Abstract
Objective
To evaluate the frequency, distribution, and clinical associations of the dilated appearance of cerebral cortical veins, termed cortical veins sign on T2*-weighted gradient recalled-echo (T2*-GRE) in the acute setting of migraine with aura attack in adult patients.
Methods
We conducted a retrospective analysis of 60 consecutive patients admitted for acute neurological symptoms with a final diagnosis of migraine with aura (42%) or probable migraine with aura (58%) who underwent emergency brain magnetic resonance imaging and 60 non-migrainous control adults. The cortical veins sign was defined as a marked hypo-intensity and/or an apparent increased diameter of at least one cortical vein. We examined the prevalence, the spatial distribution, and the associations of cortical veins sign with clinical characteristics of migraine with aura.
Results
We detected the cortical veins sign in 25 patients (42%) with migraine with aura, compared to none in the control group (p < 0.0001). The spatial distribution of cortical veins sign was characterised by the predominantly bilateral and posterior location. Presence of cortical veins sign was associated with increased severity of aura (p = 0.05), and shorter delay to MRI (p = 0.02).
Conclusion
In the setting of acute neurological symptoms, the presence of cortical veins sign is frequent in patients with migraine with aura and can be detected with good reliability. This imaging marker may help clinicians identify underlying migraine with aura.
Keywords
Introduction
Of all the patients admitted in a stroke unit for acute neurologic deficit, up to 38% are diagnosed at the end as a stroke mimic, mostly migraine with aura (MA) (1).Every clinician knows how difficult it can be to distinguish an aura from an acute ischemic stroke (AIS) or a transient ischemic attack (TIA), especially prior to the headache onset or if it is the first episode (2). Brain magnetic resonance imaging (MRI) might be useful to identify underlying MA in patients with acute persistent or transient neurological symptoms. Numerous studies using magnetic resonance perfusion imaging, PWI (Perfusion Weighted Imaging) or ASL (Arterial Spin Labeling), have documented moderate cerebral hypoperfusion in the posterior brain areas during MA (3–8). Several studies have reported a dilated appearance of the cortical veins detected on T2*-weighted gradient recalled-echo (T2*-GRE) or susceptibility weighted imaging (SWI) in children and adults with MA attack (5,9–16). A recent report has shown a spatial correlation between cortical regions affected by dilated appearance of the cortical veins and clinical symptoms of MA (17) and this radiological aspect seems frequent especially in children (15).
We therefore aimed to evaluate the dilated appearance of the cortical veins on T2*-GRE, termed cortical veins sign (CVS), as a potential imaging marker of MA in the setting of acute neurological symptoms. We assessed the frequency, distribution and clinical associations of CVS in patients admitted for acute neurological symptoms with a final diagnosis of MA who underwent brain MRI within 24 h after symptom onset, compared with control adults without migraine.
Methods
Study design
We conducted a retrospective analysis of data collected prospectively from consecutive patients admitted from May 2014 to December 2015 to Toulouse University Hospital Stroke centre for acute, persistent or transient neurological symptoms.
Standard protocol approvals, registrations, and patient consents
The study was approved by the Institutional Review Board (IRB) (number CRM-2103-145).
Study population
Patients were eligible for the study if they fulfilled the following criteria: i) aged 16–60 years; ii) having been admitted for acute neurological symptoms; iii) having had interpretable brain MRI (including T2*-GRE sequence) within 24 h after symptom onset; iv) with a final diagnosis of MA or probable MA according to criteria 1.2 and 1.5.2 of ICHD 3B (18). Patients were included even if it was theirs first attack (probable MA). Patients were excluded if: i) MRI was of inadequate quality; ii) other cerebral disease that could explain neurological symptoms (e.g. acute stroke, intra or extracranial arterial stenosis, brain tumour) was described in the MRI report.
To exclude the possibility that the dilated appearance of cortical veins was in relation with the variability of cortical veins visualisation on T2*, we formed a control group with the following criteria: i) aged 16–60 years; ii) having been admitted for acute neurological symptoms; iii) no history of migraine with or without aura; iv) who had interpretable brain MRI (including T2*-GRE sequence) within 24 h after symptom onset; v) with a final diagnosis not related to a cerebral involvement including peripheral vertigo, peripheral facial nerve palsy, or non-vascular diplopia established by a neurologist or an ENT specialist.
