Abstract
Objective
To assess cervical musculoskeletal impairments during the 4 phases of a migraine cycle in episodic migraine patients, controlling for the presence of concomitant neck pain.
Methods
Differences in cervical musculoskeletal impairments were assessed during the 4 migraine phases in episodic migraine patients and compared with healthy controls controlling for concomitant neck pain. Cervical musculoskeletal impairments were assessed as follow: cervical active range of motion; flexion rotation test; craniocervical flexion test and calculation of activation pressure score; the total number of myofascial trigger points in head/neck muscles; the number of positivevertebral segments (headache’s reproduction) during passive accessory intervertebral movement; pressure pain thresholds over C1, C2, C4, C6 vertebral segments bilaterally, trigeminal area, hand, and leg. Signs of pain sensitization were assessed by evaluating mechanical pain threshold over trigeminal area and hand, pressure pain thresholds, and the wind-up ratio. The Bonferroni-corrected p-value (05/4 = 0.013) was adopted to assess the difference between groups, while a p-value of 0.05 was considered significant for the correlation analysis.
Results
A total of 159 patients and 52 controls were included. Flexion rotation test and craniocervical flexion test were reduced in all 4 phases of the migraine cycle versus healthy controls (p < 0.001). The number of myofascial trigger points and positive vertebral segments was increased in all 4 phases of the migraine cycle versus healthy controls (p < 0.001). Flexion, extension, and total cervical active range of motion and cervical pressure pain thresholds were reduced in episodic migraine in the ictal phase versus controls (p < 0.007) with no other significant differences. Outside the ictal phase, the total cervical active range of motion was positively correlated with trigeminal and leg pressure pain threshold (p < 0.026), the number of active myofascial trigger points and positive positive vertebral segments were positively correlated with higher headache frequency (p=0.045), longer headache duration (p < 0.008), and with headache-related disability (p = 0.031). Cervical pressure pain thresholds were positively correlated with trigeminal, hand, and leg pressure pain threshold (p < 0.001), and trigeminal and leg mechanical pain thresholds (p < 0.005), and negatively correlated with the wind-up ratio (p < 0.004).
Conclusion
In all phases of the migraine cycle, independent of the presence of concomitant neck pain, episodic migraine patients showed reduced flexion rotation test and craniocervical flexion test and an increased number of myofascial trigger points and passive accessory vertebral segments. These impairments are correlated with enhanced headache duration, headache-related disability, and signs of widespread pain sensitization. Reduction in active cervical movement and increased mechanical hyperalgesia of the cervical was consistent in ictal episodic migraine patients and the subgroups of episodic migraine patients with more pronounced widespread sensitization.
Glossary of abbreviations
APS = activation pressure score; AROM = active range of motion; CCFT = cranio-cervical flexion test; CMIs =cervical musculoskeletal impairments; CNP =concomitant neck pain; CSI = central sensitization inventory; EM = episodic migraine patients; FRT = flexion rotation test; HDI-E = headache-related disability emotional; HDI-P = headache-related disability physical; MPT = mechanical pain threshold; MTrPs = myofascial trigger points; NDI: neck disability index; PAIVMs = passive accessory intervertebral movements; PPT = pressure pain thresholds; WUR = wind-up ratio;
Introduction
Migraine is a neurological disorder characterized by a cyclic increased activation of brain areas, leading to transitory headache attacks (1). Although headache is the most common, a broader spectrum of symptoms is present in patients with migraine (2). As one of the most frequent concomitant symptoms is neck pain (3,4), the role of cervical musculoskeletal impairments (CMIs) has been widely studied in migraine patients (5–8), leading to the conclusion that this subgroup of patients presents a higher prevalence of CMIs than healthy controls (9,10). However, it is still unclear which mechanism underly the presence of CMIs in the migraine population and their relation with migraine pathophysiology. On the one hand, CMIs could be considered comorbidity, a sign of concomitant neck pain, and a musculoskeletal-related headache that could be misdiagnosed as migraine (11,12). On the other hand, CMIs could be considered a physical manifestation of migraine that would be present independent of the presence of concomitant neck pain (13).
This study aimed to assess features underlying the presence of CMIs in migraine patients by evaluating their prevalence in EM patients during the 4 phases of the migraine cycle and controlling for the presence of concomitant neck pain. CMIs could be considered as: 1) comorbidity if their prevalence is not increased in episodic migraine (EM) patients in any phases of the migraine cycle independent of the presence of concomitant neck pain; 2) a transitory physical manifestation of migraine if their prevalence is increased in EM patients during a migraine attack independent of the presence of concomitant neck pain but not in other phases of the migraine cycle; 3) a persistent physical manifestation of migraine if their prevalence is increased in EM patients in every phase of the migraine cycle independent of the presence of concomitant neck pain. The latter hypothesis would be strengthened if a more increased prevalence of CMIs is found in the ictal phase when clinical manifestations of migraine are enhanced.
