Abstract
Background
The recognition of venous sinus stenosis as a contributing factor in the majority of patients with idiopathic intracranial hypertension coupled with increasing cerebral venography and venous sinus stenting experience have dramatically improved our understanding of the pathophysiologic mechanisms driving this disease. There is now a dense, growing body of research in the neurointerventional literature detailing anatomical and physiological mechanisms of disease which has not been widely disseminated among clinicians.
Methods
A literature search was conducted, covering the most recent neurointerventional literature on idiopathic intracranial hypertension, the pathophysiology of idiopathic intracranial hypertension, and management strategies (including venous sinus stenting), and subsequently summarized to provide a comprehensive review of the most recently published studies on idiopathic intracranial hypertension pathophysiology and management.
Conclusion
Recent studies in the neurointerventional literature have greatly improved our understanding of the pathophysiologic mechanisms causing idiopathic intracranial hypertension and its associated conditions. The ability to make individualized, patient-specific treatment approaches has been made possible by advances in our understanding of how venous sinus stenosis and cerebral venous hypertension fundamentally contribute to idiopathic intracranial hypertension.
Introduction
Idiopathic intracranial hypertension (IIH) is characterized by severe headache, pulsatile tinnitus, and transient visual obscurations with elevated intracranial pressure (ICP) on lumbar puncture (LP) in the absence of an intracranial mass or venous sinus thrombosis. Papilledema is a key neuro-ophthalmologic finding associated with this condition and its presence or absence is a major metric of both disease severity and treatment success (1). While previously thought to be relatively rare, this disease appears to be increasing in prevalence both as the population becomes more overweight but also with increased recognition (2). While visual loss is often the primary concern in this population, the disease frequently results in severe impairments in quality of life (3). Medical therapies may be effective in mitigating the risk of visual loss but frequently result in undesirable side effects that add to the symptom burden. Weight loss has been shown to be beneficial in improving both quality of life and ICP in obese patients with IIH (4). Medical management and weight loss continue to be the predominant focus among neurologists and headache specialists and most ongoing studies published in specialty journals aim to study and improve these treatment modalities.
The recognition of venous sinus stenosis as a radiologic finding in the majority of patients with IIH has dramatically improved our understanding of the pathophysiologic mechanisms driving this disease process (5). While there remains debate regarding whether venous stenosis is a consequence or a cause of increased intracranial pressure, the clinical response to stenting of radiologic stenosis is well-documented (6). Cerebral angiography and venography with venous manometry are now commonly performed for patients with IIH and as a result, an abundance of valuable insights into the pathophysiology of the disease have been identified. In fact, there is now a dense, growing body of research in the neuro-interventional literature detailing anatomical and physiological mechanisms of disease which has not been disseminated widely amongst neurologists. The purpose of this manuscript is to summarize the most important recent published studies on IIH that are revolutionizing our understanding of the condition for a broader audience.
The importance of venous sinus stenting in understanding IIH pathophysiology
A large and growing literature base (7–9) has focused on venous sinus stenosis as a significant contributor to the development of elevated intracranial pressure in IIH. Venous sinus outflow obstruction has been identified in anywhere from 30%–90% of patients with IIH (10–13). The increased interest in venous sinus stenosis as a pathophysiologic driver, and venous sinus stenting as a treatment option, has led to cerebral angiography and venography becoming part of the standard evaluation for patients with severe symptoms. This evaluation is usually completed after undergoing non-invasive imaging to rule out other etiologies of increased ICP (14). In this procedure, arteriographic imaging is first obtained to evaluate for arteriovenous shunting and outflow anatomy. Then, via venous access, venous manometry measurements are obtained in the superior sagittal sinus (SSS), torcula, transverse sinus (TS), transverse-sigmoid junction, sigmoid sinus (SS), internal jugular vein (IJ), and in the right atrium (central venous pressure or CVP) to delineate the patient-specific drivers of systemic and intracranial venous hypertension. The detection of a pressure differential across the region of stenosis, or trans-stenosis “gradient,” is the key factor in determining patient candidacy for endovascular treatment (3,14). Several important observations have been published from venous pressure and anatomical data obtained from these procedures that aid in understanding the foundational pathophysiologic mechanisms at play in IIH.
