Abstract
Background
Headache burden is substantial in idiopathic intracranial hypertension. The classification of idiopathic intracranial hypertension headache by the International Classification of Headache Disorders (ICHD) is an important tool for research and clinical purposes.
Methods
We phenotyped headaches and tested sensitivity and specificity of the ICHD-3 criteria for idiopathic intracranial hypertension headache in a prospective cohort of patients suspected of idiopathic intracranial hypertension at two tertiary headache centers.
Results
Sensitivity was 93% and specificity was 100% of ICHD-3 criteria for idiopathic intracranial hypertension-related headache validated in idiopathic intracranial hypertension (n = 140) and patients in whom idiopathic intracranial hypertension was suspected but disproven (n = 103). The phenotype of new/worsened headaches related to idiopathic intracranial hypertension suspicion was equally migraine-like (p = 0.76) and tension-type-like (p = 0.08). Lumbar puncture opening pressure was higher (p < 0.0001) and pulsatile tinnitus more frequent (p < 0.0001) in idiopathic intracranial hypertension patients, but neither improved the applicability of the headache criteria, nor did papilledema.
Conclusion
Headache phenotype is not distinct in idiopathic intracranial hypertension. ICHD-3 criteria for idiopathic intracranial hypertension headache are sensitive and specific, but simplicity can be improved without compromising accuracy. We propose that a new or worsened headache temporally related to active idiopathic intracranial hypertension is a sufficient criterion for idiopathic intracranial hypertension headache regardless of headache phenotype or accompanying symptoms, and that elements of idiopathic intracranial hypertension diagnostics (papilledema and opening pressure) be segregated from headache criteria.
Keywords
Introduction
Headache is a prevailing and debilitating symptom in patients suffering from Idiopathic Intracranial Hypertension (IIH) (1) and significantly hampers quality of life (2). Evidence-based headache management is among the top ten research priorities agreed upon in 2018 (3) as it barely exists. Consensus opinion for secondary headache disorders is to treat the underlying headache-inducing condition, whilst the phenotypic presentation guides symptomatic pharmacological headache management; this applies to IIH headache too (4). Mimicry of primary headache disorders, in particular chronic migraine, is common in IIH (5–7). Young adulthood, female sex, and obesity are shared risk factors in migraine and IIH (8,9). Although several studies characterize headache in IIH, few describe headaches that have clearly evolved in temporal relation to the diagnosis. Pre-existing headache in IIH is reported in 45–63% (10–12) and in the IIH Treatment Trial (IIHTT), headache was the initial presenting symptom in <40% but existed in 84% of patients at time of IIH diagnosis (13). This reflects a significant burden of headache whose relation to IIH pathology is uncertain. Hence, appropriate classification of IIH headache is essential for clinical and research purposes.
Historically, criteria for IIH headache have changed with every version of the International Headache Society’s (IHS) International Classification of Headache Disorders (ICHD). Diagnostic criteria of IIH itself have changed as well (14–16) – dynamics that reflect the complexity of this poorly understood disorder. Importantly, criteria for IIH headache are not diagnostic for IIH itself, but for attributing a headache to IIH. As a principle of all secondary headaches, IIH headache requires: i) headache, ii) IIH, and iii) evidence of causation. There has been ambiguity as to whether descriptive qualitative headache features and accompanying symptoms should be included. The Classification Committee leadership working on ICHD-4 alpha invited comments on the present ICHD-3 criteria where improvements are considered necessary (17). In this study, we phenotype specifically new or worsened headaches related to clinical suspicion of new-onset IIH in a prospective cohort and field-tested the current ICHD-3 criteria for IIH headache.
Methods
Patient population
We prospectively and consecutively included patients ≥18 years of age with clinically suspected IIH at two tertiary headache centers in Denmark (Odense University Hospital and The Danish Headache Center, Rigshospitalet) from December 2018 to November 2022. The methodology is published previously (18).
Patients were investigated with a standardized semi-structured medical interview and neurological examination, cerebral venography, and Magnetic Resonance Imaging (MRI). Lumbar puncture (LP) with opening pressure (OP) measurement was performed with the patient in an outstretched relaxed left lateral decubitus position. Cerebrospinal fluid (CSF) and blood were analyzed. An experienced neuro-ophthalmologist assessed optic nerve morphology and function by slit lamp examination, fundus photography, optical coherence tomography (OCT), automated computer perimetry (visual fields), and Ishihara color plates. IIH was diagnosed according to the revised Friedman criteria (16). Patients with IIH relapse, secondary pseudotumor cerebri syndrome (PTCS), or pregnancy were excluded. Patients were included in case of confirmed new-onset IIH (hereafter “IIH”) including the following: i) definite IIH (papilledema and OP ≥ 25 cm CSF); ii) probable IIH (papilledema, OP < 25 cm CSF, and no better explanation of optic disc swelling after thorough investigation and often repeated LPs to validate the OP); and iii) IIH without papilledema (IIHWOP). IIHWOP is defined by absence of papilledema, but OP ≥ 25 cm CSF and either abducens nerve palsy (IIHWOP) or ≥3/4 specified neuroimaging signs of elevated intracranial pressure (suggested IIHWOP) (16). We compared patients with IIH to patients in whom IIH was initially suspected, but ultimately disproven (non-IIH), regardless of final diagnosis.
Headache phenotyping and ICHD-3 testing
All headaches were systematically characterized by a semi-structured interview. Headaches were considered IIH-related in case of: i) new-onset headache or ii) worsening of a pre-existing headache related to IIH diagnostic work-up. We particularly characterize these new/worsened headaches. Quantitative information on headache worsening (two-fold or greater increase in frequency and/or severity as required by the ICHD-3) was not available; patient reported worsening was considered relevant. New/worsened headaches were classified as migraine-like, tension-type like or other/non-classifiable according to the ICHD-3 (19). Use of analgesics was registered, but frequency and duration of use was not; thus, medication overuse headache could not reliably be assessed.
ICHD-3 criteria were validated in patients with headaches; headache-free patients are irrelevant for headache criteria and omitted from ICHD-3 validation. According to the ICHD-3 (19), four main criteria (A-D) must be fulfilled to diagnose IIH headache (Table 1). We evaluated each individual sub-criterion separately in IIH and non-IIH; overall fulfillment of criteria is presented too.
