Abstract
Background/objective
Primary stabbing headache is a common but under-recognized primary headache disorder. The objectives of this review were to provide practical information for better understanding and identification of the disease, suggest an algorithm for differential diagnosis, and provide an insight into the pathophysiology of primary stabbing headache hypothesized from its clinical course.
Methods
This narrative review of primary stabbing headache is based on a literature search and the authors’ clinical reasoning.
Result
The phenotype of each stab is typically abrupt, ultrashort-lasting (<3 s), focal or multifocal, paroxysms of pain occurring sporadically or in clusters. The diagnosis of primary stabbing headache is clinical; fixed or migrating stabs without background pain or sensory abnormalities and the absence of features suggestive of other disorders (e.g., cranial autonomic symptoms or signs) can aid in the diagnosis of primary stabbing headache. The clinical patterns include monophasic, intermittent, and chronic primary stabbing headache, of which the first two are considered typical. The pathophysiology of primary stabbing headache has not yet been elucidated. In this review, we postulated the mechanism of stabbing headache, based on the pain phenotype and clinical course, and provide a clinical algorithm for the differential diagnosis of primary stabbing headache.
Conclusion
Knowledge about the typical manifestations and clinical patterns of primary stabbing headache will aid in the proper diagnosis and differential diagnosis. Treatment should be tailored by considering the clinical patterns. Further research is needed to elucidate the pathophysiological mechanisms and optimal treatment of primary stabbing headache.
Introduction
Primary stabbing headache (PSH) is a common but under-recognized primary headache disorder. PSH was formerly termed icepick pain, jabs and jolt syndrome, needle-in-the-eye syndrome, ophthalmodynia periodica, sharp short-lived head pain, and idiopathic stabbing headache (ISH) (1–3). PSH is a common cause of headache clinic visit (4–6). Although PSH is basically a benign disorder, the sudden nature of the pain negatively affects both patients and physicians: patients may be overly concerned about their symptoms, and physicians may misinterpret the pain as thunderclap headache and prescribe unnecessary imaging studies (7). Proper knowledge of this condition would help save time and costs in most cases. Differential diagnosis is also important because, although rarely, stabbing headaches can be a presenting symptom of life-threatening conditions (8–10). The objectives of this review were to provide an outline of the disease for better understanding and identification, to suggest an algorithm for differential diagnosis, and to provide an insight into the pathophysiology of PSH hypothesized from its clinical courses based on clinical reasoning.
Epidemiology
Several population-based studies have estimated the prevalence of PSH in the general population. Across studies, the lifetime prevalence of PSH has been reported differently, from 2% of 740 people from the Danish population aged 25 to 64 years (11) to 35.2% of 1779 people from the Norwegian population aged 18 to 65 years (12), probably due to differences in case definition (i.e. the location and duration of stabs). A very recent study reported a 26.7% lifetime prevalence of PSH among neurologists, which ensures a high accuracy of diagnosis (13). This finding supports a high prevalence of PSH in the general population.
Prevalence studies using hospital-based samples have also yielded various results. In single university hospital-based studies, the diagnosis of PSH was made in 1.5% of 1219 neurology outpatients in China (14), 5% of 725 patients in a Spanish headache clinic (7), 12.6% of 245 headache outpatients after excluding secondary headaches in Turkey (6), and 13% of 872 patients in an outpatient headache clinic in Taiwan (5). In a multicenter study conducted in Korea, PSH was diagnosed in 11% of 1592 first-visit patients visiting outpatient headache clinics (4). In this study, PSH was the third most common diagnosis, after migraine and tension-type headache (4). PSH is more frequent in individuals with primary headache disorders, with a reported prevalence of approximately 40% in migraineurs (1,15).
Although clinical studies reported that the prevalence of PSH was higher in women than in men (2,5,14,16,17), population studies have shown that men and women are equally affected (11,12). The age of onset is widely distributed, ranging from two to 83 years of age across clinical studies (5,17,18), and is more common in adults than in preschool or school-age children in the general population (12). Among them, patients in their 50 s or 60 s commonly presented to headache clinics (5).
Clinical manifestation
Pain phenotype
Clinical manifestations of PSH are summarized in Table 1. The pain of PSH is characterized by ultrashort-lasting, sharp paroxysms (“stabs”) occurring on a small area of the head. The typical duration of stabs is one second or less (2,5,12,16,17). Occasionally, stabs can last longer (2,5,19). In the International Classification of Headache Disorders (ICHD)-3, it is noted that 80% of stabs last three seconds or less, and rarely, stabs can last for 10–120 s (3). However, with increasing duration of stabs, more attention should be paid to the probability of primary or secondary headache disorders other than PSH (see the Diagnosis section below).