Relationship between venous and cerebrospinal fluid pressures
Animal studies performed decades ago have demonstrated that CSF is reabsorbed via pressure-dependent, unidirectional flow from the subarachnoid space into the venous sinuses through the arachnoid villi, whereby CSF removal requires a 3–5 mmHg pressure differential (15). In consequence, as intracranial venous pressure rises, the pressure within the subarachnoid space (ICP) must rise until it is 3–5 mmHg higher than the venous pressure, at which point CSF will be able drain across the arachnoid granulations, and an equilibrium will be reached. As the majority of arachnoid granulations lie along the convexities near the SSS (16), more rostral pressures (specifically within the SSS) likely have the largest influence on CSF reabsorption and therefore CSF pressures.
Human studies have now been performed confirming a similar relationship. A recent study compared venous sinus pressures to simultaneous LP opening pressures (OP) and demonstrated a strong, nearly 1-to-1 correlation between torcular pressures (in mmHg) and OP (in cm of water) in patients with diagnosed or highly-suspected IIH (17). This relationship was preserved across the ICP-spectrum and included patients with OP ranging from teens to greater than 55 cm water. This strongly supports that elevations in CSF pressure occur in concert with proportional elevations in venous sinus pressures. Patient outliers were identified that strayed from this relationship, which may be due to the inherent patient variability in terms of venous outflow anatomy, arachnoid granulation density, as well as variability in lymphatic drainage. Overall, these studies provide evidence that, in conditions where intracranial venous pressures are elevated, intracranial CSF pressures are proportionately elevated.
Intracranial venous pressures in IIH patients
While the relationship between intracranial venous pressures and IIH has been recognized for decades, only recently have anatomical and physiologic studies been performed on systemic and intracranial venous pressures in IIH patients (18). A recent study reported on measured venous pressures from the SSS through the chest in over 100 patients with IIH, without using non-invasive imaging for pre-procedure screening (therefore representing “all comers,” not just patients suspected of having TS stenosis) (19). Three critical principles were described relating to venous physiology in these patients. First, rostral venous pressures are almost always higher than more caudal venous pressures (for instance, the SSS being higher than the TS, and the TS being higher than the jugular vein, etc.). Second, CVP forms the foundation of intracranial venous pressures and, in the absence of venous stenosis and outflow impairment, CVP is about 4–5 mmHg lower than pressures measured in the SSS. Given this, it is likely that elevations in CVP, from obesity or cardiopulmonary etiology, directly lead to increases in intracranial venous pressures. Third, venous stenosis-related pressure gradients are very common and result in even higher upstream venous pressures. In the study sample, over 80% of patients had a gradient of at least 4 mmHg across a venous stenosis, with 55% having a gradient of at least 8 mmHg. While stenosis is most common at the TS, stenosis with pathological gradients were also identified in the SSS and IJ, with some patients having multiple sites of venous outflow impairment.
A study reporting on venous waveforms in these patients further corroborates the pronounced effect of sinus stenosis on venous outflow and physiology (12), providing evidence that the cerebral venous waveform is influenced by both the ICP and CVP waveforms. In the absence of stenosis, the intracranial venous waveform has a typical ICP-type waveform that becomes progressively more CVP-like (with predominant respiratory variability) as the catheter is withdrawn caudally. In the presence of a pathological stenosis, the upstream venous waveform is an ICP-type waveform with high amplitude and no respiratory variability. Once crossing the level of the stenosis, the waveform abruptly transitions to a CVP-type with low amplitude and pronounced respiratory variability (12). After treatment of the stenosis and alleviation of the gradient, the waveform morphology changes to match that of those patients without stenosis.
Previous studies had demonstrated a deleterious effect of general anesthesia on measured gradient size compared to awake values (20,21), leading experts to recommend diagnostic venography be performed awake or under conscious sedation (14). However recent data suggests the effect of general anesthesia on measured pressures can be minimized with tight blood pressure and end-tidal carbon dioxide control (22).
Intracranial venous pressures in the absence of disease
What is a normal intracranial venous pressure? While a current prospective study is underway to establish normal physiologic pressures in patients without disease, to date there are no studies specifically reporting on normal intracranial venous physiology in the absence of neurological disease. Historically, a few studies calculated SSS pressures from lumbar catheter infusion protocols, suggesting that SSS pressures likely exist below 11 mmHg in the absence of disease (23,24). Other studies of patients undergoing craniotomy or ventriculography for brain tumor diagnosis with direct venous catheterization showed that, in general, the mean awake SSS pressures are 15 mmHg or less (25,26).
