Abstract
Background
Real-world data on the effectiveness and safety of lasmiditan, a new medication for acute migraine attacks, is necessary.
Methods
We performed a prospective, observational, multi-center, real-world study. A total of 48 patients with migraine (44 females, 44.6 ± 12.9 years old) were included in this study.
Results
Twenty-three patients (47.9%) reported they were headache-free two hours after taking lasmiditan and were categorized into the responder group. In total, 44 patients (91.7%) experienced at least one side effect within two hours of taking the medication. Dizziness, somnolence, malaise, nausea, and palpitations were reported by 56.3% (n = 27), 45.8% (n = 22), 37.5% (n = 18), 20.8% (n = 10), and 14.6% (n = 7) of patients respectively. Of 48 patients, 20 (41.7%) indicated that they preferred lasmiditan to their previous acute treatment. There were no predictive factors for efficacy.
Conclusion
This real-world study demonstrated the efficacy and safety of lasmiditan. More than 90% of patients experienced side effects from lasmiditan. Approximately 40% of patients preferred lasmiditan despite the occurrence of side effects.
Introduction
Migraine is a disease with a high burden, and its prevalence in Japan ranges from 6.0–8.4% (1,2). Treatment of migraine can be divided into two categories: acute treatment and prophylactic therapy. Although many acute treatments are available, it has been reported that 35–40% of cases do not fully respond to acute treatment (3).
Lasmiditan is a selective 5-HT1F serotonin receptor agonist and a new class of acute treatment for migraine attacks (4). Based on the results of the phase 3 SAMURAI and SPARTAN trials conducted in the USA and EU, the US Food and Drug Administration has approved lasmiditan for the acute treatment of migraine headaches (5,6). The MONONOFU trial was conducted in Japan and reported the efficacy and safety of lasmiditan for migraine patients (7). At two hours (2 h) postdose, a significantly higher proportion of participants were headache pain free in the lasmiditan 100 mg groups (32.4%, 67/207; odds ratio 2.41 [1.51 to 3.83]; p < 0.001) compared to the placebo group (16.6%, 35/211). The most common treatment-emergent adverse events (TEAEs) were dizziness (38.0%; 79/208), somnolence (21.2%; 44/208), and malaise (11.1%; 23/208) in 100 mg lasmiditan groups, with no serious adverse events reported. As a result, 50 mg, 100 mg, and 200 mg dosages of lasmiditan became available in Japan in January 2022.
Lasmiditan is a drug with a relatively high rate of reported side effects (8), yet there is a paucity of literature examining the predictive factors of increased side effect burden (8,9). Dizziness is a commonly reported side effect of lasmiditan, and it has been associated with the predictive factors of higher lasmiditan dosage, being of non-Hispanic ethnicity, lower patient body mass index (BMI), and the migraine attack treated being of mild or moderate severity (10). It has been reported that the incidence of lasmiditan's side effects decreases with repeated dosing (11). We hypothesized that despite the potential for increased side effects from lasmiditan, benefits related to its efficacy might outweigh its drawbacks, leading to a preference among patients for its use.
The objectives of this study are as follows: firstly, to evaluate the safety and efficacy of lasmiditan using real-world data from a patient cohort outside of a clinical trial; secondly, to identify the predictors of lasmiditan's efficacy; and thirdly, to examine factors contributing to patient preference for lasmiditan.
Methods
Study design and setting
This is a prospective, observational, multi-center, real-world study. The Ethical Review Board of Kyoto Prefectural University of Medicine approved this study (Approval No. ERB-C-2608-1), and each of the participating four sites’ ethical review boards or ethics committees also endorsed the study. The participants were enrolled at University Hospital Kyoto Prefectural University of Medicine, Kyoto Okamoto Memorial Hospital, Saiseikai Shiga Hospital, and Keio University Hospital in Japan between October 2022 and May 2023.
