Abstract
Abstract
Background
While growing evidence suggests the efficacy of various behavioral approaches to the preventive treatment of migraine, it remains largely unclear which behavioral interventions are indicated for which type of patient. This exploratory study aimed to identify moderators for the outcome between migraine-specific cognitive-behavioral therapy and relaxation training.
Methods
In this secondary analysis of an open-label randomized controlled trial, the data of N = 77 adults (completer sample) with migraine (mean age = 47.4, SD = 12.2, 88% female), who were allocated to either migraine-specific cognitive-behavioral therapy or relaxation training, was examined. Outcome was the frequency of headache days at the 12-month follow-up. We analyzed baseline demographic or clinical characteristics and headache-specific variables (disability, emotional distress, trigger sensitivity and avoidance, pain acceptance, self-efficacy) as candidate moderators.
Results
Higher headache-related disability (assessed via the Headache Impact Test, HIT-6, B = –0.41 [95% CI: –0.85 to –0.10], p = .047), higher anxiety (assessed via the subscale Anxiety of the Depression, Anxiety and Stress scales, DASS-A, B = –0.66 [95% CI: –1.27 to –0.02], p = .056), and the presence of a comorbid mental disorder (B = –4.98, [95% CI: –9.42 to –0.29], p = .053), moderated the outcome in favor of migraine-specific cognitive-behavioral therapy.
Conclusion
Our findings contribute to an individualized treatment selection and suggest that preference for complex behavioral treatment (migraine-specific cognitive-behavioral therapy) should be given to patients with high headache-related disability, increased anxiety, or a comorbid mental disorder.
Keywords
Introduction
Psychological or behavioral factors are assumed to affect the course of migraine (1,2). For example, the experience of stress is considered to be a major trigger of migraine attacks (3,4). Comorbid mental disorders such as depression or anxiety disorders are associated with higher headache activity and disability (5). Thus, behavioral treatments have a central role among the non-pharmacological prophylactic interventions (6). The term “behavioral treatment” encompasses different categories of interventions. Common treatment categories in behavioral migraine prophylaxis are (a) patient education and lifestyle counseling, (b) relaxation training, (c) biofeedback, and (d) cognitive-behavioral therapy (7). Other categories are (e) mindfulness-based (8) and (f) exposure-based approaches (trigger management) (9). Given the different interventional categories, two fundamental questions remain unanswered (10,11). The first question is whether and if so, which behavioral interventions are overall superior compared to others in the preventive treatment of migraine. The second question is which behavioral interventions are indicated for which type of patient. The current German medical guideline on the treatment of migraine attacks and migraine prophylaxis recommends supplementing pharmacological therapy with non-pharmacological, behavioral therapy (such as relaxation or cognitive-behavioral therapy as a standard approach). In the case of remarkable migraine-related disability and/or comorbid mental disorders, pain-specific behavioral therapy should be applied as an escalation of the standard behavioral approach (12). Beyond these recommendations, the empirical evidence for a differentiated indication or individual selection of behavioral treatments (“precision therapy”) in migraine should be advanced, particularly since clinical characteristics and treatment response in migraine patients are heterogeneous (11,13). Also, it is not yet clear whether a combination of several interventions is superior to single behavioral approaches.
In our randomized controlled trial (RCT), the efficacy of a multimodal, migraine-specific cognitive-behavioral therapy (miCBT) was compared with relaxation training (RLX) only (14). Both treatments led to a statistically similar reduction of headache activity, disability, and emotional distress, and neither treatment was superior to the other overall (14). This secondary analysis aimed to investigate which type of patient benefits more from miCBT than RLX and to explore demographic, clinical, and headache-related moderators of treatment outcome. Considering the existing guideline, disability and comorbidity are included as potential moderators.
