Abstract
Background
Idiopathic intracranial hypertension (IIH) occurs more frequently in obese females of childbearing age. A link between eating disorders and poor outcome has been suggested but remains unproven.
Methods
This prospective field study at two tertiary headache centers included patients with clinically suspected IIH after standardized diagnostic work-up. Eating disorders were evaluated using validated questionnaires (EDQs). Primary outcome was the impact of eating disorders on IIH severity and outcome, secondary outcome was the prevalence and type of eating disorders in IIH compared to controls.
Results
We screened 326 patients; 143 patients replied to the EDQs and were classified as ‘IIH’ or ‘non-IIH’ patients. The demographic profile of EDQ-respondents and non-respondents was similar. Presence of an eating disorder did not impact IIH severity (lumbar puncture opening pressure (p = 0.63), perimetric mean deviation (p = 0.18), papilledema (Frisén grad 1–3; p = 0.53)) nor IIH outcome (optic nerve atrophy (p = 0.6), impaired visual fields (p = 0.18)). Moreover, we found no differences in the prevalence and type of eating disorders when comparing IIH with non-IIH patients (p = 0.09).
Conclusion
Eating disorders did not affect IIH severity or outcome. We found the same prevalence and distribution pattern of eating disorders in IIH and non-IIH patients advocating against a direct link between IIH and eating disorders.
Introduction
Idiopathic intracranial hypertension (IIH) is characterized by increased intracranial pressure (ICP) of unknown cause (1). The IIH population predominantly consists of obese women of childbearing age, with an incidence of 20/100,000 compared to 0.5–2/100,000 in the background population (2). Patients experience chronic headache, cognitive impairment (3), pulsatile tinnitus and visual disturbances, the latter due to bilateral papilledema. Fulminant disease or delayed treatment may result in permanent visual loss (4), and chronic invalidating headache, cognitive decline and impaired visual function are common long-term sequelae. Definite IIH is diagnosed based on the presence of papilledema, elevated lumbar puncture opening pressure (OP ≥25 cmH2O), normal neurological exam except abducens nerve palsy, normal cerebrospinal fluid (CSF) composition, normal neuroimaging including cerebral venography and the absence of any known cause of elevated ICP (5,6).
IIH has been linked to obesity in young females of childbearing age and in line with this, IIH in female patients with a mean body mass index (BMI) ≥30 kg/m2 is defined as ‘classic IIH’ (7). Currently, the burden of IIH is growing rapidly due to pandemic obesity (4). In Denmark, the number of overweight and obese women increased from 39.4% in 2010 to 46.2% in 2021 (8). The most common eating disorders in Denmark are anorexia nervosa, bulimia nervosa and binge-eating disorder (BED). BED is strongly associated with obesity and metabolic syndrome (9), whilst anorexia and bulimia cannot be linked to metabolic syndrome. Only recently, in 2022, BED was implemented as a diagnosis in Denmark and is estimated to affect 2–3% of the population (10). As well, a meta-analysis from 2020 found a significant association between obesity and eating disorders (11), and that young women who dieted were five times more likely to develop an eating disorder (12).
As IIH is more common in obese females of childbearing age (7) and the primary disease modifying treatment is weight-loss, it is relevant to consider its association with eating disorders. Obese individuals have a higher prevalence of eating disorders than their peers of lower weight (13), which might impede or delay successful weight loss and deteriorate the patients’ outcome. Interestingly, different types of obesity are hypothesized in IIH patients and the obese background population, as adiposity in IIH patients is mostly truncal and evidence suggests that metabolic perturbations driving obesity in IIH are distinct from those in simple obesity (14).
However, an association of eating disorders with IIH and its specific phenotype of obesity has not been investigated yet but research on this possible association is needed to optimize treatment and to minimize sequelae. We wanted to investigate the impact of eating disorders on IIH severity and outcome. Secondary outcome was the prevalence and distribution pattern of eating disorders in IIH compared to non-IIH patients.
Methods
Inclusion of participants
We performed a cross-sectional study continuously including patients from two specialized headache centers in Denmark (Department of Neurology, Odense University Hospital; Danish Headache Center, Rigshospitalet-Glostrup) with multi-disciplinary expertise in IIH from January 2018 to January 2023. The inclusion criteria were diagnosis of clinically suspected IIH, ≥18 years of age and ability to provide written, informed consent. Clinically suspected IIH was assessed by DB, RHJ, NSH, and JJK and defined as a combination of some of the following symptoms: headache, obesity, papilledema, subjective visual impairment, pulsatile tinnitus, double vision. Exclusion criteria were age < 18 years, inability to provide informed consent, absence of diagnosis, missing response to the eating disorder questionnaires (EDQs), IIH relapse, pregnancy, and secondary pseudotumor cerebri syndrome (PTCS). Exclusion based on absence of diagnosis accounted for cases where a diagnosis could not be made due to missing data. Patients fulfilling the diagnostic criteria but simultaneously presenting with an independent disease likely causing the elevation of the ICP were classified as secondary PTCS and excluded.