Clinical manifestations of primary stabbing headache.

The location of stabs has been reported to be frontal, temporal, parietal, occipital, orbital, around the vertex, and even on the face (1,2,5,16,17,20–22). It is now accepted that PSH occurs in an extratrigeminal location in approximately 70–80% of PSH patients (5,16,17,20,21), in contrast to earlier studies where anterior locations such as the orbital, supraorbital, frontal, and temporal regions were common sites of pain in patients with PSH (1,2). A recent clinic-based study reported a lower prevalence of extratrigeminal stabs (53%) in 32 patients (22). Stabs can be multifocal and migrating from one area to another, which has been reported in 23–64% of patients with PSH (1,2,5,17,20,22). In patients with migraine, stabs tend to be localized to areas where migraine attacks frequently occur, and can sometimes occur during migraine attacks. Stabs can involve the face (V2 and/or V3) (16), and facial stabs occurring exclusively within the V2 or V3 dermatome have been reported (23,24). Whether this is a variant of PSH or a new facial pain disorder should be discussed in the next version of the ICHD.
The jolt, which is a sudden movement, can accompany stabs in 38–74% of patients (5,17), which is quite characteristic for PSH. Vocalization has also been reported in 18% of patients (5). Bodily jabs have been reported infrequently in population-based surveys (1.1% in the Vågå study) (24); however, they are relatively common in clinical studies (12–13%) (5,17). Allodynia has been reported in 19–37% of patients in clinic-based studies (5,16,17). While nausea, vomiting, photophobia, and phonophobia have been reported as accompanying symptoms (5,14), they are not prominent in most patients with typical PSH that does not overlap with migraine attacks.
Clinical pattern and course
During a brief episode, stabs occur sporadically or in a clustering pattern. Such sporadic or a series of stabs can recur infrequently (e.g. once every few months or years) or as frequently as dozens or hundreds of attacks per day (5,17,22). Between stabs, patients are asymptomatic in typical cases. If headache of any degree or sensory abnormalities persist, other primary or secondary headache disorders or cranial neuropathies should be considered first (see the Diagnosis section below).
From a long-term perspective, the clinical course of PSH can be monophasic, intermittent, or chronic daily (Figure 1) (2,17,22). In monophasic PSH, attacks are more frequently located in fixed areas with acute or subacute onset and daily occurrence, whereas chronic daily PSH has more migrating locations (17). Intermittent PSH manifests as infrequent episodes of sporadic or clustered stabbing pain. This is believed to be the predominant type in the general population, as reported in migraine-free controls in a case-control study (1).
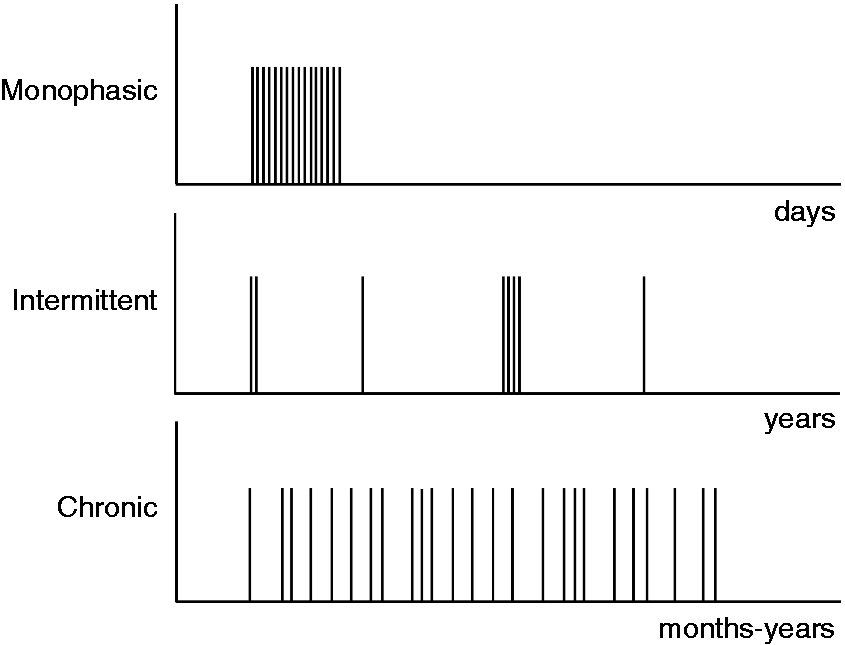
Clinical patterns of primary stabbing headache.