ICHD-3 criteria for IIH headache applied in the cohort.
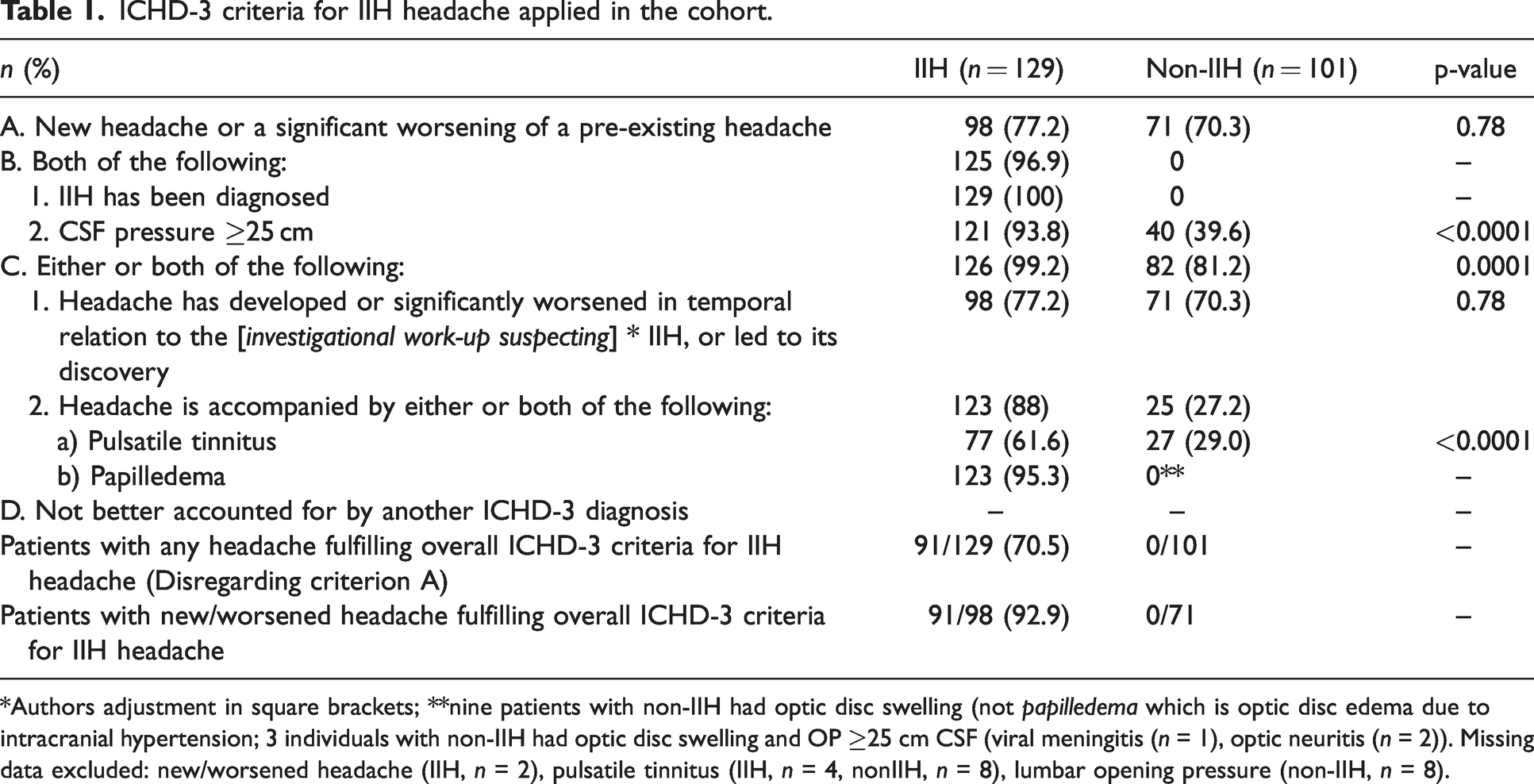
*Authors adjustment in square brackets; **nine patients with non-IIH had optic disc swelling (not papilledema which is optic disc edema due to intracranial hypertension; 3 individuals with non-IIH had optic disc swelling and OP ≥25 cm CSF (viral meningitis (n = 1), optic neuritis (n = 2)). Missing data excluded: new/worsened headache (IIH, n = 2), pulsatile tinnitus (IIH, n = 4, nonIIH, n = 8), lumbar opening pressure (non-IIH, n = 8).
We defined ‘true IIH headache’ as patients diagnosed with IIH who clearly had a new or worsened headache in temporal relation to the diagnosis.
Ethics
The study was conducted in accordance with the Helsinki Declaration (2013) and Danish data laws, and approved by the Committee on Health Research Ethics, Capital- and Southern Region of Denmark (S-20170058). Oral and written informed consent was obtained before inclusion.
Statistics
Categorical variables were tested with Fishers exact test. Student’s or Welch’s t-test were used as appropriate. For correlation analysis we used Spearman’s test. Statistical significance level was set at p < 0.002 after Bonferroni correction for multiple testing. OP exceeding possible measurement was corrected to the nearest measurable value, i.e., an OP >60 cm CSF was converted to 61 cm CSF. Statistical analysis was performed with R-studio version 4.2.2.
Results
Study population
Of 310 patients included in the cohort, 140 patients were diagnosed with “IIH” (definite IIH (n = 125), probable IIH (n = 8), IIHWOP (n = 0), and suggested IIHWOP (n = 7)). IIH was disproven in 103 patients (see Figure 1). Sixty-seven patients were excluded due to IIH relapse (n = 20), pregnancy (n = 4), uncertainty about the final diagnosis (n = 6), and secondary PTCS ((n = 37); drug-induced (n = 12), including lithium, doxycycline/tetracycline, isotretinoin, ustekinumab, and tapering of prednisolone; cerebral venous sinus thrombosis (n = 3); significant obstructive sleep apnea fulfilling criteria for secondary pseudotumor cerebri syndrome (n = 3), systemic disease (n = 8), e.g., kidney failure, dialysis, systemic lupus erythematosus, anti-phospholipid-antibody-syndrome; severe anemia (n = 5), and other causes (n = 6), e.g. genetic syndromes, complication to blood patch after iatrogenic intracranial hypotension, vascular disease, and unresolved contrast-enhancement of several cranial nerves). Patients with IIH were comparable to non-IIH regarding sex, BMI, age, and employment rate (Table 2) but had higher OP (39.0 cm CSF (SD ± 11) versus 25.0 cm CSF (SD ± 8), p < 0.0001). Elevated OP was found in 42 individuals (44%) of non-IIH, all with overweight or obesity (mean BMI 40.3). IIH and IIHWOP was carefully ruled out. Their final diagnosis was a primary headache disorder (n = 19), pseudo-papilledema (n = 7), optic neuritis (n = 2), meningitis (n = 3), sleep apnea without fulfilling criteria for PTCS (n = 2), non-classifiable headaches (n = 3), healthy (n = 3), and in three patients, no diagnostic conclusion appeared by chart review.
