Abstract
Background
Safety data on the use of migraine preventive monoclonal antibodies targeting the calcitonin gene-related peptide (CGRP) system in pregnancy are limited.
Methods
Updated pharmacovigilance assessment of the safety reports related to pregnancy associated with erenumab, galcanezumab, fremanezumab and eptinezumab, retrieved from VigiBase® as of 31 December 2021. As primary outcome, the whole group of monoclonal antibodies targeting the CGRP system was considered and sex and age subgroup disproportionality analyses using the reporting odds ratio (ROR) were conducted.
Results
286 safety reports were found: 116 (40.6%) on erenumab, 125 (43.7%) on galcanezumab, 39 (13.6%) on fremanezumab, 6 (2.1%) on eptinezumab. One hundred and forty-nine (52.1%) safety reports reported only drug exposure in relation to pregnancy while 137 (47.9%) also included ≥1 pregnancy outcomes: maternal outcomes (n = 64), spontaneous abortion (n = 63), foetal growth restriction (n = 1), prematurity (n = 8), neonatal outcomes (n = 13), and poor breastfeeding (n = 1). No specific patterns of maternal, foetal and neonatal toxicity were observed. Spontaneous abortion was not disproportionally more frequently reported with erenumab, galcanezumab, fremanezumab and eptinezumab compared with the entire database (ROR 1.1, 95% confidence interval, CI, 0.8–1.5), the entire database since 2018 (ROR 1.3, 95% CI 1.0–1.8), and triptans (ROR 1.2, 95% CI 0.8–1.9).
Conclusions
This updated safety analysis on erenumab, galcanezumab, fremanezumab and eptinezumab in pregnancy showed no signals of foeto-maternal toxicity according to VigiBase® safety reports.
Introduction
Migraine affects about 25% of women of childbearing age (1). Most women with migraine experience improvement over the course of pregnancy (2), so that migraine preventive treatments are generally not needed in this setting. However, unintended pregnancies or simply being unaware of an initial pregnancy status are very common events (3), and in these cases using migraine preventive therapies might expose pregnant women or their foetuses to potential drug-related adverse events.
Erenumab, galcanezumab, fremanezumab and eptinezumab are monoclonal antibodies recently approved for the prevention of episodic and chronic migraine (4), which target the calcitonin gene-related peptide (CGRP) system, a key mediator of migraine pain (5). Currently, the use of these agents is not recommended during pregnancy because of the limited amount of pregnancy-related safety data available in humans (6,7). Since marketing authorisation, five clinical cases of accidental exposure to erenumab during pregnancy and a series of 94 safety reports concerning the exposure to erenumab, galcanezumab and fremanezumab in relation to pregnancy, spontaneously reported in VigiBase®, the World Health Organization (WHO) pharmacovigilance database, have shown no signals of specific maternal toxicities, patterns of major birth defects, or increased reporting of spontaneous abortion (8–11).
The aim of the present study was to provide an updated pharmacovigilance assessment in VigiBase® of the safety profile of erenumab, galcanezumab, fremanezumab and eptinezumab in pregnancy by performing disproportionality analyses with reporting odds ratio (ROR) computation.
Methods
Data retrieval
De-duplicated safety reports associated with erenumab, galcanezumab, fremanezumab and eptinezumab concerning pregnancy and collected in VigiBase® as of 31 December 2021 were retrieved by using the Standardized Medical Dictionary for Regulatory Activities (MedDRA) Query (SMQ) “Pregnancy and neonatal topics” (MedDRA version 25.0). Erenumab, galcanezumab, fremanezumab and eptinezumab were selected as active ingredients when classified as suspected. Detailed description of data retrieval process, aims and structure of VigiBase® has been previously reported (11). Safety reports lacking specific terms referring to drug exposure in relation to pregnancy and/or presenting partial or missing dates (of drug administration, pregnancy status, and/or occurrence of reported events related to pregnancy), as well as safety reports involving patients of implausible or unknown sex and/or age, and/or reporting general adverse events (not necessarily related to pregnancy), were excluded from the study population (online Supplementary Table 1).