Patients were classified by DB and RHJ according to the Friedman criteria (5,7). Eligible patients were subjected to a semi-structured medical interview, neurological exam, neuro-ophthalmological exam, lumbar puncture, routine analysis of blood and CSF, electrocardiogram, and neuroimaging including Magnetic Resonance Imaging (MRI) and Magnetic Resonance-venography (MR-V) or cerebral computed tomography venography (CT-V). Neuroimaging and neuroophthalmological exams were evaluated by LH/LDM.
Patients were divided into two groups:
Patients fulfilling the diagnostic criteria were classified as definite IIH, probable IIH, IIH without papilledema (IIHWOP), suggested IIHWOP and are hereinafter called ‘IIH patients’ Patients not fulfilling the diagnostic criteria were classified as ‘non-IIH patients’
Ophthalmological assessment
Ophthalmological assessment was performed as described elsewhere (3). Ophthalmological remission was defined as remission of bilateral papilledema with stable thickness of the optic nerve measured with papillary optical coherence tomography (OCT) combined with either normal visual fields or stable impairment assessed with automated computer perimetry. Sequelae at remission were defined as optic nerve atrophy and/or impaired visual field of at least one eye.
Lumbar puncture
Lumbar puncture was done in a standardized manner as described elsewhere (6).
Radiological diagnostics
Radiological diagnostics were done as described elsewhere (6).
Psychiatric comorbidity
Patients were screened for psychiatric comorbidity as published elsewhere (3).
Identification of eating disorders/eating disorder behavior
To identify patients with eating disorders or eating disorder behavior, patients received three separate questionnaires sent automatically to their private email by the Open Patient data Explorative Network (OPEN) Redcap database: the SCOFF (Sick, Control, One, Fat Food-questionnaire), EDE-Q (Eating Disorder Examination Questionnaire) and BED-Q (Binge Eating Disorder Questionnaire), all of which in the following are named EDQs (Table 1).
Overview of the eating disorder questionnaires.

The SCOFF-questionnaire was used to screen for eating disorder symptoms/behavior. To further specify the eating disorder pathology, EDE-Q was used to assess symptoms of anorexia nervosa, bulimia nervosa and binge eating disorder (BED), according to the DSM-5 criteria. BED-Q was used to assess symptoms of BED, and thus the prevalence of eating disorder symptoms associated with being overweight. Both SCOFF and EDE-Q have been validated in Danish (15,16). The BED-Q is an inhouse questionnaire, and is yet to be validated in Danish (16).
The outcome of the questionnaires was considered positive (indicating an eating disorder), when patients presented with at least one positive eating disorder questionnaire (EDQ).
Medication associated with weight gain
As a conceivable risk factor for obesity, we assessed the use of medication associated with weight gain defined as medical drugs, used daily at time of diagnosis, where weight gain as a side effect is common (1–10%) or very common (>10%) (17). We excluded common contraceptives (progesterone only or combined common contraceptives pills, and intrauterine devices).
Statistical analysis
Primary outcome was the impact of eating disorders on IIH severity and outcome. IIH severity was presented by lumbar puncture opening pressure (OP), papilledema of the worst eye (Frisén grad 1–3 and ≥4) (15,16), perimetric mean deviation (PMD) of the worst eye and treatment modality (medical and surgical treatment). Outcome covers sequela of papilledema, herein optic nerve atrophy and impaired visual fields. Secondary outcome was the prevalence and distribution pattern of eating disorders in IIH compared to non-IIH patients.
We especially focused on eating disorders causing obesity, i.e. BED (20).
The SPSS, version 28.0.0.0 software was used. Pearson’s Chi-squared test, Fishers exact test and Mann-Whitney U test were used appropriately. To correct for multiple comparisons, the Holm-Bonferroni method was used. We considered p < 0.05 to be statistically significant. P-values, which changed from significant to non-significant due to Holm-Bonferroni correction, are marked “p′” in the tables. We used binary logistic regression to control for confounders. The upper limit of the standard issue manometer used to measure OP is 50 cm CSF; 40 patients included had an OP above 50 cm CSF. For measurements above this, the maximum readable level on the manometer was used for analysis. OP may therefore be underestimated in this study.