Clinical manifestation in special population: Children/adolescents and elderly
Stabbing headaches in children and adolescents have been described in several clinic-based studies (18,25–27). The age of onset was reported to be as young as two years (17). Although the mean age of onset differed slightly among studies, e.g. seven years (18), nine years (27), and 9.6 years (26), it seems that younger children can develop PSH, even with a higher prevalence than adults at least in clinic-based samples (18,25–27). A family history of primary headaches is common in pediatric patients with PSH, with migraine being the most common type (18,25,27). However, an association with migraine was not well documented in children and adolescents with PSH unlike adult patients (18,27), whereas episodic syndromes associated with migraine such as cyclic vomiting and recurrent abdominal pain were relatively frequent in pediatric patients in one study (18) and a recent case series reported six children with PSH and concomitant migraine or tension-type headache (28). Although different diagnostic criteria were applied according to the study period, studies showed clinical manifestations of PSH in adolescents and children were similar to those of adult PSH (29). The duration of stabs are mostly brief: <2 seconds in 53.3% (26,27) and <5 seconds in 70% of juvenile patients (18), while a study reported longer attacks of >3 minutes in 52% of children and adolescents with PSH (27). No accompanying symptoms were reported in two studies, in contrast to 47% (photophobia or phonophobia 15%, nausea 7%, vertigo 8%, and other nongastrointestinal symptoms 17%) in a study conducted by Soriani et al. (18).
PSH in the older adults has not yet been studied in detail. The Vågå epidemiological study included participants aged 18–65 years and showed that the age of onset of PSH peaks in the 20 s and declines thereafter (24). However, PSH seems to be a common problem in older adults, at least in clinics (2,5). In a multicenter study conducted in Korea, PSH was diagnosed in 22% of first-visit older adult patients (aged ≥65 years) presenting to headache clinics and was the second most frequent diagnosis after tension-type headache (30). Detailed comparisons of clinical characteristics between older adults and younger patients have not yet been made.
Pathophysiology
The pathophysiology of PSH is unknown and an animal model is lacking. Nevertheless, the pain characteristics, clinical courses, and drug responses may provide some clues to postulate its pathophysiology. Although the pain of PSH is similar to neuropathic pain in nature, it is extremely short-lasting, focal in location, and not provoked by external stimuli. These features suggest a spontaneous, temporary firing of the nociceptive nerve (A-delta or C fibers) endings originated from sensory afferents innervating the head: mainly trigeminal and occipital nerves (5,29,31,32). Given the localized, abrupt, and short-lasting nature of a PSH attack, A-delta fibers may be more involved than C-fibers (32). We will refer to the clinical patterns of PSH (see the clinical manifestation section) to generate several hypotheses based on clinical reasoning regarding why and how these nerve endings are spontaneously activated.
Most PSHs are of the intermittent type in the general population, with infrequent stabs and normal sensory function between stabs. This suggests a nonpathological process without axonal damage. First, the mechanism of spontaneous firing can be attributed to the hyperexcitability of the free nerve endings (Figure 2B). Second, nociceptive Schwann cells may play a role in PSH development. Nociceptive Schwann cells have recently been found in mice and later in humans, and their presence contradicts previous knowledge that nociceptive neurons have “free” nerve endings (Figure 2A) (33,34). Transient failure of the pain gating mechanism due to partial loss or dysfunction of nociceptive Schwann cells can be another, probably more feasible, mechanism of spontaneous firing of nerve endings (Figure 2B), as nociceptive Schwann cells seem to play a role in initiating and gating pain perception (33,34).