Safety reports’ characteristics
Characteristics of the safety reports included in the study were evaluated, which encompassed patient sex, patient age at the time of reporting, the suspected monoclonal antibody targeting the CGRP system and its indication, the country of origin of the safety report, the reporting year, and the type of reporter. In safety reports referring of exposure to erenumab, galcanezumab, fremanezumab and eptinezumab in relation to pregnancy and reporting pregnancy outcomes, the latter were clustered as follows: maternal outcomes, foetal and neonatal outcomes (including spontaneous abortion, foetal growth restriction, prematurity, birth defects, and other neonatal outcomes), and events related to lactation. A case-by-case assessment of maternal age, concomitant medications and underlying comorbidities, was performed by searching in Reprotox® database (https://www.reprotox.org/) and by screening the scientific literature (MEDLINE) in order to define potential confounding factors for the solely specific pregnancy outcomes eligible for disproportionality analysis (i.e. reported in ≥5 safety reports, as explained below).
Medians and interquartile ranges were used to describe quantitative variables, counts and percentages for qualitative variables. Missing data were omitted from the description of correspondent variables. Data management and analysis were performed using the Statistical Analysis System Software (version 9.4; SA Institute, Cary, NC).
Disproportionality analyses
Disproportionality analysis with ROR and 95% confidence interval (CI) computation was used to detect potential safety signals of increased reporting of specific pregnancy outcomes with erenumab, galcanezumab, fremanezumab and eptinezumab. Since subgroup disproportionality analyses have shown benefits in both sensitivity and precision over crude disproportionality analyses for large international databases such as VigiBase® (12), disproportionality analyses were performed in subgroups of safety reports involving female patients aged 18 to 48 years (the age range observed among the safety reports included in the study, Table 1). Three comparator groups were used: 1) the entire database (over the time frame of data retrieval); 2) the entire database with temporal restriction (that is since 2018 when the first-in-class erenumab received marketing authorisation); 3) triptans (of the anatomical, therapeutic and chemical group N02CC, in an analysis by therapeutic area to reduce confounding by indication) (13). As primary outcome, the whole group of monoclonal antibodies targeting the CGRP system was considered in the disproportionality analyses., Disproportionality analyses with the same three comparator groups were then performed separately according to the mechanism of action, i.e. for erenumab (targeting the CGRP receptor) and galcanezumab, fremanezumab and eptinezumab (targeting the CGRP ligand) and were considered secondary study outcomes. The threshold for signal detection was defined with the ROR lower limit of the 95% CI > 1 and the number of safety reports concerning the pregnancy outcome of interest ≥5 (13). All disproportionality analyses were repeated as sensitivity analyses after the exclusion of safety reports with confounding factors.
Characteristics of safety reports included in the study.

aOne child-mother safety report on fremanezumab (describing the child), one safety report concerning paternal exposure to galcanezumab, one safety report concerning a male child of unknown age exposed to erenumab during lactation, one safety report concerning a 2-year-old male child exposed to erenumab during lactation, one safety report of prematurity associated with erenumab and concerning a male child, one safety report of jaundice associated with erenumab and involving a 2-month-year-old male neonate.
bTwo safety reports of prematurity with galcanezumab; three safety reports of prematurity with erenumab; one safety report with erenumab of neonatal cerebral haemorrhage and epilepsy; one safety report with erenumab of meningomyelocele.
cAustria, Czechia, Sweden, n = 3; Argentina, Norway, Poland, Portugal, United Arab Emirates, n = 2; Belgium, Brazil, Croatia, Finland, Lithuania, Netherlands, Spain, n = 1.
Abbreviations: IQR interquartile range.
Standard protocol approvals, registrations, and patient consents
According to the Human Research Act (810.30, of 30 September 2011 – status as of 26 May 2021), from the Federal Assembly of the Swiss Confederation, ethical approval and written informed consents were not required (Art. 2: “It does not apply to research which involves anonymously collected or anonymised health-related data”).
Results
As of 31 December 2021, out of 29,113,988 safety reports in VigiBase®, 331 were associated with erenumab, galcanezumab, fremanezumab and eptinezumab and concerned pregnancy (Figure 1). Of these, 45 (13.6%) safety reports were excluded (based on the exclusion criteria stated in the Methods section, see online Supplementary Table 1 for the detailed list).

Consort diagram showing the selection process of safety reports included in the study.