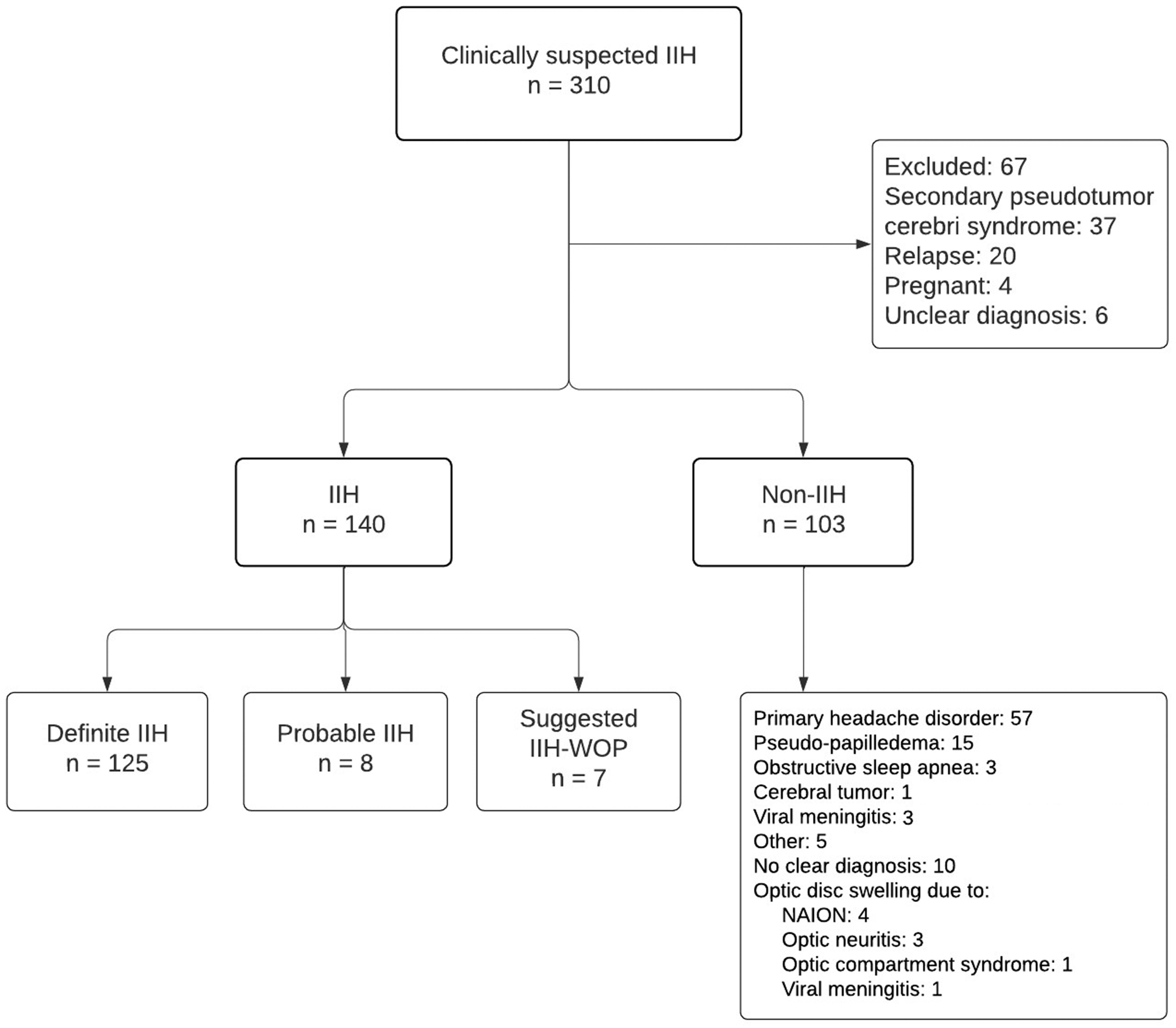
Flowchart of the cohort and final diagnoses.
Patient characteristics and prevalence of non-headache symptoms.
