Abstract
Clinic-based headache registries collect data for a wide variety of purposes including delineating disease characteristics, longitudinal natural disease courses, headache management approaches, quality of care, treatment safety and effectiveness, factors that predict treatment response, health care resource utilization, clinician adherence to guidelines, and cost-effectiveness. Registry data are valuable for numerous stakeholders, including individuals with headache disorders and their caregivers, healthcare providers, scientists, healthcare systems, regulatory authorities, pharmaceutical companies, employers, and policymakers. This International Headache Society document may serve as guidance for developing clinic-based headache registries. Use of registry data requires a formal research protocol that includes: 1) research aims; 2) methods for data collection, harmonization, analysis, privacy, and protection; 3) methods for human subject protection; and 4) publication and dissemination plans. Depending upon their objectives, headache registries should include validated headache-specific questionnaires, patient reported outcome measures, data elements that are used consistently across studies (i.e., “common data elements”), and medical record data. Amongst other data types, registries may be linked to healthcare and pharmacy claims data, biospecimens, and neuroimaging data. Headache diagnoses should be made according to the International Classification of Headache Disorders diagnostic criteria. The data from well-designed headache registries can provide wide-ranging and novel insights into the characteristics, burden, and treatment of headache disorders and ultimately lead to improvements in the management of patients with headache.
Keywords
Introduction
A patient registry is an organized method of collecting data from a group that shares a disease, condition, or exposure (1). Registry goals might be epidemiological, clinical, and/or policy related. On the population level, registries collect information of all cases that occur within a specific setting, often within a specific geographical region. As the underlying population size is generally known, such registries may allow for estimates of disease incidence and prevalence for conditions that are nearly completely ascertained (2). Registering of patients with a specific condition can also be done in the clinical setting, which is referred to as a “clinic-based” or “hospital-based” registry. The general approach, however, is similar in that all patients with a given disease or condition in that clinic or hospital should be included in the registry. Clinic-based registries are either cross-sectional or longitudinal cohort studies. They collect data from a large group of patients as they receive usual medical care (see Table 1 for a comparison of clinic-based registries vs. controlled trials vs. patient level survey data vs. electronic health record/medical claims/pharmacy data). Registry data can be obtained from multiple sources, including structured patient interviews, validated questionnaires, symptom diaries, patient reported outcomes (PRO) measures, medical records, insurance claims, and pharmacy claims (3). Patient registries might include physical examination findings, biological specimens, imaging data, physiological data, and diagnostic test results. Registries collect data that can be used for numerous purposes, including defining disease characteristics, identifying heterogeneity amongst those with a disease or condition, understanding longitudinal natural disease course or its modification by treatment, quantifying long-term tolerability, safety, and effectiveness of treatments, measuring health care resource utilization, defining and comparing different treatment strategies, identifying patients for clinical trials, identifying potentially new indications for existing therapies, discovering biomarkers that predict treatment response and disease outcomes, measuring clinician adherence to guidelines, measuring quality of care, and calculating cost-effectiveness of various care strategies (1,4,5). Registry data are valuable to numerous stakeholders, including patients and their families, healthcare providers, scientists, healthcare systems, regulatory agencies, pharmaceutical companies, healthcare payers, employers, and policymakers (6,7).
Contrasting typical features of clinic-based registries, patient surveys, electronic health record/medical claims/pharmacy data studies, and randomized controlled trials.
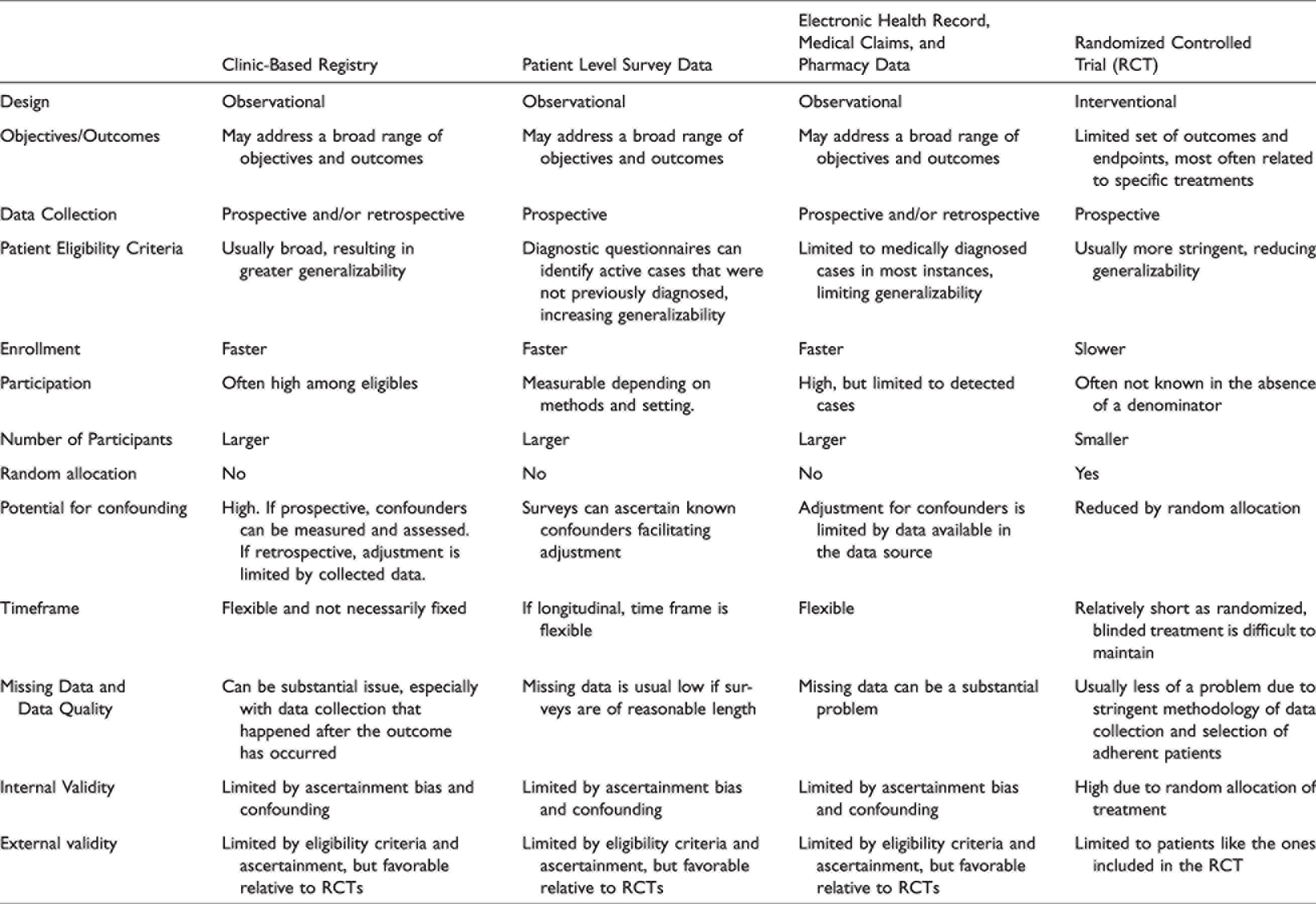
For conditions that are underdiagnosed, like migraine, certain patient identification strategies may lead to incomplete ascertainment of the disease of interest and underestimation of prevalence and incidence. For example, if registries are derived from primary care clinics, many people with migraine may go undiagnosed. Particular caution is required if there are systematic differences among persons who are ascertained and those who are not enrolled in a registry. In the US, about 60% of people with migraine report having received a medical diagnosis (8). Consultation and medical diagnosis are more likely in people with severe migraine and high disability (9,10). This problem may be amplified if a clinic-based registry is derived from headache subspecialty centers, as this would result in non-representative samples that would not allow for prevalence and incidence estimates unless the