Table 1 shows the characteristics of the 286 safety reports finally included in the study of which 116 (40.6%) with erenumab, 125 (43.7%) with galcanezumab, 39 (13.6%) with fremanezumab and six (2.1%) with eptinezumab.
In 253 (88.5%) safety reports exposure to erenumab, galcanezumab, fremanezumab and eptinezumab occurred during pregnancy, in 11 (3.8%) safety reports drug exposure occurred before pregnancy (Table 2). Of these, seven safety reports reported only exposure to either galcanezumab or fremanezumab before pregnancy without additional pregnancy outcomes and without providing further information on the time gap between the end of drug treatment and the occurrence of pregnancy. One safety report reported missed abortion (i.e. nonviable pregnancy in the absence of symptoms), gestational trophoblastic detachment and drug ineffectiveness about four months after the end of treatment with erenumab. In two safety reports associated with fremanezumab and reporting spontaneous abortion and drug infectiveness respectively, no information was available to determine the time elapsed between the end of treatment with fremanezumab and the onset of the two reported events. 149 (52.1%) safety reports only mentioned drug exposure whereas the remnant 137 (47.9%) reported drug exposure and one or more pregnancy outcomes. Overall, pregnancy outcomes were 219 clustered as follows: maternal outcomes (n = 123), spontaneous abortion (n = 63), foetal growth restriction (n = 1), prematurity (n = 9), neonatal outcomes (n = 23 of which nine had birth defects) and events related to lactation (n = 1) (Table 3).
Assessment of drug exposure in the safety reports included in the study.

aOne safety report reporting paternal exposure before pregnancy (however, unknown how long before), without pregnancy outcomes.
bWith erenumab: one safety report of prematurity and one safety report of spontaneous abortion.
With galcanezumab: four safety reports with unknown timing of maternal exposure (and without pregnancy outcomes), one safety report of spontaneous abortion, one safety report of drug ineffective.
With eptinezumab: three safety reports with unknown timing of maternal exposure (and without pregnancy outcomes).
Pregnancy outcomes reported with erenumab, galcanezumab, fremanezumab and eptinezumab as of 31 December 2021.

aSafety reports reporting only drug exposure were excluded from this analysis.
bSome safety reports reported more than one pregnancy outcome.
Maternal outcomes
Sixty-five safety reports included 124 maternal outcomes (online Supplementary Table 2). The most frequent maternal outcome was ineffectiveness of the anti-migraine treatment, which occurred in 30 (46.9%) safety reports (24 with erenumab, four with galcanezumab and two with fremanezumab). Six safety reports included local adverse events at the injection site, four nausea and/or vomiting, four constipation, two arterial hypertension and one pre-eclampsia.
Foetal and neonatal outcomes
One case of foetal growth restriction was reported with erenumab. There were eight (2.8%) safety reports of prematurity. Of these, five with erenumab and two with galcanezumab did not mention additional pregnancy outcomes and did not have confounding factors. One safety report of prematurity with erenumab involved a 37-year-old women who suffered from placental insufficiency and pre-eclampsia before delivering (maternal age as confounding factor for prematurity) (14).
Heterogeneous birth defects were reported in four safety reports with erenumab (anencephaly, congenital diaphragmatic hernia, meningomyelocele, thyroglossal cyst and spina bifida, respectively), in two with galcanezumab (talipes and atrial septal defect with anomalous pulmonary venous connection) and in one with fremanezumab (renal aplasia and gastroesophageal reflux disease) (online Supplementary Table 2). The remnant neonatal outcomes were also miscellaneous and reported with either erenumab (five safety reports) or galcanezumab (one safety report) (online Supplementary Table 2).
Sixty-three (22.0%) cases of spontaneous abortion were reported, of which 30 were without confounding factors (19 with erenumab, seven with galcanezumab, three with fremanezumab and one with eptinezumab). In 11 cases, maternal age over 35 years was the only confounding factor (15). In 10 cases spontaneous abortion occurred in younger women, however concomitant medications and/or underlying comorbidities acted as confounding factors. Lastly, 12 cases were confounded by maternal age over 35 years, concomitant medications and/or underlying comorbidities (online Supplementary Table 3) (16–35).