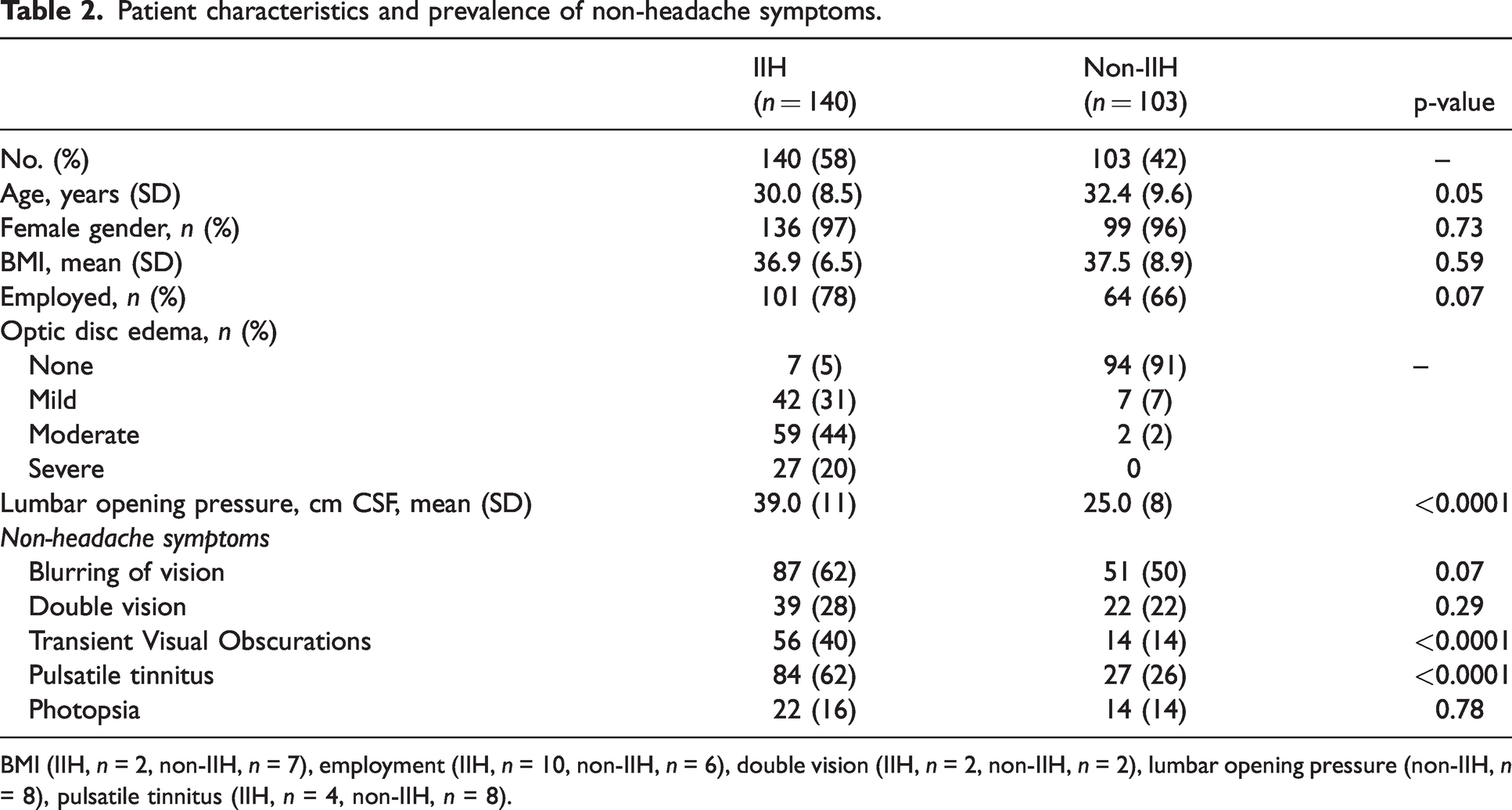
BMI (IIH, n = 2, non-IIH, n = 7), employment (IIH, n = 10, non-IIH, n = 6), double vision (IIH, n = 2, non-IIH, n = 2), lumbar opening pressure (non-IIH, n = 8), pulsatile tinnitus (IIH, n = 4, non-IIH, n = 8).
Overall, final diagnoses in non-IIH were as follows (Figure 1): primary headache disorders (n = 57), pseudo-papilledema (n = 15), optic disc swelling (n = 9: non-arteritic-acute ischemic optic neuropathy (NAION, n = 4), semi-compartment syndrome due to a crowded optic disc (n = 1), optic neuritis (n = 3), viral meningitis (n = 1)), obstructive sleep apnea (n = 3), cerebral tumor (n = 1), viral meningitis without optic disc swelling (n = 3), and other diagnoses (n = 5 idiopathic abducens palsy, chronic sinusitis, tinnitus, post-traumatic headache, and polycystic kidney disease). In 10 patients no diagnosis was concluded, but PTCS was ruled out.
Headache characteristics and phenotypes
Headache was found in 92% (n = 129) of patients with IIH and 98% (n = 101) with non-IIH (Table 3). Headache-free patients (n = 13) presented with visual symptoms (n = 6), pulsatile tinnitus (n = 3), eye pain (n = 1), and few were asymptomatic and referred from optometrists because of suspicious fundus photos taken as a routine health-check (n = 3). Headache was new or worsened in 77% with IIH and 71% in the non-IIH group (p = 0.78). New/worsened headaches were equally frequent (≥15 days monthly in 76% and 77%, p = 0.86), equally positional (32% and 37%, p = 0.51), equally aggravated by straining (54% and 62%, p = 0.35 but tended to be more relieved by lumbar puncture (46% versus 30%, p = 0.04) in IIH versus non-IIH-patients, respectively. Post-lumbar puncture headache was equally frequent (13% and 11%, p = 0.81). Headache phenotype was migraine-like in 36% and 39% (p = 0.76), tension-type like in 23% and 13% (p = 0.08), and other/non-classifiable in 41% and 33% (p = 0.20) of IIH and non-IIH patients, respectively; Table 3).
Characterization of new or worsened headaches related to IIH suspicion.
