Abstract
Objectives
To compare the clinical profiles between male and female cluster headache patients from a large cohort.
Methods
This hospital-based study enrolled patients diagnosed with cluster headache between 1997 to 2021. Participants completed structured questionnaires collecting information on demographics, clinical profiles, and quality of life. Treatment regimens and effectiveness were determined through medical chart review. All variables were compared between the sexes.
Results
In total, 798 patients (M/F:659/139) were enrolled. The male-to-female ratio was 4.7:1 for the full study period, but it declined from 5.2:1 to 4.3:1 for patients enrolled before and after 2010, respectively. The frequencies of chronic cluster headache (M:1.2%, F:1.4%) and aura (M:0.3%, F:0.7%) were low but similar between the sexes. Most headache features showed no difference between men and women. Female patients had significantly longer attack duration, shorter inter-bout duration, higher frequencies for eyelid edema, nausea and vomiting and lower frequencies for conjunctival injection and pacing. Sex difference did not influence headache-associated disability, anxiety, or depression, but poor sleep quality was significantly more common in women. Among menstruating women, 22/122 (18.0%) reported worsening headaches during menses. The effectiveness of treatment was similar between the sexes.
Conclusions
Despite a decline of male-to-female ratio in the past two decades, most clinical profiles were similar between the sexes.
Keywords
Introduction
Cluster headache (CH) is a primary headache disorder with a lifetime prevalence of 0.1% worldwide (1). The diagnostic criteria are based on the International Classification of Headache Disorders, Third Edition (ICHD-3). CH can be categorized as episodic or chronic depending on the remission period between bouts (2). Although the mechanism of CH has not been completely established, it is widely accepted that the hypothalamus and sphenopalatine ganglion participate in the pathogenesis because both circannual and circadian rhythmicity and autonomic features are prominent among patients with CH (3,4).
Sex hormones have been associated with primary headache disorders (5,6). In contrast to migraine, which is a predominantly female headache disorder, CH has a high male-to-female ratio that is especially prominent among Asians (7). However, the presumption that CH is a predominantly male disorder may lead to under-recognition of the condition among women, thus leading to misdiagnoses. Several studies have been conducted to evaluate the headache profiles between the sexes. Although findings such as a younger age at onset and a higher frequency of migrainous features in women have been consistently reported, findings concerning other clinical profiles have been conflicting or inconclusive across studies (8–16). Moreover, the numbers of female patients enrolled in Asian studies have been relatively small, ranging from 14 to 25 participants (8–12), whereas European and US studies have enrolled patient populations ranging from 32 to 318 (13–16). Hence, a gap remains in the understanding of the clinical features of CH among Asian women.
To better understand how CH differs between the sexes, we enrolled a large cohort at a headache center in Taiwan. This study aimed to compare the clinical profiles of CH and treatment effectiveness between the sexes.
Methods
This hospital-based, prospective registry study was conducted at the headache clinic at Taipei Veterans General Hospital (TPE-VGH), a tertiary medical center in northern Taiwan, from 1997 to 2021. Under Taiwan’s National Health Insurance, both veterans and civilians can visit the hospital without referral (17). Patients were recruited if they were diagnosed as having either episodic CH (ECH) or chronic CH (CCH) on the basis of the diagnostic criteria of the ICHD-3 (2). CCH was diagnosed if headaches occurred for 1 year or longer without remission or with a remission period of less than 3 months (2). All participants were asked to complete structured questionnaires, as described in our previous study (12) (see supplement material). In brief, the questionnaires consisted of three parts. The first part solicited demographic data including sex, height, weight, body mass index (BMI), age of onset, age at diagnosis, duration of diagnosis delay, personal history of migraine, and family history of CH or migraine. Year 2010 was selected to compare the trend of male-to-female ratio in the first-half and the second-half study periods. The body indices including height, weight and BMI of both male and female patients were compared with the general Taiwanese population between 18 to 65 years-old, which is similar to the age of our patients (18). Data regarding smoking, alcohol consumption, and a history of head injury were also collected. In this study, we defined “alcohol drinker” as someone who had ever drank, which is in line with the definition of the Health Promotion Administration of Taiwan (19). Only “current smoker” in our study fulfilled the definition of “smokers” from the Health Promotion Administration of Taiwan (20). Patients who reported a head injury resulting in loss of consciousness or requiring hospitalization were categorized as having a positive history of head injury. We also compared the prevalence of these risk factors in patients with CH with the general Taiwanese population from 2008 to 2009, which was the median recruitment year of our patients.