Disproportionality analysis
Pregnancy outcomes reported in ≥5 safety reports were spontaneous abortion and prematurity. However, subgroup disproportionality analyses could not be performed for prematurity because only two of the eight safety reports included in the study cohort referred to female patients aged 18–48 years. Out of 63 safety reports of spontaneous abortion with erenumab, galcanezumab, fremanezumab and eptinezumab, 21 (16 with erenumab, four with galcanezumab and one with eptinezumab) did not report patient sex and/or age, therefore were excluded from subgroup disproportionality analyses.
Reporting of spontaneous abortion with the whole group of monoclonal antibodies targeting the CGRP system in female patients aged 18–48 years was similar to reporting of spontaneous abortion by the same patient subgroup within the entire database over the time frame of data retrieval (ROR 1.1, 95% CI 0.8–1.5), within the entire database with temporal restriction (ROR 1.3, 95% CI 1.0–1.8), and with triptans (ROR 1.2, 95% CI 0.8–1.9). When subgroup disproportionality analyses were repeated after exclusion of safety reports of spontaneous abortion with erenumab, galcanezumab, fremanezumab and eptinezumab presenting confounding factors, no signals of disproportionate reporting were found either (against none of the three predefined comparator groups) (Figure 2).
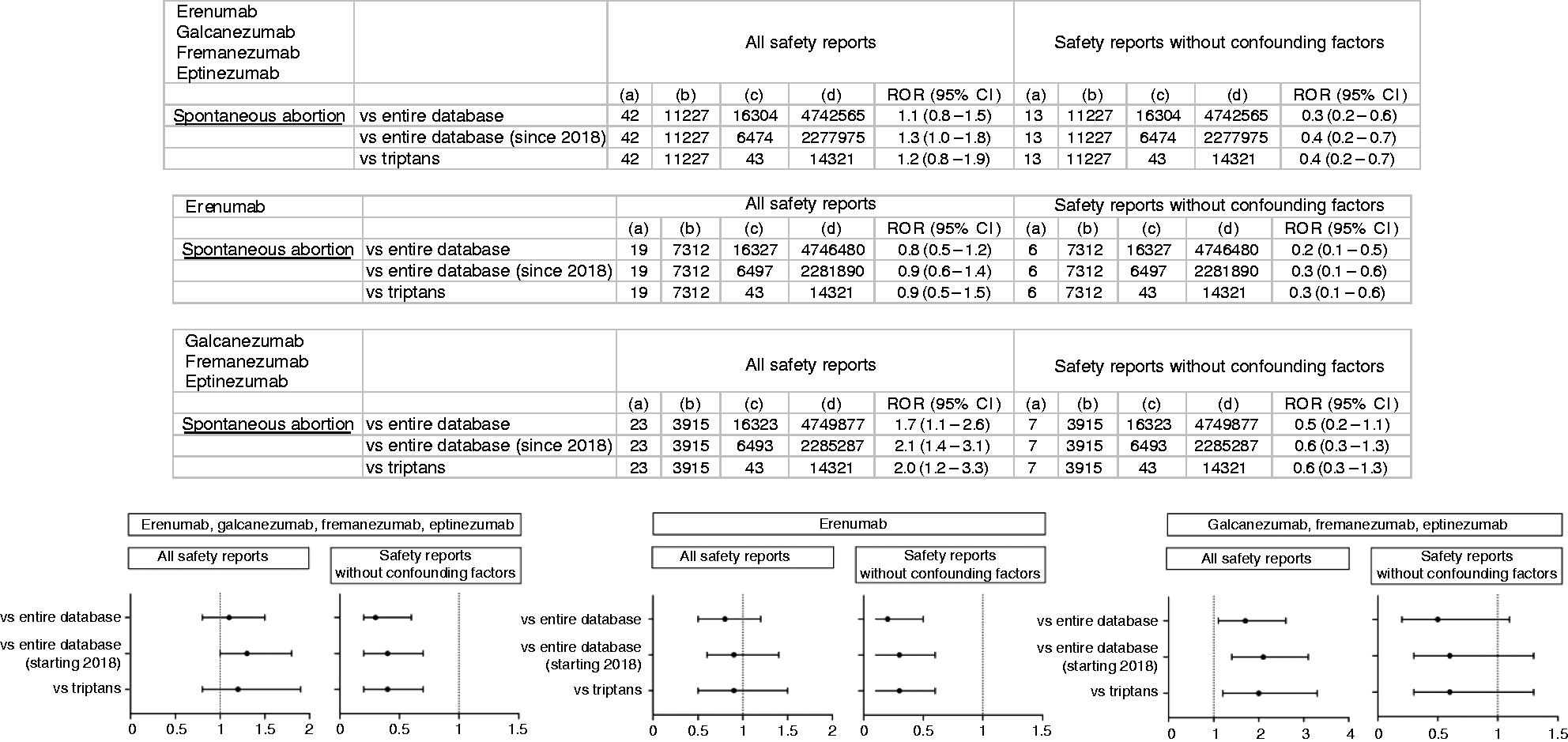
Computation and forest plots of reporting odds ratios and 95% confidence intervals in subgroup disproportionality analyses. (a) Number of safety reports of spontaneous abortion reported in association with erenumab, galcanezumab, fremanezumab and eptinezumab (all the four monoclonal antibodies targeting the calcitonin gene related peptide, CGRP, system); with erenumab (which targets the CGRP receptor); and with galcanezumab, fremanezumab and eptinezumab (which target the CGRP ligand) and (b) Number of safety reports concerning events other than spontaneous abortion reported in association with erenumab, galcanezumab, fremanezumab and eptinezumab; with erenumab; and with galcanezumab, fremanezumab and eptinezumab.