This study also aims to assess any correlation between CMIs and clinical characteristics of migraine (interval from previous/next attack, headache characteristics, disabilities, and signs and symptoms of sensitization).
Method
Design
This multicenter, cross-sectional, observational study was conducted in the Headache Center of Parma and Genova (Italy) and approved by the Ligurian (244/2018) and Area Vasta Emilia-Nord (18305/2019) regional ethic committee. Headache patients and healthy subjects were assessed between April 2019 and August 2021. All patients signed an informed consent form.
Population
Patients on waiting lists to attend their first visit to the Headache Centers (Genova or Parma) were invited to participate in this study. Men and women aged between 18 and 65 with EM (with and without aura) in the interictal, preictal, ictal, and postictal phases were included. Patients were excluded if they had: any other primary/secondary headache; any other neurologic, psychiatric, rheumatologic, or systemic pathology with medical diagnosis; history of head/neck trauma in the previous year; received cervical/head surgery; received manual therapy in the cervical spine, cervical anesthetic block, or botulin injection in the last six months; changed the prophylactic treatment in the last three months; were unable to speak and understand Italian.
Control participants were defined as healthy subjects with a maximum of two headache episodes per year that did not fulfill the criteria for migraine or any other primary headache type with no family history of primary headaches. The inclusion criteria for the control subjects were the same as the criteria used for migraine patients.
Procedure
The first screening was made by a telephone interview where migraine patients were excluded if they presented any signs of red flags (14) or they reported at least one exclusion criteria. Healthy controls were recruited from university students, hospital or university staff, and the general population. During the examination, one therapist for each recruitment center (SD and MC), blinded to the subject’s diagnosis, performed the assessment (physical examination, questionnaires, and explanation of how to fulfill a diary for the following four weeks). After four weeks from the first evaluation, headache patients were visited by a neurologist who performed a diagnosis of headache according to the International Headache Classification Criteria (15). The neurologist retrospectively assessed the diary and subgrouped patients according to the phase of the migraine cycle in which the first examination was performed (2,16,17)
Interictal: No headache attack occurred in the 48 hours before/after the evaluation Preictal: Headache attack occurred in the 48 hours after the evaluation Ictal: Headache attack during the evaluation Postictal: Headache attack occurred in the 48 hours before the evaluation
Migraine patients who fulfilled criteria to be included both in the preictal and in the postictal group were included in the preictal group if the nearest attack was the one after the evaluation and in the postictal group if the nearest attack was the one before the evaluation (Figure 1).

Flow chart.
Assessments
For each subject, general characteristics were assessed (Table 1). To assess the headache characteristics, we used a daily updated diary where patients recorded the total use of symptomatic drugs and the frequency, intensity, and duration of headache attacks. The headache side, the percentage of patients with aura, and total years lived with the headache were recorded. For those patients with headache during the assessment, the pain intensity during the current headache attack was recorded (Table 2).
General characteristics.

BMI: body mass index; EM: episodic migraine; N: number; SD: standard deviation
Headache characteristic, disability, and sensitization.

CSI: central sensitization inventory; EM: episodic migraine; g: grams; HDI-E: headache disability index emotional; HDI-P: headache disability index physical; kPa: kilopascal; MPT: mechanical pain threshold; MwoA: migraine without aura; MwA: migraine with aura; NDI: neck disability index; N: number; NPRS: numeric pain rating scale; PPT: pressure pain thresholds; SD: standard deviation; WUR: wind-up ratio
Cervical musculoskeletal impairments (CMIs)
Validated physical examination tests were used to assess the presence of CMIs (6). Physical tests aimed to assess CMIs were divided into tests aimed to assess the functionality of the cervical spine (cervical musculoskeletal dysfunctions), the presence of referred pain from the head/neck region, and cervical mechanical hyperalgesia.
Cervical musculoskeletal dysfunctions:
Active range of motion (AROM): cervical AROM (extension, flexion, left/right lateral flexion, left/right rotation, and total) was recorded in degrees of movement with the cervical range of motion (CROM) device (7,18,19). Flexion rotation test (FRT): FRT (left, right, and total) was used to record passive mobility of the upper cervical spine in degree using the CROM device (7,20,21). Craniocervical flexion test (CCFT): CCFT was used to assess the function of deep cervical flexors muscles using a pressure biofeedback Unit 20–30 mmHg. Subjects were in a supine position and performed craniocervical flexion in five incremental stages (one stage every 2 mmHg). The mmHg value held for 10 seconds without compensation was recorded as the activation pressure score (APS) (22–24).