Patients with migraine who visited participating study sites and were prescribed 100 mg of lasmiditan as part of their migraine treatment were recruited. Participants were 20 years old or older, received a prior oral explanation, and provided written consent before enrollment. Of the 72 migraineurs who agreed to participate, 48 completed the study by submitting questionnaires during the observation period.
Diagnosis of migraine
Diagnoses of migraine with or without aura were made by headache specialists from each study site, following The International Classification of Headache Disorders, 3rd edition criteria (12).
Clinical evaluation and data extraction
Patients who agreed to participate in this study were prescribed 100 mg of lasmiditan and were given a paper questionnaire. Patients were instructed to take lasmiditan when headache attacks occurred. The questionnaire collected information on each patient's first dose of lasmiditan for analysis. The questionnaires were filled out by the patients and collected at the follow-up visit. Information on demographic data (sex, age, height, and weight), history of migraine with or without aura, taking migraine prophylaxis, using anti-calcitonin gene-related peptide monoclonal antibody (anti-CGRP mAb) in the month prior to the medication, and previous acute medication for headache were extracted from questionnaire data and/or medical charts. Monthly headache days (MHD) were extracted from medical charts or headache diaries. In most cases, MHD is noted based on information from headache diaries, but in the case of patients who refuse to keep a headache diary, the physician notes MHD in the medical chart based on the patient's own report. The participants also reported their headache severity, associated headache symptoms, duration of time between headache onset and taking lasmiditan (at baseline/the time of pre-medication, and at 30/ 60/ 90/ 120 min after taking lasmiditan). Patients selected the severity of the headache from options ‘severe [3],’ ‘moderate [2],’ ‘mild [1],’ and ‘none [0].’ They also indicated the presence of associated headache symptoms: ‘nausea/vomiting,’ ‘photophobia,’ and ‘phonophobia.’ For side effects, the presence or absence of ‘dizziness,’ ‘somnolence,’ ‘malaise,’ ‘nausea,’ and ‘palpitations’ were selected for each time period (at baseline/the time of pre-medication, and 30/ 60/ 90/ 120 min after the medication). Although other side effects could be freely described at each time point, they were not included in this analysis because they were rarely described. Additionally, each patient reported their preference between lasmiditan and their previous acute medication, and indicated their justification among the following: ‘stronger efficacy,’ ‘longer duration of efficacy,’ ‘fewer side effects,’ and ‘lower cost.’ Participants were allowed to select multiple reasons for their medication preference.
Definition of responder
Based on the information on headache intensity at pre-medication and 120 min, we defined a responder as a patient whose headache severity became ‘none [0]’ at 120 min, per the Guidelines of the International Headache Society for Controlled Trials of Acute Treatment of Migraine Attacks in Adults (13). As a sensitivity analysis, we performed the same analysis while changing the definition of responder. As described in the Online Supplementary material (Supplemental Table 1), we defined the headache improvement group as patients who achieved pain relief defined as a reduction in self-reported headache severity from ‘severe [3]’ or ‘moderate [2]’ at baseline, to ‘mild [1]’ or ‘none [0]’, or patients who achieved a reduction in headache severity from ‘mild [1]’ at baseline to ‘none [0]’.
Statistical analysis
Descriptive statistics are presented as mean (standard deviation [SD]) or number (percentage). Patients’ characteristics and other variables were compared by logistic regression analysis models adjusted for sex and age, because there are no established predictors of lasmiditan efficacy thus far. Similarly, regarding the patients’ preference for acute treatment (lasmiditan versus their prior acute migraine treatment), variables were compared by logistic regression analysis adjusted for sex and age. A p-value <0.05 on two-sided tests was considered significant. Data were analyzed using the R statistical computing program (version 4.2.2, https://www.r-project.org/).
Results
Baseline characteristics
Forty-eight patients consented to the study and submitted questionnaires. Participants were predominantly female (n = 44, 91.7%), with migraine without aura (n = 33, 68.8%), and were concurrently using a migraine prophylactic treatment (n = 28, 58.3%). The mean MHD in total was 8.1 ± 6.5. There were 15 (31.2%) patients who were under the treatment of an anti-CGRP mAb migraine prophylactic. Twenty-two patients (45.8%) reported severe headache before the treatment, 24 (50.0%) had moderate headache, and 2 (4.2%) had mild headache (Table 1).