Subgroup disproportionality analyses performed separately by mechanism of action did not detect signals of disproportionate reporting of spontaneous abortion with erenumab (which targets the CGRP receptor) compared to none of the three predefined comparator groups, neither when all safety reports of spontaneous abortion with erenumab were used to compute the ROR, nor after exclusion of safety reports with confounding factors (Figure 2). Conversely, signals of disproportionate reporting for spontaneous abortion with galcanezumab, fremanezumab and eptinezumab (which target the CGRP ligand) were detected in the comparisons with the entire database over the time frame of data retrieval (ROR 1.7, 95% CI 1.1–2.6), the entire database with temporal restriction (ROR 2.1, 95% CI 1.4–3.1), and triptans (ROR 2.0, 95% CI 1.2–3.3). However, these signals were not confirmed after excluding safety reports with confounding factors (Figure 2).
Discussion
This updated analysis in VigiBase® of safety reports gathered up to 31 December 2021 shows no specific patterns of maternal, foetal or neonatal toxicity reported with erenumab, galcanezumab, fremanezumab and eptinezumab and no signals of increased reporting of spontaneous abortion for the whole group of monoclonal antibodies targeting the CGRP system.
Knowledge of drug safety in pregnancy is often limited due to general exclusion of pregnant women from clinical trials because of teratogenicity risks. Therefore, alternative sources of information in the post-marketing setting, such as pregnant women inadvertently exposed to drugs or in whom the risks of stopping treatment are perceived to outweigh any potential risks to the baby, are of paramount importance. These include pregnancy registries established by manufacturers, academic groups and regulatory authorities, published clinical cases and spontaneous reporting systems. The value of the spontaneous reporting system VigiBase® as data source to gain information on drug safety in pregnancy is demonstrated by the progressive increase in the number of pharmacovigilance studies so far published that used disproportionality techniques to detect potential safety signals (11,36–40). Moreover, VigiBase® has been tested recently and validated as complementary reference source in the evaluation of safety signals detected in the EUROmediCAT database which collects data from European population-based registries on medication exposure in pregnancies with a congenital anomaly (41).