Referred pain from head/neck region
Passive accessory intervertebral movements (PAIVMs): the number of positive vertebral segments (headache’s reproduction in control, typical migraine pain’s reproduction in EM patients) was assessed with PAIVMs over to C-0/C-1 and C-2/C-3 vertebral segments bilaterally (7,25,26) Myofascial Trigger Points (MTrPs): The presence of MTrPs was assessed bilaterally in the temporal muscles, masseter muscle, sternocleidomastoid muscle, suboccipital muscles, splenius muscles, and in the trapezius muscle. The total number of active and latent trigger points was recorded (8,27,28).
Cervical mechanical hyperalgesia
Pressure pain threshold (PPT): PPT to hand-held algometry (Somedic AB, Sweden, probe area 1 cm2, 30 kPa/s force increase) were assessed over eight areas of the cervical spine, and the sum of both sides was calculated (29,30): right/left posterior arch of the atlas (C1); right/left articular pillar of the axis (C2); the right/left articular pillar of the fourth cervical vertebral segment (C4); the right/left articular pillar of the sixth cervical vertebral segment (C6)
Concomitant neck pain (CNP)
A standardized questionnaire was conducted to identify subjects with CNP. Patients were asked to answer the following questions, not referring to a migraine attack. They were asked if they had neck pain of low, moderate, or high intensity or no neck pain or if they had neck pain during one of the following daily life activities: personal care, lifting, reading, driving, recreation. Subjects with moderate or high neck pain or subjects with neck pain during at least two daily life activities were categorized as “subjects with CNP”; if not, they were categorized as “subjects without CNP.” This quality criterion was adopted to avoid an episode of not disabling low-intensity neck pain that could frequently occur in the general population (31).
Disability
Headache-related disability was assessed using the emotional (HDI-E; total score 0–52) and the physical (HDI-P; 0–48) headache disability index (32–34). Neck-related disability was assessed using the neck disability index (NDI 0–100%) (35,36). The higher the score, the higher the disability.
Symptoms related to sensitization
The central sensitization inventory (CSI) questionnaire was used to assess symptoms related to sensitization. The higher the score more symptoms related to central sensitization are present (0–100 total score) (37–39).
Signs related to sensitization
Pressure pain threshold. PPTs were assessed over three different areas: trigeminal area (symptomatic side in patients with unilateral migraine; dominant side in patients with side/shift or bilateral migraine and in controls); distal pain-free areas (second metacarpophalangeal joint of the dominant hand; tibialis anterior muscle of the dominant leg).
Mechanical pain threshold (MPT)
MPT was used to assess mechanical pain threshold to 7 pinpricks stimulation (ranging from 0.80 g to 50.1 g nylon filaments) (40) over the following areas: trigeminal area; distal pain-free areas (thenar eminence of the dominant hand).
Wind-up ration (WUR)
The WUR was used to assess the temporal summation of mechanical pinprick pain (50.1 g nylon filament). The subject was instructed to give a pain rating (11-point Numeric Rating Scale) for the first and last stimulus of 10 stimuli. The difference between the pain rating of the ten stimuli series and the pain rating of the first stimulus was calculated (41). WUR was assessed over the trigeminal area. A positive WUR was a sign of increased temporal pain summation.
Detailed of the assessment procedure are reported in the appendix (Appendix 1).
Statistical analysis
After a pilot study was conducted to calculate the effect size, a sample size calculation was performed using G*Power 3.1. A total of 109 patients were required for the correlations and 185 patients for regression models to achieve a medium effect size (r: 0.30; f2: 0.15) with a power of 90% and an alpha level of 0.05 and 0.013 due to multiple comparison correction. Mean (standard deviation) of assessment results were presented among controls and patients at different phases of the migraine cycle. A linear regression model was used to compare CMIs results of patients at specific migraine phases to controls while adjusting for possible confounders (age, presence of CNP, gender, body mass index, use of preventive pharmacological therapy, and use of acute treatment in the previous 24 hours) and making appropriate transformations when normality assumption was not fulfilled. Spearman partial correlation adjusted for age and headache frequency were assessed between CMIs results and time relative to the last or the next migraine attack in interictal, preictal, and postictal EM patients pooled together. Age-adjusted Spearman partial correlations were calculated between CMIs results and the pain intensity during the current headache attack, headache characteristics, disability, and signs and symptoms related to sensitization in ictal EM. Spearman partial correlations adjusted for age and the time relative to the last/next migraine attack were calculated between CMIs results and headache characteristics, disability, and signs and symptoms related to sensitization in interictal, preictal, and postictal EM patients pooled together. Subjects with one or more missing values were excluded from the correlation analysis. Data were adjusted for age as previous studies found a high correlation between CMIs results and age (42–45), for headache frequency as the underlying headache frequency of each individual is directly related to the probability that they would be observed in a certain headache phase, and for time relative to the last/next migraine attack as PPTs could change in the proximity to a headache attack (46,47). As multiple between-group comparisons were conducted, a Bonferroni-corrected p-value was adopted to assess the difference between groups in the regression model (0.05 divided for the 4 comparisons performed = 0.013). The threshold accepted for statistical significance in the correlation analysis was p < 0.05, and tests of statistical significance were two-tailed. Statistical analyses were performed using the SPSS software (version 24).