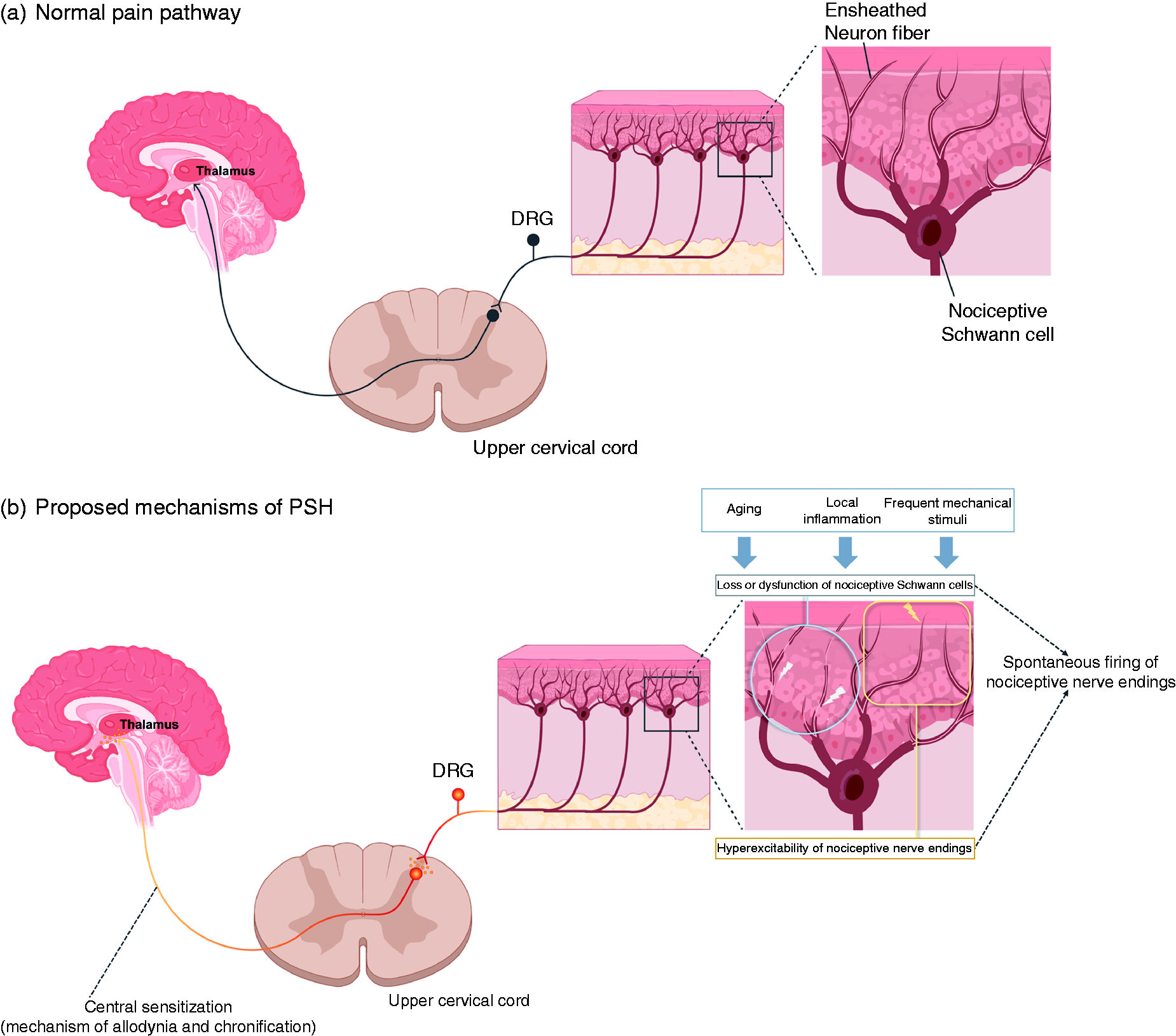
Proposed mechanisms of primary stabbing headache.
These two hypotheses, i.e. hyperexcitability of free nerve endings and loss or dysfunction of nociceptive Schwann cells, might be caused by aging, mechanical stimuli, inflammation, and central sensitization, and each of them may serve as a background for spontaneous stabs alone or in combination with others (Figure 2B). The observation that most monophasic PSHs, defined by a short (<3 months) clinical course, have side-locked and fewer migrating locations, high frequency stabs, and high response rate to corticosteroids, suggests an inflammatory process around the nerve endings (17). Central sensitization secondary to peripheral mechanisms can be another proposed mechanism of intermittent and chronic daily PSH, which may explain its long-term course, migrating location, allodynia, and bodily jabs (17). The high prevalence of PSH in people with migraine further supports this hypothesis.
Apart from the nerve ending hyperexcitability and nociceptive Schwann cell hypotheses, findings of secondary stabbing headache caused by brain disorders, although rare, provide another perspective of the origin of pain in PSH. In the relapsing phase of multiple sclerosis, stabbing headache has been reported and is thought to be caused by the inflammation of the subpial gray matter and meninges (35). Contralateral stabbing headaches have also been reported in hemorrhagic or ischemic strokes, probably caused by the disinhibition of the pain modulation pathway (10). Furthermore, stabbing headaches have been reported in cervical myelitis involving the spinal nucleus of the trigeminal nerve (36). Taking these findings together, although rare, a focal central lesion causing irritation of the nociceptive pathway or disinhibition of central pain modulation pathway may allow spontaneous synchronous discharges of peripheral nociceptive neurons and lead to stabbing pain in an extremely focal area (21,37).