Standard protocol approvals, registrations and patient consent
The study was approved by the Ethics Committee of Southern Denmark and the Danish Data Protection Agency. All participants provided written informed consent before inclusion. Data reporting was in line with the STROBE (Reporting of Observational Studies in Epidemiology) recommendations (21).
Data availability
De-identified participant data can be shared upon request from qualified researchers who provide a methodologically sound proposal. Legal requirements state that a data processing agreement is signed and approved by the data protection office in the Region of Southern Denmark. Data is available two years after publication. Requests should be directed at the corresponding author.
Results
We screened 326 patients with clinically suspected IIH (Figure 1). We excluded 177 (54%) patients who did not respond to the EDQs; 149 patients responded to the EDQs yielding a response rate of 46% (Figure 1). Of the 149 responders, a total of 28 patients were excluded, due to missing data impeding diagnosis (n = 6) or a diagnosis of secondary PTCS (n = 22; Figure 1).

Patient inclusion.
The remaining 121 patients were classified as definite IIH (n = 70), probable IIH (n = 6) and suggested IIH without papilledema (n = 1) (hereinafter referred to as IIH patients; Table 2). In 44 patients, the diagnosis was disproven (non-IIH patients; n = 44; Figure 1 and Table 2). The alternative diagnoses in the non-IIH group included episodic or chronic migraine/tension-type headache (n = 24; 54.5%), secondary headache disorders such as posttraumatic headache (n = 1; 2.3%), pseudopapilledema (n = 2; 4.6%), as well as other secondary causes (n = 5; 11.4%). In 12 patients, IIH was ruled out, but a definitive diagnosis was not reached due to nonspecific and intermittent symptoms (27.3%).
Assessment for selection bias.

*completed university degree; standard deviation (SD), body mass index (BMI)
Missing answers excluded: Age (EDQ-complete [n = 1], EDQ-incomplete [n = 4]), Weight (EDQ-complete [n = 4], EQD-incomplete [n = 10]), BMI (EDQ-complete [n = 4], EQD-incomplete [n = 10]), Smoking (EDQ-complete [n = 7], EQD-incomplete [n = 5]), Alcohol (EDQ-complete [n = 7], EQD-incomplete [n = 9]), Drug abuse (EDQ-complete [n = 23], EQD-incomplete [n = 31]), Region (EDQ-complete [n = 8], EQD-incomplete [n = 9]), Education (EDQ-complete [n = 38], EQD-incomplete [n = 38]).
To assess a potential selection bias with our reported results, we compared the patients who completed the EDQs (EDQ-complete) with the patients who did not (EDQ-incomplete; Table 2). Patients with missing data impeding diagnosis (n = 2 (EDQ-incomplete), n = 6 (EDQ-complete); Figure 1) were excluded from the analyses. Patients responding to the EDQs did not differ from non-responding patients in terms of demographic variables (Table 2).
Disease severity and outcome of IIH patients is not affected by concomitant eating disorders or eating disorder behavior
To investigate the impact of eating disorders and eating disorder behavior on the outcome of IIH patients, we compared IIH patients suffering from an eating disorder or eating disorder behavior (IIH + E) with IIH patients without (IIH − E) (Table 3). Patients in both groups were obese (BMI 38.5 vs. 35.3 kg/m2; p′ = 1), largely female (97.6 vs. 97.1%; p = 0.9) and of childbearing age (29 vs. 33 years; p′ = 0.44) (Table 3). Moreover, there were no differences between the two groups in terms of smoking, consumption of alcohol, drug abuse, psychiatric comorbidity or use of medication related to weight gain (Table 3).
Comparison between IIH + E and IIH − E patients.

Standard deviation (SD), perimetric mean deviation (PMD), ventriculo-peritoneal shunt (VP-shunt),
ventriculo-atrial shunt (VA-shunt), optic nerve sheath fenestration (ONSF), body mass index (BMI)
Missing answers excluded: Headache >15 days/month (IIH + E [n = 6], IIH − E [n = 9]), Smoking (IIH + E [n = 1], IIH − E [n = 1]), Alcohol (IIH + E [n = 1], IIH − E [n = 1]), Drug abuse (IIH + E [n = 7], IIH − E [n = 4]), Papilledema worst eye (IIH + E [n = 4], IIH − E [n = 4]), PMD worst eye (IIH + E [n = 19], IIH − E [n = 16]), Medical treatment (IIH + E [n = 2], IIH − E [n = 1]), Surgical treatment (IIH + E [n = 2], IIH − E [n = 2]), Loss of weight at remission (IIH + E [n = 9], IIH − E [n = 10]), Time to remission (IIH + E [n92], IIH − E [n = 7]), Sequalae optic nerve atrophy (IIH + E [n = 15], IIH − E [n = 11]), Sequalae impaired visual field (IIH + E [n = 14], IIH − E [n = 11]), BMI (IIH − E [n = 2]), Use of medication related to weight gain (IIH + E [n = 13], IIH − E [n = 17]).
