Abstract
Background
Cluster headache presents in an episodic and chronic form, between which patients can convert during the course of disease. We aimed to quantify the rate of cluster headache patients changing phenotype within one and five years and investigate the earlier proposed association between chronification and having side-shifting attacks.
Methods
In total, 430 cluster headache patients well-characterized according to current International Classification of Headache Disorders criteria, who were all participants in a prior transition-study, were re-interviewed in an observational, retrospective, cross-sectional follow-up study design at the Danish Headache Center.
Results
The transition rate for the whole cohort was 6.5% within one year and 19.8% within five years. The risk of becoming chronic if episodic was 4.0% within one year and 12.3% within five years. For conversion from chronic to episodic, the corresponding risk was 11.1% and 25.0%, respectively. Alterations in attack-side were reported in 32% of all chronic patients, generating an odds ratio of 2.24 of being chronic as opposed to episodic if experiencing side-shifting attacks.
Conclusions
A higher transition rate since the original cross-sectional study demonstrates cluster headache as a non-static condition. Identifying a risk of transition within one and five years, based on current phenotype along with high odds of being chronic when experiencing a shift of attack-side, offers a valuable clinical compass in the dialogue with the patient.
Introduction
Recognized to be the most painful primary headache disorder, cluster headache (CH), constitutes a substantial burden for its patients (1–4). Diagnostically, CH is categorized into two phenotypes: episodic (eCH) and a chronic (cCH). Although 80–90% of patients have an episodic variant with longer attack free remission periods in between bouts, the remaining 10–20% suffer from the chronic variant with attack free periods not exceeding three consecutive months per year (3,5). Hence, phenotyping in CH is not only an exercise in diagnostics, but also an indicative factor of the CH patient's disease burden (4).
It is a well-established fact that CH is a dynamic condition with patients changing diagnostic phenotype over the course of the disorder, from primary episodic (pECH) to secondary chronic (sCCH) or from primary chronic (pCCH) to secondary episodic (sECH) (6–8). Previous cross-sectional studies have provided heterogenous estimates ranging from 2–32% of the transition rate. Therefore, a larger cohort study was conducted by our group in the Danish Headache Center (DHC) in 2022, reporting a total transition rate of 20.7% during the course of the disorder (9). However, whether this number covered only single or multiple changes of phenotype within the same individual remained unexplored. Furthermore, even more clinically pertinent, we are still unable to provide patients with an estimate of the risk of phenotype transition within a measurable future.
In search of clinically useful predictors for phenotype transition, several studies have examined a broad array of demographic factors and features of disease burden as possible indicators for transitioning (7,10). The conducted study from DHC found a history of shifting side of CH attacks as a possible association for pECH converting into sCCH. However, the study did not investigate when this side-shifting occurred in the patient´s entire course of disease, nor whether it debuted in proximity with the phenotype-transition. Furthermore, several other factors remained unexplored, such as the influence of medical therapy, concurrent head trauma or external stressors.
We sought to address these uncovered fields of research by conducting a follow-up study of our cohort of formerly interviewed CH patients.
Our aim was to propose a one-year and five-year rate of patients with CH changing phenotype. Additionally, we aimed to investigate possible factors for transitioning, among these the previously suggested shifting side of attacks as an indicator for transitioning to sCCH. Our expectation was to find an approximately one-year and five-year transition rate of 3% and 10%, respectively.
Methods
Participants
We performed an observational, retrospective, cross-sectional, single-center follow-up cohort study of 430 patients diagnosed with CH, first interviewed in a genetics-study (H22010122) with continuous patient-inclusion between 2017–2023. The study was carried out at the DHC, a specialized tertiary referral center, with collection of data between 2022–2023. Being a follow-up study, all interviewed participants in the original transition study were contacted anew and underwent a semi-structured interview by telephone.