The second part of the questionnaire inquired into clinical features, including headache severity (scored 0 to 10/10 on a numeric rating scale), mean attack frequency and duration, mean bout frequency and duration, mean interbout duration, headache quality, laterality, locations, and periodicity. ICHD-3-defined cranial autonomic symptoms (CAS), restlessness, agitation, pacing during attacks, as well as migrainous features including nausea, vomiting, photophobia, phonophobia, and aura, were also included in this section.
The third part of the questionnaire was aimed at evaluating quality of life. The third part of the questionnaire comprised the hospital anxiety and depression scale (HADS) (21), migraine disability assessment (MIDAS) (22), Pittsburgh sleep quality index (PSQI) (23), a suicide questionnaire, and headache impact test-6 (HIT-6) (24). A severe disability was defined as MIDAS ≥21 or HIT-6 score ≥60. For menstruating females, an additional section inquired into changes in CH quality during perimenstrual periods.
We performed a chart review of all medical records and compared the prescribed medications and their effectiveness between the sexes. The utility rate was defined as the number of patients prescribed a medication divided by the number of patients with available medical records. Effectiveness was defined as the ratio of the number of medical records indicating “effective” among all patients prescribed the medication. The highest prescribed doses of the medications were also analyzed.
To compare the differences between sexes, we used a Student’s t test to compare the means for continuous data and a Chi-square test to assess the frequencies for categorical data. The results were analyzed with R (R Core Team, 2019), and p < 0.05 was set as the threshold for statistical significance.
This study was approved by the Institutional Review Board of TPE-VGH (TPEVGH IRB No.: 2022-06-007CC). All the participants who filled out the questionnaires consented to participate in this study.
Results
Demographics
A total of 798 patients with CH (659 men, 139 women) who visited our headache clinic at any time between 1997 and 2021 were enrolled in this study. The male-to-female ratio from the whole study period was 4.7:1 (Table 1). However, this ratio declined from 5.2:1 for patients enrolled between 1997 to 2010 to 4.3:1 for those enrolled between 2011 to 2021 (p = 0.355). The average height (male: 173.2 ± 5.8 cm, female: 161.6 ± 5.7 cm, p < 0.001), weight (male: 73.0 ± 11.2 kgs, female: 56.7 ± 8.3 kgs, p < 0.001) and BMI (male: 24.3 ± 3.5, female: 21.7 ± 3.1, p < 0.001) were significantly higher in males. When compared with the age-similar general population, both male (general population: 169.3 cm, p < 0.001) and female (general population: 157.4 cm, p < 0.001) patients were significantly taller. The body weight, when compared with the general population, was significantly heavier in male patients (general population: 71.7 kgs, p < 0.001) but significantly lighter in female patients (general population: 58.3 kgs, p = 0.026). Moreover, both male (general population: 25.0, p < 0.001) and female (general population: 23.5, p < 0.001) patients had significantly lower BMIs than the general population. CCH was identified in two (1.4%) women and eight (1.2%) men (p = 1.000). The average age of onset was lower in women than in men (male: 26.7 ± 10.0 [range 10 to 63], female: 23.6 ± 9.3 [range 10 to 55], p < 0.001), whereas the average age at diagnosis was similar for both sexes. The duration from disease onset to diagnosis was similar for both sexes (male: 10.4 ± 8.0 years, female: 12.1 ± 10.2 years, p = 0.059).
Demographic data, personal and family history and headache profiles.