headache condition is rare. While these factors do not undermine the value of clinic-based registries in tracking clinical course, health care utilization or barriers to treatment among people with headache who are identified, they may limit generalizability of findings and must be considered when interpreting results. Nonetheless, there are some advantages associated with patient enrollment from headache subspecialty centers, such as the ability to include rarer headache disorders and greater patient willingness to provide data that deeply phenotypes their condition. Headache subspecialty registries may also be particularly advantageous for studying rare disorders which may not be diagnosed accurately outside of subspecialty centers, such as paroxysmal hemicrania or short-lasting unilateral neuralgiform headache attacks with conjunctival injection and tearing (SUNCT) and for studies of treatments primarily given in subspecialty care, such as onabotulinumtoxinA and nerve blocks.
This International Headache Society guideline on Clinic-Based Headache Registries was developed by a panel of authors who have experience with developing and managing clinic-based headache patient registries. Of note, clinic-based registries are sometimes referred to as “patient databases”. The document may serve as guidance when developing clinic-based headache patient registries, including recommendations about developing a registry, data sources, data harmonization and quality, human subject protection, patient engagement, data and specimen sharing, and result dissemination and publication. Table 2 summarizes key considerations when developing clinic-based headache registries.
Key considerations for a clinic-based headache registry.

ICHD = International Classification of Headache Disorders.
Developing a clinic-based headache registry
Existing guidelines/advice on registry development
There is existing guidance on patient registry development from the United States Food and Drug Administration (FDA), the European Medicines Agency (EMA), the Agency for Healthcare Research and Quality (AHRQ), the Patient Centered Outcomes Research Institute (PCORI), and other agencies and medical societies (1,11
–13). This International Headache Society guideline incorporates recommendations from these other agencies, but is unique since it includes guidance specific to clinic-based
Setting goals for the registry
Overarching goals for a registry are determined prior to developing the research and data collection methods. Doing so ensures that the chosen methodology, data collected, and sample size are optimal for addressing the overarching registry goals. Goals may be very narrow or broad. For example, a narrow goal might be to assess the safety of a particular migraine treatment during pregnancy. Such a registry would enroll pregnant women, some of whom were exposed to and others who were unexposed to the migraine treatment of interest. Another narrow registry might seek to identify genotype-phenotype correlations among people with migraine. Such a registry might deeply phenotype each enrolled patient and include their genetic information. An example of a registry with broad goals is one that seeks to characterize the clinical course of migraine and predictors of migraine outcomes. Such a registry might enroll and follow a broad, representative sample of people with migraine.
It is recommended that specific research aims be determined when developing the registry, to ensure that the data that are then collected will allow those research aims to be addressed. However, additional specific research aims might first be identified after the registry is populated with data. Of note, these additional analyses might require approval by the Institutional Review Board (IRB)/Medical Ethics Committee (METC). Whether aims are determined prior to or after data collection, when registry data are used for analyses, the specific aims of the analyses should be determined a priori and contained within a formal document.
Registry team composition and governance
The optimal composition of the team developing and managing a headache registry depends on the size of the registry and its goals and methods. Large registries may include headache specialists, general practitioners, registry experts, information technology experts, epidemiologists, statisticians, data managers, patients who have the disease of interest, lawyers with expertise in clinic-based registries, and registry administrators. If relevant, the team also includes experts in biospecimen collection and processing, genetics, neuroimaging, economics, survey design and administration, and healthcare policy. The registry team can be divided into committees:
(1) (2) (3) (4) (5) (6)
The size, areas of expertise, and complexity of the registry team should reflect the magnitude, research methods, and goals of the registry itself. Smaller, less complex registries might be led by a smaller number of individuals and fewer committees, as long as there is sufficient leadership and expertise to oversee the registry functions.
Protocols and proposals
Registry development and use of registry data require proper documentation and should follow a standardized process. Registries typically have general overarching aims, such as studying the course of a headache disorder, as well as more specific aims that test focused hypotheses, such as persistence with specific therapies or the predictive ability of a specific biomarker.