Results
After 671 subjects were initially screened, 211 were included for the final analysis to detect differences between EM patients and controls. Then, 159 EM patients were included in the correlation analysis (Figure 1). General characteristics of all included subjects are reported in Table 1, while headache characteristics, disability, and signs and symptoms related to sensitization of EM patients are reported in Table 2.
Cervical musculoskeletal impairments
Ictal EM, compared to controls, had a reduction in extension (p = 0.004), flexion (p = 0.005), and total (p = 0.002) AROM with no significant differences in other active cervical movements. Interictal, preictal, and postictal EM had no significant differences in any active cervical movement compared to controls. Total FRT and APS were reduced in interictal, preictal, ictal, and postictal EM compared to controls (p < 0.001). The number of total MTrPs (active plus latent) and of positive PAIVMs were higher in interictal, preictal, ictal, and postictal EM compared to controls (p < 0.001). Compared to controls, Ictal EM had reduced PPTs over C1 (p = 0.007), C2 (p = 0.002), C4 (p = 0.003), and C6 (p = 0.005) vertebral segments. No differences in PPT over any vertebral segment were found between controls and interictal, preictal, and postictal EM (Table 3; Supplemental material S1).
Linear regression models using MSK impairment results as dependent variables and 10 predictors: age, presence of neck pain, gender, BMI, use of preventive pharmacological therapy, and use of symptomatic drugs in the 24 hours before the evaluation were first included in the models as covariate then four dummy variables (controls against EM patients in each phase) were included.

APS: activation pressure score; AROM: active range of movement, BMI: body mass index; EM: Episodic migraine; FRT: flexion rotation test; kPa: kilopascal; PAIVMs: passive accessory intervertebral movements; PPT: pressure pain threshold; SD: standard deviation; TrPs: trigger points *significant atp < 0.013 vs. Control; †= data were log-transformed for statistical analysis; ‡ data were square-root transformed for statistical analysis.
Correlations
In preictal, interictal, postictal EM pooled together, a significant negative correlation was found between the number of active MTrPs and time relative to the next headache attack (p = 0.034), and a significant positive correlation was found between the number of positive PAIVMs and time relative to the last headache attack (p = 0.014). No other significant correlations were found between time relative to the last/next migraine attack and CMIs (Table 4).
Spearman Partial correlations adjusted for age and headache frequency in preictal, interictal, postictal EM polled together.

APS: activation pressure score; AROM: active range of movement, FRT: flexion rotation test; PAIVMs: passive accessory intervertebral movements; PPT: pressure pain threshold; TrPs: trigger points *: significant two-tailed correlation at p<0.05
In ictal EM, a significant negative correlation was found between total AROM and hand PPT (p = 0.039) and between the total FRT and the pain intensity during the current headache attack (p = 0.037). APS was negatively correlated with headache duration (p = 0.046) and positively correlated with NDI (p = 0.042) and CSI questionnaires (p = 0.037). A significant positive correlation was found between the number of active MTrPs and headache duration (p = 0.005), monthly use of symptomatic drugs (p = 0.036), HDI-E (p = 0.009), and CSI questionnaires (p = 0.046). A significant positive correlation was found between the number of positive PAIVMs and headache frequency (p < 0.001) and monthly use of symptomatic drugs (p = 0.003). A significant negative correlation was found between PPT over C4 and headache duration (p = 0.035). PPT over C1, C2, C4, and C6 were negatively correlated with NDI (p < 0.042), CSI (p < 0.024), and WUR (p < 0.019). PPT over C1, C2, C4, and C6 were positively correlated with trigeminal, hand, and leg PPT (p < 0.001) and trigeminal MPT (p < 0.041) (Table 5).
Age-adjusted Spearman partial correlations in ictal EM.