To date, the most robust data on ‘normal’ intracranial venous pressures is derived from studies on IIH patients that included intracranial pressures on the lower end of the ICP spectrum. Two recent studies, which included a small number of diagnosed (or highly suspected) IIH patients with OP on spinal tap of 20 cm of water or less that underwent venous manometry (a total of 20 patients combined), suggested that intracranial SSS pressures in patients with ICP of 20 or less should exist at or below 16–18 mmHg (17,19). This range makes intuitive sense given the previously defined relationship between venous pressures and CSF OP, as venous pressures exceeding this range would be expected to result in ICP over 20.
Obesity causes increases in CVP
There is significant evidence suggesting a strong association between IIH and obesity, with hormonal factors, chronic inflammation and cortisol levels all likely contributing but, most importantly, the relationship between obesity and an elevated CVP (19,27–31). As intra-abdominal pressure increases with obesity, the cardiac and pleural filling pressures also become elevated. This results in decreased venous outflow, increased intracranial venous pressure due to venous congestion, and a subsequent increase in pressure within the subarachnoid space (ICP) to facilitate CSF absorption (32). This correlation of CVP and BMI has been demonstrated in a study looking specifically at patients with IIH, which found that the CVP could be predicted by using the BMI with the following formula- CVP = 5.26 + 0.18 * BMI (19). Raper et al. (33) also demonstrated that an increasing BMI was significantly correlated with elevated intracranial venous pressures, as well as a higher-pressure gradient across the stenosis prior to treatment. Further, the hypothesized relationship between obesity and CVP, and the association of CVP with IIH, may also be supported by the efficacy of bariatric surgery for improving the symptoms seen in IIH (10,34,35). One systematic review demonstrated that with bariatric surgery 90% of patients had significant improvement or complete remission of headaches, 29% for visual symptoms, 96% for pulsatile tinnitus, and 88% demonstrated resolution or stabilization of papilledema (34). A randomized trial of bariatric surgery versus weight loss in 66 IIH patients demonstrated clear superiority of bariatric surgery in ICP reduction and improvement in quality of life (4).
An additional consideration is the contribution of obesity-associated comorbidities, such as obstructive sleep apnea (OSA), seen in 50–60% of obese patients, to the disease (36,37). Significant nocturnal intracranial pressure spikes have been demonstrated in patients with OSA. As we will discuss, the development of venous stenosis is most commonly thought to be related to extramural compression from high ICP, which initiates a positive feedback loop manifesting as high ICP, venous stenosis, and venous congestion. Recurrent, nightly elevations in ICP due to obesity-related OSA may contribute to this process and intracranial venous congestion that continue to further elevate ICPs (38).
Venous sinus stenosis
The recognition of intracranial venous hypertension as a key factor in IIH and the common practice of stenting in those with sinus stenosis and a trans-stenosis pressure gradient have dramatically improved our understanding of the pathophysiology of IIH. There is now increasing evidence that extramural (extrinsic) venous stenosis occurs in IIH secondary to a positive feedback loop mechanism that is initiated by elevated ICP (Figure 1). The source of compression of the TS is thought to be from the intracranial contents themselves (brain and CSF); higher ICP exerts a more pronounced compressive effect on the susceptible, or exposed, portion of the sinus inducing stenosis. The presence of extramural compression from increased ICP causes progressive venous outflow obstruction, causing higher upstream venous pressures and therefore higher CSF pressure reabsorption at the arachnoid granulations. This positive feedback loop progresses until severe TS stenosis, high upstream venous pressures with a consequent pathological gradient, and high ICP are present (39,40). It is likely the process continues until the intramural and extramural forces equilibrate (intramural pressure from venous congestion opposing the extramural compression from elevated ICP) (red arrow in Figure 1), stabilizing at a higher ICP and venous pressure. Interestingly, the degree of TS stenosis necessary to cause a significant trans-stenosis gradient is quite low, with one study showing that development of a significant pressure gradient can be found with angiographic stenosis of only 30–35% (12).