Formal research protocol for developing a clinic-based registry
A formal research protocol is developed prior to initiating a registry. The protocol includes the evidence gap and rationale for conducting the registry, global goals and initial specific aims, study design and its limitations, participant eligibility criteria, selection of data elements and methods for data collection, data sources that will be used, methods for data cleaning and ensuring data quality, plans for protecting human subjects and their data and specimens, and the registry governance structure. If multiple data sources are used, methods for data harmonization are included. A statistical analysis section for addressing key research aims and sample size calculations for the overarching aims should be included. Plans for publications, result dissemination, and data sharing are included. The duration of time that registry data and specimens will be collected and stored are included in the protocol. Registration of the protocol within a publicly available database, such as the European Network of Centres for Pharmacoepidemiology and Pharmacovigilance (ENCePP) (https://www.encepp.eu/encepp/resourcesDatabase.jsp) or clinicaltrials.gov, should be considered and might be required depending on the research aims, funding sources, locations from which patients are enrolled, and journal publication policies.
Formal research protocol for using data from a clinic-based registry
Use of data from the clinic-based registry should be requested by submitting a formal proposal to the Use, Access, and Publication Committee. This proposal should include the specific scientific questions, the data needed, the methods that will be used to analyze the data, a power estimate for the analysis, and the sources of study funding. Requesters must provide assurance that registry data and/or specimens will be protected from unauthorized or inappropriate use. Although the method of providing assurance might vary, this is typically accomplished through IRB/METC approvals, and data and materials transfer agreements.
Registry infrastructure
New headache registries should be supported by a strong electronic infrastructure. Although paper forms might still be used in some registries, direct entry of data into electronic forms and the use of mobile apps has several advantages. Direct data entry into electronic forms and transfer of electronic medical record data into the registry without the need for human intervention can reduce data entry errors and the time required for manual data entry. Furthermore, direct electronic entry allows for automated data checking and warnings when there are missing data and when data are not within pre-determined ranges, suggesting they might be erroneous. Direct electronic entry facilitates participation by individuals who are physically distant from the research center while obviating the need for mailing paper forms or attachments by e-mail. However, registries that require direct electronic data exclude participation from individuals who do not have access to devices or the internet, and thus risk excluding patients of lower socio-economic status, the elderly, and other groups (14). In addition, some patients who have extreme photosensitivity might find it difficult to view digital screens. To not exclude certain individuals, when feasible and consistent with the registry goals, registries could include options for data collection that do not require electronic data entry. All data entries should be time and date stamped and fully auditable, meaning there are records of who entered the data and of all data revisions. It is important that the registry has proper data management support and oversight, ideally from a dedicated data manager.
Registry design
Patient registries are observational studies. Depending on the specific goals for the registry and the resources available, registries can be cross-sectional or longitudinal, and data can be collected prospectively or retrospectively. Commonly, a combination of prospective (e.g., patients answering questionnaires, maintaining headache diaries) and retrospective data collection (e.g., data from medical records) is used. The limitations of retrospective data need to be recognized, especially differential misclassification if data were not collected using standardized methods. In rare occasions, registries can be part of a cluster randomized trial in which the clusters are compared. However, each individual registry remains observational.
One of the advantages of a clinic-based registry is to have an available target population for enrollment into randomized clinical trials. In addition, causal inference from large observational registries can be used to emulate a target trial when the randomized experiment cannot be conducted (15,16).
Patient Recruitment, Eligibility, and Retention
Specific inclusion and exclusion criteria need to be defined a priori and included within the registry protocol. The specifics of the criteria depend on the overarching goals for the registry. However, large patient registries typically have broad eligibility criteria that allow for a wide range of patients with the same disease or condition to be studied, an approach that strengthens the external validity/generalizability of registry results. Headache registries might focus on one or more specific ICHD headache diagnoses or enroll individuals with any headache type.
Offering enrollment into a patient registry is often achieved via convenience sampling, which however, may result in selection bias. Patients might be enrolled as they are seen in clinical practice or be recruited from healthcare organizations or pre-existing research panels. The settings from which individuals are recruited, the proportion offered enrollment, and the proportion of those offered enrollment who participate, all impact the representativeness of the enrolled participants and the generalizability of registry results. Thus, details regarding the settings from which individuals are recruited, the number of patients screened, enrolled, and excluded should be collected and reported. When possible, comparing characteristics of patients who do not enroll into the registry with those who do enroll may help determine the generalizability of registry results. Methods of enrollment into registries should include diversity, equity, and inclusion plans that assess the characteristics of registry patients relative to the source population that forms the sampling frame. To the extent that full representation is not achieved, the influence of non-representativeness on registry results should be carefully considered and reported.
In longitudinal registries, retention of participants is crucial. Several methods might increase retention rates, including but not limited to 1) communications with registry participants about the importance of the registry and how the data will be used to advance headache science, disease management, and treatment (as applicable); 2) providing feedback to participants about the data they provide to the registry. For example, the registry could provide summaries of a patient’s headache diary data and longitudinal patterns. Participants might be able to share these summaries with their headache clinician, enhancing the patient-clinician encounter; 3) providing reminders to participants about completing their registry responsibilities, such as completing questionnaires and diary entries; 4) providing compensation to participants for the time they spend participating in registry activities; 5) when possible, allowing flexibility on how follow-up data are collected (e.g., in-office visit, telephone visit, mailed questionnaires) (17).