Baseline characteristics.

Data were presented as unweighted number (percentage) of patients unless otherwise indicated.
*headache intensity before the medication. †usage of any preventive medications. ‡usage of any Anti-CGRP Ab.
Abbreviations; SD: standard deviation, BMI: body mass Index, MwA: migraine with aura, MwoA: migraine without aura, MHD: monthly headache days, Anti-CGRP mAb: anti-calcitonin gene-related peptide monoclonal antibody.
Lasmiditan efficacy over time after medication
As shown in Table 2 and Figure 1, 23 of the patients (47.9%) reported their headache severity was ‘mild [1]’ or ‘none [0]’ at 60 min after taking lasmiditan. Headache disappeared (i.e., was described as severity ‘none [0]’) in 6 patients (12.5%) after 60 min, 16 (33.3%) after 90 min, and 23 (47.9%) after 120 min.

Change in the number of patients with headache severity after taking Lasmiditan.
Patient distribution of headache intensity at each time point after taking lasmiditan.

*Time (minutes) after taking the medication. Data were presented as unweighted numbers (percentage) of patients unless otherwise indicated.
Predictive factors of responder group
Twenty-three patients (47.9%) were categorized into the responder group, and 25 (52.1%) were categorized into the non-responder group (Table 3). To determine the predictive factors for patients being responders, baseline characteristics and clinical features were compared between the responder and non-responder patient groups. There were no significant differences between the responder group and the non-responder group in baseline characteristics (including sex and BMI), headache intensity before treatment, MHD, usage of preventive treatment, duration of headache onset to treatment with lasmiditan, nausea/vomiting, photophobia, phonophobia, and the side effects (including dizziness, somnolence, malaise, nausea, palpitation). In the sensitivity analysis that used a less stringent definition to classify the responder group, the results of the predictive factors for the improvement group were similar, except for age. Older age emerged as a positive predictive factor for headache relief (adjusted odds ratio: 1.072 [1.008–1.142], p = 0.028) (Online Supplemental Table 1).
Predictive and associated factors of responder.

Data were presented as unweighted numbers (percentage) of patients unless otherwise indicated. Significant differences in p values are indicated in bold. *headache intensity before the medication, †usage of any preventive medications, ‡usage of any Anti-CGRP monoclonal antibody injection, §accompanying symptom before the medication, ¶side effects that appeared within 2 h after the medication.
Abbreviations; SE: standard error, OR: odds ratio, CI: confidence interval, SD: standard deviation, BMI: body mass index, MwA: migraine with aura, MHD: monthly headache days, Anti-CGRP mAb: anti-calcitonin gene-related peptide antibody.
Lasmiditan side effect profile over time after the medication
Table 4 and Figure 2 summarize the time course of the appearance of side effects including dizziness, somnolence, malaise, nausea, and palpitations every 30 min after oral administration up to two hours. The cumulative column in Table 4 describes the total number and percentage of patients who reported side effects in 2 h after taking lasmiditan. In Figure 2, the points represent the frequency of side effects at each time point, and the bars represent the cumulative frequency of side effects up to that time.

Time course of frequency of patients with side effects and accumulation of side effects up to that time.
Side effects over time after the medication.