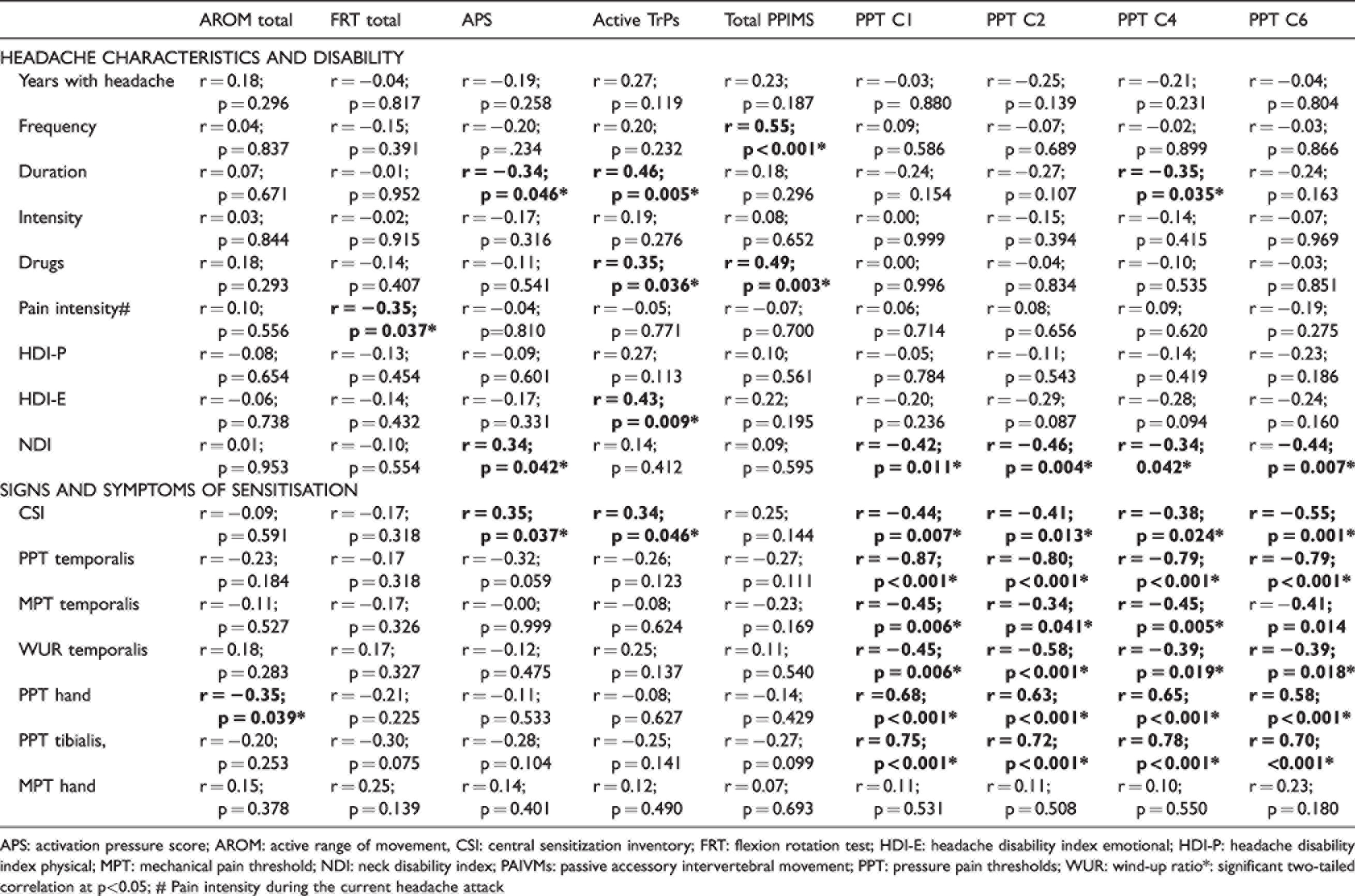
APS: activation pressure score; AROM: active range of movement, CSI: central sensitization inventory; FRT: flexion rotation test; HDI-E: headache disability index emotional; HDI-P: headache disability index physical; MPT: mechanical pain threshold; NDI: neck disability index; PAIVMs: passive accessory intervertebral movement; PPT: pressure pain thresholds; WUR: wind-up ratio*: significant two-tailed correlation at p<0.05; # Pain intensity during the current headache attack
In interictal, preictal, and postictal EM pooled together, the total AROM was positively correlated with trigeminal PPT (p = 0.005) and leg PPT (p = 0.026) and negatively correlated with NDI (p = 0.008). The total FRT was positively correlated with WUR (p = 0.028) and hand PPT (p = 0.049). The number of active MTrPs was positively correlated with headache duration (p = 0.008), HDI-P (p = 0.031), and CSI (p = 0.001) questionnaires. The total number of symptomatic PAIVMs was positively correlated with headache frequency (p = 0.045), headache duration (p = 0.007), and CSI questionnaires (p = 0.014). A significant negative correlation was found between PPT over C4 and headache intensity (p = 0.029). PPT over C1 was negatively correlated with NDI (p = 0.006) and CSI (p = 0.016) questionnaire and was positively correlated with trigeminal, hand, and leg PPT (p < 0.001), and hand MPT (p = 0.003). PPTs over C2, C4, and C6 were negatively correlated with WUR (p < 0.020) and positively correlated with trigeminal, hand, and leg PPT (p < 0.001) and trigeminal and hand MPT (p < 0.025) (Table 6)
Spearman Partial Correlations adjusted for age, time from last headache attack, and time from next headache attack in preictal, interictal, postictal EM polled together.