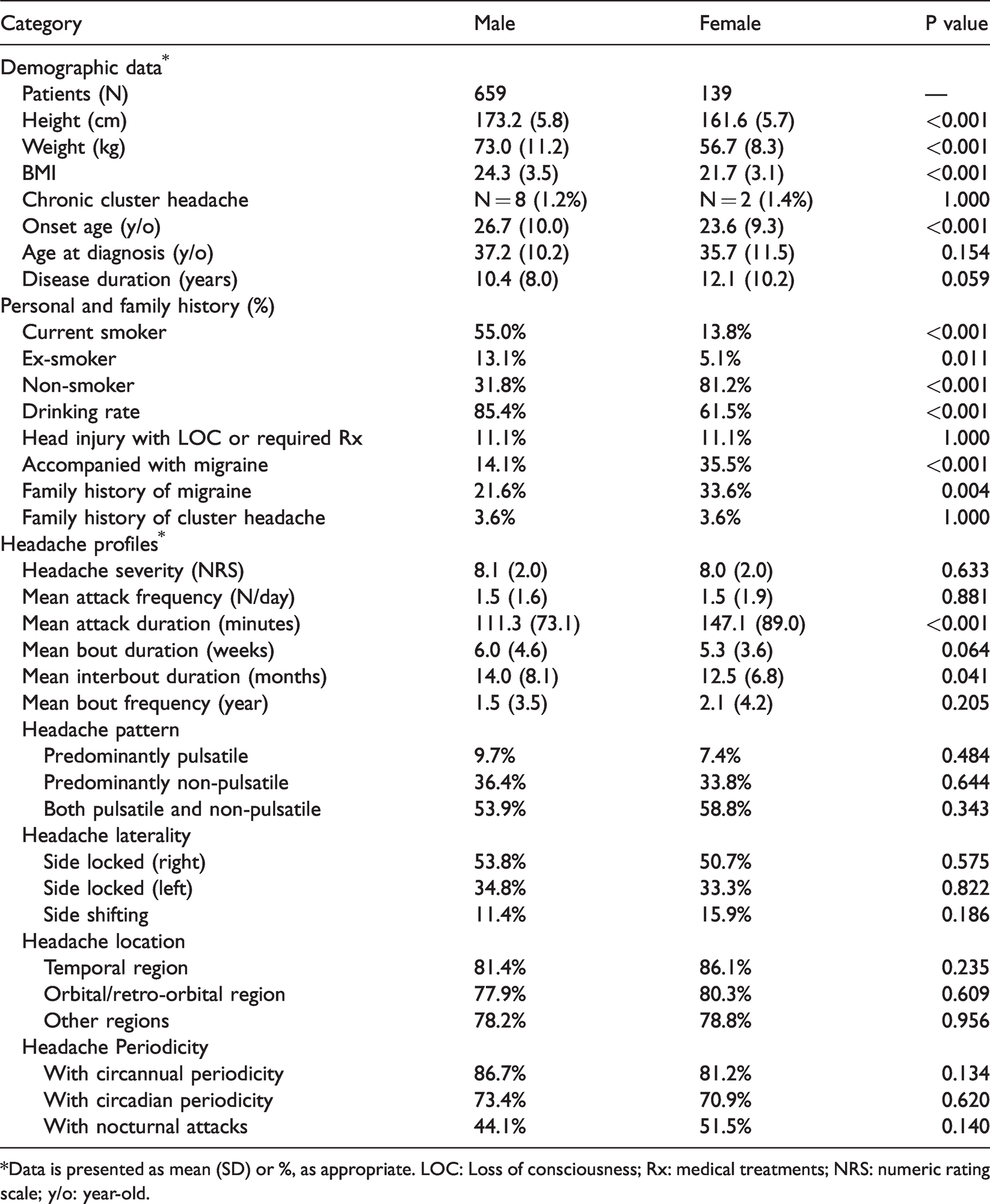
*Data is presented as mean (SD) or %, as appropriate. LOC: Loss of consciousness; Rx: medical treatments; NRS: numeric rating scale; y/o: year-old.