Eligible patients with MA, and controls were identified from our ongoing prospective observational stroke registry, which included patients with ischemic stroke, transient ischemic attack (TIA), intracerebral haemorrhage and stroke mimics.
Data collection
The medical files were retrospectively evaluated and migraine diagnosis were adjudicated blinded of MR results by a neurologist head of the headache clinic in our academic centre (NF) who did not participate in initial patient management and follow-up. We recorded and analysed demographic and clinical data including age, sex, history of migraine with/without aura, cardiovascular risk factors (hypertension, dyslipidaemia, diabetes, and smoking status). For patients with MA, we also collected information on type of aura symptoms (sensory, visual, aphasia, and motor), symptom duration, delay between the onset of aura and the MRI acquisition, and delay between the end of aura and the MRI acquisition.
MRI acquisition and analysis
MR images were acquired on a 3 Tesla scanner: Phillips Achieva Quasar Dual (Netherlands) (MR1) or Siemens Magnetom Skyra (Germany) (MR2). Study patients have a standardised vascular protocol as part of neurovascular management that included diffusion-weighted imaging (DWI), T2*-GRE, fluid-attenuated inversion recovery (FLAIR) images, and a three-dimensional (3D) time-of-flight (TOF) MR angiography. The T2* sequences were performed with full-head coverage with 32 slices, TR = 516 ms, TE = 20 ms, FOV = 160 mm, section thickness = 3 mm on Philips and with 38 slices, TR = 730 ms, TE = 14,80 ms, FOV = 230 mm, section thickness = 4 mm on Siemens.
By visual analysis, we systematically studied on T2* sequences the entire cerebral venous system, including cortical veins, trans-cerebral veins, and dural sinuses. The presence and extent of the CVS were visually assessed on T2*-GRE by two trained senior raters (GA and AV), independently. T2*-GRE images were anonymised and were randomly analysed to ensure the raters were blind to study group status. The CVS was defined as a marked hypointensity and/or an apparent increased diameter of at least one cortical vein, compared to other cortical veins in remote, homo, or contralateral sulci. In case of disagreement between the two raters, T2*-GRE images were discussed and rated by consensus with a third independent neuroradiologist (FB). We established a visual score of no CVS, mild, or obvious to rate their appearance based on the degree of their T2* low signal intensity, the enlargement of the apparent diameter of veins, and the extent on the brain surface. See Figure 1 for examples of classification of cortical veins’ appearance. Trans-cerebral veins were considered abnormal when they were easily visible as linear hypointensity in deep white matter. For transverse and superior sagittal sinuses, marked hypointensity or an apparent increase of their calibre was considered abnormal.

Examples of classification of cortical veins sign (CVS). Axial T2*-GRE images showing a dilated veins appearance (white arrowhead), rated (a) mild CVS: Hypointense signal concerning several contiguous veins localised in the left temporoparietal lobe, (b) obvious CVS: Deep hypointensity of many cortical veins localised in the left temporal, parietal, and occipital lobes.
To better assess the extent of this abnormality, we further analysed i) the brain lobes affected with a maximum of 10 anatomic territories (five in each hemisphere): The frontal, parietal, occipital, temporo-insular, and temporo-medial regions; ii) the side affected, unilateral when the sign concerned only one hemisphere, or bilateral. When an equal number of territories were involved in each hemisphere, the distribution was qualified as symmetric.
Statistical analysis
Descriptive analyses were performed with frequency and percentage for qualitative characteristics and mean, standard deviation (SD), minimum, and maximum (median and interquartile range) for quantitative characteristics.
Clinical and imaging characteristics of patients with MA were compared with controls using χ2 test for qualitative variables (Fisher’s exact test as appropriate) and Wilcoxon rank-sum test for quantitative variables.
Inter-rater reliability for the presence of the CVS was evaluated using Cohen’s kappa coefficient (and 95% confidence interval [CI]). The kappa coefficient ranges from 0 to 1, where the agreement strength was interpreted using as follows: <0.20, poor; 0.21 to 0.4, fair; 0.41 to 0.6, moderate; 0.61 to 0.80, good; 0.81 to 1, very good.