With regard to the monoclonal antibodies targeting the CGRP system, current evidence on their safety in pregnancy in humans is still limited (8–11). Five clinical cases of women exposed to erenumab during pregnancy have been described without complications, abnormalities or adverse events in the mother and the child (8–10). A previous pharmacovigilance study in VigiBase® by our group identified 94 safety reports associated with erenumab, galcanezumab and fremanezumab and related to pregnancy by the end of 2019 without observing specific maternal toxicities, patterns of major birth defects or increased reporting of spontaneous abortion (11). A peculiarity of spontaneous reporting systems such as VigiBase® is that the number of safety reports collected increases over time thus offering the possibility of retrieving updated series of safety reports of interest as well as of routinely applying data mining techniques aimed at detecting potential safety signals. Therefore, two years later, we queried VigiBase® to provide an updated pharmacovigilance assessment of the safety profile in pregnancy of the monoclonal antibodies targeting the CGRP system, which to date include more than erenumab, galcanezumab and fremanezumab, such as the most recently approved eptinezumab. New safety reports collected in the period 1 January 2020–31 December 2021 were analysed in combination with the previously published 94 safety reports.
With regard to reported maternal outcomes, new analyses confirm that there are no specific toxicity patterns in the mother, for whom the most frequently reported event continues to be the lack of efficacy of the anti-migraine treatment. Although migraine has been associated with increased risk of vascular disorders in pregnancy, particularly gestational hypertension and preeclampsia (42), the CGRP system is involved in the maintenance of normal gestational blood pressure and its levels increase during pregnancy up to the last trimester presumably to regulate the foeto-placental vascular tone (43,44). Preclinical studies have shown that CGRP blockade during pregnancy results in an increase of systolic blood pressure and foetal mortality (45,46), thus posing the challenge of a potential increase in gestational hypertension, pre-eclampsia and eclampsia in migraine patients treated with monoclonal antibodies targeting the CGRP system. Consistently, two safety reports of maternal hypertension associated with erenumab were included in the present study, while no safety reports of pre-eclampsia other than the one previously described with erenumab were collected (11). Noteworthy, in these three cases, exposure to erenumab occurred during pregnancy, however, with no information on the exact trimester of gestation. With half-life time of about one month, if drug exposure had occurred during the first trimester, any adverse effects on the subsequent course of pregnancy (including pre-eclampsia and eclampsia) may have been missed. Conversely, drug exposure in the following trimesters could have resulted in the occurrence of such events. Therefore, the use of the monoclonal antibodies targeting the CGRP system for the entire duration of pregnancy might not always be safe.
Among reported foetal/neonatal outcomes, there was one newly-added safety report of foetal growth restriction and a few more safety reports of prematurity compared with the previous study (11). Due to the paucity of clinical information, neither the exact gestational period of drug exposure nor the neonatal consequences of these premature births reported in VigiBase® are known. Nevertheless, the fact that in none of the five published clinical cases did a premature birth occur (8–10) may suggest that this particular event deserves to be monitored over time as potential safety signal with monoclonal antibodies targeting the CGRP system. In this study, as in the previous one (11), no patterns of major birth defects were found.
Spontaneous abortion was confirmed as the most frequently reported foetal outcome. Even with a higher number of safety reports, no safety signal with the whole group of monoclonal antibodies targeting the CGRP system was found from disproportionality analyses. Furthermore, the reporting of spontaneous abortion with the whole group of monoclonal antibodies targeting the CGRP system did not increase either in the comparison with the entire database (with and without the time restriction from 2018 onwards) or with triptans when only safety reports without confounding factors for spontaneous abortion were considered. By contrast, ROR values from the latter analyses were far below one suggesting that maternal age, concomitant medications and underlying comorbidities likely contribute to increased reporting of spontaneous abortion. Since erenumab is the only monoclonal antibody targeting the CGRP receptor while galcanezumab, fremanezumab and eptinezumab target the CGRP ligand, disproportionality analyses for spontaneous abortion were performed separately by mechanism of action. Although no signals for spontaneous abortion were found with erenumab, ROR values obtained from safety reports without confounding factors were lower than those from all safety reports. Such results mirror those obtained with the whole group of monoclonal antibodies targeting the CGRP system and are similarly explainable. Noteworthy, increased reporting of spontaneous abortion did emerge with galcanezumab, fremanezumab and eptinezumab albeit not confirmed after excluding safety reports with confounding factors. We therefore performed separate disproportionality analyses for the three monoclonal antibodies targeting the CGRP ligand. No signals for spontaneous abortion emerged with galcanezumab, whereas the reporting of spontaneous abortion was found to be disproportionately increased with fremanezumab (online Supplementary Table 4). In this case, the analysis of safety reports without confounding factors was not possible due to insufficient numbers, preventing any definitive conclusions. Also, only one safety report of spontaneous abortion was reported with eptinezumab, preventing any computation.