Antidepressants, such as selective serotonin reuptake inhibitors, serotonin–norepinephrine reuptake inhibitors, noradrenergic and specific serotonergic antidepressants, and tricyclic antidepressants, as well as second-generation antipsychotics, lithium, and pregabalin or gabapentin (treatment of pain or anxiety).
Definite IIH was the most frequent diagnosis in both groups (90.5 vs. 91.4%; p = 0.46). Unexpectedly, we did not find any differences between IIH + E and IIH − E patients regarding disease severity at time of diagnosis as reflected by OP (38 cm CSF versus 37 cm CSF; p = 0.63), papilledema of the worst eye (Frisén grade 1–3: p = 0.53/Frisén grade ≥4: p = 0.31), PMD of the worst eye (−5.96 db versus −5.76 db; p = 0.18) and treatment modality (medical treatment (p = 0.25), surgical treatment (p = 0.61)). Both groups reported chronic headaches (69.4% versus 65.4%; p = 0.74) (Table 3).
In line, IIH + E and IIH − E were similar concerning weight loss at ophthalmological remission (7.8 kg versus 5.98 kg; p = 0.44), time to remission (11.2 months versus 9.46 months; p = 0.95) and sequela (optic nerve atrophy (37% versus 25%; p = 0.60), impaired visual fields (50% versus 25%, p = 0.18)) (Table 3).
Disease severity and outcome of ‘classic’ IIH patients is neither affected by concomitant eating disorders nor eating disorder behavior
In order to exclude ‘atypical IIH patients’ as possible confounders, we excluded men (n = 2) and patients with a BMI < 30 kg/m2 (n = 12) and performed sub-analyses of ‘classic’ female IIH patients with a BMI ≥ 30 kg/m2. We then compared classic IIH+E with classic IIH-E patients (Table 4). There was no difference in terms of mean age (29 years versus 33 years, p′ = 0.48) and BMI (39.4 versus 37.2 kg/m2, p = 0.15), nor in smoking, consumption of alcohol, drug abuse, psychiatric comorbidity or use of medications related to weight gain. Definite IIH diagnosis (89.2% versus 88.9%, p = 0.46) was the most frequent diagnosis. Patients presented equally frequent with chronic headache (71% versus 61.1%, p = 0.48). Also, we did not find any differences regarding disease severity, sequelae (Table 4), time to remission or loss of weight at remission.
