Abstract
Abstract
Background
Delayed-onset of headache seems a specific feature of cerebrovascular events after COVID-19 vaccines.
Methods
All consecutive events reported to the United States Vaccine Adverse Reporting System following COVID-19 vaccines (1 January to 24 June 2021), were assessed. The timing of headache onset post-vaccination in subjects with and without concomitant cerebrovascular events, including cerebral venous thrombosis, ischemic stroke, and intracranial haemorrhage was analysed. The diagnostic accuracy in predicting concurrent cerebrovascular events of the guideline- proposed threshold of three-days from vaccination to headache onset was evaluated.
Results
There were 314,610 events following 306,907,697 COVID-19 vaccine doses, including 41,700 headaches, and 178/41,700 (0.4%) cerebrovascular events. The median time between the vaccination and the headache onset was shorter in isolated headache (1 day vs. 4 (in cerebral venous thrombosis), 3 (in ischemic stroke), or 10 (in intracranial hemorrhage) days, all P < 0.001). Delayed onset of headache had an area under the curve of 0.83 (95% CI: 0.75–0.97) for cerebral venous thrombosis, 0.70 (95% CI: 0.63–76) for ischemic stroke and 0.76 (95% CI: 0.67–84) for intracranial hemorrhage, and >99% negative predictive value.
Conclusion
Headache following COVID-19 vaccination occurs within 1 day and is rarely associated with cerebrovascular events. Delayed onset of headache 3 days post-vaccination was an accurate diagnostic biomarker for the occurrence of a concomitant cerebrovascular events.
Background
Headache is one of the most frequent adverse events following COVID-19 vaccination. In the randomized controlled trials of the BNT162b2 (Pfizer®), mRNA-1273 (Moderna®) and Ad26.COV2.S (Janssen®) vaccines, headache was reported by 52%, 59% and 39% of the vaccinated individuals, respectively (1 –3). The pathophysiology of headache in vaccine recipients seems related to the systemic immune response triggered by the vaccination (4). This could be supported by the observation that in most cases, headache following COVID-19 vaccines starts within the first 72 hours (1,5). The presence of headache following COVID-19 vaccines has been associated with prior history of headache following prior influenza vaccination (odds ratio (OR): 4.8), prior history of headache during COVID-19 infection (OR: 4.3) and prior history of primary headache disorders (OR: 2.1) (6).
Cases of severe thrombotic events with thrombocytopenia have been described in individuals vaccinated with the ChAdOx1-S (AstraZeneca®) and Ad26.COV2.S vaccines (7,8), with a crude rate of thrombosis with thrombocytopenia syndrome (TTS) between 1 to 2 cases per 100,000 ChAdOx1-S doses and 0.3 to 1.1 per 100,000 Ad26.COV2.S doses (7 –11). Whereas most studies and pharmacovigilance systems have not observed an increased rate of vascular events with mRNA-based vaccines (7 –11), few studies have reported slightly increased rates of arterial thromboembolisms and cerebral venous thrombosis (CVT) following mRNA-based (BNT162b2 or mRNA-1273) vaccines (12,13).
The most frequent localization of the thromboses in TTS are the cerebral veins, but cases of ischemic stroke (IS) and intracranial haemorrhage (ICH) have also been reported (7 –11,13,14). Headache is a common symptom in cranial and cervical vascular disorders (15 –17). In the case of headache attributed to cerebral ischemic event, non-traumatic intracranial haemorrhage or CVT, the evidence of causation is based on 1) the development of the headache in close temporal relationship with other symptoms/signs pointing to a cerebrovascular event, 2) the fact that the headache led to the diagnosis of the disorder, or 3) the significant improvement of the headache in parallel with the disorder amelioration (18). Since headache may have variable phenotypes, the diagnostic suspicion of these secondary headache disorders is based on the presence of red flags (19,20).
One of the remarkable elements of TTS is the delayed onset of symptoms after the vaccination (7 –11,17,21,22). In contrast to the headache observed in other vaccine recipients, which usually starts early and resolves within a few days, headache and other symptoms of TTS usually starts around one week after the vaccination (21,22). This is due to immune-mediated production of anti-platelet factor 4 antibodies (anti-PF4), resulting in platelet activation, aggregation, consumption, and thrombosis (14,17,22,23). The delayed onset would be explained by the needed time to produce the anti-PF4 antibodies (23). For this reason, most clinical guidelines establish three days as temporal cut-off for classifying patients as possible TTS (21).
