recent ischemic stroke, ischemic heart disease, vasospastic angina, and uncontrolled hypertension. The lack of evidence that triptans increase the risk of heart attacks or stroke should be acknowledged (276).
DHE is an ergot alkaloid that acts on serotonergic (5HT-1A, -1B, 1F, -2A, and -2C), dopaminergic (DA-1 and -2) and adrenergic (alpha-1 and -2) receptors. DHE has a prominent vasoconstriction effect and affects extracranial vessels more than the intracranial ones, with no significant effects on cerebral blood flow (277). Intense consumption of ergot derivatives may be associated with an increased risk of serious ischemic complications including ergotism and therefore DHE is contraindicated in individuals with CVDs (278).
Lasmiditan is a selective serotonin 5H-1F receptor agonist, part of the ditan class. It has been shown to be effective in the acute treatment of migraine attacks, even in people with cardiovascular risk factors (279). Available evidence suggests that lasmiditan has a low risk of cardiovascular side effects, due to its lack of vasoconstrictive properties (280) and can thus be prescribed in individuals with comorbid CVDs (281).
Due to its vasodilating properties, calcitonin gene-related peptide (CGRP) plays an important role not only in migraine but also in physiological and pathological cardiovascular conditions. Antagonising the CGRP receptor for the acute treatment of migraine is now possible using second and third generation gepants, such as ubrogepant, rimegepant, and zavegepant (282). Specifically, gepants have demonstrated efficacy in relieving migraine in clinical trials without causing direct vasoconstriction, and there is no contraindication in their labels to use in people with stroke or myocardial infarction (283,284). Consequently, at this time, there is insufficient evidence that gepants should be avoided in those with CVDs including stroke or myocardial infarction. Vasodilation may be an important CGRP-mediated mechanism of ischemia, especially in people with small vessel disease, therefore CGRP antagonists should be used with caution in this context (285).
Guidelines have started to include comments on gepant use including those released by the American Headache Society or the Taiwan Headache Society, which highlighted that gepants do not cause vasoconstriction, hence they could be used in the subjects with cardiovascular contraindications to triptans. The German Headache Society is taking the contrary position and advising caution in their use. Overall, it is wise to carefully consider the use of gepants in people with a high risk of vascular accident or who are already presenting symptoms of vascular impairment, Raynaud phenomenon or small vessel disease (286).
Q17 – What are possible treatment approaches to menstrual migraine?
Recommendations
Optimal
In women with menstrual migraine we suggest non-steroidal anti-inflammatory drugs or triptans as first-line drugs. The combination of triptans with non-steroidal anti-inflammatory drugs, or triptans with antiemetics, as well as of non-steroidal anti-inflammatory drugs with antiemetics can be used in case of failure of individual drugs.
Lasmiditan and gepants may represent an additional option to consider.
If these options are not successful, we suggest short-term prevention* with naproxen or frovatriptan in women with regular cycles.
When all of the above are ineffective, hormonal treatment with a continuous regimen of combined hormonal contraceptives or progesterone-only contraceptives can be considered in migraine without aura, as well as a lower threshold of monthly headache days to start regular preventive treatment.
Essential
In women with menstrual migraine we suggest non-steroidal anti-inflammatory drugs or triptans as first-line drugs. The combination of triptans with non-steroidal anti-inflammatory drugs, or triptans with antiemetics, as well as of non-steroidal anti-inflammatory drugs with antiemetics can be used in case of failure of individual drugs.
In case of failure of the above, in women with regular cycles, we suggest short-term prevention* with naproxen or, if available, frovatriptan.
When all of the above are ineffective, hormonal treatment with a continuous regimen of combined hormonal contraceptives or progesterone-only contraceptives (if available) can be considered in migraine without aura, as well as a lower threshold to start regular preventive treatment.
*Short-term prevention should be started 2–3 days prior to the first day of menses and continued during menstruation.
Background
Menstrual migraine includes pure menstrual migraine (MM) and menstrually related migraine (MRM), which may occur with or without aura. Changes in hormone levels, typically oestrogens, are associated with both MM (occurring exclusively on day 1 ± 2 [i.e., days −2 to +3 of menstruation] in at least two out of three menstrual cycles and at no other times of the cycle) and MRM (occurring on day 1 ± 2 [i.e., days −2 to +3] of menstruation in at least two out of three menstrual cycles, and additionally at other times of the cycle) (6,287). MM most frequently occurs in the second decade of life around the onset of menarche but may change over a woman’s reproductive cycle. MM prevalence varies from 4% to 70% of women with migraine, peaking around 40 years and declining towards menopause (288).