Methods
Study design and participants
This study is a secondary analysis of an open-label RCT (14). The first stage of this trial comprised three conditions (miCBT, RLX, and a waiting-list control group, WLC). Eligible participants were randomly assigned with a 1:1:1 ratio to miCBT, RLX, or the WLC. The second stage (four- and 12-month follow-up) comprised two conditions (miCBT, RLX) since the participants, who successfully passed the WLC by providing a post measurement, were randomly assigned to the miCBT- or RLX-condition (1:1 ratio, Figure 1). The treatment was carried out at the psychotherapy outpatient clinic of the Department of Psychology (University of Mainz, Germany). The protocol was approved by the competent Ethics Committee of the State Chamber of Medicine in Rhineland-Palatinate, Germany, reference number 837.291.16 (10610). All participants gave their written informed consent.

Study design and assessments. In total, four assessments were carried out (baseline, post-treatment, 4-month follow-up, and 12-month follow-up).
Participants were recruited via several newspaper articles, social media, and one TV clip as well as through the distribution of flyers in local medical practices (general practitioners, neurologists, pain therapists). Eligible participants were adults, meeting the International Classification of Headache Disorders (3rd edition, beta version, ICHD-3 beta) criteria (15) of either migraine without aura, migraine with aura, or chronic migraine for at least one year, with a minimum of four headache days per month, and a pattern of migraine symptoms stable over last six months. Exclusion criteria included a diagnosis of medication-overuse headache, a previously completed or current psychotherapy, a severe mental disorder or medical comorbidity, suicidal tendency, and pregnancy or lactating. Participants were excluded if they were currently taking a headache prophylactic medication or having therapy with botulinum toxin or neuromodulation. The face-to-face screening included a structured interview to validate the migraine diagnosis according to the ICHD-3 beta (15) as well as to assess comorbid mental disorders according to the Diagnostic and Statistical Manual of Mental Disorders (5th edition, DSM-5) (16).
Interventions
Both treatments (miCBT, RLX) comprised seven group sessions, each lasting 90 min. The sessions were carried out weekly. Both interventions are described in detail elsewhere (14), and the miCBT is published as a treatment manual (17). The miCBT comprised different behavioral approaches (e.g., patient education, lifestyle counseling, cognitive-behavioral techniques to improve coping with stress and fear, and trigger management, Figure 2). At the end of each session, the participants were taught a brief relaxation method. The RLX centered around progressive muscle relaxation (PMR) as an easy-to-learn and easy-to-apply relaxation technique with Grade A evidence in migraine prophylaxis (Figure 2) (18,19).

Description of treatment programs (miCBT, RLX).
Assessments
After completion of the baseline measure, the treatment (miCBT or RLX) or the waiting period started. Immediately after the last treatment session (miCBT or RLX) or after seven weeks (WLC), the post-assessment was conducted (Figure 1). The WLC participants were randomized to one of the treatments after completion of the post-assessment. Follow-up assessments were carried out 4 and 12 months after completion of the treatment. Each assessment comprised a four-week online headache diary as well as an online survey, which included a set of questionnaires on headache-related factors.
In this study, we analyzed headache days at 12-month follow-up as measure of treatment effect (i.e., as outcome). The 12-month follow-up was selected to investigate the long-term effects of behavioral treatments (which are rarely studied in RCTs). The results of our RCT showed that treatment effects tended to improve even further from 4-month to 12-month follow-up. Thus, we assumed that moderation effects could be better determined for the 12-month follow-up. These long-term improvements were explained by the participants' ability to select appropriate coping strategies and successfully transfer them into everyday life. The increasing improvements were observed in all four main outcomes (headache days, disability, emotional distress, and self-efficacy) and were similar in both treatments (14). Since a reduction in headache or migraine days is still recommended as a primary endpoint in the respective guidelines (20,21), this parameter was selected as the measure of effect in the current study.