Within subjects with MA, clinical and imaging characteristics were compared between patients with versus without CVS using χ2 test for qualitative variables (Fisher’s exact test as appropriate) and Wilcoxon rank-sum test for quantitative variables. Next, multivariate logistic regression models were used to identify factors associated with the presence of the CVS. The characteristics tested previously with usual tests and statistically significant at 30% were potential factors for models. Quantitative variables were dichotomised with median for reference. First, these factors were tested in a univariate model with presence of CVS as the response variable and one factor as the independent variable. Then, factors with p < 30% in univariate model were inserted in the multivariate model. The candidate variables thus retained were age (ref: <30 years), gender (ref = male), duration of aura symptoms (ref: <60 min), delay from end of aura to MRI (ref.: <170 min), and multiple aura symptoms (ref: <3). The final model was obtained with a backward elimination strategy. No adjustment was made to any model. The final model contained only factors significant at 5%. SAS statistics software, version 9.4 (SAS Institute Inc., Cary, NC) was used for all analyses. A two-sided p < 0.05 was considered statistically significant.
Results
During the study period, a total of 110 patients were admitted to our stroke centre for acute neurological symptoms attributed to potential MA at discharge. Of those, 32 did not meet the criteria for a confirmed or probable MA after file review, and 18 were not imaged with brain MRI, had MRI of inadequate quality or without T2*, and were excluded. The final cohort consisted of 60 adult patients with MA with available brain MRI performed within 24 h after symptom onset, and 60 non-migrainous controls imaged with MRI within 24 h after neurological symptoms not attributed to a cerebral involvement (peripheral vertigo, n = 37; peripheral facial nerve palsy, n = 16; non-vascular diplopia, n = 7). Thirty five (58%) MA patients had not experienced a previous migraine attack and were classified as probable migraine. The two MR scanners were used equivalently (60 MR1 and 60 MR2) and there was no difference between the MA group (31 MR1 and 29 MR2) and control group (29 MR1 and 31 MR2).
Clinical characteristics
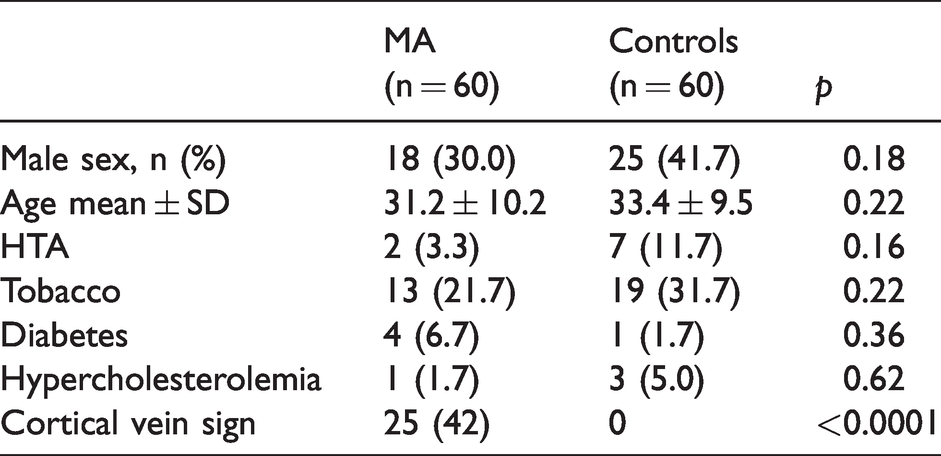
Baseline clinical characteristics of patients with MA compared to controls are summarised in Table 1. Patients with MA and controls were comparable for age, sex, and vascular risk factors. The clinical characteristics of aura and the delay from aura symptoms to MRI for the entire migrainous group and according to the presence of CVS are described in Table 3. Among the 60 patients with MA, 52 had gradually developing symptoms (≥5 min) and 57 had at least one positive aura symptom. The auras were severe and prolonged. The median total duration of aura was 60 min. Only 26% of patients suffered from one single clinical symptom and 28% have three or more aura symptoms. Nine out of 60 (15%) patients reported motor symptoms that were either moderate or subjective. For six patients, the clinical examination was normal on arrival but they reported a sensation of motor impairment in the arm (four patients) or the face (two patients). For the three other patients, clinical examination on arrival revealed a slight facial asymmetry (one patient), a moderate motor deficit of the arm (one patient) and the hand (one patient). All had associated sensory symptoms and five had visual symptoms. For six patients, it was the first episode of MA and three patients had a history of aura but without motor symptoms. Every case was reviewed by our headache clinic director (NF), who concluded that none of them fulfilled the hemiplegic migraine diagnosis criteria and therefore no genetic analysis was performed. The symptoms were clinically related to the left hemisphere in 46 (77%) patients and bilateral in 14 (23%) patients. The median delay from aura onset to MRI was 270 min and 170 min from end of aura to MRI. MRI was performed during aura in seven (11.7%) patients, of whom five had a CVS (71%).