Headache diagnoses
Headache registries should assign headache diagnoses according to International Classification of Headache Disorders (ICHD) diagnostic criteria (18). If new ICHD criteria are published while a patient registry is ongoing, new diagnoses should be made using the most recent criteria, with documentation of which ICHD version was used for each patient. Patients must receive all appropriate ICHD diagnoses, meaning some patients will have more than one diagnosis (e.g., chronic migraine plus migraine with aura plus migraine without aura). Since an individual’s headache diagnoses can change over time, longitudinal registries must allow for new ICHD diagnoses to be added. Some registries may be populated with pre-existing data, such as data extracted from medical records. In these situations, the diagnostic criteria that were used might not be known, and this diagnostic uncertainty needs to be included as a limitation when reporting the results of scientific studies using registry data. When possible, pre-existing diagnoses should be validated using ICHD criteria (e.g., using patient-reported symptoms to assign an ICHD diagnosis).
Inclusion of validated questionnaires, patient reported outcomes, and common data elements
Validated questionnaires are included in a headache registry. Numerous validated questionnaires relevant to headache, associated symptoms, and comorbidities are available (19,20). In addition, many questionnaires have been validated specifically within headache populations, most commonly amongst individuals with migraine (19 –21). When validated questionnaires for the headache under evaluation are not available, registries should include questionnaires that have been validated within populations that most closely resemble the patients who will be enrolled into the registry. When possible, questionnaires that have been validated in the language spoken by the research participants should be used. Headache registries also commonly include non-validated questionnaires, such as structured interviews used to collect information about headache characteristics and headache history. If newly designed questionnaires are included in the registry, validation against existing validated questionnaires and/or clinician diagnoses as the gold-standard is expected.
Patient reported outcome (PRO) measures can be included in headache registries. PROs measure health, quality of life, or functional status associated with health care or treatment as reported by patients themselves (22). PROs do not require interpretation of patient responses by a clinician or scientist. PROs are particularly important when studying headache since many of the outcomes of interest are relatively subjective and dependent on patient self-report (e.g., headache intensity, headache-related disability). A systematic review of headache PROs published in 2018 found twenty-three evidence-based PRO measures, including five specific to health-related impacts from migraine, six to health-related impacts from headache, six which assess migraine-specific treatment response or satisfaction, and six generic measures (23).
When available, common data elements (CDEs) are included in a registry. CDEs use standardized language and associated values for specific data elements. Use of CDEs allows for sharing data across studies and for performance of meta-analyses. Headache CDEs are available from the National Institutes of Health (NIH) National Institute of Neurological Disorders and Stroke (NINDS) (20) [https://www.commondataelements.ninds.nih.gov/headache]. When a goal for the registry is to determine response to treatments, data that correlate with endpoints included in the International Headache Society clinical trials guidelines for acute and preventive therapy should be collected (24 –29). When assessing real-world effectiveness of treatments is a goal, inclusion of additional clinically meaningful and patient-centered outcome measures is recommended.
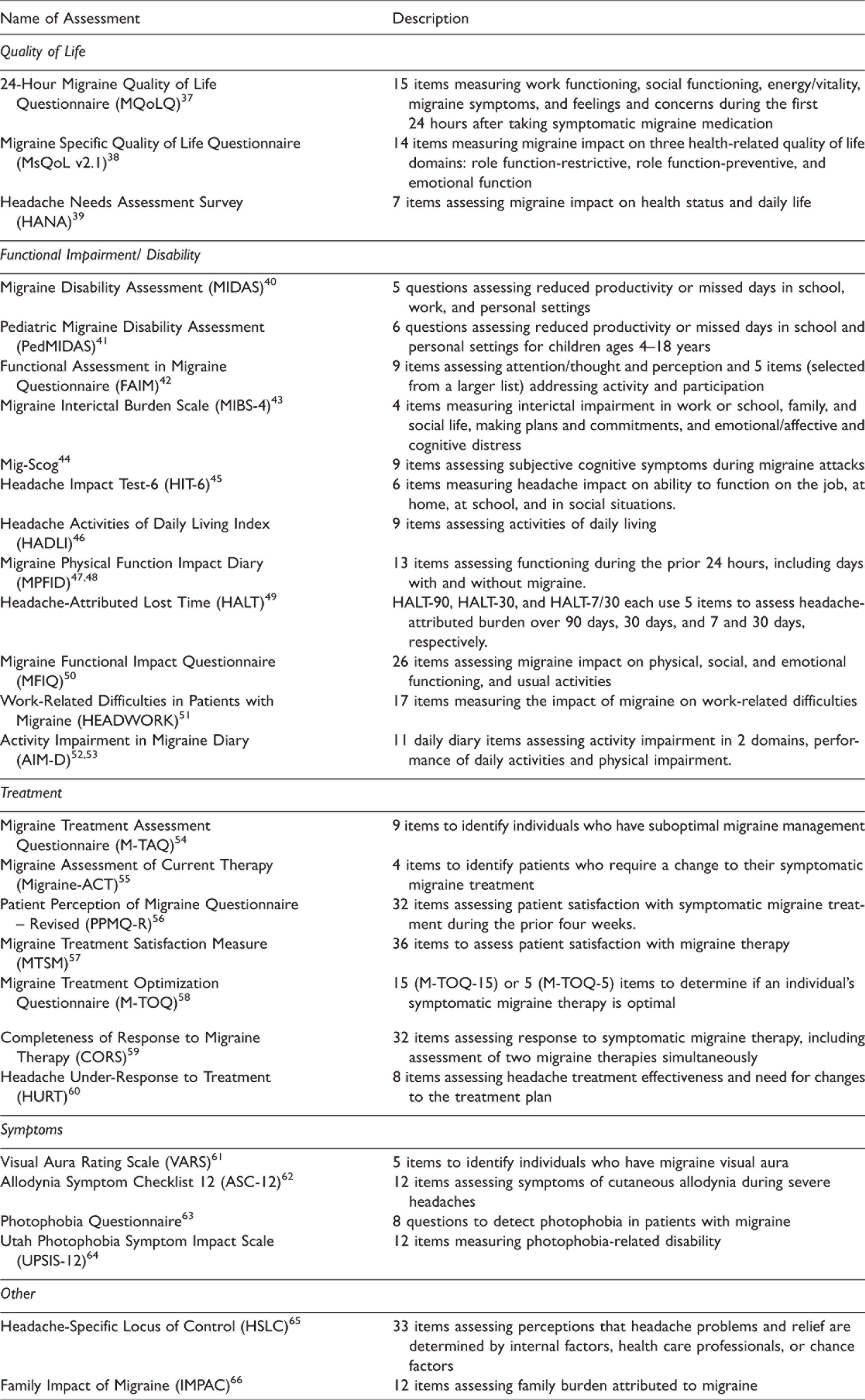
Table 3 lists and describes headache-specific questionnaires and PROs that might be included in a headache registry. Additional assessments may be included that obtain information about socio-demographics, patient behaviors (e.g., diet, exercise, alcohol), prior medical history, family medical history, comorbid and coexistent diseases (e.g., depression, anxiety), treatments, exposure to headache treatments during pregnancy and pregnancy outcomes, and general quality of life. However, it is crucial to avoid overburdening patients with extensive data entry, so the value added with each additional questionnaire should be weighed against the drawback of incomplete data collection.