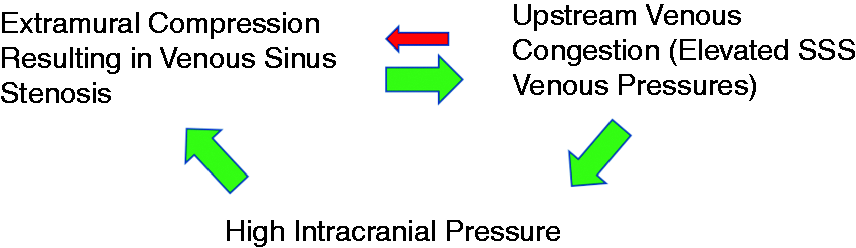
Positive feedback loop hypothesis for the development of extramural venous sinus stenosis. Increases in intracranial pressure cause compression of the sinus at a susceptible site resulting in worsened stenosis and resultant upstream venous congestion, thereby leading to higher intracranial pressures (green arrows). High venous pressures ‘push back’ against intracranial pressures (red arrow).
The feedback loop hypothesis is supported by several studies evaluating sinus calibers before and after lowering of ICP with CSF removal (41–43). One of the most interesting case reports used intravascular ultrasound and venous manometry to evaluate severe venous sinus stenosis in a patient with a large gradient. After measuring venous caliber and the gradient, a high-volume LP was performed and then the study was immediately repeated. Following LP, the gradient and venous stenosis completely resolved and intracranial venous pressures dropped dramatically (42).
Why does this process most commonly selectively occur at the lateral TS in patients with IIH, but not in individuals without this disease? While there has yet to be anatomical evidence supporting the reasons for this phenomenon, some important insights have been provided. Venous sinus stenosis appears to be a diffuse process through which the effect of ICP occurs across multiple segments of the venous sinuses, supported by the identification of patients with multiple sites of venous stenosis and those who develop de novo or stent-adjacent stenosis after venous sinus stenting (44,45). In fact, one study demonstrated that the caliber of the TS and SSS are significantly smaller in those with IIH relative to those without (46). One hypothesis states that varying regions of the sinuses may be more susceptible than others to extramural compression in the setting of elevated ICP, wherein the torcula and sigmoid sinus are most resistant to compression and the lateral TS is most susceptible (44). Once significant stenosis manifests at the TS, venous congestion upstream “pushes back” against ICP and therefore upstream stenosis is usually not identified. So, while other areas of the sinuses may be exposed to potential compression from elevated ICP, this hypothesis does explain why in most patients the lateral TS is the only identified site of significant stenosis, and why patients may develop de novo stenosis after the TS is rigidly reinforced with a stent.
Venous sinus stenting
Venous sinus stenting is on the rise and has gained acceptance as a primary means of treating severe IIH in patients with demonstrated venous sinus stenosis with resultant pathologic pressure gradient (Figure 2) (47). This trend is based on both the efficacy and safety of stenting, as well as the traditionally high rate of failure for CSF shunting in this population, which has not improved even with the advances in valve technologies and the use of neuro-navigation. The rate of shunt revision in the literature, in the setting of shunt malfunction or infection, ranges from 38% to 85.7% (48,49). In fact, meta-analyses reporting on a series of IIH patients undergoing venous sinus stenting suggest high rates of improvement in headache, tinnitus, and vision (Table 1). Nearly 80% of patients had improved headache, 90% had improved tinnitus, and nearly 94% had improved papilledema, with treatment failure (need for repeat procedure or shunt) only occurring in 12% (50). Other studies have reported similar rates (48,51),

Example patient with severe left transverse sinus stenosis and 21 mmHg gradient across the site of stenosis (left panel; arrows denote venous pressures at each location in mmHg). Following transverse sinus stenting, the stenosis has resolved, the gradient has been reduced to 1 mmHg, and upstream venous pressures have reduced from 31 mmHg to 14 mmHg.
Meta-analyses reporting on outcomes after venous sinus stenting.