Potential moderators
Demographic moderators included sex and age, clinical characteristics included disease duration, migraine type, as well as comorbid mental disorders or somatic conditions. Headache-related disability was assessed via the Headache Disability Inventory (HDI/German version: IBK, Cronbach’s alpha, α = 0.90, range 0 to 4) (22), the Headache Impact Test (HIT-6, α = 0.83, range 6 to 13) (23), and the Pain Disability Index (PDI, α = 0.88, range 0 to 10) (24). Headache-specific self-efficacy was assessed with the short form of the Headache Management Self-Efficacy Scale (HMSE-G-SF, α = 0.72, range 1 to 7) (25). Emotional distress was assessed with the Depression Anxiety Stress Scales (DASS, range 0 to 3) (26), with the subscales Depression (DASS-D, α = 0.92), Anxiety (DASS-A, α = 0.78), and Stress (DASS-S, α = 0.81). Sensitivity to and avoidance of headache triggers was measured with the Headache Triggers Sensitivity and Avoidance Questionnaire (HTSAQ, α = 0.83 to 0.86, range 1 to 5) (27). Acceptance of chronic pain was measured with the 20-item Chronic Pain Acceptance Questionnaire (CPAQ, α = 0.84, range 0 to 6) (28). All questionnaires were provided in a German version (Cronbach’s alphas are each from the credited source).
Statistical analyses
Descriptive statistics (mean, standard deviation, frequency, and percentage) were calculated to describe sample characteristics. To assess differences of subsamples (completer vs drop-out, miCBT vs RLX) regarding baseline characteristics (including candidate moderators), chi-square tests for categorical variables or t-tests for continuous variables (for independent samples) were conducted.
Moderation analysis was performed using the PROCESS macro (v. 3.5.3) in SPSS (v. 23) using ordinary least squares regression to estimate unstandardized coefficients (29). It was analyzed if the effect of treatment (predictor X with two levels: 0 = RLX, 1 = miCBT) on headache days at 12-month follow-up (criterion Y) was moderated by further variables when controlled for pre-treatment headache days. For each moderator, a separate moderation model was run (model 1 with 95% confidence intervals [95% CI] based on 10,000 bootstrapped samples, and heteroscedasticity consistent standard errors [HC3]). In the case of borderline p-values (around .05), the bootstrapped 95% CIs were taken into account, since a CI outside zero can be regarded as an alternative indicator of significance (30). In doing so, a significant moderation effect was qualified either by p < .05 and/or the bootstrapped 95% CI outside the value zero. Analyses were based on the complete-case sample (n = 77), including participants who (a) provided data at all measurement points, (b) completed the treatment, and (c) still met all defined inclusion and exclusion criteria (i.e., participants who became pregnant or started medication prophylaxis, e.g., with a monoclonal antibody, were excluded from further analyses).
In addition to the complete-case analyses, all participants who were randomly assigned to one of the two conditions (intention-to-treat sample, ITT) were analyzed. Since two participants of the WLC could not be randomized to a treatment (due to severe illness or a missing post-measurement), the ITT sample included n = 104 (instead of n = 106 for the pre-post analyses of the RCT) (14). Missing data at follow-up (18.9%) was replaced using last-observation-carried forward (LOCF), as a reproducible and conservative approach to missingness in RCTs, also reducing the risk of false positive values (31). Another reason for preferring the LOCF method was that multiple imputed data cannot be entered in the PROCESS macro, which can be considered one of the most appropriate tools for moderation analyses.
Results
Participant characteristics
Between January 2017, and April 2019, N = 121 participants were randomly assigned to miCBT (40), RLX (41), or WLC (40). The flowchart is displayed in the original study (14). The completer sample (N = 77) included mostly female participants (88%), having migraine with or without aura (97%) as headache diagnosis (Table 1). The mean age was 47.4 years (SD = 12.2, range 21 to 71). Both treatment groups (miCBT vs. RLX) did not differ statistically in candidate moderators or baseline characteristics. As well, the completer and the drop-out sample did not differ statistically in candidate moderators or baseline characteristics, except for headache days (Online Supplementary Table 1). The frequency of headache days was significantly higher (M = 11.1, SD = 5.2 vs M = 7.5, SD = 3.7) in the drop-out sample.
