Abstract
At present, it is contradictory to determine if the combination of certain prothrombotic polymorphisms and migraine increases the risk to develop ischaemic cerebrovascular disease. Recently, the common Val34Leu polymorphism of the A-chain factor XIII gene, associated with variations in factor XIII activity, has been suggested to play a significant role in the development of arterial and venous thrombotic disorders. We analysed the prevalence of this polymorphism in 17 patients with coexisting ischaemic cerebrovascular disease and migraine (5 with aura, and 12 without aura), 89 patients with migraine (43 with aura, and 46 without aura), 116 patients with ischaemic cerebrovascular disease, and 467 healthy Caucasian controls from the South of Spain. Genomic PCR amplification, using a mutated oligonucleotide, and allele-specific restriction assays were used for genotyping. The factor XIII Leu 34 variant was present in 47.1; 40.5; 34.9; and 35.1% of patients with coexisting ischaemic cerebrovascular disease and migraine, ischaemic cerebrovascular disease, migraine, and control subjects, respectively. These data suggest that the factor XIII Leu 34 allele does not play a protective role against these disorders in our population.
Introduction
Factor XIII (FXIII) has a pivotal role in the coagulation process. Activated FXIII catalysed the formation of covalent γ-glutamyl-ε-lysine bonds between fibrin monomers, and is involved in the formation of these bonds between other proteins, such as fibronectin and collagen. All these effects increase resistance of fibrin to degradation by plasmin, and improve clot adherence to the vessel wall (1). The relevance of FXIII is demonstrated by the severe bleeding tendency of patients with congenital FXIII deficiency (2). Recently, it has been identified one common polymorphism (G/T) in exon 2 of the FXIII A-subunit gene causing a Val34Leu amino acid change (3). The Leu 34 variant associates with increased FXIII specific activity (3). Interestingly, the Leu 34 allele has been suggested to play a significant role in the development of arterial and venous thrombotic disorders (4–7).
Stroke is a multifactorial disorder in which the influence of genetic and environmental factors could affect the risk of a thromboembolic disease (8). Among the recognized risk factors for stroke, migraine has been considered an independent risk factor for ischaemic cerebrovascular disease (CVD) (9, 10). At present, it is contradictory to determine if the combination of certain prothrombotic polymorphisms and migraine increase the risk to develop CVD (11, 12).
The objective of this study was to evaluate the role of the recently characterized FXIII V34L polymorphism in migraine, and specially the relevance of this polymorphism in the risk to develop CVD in those patients with personal antecedents of migraine.
Methods
Patients
The present study was approved by the local Ethics Committee, and all participants gave their informed consent to be studied.
This study involved 17 patients with coexisting CVD and personal antecedents of migraine (5 with aura, and 12 without aura) (Table 1). We also enrolled 116 consecutive patients with CVD referred to our institution (Table 1). Diagnosis was attained according to previous reports (13). Classification of ischaemic strokes among atherothrombotic, cardiac emboli, small artery disease, others, and undetermined was performed following criteria indicated elsewhere (14). Additionally, we studied 89 patients with migraine (43 with aura, and 46 without aura) (Table 1). Diagnosis of migraine was achieved according to the International Headache Society (15).
Characteristics of subjects included in this study

CVD, Ischaemic cerebrovascular disease. M, Migraine.
Furthermore, to determine the frequency of the studied polymorphism in our area, genetic analyses were also performed in 467 nonrelated and unselected subjects without documented history of migraine, venous and/or arterial thrombosis (Table 1).
Genotype analysis
Genomic polymerase chain reaction (PCR) of the FXIII exon 2 gene was performed
using the following primers: 5′ gaccttgtaaagtcaaaaatgtc 3′
and the mutagenic 5′ t
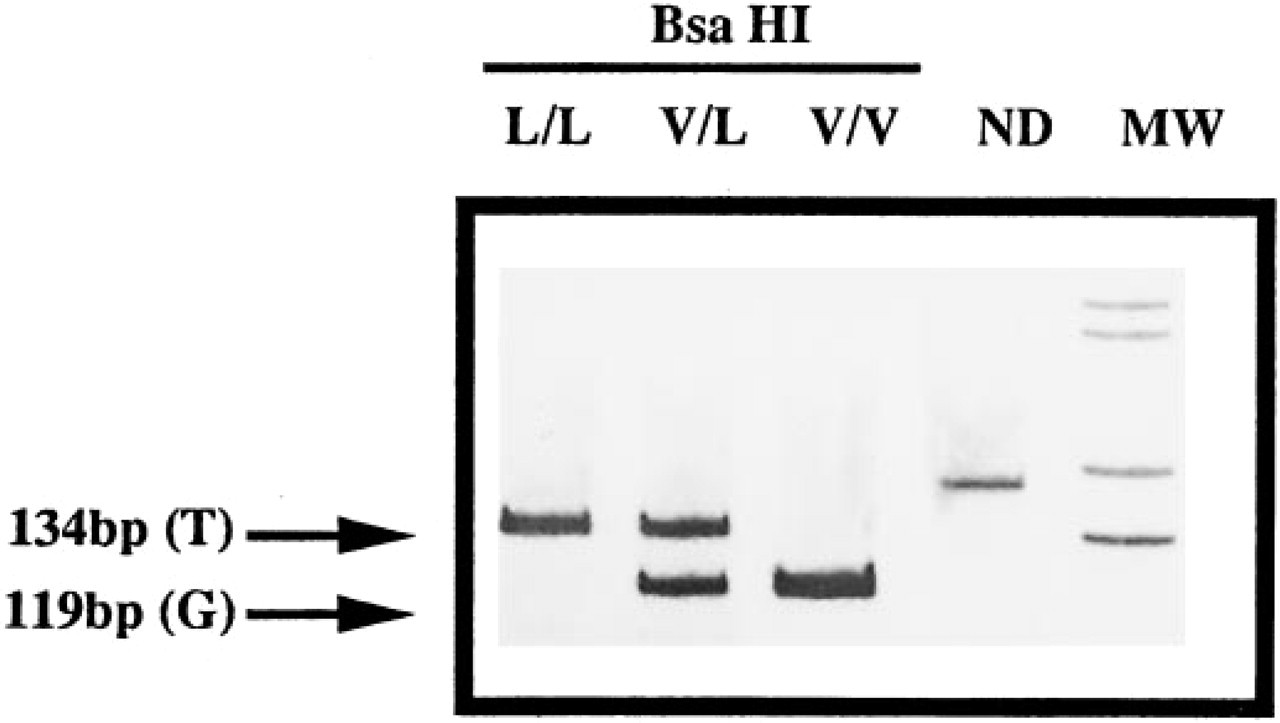