Diagnosis
Diagnostic criteria
In 1988, the ICHD-1 suggested the diagnostic criteria for ISH (38). ISH was defined as a single or series of stabs confined to the first division of the trigeminal nerve (orbital, temporal, and parietal area) for a few seconds with irregular intervals (hours to days). Structural changes at the site of pain and in the distribution of the affected cranial nerve should be excluded. In ICHD-2, published in 2004 and revised in 2005, ISH was renamed PSH (39,40). The diagnostic criteria for the nature and location of pain of PSH in ICHD-2 were the same as ISH in ICHD-1; however, ‘no accompanying symptoms’ was emphasized to differentiate it from other primary headache diseases such as trigeminal autonomic cephalalgias (TACs).
The ICHD-3β was published in 2013, followed by ICHD-3 in 2018 which is currently the most updated version (3,41). The ICHD-3 diagnostic criteria for PSH (Table 2) had two major changes compared with that of ICHD-2. First, the pain location of PSH was restricted to the first branch of the trigeminal nerve in ICHD-2, and it was expanded to ‘head’ in ICHD-3. Previous studies have reported that most patients with PSH had pain in the extratrigeminal region (5,21,42). Second, the ICHD-3 indicated that no cranial autonomic symptoms were accompanied by stabbing headaches. In ICHD-2, no accompanying symptoms were allowed for the diagnosis of PSH. However, subsequent studies have revealed that symptoms such as nausea, vomiting, photophobia, phonophobia, dizziness, ictal allodynia, and vocalization are not uncommon in patients with PSH (5,14,16,18). While incorporating these features into a spectrum of PSHs, the lack of cranial autonomic symptoms can be helpful in differentiating PSH from short-lasting unilateral neuralgiform headache attacks with autonomic symptoms/short-lasting unilateral neuralgiform headache attacks with conjunctival injections and tearing (SUNA/SUNCT) and maintain the sensitivity of the diagnosis of PSH. In addition, responsiveness to indomethacin was described in the ‘comments’ of ICHD-1 and ICHD-2 (38–40); however, it was removed from ICHD-3β as most cases are self-limiting and other agents can also help (41).
Diagnostic criteria of primary stabbing headache in the ICHD-3.

1Studies show 80% of stabs last three seconds or less; rarely, stabs last for 10–120 seconds.
2Attack frequency is generally low, with one or a few per day. In rare cases, stabs occur repetitively over days, and there has been one description of status lasting one week.
Researchers should be careful when interpreting previous studies on PSH or ISH because of the modifications in the diagnostic criteria for PSH. Theoretically, SUNA/SUNCT might have been falsely classified as ISH in studies performed prior to ICHD-2. Furthermore, PSH with accompanying symptoms or pain outside the first branch of the trigeminal nerve could have been excluded in studies using ICHD-2. One field-testing test using ICHD-3β showed that only 9.3% of patients diagnosed with PSH based on ICHD-3β fulfilled the ICHD-2 criteria for PSH (16).
Differential diagnosis
The major differential diagnoses of PSH are epicranial headaches (headaches originating from the outside of the skull) or cranial neuralgias: occipital neuralgia, nummular headache, and cervicogenic headache. Although they have distinct characteristics, patients may have overlapping features. Cervicogenic headaches can be more long-lasting, have mixed (i.e., nociceptive and neuropathic) pain types, and worsen with neck movement. In addition, excessive forward head position, reduced cervical range of motion and myofascial trigger points can be accompanied, and pain can be precipitated by external pressure over the upper cervical or occipital area on the symptomatic side in patients with cervicogenic headache (43). The pain of occipital neuralgia is distributed within the dermatome of the occipital nerve branch, while the pain of PSH is more focal. Occipital neuralgia is often accompanied by dysesthesia, allodynia, and tenderness in the affected nerve branch (3), whereas interictal PSH has no symptoms or signs. A diagnostic nerve block can help the differential diagnosis between occipital neuralgia and PSH. Nummular headache is located in a round or elliptical area and is one to six centimeters in diameter with a sharp margin. Nummular headache presents with persistent pain more frequently; however, it can also present with transient stabs, which resemble PSH, according to the ICHD-3 definition (3). A clearly demarcated pain area is a hallmark of nummular headache, and if present, focal tenderness or sensory abnormalities within the pain area are highly suggestive of nummular headache, differentiating it from PSH (44).