Headache Questionnaires and Patient Reported Outcome Measures (PROs). Headache questionnaires and PROs that are used specifically to assess individuals with headache. Assessments that are not specific for headache, such as those that interrogate general health (e.g. Patient Global Impression of Change), overall quality of life (e.g. SF-36, EQ5D), psychological symptoms (e.g. Patient Health Questionnaire, Generalized Anxiety Disorder 7), comorbid conditions, exposure to headache treatment during pregnancy and pregnancy outcomes, and pain other than headache, are not included in this Table, but are often included in headache registries. This table is intended to be used as a resource to help with identifying and choosing assessments for a headache registry. This table is not intended to be all-inclusive, and development of new questionnaires and PROs is anticipated.
Statistical considerations
Prior to starting data collection, the sample size needed to address the overarching aims of the registry should be determined. When analyses of the collected data are proposed, specific research aims, data analysis plans, sample sizes, and statistical power need to be determined a priori. Sample size calculations might be based on minimally important differences that need to be detected or the precision with which the outcome must be measured (1). Loss to follow-up and missing data need to be taken into consideration for each analysis. Revisions to statistical plans should be documented. Since registries often include large datasets from which numerous analyses are performed, statistical analysis should consider the risk of false positive discovery due to multiple comparisons.
Data sources
Registries utilize single or multiple data sources, including but not limited to data provided by the patient, legal guardian, clinician/researcher, medical records, and pharmacy and insurance claims, as well as biospecimens, neuroimaging, and other data types.
Patient provided data
Patient provided data include patient-completed questionnaires, headache diary data, and data derived from passive monitoring, such as that collected with mobile device applications and wearable devices (e.g., physical activity quantification, heart rate monitors, sleep monitoring). The section above, titled “Inclusion of validated questionnaires, patient reported outcomes, and common data elements” discusses these concepts further.
Headache diaries prospectively collect information about individual headache attacks (30,31). Most commonly, diary information is entered daily. Data on non-attack days is often captured for comparative purposes and to assess interictal symptoms. Headache diary entries should be “time-locked” so that the exact day and time of data submission is known. Since headache diaries are intended for prospective data collection, they usually include limits on the amount of time that can pass between a calendar day and submission of data for that day. For example, patients might be able to retrospectively enter data into the headache diary for up 24 to 72 hours after a calendar day. Longer time intervals result in more recall bias. In addition to collecting information about headache presence, data on headache intensity and duration, associated symptoms (e.g., photophobia, phonophobia, nausea, vomiting, cranial autonomic features), aura symptoms, premonitory and postdrome symptoms, headache-related disability, general well-being (during and between headache attacks), use of acute and preventive treatments including over-the-counter therapies, side effects, and perceived headache triggers might be collected. To optimize compliance with diary maintenance it is necessary to find the right balance between collecting all desired information and avoiding overburdening the patient. This might be accomplished by limiting the number of questions, making the diary user-friendly with easy to answer questions, and rotating some of the questions from day-to-day to not ask every question every day. Although paper diaries are still feasible for smaller studies and for participants who do not have access to electronic devices, electronic headache diaries that are completed via the internet and/or mobile applications are typically preferred for large headache registries.
It is essential to provide participants with clear and understandable education on how to complete registry questionnaires and headache diaries. Such education might be provided in-person, via printed materials, using videos, and by placing instructions within headache registry electronic platforms. It is recommended that patient partners participate in the development and review of educational materials, helping to ensure that the materials will be useful and understood by registry participants.
Even if initial recruitment is tied to the clinic, it may be helpful to allow patient follow-ups to occur outside of the usual clinical setting using remote data capture methods. For example, questionnaires and PROs might be sent directly to participants via e-mail or mobile apps. Research visits might occur via telehealth methods. Automated data capture with devices may also be used. For example, actigraphy can be used to measure physical activity and sleep parameters. Heart rate monitors and skin impedance can be used to capture physiological stress. GPS monitoring can be used to capture location which in turn can be linked to weather or air pollution data. Robust protocols are required to optimize data quality and to derive reliable, quantitative indices of the measures of interest.
Clinician/researcher provided data
Commonly, the research team enters data into the registry such as the patient’s headache diagnoses, patient answers given during structured interviews, and diagnostic tests performed and their results, if such results are not populating the registry directly from an electronic medical record. When biospecimens are being collected, information about the specimen collection and processing must be entered.
Medical record data
Medical record data might be extracted from electronic medical records via ‘data pulls/pushes’ that directly populate the registry, or the research team might manually enter medical record data into the registry. Medical record data must be collected in a standardized fashion. A data dictionary is required, describing in detail each piece of data that is being extracted from the medical record so that current and future registry data users will understand the terms and vocabulary used. The data dictionary should include definitions for any labels that might be used to identify each piece of data, a detailed description of what the data represents, and the data source(s).