Comparison between IIH + E and IIH − E patients, BMI ≥30 kg/m2.
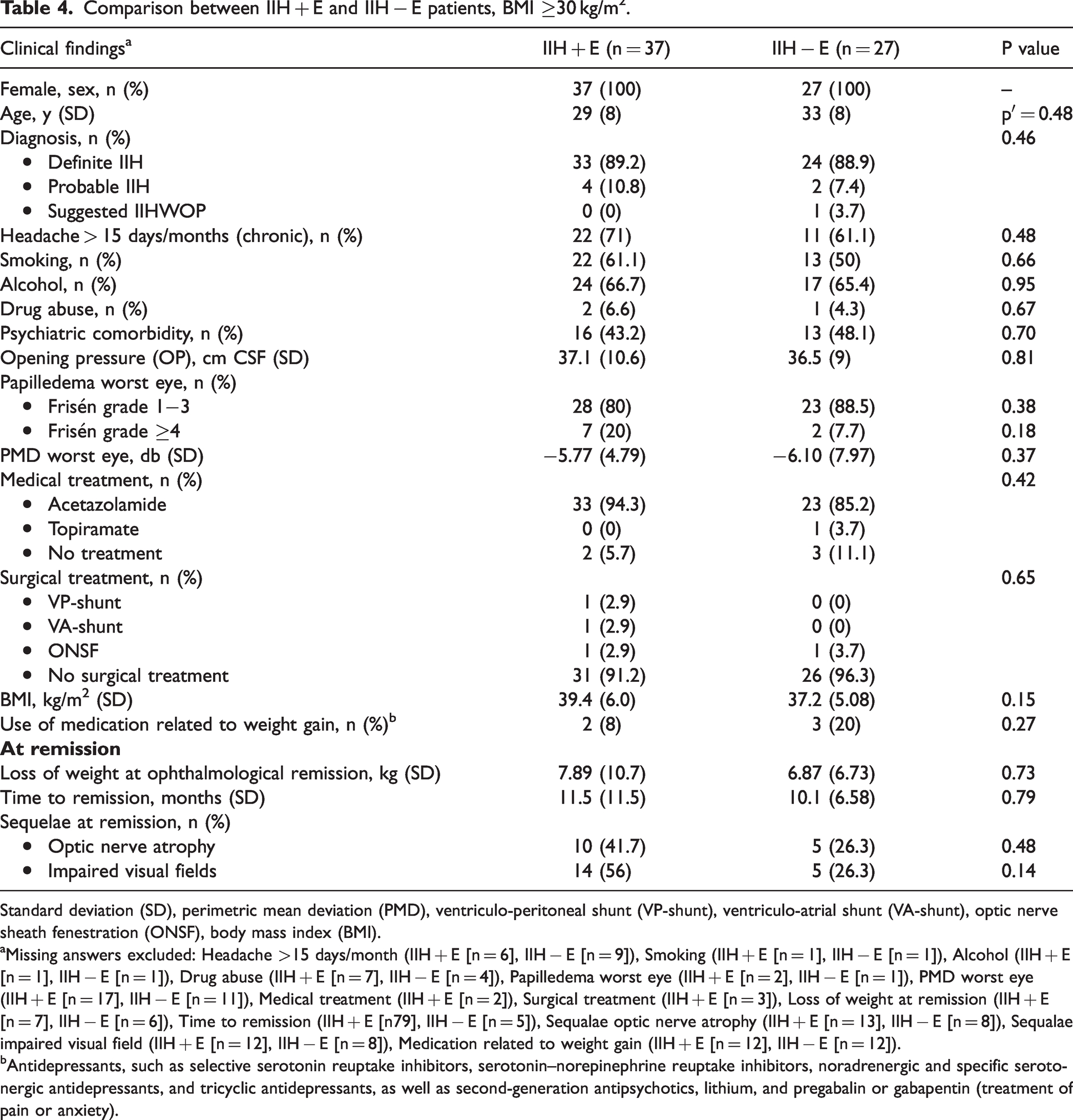
Standard deviation (SD), perimetric mean deviation (PMD), ventriculo-peritoneal shunt (VP-shunt), ventriculo-atrial shunt (VA-shunt), optic nerve sheath fenestration (ONSF), body mass index (BMI).
Missing answers excluded: Headache >15 days/month (IIH + E [n = 6], IIH − E [n = 9]), Smoking (IIH + E [n = 1], IIH − E [n = 1]), Alcohol (IIH + E [n = 1], IIH − E [n = 1]), Drug abuse (IIH + E [n = 7], IIH − E [n = 4]), Papilledema worst eye (IIH + E [n = 2], IIH − E [n = 1]), PMD worst eye (IIH + E [n = 17], IIH − E [n = 11]), Medical treatment (IIH + E [n = 2]), Surgical treatment (IIH + E [n = 3]), Loss of weight at remission (IIH + E [n = 7], IIH − E [n = 6]), Time to remission (IIH + E [n79], IIH − E [n = 5]), Sequalae optic nerve atrophy (IIH + E [n = 13], IIH − E [n = 8]), Sequalae impaired visual field (IIH + E [n = 12], IIH − E [n = 8]), Medication related to weight gain (IIH + E [n = 12], IIH − E [n = 12]).
Antidepressants, such as selective serotonin reuptake inhibitors, serotonin–norepinephrine reuptake inhibitors, noradrenergic and specific serotonergic antidepressants, and tricyclic antidepressants, as well as second-generation antipsychotics, lithium, and pregabalin or gabapentin (treatment of pain or anxiety).
IIH patients and non-IIH patients show the same prevalence and distribution pattern of eating disorders and eating disorder behavior
Demographic data
We did not find any differences between IIH- and non-IIH-patients in terms of sex (97.4 vs. 100% female; p = 0.28), age (31 vs. 32 years; p = 0.32), weight (BMI 37.1 vs. 37.5 kg/m2; p0.87), smoking, consumption of alcohol, drug abuse, psychiatric comorbidity, use of medication related to weight gain or place of residence (Table 5). As expected, IIH patients presented with elevated OP (37.5 versus 25.4 cm H2O, p < 0.001) and papilledema (p < 0.001). Both groups presented with chronic headache (67.7 vs. 75%; 0.45). Of the non-IIH patients, 12 patients (28.6%) received IIH specific medical treatment, in the form of acetazolamide due to a suspected IIH diagnosis, which was later disproven in all of them (Table 5).
Comparison between IIH and non-IIH patients.