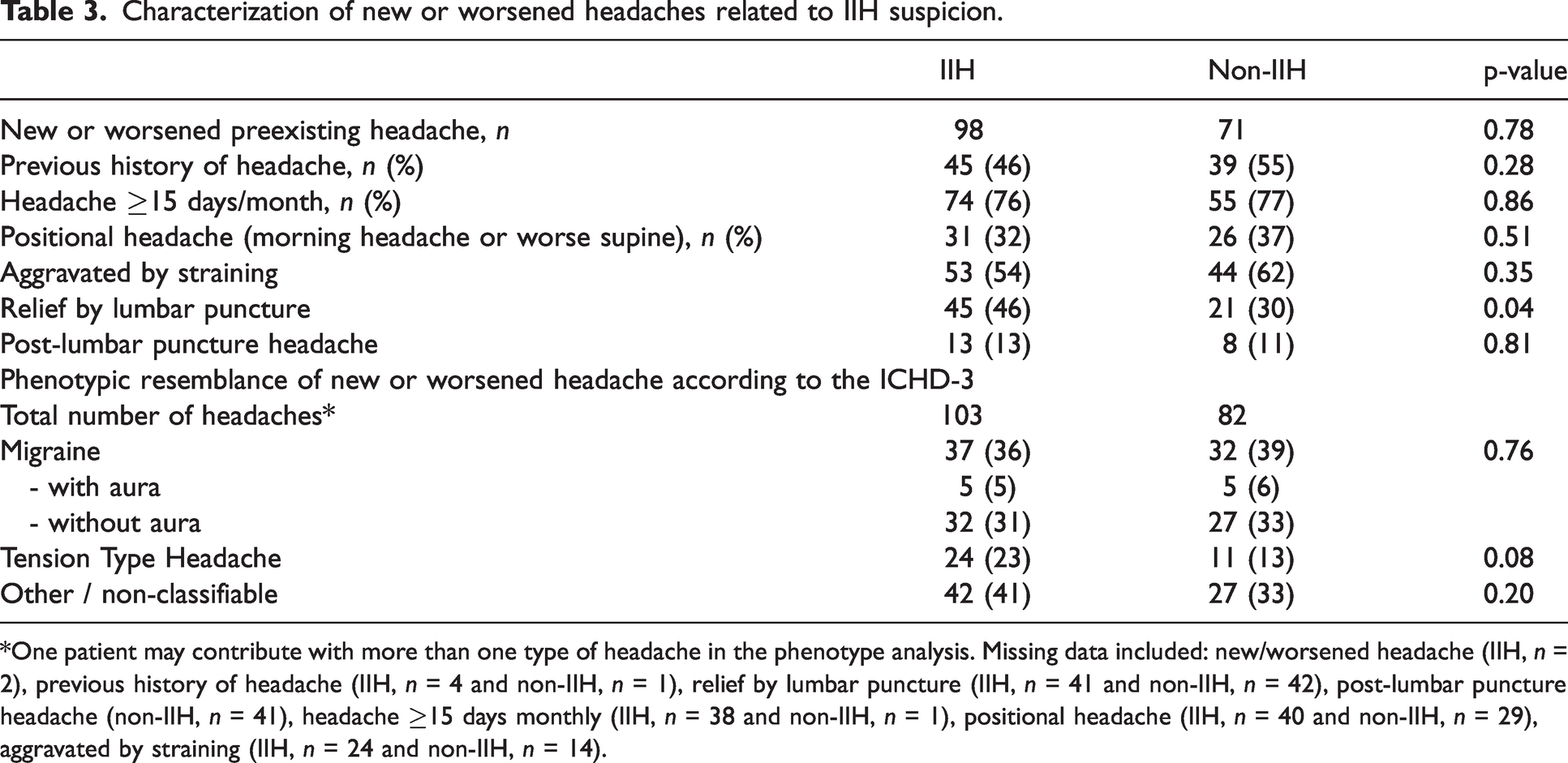
*One patient may contribute with more than one type of headache in the phenotype analysis. Missing data included: new/worsened headache (IIH, n = 2), previous history of headache (IIH, n = 4 and non-IIH, n = 1), relief by lumbar puncture (IIH, n = 41 and non-IIH, n = 42), post-lumbar puncture headache (non-IIH, n = 41), headache ≥15 days monthly (IIH, n = 38 and non-IIH, n = 1), positional headache (IIH, n = 40 and non-IIH, n = 29), aggravated by straining (IIH, n = 24 and non-IIH, n = 14).
ICHD-3 criteria applicability
In those with IIH and a new or worsened headache temporally related to the diagnosis, 92.9% fulfilled the ICHD-3 criteria (91/98, Table 1). Including those with stable pre-existing headache, 70.5% of patients with IIH fulfilled the criteria (91/129). No non-IIH patients met the criteria overall. Sensitivity of ICHD-3 criteria for IIH headache was 92.9%, and specificity was 100% (Figure 2). Criteria were met by three of seven patients with suggested IIHWOP; four did not fulfill the criteria: one had no headache and three had headaches but not new/worsened, hence failing criterion A.
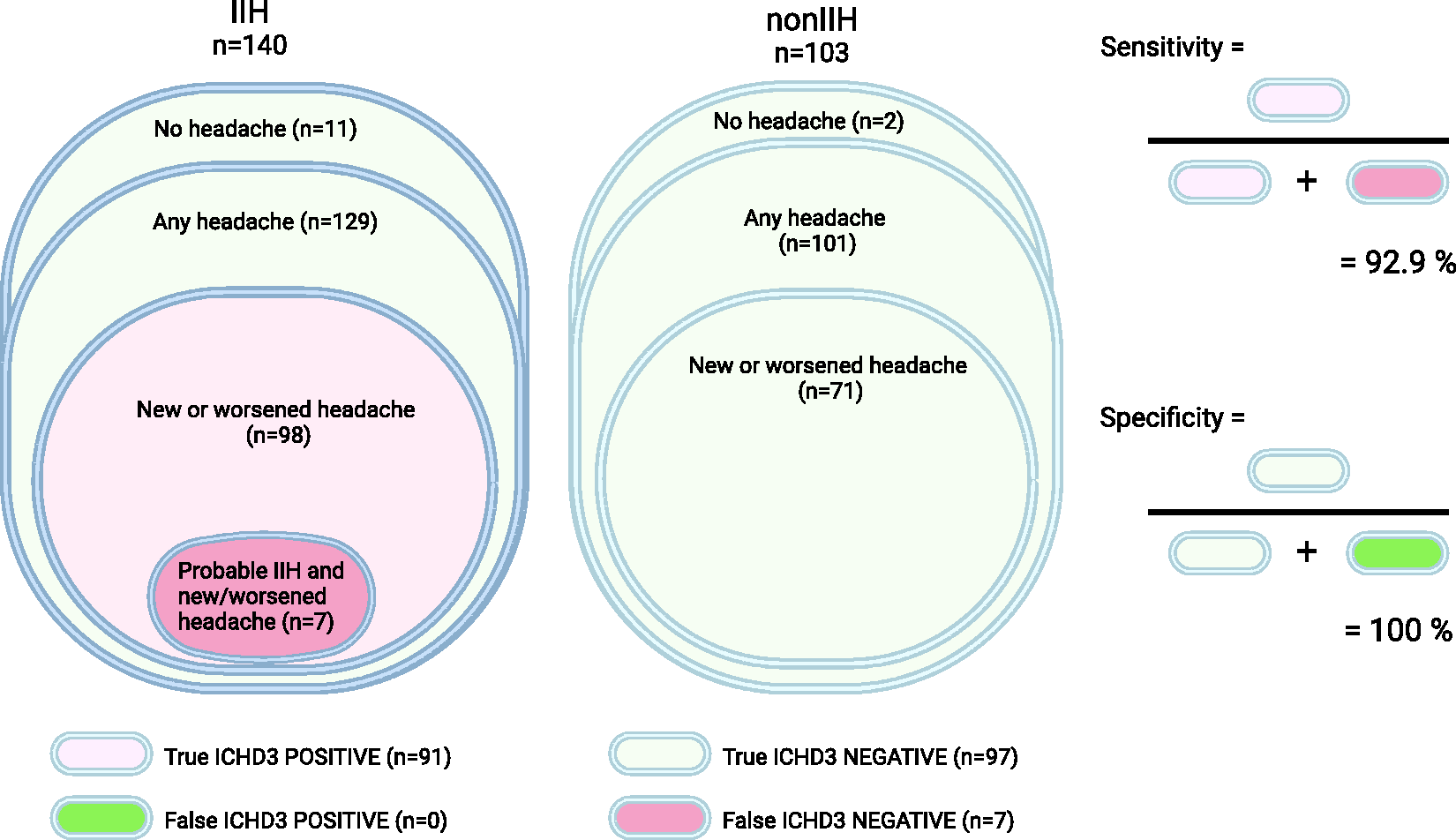
Application of ICHD-3 criteria for IIH headache in the cohort: Sensitivity and specificity. Created with Biorender.