Health care claims data
Health care claims data can be helpful for identifying patients with a specific headache type and understanding health care resource utilization. Pharmacy claims data provide medication names, doses, supply, fill dates, and prescriber codes, allowing for longitudinal assessments of medication compliance and persistence. Non-prescription medications are not captured within pharmacy claims data, a limitation when trying to investigate total medication use. In some countries, medical claims data provide information about all inpatient and outpatient services, including diagnostic codes, services provided, procedures, tests, and treatments. Claims data can be used to collect information on large groups of patients with specific diagnostic codes. Limitations of claims data must be recognized, such as a lack of information about patient level encounters prior to enrollment in the health plan, “rule-out” diagnoses (e.g., a diagnostic claim is made but then ruled out by diagnostic testing), missing data, selection bias, underreporting of certain diagnoses (e.g., medication overuse headache), and lack of correspondence between some diagnostic codes with ICHD terminology and criteria (32). When possible, registries should include methods to address these and other limitations.
Biospecimens
When biospecimens are collected, a ‘lab manual’ that details biospecimen collection techniques, processing, storage, retrieval, and shipping is required. Depending on the intended use for the specimens, data on the timing of specimen collection in relation to the patients most recent headache (e.g., ictal vs. interictal specimen collection), use of medications, hormonal status (e.g., date of last menstrual period, (peri)menopausal status), and dietary intake might be collected. The date and time of specimen collection, the handling methods, and duration between specimen storage, processing, and analysis are recorded. Details about specimen processing (e.g., speed of centrifugation, storage temperature) and assays used (e.g., sensitivity and specificity) are documented. Results from specimen analyses are included within the registry, facilitating the sharing of such results in addition to or instead of sharing the specimens themselves. Existing guidance on developing biorepositories is available (33,34). Prior to including a biospecimen in a registry, patients need to be made aware of how their specimens and resulting data will be used and shared, with careful attention to informed consent.
Neuroimaging
Neuroimaging data collected as part of clinical care or in a standardized fashion as part of the registry research might be included. The registry should document the dates that images were collected, the imaging sequences performed, and the type of scanner used. Visual inspection of the images might be used for studies interrogating brain structural findings, such as white matter hyperintensities, cerebral infarctions, small vessel disease markers, and cerebral atrophy. Advanced analyses of structural data might interrogate other measures such as cortical thickness, regional volumes, and white matter tract integrity. When brain imaging data are collected, the registry should document the timing of headache in relation to collecting the images (e.g., ictal vs. interictal, time since end of last headache and time to the next attack). Optimally, a standard imaging protocol is predetermined and used for collecting all images. These protocols include details about the imaging equipment, imaging sequences and their parameters, methods to measure head movement, methods to collect or account for physiologic data (e.g., respirations, pulse) during functional imaging, and any tasks that participants perform during imaging. When standardized methods for collecting imaging data are not possible, the analysis of registry imaging data must consider the use of multiple scanners and different acquisition techniques. If neuroimaging data are shared, care must be taken to fully anonymize the images, the medidata associated with the images, and the image file names.
Other data
A headache registry might include other data types, depending on the registry goals. Examples include neurophysiological test data, physical and neurological examination findings, vital signs, and data from other registries or data sets. Collection of data on adverse events might be included within a registry. If so, it is essential to have a well-defined plan for identifying when adverse events are entered into the registry and reporting the adverse events according to current rules and regulations. Registries might also collect data on exposure to headache therapies during pregnancy. In many instances, regulatory approval of new medications comes with a mandate for post-approval pregnancy exposure registries that follow regulatory agency guidelines. Registries that collect data on pregnancy exposures should report such data to these post-approval pregnancy exposure registries. Resources for identifying and contacting these pregnancy exposure registries can be found at https://clinicaltrials.gov/ and https://www.fda.gov/science-research/womens-health-research/list-pregnancy-exposure-registries.
Data harmonization and quality
Data harmonization
When more than one data source is used, a comprehensive plan for data harmonization is required. Optimally, all data elements from all registry data sources are standardized prior to their collection. For example, it is preferred that each data element has the same label/name as it populates the registry, and that each data element is being reported using the same unit of measurement. However, this might not always be possible, such as when data are being extracted from multiple different medical record systems. In such a situation, the registry must map and transform each set of data to a core data element. For example, one data source may rate pain intensity on a 0-3 interval scale, while another rates pain on a 0-10 anchored scale, and a third data source uses a 100 mm visual analogue scale. Systematic strategies are required for mapping one measurement set onto another, ideally by using data which include nearly simultaneous measurement using more than one strategy to develop the calibration methodology (35,36). Challenges related to collection of registry data using more than one language also need to be considered, especially if there is concern that participants who speak different languages might perceive certain questions differently.
It is essential that all data and specimens derived from a single patient can be linked back to that individual. Furthermore, when using multiple data sources, some data might be duplicated for a single patient. Duplicative data must be identified and handled during the data harmonization process.
Data quality
Headache registries often include large datasets derived from multiple centers and from multiple data sources that have differing levels of quality. A registry must have a comprehensive data monitoring plan for assessing data quality, such as consistency, completeness, accuracy, and timeliness, and for handling low quality data (e.g., excluding such data, methods for managing missing data, and improving the quality of such data) (13). Registry protocols establish methods for limiting, quantifying, and handling missing data. The magnitude of missing data and data imputation can substantially impact the quality of registry data. When possible, automated data quality checks should be built into the database itself, providing alerts when there are out of range data or missing data. When this is not possible, data quality assurance measures should be conducted intermittently (e.g., counting missing data, plotting values to identify outliers). When data are entered manually, double data entry can optimize accuracy. Ideally, each registry has a responsible data manager. The person entering data into a registry should be documented and auditable, as should the date and time of data entry. Internal or external audits of registry data can be performed, and key performance indicators reported.