Baseline characteristics (completer sample, N = 77). a
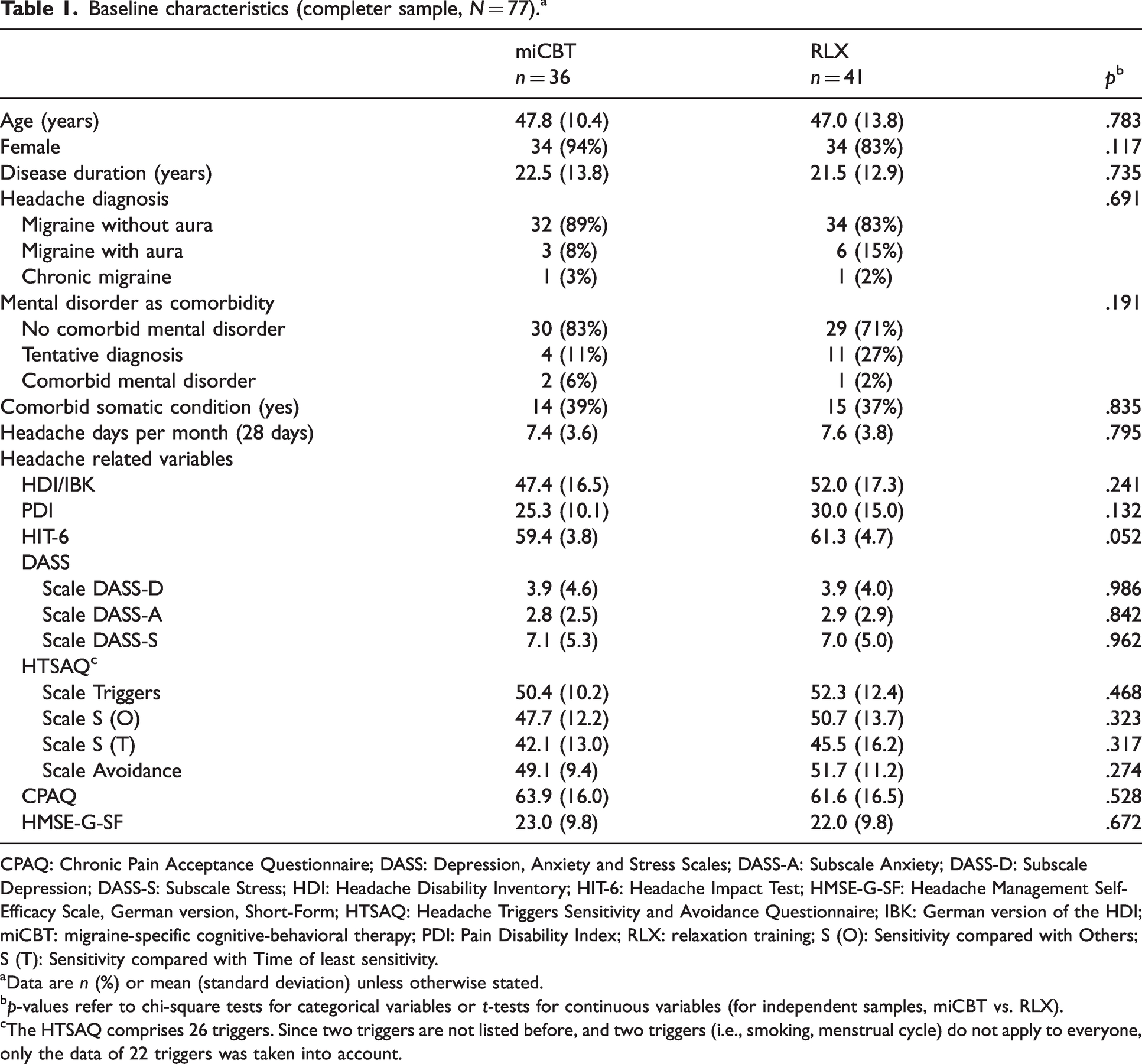
CPAQ: Chronic Pain Acceptance Questionnaire; DASS: Depression, Anxiety and Stress Scales; DASS-A: Subscale Anxiety; DASS-D: Subscale Depression; DASS-S: Subscale Stress; HDI: Headache Disability Inventory; HIT-6: Headache Impact Test; HMSE-G-SF: Headache Management Self-Efficacy Scale, German version, Short-Form; HTSAQ: Headache Triggers Sensitivity and Avoidance Questionnaire; IBK: German version of the HDI; miCBT: migraine-specific cognitive-behavioral therapy; PDI: Pain Disability Index; RLX: relaxation training; S (O): Sensitivity compared with Others; S (T): Sensitivity compared with Time of least sensitivity.