Val34Leu polymorphism of the A-chain FXIII gene. L, Leucine 34; V, Valine 34; MW, Molecular Weight marker (1 Kb ladder Gibco-BRL), bp, base pairs. ND, not digested.
Statistical Analysis
The χ2 test was used to compare frequency distributions. The strength of the association between the polymorphism and migraine with the occurrence of CVD was estimated by calculation of the odds ratio (OR) with the EpiInfo software and the Cornfield method for the calculation of 95% confidence intervals (CI).
Results
We have developed a rapid, simple, and reproducible method to identify the 214 G/T (Val34Leu) polymorphism of the factor XIII gene. The use of a mutagenic primer in the PCR amplification allows the identification of the genotype by a simple restriction assay with Bsa HI. Thus, we avoid the use of more complex and less reproducible techniques of genetic identification (SSCP, allele-specific PCR, of solid-phase minisequencing).
Table 2 summarizes genotype and allele distribution in all established groups of patients and controls. Statistical comparisons were established with controls. The frequency of the Leu 34 allele did not significantly differ between any group of patients and controls.
Genotype and allele frequencies of the FXIII V34L polymorphism

CVD, Ischaemic cerebrovascular disease; AT, Atherothrombotic; CE, Cardiac Emboli; SAD, Small Artery Diseases; UD, Undetermined; M, Migraine; W, With; Wt, Without; OR, Odds ratio; CI, Confidence Interval.
Discussion
It is now well demonstrated that the FXIII V34L polymorphism associates with FXIII specific transglutaminase activity, due to the thrombin-activation rate of FXIII (3, 18–22). Paradoxically, the less frequent Leu 34 allele has been identified as a protective genetic factor against coronary artery disease (4, 5) and venous thrombosis (6, 7). Also, a preliminary study indicated a slightly higher incidence of the FXIII Leu 34 variant in 62 patients with primary intracerebral haemorrhage compared with controls (23). However, contradictory results have been obtained by our group including more patients with primary intracraneal haemorrhage (24), coronary heart disease, and venous thrombosis (25) that do not support the previous conclusion.
Our results in CVD patients suggest that FXIII Leu 34 allele does not play a protective role in CVD patients from our area, independently of the aetiologic varieties. However, stroke is a multifactorial disorder, genetic and environmental factors act additively to determine the risk of an individual of developing thrombosis (8). Recent studies have suggested that migraine (an independent risk factor for CVD) could be synergistic with other risk factors for thrombosis (9, 10). We speculated that FXIII Leu 34 when combined with migraine could increase the likelihood of CVD. Firstly, we found that this polymorphism has no a significant role in the development of migraine (with or without aura). Finally, our data suggest that the Leu 34 allele does not protect against CVD in patients with coexisting migraine.
Considering the present study, several points should be borne in mind when interpreting results. First, the study was performed in survivors. Therefore, a survival bias cannot be avoided in the disease-association study, and likely early mortality from CVD in patients could lead to an underestimation of the prothrombotic polymorphism. Second, our study has been performed in Caucasian subjects from the Mediterranean area. Since the prevalence of this polymorphism could vary geographically, and it is linked to other FXIII polymorphisms also associated with the activity of FXIII, the FXIII Val34Leu polymorphism may have a different predictive value for thromboembolic disease in diverse populations. Therefore, these data could not exclude the possibility of association between the Val34Leu FXIII and thrombotic risk in other populations, and consequently, the relevance of FXIII polymorphism should be further investigated in other populations, and with prospective and family studies. Third, the size of our sample is small, and thus, new studies including more patients should be performed.
Footnotes
Acknowledgements
The study has been supported by FIS 00/0328. JC is Contratado de Investigación FIS-Servicio Murciano de Salud. R G-C is Postoctoral fellow of the University of Murcia.