The smoking rate was significantly higher in men (current smoker: male: 55.0%, female: 13.8%, p < 0.001; ex-smoker: male: 13.1%; female: 5.1%, p = 0.011). When compared with the general population in Taiwan (20), the prevalence of current smoker among CH patients was 1.4-fold higher in men and 2.9-fold higher in women. The rate of alcohol consumption was also significantly higher in men (male: 85.4%, female: 61.5%, p < 0.001). The drinking rate of CH patients was 1.4-fold higher in men and 1.9-fold higher in women when compared with the general population (19). Both sexes had similar rates of previous head injury (male: 11.1%, female: 11.1%, p = 1.000). However, the prevalence of head injury among CH patients was 13.1-fold higher in men and 34.6-fold higher in women when compared with the general population (25,26).
Women were more likely to have coexistent migraine than men (male: 14.1%, female: 35.5%, p < 0.001). Both sexes had similar rates of family history for CH (male: 3.6%, female: 3.6%, p = 1.000), but more women than men had a positive family history of migraine (male: 21.6%, female: 33.6%, p = 0.004).
Headache profiles
There were no significant differences in headache pain severity (on a 0 to 10 scale; male: 8.1 ± 2.0, female: 8.0 ± 2.0, p = 0.633) and mean attack frequency per day (male: 1.5 ± 1.6, female 1.5 ± 1.9, p = 0.881) between sexes. (Table 1) However, the attack duration was significantly longer in female patients than in male patients (male: 111.3 ± 73.1 minutes, female: 147.1 ± 89.0 minutes, p < 0.001). Mean bout duration was similar for the sexes. Nevertheless, mean interbout duration was significantly longer in male patients than in female patients (male: 14.0 ± 8.1 months, female: 12.5 ± 6.8 months, p = 0.041).
Headache pattern did not differ between the sexes nor did headache laterality, and both sexes had higher frequency of right-side-predominant CH (Table 1). Headache locations were similar between the sexes, with the temporal region being the most frequent area followed by the orbital/retro-orbital regions.
There were no differences in the proportions of patients with circadian or circannual periodicity and nocturnal attacks between the sexes (Table 1).
Cranial autonomic symptoms
Men had an average of 2.3 ± 1.2 ICHD-3 autonomic features, and women had 2.2 ± 1.2. (p = 0.721; Table 2) In both sexes, the most common CAS was lacrimation, with similar frequencies (male: 79.9%, female: 83.9%, p = 0.338). Conjunctival injection was significantly less common in women than in men (male: 54.4%, female: 36.5%, p < 0.001). Female patients presented significantly more eyelid edema (male: 17.3%, female: 25.5%, p = 0.034). Other CASs showed similar results between the sexes.
Cranial autonomic symptoms and accompanying symptoms.

*Data is presented as mean (SD) or %, as appropriate. CAS: Cranial autonomic symptoms.
Accompanying symptoms
Aura was identified in two men and one woman, none of whom had coexistent migraine (Table 2). Female patients had significantly higher percentages of nausea (male: 49.8%, female: 77.4%, p < 0.001) and vomiting (male: 31.2%, female 49.3%, p < 0.001) than did male patients. The proportion of patients reporting photophobia, unilateral photophobia, or phonophobia did not differ between the sexes.
Other accompanying symptoms, including restlessness (male: 47.4%, female: 39.7%, p = 0.121), agitation (male: 41.5%, female: 54.8%, p = 0.177), and headaches aggravated by physical activity (male: 65.6%, female: 73.5%, p = 0.091), did not differ between the sexes. Men tended to pace more than women (male: 41.6%, female: 19.0%, p < 0.001).
Menstruation
Among menstruating patients (N = 122), 18.0% (22/122) reported worsening of headaches during menstrual cycles, and 40.9% (9/22) of them had coexisted migraine. Besides, headache worsening during menstrual cycles was also more commonly seen in patients with coexisting migraine, though there was no statistical significance (with migraine: 24.3%, without migraine: 13.6%, p = 0.285). The most common timing of worsened headaches was 1 to 2 days prior to menstruation (14/22, 63.6%), followed by the first day of the menstrual cycle (10/22, 45.5%).