In the present study, we aimed to validate the hypothesis that the headache in individuals who experience a cerebrovascular event following a COVID-19 vaccination has a delayed onset beyond three days. For this aim, we evaluated the frequency of CVT, IS, and ICH in vaccine recipients in the United States of America (USA) and compared the interval between the vaccination and the headache onset in individuals with and without a cerebrovascular event. We assessed how accurate the 3-days cut-off proposed by the TTS guidelines (21) is in the discrimination between headache with or without a concomitant cerebrovascular disorder.
Methods
Study design
This is retrospective review of data collected in the Vaccine Adverse Reporting System (VAERS)4 database between January 1, 2021–June 14, 2021. The studied population were US residents that received at least one dose of a Food and Drug Administration (FDA) approved vaccine to prevent COVID-19 between January 1, 2021 and June 14, 2021.
Data sources
Data were collected from the USA VAERS database, which includes self-reports and notifications from healthcare providers, vaccine manufacturers, and subjects who self-report, regardless of assessment for causality (24). Notification is mandatory for vaccine manufacturers, and it is encouraged for healthcare providers.
Recruitment and sampling
The VAERS database is managed by the USA Centers for Disease Control and Prevention (CDC) and the Food and Drug Administration. The clinical information included in the individual adverse event reports, both as free text and automated symptom coding, was manually reviewed by neurology-trained clinicians. All consecutive adverse events reported within 42 days of vaccination with one or two doses of BNT162b2, mRNA-1273, or Ad26.COV2.S COVID-19 vaccines during the study period were screened. This 42-day period was selected as a plausible timeframe within which symptoms could be associated with the immunization based upon prior causality assessment data (25). Booster shots were not approved nor available during the timeframe of this study. The present study is a sub-analysis of the individuals who reported headache and the details of the main study were published elsewhere (25).
Eligibility criteria
Subjects were included if 1) they had received the first or second dose of Pfizer®, Moderna® or the first dose of Janssen® vaccines to prevent COVID-19; 2) had an interval between the vaccination and the report of six weeks (42 days); 3) were older than 12 years (the minimum FDA-approved age for COVID vaccination during the study time frame); 4) reported headache following vaccination. Subjects were excluded if type of vaccine was not specified or if the timing from vaccine to symptom onset was not noted.
Variables
Age (years), sex, type of vaccine (Pfizer® BNT162b2, Moderna® mRNA-1273 or Janssen® Ad26.COV2.S), the interval between the vaccination and the symptom onset, and the presence of the targeted neurological disorders were analysed, namely acute CVT, acute IS and/or acute ICH, including subarachnoid haemorrhage (SAH), intraparenchymal haemorrhage (IPH) and subdural hematoma (SDH). No other vaccines were studied since these were the three approved vaccines in the USA during the studied period. The total number of individuals vaccinated, and the total number of doses administered during the study period in the USA were gathered from publicly available CDC data (26).
Statistical analysis
We present categorical data as frequency and percentage and numeric data as mean and standard deviation or median and inter-quartile range when the distribution was not normal. We calculated the frequency of reported cases of headache per 1 million doses during the studied period. We compared age, sex and time intervals between the vaccination and symptoms onset across the three COVID-19 vaccine types. We also compared headache sufferers with or without a reported cerebrovascular event (CVT, IS, ICH). Whenever applicable, subjects with a combination of CVT plus haemorrhage or IS plus haemorrhage were also evaluated. We used two-tailed chi-square tests for hypothesis testing of categorical variables, and independent samples Student’s t-test and analysis of variance test for numeric continuous variables when the distribution was normal and Mann-Whitney U test and Kruskal-Wallis otherwise.
We assessed the diagnostic accuracy of the three-day temporal cut-off for the differentiation of headache individuals with versus without acute CVT, acute IS, or acute ICH (21). We estimated the sensitivity, specificity, positive likelihood ratio, negative likelihood ratio and Area Under the Curve (AUC). Based on the disease prevalence in the sample, positive predictive value, negative predictive value, and diagnostic accuracy were also calculated. All these parameters were calculated within the 95% confidence intervals (CI) (27).