Because the mentioned genetics study is an ongoing study at DHC, an additional 34 patients had been included since 2021, where our first transition study was carried out. These patients also answered the follow-up interview and were added to the original cohort.
Inclusion criteria included a diagnosis of CH according to current International Classification of Headache Disorders (ICHD) criteria (ICHD-3-beta or ICDH-3), validated by a headache specialist, and the ability to tell CH attacks apart from other types of headaches. A still-active diagnosis of CH according to ICHD-3 criteria was confirmed by the interviewer at the beginning of each interview. In case of uncertainty or a rejection of the CH diagnosis, the patient was excluded. There were no additional criteria for exclusion, concordantly with the inclusion procedure reported previously (9).
Study size
A power calculation for sample size was not performed because all patients with CH and affiliated to DHC during 2017–2021 were consecutively invited to participate with the aim of including as many as possible.
Interview
For the present study, a structured interview, based on clinical experience and previous epidemiological questionnaires (9,11), was constructed by four of the investigators (MKS, ASP, NL and RJ). Validation was performed by a randomly selected population of five CH patients representing different ages, gender and CH phenotype (eCH and cCH), where changes based on their feedback were implemented accordingly.
The interview contained up to 71 questions in eight sections: verification of headache diagnosis, CH phenotype, phenotype transition, medical treatment, CH attack variants, CH background headache, medication overuse headache and comorbidities within the last year (see Supplementary Material). All patients underwent the same interview, apart from the transition-section, which was applied according to current CH phenotype: pECH, sECH, pCCH or sCCH. Medical used terms and definitions such as ‘chronic CH’, ‘episodic CH’ and ‘remission’ were explained at each patient contact. Estimated time of completion of the interview was 30 min. Interviews were performed by two of the investigators (MKS and ASP) and three purpose-specific trained medical students, consulting one of the investigators (ASP) in cases of ambiguity. All current phenotypes were compared with phenotype status in the original study for consistency. Subsequent auditing by two of the investigators (MKS and ASP) was performed in cases of possible discrepancies between self-reported data and personal medical charts.
Study data were collected and managed using REDCap electronic data capture tools hosted by the capital region of Denmark (12,13).
Outcomes
Our primary outcome was the number of patients who had changed CH phenotype to either sECH or sCCH within the last year and the last five years, according to present ICHD-3 criteria. Furthermore, we calculated a resultant risk for changing phenotype within one and five years if being respectively episodic or chronic.
The subgroup of patients with a registered transition of CH phenotype was also described by various secondary outcomes: most importantly, the prevalence of side-shifting attacks. First, patients were asked if they had ever experienced a shift in attack-side during their entire course of disease. In such a case, a timewise characterization of the side-changing attacks was given (side-shift during an attack, between attacks or both, debut of side-shifting attacks concomitant with time of phenotype transition, increase in frequency of already known side-shifting attacks concomitant with time of phenotype transition, debut of bilateral attacks concomitant with time of phenotype transition). Furthermore, a groupwise comparison of the episodic and chronic patient group provided odds ratios for being either episodic or chronic when experiencing the mentioned parameters.
Both sCCH and sECH patients were described by features of their disease burden immediately before transition versus priorly (change in average maximum pain level during attacks, change in duration of attacks, diminishing effect of abortive, preventive and transitional treatment). sCCH patients were further characterized by traits of their last three bouts before chronification versus priorly (change in duration of bout, change in duration of remission in between bouts). Both groups were additionally described by external life circumstances immediately before the time of phenotype transition, with that being the prevalence of head trauma within two weeks before time of transition, presence of external stressors, changes in job-affiliation and changes in smoking habits.
Patient consent and registration
In compliance with the Declaration of Helsinki, all participants provided their verbal and written formal consent. The study was approved by the Danish data protection agency (P-2022-607) and was reported to the ethics committee but did not need a formal ethics approval according to the Danish law, being a non-interventional study.