Evaluating each criterion separately (Table 1), criterion A (a new or significantly worsened headache) was met by 77.2% of patients with IIH and 70.3% non-IIH (p = 0.78). Criterion B (IIH diagnosed and OP ≥ 25 cm CSF) was met by 96.9% with IIH and 0% non-IIH, but 39.6% (n = 40) of non-IIH with headache had an OP ≥ 25 cm CSF. Criterion C (new/worsened headache in temporal relation to IIH diagnostic work-up or headache with pulsatile tinnitus and/or papilledema) was met by 99.2% with IIH and 81.2% of non-IIH (p < 0.0001). Headache with pulsatile tinnitus (criterion C.2.a) was significantly more prevalent among those with IIH versus non-IIH (61.6% versus 29.0%, p < 0.0001), but these patients already fulfilled criterion C by means of their headache developing in relation to the diagnosis (criterion C.1).
Discussion
In line with previous findings (10,11,13,20), we report that 92% of patients with new-onset IIH had headache, and in 77% the headache was new or worsened at time of diagnosis.
Sensitivity and specificity of ICHD-3 criteria for IIH headache
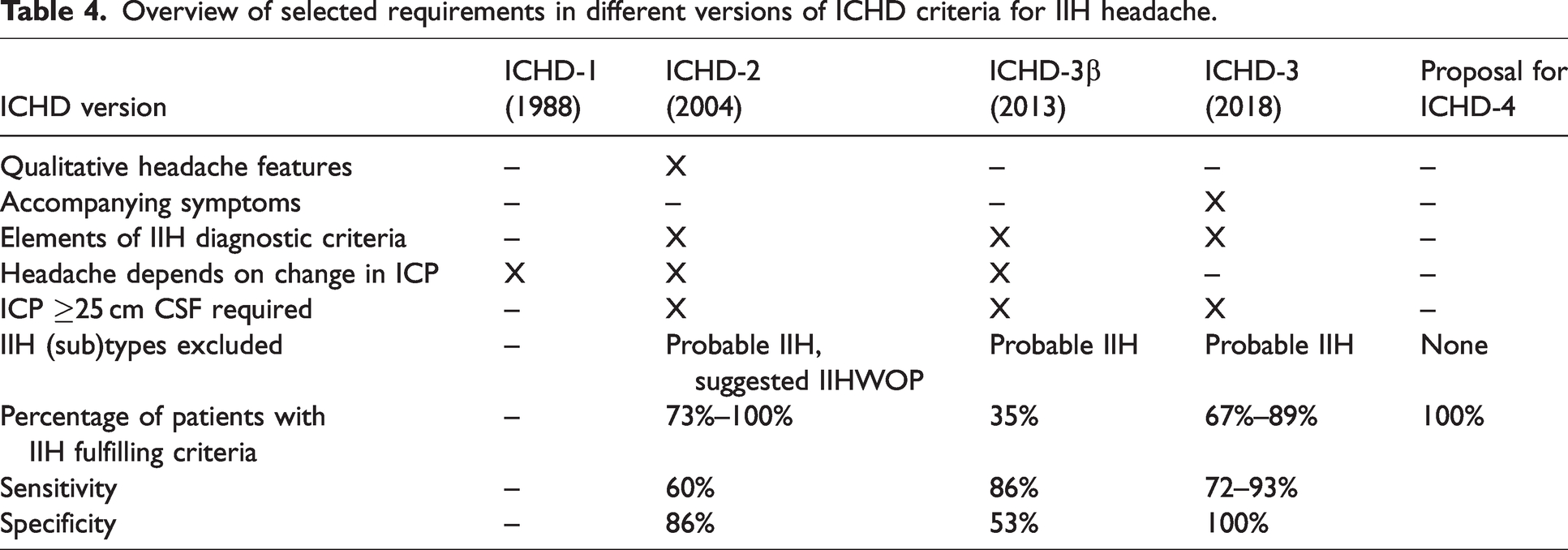
To our knowledge, the ICHD-3 criteria for IIH headache have never been field-tested in adults with IIH. We found a sensitivity of 92.9% and a specificity of 100%. Sensitivity was limited by the subgroup of patients with probable IIH failing to meet the criterion of raised OP (B2). In principle, IIHWOP may well fulfill the ICHD-3 criteria if considered “IIH”, and three of seven did so in our cohort. Non-IIH did not meet the ICHD-3 criteria due to the absence of an IIH diagnosis; ignoring this requirement brought 28% (n = 28) of non-IIH to meet the criteria overall (i.e., non-IIH with a new/worsened headache and OP ≥ 25 cm CSF). This would apply in cases of erroneous IIH over-diagnosis which is frequent (21). Our data suggest superiority of the current ICHD-3 criteria for IIH headache compared previous ICHD versions (Table 4). In the first version of ICHD, specific criteria for IIH headache were not formulated. In ICHD-2, sensitivity was 60% and specificity was 86% when tested in 44 patients with new-onset IIH (22). With ICHD-3 beta, sensitivity increased to 86% but specificity decreased to 53% (22) in the same IIH population. In 2015, new criteria were proposed (22) with a more extensive list of qualitative headache features and accompanying symptoms which achieved a more favorable sensitivity of 95% and specificity of 65%. Suggested sub-criteria of pulsatile tinnitus and/papilledema were implemented in the following ICHD-3 version; these symptoms did not add to sensitivity or specificity in our cohort, though. This may be because we evaluated patients only at time of diagnosis: here, criterion A and C1 are identical and the criterion of pulsatile tinnitus and/or papilledema co-existing with headache (C2) is redundant. But in the ICHD-3 criteria, headache is not required to evolve in temporal relation to the IIH diagnosis, nor does headache necessarily coincide with papilledema if an IIH diagnosis has been given at some point. In this scenario, OP ≥ 25 cm CSF and pulsatile tinnitus are markers of presumed active IIH and may, therefore, be relevant in headache related to IIH relapse. Our data supports that pulsatile tinnitus is a marker of IIH, as well as transient visual obscurations (Table 2). To elucidate the importance of such accompanying symptoms, ICHD-3 criteria for IIH headache could be field-tested in a heterogenous IIH population with active IIH of varying duration. Specificity is not expected to diminish, as a diagnosis of IIH is required.