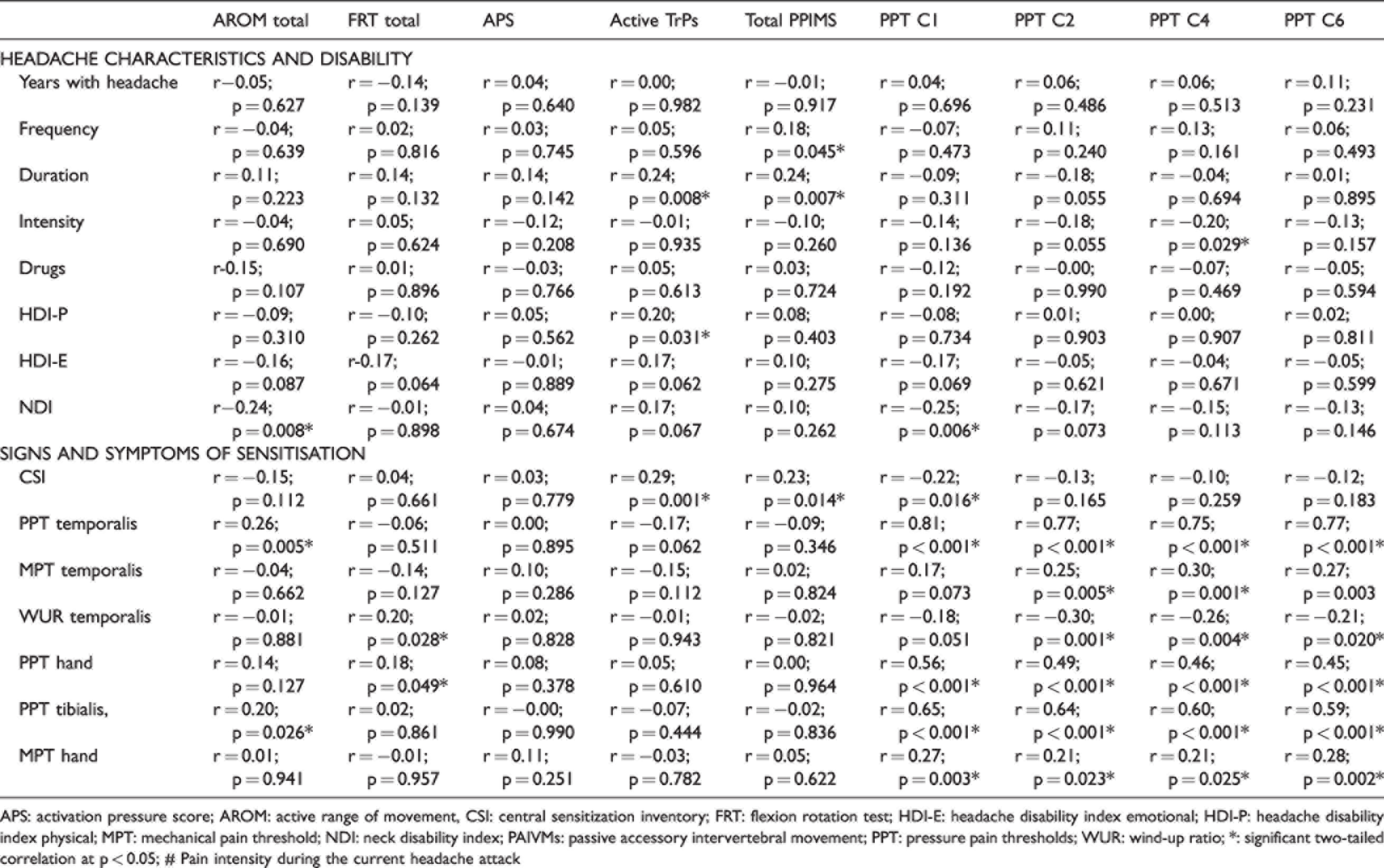
APS: activation pressure score; AROM: active range of movement, CSI: central sensitization inventory; FRT: flexion rotation test; HDI-E: headache disability index emotional; HDI-P: headache disability index physical; MPT: mechanical pain threshold; NDI: neck disability index; PAIVMs: passive accessory intervertebral movement; PPT: pressure pain thresholds; WUR: wind-up ratio; *: significant two-tailed correlation at p < 0.05; # Pain intensity during the current headache attack
Discussion
Patients with EM in all phases of the migraine cycle, independent of the presence of concomitant neck pain, had a reduction in deep neck flexor muscles functionality, in upper cervical-spine passive mobility, and had increased number of myofascial and articular areas able to produce referred pain when stimulated. Restricted cervical active range of motion and reduced cervical pressure pain thresholds were present in EM patients during the ictal phase but not in other headache phases.