Psychological disturbance, disability, and sleep
Male and female patients exhibited no significant difference in total HADS (male: 14.7 ± 6.5 [N = 579], female: 14.8 ± 5.8 [N = 121], p = 0.851), MIDAS (male: 24.0 ± 32.9 [N = 525], female: 29.1 ± 35.4 [N = 106], p = 0.175), or HIT-6 score (male: 64.3 ± 7.6 [N = 165], female 66.9 ± 7.5 [N = 39], p = 0.056) (Table 3). The proportion of patients reporting suicidal ideation (male: 16.0% [N = 34/213], female: 27.1% [N = 13/48], p = 0.109) or an attempt at suicide (male: 3.8% [N = 8/212], female: 2.0% [N = 1/49], p = 0.869) was similar in both sexes. Women had a significantly higher PSQI score (male: 8.0 ± 4.0 [N = 402], female: 9.7 ± 4.2 [N = 84], p = 0.001) and a lower sleep efficacy (male: 83.8 ± 14.7% [N = 402], female: 78.3 ± 16.9% [N = 84], p = 0.006) than men did.
Quality of life, disability and psychological disturbance.

*Data is presented as mean (SD) or %, as appropriate. HADS-A: Hospital anxiety and depression scale-anxiety; HADS-D: hospital anxiety and depression scale-depression; MIDAS: migraine disability assessment test; HIT-6: headache impact test-6; PSQI: the pittsburgh sleep quality index.
Treatment regimens and effectiveness
In our cohort, data on treatment regimens were available for 571 (86.6%) men and 127 (91.4%) women (Table 4). Among them, the most commonly prescribed acute medication was sumatriptan tablets for both sexes (male: 49.9%, female: 53.2%, p = 0.572), and oxygen therapy was the least commonly used (male: 10.5%, female: 11.1%, p = 0.969). The utility rates and treatment effectiveness in abortive treatments were similar between the sexes.
The utility rate and effectiveness of the treatments.
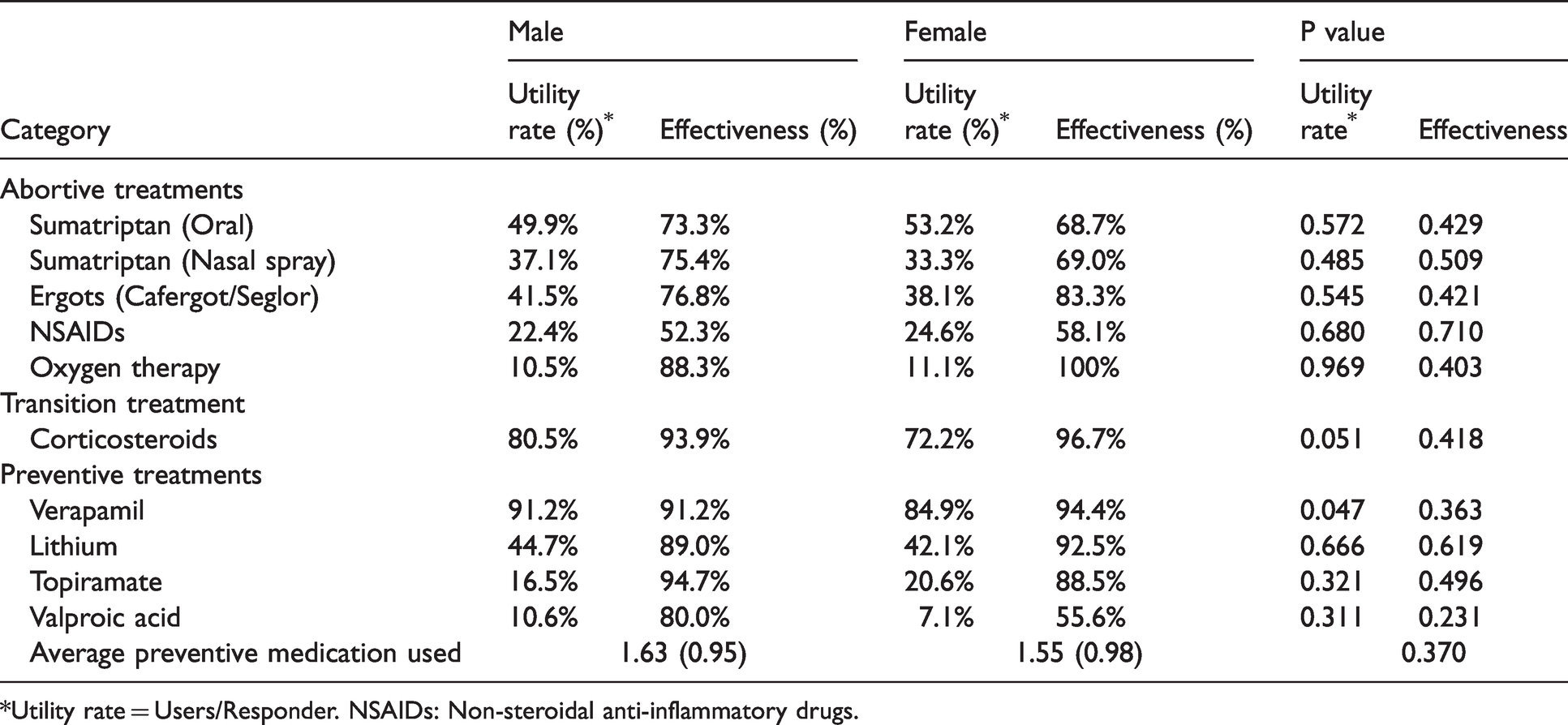
*Utility rate = Users/Responder. NSAIDs: Non-steroidal anti-inflammatory drugs.
The proportions of patients prescribed oral prednisolone as a transitional therapy (male: 80.5%, female: 72.2%, p = 0.051) as well as its effectiveness (male: 93.9%, female 96.7%, p = 0.418) were similar between sexes. The average dosages of prednisolone per kg of body weight were similar between the sexes (male: 0.88 mg/kg, female: 0.87 mg/kg, p = 0.803).
Verapamil was the most commonly used preventive medication, and male patients had a significantly higher prescription rate than did female patients (male: 91.2%, female: 84.9%, p = 0.047), though treatment efficacy did not differ between the sexes. Other preventive medications such as lithium, valproic acid, and topiramate had similar utility rates and effectiveness. On average, the number of types of preventive medications did not differ between the sexes (male: 1.63 ± 0.95, female: 1.55 ± 0.98, p = 0.370).