Human subject and human subject data protection
Patient registries require IRB/METC approval or a waiver. For some registries, such as those that solely extract clinically collected anonymized information, waiver of informed consent might be allowed. As with all research, the potential risks and benefits need to be thoughtfully considered by the research team and potential participants, so that they can comprehensively assess such risks and benefits. Specific regulatory requirements vary from region-to-region and it is essential to adhere to local, national, and international regulations. IRB or METC approval should cover the broad aims of the registry so that individual analyses of registry data might not require additional approvals.
Protection of data and specimens against unauthorized use is of the utmost importance. Methods to protect registry data must be included in the study protocol and built into the methods being used for data collection, data access, and data sharing. Appropriate anonymization and coding of data, specimens, and neuroimages is essential for protecting privacy. Rules regarding who has access to non-anonymized or anonymized data are determined and the registry must monitor all data access. Data protection regulations must be carefully followed, such as General Data Protection Regulation (GDPR) in the European Union. This may also have implications if data are pooled across registries.
When informed consent is required (i.e., a waiver of informed consent has not been granted by the IRB or METC), patients should be made aware of intent to share data and specimens via the initial consent form. The informed consent should include consent for anonymized data storage and sharing. It should be made clear who will have access to the data and under which conditions they may use the data. Patients should be made aware of situations in which there is mandatory reporting, such as serious adverse events attributed to a medication. Patients should also be informed as to whether they will receive their own registry results such as questionnaire scores, neuroimaging results, and results of genetic analyses. Depending on the extent to which registry research methods are the same as clinical methods or validated for clinical use, some registry data (e.g., that derived from research imaging sequences, genetic analyses) might not be appropriate for clinical interpretation and sharing with patients. The consent form should describe the intended use of biospecimens, maximum storage duration, and the patient’s right to request specimen destruction should they decide to withdraw their consent.
Patient engagement
A patient engagement plan is recommended to be part of all headache registries in which individuals with headache are actively involved (e.g., prospective registries that actively enroll patients, as opposed to registries that only pull data from existing databases). Individuals who are participating as patients in the study, as well as individuals with headache who are not serving as research participants, might be included. Patient engagement with planning the registry, conducting the research, interpreting results, and disseminating research conclusions should be strongly considered. In addition, active engagement with research participants is likely to improve compliance with registry procedures, such as completing all registry questionnaires, and adhering to longitudinal follow-up schedules.
Compensation may be used to remunerate participants for their time and to encourage full participation with registry procedures. Providing feedback to patients about their own headache patterns, such as summarized headache diary data or changes in questionnaire scores over time, can also enhance participation.
Data and specimen sharing
Data sharing is encouraged for headache registries. Data sharing increases the scientific productivity of a registry and helps to advance the headache field in an expeditious and collaborative manner. IRB or METC approval for data sharing must be sought. Regulations about data sharing, such as those provided by the General Data Protection Regulation (GDPR), must be followed (https://gdpr-info.eu/). Data sharing plans are included in the registry protocol prior to launching the registry. In some settings, formal contracts need to be in place before data can be shared. Templates of such contracts should be developed within the registry protocol. Data sharing plans include: 1) timing of data sharing in relationship to collecting the data. Often, registry investigators can access data for a defined period of time prior to making data available to outside researchers; 2) methods for investigators to submit a data access request; 3) plans for reviewing investigator data access requests (as described in the Use and Access committee section above); and 4) methods for safe, compliant data sharing. The registry determines if raw data, summarized data, or data that have already been analyzed by registry statisticians will be shared. If raw data are shared, the recipient should be informed if data quality checks and data cleaning have been conducted prior to sharing. For the protection of registry participants, full data anonymization is essential prior to sharing. The registry keeps a log of all investigators and institutions with whom data are shared. Research specific aims associated with each project for which data are shared are documented to avoid duplication of efforts amongst researchers.
The rules for sharing limited resources, such as biospecimens, typically differ from rules used for sharing data. If specimens themselves are shared, the registry might require that all data derived from the analysis of specimens be sent back to the registry for future use. If the registry has already processed and analyzed the specimens, the registry might decide to share the resulting data rather than the specimens themselves. Legal agreements regarding data and specimen use are often required.
Result dissemination and publication policy
Headache registries typically have rules regarding the dissemination, presentation, and publication of registry results. Authorship policies are predetermined and consistent with authorship guidance from agencies such as the International Committee of Medical Journal Editors (ICMJE) (http://www.icmje.org/recommendations/browse/roles-and-responsibilities/defining-the-role-of-authors-and-contributors.html). The registry team documents all dissemination activities, presentations, and publications resulting from registry work to avoid duplication of efforts and for tracking registry productivity. Statements of attribution to the registry should be included when results are disseminated and within publications utilizing registry data.
Conclusions
When data are properly collected, clinic-based registries can provide substantial insights into headache, including disease characteristics and heterogeneity, longitudinal outcomes and predictors of such outcomes, treatment safety, tolerability and effectiveness, healthcare resource utilization, and quality of care. The general principles for registry planning, governance, data collection, quality assurance, human subject and data protection, and data sharing outlined in this International Headache Society guideline can assist investigators with meeting the goals for their clinic-based headache registry.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TJS: Within the prior 24 months, TJS has received personal income from Abbvie, Allergan, Biodelivery Science, Biohaven, Click Therapeutics, Eli Lilly, Equinox, Ipsen, Lundbeck, Novartis, and Satsuma. He holds stock options in Aural Analytics and Nocira and has received royalties from UpToDate. He has received research funding from American Migraine Foundation, Amgen, Henry Jackson Foundation, National Institutes of Health, Patient Centered Outcomes Research Institute, SPARK Neuro, and the United States Department of Defense.