Overview of selected requirements in different versions of ICHD criteria for IIH headache.
For future classification, ‘active IIH’ should be clearly defined, and it should be clarified whether headache that evolves after ocular remission should be attributed to IIH. Friedman (23) showed a significant emergence of headaches in patients after stabilizing treatment or remission of IIH symptoms.
To conclude, sensitivity and specificity of current ICHD-3 criteria for IIH headache are excellent in new-onset IIH. Our finding agrees well with the sensitivity of 89% found in a pediatric IIH population (24) (n = 27).
Cerebrospinal fluid pressure and headache
CSF OP has been included in all versions of ICHD-criteria for IIH headache (Table 4); it is important to clarify the eligibility hereof. Insights into mechanisms governing ICP and how it relates to headache and the lumbar OP are currently very limited.
Historically, either an OP ≥ 25 cm CSF or a dynamic dependency and reversibility of headache as function of CSF pressure has been mandatory in IIH headache criteria. In the first version of ICHD, headache attributed to “high cerebrospinal fluid pressure” required “headache intensity and frequency related to variations of intracranial pressure with a time lag of less than 24 hours”. ICHD-2 and ICHD-3 require OP ≥ 25 cm CSF, whereas ICHD-3-beta required relief/worsening of headache by reduction/increase in ICP.
Patients with IIH in our cohort had significantly higher lumbar OP than clinical mimicries, but 41% (n = 42) of non-IIH had OP ≥ 25 cm CSF. This may be explained by their mean BMI of 40.3, and some had intracranial pathologies. Elevated OP has previously been found in 25–43% (25,26) of patients with chronic headache which was a significant morbidity amongst non-IIH in our cohort. Also, obesity has been linked to elevated OP. Reports are divergent, though (27–30) and most studies are limited by lack of standardization and missing information on OP measurement technique. The only study (29) specifying, that patients were outstretched (neck and hip extension) in a lateral decubitus position during the lumbar puncture found no correlation between BMI and OP. In our cohort, IIH and non-IIH were equally obese. Curiously, we found that OP correlated with BMI only in the non-IIH group (r = 0.42, p < 0.0001), but not in the IIH group (r = 0.03, p = 0.73). This phenomenon has been observed previously (28). It may suggest loss of compensatory mechanisms governing OP in IIH or reflect flattening of the pressure-volume curve due to tissue compliance alterations at the extreme values of OP seen in IIH (31).
Reversibility of headache as a function of cerebrospinal fluid pressure
In our cohort we saw a tendency towards more pronounced relief by LP in IIH than in non-IIH (46% vs. 30%, p = 0.04) in agreement with previous findings (22). Curiously, De Simone et al. (26) found that 77% of patients with migraine also experienced headache relief by lumbar puncture.
Furthermore, despite remission of papilledema, a significant proportion of IIH patients experience headache chronification with persistent headaches described in 43–69% of patients (5,10,11,23,32,33). This argues against dynamic reversible relations between ICP change and headache as a rule-of-thumb in IIH, as a reduced ICP is expected to condition ocular remission.
Also, it emphasizes that criteria of IIH headache should be reserved for states of active disease. If headache persists despite ocular remission and normalization of ICP, we suggest that the Appendix A7.9 criteria of Persistent headache attributed to past non-vascular intracranial disorder should be applied. A substantial proportion of the IIH population is expected to fulfill these and future investigations of these appendix criteria are warranted. This will differentiate IIH headache during active disease from post-IIH headache which has implications for pharmacological treatment: acetazolamide is primarily relevant during active IIH, whereas other headache preventives may be more appropriate and beneficial in chronified post-IIH headache.
Caution has been raised about including OP in the context of headache criteria (34). We agree that OP is inappropriate in the context of headache classification. As the OP is already covered by the diagnostic criteria of IIH, we advocate for the segregation of diagnostic criteria from headache criteria in IIH. This will allow for flexible independent adjustments of each as evidence evolves; recently, revised diagnostic criteria were proposed for IIH (18).
Headache phenotype
We found that IIH headache resembled migraine in 36%, tension-type headache in 23% and was non-classifiable in 41%, and that headache phenotype did not aid in differentiating IIH from non-IIH. Importantly, positional (supine/morning) headache and aggravation by straining was not more common in IIH.
Similarly, in a population of 108 patients with new-onset IIH, migraine-like headache debuted after the IIH diagnosis in 37% previously ‘migraine-naïve’ (12). Previous studies have reported higher rates of migraine-like headache in IIH ranging between 52–90% (5–7,11). These studies did not distinguish new/worsened headaches from stable pre-existing ones, though, and thus reported the cumulative headache burden at varying times throughout the disease. Our finding of 23% tension-type headache in IIH is nearly similar to the IIHTT (22%) (11). Headache phenotype in IIH has to some degree also been shown to change over time (10). Onset and evolution of pathophysiology and symptoms in IIH is largely unknown and this challenges the interpretation of causality and temporality regarding headaches. Diagnostic delay could mask a temporal relation and prolong the headache phase for months or years. Meanwhile, the headache could erroneously be interpreted as a primary headache disorder, and it may have changed phenotypic presentation at time of eventual diagnosis. This complicates IIH headache research.