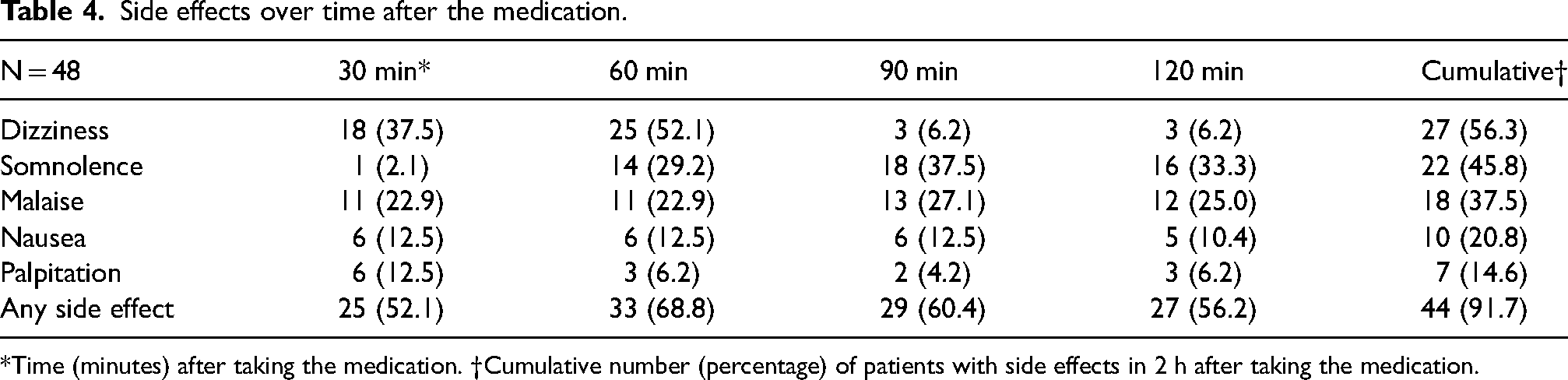
*Time (minutes) after taking the medication. †Cumulative number (percentage) of patients with side effects in 2 h after taking the medication.
Dizziness was most frequently reported at 60 min (n = 25, 52.1%) after administration and disappeared drastically in most patients within 90 min (reported only by 3, 6.2%). Somnolence as a side effect was most reported at 90 min (n = 18, 37.5%) and did not increase much thereafter.
In total, somnolence was reported by 22 patients (45.8%), malaise was reported by 13 patients (27.1%), nausea was reported by 10 patients (20.8%), and palpitations were reported by seven patients (14.6%) in total. The proportion of patients reporting any side effects within 2 h after administration peaked at 60 min (n = 33, 68.8%). In total, 44 patients (91.7%) experienced some kind of side effect within two hours after administration.
Patients’ preference for acute treatment
Table 5 shows the results of a question asking whether the patient preferred their previous acute medication or lasmiditan. The previously used acute treatment was triptans in 12 patients, non-steroidal anti-inflammatory drugs in four, over-the-counter pain medication in one, and the information was unavailable for three patients. The latest treatments, such as gepants, are not yet available in Japan. Of 48 patients, 20 (41.7%) indicated that they prefer lasmiditan. A logistic regression model adjusted for age and sex was performed to identify the predictors of preference for lasmiditan. In comparing patients who indicated they preferred lasmiditan to those who indicated they did not, there were no significant differences in baseline characteristics, headache intensity before treatment, MHD, and the usage of preventive treatment or anti-CGRP mAb. The proportion of patients who were responders was higher (n = 12, 60.0%) in the group that preferred lasmiditan, but not significantly so. Regarding the reason for the preference, 13 participants (65.0%) in the group that preferred lasmiditan reported it had ‘stronger efficacy.’ Conversely, 22 participants (78.6%) in the group that preferred an acute medication other than lasmiditan cited that their other abortive treatment had ‘fewer side effects.’ Finally, 17 patients (85.0%) preferred lasmiditan despite experiencing side effects.
Patient's preference for acute treatment.

Data were presented as unweighted numbers (percentage) of patients unless otherwise indicated. Significant differences in p values are indicated in bold.
*headache intensity before the medication, †usage of any preventive medications, ‡usage of any Anti-CGRP Ab, §side effects that appeared within 2 h after the medication.
Abbreviations; SE: standard error, OR: odds ratio, CI: confidence interval, SD: standard deviation, BMI: body mass index, MwA: migraine with aura, MHD: monthly headache days, Anti-CGRP mAb: anti-calcitonin gene-related peptide antibody, NA: not available, NSAIDs: non-steroidal anti-inflammatory drugs, OTC: over-the-counter drugs.