CT: Within the last 2 years, CT received fees from Allergan/Abbvie, Eli-Lilly, Lundbeck Novartis, Teva, and WebMD for participating in advisory boards or for lecturing. Her Institute has received income for clinical trials sponsored by Allergan/Abbvie, Eli-Lilly, Lundbeck, Novartis, and Teva. CT has no ownership interest and does not own stocks of any pharmaceutical company. She has received research funding from ERA-NET schemes, Migraine Research Foundation, and the Italian Ministry of Health.
SDS received honoraria for participation in clinical trials, contribution to advisory boards or oral presentations from: AEON, Amgen, Cambridge University Press. Clexio, Cooltech, Ctrl M, Allergan (Abbvie), Biohaven, Impel, Lundbeck, Promius, eNeura, Equinox, Medlogix Communications, Lilly, Lundbeck, Miller Medical Communication, Novartis, Promius Satsuma, Theranica, Nocira, Oxford University Press, Revance, TEVA, WebMD Health/Medscape, Wolters Kluwer.
CLS has received research/grant support from Miles for Migraine, the National Institutes of Health/National Institute of Neurological Disorders and Stroke (K23 NS102521), and the US Food and Drug Administration (1U18FD006298). CLS or her institution have received compensation for her consulting work for Allergan/AbbVie Inc; Impel NeuroPharma, Inc; Eli Lilly; Lundbeck; Teva Pharmaceutical Industries Ltd; and Upsher-Smith Laboratories, LLC.
TK: received outside of the submitted work research grants from the German Joint Committee and the German Ministry of Health; personal compensation form Eli Lilly & Company, Teva, TotalEnergeis and the BMJ.
PPR: within the last 24 months, PPR has received honoraria as a consultant and/or speaker for AbbVie, BCNPeptides, Biohaven, Chiesi, Eli Lilly, Lundbeck, Medscape, Neurodiem, Novartis, Pharvaris, Praxis, and Teva Pharmaceuticals. Her research group has received research grants from AGAUR, EraNet Neuron, FEDER (RIS3CAT), la Caixa foundation, International Headache Society, Instituto Investigación Carlos III, AbbVie, Novartis, Teva Pharmaceuticals; and has received funding for clinical trials from AbbVie, Amgen, Biohaven, Eli Lilly, Lundbeck, Novartis and Teva. PPR serves as an Associate Editor for Cephalalgia, Headache, Frontiers in Neurology, Neurologia and Revista de Neurologia. PPR is the Honorary Secretary of the International Headache Society and serves as a member of the Clinical Trials Committee and the Scientific Committee of the IHS. She is the founder of ![]() PPR does not own stocks from any pharmaceutical company.
PPR does not own stocks from any pharmaceutical company.
FMA received honoraria as an advisory board member and speaker for Lundbeck, Novartis, Eli Lilly and Teva. FMA serves a member of the national headache guidelines committee in Denmark.
RBL receives research support from the NIH, FDA as well as the National Headache Foundation and the Marx Foundation. He also receives research support from Allergan/Abbvie, Amgen, Eli Lilly and Electrocore. He receives personal fees as a consultant or advisor from Allergan/Abbvie, Amgen, Biohaven Holdings, Dr. Reddy’s, GlaxoSmithKline, Grifols, Impel NeuroPharma, Novartis, Lundbeck, Merck, and Teva Pharmaceuticals. He holds stock or options in Biohaven Holdings and CtrlM Health.
DWD reports the following conflicts within the past 36 months: Consulting: AEON, Amgen, Clexio, Cerecin, Cooltech, Ctrl M, Allergan, Alder, Biohaven, Linpharma, Lundbeck, Promius, Eli Lilly, eNeura, Novartis, Impel, Satsuma, Theranica, WL Gore, Nocira, XoC, Zosano, Upjohn (Division of Pfizer), Pieris, Praxis, Revance, Equinox. Honoraria: CME Outfitters, Curry Rockefeller Group, DeepBench, Global Access Meetings, KLJ Associates, Academy for Continued Healthcare Learning, Majallin LLC, Medlogix Communications, MJH Lifesciences, Miller Medical Communications, Southern Headache Society (MAHEC), WebMD Health/Medscape, Wolters Kluwer, Oxford University Press, Cambridge University Press. Research Support: Department of Defense, National Institutes of Health, Henry Jackson Foundation, Sperling Foundation, American Migraine Foundation, Patient Centered Outcomes Research Institute (PCORI). Stock Options/Shareholder/Patents/Board of Directors: Ctrl M (options), Aural analytics (options), ExSano (options), Palion (options), Healint (Options), Theranica (Options), Second Opinion/Mobile Health (Options), Epien (Options/Board), Nocira (options), Matterhorn (Shares/Board), Ontologics (Shares/Board), King-Devick Technologies (Options/Board), Precon Health (Options/Board). Patent 17189376.1-1466:vTitle: Botulinum Toxin Dosage Regimen for Chronic Migraine Prophylaxis.
MA is a consultant, speaker or scientific advisor for AbbVie, Allergan, Amgen, Eli Lilly, Lundbeck, Novartis, and Teva, principle investigator for AbbVie/Allergan, Alder, Amgen, Eli Lilly, Lundbeck, Novartis and Teva trials. MA has no ownership interest and does not own stocks of any pharmaceutical company. MA serves as associate editor of Cephalalgia, associate editor of the Journal of Headache and Pain, and associate editor of Brain.
HCD received honoraria for participation in clinical trials, contribution to advisory boards or oral presentations from: Lilly, Novartis, Pfizer, Teva and WebMD. The German Research Council (DFG), the German Ministry of Education and Research (BMBF) and the European Union support his headache research. HCD serves on the editorial boards of Cephalalgia and Lancet Neurology. HCD is member of the Clinical Trials Committee of the IHS.
GMT reports consultancy support from Novartis, Allergan, Lilly, and Teva, and has received royalties from UpToDate. She has received independent support from the Dutch Organization for Scientific Research, the Dutch Heart & Brain Foundations, IRRF and Dioraphte.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: CLS’s time working on this manuscript was supported by NIH K23 NS102521.