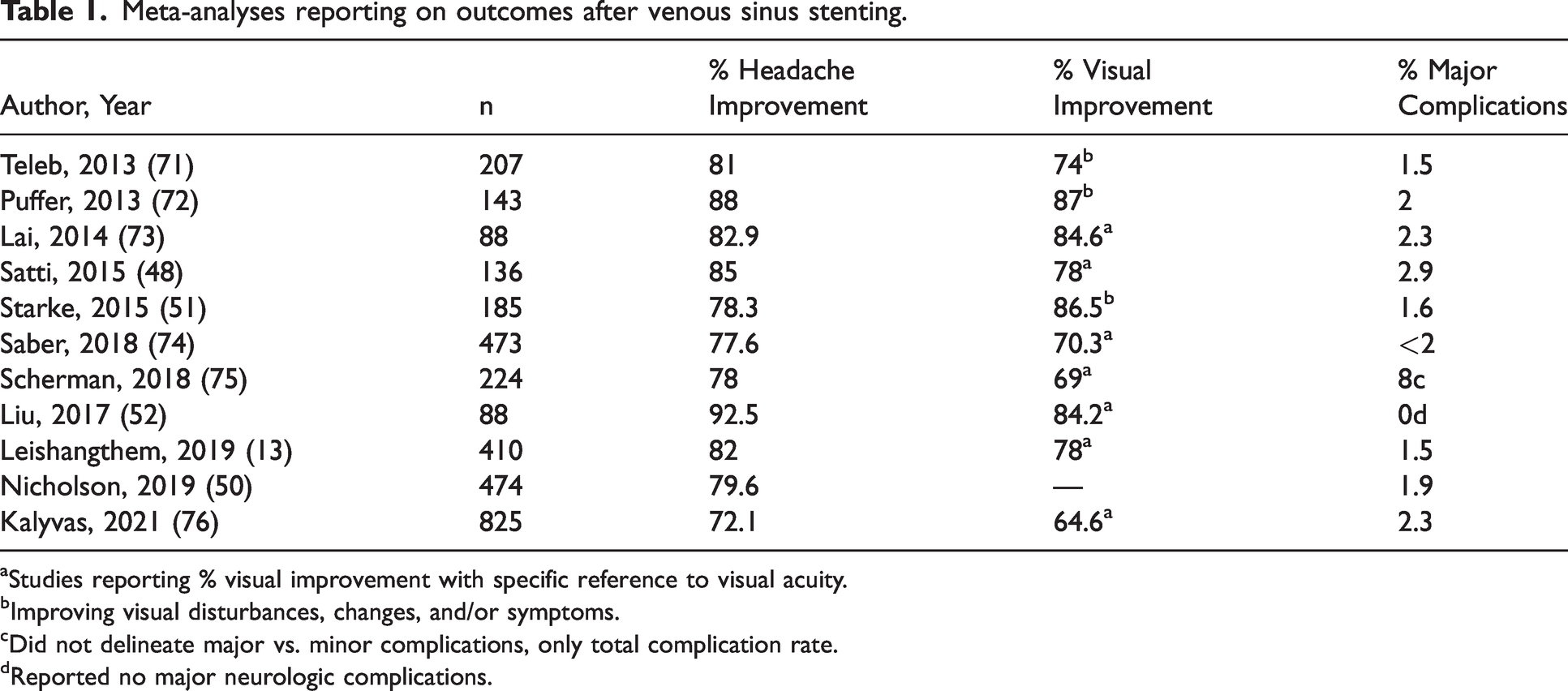
aStudies reporting % visual improvement with specific reference to visual acuity.
bImproving visual disturbances, changes, and/or symptoms.
cDid not delineate major vs. minor complications, only total complication rate.
dReported no major neurologic complications.
Most studies on VSS report using a gradient >8 mmHg for selecting candidates for stenting, with evidence supporting safety and efficacy (14,44,50,51). Prospective studies in which patients underwent stenting with an intraparenchymal pressure monitor in place demonstrated immediate reductions in ICP with stenting (32,52). Further, studies have also reported long term reductions on ICP as well. Patsalides et al. (53) measured CSF OP three months before and after venous sinus stenting. Their study demonstrated a statistically significant decrease (45% mean reduction) in the OP in patients with IIH three months post-stent placement, and found the effect to be independent of acetazolamide use and weight loss.