Discussion
This is the first multi-center prospective observational real-world analysis evaluating the efficacy and tolerability of lasmiditan as an acute treatment for migraine attacks. The main finding of this study is that lasmiditan achieved pain-free status for 47.9% of migraine attacks within 2 h after treatment for patients in the real world. This result is higher than that of the previous phase 2 or 3 clinical trials, which reported that the rate of pain-free achievement at 2 h after administration was between 28.2% and 32.4% (5,–7). The achievement of such a high headache-free rate is one of the most important characteristics of acute migraine medications because inadequate treatment is a risk factor for the chronification of migraine (14).
There were no significant differences in baseline characteristics between the responder and non-responder groups. Takeshima et al. also reported that the efficacy of lasmiditan is generally not influenced by patient characteristics (9), based on the MONONOFU clinical trial in Japan (7). It has been reported that the factors associated with having headache resolution within 2 h after use of common acute medications are: being female, having low BMI, reporting a lower numerical rating scale (NRS) of headache severity, and using prophylactic medications (15). The report by Lipton et al. is based on the Migraine Treatment Optimization Questionnaire surveys from 2006 collected as part of the American Migraine Prevalence and Prevention (AMPP) study, a longitudinal, population-based study in the United States (15). We found no significant difference in patient sex, BMI, headache severity, prophylactic medication utilization, or other baseline characteristics between responders and non-responders. The absence of such predictive factors in our study and previous studies may suggest that lasmiditan has different properties than previous acute-phase migraine medications. Similarly, there were no predictors for treatment response among the characteristics of headache, migraine preventive treatment used with lasmiditan, and side effect profile. These results are consistent with those reported in the post-hoc analyses of previous clinical trials, where the efficacy of lasmiditan was unaffected by patient characteristics, migraine disease history, or migraine attack characteristics (8,–10). Of note, the occurrence of side effects would not be positive predictors in general.
Notably, there was no significant difference in the time from headache onset to the administration of lasmiditan between the responder and non-responder groups (178.2 ± 340.0 vs 91.4 ± 120.5 min, adjusted odds ratio: 1.001 [0.998–1.005], p = 0.446). This result is compatible with previous reports. Tepper et al. reported that lasmiditan is effective even when dosing two or more hours after the onset of headache (8). Takeshima et al. also reported that lasmiditan dosing one hour after the onset of headache is effective (9). In contrast, triptans have been reported to be more efficacious when used earlier during the attack (16). Our results suggest that this may not be the case for lasmiditan. Therefore, lasmiditan could also be used as a second-line acute treatment in clinical practice, since the efficacy of other acute medicine decreases with increased time between headache onset and dosing.
Older age was a positive predictive factor for headache relief (Online Supplemental Table 1). This result may be a statistical fluke, but if true, the elderly may be a more apparent target for lasmiditan treatment, especially because it is safe for patients with vascular risk factors. Consistent with previous studies, the results of our study support the idea that lasmiditan is an effective acute treatment option for migraine.