SCOFF (Sick, Control, One, Fat Food)-questionnaire, EDE-Q (Eating Disorder Examination Questionnaire), BED-Q (Binge Eating Disorder Questionnaire), standard deviation (SD), perimetric mean deviation (PMD), ventriculo-peritoneal shunt (VP-shunt), ventriculo-atrial shunt (VA-shunt), optic nerve sheath fenestration (ONSF), body mass index (BMI).
Missing answers excluded: Weight (IIH [n = 2], Non-IIH [n = 1]), BMI (IIH [n = 2], Non-IIH [n = 1]), Smoking (IIH [n = 2], Non-IIH [n = 2]), Alcohol (IIH [n = 2], Non-IIH [n = 2]), Drug abuse (IIH [n = 11], Non-IIH [n = 8]), Region (IIH [n = 6], Non-IIH [n = 1]), Headache >15 days/month (IIH [n = 15], Non-IIH [n = 8]), OP (Non-IIH [n = 5]), Papilledema worst eye (IIH [n = 9, papilledema was confirmed, but not graded according to Frisen], Non-IIH [n = 2]), PMD worst eye (IIH [n = 35], Non-IIH [n = 44]), Medical treatment (IIH [n = 3], Non-IIH [n = 2]), Surgical treatment (IIH [n = 5], Non-IIH [n = 4]), Use of medication related to weight gain (IIH [n = 30], Non-IIH [n = 15]).
EDQs were sent to all patients simultaneously, thus the EDQs were done at different time-points in the course of the disease of the respective patients and not necessarily at baseline.
Antidepressants, such as selective serotonin reuptake inhibitors, serotonin–norepinephrine reuptake inhibitors, noradrenergic and specific serotonergic antidepressants, and tricyclic antidepressants, as well as second-generation antipsychotics, lithium, and pregabalin or gabapentin (treatment of pain or anxiety).
12 of the 12 patients received acetazolamid due to a suspicion of IIH, which was later disproven in all of them.
Prevalence and pattern of eating disorders and eating disorder behavior
IIH patients showed the same pattern and frequency of eating disorders and eating disorder behavior as their non-IIH counterparts of comparable age, weight and sex (Table 5). Of IIH patients, 54.5% presented with an indication of an eating (behavior) disorder compared to 70.5% in the non-IIH group (p = 0.09). Further 45.5% of IIH patients presented with positive SCOFF compared to 59.1% of the non-IIH controls (p = 0.15). BED-Q was positive in 16.9% of the IIH compared to 18.2% in the non-IIH group (p = 0.86). Remarkably, only 45.5% of IIH and 29.5% of non-IIH patients presented without eating (behavior) disorders at all (Table 5).
When comparing IIH patients with eating disorders and eating disorder behavior to non-IIH patients with eating disorders and eating disorder behavior (IIH + E vs. non-IIH + E, Table 6), IIH + E patients presented with papilledema Frisén grade 1−3 (81.6 versus 0%; p′ = 0.017) and elevated OP (mean 38 cm CSF versus mean 25 cm CSF, respectively; p′ = 0.017). Both groups presented with chronic headache (69.4 versus 70.8%; p = 0.91). However, demographic data were similar (Table 6).
Comparison between IIH + E and non-IIH + E patients.

SCOFF (Sick, Control, One, Fat Food)-questionnaire, EDE-Q (Eating Disorder Examination Questionnaire), BED-Q (Binge Eating Disorder Questionnaire), standard deviation (SD), perimetric mean deviation (PMD), ventriculo-peritoneal shunt (VP-shunt), ventriculo-atrial shunt (VA-shunt), optic nerve sheath fenestration (ONSF), body mass index (BMI).
Missing answers excluded: Headache >15 days/month (IIH + E [n = 6], Non-IIH + E [n = 7]), Smoking (IIH + E [n = 1], Non-IIH + E [n = 1]), Alcohol (IIH + E [n = 1], Non-IIH + E [n = 1]), Drug abuse (IIH + E [n = 7], Non-IIH + E [n = 6]), OP (Non-IIH + E [n = 3]), Papilledema worst eye (IIH + E [n = 4], Non-IIH + E [n = 1]), PMD worst eye (IIH + E [n = 19], Non-IIH + E [n = 31]), Medical treatment (IIH + E [n = 2], Non-IIH + E [n = 2]), Surgical treatment (IIH + E [n = 4], Non-IIH + E [n = 3]), BMI (Non-IIH + E [n = 1]), Use of medication related to weight gain (IIH + E [n = 13], Non-IIH + E [n = 11]).
EDQs were sent to all patients simultaneously, thus the EDQs were done at different time-points in the course of the disease of the respective patients and not necessarily at baseline.