Statistical significance threshold was <0.05 after adjusting for multiple comparisons according to the False Discovery Rate (FDR), estimated with the Benjamini-Hochberg procedure (28). Missing data was managed by complete case analysis. There was no sample size estimation, and the analysis included all available data during the study period.
Ethics
We used publicly available de-identified data. According to the New York University Grossman School of Medicine Institutional Review Board, this study was not subject to institutional review board, or patient consent.
Results
During the study period, 314,610 adverse effects were reported to VAERS out of 306,907,697 COVID-19 vaccine doses. There were 140/314,610 (0.04%) cases of CVT, 1805/314,610 (0.57%) cases of IS and 309/314,610 (0.09%) cases of ICH. Headache was reported in 41,952/314,610 (13.3%) and 41,700 fulfilled eligibility criteria. Figure 1 describes the flow diagram of studied cases. The number of reported cases of headache per 1 million doses was higher among the Janssen® vaccine recipients compared to either Pfizer® or Moderna® (0.07% of doses vs 0.01% or 0.01% of doses, respectively, P < 0.001).

Flow diagram of the study.
Table 1 summarizes the demographic variables of the entire study sample of individuals with headache and each of the vaccine groups. Subjects in the Janssen group were younger, related to the approval strategies, and more often male, compared to the Pfizer and Moderna groups. The median time between the vaccination and headache onset was similar in the three groups, 1 [0–1] for Pfizer® and Moderna®, and 0 [0–1] for Janssen®. There were 178 (0.4%) cases of cerebrovascular disorders within patients with headache, including 31 cases of CVT, 107 cases of IS and 50 cases of ICH. Of note, four patients had a combination of IS and ICH and six patients had CVT and ICH. The relative proportion of cases of CVT, ICH and IPH within the sample was higher among Janssen® vaccine recipients, and the proportion of IS was slightly lower within the Pfizer® vaccine recipients (Table 1).
Demographic parameters and frequency of cerebrovascular disorders.

FDR: False Discovery Rate. P values after FDR adjustment for multiple comparisons.
Time to headache onset
The number of days between the vaccination and the headache onset was higher in subjects with CVT, IS, ICH or a combination of CVT and ICH or IS and ICH (all P < 0.001) (Figure 2).

Boxplot of median time between the vaccination and the headache onset.
Figure 3 represents the cumulative proportion of individuals who reported headache from the first day until day-42 following immunization, comparing the groups of CVT, IS and ICH.

Cumulative proportion of individuals with headache over time.
Diagnostic accuracy of three days as temporal cut-off
A cut-off of ≥3 days from vaccination to headache onset was 87.1% specific for associated cerebrovascular events and the negative predictive value for headache onset in <3 days was 99.98%, 99.86% and 99.95% for CVT, IS and ICH, respectively. Table 2 presents the diagnostic accuracy of the 3-day cut-off for each of the studied cerebrovascular disorders.
Diagnostic accuracy of delayed onset of the headache (≥day 3) as red flag.

*These values are dependent on disease prevalence. CI: Confidence interval.
Across sub-categories of cerebrovascular events, there were no differences in age or sex among the three vaccine types. However, subjects with an IS or an ICH were older than patients than patients without vascular events (Table 3).
Comparison of demographic parameters and interval between the vaccination and the headache onset in subjects with cerebrovascular events, stratified by vaccine type.