Other differential diagnoses include trigeminal neuralgia and TACs. Trigeminal neuralgia is characterized by pain with severe intensity lasting less than two minutes precipitated by innocuous stimuli within the affected trigeminal distribution (3). Precipitation by innocuous stimuli followed by a refractory period with or without concomitant continuous pain can be a differential point that favors trigeminal neuralgia and excludes PSH. Trigeminal neuralgia is usually severe or extremely severe, while PSH is not uniformly severe; it can be mild or moderate, at least in the general population (7,45). In TACs, pain is more widely distributed, located more within the V1 territory, and longer lasting than in PSH (46). Cranial autonomic symptoms ipsilateral to the headaches are characteristic (3). The duration of pain is 15–180 minutes for cluster headache, 2–30 minutes for paroxysmal hemicrania, and 1–600 seconds for SUNA/SUNCT (3), which are mostly longer than that in PSH, which is of a few seconds (typically 3 s or less but reportedly up to 10–600 s in the ICHD-3 [3]). However, it can overlap with PSH in some cases with SUNA/SUNCT.
Although rarely, stabbing headache can also occur secondarily to structural intra- or extracranial diseases resulting in focal irritation or damage of the nociceptive or pain modulation pathways, such as giant cell arteritis (47), pituitary tumors (48), meningiomas (49), ocular trauma (2), ischemic stroke (50), cavernous hemangioma of the frontal bone (51), herpetic zoster infection (52,53), Bell’s palsy (54), idiopathic intracranial hypertension (55), intracranial hemorrhage (9,10), and multiple sclerosis (56). Stabbing headaches have also been reported in systemic autoimmune disorders including Sjögren’s disease, systemic lupus erythematosus, Behçet's disease, autoimmune vasculitis, and antiphospholipid antibody syndrome (57). It is uncertain whether these disorders have caused or predispose stabbing headaches and whether stabbing headaches secondary to structural or systemic disorders have characteristics distinct from PSH. Nevertheless, awareness of these potentially life-threatening disorders can be helpful when patients with suspected PSH show atypical features or a progressive course.
Suggested diagnostic flow and work up
We proposed an algorithm for the diagnostic flow of stabbing headaches, as shown in Figure 3. If headache has typical characteristics of a PSH, that is, migrating location and less than 3 s duration without background pain or sensory loss, clinicians can monitor it with/without medical treatment without further investigation. However, if the pain is in a fixed location, other primary headaches and secondary causes should be considered. If cranial autonomic symptoms occur with fixed pain, TACs such as SUNA/SUNCT should be differentiated. Background pain between stabbing or sensory abnormalities in the same area of pain may suggest neuropathies provoked by peripheral or central causes. Pain precipitated by innocuous stimuli, such as touching the face, chewing, speaking, or brushing teeth, is suggestive of trigeminal neuralgia. Occipital neuralgia should be considered if tenderness is present in the occipital nerve distribution or if improvement is shown after occipital nerve block; although, tenderness of the occipital nerve distribution is possible in PSH. Patients with atypical presentation, such as longer duration, substantial background pain, or other red flag signs or uncommon (i.e. chronic daily or progressive) courses require further investigation (Figure 3) (58).