MM tends to be more severe (with associated nausea and vomiting, photophobia and phonophobia), longer lasting, more disabling and refractory to treatment than non-MM attacks (289). A headache diary is important in the diagnosis of MM. Prospective documentation of headache days, the severity of attacks, and the relationship of headaches to menses is critical. Acute treatment has proven effective in randomized clinical trials. In women with regular menstrual periods, the short-term preventive treatment is a reasonable approach (see below) (290).
Evidence
Pharmacologic therapy
Acute treatment options used for all types of migraines can also be used to treat MM. Barring contraindications, most people should be prescribed migraine-specific agents (triptans) as first-line therapy (290). Other treatment options include various analgesics and antiemetic agents, NSAIDs, COX-2 inhibitors and ergot derivatives, and some guidelines include them all (291). Combination drugs could be used for those in whom monotherapy does not work effectively.
Several studies evaluating triptans for the acute treatment of MM showed good evidence for sumatriptan and combination sumatriptan-naproxen (85–500 mg), zolmitriptan, rizatriptan, frovatriptan, or almotriptan with pain relief at 1–4 hours and a responder rate ranging from 30% to 81% (289). Various triptans can be recommended for treating MM attacks, particularly long-lasting triptans such as frovatriptan and naratriptan (292–294). These can also be prescribed as a short-term prevention, to be started two days before the expected first day of menstruation and continued for five days (188).
Combination therapies should also be recommended due to their good response, high tolerance and accessibility. Common combinations of drugs include a triptan with an NSAID (such as sumatriptan 85 mg and naproxen sodium 500 mg), or an NSAID or acetaminophen with metoclopramide. DHE 2 mg nasal spray may also be offered (295–298).
A post-hoc analysis of two RCTs found that treatment of perimenstrual migraine attacks with lasmiditan was associated with freedom from migraine pain at two hours, early onset of efficacy, and sustained efficacy. Lasmiditan 200 mg produced the best effect compared to other doses, particularly among subjects with migraine with cardiovascular or asymptomatic cerebrovascular disease (299). Ditans exert central inhibitory effects, which may impair the patient’s ability to assess their driving competence and the extent of drug-induced harm. Patients should be advised to refrain from driving for at least eight hours after taking the medication (40).
Short-term prevention of menstrual migraine
Women suffering from MM who do not receive a complete benefit from abortive therapies may be candidates for preventive treatment. Women with a regular menstrual cycle and MM may benefit from short-term prevention with drugs administered a few days before the start of, and continuing through, menses. Naproxen sodium proved superior to placebo in reducing the impact of MM in two RCTs (300,301). Mefenamic acid (500 mg three times daily) may be considered for the treatment of MM (302). Gastrointestinal side effects may limit NSAID use in some subjects.
Triptans have been used for the short-term prevention of menstrual migraine but are not approved for the indication. Naratriptan (1 mg bid per day for five days starting two days prior to the expected onset of menses) and frovatriptan (2.5 mg bid given for six days perimenstrually) showed superiority over placebo (26,293,303–305). There is also some evidence for oestrogen replacement therapy using, for instance, transdermal estradiol (<100 ug given for six days perimenstrually as a gel or a patch) (306,307). The evidence is however inconclusive as another study failed to detect efficacy of hormone replacement with respect to attack frequency during the whole menstrual cycle (308).