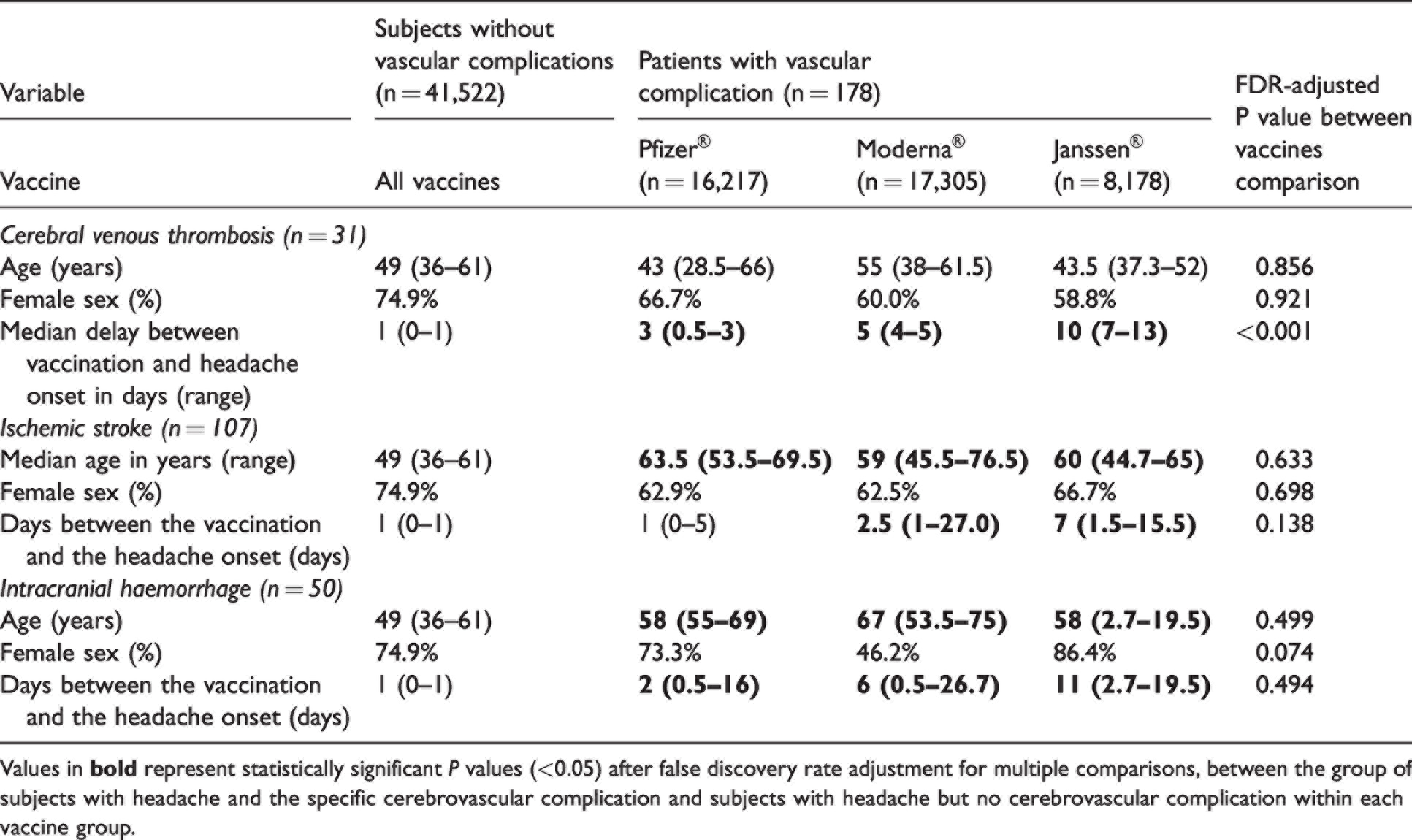
Values in
Discussion
In this study, we assessed 314,610 VAERS reports accrued during the first 6.5 months of 2021, representing adverse events reported in over 306 million COVID-19 vaccine doses in the United States. We identified 41,700 cases where headache was one of the reported symptoms, and we evaluated the frequency of cerebrovascular disorders and timing of headache. The main finding of the present study is that a delayed time from vaccine to headache onset of ≥3 days may be suggestive of a concomitant cerebrovascular event such as CVT, IS or ICH.
Despite the pivotal trials of the BNT162b2 (Pfizer®), mRNA-1273 (Moderna®) and Ad26.COV2.S (Janssen®) vaccines which included 18,556; 15,168; and 21,869 patients, respectively, there were no indication for an increased risk of cerebrovascular disorders (1 –3). This could be related to the sample size (1 –3), which was insufficient to detect very infrequent adverse events. The association between non-replicant adenovirus vector-based vaccines and thrombotic events was based on an observed rate higher than the expected at a population level in several European countries and the US (7,8,29), the specific clinical presentation with a worse outcome than the CVT observed in the general population (14,17,30), the presence of severe thrombocytopenia (13,14,17) and the presence of anti-PF4 antibodies (13,14,17,23). Still, the rates have been within the range of 1 case per 100,000 vaccine recipients (7 –11), which requires country-level analyses to evaluate a possible association, based on an increased number of observed cases, compared to the expected background rate.