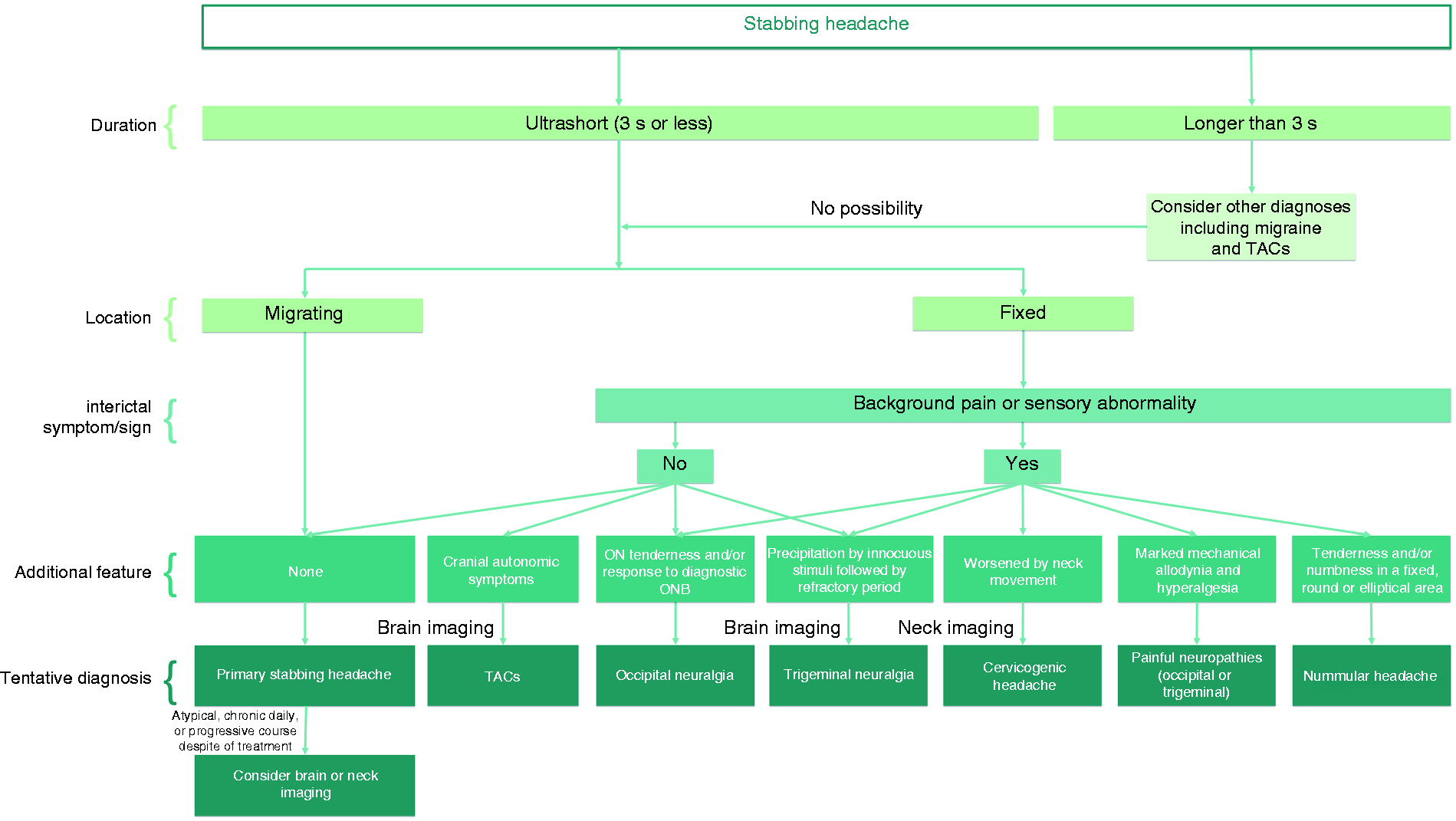
Suggested algorithm for the differential diagnosis of primary stabbing headache.
Plain radiography, computed tomography, or magnetic resonance imaging of the brain or cervical spine can be used to exclude structural lesions or cervical spine abnormalities (10,47,49) in patients with atypical features or courses. Such radiological investigations show normal structures in pediatric and adult patients with PSH (56). If stabbing headache occurs with a substantial background pain in patients aged >50 years, temporal artery tenderness, jaw claudication, and erythrocyte sedimentation rate can be helpful in the differential diagnosis of giant cell arteritis. In two pediatric studies, abnormal findings in the electroencephalogram (EEG) were found in 26–30% of patients with PSH (59,60); however, the diagnostic value of EEG is unclear and the use of EEG for the diagnosis or differential diagnosis of PSH is generally unnecessary.
Treatment
In both general population and patients seeking medical advice, most PSHs are self-limiting and do not require medical treatment (17,61). However, treatment can be initiated according to the patients’ need from headache impact, disability, or fear of symptoms. In general, patients with high-frequency or chronic attacks (i.e. recent onset but innumerable attacks or chronic daily occurrence with variable frequency) are potential candidates for medical treatment.
PSH has been recognized as one of indomethacin-responsive headaches (61,62). However, several other agents have also shown similar efficacy in the treatment of PSH. These include non-steroidal anti-inflammatory drugs (NSAIDs) (naproxen and ibuprofen) (16), selective cyclooxygenase-2 inhibitors (rofecoxib, celecoxib, and etoricoxib) (5,50,63), prednisolone (17), gabapentin (16,17), tricyclic antidepressants (amitriptyline or nortriptyline) (16,17,64), carbamazepine (26), and melatonin (65).