Statistical analysis
Data were collected retrospectively with subsequent data management and statistical analyses performed using SAS, version 9.4 (SAS Institute Inc., Cary, NC, USA). Methods of statistical analysis were discussed with a biostatistical consultant from the University of Copenhagen. p < 0.05 (two-tailed) was considered statistically significant.
To evaluate side-shift of attacks as a potential indicator for transition, patients were compared groupwise as in the original study; pECH were compared with sCCH and pCCH were compared with sECH. Assessing risk of changing phenotype within one and five years, respectively, an equation was formulated, exemplified for the risk for episodic patients turning chronic within one year: total number of sCCH patients/(number of pECH patients + number of sCCH patients + number of sECH patients answering no to a phenotype transition within the last year).
For quantitative continuous variables, we used the mean ± SD or median and interquartile range, depending on the distribution of data.
For categorical data, group comparisons were assessed using a chi-squared test or Fisher’s exact test as appropriate. For calculation of risk of changing phenotype within one and five years according to which phenotype one was at baseline, we calculated the 95% confidence interval (CI) using exact values under a binomial distribution assumption. Patients with missing data were omitted from analysis of the examined variable. No sensitivity analysis was performed.
All findings were reported according to the Strengthening the Reporting of Observational studies in Epidemiology (STROBE) guidelines (14).
Results
In total, 546 patients were invited to participate in the study, of whom 430 completed the interview, constituting a follow-up-rate of 79%.
Since the first interview, 19 patients had died, two were lost to follow-up and 24 could not participate for various other reasons (i.e. no accessible updated contact-information, withdrawal of CH diagnosis, fulfilling interview not possible because of cognitive problems or language barrier). At interview, 20 declined participating and 96 patients could not be reached (Figure 1). The non-responder group had a male-to-female ratio of 1.9:1 and was made up of 38.5% episodic and 61.5% chronic patients according to their phenotype status at the first interview. Median follow-up time from first to second interview was 4.6 (interquartile range 3.0–5.5) years.

Flowchart of recruitment and inclusion of patients.
In total, 430 patients were included for final analysis, composed of 283 (65.8%) episodic and 147 (34.2%) chronic patients. Patients were subcategorized into four groups according to reported transition during course of disease: pECH, sCCH, pCCH and sECH (Table 1). Mean ± SD age for the entire cohort was 51.0 ± 13.6 years with a male-to-female ratio of 2.1:1.
Clinical patient characteristics and shifting side of attacks.

pECH: primary episodic cluster headache; sCCH: secondary chronic cluster headache; pCCH: primary chronic cluster headache; sECH: secondary episodic cluster headache.
Transition of phenotype
The total transition rate for the cohort was 31.6%, as change of phenotype into sECH was reported by 61 (14.2%) patients and to sCCH by 75 (17.4%) patients. The remainder of the population was distributed as 222 (51.6%) pECH and 72 (16.7%) pCCH (Table 1).
One- and five-year transition rate
A conversion of phenotype within the first year was reported by 28 patients, constituting a one-year transition rate of 6.5%. Within this year, 11 patients (2.6%) had become sCCH and 17 (4.0%) had become sECH.
The corresponding five-year transition rate for the entire cohort was 19.8%. Here the distribution was 39 patients (9.1%) changing into sCCH and 46 patients (10.7%) changing into sECH. Of these patients, 16 reported multiple shifts back and forth in phenotype during the past five years, from either episodic to chronic to episodic again (n = 7) or from chronic to episodic to chronic anew (n = 9), respectively.
Risk of conversion
The risk for an episodic patient turning chronic within one year was 4.0% (95% CI = 2.0–7.0%). Analogously, the risk for an episodic patient turning chronic within five years was 12.3% (95% CI = 8.9–16.4%).
Correspondingly, the chance of changing phenotype from chronic to episodic within one and five years was 11.1% (95% CI = 6.6–17.2%) and 25.0% (95% CI = 18.9–31.9%), respectively (Figure 2).