In the present study, the number of administered doses was 306,907,697; which allows for detection of very rare adverse events. The number of cases of Guillain-Barré syndrome, CVT and seizures occurred at higher rate than the expected background rate, but only in patients vaccinated with the Janssen® vaccine (25). TTS has not been reliably reported in patients with mRNA vaccines (7 –11). In the case of cerebrovascular disorders, in patients who complained of headache within the presenting symptoms, we observed 17 cases of CVT, 24 cases of IS and 14 cases of isolated ICH out of 11,606,202 Janssen® vaccine doses. This would lead to an incidence rate between 0.13 (95% CI: 0.07–0.20) cases of CVT per 100,000 and 0.32 (95% CI: 0.25–0.40) cases of any cerebrovascular disorder per 100,000 Janssen® vaccine doses, in line with prior US studies and reports (8,10). The frequency of TTS can be influenced by the profile of population that is immunized, since the risk is higher in younger patients (7 –10), in contrast with ischemic stroke and intracranial haemorrhage, which occurred in the usual age, being patients with cerebrovascular disorders older than patients with non-complicated headache.
Even though the frequency of thrombotic events is extremely low, the occurrence of CVT and vaccine-induced immune thrombotic thrombocytopenia (VITT) has caused significant vaccine hesitancy (31,32). Headache has been listed as a main presenting symptom of TTS (21,33,34) and is a common symptom in patients with cerebrovascular disorders (15 –17). In the USA, headache is the fourth most frequent reason for a visit to the emergency room, when including all causes (35,36). For all these reasons, frontline healthcare workers must be prepared to identify which individuals with new-onset headache may have an underlying life-threatening cause with a need of emergency management. The presence of red flags, such as analgesic resistance, progressive worsening, change in the phenotype of a pre-existing headache or the presence of associated symptoms (19,20) should alert about the possibility of a secondary cause (19,20).
Delayed onset of headache is an additional tool to differentiate subjects with an emergent cause of the headache. It was proposed by the Secondary Headache Special Interest Group of the International Headache Society (22) and was included in the World Health Organization guidelines of TTS management (21). It should be used in combination with the traditional headache-specific red flags (19,20), and despite being an accurate discriminator, the sensitivity and specificity were not absolute. Patients with these red flags should be adequately evaluated with the optimal imaging modalities, which should include visualization of cerebral arteries and veins.
We found that the number of reported cases of headache was five times higher among the Janssen® vaccine recipients, however, this finding should be interpreted cautiously. Healthcare workers are required to report to VAERS all adverse events following vaccination that are listed on the VAERS Table of Reportable Events, including death and life-threatening adverse events, hospitalization, persistent or significant incapacity, congenital anomalies, medical events post-vaccination that may jeopardize an individual or require medical or surgical intervention, vasovagal syncope, or shoulder injury (24). In theory, cerebrovascular disorders would meet the above-mentioned criteria and therefore should be reported to VAERS. In the case of headache, the observed frequency in this study was far from the 39–59% frequency described in the pivotal clinical trials (1 –3), despite the fact that it was the most frequently reported neurological symptom/disorder to VAERS (25). This could be related with the spontaneous recovery of most cases. The asymmetrical frequency of cerebrovascular disorders within the different vaccines could be related to a more pronounced attention in social media (31,32,37).
Some studies have also reported an increased rate of CVT following other COVID-19 vaccines. A study including almost 30 million people vaccinated in England reported an increased risk ratio for CVT 3.6 (95% CI: 1.4–9.3), ischemic stroke 1.1 (95% CI: 1.04–1.2) and arterial thromboembolism 1.06 (95% CI: 1.01–1.10) 15–21 days following the Pfizer® vaccine, being these risks higher following the AstraZeneca vaccine® (12). An analysis of EudraVigilance database reported a frequency of CVT 10 times higher in patients vaccinated with AstraZeneca® vaccine compared with patients vaccinated with Pfizer® (38). Another study in Europe, assessing EudraVigilance data, described a reporting rate of CVT of 1.9 (95% CI: 1.7–2.1) following the Pfizer® vaccine, 5.6 (95% CI: 4.7–6.6) following the Moderna® vaccine, 21.6 (95% CI: 20.1–23.1) following the AstraZeneca® vaccine and 11.5 (95% CI: 9.6–13.7) following the Janssen® vaccine (39). In a recent study reporting the frequency of neurological adverse effects the reported rate of CVT was 12 times higher following the Janssen® vaccine than following the Pfizer® or Moderna® vaccines (25). This has been related with the systemic response triggered by the immunization, with headache being one of the most common symptoms.