Prevalence of cervical musculoskeletal impairments in episodic migraine patients
Musculoskeletal dysfunctions
EM patients had reduced passive mobility and altered cervical motor recruitment during the 4 phases of the migraine cycle independent of the presence of concomitant neck pain, suggesting these dysfunctions were more likely to reflect a physical manifestation of migraine than comorbidity of an independent neck pain problem. As a reduction in active cervical mobility was present in ictal EM but not in EM patients during other phases, a migraine attack could act as a nociceptive input able to impair active cervical mobility. On the one hand, Migraine pain is aggravated by routine physical activity (15), and patients would avoid neck movement during the ictal phase to not increase headache. On the other hand, as experimental pain could alter the motor output in target and not target muscles (48–51), the migraine attack could constitute a nociceptive input able to alter the motor control of neck muscles leading to a reduction in cervical mobility. As the effect of clinical pain on motor output is less consistent (51–53), and no study that we are aware of ever assessed the effect of migraine-like pain on cervical motor output, future studies should assess the mechanisms by which migraine-like pain affects the cervical motor output.
Even if changes in mechanical behaviors occurring during acute pain could have a short-term benefit, they often cause long-term mechanical consequences, as increased muscle stiffness and reduced movement variability (54). Thus, the reduction in upper cervical spine passive mobility (7,9,10,55–57) and the alteration in cervical motor recruitment (7,9,10,58,59) observed in migraine patients outside the ictal phase could be a consequence of changes in mechanical behaviors observed during the headache phase. This will lead to a vicious cycle where 1) a migraine attack could affect cervical mechanical behaviors with a transitory reduction in active movement and a long-lasting impairment in passive mobility and motor control (54). As a consequence, a persistent reduction of neck load tolerability to external mechanical stressors could occur (60,61); 2) thus, it will be more likely that an external mechanical stressor applied to the cervical spine outside a headache attack (i.e., a sustained muscle contraction that could occur during daily life activity), could exceed the neck load tolerability triggering a migraine attack (62) and increasing migraine attack frequency; 3) increase in migraine frequency will further impair cervical mechanical behaviors (57,58,63).
Thus, clinicians should address cervical mechanical behaviors restoring mobility and neck load tolerability to reverse this vicious cycle.
Referred pain from head/neck region
As EM patients have an increased number of myofascial and articular areas that reproduce referred pain when stimulated during all 4 phases of the migraine cycle independent of concomitant neck pain, it is likely that these impairments reflect a physical manifestation of migraine. Even if many studies confirm the increased number of MTrPs in EM patients (7,8,26,64), the relationship existing between MTrPs and migraine is still controversial. On the one hand, the alteration of cervical mechanical behaviors observed in migraine patients could lead to muscle overuse and the formation of MTrPs (65), which could become a nociceptive input able to increase sensitization mechanisms (66–68), exacerbating the headache’s symptomatology (69–72) and further altering the cervical mechanical behaviors (73–78). On the other hand, as referred pain is a central phenomenon that may reflect increased sensitization of spinal and supraspinal neural mechanism (79,80), and migraine patients have been shown to have enhanced sensitization of neurons in the trigeminocervical complex, (81–83) the increased number of head/neck MTrPs could be seen as a physical manifestation of the increased sensitization of the trigeminocervical complex (13,84).
Cervical mechanical hyperalgesia
As cervical mechanical hyperalgesia (reduced PPT) was found, independent of the presence of concomitant neck pain, in ictal EM but not in other phases of the migraine cycle, it could be seen as a psychophysical pain manifestation of a migraine attack. The migraine attack is characterized by increased sensitization of second-order neurons in the trigeminocervical complex (85–88), where ipsilateral and contralateral input from the neck converges (89–92), and the sensitization of those neurons could lead to secondary hyperalgesia in the cervical area (81,93). As previous studies that did not control for the presence of concomitant neck pain found reduced cervical pressure pain threshold in EM in all phases (81,93), cervical mechanical hyperalgesia outside the headache attack could be seen as a sign of concomitant neck pain. Future studies should assess differences in cervical mechanical hyperalgesia between EM patients with and without neck pain.