Graphical abstract of risk of transition within one and five years based on current phenotype
Side-shifting attacks
We identified 22.1% of the total cohort with a history of shift in laterality of their attacks. A substantial share of all subgroups had experienced these lifetime alterations in attack-side, but this was most prevalent in the chronic population, at 38.9% of the pCCH group and 25.3% of the sCCH group, respectively (Table 1).
Ascertaining side-shift as an indicator for phenotype change, we performed a groupwise comparison. No statistically significant difference was found for either pECH compared to sCCH (p = 0.05) or pCCH with sECH (p = 0.08), respectively. In terms of odds, the odds ratio of being sCCH when having side-shifts compared to pECH were 1.9 (95% CI = 1.0–3.5). Correspondingly, the odds ratio for being sECH when having side-shifts was 0.5 (95% CI = 0.2–1.1) compared to pCCH (Table 1).
Comparing all current eCH patients with all cCH patients, we found that 32.0% of the chronic population had a history of side change compared to 17.3% of the episodic population, constituting an odds ratio of 2.24. Thus, experiencing a side change increases the odds 124% of having a chronic phenotype compared to an episodic phenotype (odds ratio = 2.24, 95% CI = 1.41–3.59, p < 0.001).
Subcategorized according to timing, we found no significant difference in the distribution between groups of experiencing side-shifts during attacks, between attacks or both of the aforementioned (Table 1).
Timewise correlation of side change to phenotype transition
Regarding the debut of side-shifting attacks, only 7.9% of sCCH and 1.7% of sECH patients reported a timewise relation to their phenotype change. Neither sCCH, nor sECH patients reported any increase in frequency of already known side-shifting attacks and only one sCCH described debut of bilateral attacks in immediate relation to the time of phenotype transition (Table 1).
Transition concomitant disease burden