The recommendations and statements addressing the treatment of MM in the guidelines and guidance documents assessed in this manuscript are reported in Online Supplementary Table 17S.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024241252666 - Supplemental material for International Headache Society global practice recommendations for the acute pharmacological treatment of migraine
Supplemental material, sj-pdf-1-cep-10.1177_03331024241252666 for International Headache Society global practice recommendations for the acute pharmacological treatment of migraine by Francesca Puledda, Simona Sacco, Hans-Christoph Diener, Messoud Ashina, Haidar M. Al-Khazali, Sait Ashina, Rami Burstein, Eric Liebler, Andrea Cipriani, Min Kyung Chu, Alexandra Cocores, Freda Dodd-Glover, Esme Ekizoğlu, David Garcia-Azorin, Carl Göbel, Maria Teresa Goicochea, Amr Hassan, Koichi Hirata, Jan Hoffmann, Bronwyn Jenkins, Katharina Kamm, Mi Ji Lee, Yu-Hsiang Ling, Marco Lisicki, Daniele Martinelli, Teshamae S. Monteith, Raffaele Ornello, Aynur Ozge, Mario Peres, Patricia Pozo-Rosich, Volodymyr Romanenko, Todd J. Schwedt, Marcio Nattan P. Souza, Tsubasa Takizawa, Gisela M. Terwindt, Janu Thuraiaiyah, Mansoureh Togha, Nicolas Vandenbussche, Shuu-Jiun Wang, Shengyuan Yu and Cristina Tassorelli in Cephalalgia
Supplemental Material
sj-pdf-2-cep-10.1177_03331024241252666 - Supplemental material for International Headache Society global practice recommendations for the acute pharmacological treatment of migraine
Supplemental material, sj-pdf-2-cep-10.1177_03331024241252666 for International Headache Society global practice recommendations for the acute pharmacological treatment of migraine by Francesca Puledda, Simona Sacco, Hans-Christoph Diener, Messoud Ashina, Haidar M. Al-Khazali, Sait Ashina, Rami Burstein, Eric Liebler, Andrea Cipriani, Min Kyung Chu, Alexandra Cocores, Freda Dodd-Glover, Esme Ekizoğlu, David Garcia-Azorin, Carl Göbel, Maria Teresa Goicochea, Amr Hassan, Koichi Hirata, Jan Hoffmann, Bronwyn Jenkins, Katharina Kamm, Mi Ji Lee, Yu-Hsiang Ling, Marco Lisicki, Daniele Martinelli, Teshamae S. Monteith, Raffaele Ornello, Aynur Ozge, Mario Peres, Patricia Pozo-Rosich, Volodymyr Romanenko, Todd J. Schwedt, Marcio Nattan P. Souza, Tsubasa Takizawa, Gisela M. Terwindt, Janu Thuraiaiyah, Mansoureh Togha, Nicolas Vandenbussche, Shuu-Jiun Wang, Shengyuan Yu and Cristina Tassorelli in Cephalalgia
Supplemental Material
sj-pdf-3-cep-10.1177_03331024241252666 - Supplemental material for International Headache Society global practice recommendations for the acute pharmacological treatment of migraine
Supplemental material, sj-pdf-3-cep-10.1177_03331024241252666 for International Headache Society global practice recommendations for the acute pharmacological treatment of migraine by Francesca Puledda, Simona Sacco, Hans-Christoph Diener, Messoud Ashina, Haidar M. Al-Khazali, Sait Ashina, Rami Burstein, Eric Liebler, Andrea Cipriani, Min Kyung Chu, Alexandra Cocores, Freda Dodd-Glover, Esme Ekizoğlu, David Garcia-Azorin, Carl Göbel, Maria Teresa Goicochea, Amr Hassan, Koichi Hirata, Jan Hoffmann, Bronwyn Jenkins, Katharina Kamm, Mi Ji Lee, Yu-Hsiang Ling, Marco Lisicki, Daniele Martinelli, Teshamae S. Monteith, Raffaele Ornello, Aynur Ozge, Mario Peres, Patricia Pozo-Rosich, Volodymyr Romanenko, Todd J. Schwedt, Marcio Nattan P. Souza, Tsubasa Takizawa, Gisela M. Terwindt, Janu Thuraiaiyah, Mansoureh Togha, Nicolas Vandenbussche, Shuu-Jiun Wang, Shengyuan Yu and Cristina Tassorelli in Cephalalgia
Footnotes
Authors’ Note
Please note that the reference list also includes material cited within the Supplemental Material.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:
Francesca Puledda declares speaker and consultancy fees from TEVA.
Simona Sacco declares grants/contracts, consulting fees or honoraria from Novartis, Uriach, Allergan-AbbVie, Teva, Lilly, Lundbeck, Pfizer, NovoNordisk, Abbott, AstraZeneca, Pfizer.