Symptom recurrence after stenting
Most meta-analyses report need for retreatment after stenting to be less than 20% (Table 1). Studies looking into the reason for stent failures have suggested patients with higher BMI (and therefore higher CVP) are at higher risk of symptom recurrence, as well as female patients with extramural compression and higher OP (33,54,55). Further, larger diameter stents have been implicated as a risk factor for developing stent-adjacent stenosis through mechanical stretching of the sinus (56). Remote de novo or stent-adjacent stenosis, often times occurring in the SSS, are now well-described and thought to be related to the development of a new positive feedback loop at susceptible regions of the sinus from recurrent ICP elevations (44,54,57).
Interestingly, the relationship between ICP reduction and symptoms is not always consistent. Garner et al. (3) demonstrated that more than half of the 81 patients in their study who underwent stenting developed symptom recurrence (usually headache) at a median of seven months. Interestingly the vast majority of patients with symptom recurrence had sizable reductions in OP compared to pre-stenting (3). The authors named this curious observation the “re-equilibration phenomenon,” in which patients often had headache recurrence even in light of lowered ICP. A similar phenomenon has been identified with medically managed patients in which chronic headaches are common even after papilledema resolution, suggesting that this phenomenon may be a characteristic of the disease process itself and not necessarily a complication related uniquely to stenting (58).
Jugular vein stenosis
Intracranial venous hypertension can also be secondary to extracranial venous stenosis, specifically the internal jugular vein (IJV) (59). This can be in the setting of a congenitally diminutive IJV, or secondary to extrinsic compression, such as by the styloid process, a cervical rib, hypertrophic scale muscles, or the mastoid process (60,61). At present, there is limited evidence on the surgical treatment of intracranial venous congestion secondary to internal jugular vein stenosis, with no randomized trial data as of yet. Initial experience in treating IJV stenosis for IIH was described, with a follow up case series in 2019, from the same center (62,63). In a 2021 review, only 17 studies were identified that involved surgical intervention, with only 100 patients total having undergone a procedure (60). From that review, they found that the most common intervention used was styloidectomy (64.7%), followed by IJV stenting (52.9%), with others including angioplasty, myomectomy, and resection of a compressive mass or osteophyte (60). As noted in the review and demonstrated by the limited data available, the role of surgical treatment of IJV stenosis is still being defined. Additional experience will be necessary to assess the efficacy and durability of venous interventions targeting the IJV, which may serve as an additional potential avenue in addressing intracranial venous hypertension.
CSF leaks and pulsatile tinnitus
There is a subset of patients with IIH that often present to otolaryngologists rather than neurologists because headache is less prominent. These patients often have loud pulsatile tinnitus or skull base CSF leaks with rhinorrhea or otorrhea and frequently have other milder symptoms of IIH. We now understand that pulsatile tinnitus is frequently a result of transverse sinus, sigmoid sinus, or internal jugular venous anomalies or stenosis (64). Interestingly, manual compression of the ipsilateral jugular vein often results in temporary tinnitus resolution, which can be useful in confirming this relationship (65). There is increasing evidence suggesting that patients who develop spontaneous skull base CSF leaks often times have underlying IIH (66,67), leading some authors to question whether most patients with spontaneous CSF rhinorrhea or otorrhea have contributing underlying intracranial hypertension (67,68). Many of these patients have OP that are only mildly elevated, especially if they are actively leaking at time of LP. Many of these patients should be considered as having an IIH-spectrum condition and can be treated similarly to traditional IIH patients.
Classifying patients based on individualized drivers of intracranial venous hypertension
Intracranial venous hypertension, leading to a proportional elevation in the intracranial CSF pressures, is a defining pathologic characteristic of IIH. Given that we now understand that intracranial venous hypertension underlies the symptoms, authors have suggested changing the nomenclature to a more self-descriptive, cause-based term, with “chronic intracranial venous hypertension syndrome” (CIVHS) as one such proposed name (10). This nomenclature classifies patients into four different groups based upon individual drivers of their intracranial venous hypertension for treatment purposes.