Clinical characteristics and presence of the cortical veins sign in migraine aura patients and non-migrainous controls.
MRI characteristics of the 25 migraine aura (MA) patients with the cortical veins sign (CVS).

Cortical vein sign: Prevalence, distribution, and reliability
We detected the presence of the CVS in 25 patients (42%) with MA compared to none of the controls (p < 0.0001). We observed no venous abnormality affecting trans-cerebral veins or dural sinuses. Inter-rater reliability for the presence of the CVS was good (kappa = 0.78, 95% CI 0.64–0.92). Nine out of 120 (7%) cases were rated by consensus, six in the MA group and three in the control group. MRI characteristics of the CVS are described in Table 2. The CVS was mild in 11 (44%) patients and obvious in 14 (56%). The CVS could be observed in all cerebral lobes and affected several lobes in most patients (at least three of 10 territories in 84% of patients). The CVS predominantly affected the posterior regions of the brain without a good overlap with the posterior cerebral artery territory. The temporo-insular region was affected in 96% of patients with MA and the occipital lobe in 88%, whereas the CVS was detected in the frontal lobe in 28%. The interhemispheric distribution of the CVS was bilateral in 13 (52%) patients, and restricted to the left hemisphere in 12 (48%) patients, but was not restricted to the right hemisphere in any patient. Also, among the 25 patients with CVS, the symptoms were clinically related to the left hemisphere in 20 (80%) patients and bilateral in five (20%) patients (all with bilateral CVS). In all cases, the cortical area corresponding to the main clinical symptom was involved, but the CVS was often bilateral even if the symptoms were lateralised and more extensive over the cerebral cortex than suggested by clinical symptoms. Figure 2 illustrates a typical example.

Illustrative case of cortical veins sign (CVS) and temporal evolution. Baseline (2 h after aura symptoms) and follow-up (24 h after aura symptoms) MRI in a 18-year-old woman with history of migraine presenting with visual positive symptoms followed by mild aphasia. On baseline T2*-GRE ((a), (b), (c)), CVS appeared as dilated hypointense veins (rated mild) in the temporal and occipital lobes (white arrowhead). Follow-up T2*-GRE ((d), (e), (f)) demonstrated disappearance of CVS.
Factors associated with the presence of the cortical vein sign
The CVS detection rate in the MA group was comparable using either the MRI1 or the MRI2, respectively 14/31 (45%) and 11/29 (38%) (p = 0.57). In the univariable analysis, the presence of the CVS was associated with male sex (p = 0.05), longer duration of aura symptoms (p = 0.04), multiple aura symptoms (n > 2) (p = 0.02), shorter time from aura symptom onset to MRI as well as end of aura to MRI (p = 0.004) (Table 3).
Clinical characteristics of the entire migraine aura (MA) group and according to the presence of the cortical veins sign (CVS). Statistically significant results in bold.

In multivariable analysis, the presence of CVS was independently associated with the presence of multiple (n > 2) aura symptoms (odds ratio [OR] 3.57, 95% CI 1.06–13.19; p = 0.05) and a shorter time from the end of aura symptoms to MRI (OR 0.26, 95% CI 0.08–0.78; p = 0.02).
The prevalence of the CVS was 68% in patients with MA imaged during aura or within 2 h after end of symptoms compared to 43% when MRI was performed between 2 and 4 h, and 22% in patients imaged beyond 4 h (Figure 3). Three patients had a control MRI the next day, which was normal.

Prevalence of the CVS according to the time from the end of aura symptoms to brain MRI (p = 0.01; global χ2 test).
Discussion
The results from this study in adults with migraine with aura attacks, imaged with MRI within 24 h after aura symptoms onset revealed several important findings. A dilated appearance of cortical veins, termed cortical veins sign (CVS) was detected on T2*-GRE in up to 42% of patients with MA, and reached 68% when MRI was performed within 2 h after symptom onset. The spatial distribution of the CVS was characterised by the predominance of the posterior regions of the brain, involving usually several cerebral lobes, exceeding the boundaries of the arterial vascular territories, and harboured a bilateral pattern distribution in half of patients with CVS. This imaging marker was easily identified on clinical brain MRI with good reliability. Taken together, our findings suggest that the CVS may help clinicians identify underlying MA in the setting of acute neurological symptoms.