We evaluated the time course of the occurrence and resolution of adverse effects associated with lasmiditan. As shown in Table 4, the overall occurrence rate of adverse drug effects within 2 h was as follows. The cumulative incidence of side effects within 2 h is 56.3% (n = 27) for dizziness, 45.8% (n = 22) for somnolence, 37.5% (n = 18) for malaise, 20.8% (n = 10) for nausea, 14.6% (n = 7) for palpitations, and 91.7% (n = 44) for any side effect. The frequency of each side effect was higher than those reported in the SPARTAN/ SAMURAI/ MONONOFU trials (5,–7). In these prior studies, reported side effects included dizziness (reported by 12.5–38.0% of patients), somnolence (4.6–21.2%), malaise (4.1–11.1%), nausea (3.0–5.3%), and palpitations (0–0.3%). When comparing the three previous clinical trials, the MONONOFU trial conducted in Japan reported more side effects in all categories. Our study results suggest that adverse drug effects are more frequently reported in the real-world and may be more frequently reported among the Japanese population. Another possible reason for the higher frequency of adverse drug reactions reported in our study than in past MONONOFU studies in Japan is the differences in how information is collected. In our study, the side effects to be selected were dizziness, somnolence, malaise, nausea, palpitation, and free writing only, whereas in the MONONOFU study (NCT03962738), more items could be described, which may have resulted in a more varied selection. For example, the MONONOFU study described the report of malaise, fatigue, asthenia, and muscular weakness separately, which may correspond to same category in our study. We should especially note that almost half of the participants in this study reported somnolence as a side effect, which is a much higher frequency than was reported in the trial data. The side effect of somnolence has significant potential risks, especially for driving. A study evaluating the effects of lasmiditan on the ability to drive a car in healthy subjects with healthy sleep rhythms found that lasmiditan affected driving ability after 1.5 h of oral administration, but that this effect abated 8 h after oral administration (17). We believe that physicians need to fully inform patients about the potential side effects of lasmiditan, especially somnolence, in real-life clinical practice where a wide variety of patients take the drug.
We also evaluated patients who experienced any side effects (including dizziness, somnolence, malaise, nausea, or palpitations). We found that 91.7% of patients reported at least one of these side effects during the 120-min period. Of note, this study included patients on their first dose of the drug. The open-label long-term safety study of lasmiditan in the acute treatment of migraine (GLADIATOR) study reported that the incidence of side effects decreases with repeated oral administration. According to the GRADIATOR study, treatment-emergent adverse events (TEAEs) occurred in 20.9% of patients receiving the first dose of 100 mg of lasmiditan, while it decreased to 11.3% after the fifth dose (11). Similarly, phase 3 placebo-controlled study designed to assess the efficacy, consistency, and safety of lasmiditan for the acute treatment of four migraine attacks with or without aura (CENTURION study) reported that TEAEs tended to decrease in frequency across the four attacks (18). Future studies should evaluate this trend in a real-world patient population.
Regarding the time course of common side effects, dizziness was more frequently reported at 30 and 60 min, and disappeared in most patients by 90 min. These results are compatible with post-hoc analyses of clinical trials. Tepper et al. reported that the median time to onset of dizziness after dosing was generally 30–40 min, and the median duration was 1.5–2 h. Overall, dizziness as a side effect is frequent but disappears relatively quickly. Conversely, somnolence remained a relatively long-lasting side effect, with 33.3% of patients still experiencing symptoms at 120 min.
Lastly, participants were asked about their preference between lasmiditan and their previous acute treatment. Surprisingly, 41.7% of patients (n = 20) preferred lasmiditan to their previous medications, even though 91.7% (n = 44) of them experienced some side effect. As seen in Table 5, we compared lasmiditan preferred group and the other medication group. There were no significant differences in baseline characteristics, headache characteristics, category of their other acute medications, and the occurrence of side effects between patients who preferred lasmiditan and those who preferred their other acute treatment. Patients in the lasmiditan group were more likely to cite the strength and duration of medication effect as reasons for the preference. Conversely, those who preferred their previous medication were more likely to cite fewer side effects as the reason for their preference.
Limitation
This study has several limitations, and its results should be interpreted with caution. One of the biggest limitations is the relatively small number of enrolled subjects, which might impact the statistical power of the study. Although the logistic regression model was adjusted for gender and age, other patient characteristics that were not collected—like comorbidities and concomitant medications—could also influence treatment outcomes and may affect results. As we collected information using a paper questionnaire, there is no data on the time between the lasmiditan oral administration and when the survey was completed, and the potential for recall bias cannot be ascertained. The questionnaire utilized was newly created and is not validated. Selection bias is possible, since the real-world patients who were interested in participating in the study may have been more likely to be dissatisfied with their previous acute medication. If the study had included patients who were highly satisfied with their previous acute medications, the preference for lasmiditan may have been lower than what we reported (40%). We did not collect data on the severity of side effects. Although a free-text box was available on the questionnaire, most patients reported side effects using the provided multiple-choice format, which may have influenced the results. Additionally, the side effect of nausea after intake is subjective to the patient, and may be difficult to distinguish from concomitant symptoms of migraine attack. The preferences of patients in Table 5 do not include information on previous changes in acute care medications or previous occurrence of side effects. When interpreting these results, consider two opposing biases: some patients might feel inclined to prefer any newer drug to older drugs that are generally available, while others may avoid lasmiditan because they have a stable prescription history with the previous drug of choice.