Data are n (%) or mean (standard deviation) unless otherwise stated.
p-values refer to chi-square tests for categorical variables or t-tests for continuous variables (for independent samples, miCBT vs. RLX).
The HTSAQ comprises 26 triggers. Since two triggers are not listed before, and two triggers (i.e., smoking, menstrual cycle) do not apply to everyone, only the data of 22 triggers was taken into account.
Moderation analyses
Headache-related disability (assessed by the HIT-6) significantly moderated the effect of miCBT vs. RLX on headache days at 12-month follow-up (B = –0.41, SE = 0.20, p = .047, 95% CI: –0.85 to –0.10). With regard to the coding of the predictor (here: treatment group, with 0 = RLX and 1 = miCBT), the direction of the moderation effect means, miCBT was associated with better outcomes with increasing HIT-6 scores, whereas RLX was associated with better outcomes at lower HIT-6 scores (Figure 3a). For the DASS-A score, the confidence interval of the interaction effect was outside zero (B = –0.66, SE = 0.34, p = .056, 95% CI: –1.27 to –0.02), which indicates a moderation of the treatment effect in a similar direction. That means, miCBT was associated with better outcomes with increasing DASS-A scores, whereas RLX was associated with better outcomes at lower DASS-A scores (Figure 3b). As well, for the diagnosis of a comorbid mental disorder, the confidence interval of the interaction effect was outside zero (B = –4.98, SE = 2.53, p = .053, 95% CI: –9.42 to –0.29), indicating that miCBT was associated with better outcomes in persons with at least one comorbid mental disorder, whereas RLX was associated with better outcomes in persons without any comorbid mental disorder (Figure 3c).

Visualization of the moderating effect of (a) headache-related disability (assessed by the HIT-6), (b) anxiety (assessed by the DASS-A), and (c) comorbidity of any mental disorder (yes/no), depicted each on the x-axis, on the treatment efficacy (miCBT vs. RLX) regarding headache days at 12-month follow-up as outcome (y-axis). The outcome is controlled for its baseline scores.
Figure 3 is a model visualization of the moderation effects since the two lines of the outcome (headache days at 12-month follow-up, controlled for headache days at baseline) are constituted by only three (Figures 3a, 3b) or two (Figure 3c) reference markings. The three reference markings in Figure 3a and 3b are −1 standard deviation, the mean, and +1 standard deviation since the moderator is continuous. The two reference markings in Figure 3c are the presence (“yes”) or absence (“no”) of any mental disorder (as a dichotomous variable). The figures show that with an increasingly higher disability (HIT-6) or an increasingly higher anxiety (DASS-A), the miCBT tends to be more effective than the RLX (in terms of reduced headache activity), and vice versa. For the dichotomous moderator “mental disorder” (yes/no), it becomes apparent that in the presence of at least one comorbid mental disorder, the miCBT tends to lead to a higher reduction of headache activity compared to the RLX (and vice versa).
The p-values of all candidate moderators are shown in Table 2 (see Table 3 for complete moderation models). No further significant moderation effects were observed in the completer sample. In the ITT sample, no significant p-values were observed, however, the confidence intervals of the interaction effects of comorbid mental disorder as well as of sex were outside zero (Online Supplementary Table 2).
Univariate moderation analysis for the outcome headache days at 12-month follow-up (controlled for headache days at baseline) and treatment group as independent variable (miCBT vs RLX) for the completer sample (n = 77).