Discussion
The current study has several advantages enabling it to improve our understanding of Asian female patients with CH. First, the study population represents the largest Asian female cohort to be studied regarding CH (8–12). Second, we used a unified questionnaire spanning two decades, as discussed in our 2004 article, which provided reliable consistency (12). We compared our results with others, and Table 5 summarizes the results of studies recruiting more than 80 female patients, including studies from the United States (N = 318), Italy (N = 82), and Denmark (N = 116) (15,16,27).
The comparison of clinical features with other studies enrolling >80 females.

*In the original article, the description of the respective data was “need to move” for restlessness, “irritability” for agitation, and “feeling uneasy” for pacing.
✧Ptosis and/or miosis. CCH: Chronic cluster headache; a/w: accompanied with; FHx: family history; NA: not available.
In this study, we noticed a declined male-to-female ratio for patients diagnosed after 2010, when compared to those diagnosed before, though no statistical significance between the two periods. This trend has been observed in other studies, and the increases in smoking and employment rates among women have been proposed as plausible factors (8,28–30). However, the male-to-female ratio of current smokers increased from 14.9:1 to 21.6:1 for patients diagnosed before and after 2010, respectively, which makes smoking less likely to be the factor affecting the sex distribution among our patients.
Among our patients, men were significantly taller, heavier and with higher BMI than women. When compared with the general population, though heavier and taller, male patients had a lower BMI. In female patients, both significantly lower weight and taller height led to the lower BMI when compared with the general population. In a Danish study, though both male and female CH patients shared similar BMI, both sexes had higher mean BMI when compared to control subjects, which differed from our results (31). Some studies have linked an unhealthy lifestyle to the development of CH, but further studies are required to elucidate the association due to conflicting results (31,32).