VigiBase® as spontaneous reporting system suffers from inherent limitations among which are reporting biases, partial and missing information, paucity of clinical details (in this particular study concerning, among others, drug doses, treatment duration, the exact gestational period of drug exposure), and confounding issues (47). In VigiBase®, the total number of safety reports for a specific drug is known as it is the number of safety reports with a specific adverse event, however information on the number of patients exposed to a certain drug (or group of drugs) not affected by (and not reporting) adverse events is lacking. Accordingly, the ROR is not a measure of risk and does not inform about the incidence of an adverse event of interest, rather, as signal of disproportionate reporting, it indicates that a statistically significant disproportionality exists in the reporting frequency of that adverse event (48).
Conclusions
This updated safety analysis on erenumab, galcanezumab, fremanezumab and eptinezumab in pregnancy according to VigiBase® safety reports showed no consistent signals of foeto-maternal toxicity. Continuous monitoring through spontaneous reporting systems together with results from pregnancy registries and future pharmacoepidemiological studies will contribute to a more complete picture of the safety of these drugs in pregnancy. Until then and according to their current labels, use of monoclonal antibodies targeting the CGRP system during pregnancy should still be avoided. Given their half-life of about one month, women wishing to become pregnant should be advised to discontinue these anti-migraine treatments at least five months before conception, and immediately in case of unplanned pregnancy.
Article highlights
This updated pharmacovigilance assessment in VigiBase® of the safety profile of erenumab, galcanezumab, fremanezumab and eptinezumab in pregnancy showed no consistent signals of foeto-maternal toxicity. Continuous monitoring through spontaneous reporting systems together with results from pregnancy registries and future pharmacoepidemiological studies will contribute to have a more complete picture of the safety of these drugs in pregnancy.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024231158083 - Supplemental material for Safety profile of monoclonal antibodies targeting the calcitonin gene-related peptide system in pregnancy: Updated analysis in VigiBase®
Supplemental material, sj-pdf-1-cep-10.1177_03331024231158083 for Safety profile of monoclonal antibodies targeting the calcitonin gene-related peptide system in pregnancy: Updated analysis in VigiBase® by Roberta Noseda, Francesca Bedussi, Claudio Gobbi, Alessandro Ceschi and Chiara Zecca in Cephalalgia
Supplemental Material
sj-pdf-2-cep-10.1177_03331024231158083 - Supplemental material for Safety profile of monoclonal antibodies targeting the calcitonin gene-related peptide system in pregnancy: Updated analysis in VigiBase®
Supplemental material, sj-pdf-2-cep-10.1177_03331024231158083 for Safety profile of monoclonal antibodies targeting the calcitonin gene-related peptide system in pregnancy: Updated analysis in VigiBase® by Roberta Noseda, Francesca Bedussi, Claudio Gobbi, Alessandro Ceschi and Chiara Zecca in Cephalalgia
Supplemental Material
sj-pdf-3-cep-10.1177_03331024231158083 - Supplemental material for Safety profile of monoclonal antibodies targeting the calcitonin gene-related peptide system in pregnancy: Updated analysis in VigiBase®
Supplemental material, sj-pdf-3-cep-10.1177_03331024231158083 for Safety profile of monoclonal antibodies targeting the calcitonin gene-related peptide system in pregnancy: Updated analysis in VigiBase® by Roberta Noseda, Francesca Bedussi, Claudio Gobbi, Alessandro Ceschi and Chiara Zecca in Cephalalgia
Supplemental Material
sj-pdf-4-cep-10.1177_03331024231158083 - Supplemental material for Safety profile of monoclonal antibodies targeting the calcitonin gene-related peptide system in pregnancy: Updated analysis in VigiBase®
Supplemental material, sj-pdf-4-cep-10.1177_03331024231158083 for Safety profile of monoclonal antibodies targeting the calcitonin gene-related peptide system in pregnancy: Updated analysis in VigiBase® by Roberta Noseda, Francesca Bedussi, Claudio Gobbi, Alessandro Ceschi and Chiara Zecca in Cephalalgia
Footnotes
Data Availability Statement
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