Hans-Christoph Diener received honoraria for participation in clinical trials, contribution to advisory boards or oral presentations from: AbbVie, Lilly, Lundbeck, Novartis, Pfizer, Teva and WebMD. The German Research Council (DFG) supports headache research by HCD. HCD serves on the editorial boards of Cephalalgia, Lancet Neurology and Drugs.
Messoud Ashina is a consultant, speaker, or scientific advisor for AbbVie, Amgen, Astra Zeneca, Eli Lilly, GlaxoSmithKline, Lundbeck, Novartis, Pfizer, and Teva; a primary investigator for ongoing AbbVie and Pfizer trials; and is the past president of the International Headache Society. MA is supported through the Lundbeck Foundation Professor Grant (R310-2018-3711) and received institutional research grant from Lundbeck and Novartis. MA serves as associate editor of the Journal of Headache and Pain, and associate editor of Brain.
Sait Ashina has received honoraria for consulting from Allergan/AbbVie, Eli Lilly, Impel NeuroPharma, Linpharma, Lundbeck, Pfizer, Satsuma, Teva, Theranica. SA is an Associate Editor for Neurology Reviews, BMC Neurology, and Frontiers in Neurology, and serves on the Editorial Board for the Journal of Headache and Pain. SA serves as a Trustee on the Board of the International Headache Society and is a member of the Education Committee of the International Headache Society.
Eric Liebler declares consulting fees from ElecroCore, Inc.
Andrea Cipriani is supported by the National Institute for Health Research (NIHR) Oxford Cognitive Health Clinical Research Facility, by an NIHR Research Professorship (grant RP-2017-08-ST2-006), by the NIHR Oxford and Thames Valley Applied Research Collaboration and by the NIHR Oxford Health Biomedical Research Centre (grant NIHR203316). The views expressed are those of the authors and not necessarily those of the UK National Health Service, the NIHR, or the UK Department of Health.
Min Kyung Chu was a site investigator for a multicenter trial sponsored by Biohaven Pharmaceuticals, Allergan Korea, and Ildong Pharmaceutical Company. He received lecture honoraria from Eli Lilly and Company, Handok-Teva, and Ildong Pharmaceutical Company over the past 24 months. He received grants from Yonsei University College of Medicine (6-2021-0229), the Korea Health Industry Development Institute (KHIDI) (Grant No.: HV22C0106), and a National Research Foundation of Korea (NRF) grant.
Alexandra Cocores declares grants/contracts from AbbVie, Eli Lilly, Rehaler.
Esme Ekizoğlu declares honoraria from Allergan‐AbbVie.
David Garcia-Azorin declares grants/contracts from Carlos II Research and Castilla and Leon Regional Health, consulting fees from Eli-Lilly, WHO, Lundbeck or honoraria from AbbVie, Teva, Novartis, Pfizer.
Carl Göbel declares honoraria from Fielmann AG and German Society for the Study of Pain.
Maria Teresa Goicochea, personal fees as speaker or advisory board and/or non-financial support from Pfizer, AbbVie and Teva.
Amr Hassan declares consulting fees or honoraria from Novartis, Sanofi Genzyme, Biologix, Merck, Hikma Pharma, Janssen, Inspire Pharma, Future Pharma, Elixir pharma, Allergan, Merck, Biologix, Roche, Bayer, Chemipharm, Al Andalus, and Clavita pharm.
Koichi Hirata declares consulting fees from Otsuka Pharma, and honoraria from Amgen Astellas BioPharma KK, Daiichi Sankyo, Eisai, Eli Lilly Japan KK, MSD, Otsuka Pharma, Pfizer Japan, and Sawai Pharma.
Jan Hoffmann declares grants/contracts from Bristol Myers Squibb, IHS, National Institute of Health and Care Research (NIHR), Medical Research Council (MRC) and Migraine Trust, consulting fees or honoraria from AbbVie, Cannovex, Eli Lilly, Novartis, Teva, Allergan, Chordate Medical AB, Lundbeck, Sanofi, MD Horizonte, Pfizer, Sage, NEJM Journal Watch Neurology, Oxford University Press, Quintessence Publishing, Springer Healthcare. He holds stock options from Chordate Medical AB.
Bronwyn Jenkins declares honoraria from Allergan/AbbVie, Eli-Lilly, Novartis, Pfizer, Teva, Viatris.