Considering this heterogeneous dynamic phenotypic presentation, eligibility of specific headache features in ICHD criteria is questionable. In ICHD-2, a minimum of one specific qualitative headache feature was required, namely, i) daily occurrence, ii) a diffuse and/or constant (non-pulsating) pain and/or iii) aggravation by coughing or straining. These criteria were field tested among 22 patients with IIH of which 14 patients had headache, all of whom fulfilled the criteria (35). Other studies have found 73% (36) and 80% (37) of patients with IIH (n = 40) to fulfill the ICHD-2 criteria. Curiously, in 44 patients with chronic migraine and sinus stenosis (but no papilledema) (26) almost the same rate (70.5%) fulfilled ICHD-2 criteria. This stresses the need for testing sensitivity and specificity of criteria. Requirements of qualitative headache features were removed in the more simplified ICHD-3-beta version and not implemented in ICHD-3. Our data support the appropriateness of this as IIH and non-IIH patients had equally frequent headache (≥15 days monthly), were equally aggravated by straining, and presented with identical ICHD phenotype. In line with the preface of secondary headaches in the ICHD-3, colleagues have argued that qualitative headache characteristics are informative and helpful, and they described the disappearance of these from the current ICHD-3 as a regression (38). However, it seems that there simply is no homogenous clinical phenotype in IIH headache.
In clinical practice, any new or worsened headache that develops in temporal relation to discovery of IIH suggests causality regardless of phenotype and accompanying symptoms. On the other hand, IIH and (chronic) migraine share several phenotypic features (young, female, obese), and the question is whether these disorders co-exist incidentally or exist in a common spectrum of shared underlying pathophysiology where high ICP may act as a trigger of a latent primary headache disorder by lowering the threshold for onset or worsening.
Methodological considerations
We characterize a large prospective cohort of patients with new-onset IIH and compare to clinical mimicries. Extensive standardized diagnostic work-up was done by IIH specialists at tertiary centers of headache and neuro-ophthalmology. This ensured high quality; however, it may have introduced selection bias of the patient population towards more severely affected patients with IIH and more complex clinical mimicries favoring those with a substantial headache burden with certain features including morning- and positional headaches. Thus, it is no surprise that non-IIH patients were equally burdened by headache. The non-IIH population cannot serve as healthy controls as they are characterized by a proportional burden of headache-, neurological-, and/or ophthalmological disorders. It is, however, crucial to include such a group as they constitute the actual real-world diagnostic dilemma. IIH and mimicries were comparably obese, and obesity is a risk factor for migraine and its chronification (8) which may explain the high proportion of chronic headache in non-IIH. It also shows that headache is an important symptom eliciting clinical suspicion of IIH.
Medication overuse is prevalent in IIH with reports of 37% at time of diagnosis (11) and 48% long term (39). Our inability to assess medication overuse is an important limitation. IIH headache may lead to long-term analgesic overconsumption preceding diagnostic work-up. This may blur the phenotypic presentation and headache severity.
Conclusion
We validate for the first time ICHD-3 criteria for IIH headache in new-onset adult IIH We demonstrate high sensitivity and specificity of criteria but propose a simplification by omission of pulsatile tinnitus, papilledema, and cerebrospinal fluid opening pressure. The latter two are covered by diagnostic criteria of IIH, which we suggest segregated from headache criteria. Pulsatile tinnitus did not improve criteria accuracy, and headache phenotype in IIH is heterogenous and unspecific. Thus, a new or significantly worsened headache evolving in temporal relation to active IIH is proposed as future criteria (Table 5).
Proposed future criteria for IIH headache.

ICHD-3 criteria should be tested in cohorts covering the entire course of active disease including relapse. If headache persists despite remission of papilledema, the Appendix A7.9 criteria of Persistent headache attributed to past non-vascular intracranial disorder should be applied and future validation hereof is warranted.
Article highlights
ICHD-3 criteria for IIH headache are sensitive and specific. Further simplification is possible without compromising accuracy. We suggest segregation of IIH diagnostic criteria from IIH headache criteria. Opening pressure and papilledema are covered by the diagnostic criteria and may be omitted from headache criteria. Opening pressure is uncertain in the context of headache disorders.
Footnotes
Authors contributions
NSH drafted the first manuscript and analyzed the data; NSH, JJK, and DB collected and cleansed the data; DB and RHJ supervised data registration and interpretation; all authors contributed with the design of the study and with critical revisions of the manuscript and gave their final approvement.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: NSH received funding from the Novo Nordic Foundation during the conduction of the work and gave lectures for Pfizer, University of Southern Denmark, and University of Copenhagen. JJK received funding from the Lundbeck Foundation, Rigshospitalet-Glostrup, and Odense University hospital for the duration of the study. HMY received support from Abbvie for participation in non-related courses. RHJ gave lectures for Pfizer, Eli-Lilly, Merck, TEVA, Novartis, Lundbeck and Allergan; was investigator in clinical trials with Eli-Lilly, Novartis and Lundbeck. She is the Director of Danish Headache Center, Lifting The Global Burden of Headache and Founder of Master of Headache Disorders at University of Copenhagen and received research funding from University of Copenhagen, Rigshospitalet, Lundbeck Foundation, The Medical Society in Copenhagen, NovoNordisk Foundation and Tryg Foundation. DB gave lectures for TEVA, Novartis, Pfizer. Received travel support from Allergan, TEVA, Pfizer, Abbvie; was in advisory boards for Novartis, Lilly, Teva, Lundbeck, Pfizer, Abbvie and participated in clinical trials for TEVA, Lundbeck, Novartis, Lilly, Novo Nordic Foundation outside the submitted work. Received grants from Odense University Hospital and Rigshospitalet, University of Copenhagen during the conduction of the work.
Funding
The authors received the following financial support for the research, authorship, and/or publication of this article: NSH received funding from the Novo Nordis Foundation during the conduction of the work. JJK received funding from the Lundbeck Foundation, Rigshospitalet-Glostrup, and Odense University Hospital for the duration of the study. RHJ received research funding from University of Copenhagen, Rigshospitalet, Lundbeck Foundation, the medical society in Copenhagen, Novo Nordis Foundation, Tryg Foundation. DB received grants from Odense University Hospital and Rigshospitalet-Glostrup during the conduction of the work.