CCH was diagnosed in 10 patients (1.3%) in this study. Whether the frequency of CCH is similar between the sexes has remained inconclusive because some studies have reported affirmative data as ours (8,10,13) while others have not (11,15,33). Still, this study confirmed a lower frequency of CCH in Taiwanese patients, as demonstrated in our previous report (12), and other East Asian populations ranging from 0% to 7.5% (7,10,11,34–36), than the rates between 10.2% and 36.8% seen in Western populations (7,12,31,37).
The average onset age for women was 3.2 years earlier than that of men, which is consistent with the results from US and Italian studies (16,27) but contrary to the results of several other studies (8–11,15,34). In our cohort, there was no significant difference in diagnostic delay between the sexes, although both sexes had delays of more than 10 years. Two studies utilizing regression models have indicated that coexisting migrainous features were the actual factors contributing to delayed diagnoses rather than sex (38,39).
Smoking, alcohol consumption, and head injury are proposed risk factors for CH (3,40). For a predominantly male disease like CH, it is not unexpected to see consistently higher smoking rates among male patients across studies, including ours (8,9,12,16,27,41) (Table 5). Rates of alcohol consumption and head injury have rarely been compared between the sexes. Nevertheless, three studies have shown a male predominance in alcohol consumption (8,16,41), and one study reported a similar prevalence of head injury between the sexes (27). These four studies are consistent with our own findings. Notably, although men showed a higher prevalence of smoking and alcohol consumption in absolute terms, the prevalence ratio, as standardized to the general population, was higher in women. This finding was also reported in Danish (male: 5.0-fold, female: 6.5-fold) and Korean studies (male: 1.2-fold, female: 4.3-fold) (9,31). These results may indicate that the disease-causing effects of these potential exposures are mediated by sex.
Clinically, our female patients differed from our male patients in having a longer attack duration and a shorter interbout duration; however, other headache profiles exhibited no differences between the sexes. The majority of studies have demonstrated no sex difference in attack duration (8,10,13–15,42), although an Italian study reached an opposite result to ours (27). Similar interbout intervals between sexes were reported in two studies (8,16) (Table 5). One of the cardinal symptoms of CH was CAS. The lower frequency of conjunctival injection among women was reported in two prior studies (12,43) in addition to our own, whereas other studies have reported similar frequencies between the sexes (8,10,15). Moreover, our finding that women had a higher frequency of eyelid edema was also reported in a Danish study (15). Pacing during attacks, as opposed to the activity-exacerbating headache seen in migraine, is also characteristic of CH. Women demonstrated a lower frequency of pacing in both the current and a US study (16). Our study is consistent with previous studies that women presented more migrainous features, especially nausea and vomiting (13,16,27), when compared with men. However, the differences between the sexes were not significant for photophobia or phonophobia (9,10,12–15). As mentioned in the previous section, the presence of migrainous features is an independent factor for a delayed diagnosis (38,39).
In our cohort, two men and one woman were identified as having aura. In Western studies, the frequency of visual aura ranged from 13% to 22% in men and 18% to 19% in women (14,16). Although no sex difference was reported in the Western studies, these results reflect the scarcity of aura in Asian patients with CH (7,14,16).
Among our menstruating patients, 18.0% reported headache worsening during menses; other studies have reported frequencies between 1.2% and 27% (14,16,27,44). However, these rates were much lower than the frequency of menstrual migraine, which has been reported to be 22% to 79% (45,46). Nevertheless, the frequency of headache worsening during menses was higher in females with coexisting migraine, while that in females with pure CH was lower. This disparity might reflect the notion that testosterone rather than female hormones participates more in the pathogenesis of CH (6,14,44). Less female hormone involvement in patients with pure CH may in turn result in the reduced effect of menstruation on CH.