Conclusion
This real-world study showed that lasmiditan has high efficacy in reducing the severity of headache attacks or achieving headache resolution. Overall, 91.7% of patients reported side effects related to lasmiditan. Yet, 41.7% of patients preferred lasmiditan to their previous acute migraine treatment despite the occurrence of side effects. Lasmiditan may also have particular clinical utility as a backup rescue medication due to its therapeutic properties.
Key findings
Lasmiditan is effective in achieving headache-free status as an acute treatment for migraine.
More than 90% of patients experienced side effects of lasmiditan.
Approximately 40% of patients preferred lasmiditan despite their side effect burden.
Supplemental Material
sj-xlsx-1-cep-10.1177_03331024241258695 - Supplemental material for Efficacy and tolerability of 100 mg of lasmiditan for migraine: A multi-center, prospective observational real-world study in Japan
Supplemental material, sj-xlsx-1-cep-10.1177_03331024241258695 for Efficacy and tolerability of 100 mg of lasmiditan for migraine: A multi-center, prospective observational real-world study in Japan by Ryotaro Ishii, Kei Ishizuchi, Narumi Watanabe, Ryosuke Fukazawa, Meesha Trivedi, Jin Nakahara and Tsubasa Takizawa in Cephalalgia
Footnotes
Acknowledgements
The authors gratefully acknowledge the patients, medical staff, and clinicians who participated in this registry. In addition, they would like to thank for kind support of the representative of each site, including Dr Masahiro Makino of Department of Neurology, Kyoto Okamoto Memorial Hospital, Kyoto, Japan, and Dr Akihiro Fujii of Department of Neurology, Saiseikai Shiga Hospital, Shiga, Japan. The authors would like to thank Dr Koji Sekiguchi of Department of Neurology, Keio University School of Medicine for insightful discussion about the study.
Authors’ note
The principal investigator is Ryotaro Ishii, who had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RI has served as a consultant for Amgen K.K., Eli Lilly Japan K.K., Daiichi Sankyo Co., Ltd., and Otsuka Pharmaceutical Co., Ltd. He has received lecture fees from Amgen K.K., Eli Lilly Japan K.K., Daiichi Sankyo Co., Ltd., and Otsuka Pharmaceutical Co., Ltd.
KI has nothing to disclose.
NW has nothing to disclose.
RF has nothing to disclose.
MT has nothing to disclose.
JN has received honoraria and research scholarships from Amgen K.K. and Daiichi Sankyo Co., Ltd.
TT served as a consultant/advisor and/or served on an advisory board for Eli Lilly Japan K.K., Otsuka Pharmaceutical Co., Ltd., Amgen K.K., Teijin Pharma Limited, and Pfizer Japan. He received honoraria from Eli Lilly Japan K.K., Daiichi Sankyo Co., Ltd., Otsuka Pharmaceutical Co., Amgen K.K., Kowa Co., Ltd., Kyowa Kirin Co., Ltd., Eisai Co., Ltd., UCB Japan Co., Ltd., Takeda Pharmaceutical Co., Ltd., and Santen Pharmaceutical Co., Ltd.. He received funds from Eli Lilly Japan K.K., Pfizer Japan Inc., and Tsumura & Co. outside the submitted work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was conducted without funding sources such as a pharmaceutical company. All expenses were covered by an allocated university budget of each site for the Department of Neurology.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