CI: Confidence interval; CPAQ: Chronic Pain Acceptance Questionnaire; DASS: Depression, Anxiety and Stress Scales; DASS-A: Subscale Anxiety; DASS-D: Subscale Depression; DASS-S: Subscale Stress; HDI: Headache Disability Inventory; HIT-6: Headache Impact Test; HMSE-G-SF: Headache Management Self-Efficacy Scale, German version, Short-Form; HTSAQ: Headache Triggers Sensitivity and Avoidance Questionnaire; IBK: German version of the HDI; miCBT: migraine-specific cognitive-behavioral therapy; PDI: Pain Disability Index; RLX: relaxation training; S (O): Sensitivity compared with Others; S (T): Sensitivity compared with Time of least sensitivity.
Moderator x treatment condition (0 = RLX, 1 = miCBT) interaction. Each line refers to a separate model.
Headache days refers to a 28-day period.
n = 75 (2 participants with chronic migraine excluded).
n = 75 (2 participants with tentative diagnosis excluded).
The HTSAQ comprises 26 triggers. Since two triggers are not listed before, and two triggers (i.e., smoking, menstrual cycle) do not apply to everyone, only the data of 22 triggers was taken into account.
Full moderation models, i.e., (a) disability by HIT-6, (b) anxiety by DASS-A, and (c) comorbidity of any mental disorder (yes/no), each between treatment conditions (RLX vs. miCBT) a for the outcome (headache days at 12-months follow-up, controlled for headache days at baseline) in the completer sample (n = 77).

DASS-A: Depression, Anxiety and Stress Scales, subscale Anxiety; HIT-6: Headache Impact Test; miCBT: migraine-specific cognitive-behavioral therapy; RLX: relaxation training.
Treatment: 0 = RLX, 1 = miCBT.
Discussion
In an effort to facilitate patient-tailored treatment decisions, this secondary analysis evaluated candidate moderators of the efficacy of a newly developed, cognitive-behavioral migraine prophylaxis (miCBT) compared to relaxation training (RLX). The previous primary analysis of this RCT showed comparable efficacy for the two treatment programs miCBT and RLX (14). Both treatment programs led to an average reduction of about two headache days per month (from pre to 12-month follow-up, responder rate each about 44%). Our current findings demonstrate that patients with high disability, increased anxiety, or comorbid mental disorder(s) benefited more from the miCBT compared to RLX, while patients with relatively lower disability or anxiety, as well as those without comorbid mental disorder(s) benefited more from RLX.
Our results indicate that the challenges of patients with higher psychosocial burden are probably not sufficiently addressed with relaxation training alone. Since high disability, anxiety, or a comorbid mental disorder are usually associated with dysfunctional behavioral patterns, a cognitive-behavioral treatment, which covers a wider range of coping strategies, is probably better suited than relaxation, which targets “only” the level of arousal. In contrast, RLX may be superior in patients who are less burdened. PMR is an effective, easy-to-learn technique for migraine prophylaxis. A strength of the RLX was its focus on this one technique. The repeated exercise of PMR contributed to the transfer to everyday life. Interventions for other issues (e.g., session 3 of the miCBT, coping with fear of attacks) could have a distracting effect on patients, who are not affected by the respective issue (here: fear of attacks). For less burdened patients it is probably better to focus on learning and implementing PMR (according to the principle “more from less”).
Our results provide a preliminary indication that different behavioral interventions have different effects depending on the subgroup of migraine. This finding supports the current German medical guideline on migraine prophylaxis, in which different escalation levels of behavioral treatments are recommended (12). Future headache diagnosis should take into account at least these psychosocial parameters, which are relevant for the selection of behavioral interventions. Martin (13) suggests a multiaxial system beyond categorical headache diagnosis by recommending an assessment of (a) disability, (b) trigger sensitivity and avoidance, and (c) other psychosocial parameters such as lifestyle or dysfunctional coping. Contrary to expectations, trigger sensitivity and avoidance (assessed by the HTSAQ) could not be identified as moderators between treatments in our study. In the miCBT, the dose of trigger management with just one session may have been too low to lead to relevant effects, thus a higher number of sessions addressing this issue should be used for future studies.