While women exhibited more sleep disturbance in both our study and a Danish study, there were no sex differences in HADS, MIDAS, HIT-6 scores, suicidal ideation or attempts (15). Our results are consistent with those of previous studies finding no sex difference in disability, psychological disturbance, or suicidality (8,16,47). Of note, the MIDAS and HIT-6 are initially designed for migraine patients, and our study use them to evaluate disability in CH patients because there is still lack of a widely utilized disability instrument specific for CH.
Regarding treatments, women had a lower prescription rate for verapamil but a similar treatment effectiveness. Other acute and preventive medications demonstrated comparable utility rates and effectiveness between the sexes. Prior studies have mostly identified no sex difference regarding treatment utility and effectiveness; however, a higher oxygen utility rate and a better response to nasal spray among men have been reported in two separate studies (14,48,49). Despite its widely accepted effectiveness, oxygen therapy was relatively underutilized in patients of both sexes in our study when compared with the data from the UK and Germany (14,48). This finding may reflect the inaccessibility of medical oxygen devices, the inconvenience of setup, and the higher cost of oxygen use when compared with other medications that are fully reimbursed by Taiwan’s National Health Insurance (14,48). Of note, subcutaneous form sumatriptan is not available in Taiwan.
This study has several limitations. First, this study was based on a structured questionnaire; therefore, recall bias may have affected the results. In addition, though the participants provided their biological sex on the questionnaire, we did not carry out tests to check for biological sex. Second, the cluster attacks of many patients were not witnessed by medical professionals; therefore, CAS may not have been correctly reported. Third, we collected our data in a tertiary hospital in Taiwan, which may restrict the ability to generalize these findings to the overall population in our society. Nevertheless, given that patients in Taiwan could visit a tertiary center without referral, our cohort may still well represent the general population in Taiwan (17). Finally, a thorough discussion of how sex hormones affect the CH phenotype is beyond the scope of this study, and future studies are required for further investigation.
Conclusions
In this study, the clinical profiles, psychological disability, and treatment effectiveness of CH were largely similar between the sexes. Moreover, the frequency of women with CH being affected by menstruation was less than 20%. Overall, the CH phenotype did not exhibit major differences between the sexes.
Article highlights
The male-to-female ratio of CH declined from 5.2:1 to 4.3:1 for patients enrolled before and after 2010, respectively. Generally, women showed largely similar clinical presentations and treatment effectiveness to men. Women had longer attack duration and shorter inter-bout duration, more migrainous features and eyelid edema, and less lacrimation and pacing during attacks. Among menstruating women, only 18.0% reported worsened headaches during menses. Still, females with coexisting migraine were more likely to have headache worsening during menses.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024221120054 - Supplemental material for Sex-related differences in cluster headache: A hospital-based study in Taiwan
Supplemental material, sj-pdf-1-cep-10.1177_03331024221120054 for Sex-related differences in cluster headache: A hospital-based study in Taiwan by Yi-Chia Liaw, Yen-Feng Wang, Wei-Ta Chen, Shih-Pin Chen, Jr-Wei Wu, Shu-Ting Chen, Kuan-Lin Lai, Jong-Ling Fuh and Shuu-Jiun Wang in Cephalalgia
Footnotes
Acknowledgments
We would like to thank the study participants for actively participating.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: YCL, YFW, WTC, SPC, JWW, STC, KLL and JLF declare no potential conflicts of interest. SJW reports grants and personal fees from Novartis Taiwan, personal fees from Daiichi-Sankyo, grants and personal fees from Eli-Lilly, personal fees from AbbVie/Allergan, personal fees from Pfizer Taiwan, and personal fees from Biogen Taiwan, outside the submitted work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Ministry of Science and Technology of Taiwan (MOST108-2314-B-010-023-MY3 and MOST111-2321-B-A49-004 to SJ Wang, MOST110-2314-B-075-041-MY3 to YF Wang, and MOST108-2314-B-010-022-MY3 and MOST110-2326-B-A49A-501-MY3 to SP Chen), Taipei Veterans General Hospital (V111C-111 and V111E-006-1 to SJ Wang, V111C-161 to YF Wang, and V111C-158 and V109D52-001-MY3-3 to SP Chen) and Brain Research Center, National Yang-Ming University from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan (SJ Wang).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