CH attacks in proximity to the time of phenotype transition were described by sCCH patients as more painful (44.4%) and of longer duration (39.7%) than previously (Table 2). Similarly, the last three cluster bouts before becoming chronic were reported to be of longer duration (by 22.2%), with increased attack frequency (by 38.1%) and adjacent shorter remission periods (by 41.2%).
Circumstances immediately before time of transition.
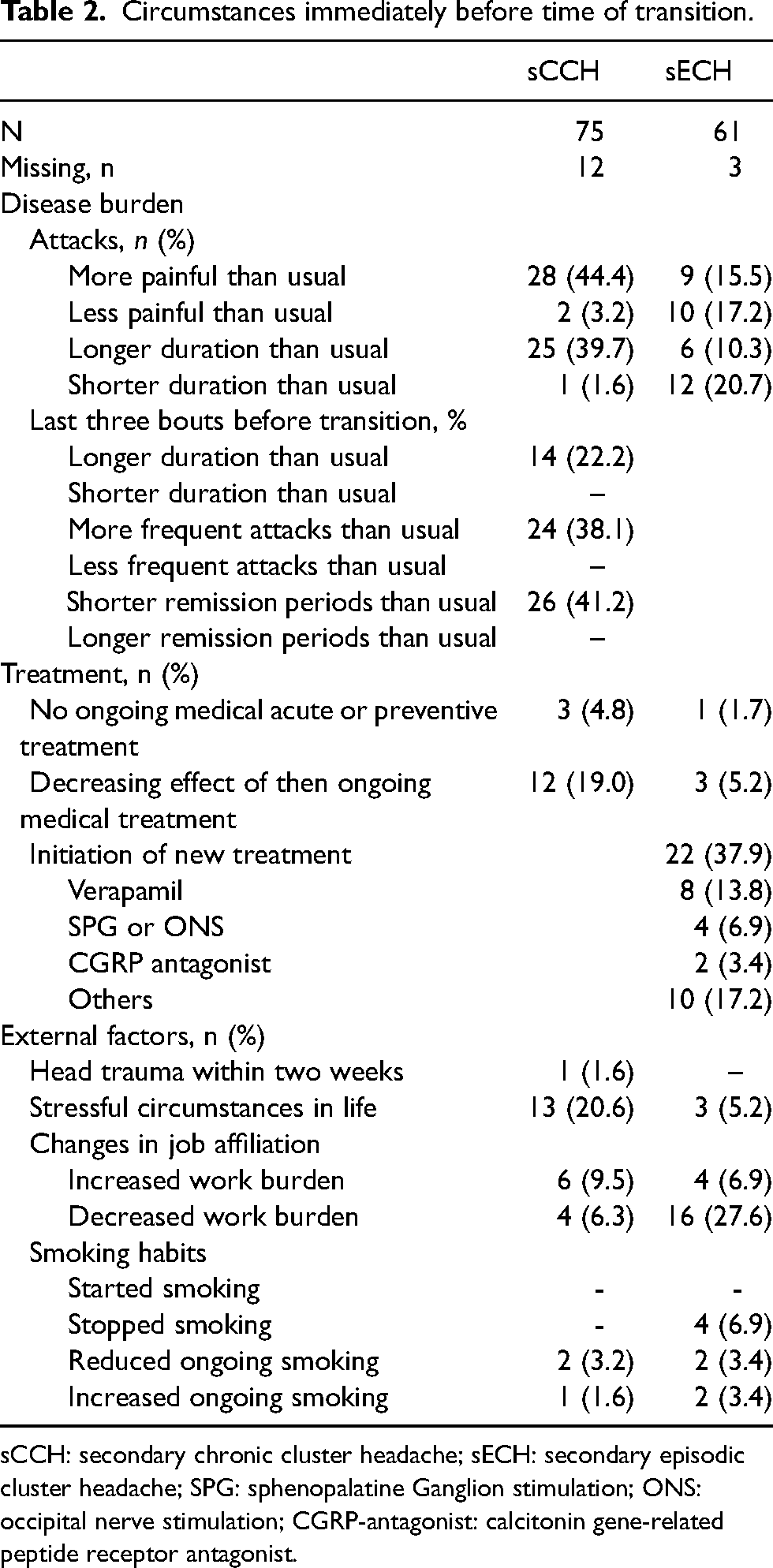
sCCH: secondary chronic cluster headache; sECH: secondary episodic cluster headache; SPG: sphenopalatine Ganglion stimulation; ONS: occipital nerve stimulation; CGRP-antagonist: calcitonin gene-related peptide receptor antagonist.
Depicting the sECH patients’ disease burden immediately before the time of transition, the trend was more unclear: 15.5% experienced more painful attacks, whereas 17.2% less painful attacks compared to previously. Attack-duration was likewise reported both longer (10.3%) and shorter (20.7%) than priorly. Of the sECH patients, 37.9% reported their de-chronification to be a result of the initiation of medical treatment, of whom 13.8% reported verapamil (Table 2).
External factors affecting phenotype transition
In proximity to the reported time of phenotype transition, we detected an increased work burden in 9.5% of sCCH and, inversely, a decreased work burden in 27.6% of sECH. Presence of stressful circumstances in life were reported by 20.6% sCCH patients, whereas no pronounced changes in smoking habits were exposed, nor was there a history of head trauma within two weeks from the time of transition (Table 2).
Discussion
The present study certifies transition of phenotype as a common phenomenon in CH patients with a total prevalence of 31.8% in the study population. Our data offer multiple novel findings, encompassing a one- and five-year transition rate of 6.5% and 19.8%, respectively, as well as a risk of transition within one and five years based on current phenotype: The risk of becoming chronic if episodic was 4.0% within one year and 12.3% within five years. For conversion from chronic to episodic, the corresponding chance was 11.1% within one year and 25.0% within five years. Lastly, we presented an odds ratio of 2.24 for being chronic if experiencing side-shifting attacks.