Antidepressants, such as selective serotonin reuptake inhibitors, serotonin–norepinephrine reuptake inhibitors, noradrenergic and specific serotonergic antidepressants, and tricyclic antidepressants, as well as second-generation antipsychotics, lithium, and pregabalin or gabapentin (treatment of pain or anxiety).
To account for the possible interaction between chronic headache, psychiatric disorders and obesity, we performed a binary logistic regression controlling for chronic headache, psychiatric disorders, BMI, and age, which confirmed the null association in line with recent publications (3). Moreover, we repeated our analyses including only patients with eating disorders or eating disorder behavior with ≥2 positive questionnaires. However, this did not change the results (Online Supplementary Table 1−4).
Discussion
Our study suggests that eating disorders and eating disorder behavior seem not to affect disease severity, weight loss or short-term ophthalmologic outcome in IIH patients. Moreover, we found a similar prevalence and distribution pattern of eating disorders and eating disorder behavior in IIH and non-IIH patients arguing against a direct link between IIH and eating disorders.
Our study is the first prospective cross-sectional multicenter study conducted on the relationship between IIH and eating disorders in adults. Previous case control studies have been performed in children (22). In our study, IIH patients from two tertiary headache centers in Denmark were compared to non-IIH control patients of corresponding age, sex and weight, which is one of the most important strengths. Moreover, diagnosis was made according to standardized diagnostic workup. In addition, eating disorders were diagnosed using validated standardized EDQs. No difference was found between the group of patients replying to the EDQ’s and the patients who did not, attributing to the study’s high internal validity and minimal selection bias. However, as only 45.7% of the patients responded to the questionnaires, we cannot exclude a reporting bias, which could partly explain the high rates of eating disorders in our patients. However, this bias was without impact on our primary and secondary outcomes.
Over the last years, a high incidence of psychiatric comorbidities among patients with IIH has been described by several groups (3,22) and linked psychiatric comorbidities to obesity or chronic headache rather than IIH as such (3). Moreover, IIH has also been linked to an increased incidence of eating disorder behaviors and eating disorders (22,23). However, the results of the studies investigating eating disorders were limited by small numbers of patients and none-obese control groups. When we compared IIH patients with non-IIH patients of comparable sex, age, and weight, we did not find increased morbidity of eating disorders.
However, we found a high prevalence of eating disorders in both ‘non-IIH patients’ and ‘IIH patients’, where more than 45% of patients were screened positive (SCOFF), and 18.2% (non-IIH)/16.9% (IIH) of the patients presented with a positive questionnaire for binge eating (BED-Q) respectively. These results are in line with a meta-analysis, which found an overall prevalence of binge eating of 22.2% and loss of control eating of 31.2% in children and adults with obesity (24). Other groups reported a prevalence of binge eating disorder and night eating syndrome of 10-15% in obese (25) and of 18.3% among female Korean student with excess weight (BMI ≥25 kg/m2) (26). A French group screened 47% of all participants positive for eating disorder behavior in a large (n = 330), predominantly female cohort with a mean BMI of 40 kg/m2 and a mean age of 51 years (27). However, the prevalence of eating disorders is much lower in the background population covering all classes of BMI. Another meta-analysis found an overall prevalence of 1.32% binge eating disorder and 3.0% subclinical binge eating disorder in children and adolescence background population (28). Moreover, a Danish study determined the life-time risk in females for developing an eating disorder by the age of 50 years to be 3.0% (29) in line with European data (30). Thus, eating disorders seem more prevalent in IIH and equally obese clinical mimicries compared to the background population, but not compared to the obese background population.
Moreover, we found a high prevalence of chronic headache and psychiatric disease in both non-IIH patients and IIH patients. An association between eating disorders and psychiatric disease and vice versa (31) has been reported, and chronic migraine, has been linked to psychiatric disease, obesity and eating disorders (32,33). In line, our data show a coincidence of chronic headache, obesity, psychiatric disease and eating disorders but the lack of a headache-free obese control group is a limitation to our study.