Katharina Kamm declares grants/contracts from the German Headache and Migraine Society, Novartis, consulting fees from Hormosan, and honoraria from Lundbeck, TEVA, derCampus, Novartis.
Mi Ji Lee declares onsulting fees or honoraria from Eli-Lilly, Lundbeck, Pfizer, NuEyne Co, Teva AbbVie, SK Chemical, CKD Pharma, and YuYu.
Marco Lisicki declares honoraria from Teva, AbbVie, Novartis, Pfizer and Allergan.
Daniele Martinelli declares honoraria from AbbVie and Lundbeck.
Teshamae S. Monteith has the following disclosures over the past 3 years: clinical trial site principal investigator for studies sponsored by Eli Lilly and AbbVie (all paid to the institution), participation in an advisory board/consultancy for AbbVie, Teva, Linpharma, e-Neura, Novartis, Merz, and Pfizer, Educational grant from Amgen and AbbVie, personal fees from Medscape, Massachusetts Medical Society, American Headache Society, American Academy of Neurology, Neurodiem, Academic CME, AbbVie, Novartis, unpaid co-author for research funded by AbbVie, Pfizer/Biohaven, and Theranica. She is an associate editor for Cephalalgia and Continuum Audio, and is on the editorial board for Neurology, American Migraine Foundation, and Brain and Life Magazine. TSM has provided unpaid service on the board of directors for the International Headache Society (2021–2023) and currently on the executive board for the Florida Society of Neurology.
Raffaele Ornello declares grants/contracts from Novartis, consulting fees/honoraria from Eli Lilly, AbbVie, Pfizer, and Teva.
Aynur Ozge declares consulting fees or honoraria from AbbVie Lilly, Teva, Abdi Ibrahim, İlko and Drogsan.
Mario Peres accepted honoraria for lectures, presentations, speakers bureaus, manuscript writing and educational events from Pfizer, Teva, AbbVie-Allergan, Lundbeck. Accepted travel support from Teva. Participated in advisory boards, AbbVie-Allergan, Eli Lilly, Pfizer, Kenvue, Eurofarma, Sanofi-Aventis. Mario Peres is president of ABRACES.
Patricia Pozo-Rosich declares grants/contracts from ERANet Neuron, Instituto Salud Carlos III, Novartis, Teva and AbbVie and consulting fees or honoraria from AbbVie, Almirall, Eli Lilly, Lundbeck Medscape, Novartis, Pfizer, Teva.
Todd J. Schwedt declares grants/contracts with AHA, Amgen, Henry Jackson Foundation, NIH, Patient Centered Outcomes Research Institute, Pfizer, Spark Neuro, US Dept. of Defense, AMF, NHF and Mayo Clinic with consulting fees from AbbVie, Allergan, Amgen, Axsome, Biodelivery Science, Biohaven, Click Therapeutics, Collegium, Eli Lilly, Equinox, Ipsen, Unpharma, Lundbeck, Novartis, Satsuma, Scilex, Theranica, and Tonix.
Marcio Nattan P. Souza declares consulting fees or honoraria from Teva, Libbs, Pfizer, Sandoz, Lundbeck, Allergan/AbbVie, Lilly.
Tsubasa Takizawa declares grants/research funds from Eli Lilly, Pfizer and Tsumura and advisory/consulting fees and/or honoraria from Eli Lilly, Daiichi Sankyo, Otsuka, Amgen, Teijin, Pfizer, Kyowa Kirin, Elsai, Kowa, UCB Japan, Takeda, and Santen Pharma.
Gisela M. Terwindt declares grants/contracts from European Community, Dutch Heart Foundation, IRRF, Dioraphte and Dutch Brain Council with consulting fees or honoraria from Novartis, Lilly, Teva, AbbVie/Allergan, Lundbeck, Pfizer, Spring Media, Ashfield MedComms, Remedica Cygnea.
Nicolas Vandenbussche declares consulting fees or honoraria Pfizer, AbbVie, Novartis, Teva, Babinski VZW, ACREHAB UGent.
Shuu-Jiun Wang declares honoraria from AbbVie, Pfizer and Biogen.
Cristina Tassorelli declares grants/contracts from AbbVie with consulting fees or honoraria from Eli Lilly, Dompé, Teva, Lundbeck, Pfizer, Medscape.
All remaining authors report no disclosures or conflicts of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