Multiple estimates had changed since our original cross-sectional study. First, the overall transition rate had increased from 20.7% to 31.6%. The discrepancy is plausibly explained by the follow-up design because an increasing share of patients will expectably experience a phenotype conversion proportionally with the number of years they are observed. Illustrating this trend, a preceding cohort study demonstrated how the cumulative proportion of sCCH patients increased from 9.3% to 36% over an observation period of 30 years (7). Concordantly, the secondary phenotypes in our cohort had increased to a higher percentage since the original study, from 14.4% to 17.6% for sCCH and from 6.3% to 12.9% for sECH, respectively. Together with the finding of several patients with multiple shifts in phenotype over five years, we observed a changeability within the same cohort underlining the fluidity of the CH phenotype over the course of disease. Whether the changeability is applicable to the general CH population remains discussable. Because the recruited cohort represents a specialized treated group with a probable increased disease burden, our findings may hold a potential overestimation. Contributing to a possible overestimation is the inclusion of patients with phenotype transition reported as a result of treatment because these patients could be regarded as not being representative of a true, spontaneous transition in the natural course of disease.
In our cohort, we found transition into sCCH (17.6%) to be more common than into sECH (14.2%), concordant with our original study. Although the transition rate for sCCH was in agreement with earlier studies estimates (2–19%), the transition rate for sECH was interestingly considerably lower than findings in previous studies (22–32%) (7,8,10). As discussed in our original study, this discrepancy may still be explained by the implementation of the current ICHD-3 criteria, introduced after the previous studies were performed. In terms of selection bias, we additionally recognize an active treatment affiliation to DHC as an incentive for participation. Hence, we can hypothesize a possible lower participation rate amongst the episodic patients and inversely a higher participation rate in the chronic cohort.
As new findings, the present study identified an approximate risk of changing phenotype within one and five years. The risk of changing phenotype to sECH was highest at both time points. Although not posing a new therapy in a classical sense, we consider that the present estimates answer a frequently asked question in the clinic. Thus, it constitutes an important prognostic piece of information for the individual CH patient, although we are still unable to predict their entire lifetime course.
Re-examining risk factors for phenotype conversion, we could not convincingly reproduce the previously found association between side-shifting attacks and a secondary chronification (9). Even so, the distribution clearly showed a trend of higher prevalence in the chronic groups. Adding to this observation, the odds ratio of 2.24 demonstrated an explicit pattern of being chronic if experiencing changes in attack laterality. These observations indicate how side-shift is a more frequent trait within the chronic spectrum, possibly serving as a useful clinical compass. Moreover, our findings are supportive of the theory of the chronic phenotype being a CH ‘wild-type’, not subjected to the same chronobiologic mechanisms as the episodic variant (6,11), although the underlying pathophysiologic mechanisms are still vastly uncharted (15,16). Contributing to this hypothesis are earlier findings of bilateral attacks predicting refractoriness to treatment and case reports of primarily chronic patients with transient side-shifting attacks upon greater occipital nerve injection (17,18).
Regarding a timewise correlation with phenotype transition, we did not demonstrate an apparent overlap with the debut time of side-changing attacks. Thus, our follow-up data do not indicate the debut of side-shifting attacks in an episodic patient as an omen of an immediately forthcoming chronification. Because patients were asked about side-shifting attacks during their entire course of disease, we could not determine precisely when the side-shifting attacks debuted, including whether the patient was episodic or chronic at the time. Since we conducted our original study, no new studies have investigated the relation between side-shift and chronification, neither validating, nor opposing our findings. Hence, a prospective study of the debut of side-shifting attacks in relation to phenotypic changes and underlying pathophysiology remains desirable.