Binge eating was suggested to have a negative impact on weight loss (22) and to be associated with higher OP (>200 mm H2O) (23) in IIH patients and could thus apply to increased time to remission in IIH patients. We did not confirm this finding, nor did we find an effect of eating disorders on grade of papilledema, treatment modality (reflected by medical/surgical treatment), weight loss at ophthalmological remission, time to remission or sequelae (optic nerve atrophy, impaired visual fields) and binge eating was not more prevalent in either group, when comparing IIH and non-IIH patients. We wondered if the impact of an eating disorder was increased in the most obese population. Therefore, we stratified for non-obese and atypical IIH patients which did not change the results; only 12 patients were atypical, though. One explanation for the lack of a clinical impact could be the multidisciplinary treatment of the patients at two tertiary headache centers, which may not reflect the treatment results in other less specialized settings or in primary care. Moreover, we did not perform a long-term follow-up regarding the duration of weight loss or risk of relapse in IIH patients with an eating disorder compared to IIH patients without, which would be highly relevant in the clinical setting and might imply a differentiated weight management approach depending on the presence of an eating disorder or not. However, acute treatment of IIH may focus on simple weight loss to reduce intracranial pressure, followed by psychiatric treatment that focuses primarily on the eating disorder rather than body weight at a later stage.
Limitations
We used standardized EDQs, all validated in Danish, except for BED-Q. Moreover, the questionnaires are only indicative for an eating disorder. Use of diagnostic codes and detailed interviews would account for uncertainty and lack hereof is a limitation. Another limitation is the varying stages of disease and treatment periods represented. Patients responded to the questionnaires at varying time points after initial diagnosis or treatment. This hampers evaluation of causality. Additionally, our control group does not reflect the “ordinary” obese person, considering that non-IIH patients were recruited at a tertiary headache center, suffered from severe, chronic headache and presented with slightly elevated OP, which is well-accepted in patients suffering from chronic headache or simple obesity (34). In line, the cut-off point is still a matter of debate with a postulated ‘grey zone’ between 25–30 cm H2O (7). In addition, the differential diagnosis of IIH without papilledema is very rare compared to other primary headache disorders, and chronic migraine is highly prevalent and obesity is a risk factor for migraine in young women (35).
Conclusion
Eating disorders did not affect disease severity at time of diagnosis or outcome in IIH patients. Moreover, we found the same prevalence and distribution pattern of eating disorders in IIH and non-IIH patients advocating against a direct link between IIH and eating disorders.
Public health relevance
We showed that IIH patients had the same prevalence and pattern of eating disorders as their non-IIH controls with the same age, weight and sex, indicating a lack of association between eating disorders and IIH. Furthermore, eating disorders had no influence on the severity or outcome of IIH. Thus, acute treatment of IIH may solely focus on simple weight loss to reduce intracranial pressure, followed by psychiatric treatment that focuses primarily on the eating disorder rather than body weight at a later stage.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024241237237 - Supplemental material for The impact of eating disorders on idiopathic intracranial hypertension
Supplemental material, sj-pdf-1-cep-10.1177_03331024241237237 for The impact of eating disorders on idiopathic intracranial hypertension by Therese Wallentin, Jakob Linnet, Mia B Lichtenstein, Nadja S Hansen, Johanne J Korsbæk, Lisbeth Høgedal, Snorre M Hagen, Laleh D Molander, Rigmor H Jensen and Dagmar Beier in Cephalalgia
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: T.W.: No conflicts of interest. J.L.: No conflicts of interest. M.B.L.: No conflicts of interest. N.S.H.: Received funding from the Novo Nordic Foundation (NNF20OC0065926) during the conduction of the work and gave lectures for Pfizer, University of Southern Denmark, and University of Copenhagen. J.J.K.: Received funding from the Lundbeck Foundation, Odense University Hospital and Rigshospitalet during the study. L.H.: No conflicts of interest. S.M.H.: Received grants from Fight for Sight, Denmark and the Synoptik Foundation. L.D.M.: No conflicts of interest. R.H.J.: Gave lectures for Pfizer, Eli-Lilly, ATI, Merck, TEVA, Novartis, Lundbeck and Allergan. Investigator in clinical trials with ATI, Eli-Lilly, Novartis and Lundbeck. Director of Danish Headache Center, Lifting The Global Burden of Headache and Founder of Master of Headache Disorders at University of Copenhagen. Recived research funding from University of Copenhagen, Rigshospitalet, ATI, Lundbeck Foundation, The Medical Society in Copenhagen, NovoNordisk Foundation and Tryg Foundation. D.B.: Participated in clinical trials and served with lectures as well as on advisory or speaker boards for Novartis, Eli Lilly, Lundbeck, Pfizer, Abbvie and Teva outside the submitted work (migraine). Received grants from Odense University Hospital and Rigshospitalet, University of Copenhagen during the conduct of the study.
Funding
The authors disclose receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the Lundbeck Foundation (LBF R-276-2018-403-4), Candys Foundation (2015-146), Odense University Hospital and Rigshospitalet (25-A1320 and 69-A3346), Odense University Hospital (e-fond 177), Novo Nordic Foundation (NNF20OC0065926).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