In addition to the evaluation of complex, multicomponent treatments such as miCBT, more research (e.g., dismantling studies) is needed to identify moderators for specific treatment components such as trigger management or specific CBT strategies (e.g., coping with fear of attacks). When analyzing the treatment effects of specific behavioral treatments in migraine, the aspect of deterioration and negative effects should also be taken into account (32).
For migraine patients, who do not show any increased scores in a psychological assessment, headache education, in combination with lifestyle recommendations and the regular practice of relaxation may be the better choice. For migraine patients who show increased scores in certain problem areas (e.g., high attack-related fear), the corresponding behavioral intervention (e.g., CBT techniques to cope with attack-related fear) should be recommended.
Limitations
While interpreting our findings, certain limitations have to be considered. First, our explorative study was not powered adequately to detect small-sized interaction effects. Second, since our analyses focused on the completer sample and moderation effects could not be replicated in the ITT sample, the generalizability to a larger patient population with different treatment (non)response is limited. The drop-out sample differed from the completer sample only with regard to a higher frequency of headache days at the beginning of treatment. The two main reasons for a drop-out were declining a measurement (n = 7) and the start of a prophylactic medication (n = 7). Presumably, at least in the latter case, there was a high headache activity (otherwise medication prophylaxis would not have been initiated), which may explain the overall higher headache frequency in the drop-out sample. As a third limitation, additional unmeasured variables such as attack-related fear could have also moderated the effect of miCBT. Since there are now more specific questionnaires with good psychometric properties (e.g., the Fear of Attacks in Migraine Inventory, FAMI, [33]), these parameters should be investigated further.
Conclusion
This secondary analysis of an RCT identified headache-related disability, anxiety, and mental disorder comorbidity as moderators of the effect of behavioral treatment (miCBT vs. RLX) on headache days in the long-term. While a more complex, migraine-specific treatment with several approaches (miCBT) may be indicated for patients with higher disability, anxiety, or a comorbid mental disorder, a simpler treatment approach (RLX) may be better for patients, who are less burdened. Further, adequately powered studies are needed to confirm these preliminary results and advance tailored multimodal treatment selection for migraine.
Clinical implications
In migraine prophylaxis, it is still unclear which behavioral approach is best suited for which type of patient. Disability, anxiety, and comorbid mental disorder were identified as moderators of headache days at the 12-month follow-up between miCBT or RLX. Higher-burdened patients benefited more from miCBT whereas lower-burdened patients benefited more from RLX. Prospective studies are needed to confirm these preliminary results.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024231178237 - Supplemental material for Behavioral treatment for migraine prophylaxis in adults: Moderator analysis of a randomized controlled trial
Supplemental material, sj-pdf-1-cep-10.1177_03331024231178237 for Behavioral treatment for migraine prophylaxis in adults: Moderator analysis of a randomized controlled trial by Timo Klan, Charly Gaul, Eva Liesering-Latta, Michael Witthöft and Severin Hennemann in Cephalalgia
Footnotes
Acknowledgments
We are grateful to all participants who completed the treatment and the assessments. We thank the Department of Psychology (University of Mainz), the research staff, and the therapists for their contributions to the study. Also, we thank the sapite GmbH (Klein-Winternheim, Germany) for cooperation, providing their software, and assistance in the conception of the headache diary.
Data sharing
De-identified data of the current study is available online at the Open Science Framework (OSF): Klan, T. (2022, December 29). Behavioral Treatment for Migraine Prophylaxis. Retrieved from osf.io/m9r7d
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CG has received honoraria for consulting and lectures within the past three years from Abbvie, Lilly, Novartis Pharma, Hormosan Pharma, Grünenthal, Sanofi-Aventis, Weber & Weber, Lundbeck, Perfood, and TEVA. CG is honorary secretary of the German Migraine and Headache Society. ELL has received honoraria for lectures within the past three years from Allergan Pharma, Lilly, and TEVA. ELL and TK published a treatment manual about CBT for migraine. All other authors declare no competing interests.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The RCT was funded by the German Migraine and Headache Society (DMKG) and by internal funds (Department of Psychology, Johannes Gutenberg University of Mainz).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