Cervical musculoskeletal impairments and interval between headache attack
A positive correlation was found between the number of active MTrPs and time to next headache attack suggesting that the number of active MTrPs increases approaching the next headache attack. As referred pain from MTrPs could be considered a central phenomenon that may reflect increased sensitization of spinal and supraspinal neural mechanism (79,80) the number of MTrPs could increase approaching the next headache attack as a sign of increased sensitization mechanisms that occur before the headache attack (46,47).
Cervical musculoskeletal impairments and headache characteristics, disability, and signs and symptoms of sensitization
In ictal EM, the lower the upper cervical spine mobility, the higher the current headache intensity and the lower the muscle functionality, the longer the duration of headache attacks, supporting a relationship between the migraine attack and impairments in cervical mechanical behavior (57,58).
During the 4 phases of the migraine cycle, a higher number of positive PAIVMs and active MTrPs were correlated with worse headache characteristics, worse headache-related disabilities, and higher symptoms related to sensitization. A higher cervical mechanical hyperalgesia was correlated with worse headache characteristics (82), worse disabilities due to neck pain, and worse signs and symptoms of sensitization (94). Thus, physical examination of the cervical spine could be used as a proxy to identify a subgroup of migraine patients worse afflicted by their disease.
Outside the headache attack, the lower active cervical movement was correlated with worse signs of sensitization over multiple areas and worse disability due to neck pain. Thus, the more impaired the cervical mechanical behavior, the more increased the sensitization (95,96) and the more the disability due to neck pain. The presence of a migraine phenotype with concomitant neck pain, lower active cervical mobility, and more widespread sensitization (55) could explain why studies assessing signs of widespread sensitization in migraine patients find heterogamous results (82,93,97–99). Future studies should compare the cervical active range of motion and signs of sensitization between interictal migraine patients with and without neck pain.
Limitation
The population was recruited from a specialized headache center, and over half of the patients were excluded for age, concomitant pathologies, and presence of other headache types. Thus, the external validity of these results should be interpreted with caution.
As no gold standard exist to evaluate the presence of concomitant neck pain in the migraine population, we used a structured interview that was never used before. The percentage of migraine patients and controls with concomitant neck pain were similar to previous studies (31,55), supporting the method's validity.
The blindness of the assessor was not maintained for the entire evaluation of every patient. To reduce the duration of the assessment, we decided to evaluate the trigeminal pain threshold only in one side, incurring the possibility that, for those patients with a unilateral headache on the non-dominant side, the blindness could be lost. Our decision was supported by the fact that the assessor would be blinded regarding the headache type and phase because of the study's design.
As the study has a between-subject cross-sectional design and not a within-subject longitudinal design, comparisons between the different phases of the migraine cycle should be avoided. Thus, differences observed only between one migraine subgroup and healthy controls should not be interpreted as general differences between migraine subgroups.
Conclusion
A large cohort of EM patients in the 4 phases of the migraine cycle had a reduction in the functionality of deep neck flexor muscles, upper cervical-spine passive range of motion, and an increased number of myofascial and articular areas able to reproduce referred pain. Restricted cervical active range of motion and increased cervical pressure hyperalgesia were present in EM patients during the ictal phase but not in other headache phases. As cervical musculoskeletal impairments were present independent of concomitant neck pain, and their prevalence further increased during the migraine attack, they should be considered a physical manifestation of migraine. EM patients with higher cervical mechanical hyperalgesia exhibited higher signs of widespread sensitization and more facilitated temporal summation of pain.
Clinical implications
In all phases of the migraine cycle, EM patients showed a reduction in cervical muscles functionality, upper cervical-spine passive mobility, and an increased number of myofascial and articular areas able to reproduce referred pain. During the ictal phase, EM patients showed reduced cervical active mobility and increased cervical pressure hyperalgesia. EM patients with a higher number of myofascial and articular areas able to reproduce referred pain are worse affected by the disease. EM patients with higher cervical pressure hyperalgesia and lower cervical active range of motion showed higher signs of widespread sensitization
Supplemental Material
sj-pdf-1-cep-10.1177_03331024221082506 - Supplemental material for Cervical musculoskeletal impairments in the 4 phases of the migraine cycle in episodic migraine patients
Supplemental material, sj-pdf-1-cep-10.1177_03331024221082506 for Cervical musculoskeletal impairments in the 4 phases of the migraine cycle in episodic migraine patients by Stefano Di Antonio, Lars Arendt-Nielsen, Marta Ponzano, Francesca Bovis, Paola Torelli, Cinzia Finocchi and Matteo Castaldo in Cephalalgia
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Center for Neuroplasticity and Pain (CNAP) is supported by the Danish National Research Foundation (DNRF121). Francesca Bovis reports personal fees from Eisai, personal fees from Novartis, during the conduct of the study.
Supplemental material
Supplemental material for this article is available online
Appendix 1. Assessment
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