For instance, an elevated CVP in the setting of morbid obesity can lead to intracranial venous hypertension. Individuals with pulmonary hypertension or congestive heart failure with an otherwise normal weight also have increased cerebral venous pressures. Further, a subset of patients have significant venous sinus stenosis as their underlying etiology, while others have both an elevated CVP and venous stenosis contributing. Fargen et al. delineated four types of CIVHS to reflect these etiologies. These four types include craniocervical (elevated ICP secondary to venous sinus stenosis, without an elevated CVP), central (elevated ICP in the setting of isolated elevation in CVP without sinus stenosis), mixed (elevated ICP secondary to both an elevated CVP and trans-stenosis pressure gradient), and post-venous thrombosis (10).
Importantly, this classification scheme also identifies patients as IIH-spectrum with clear symptoms of elevated intracranial pressure that have OP below the 25 cmH20 threshold for IIH diagnosis (7). These patients, with an OP within the range of 14–24 cmH2O, present with symptoms consistent with IIH and have symptom relief through pressure-lowering means, such as an LP (10). This phenotype is anecdotally often seen in patients with connective tissue disorders, most commonly Ehlers-Danlos syndrome, which often manifests with profound impairment in quality of life due to hypersensitivity to elevated intracranial pressure, as well as associated TS and IJ stenosis, postural orthostatic tachycardia syndrome, and CSF leaks (69).
Rethinking the way we manage and treat IIH patients
The defining hallmark of those with IIH is intracranial venous hypertension, but the etiology varies. Given that patients have unique drivers of elevated intracranial venous pressures, individualized, etiology-targeted interventions and treatment strategies are important. Ideally, the treatment paradigm for each patient would target patient-specific drivers of the venous hypertension and therefore classification of patients into the subtypes of CIVHS makes this distinction of treatment pathways most feasible. Those patients with demonstrated venous outflow impairment, without an elevation of CVP proximally, would see benefit from stenting. However, they would not benefit from treatment options targeting more proximal causes of venous congestion, as these are not the culprits driving the venous hypertension. In contrast, in the absence of sinus stenosis but with a proximal cause of elevated CVP, stenting has no role given the absence of a focal, treatable venous stenosis. However, this subset of patients would potentially benefit from options targeting their central cause of elevated CVP, such as weight loss or diuretics. Those patients of a mixed-etiology subtype are those that serve as the greatest challenge. Treating sinus stenosis in the mixed-etiology subtype does not address the central cause of systemically high venous pressures, which ultimately contributes to elevated intracranial venous pressures as well and may result in refractory elevations in intracranial venous pressures even after successful stenting. Separating patients into classes based on their pathophysiologic cause of venous hypertension does not mean that everyone will perfectly fit into category-specific treatment algorithms, but it does provide a framework to guide practitioners instead of a traditional “shotgun”-style approach.
Furthermore, our new understanding of the function of intracranial venous hypertension in IIH necessitates a reexamination of the pathophysiologic roles of historical, long-standing associations with the condition. Specifically, how things such as vitamin A, retinoids, iron deficiency anemia, antibiotics (tetracycline, doxycycline, sulphonamides), hormonal factors, corticosteroids, or anecdotally reported triggers such as traumatic head injuries or spine surgery alter either venous pressures or the reabsorption of CSF based upon our new understanding of this disease (70).
Figure 3 provides a general summary strategy for the interventional management of IIH patients from diagnosis through post-stent care.

Interventional management algorithm for patients with severe IIH symptoms refractory to medical management.
Future
The ability to make individualized, patient-specific treatment approaches has been made possible by advances in our understanding of how venous sinus stenosis and cerebral venous hypertension fundamentally contribute to IIH. It also serves as a roadmap for future research, including the study of normal intracranial venous physiology in the absence of disease and the inherent predilections for the development of stenosis in certain patients. Refocusing therapy of patients with IIH on the disease's fundamental mechanism (intracranial venous hypertension) is critical to enhancing the care we provide them.
Article highlights
Medical management and weight loss continue to be the predominant focus among neurologists and headache specialists and most ongoing studies published in specialty journals aim to study and improve upon these treatment modalities. The recognition of venous sinus stenosis as a contributing factor in a large percentage of patients with IIH has dramatically improved our understanding of the pathophysiologic mechanisms driving this disease process. Cerebral angiography and venography with venous manometry are now commonly performed for patients with IIH and as a result, an abundance of valuable insights into the pathophysiology of the disease have been identified. The purpose of this manuscript is to summarize the most important recent published studies on IIH that are revolutionizing our understanding of the condition for a broader audience.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