Indomethacin has been used to treat PSH at reported doses of 50, 75, and 150 mg daily (2,5,66). Its efficacy is not absolute, yielding a 30–50% success rate of complete remission in most case series (2,5,17). Although some patients may respond dramatically to indomethacin, its unfavorable safety profile limits the use of indomethacin as a first-line therapy. The side effects of indomethacin include gastrointestinal disturbances, acute renal failure, and hypertension, which limit its long-term use. Concomitant medications such as aspirin and anticoagulants may further limit indomethacin use.
The choice of medication should be based on the symptom severity and chronicity. We propose a pattern-based treatment algorithm as follows. For recent onset, high-frequency PSH, anti-inflammatory agents such as naproxen, ibuprofen, indomethacin, and steroids can be used according to symptom severity. Intermittent PSH does not usually require medical treatment. Rapid-acting NSAIDs, such as ibuprofen or indomethacin, can be used as needed when a series of stabs recur. For chronic PSH, the treatment can be long-term. Thus, treatment should be administered only when the benefits clearly outweigh the risks of long-term medication use. Medications with proven safety for long-term use, such as gabapentin, amitriptyline, and melatonin, can be considered in such cases. The efficacy of botulinum toxin has been demonstrated in an open-label study (67). It can be considered for chronic PSH with fixed or nearly fixed locations when medications fail in terms of efficacy or tolerability, although, the use of botulinum toxin for headaches, other than chronic migraine, is not supported by high-quality evidence and currently off-label in most countries (68).
Less information is available regarding treatment of PSH in children and adolescents with PSH. Although the number of reported cases is small, the general treatment strategy should be similar with that of adult patients. The use of pharmacological prophylaxis is relatively uncommon in children and adolescents with PSH, probably due to the potential adverse effects of drugs and benign nature of the disease. Drugs reported as effective are indomethacin (26,69), carbamazepine (26), amitriptyline (64), L-5 hydroxytryptophan plus riboflavin (26), and paracetamol (27).
Prognosis
There have been no studies specifically regarding the natural history of PSH. Results from epidemiological studies, clinical investigations, and a study evaluating clinical patterns and courses showed that most cases are benign and self-limiting without treatment (2,5,11,12,17). In a clinical, prospective follow-up study of PSH, all patients with recent-onset PSH eventually remitted; remission was spontaneous in half of the patients and occurred after medical treatment in the other half, and therefore this pattern was named ‘monophasic’ (17). Recurrence of monophasic PSH has also been previously reported. In contrast, the majority of patients with chronic PSH did not respond to treatment and continued to have chronic daily stabs (17). The results of this study suggest that a chronic refractory form may exist. The intermittent subtype can relapse at variable frequencies over several years. It can progress to the chronic daily subtype, although this has been reported in a very small number of patients (17). However, the exact rate of progression is yet unknown.
Conclusion
Stabbing headache is common in general population, and PSH is a frequent cause of headache clinic visit. Since PSH was first introduced as a disease entity, there have been numerous revisions in the diagnostic criteria of this disorder. Although PSH has various manifestations, knowledge about the typical manifestations and clinical patterns will aid in the proper diagnosis and differential diagnosis. Treatment should be tailored by considering the clinical patterns. Further research is needed to elucidate the pathophysiological mechanisms and optimal treatment of PSH.
Clinical implications
While primary stabbing headache is a common cause of headache clinic visit, its abrupt mode of onset can negatively affect both patients and physicians: patients may be overly concerned about their symptoms, and physicians may misinterpret the pain as thunderclap headache and prescribe unnecessary imaging studies. Typical features of primary stabbing headache are ultrashort-lasting paroxysms of focal stabbing pain occurring scalp or face, frequently migrating in location, and the absence of background pain or sensory abnormalities. Cases with longer duration, fixed location, and background pain or sensory abnormality warrant the differential diagnoses such as occipital neuralgia, nummular headache, trigeminal neuralgia, painful neuropathies involving occipital or trigeminal nerves, cervicogenic headache, and trigeminal autonomic cephalalgias, and secondary causes should be investigated when a patient shows atypical features or courses. Most cases are self-limiting, while medications can be initiated based on symptom severity and chronicity.
Footnotes
Acknowledgement
This study was supported by National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIP; No. 2020R1A2B5B01001826 to MJL).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