The present study has noteworthy limitations. Diagnoses may not be accurate, since both healthcare providers, vaccine manufacturers and subjects may complete the notifications, with a variable accuracy and completeness (24). Because the available data is de-identified and contains a limited number of variables, we were unable to evaluate factors that may have improved predictive modelling, such as the presence of other symptoms, prior medical history including risk factors, the interval between the headache onset and the cerebrovascular disorder diagnosis, or some headache-specific variables, (i.e., phenotype, associated symptoms, red flags) which were not consistently reported. Since data was anonymized, we could not review individual medical records to verify the accuracy of diagnoses. The cross-sectional nature of the VAERS system limits the longitudinal evaluation of subjects, and some individuals that had immediate headache could develop later a cerebrovascular disorder, however, the headache onset in patients with cerebrovascular disorders was clearly defined in all cases as a new-onset headache, and this is how it should be interpreted in clinical practice, as a new-onset headache or a clear worsening from a pre-existing headache (19,20). Though most reports provide information about when the symptoms started, some cases may be reported separately with limited information about the immediate outcome of patients. Reporting bias is a well-known limitation with passive reporting systems. Because post-vaccination CVT received a great deal of media coverage, there may have been a bias toward reporting these events more often. On the other hand, passive surveillance systems may be affected by underreporting. It is possible that benign symptoms, such as headache that occurred in a delayed fashion without other sequelae may not have been reported. This would lead to inflated specificity and NPV values for the timing of headache as an indicator of concomitant cerebrovascular disease. The inputs of this study were accomplished in a single country, where not all the available COVID-19 vaccines (e.g., AstraZeneca® ChAdOx1-S vaccine) were approved. Despite these limitations, the present sample included >40,000 cases of headache without cerebrovascular disorders and the demographic and clinical profile were similar to that of the published randomized clinical trials (1 –3) and real-life studies (5,6), with an extensive age, gender and racial distribution representing the US population.
Conclusion
Headache following vaccination to prevent COVID-19 typically presents within one day of inoculation. The frequency of cerebrovascular disorders among individuals with post-vaccine headache was extremely rare. Delayed onset of headache symptoms ≥3 days may help differentiate between subjects with and without cerebrovascular events, mainly cerebral venous thrombosis, which may not necessarily be related to the vaccination.
Clinical implications
Cerebrovascular disorders are very rare in subjects with headache following COVID-19 vaccines. Delayed-onset of headache following vaccination to prevent COVID-19 should be considered as a red flag of a possible concomitant cerebrovascular disorder. The three-day threshold showed a good diagnostic accuracy in the differentiation of isolated headache and headache in the context of a cerebrovascular disorder following COVID-19 vaccines. Patients with a new-onset headache or change in characteristics of a pre-existing headache, with delayed-onset following COVID-19 vaccination or red flags should be evaluated for a secondary cause of the headache.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024221099231 - Supplemental material for Timing of headache after COVID-19 vaccines and its association with cerebrovascular events: An analysis of 41,700 VAERS reports
Supplemental material, sj-pdf-1-cep-10.1177_03331024221099231 for Timing of headache after COVID-19 vaccines and its association with cerebrovascular events: An analysis of 41,700 VAERS reports by David Garcia-Azorin, Betül Baykan, Ettore Beghi, Mohamed F Doheim, Cesar Fernandez-de-las-Penas, Hasim Gezegen, Alla Guekht, Fan Kee Hoo, Michele Santacatterina, James Sejvar, Arina A Tamborska, Kiran T Thakur, Erica Westenberg, Andrea S Winkler, Jennifer A Frontera and Contributors from the Global COVID-19 Neuro Research Coalition in Cephalalgia
Footnotes
Acknowledgements
We would like to thank the patients and clinicians who contributed information to the VAERS dataset and t
Data sharing statement
De-identified datasheets are available for other researchers upon reasonable request to the corresponding author. DGA had full access to all the data in the study and takes full responsibility for the integrity of the data and the accuracy of the data analysis.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DGA received honoraria from the World Health Organization. The rest of the authors and coalition members declare no conflicts of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