Although not demonstrating concurrent changes in smoking habits and the occurrence of head-trauma as an association for transition, we uncovered possible patterns in disease burden immediately before the time of phenotype transition. The sCCH group showed a clear tendency of longer cluster bouts and shorter remission periods and attacks of increased frequency, duration and pain-level compared to previously. This indicates the transition phase as a disease continuum for this subset of patients. Moreover, 19.0% of patients reported diminishing effect of ongoing medical treatment in the time before transition. Although no statistical testing was applied as a result of small numbers, these findings strengthen speculation of whether decreasing treatment response could be an association for transitioning into a secondary chronic form. Alternatively, it could be interpreted as disease aggravation in episodic patients because of medication overuse as seen in mgraine (19,20). The clinical pattern of the time before transition was more heterogenous for the sECH group. Thus, we saw no clear tendency in changes within traits of the single attack. Most interestingly, a substantial part of the sECH patients reported their de-chronification as a result of initiation or adjustment of medical treatment, with the preventative verapamil reported as the responsible drug in 36.4% of these cases.
Strengths and limitations
The present study holds several methodological strengths, including being the largest cohort examined in a follow-up design for transition to date, patient recruitment from DHC with diagnostic validation by headache specialists and data collection with thorough audit by two investigators. Limitations include collection of data from a tertiary specialized center, as well as introducing selection bias as a result of possible over-representation of severely affected and chronic patients. The retrospective design constitutes a major limitation due to recall bias, whereas the interview design holds a possibility for interviewer bias, although attempts were made to minimize both via audit of personal medical charges and validation of phenotypes by a headache specialist. Furthermore, we acknowledge that performing the interview by telephone rather than in person introduces a risk of simplified answers, especially considering questions about personal life and stressful life events. Lastly, some of the examined subgroups were small in numbers, resulting in a risk of not exposing significant differences between groups in the data analysis, solely because of small sample size.
Conclusions
In conclusion, our large, interview-based follow-up study exposed an increasing transition rate since the original cross-sectional study, confirming CH as a dynamic condition. We identified a one- and five-year transition rate together with a risk of transition within one and five years based on current phenotype, offering a valuable clinical tool in the dialogue with the CH patient. Likewise, the high odds of being chronic if experiencing side-changing attacks constitutes a useful clinical compass, if not proposing alterations of attack side as a primarily chronic trait.
Clinical implications
The total transition rate for the entire cohort was 6.5% within one year and 19.8% within five years. The risk of becoming chronic, if episodic, was 4.0% within one year and 12.3% within five years. The risk of becoming episodic, if chronic, was 11.1% within one year and 25.0% within five years. Side-shifting attacks increases the odds of being chronic compared to episodic by 124%.
Footnotes
Acknowledgments
We thank all of the study participants.
Declaration of conflicting interests
Marie-Louise Kulas Søborg is a previous sub-investigator in trials for Lundbeck. Anja Sofie Petersen is a previous sub-investigator in trials for Lundbeck and received personal fees for teaching from Pfizer. Nunu Lund has received a restricted grant from Region Hovedstadens forskningsfond and Lundbeck and has given lectures for Pfizer and Dagens Medicin. Mads Christian Johannes Barløse has no conflicts of interest to report. Rigmor Højland Jensen has served as principal investigator in studies sponsored by Eli Lily, Novartis, Autonomic Technologies (ATI) and Lundbeck and has given lectures for Eli-Lily, ATI, Lundbeck, Novartis, AbbVie, Pfizer and TEVA.
Funding
This work was supported by Rigshospitalets Research Foundation and Lundbeck Pharma by an unrestricted study grant.
Data availability statement
The Danish Cluster Genetics Study contains sensitive information and cannot be shared in full form according to Danish data protection law. Deidentified data can be shared on request.
Ethical statement
In compliance with the Declaration of Helsinki, all participants provided their verbal and written formal consent. The study was approved by the Danish data protection agency (P-2022-607) and was reported to the ethics committee but did not need a formal ethics approval according to the Danish law, being a non-interventional study.
Supplemental material
Supplemental material for this article is available online.