As previously reported (9–17,19), the CVS is defined by a dilated and hypointense appearance of the cortical veins on T2* sequence, contralateral to symptoms of aura or bilateral. This cortical venous aspect in T2* was also described during acute ischemic stroke (AIS) (20–22). However, in MA, contrary to AIS, CVS never involved the trans-cerebral veins, had no arterial territory systematisation, and was not associated with vessel abnormality or abnormal parenchymal signal. Our series allowed its frequency and radiological characteristics to be described more precisely. We found a CVS frequency of 42%, more than twice that in the only other series in adults (16), which is probably explained by the rapid completion of MRIs as part of emergency stroke management. Hence in our study, delays between symptoms and MRI were shorter than in previous studies (16) (median delay: 270 [180–497]), and CVS prevalence was strongly associated with the delay of MRI acquisition, frequent when MRI is performed during aura or in the first 2 h following the end of aura (68%), then quickly decreasing. Conversely, two paediatric retrospective series using SWI sequences reported a higher frequency of prominent hypointense veins, 58% and 62%, respectively (5,15). In the largest (15), age was inversely correlated with SWI abnormalities, which may explain the lower relative frequency in our population of young adults, despite shorter delays. The CVS therefore seems to be a frequent radiological sign in adults during MA attacks, if MRI could be performed during the aura or the following hours.
The radiological topography of CVS was remarkable in several regards. First, it did not respect the arterial vascular territories, and overlapped onto adjacent areas as previously described. Interestingly, it followed the same distribution of the perfusion abnormalities previously described during MA (6–8). Second, the occipital and temporo-insular areas were almost invariably affected. This is, in our experience, a typical radiological pattern regardless of aura symptoms (see Figure 2). Third, the CVS was bilateral in half of the cases (52%), while the aura symptoms were unilateral in 80% of cases. Bilateral venous abnormality on SWI sequences was previously reported in only 8.2% of paediatric patients with aura (15). One explanation could be that we used T2* images and not SWI. Using SWI, every cortical vein is noticeable and abnormalities are scored using a scale of asymmetry. Patients with bilateral but asymmetric prominent veins can be rated as unilateral with SWI and bilateral with T2*.
In current medical practice, MA diagnosis is defined on clinical criteria by ICHD (18). Brain imaging is sometimes performed to exclude potential differential diagnoses (23). Through this sign, an early MRI performed for the evaluation of acute neurological symptoms offers an imaging argument to support the diagnosis of MA. The CVS was visible on a sequence systematically realised in the exploration of a focal deficit by MRI, does not require gadolinium-based contrast agent injection, and is easily recognisable with a good inter-observer kappa. In addition to clinical data, CVS contributes to a positive diagnosis of MA and spares patients from potentially dangerous treatment and unnecessary etiological investigations. More generally, our study supports the yield of MRI for acute neurological deficit management, for stroke, but also and particularly for diagnoses of stroke mimics.
T2* weighted MRI sequences are performant in visualising venous structure using paramagnetic properties of various iron forms of haemoglobin (24,25). Deoxyhaemoglobin contained in the veins increases magnetic susceptibility, indirectly reflecting blood deoxygenation levels and an increase in oxygen extraction (26). An appearance with prominent veins in T2* has been mostly reported in the critical situation of cerebral hypoperfusion during an ischemic stroke due to proximal occlusion (27–29). As in AIS, during migraine aura, a dilated appearance of cortical veins is probably related to an increase in oxygen extraction, reflecting cerebral hypoperfusion. Several studies with MRI perfusion (6,7), ASL (8) and BOLD MRI (31) on human brains showed cerebral hypoperfusion during migraine aura. In the study by Cobb-Pitstick et al., using ASL sequences, the seven patients with prominent veins had cerebral hypoperfusion in the same areas. The good correlation between areas of SWI abnormality and perfusion maps seems most evident using perfusion TTP imaging (7,15).
The CVS reflecting prolonged hypoperfusion without ischemic lesion is concordant with the current models of MA focused on the cortical spreading depression (CSD) phenomenon (32,33). It is established that CSD generates several major cerebral hemodynamic modifications. In the last stage after a CSD, a prolonged oligemia with distal vasoconstriction for multiple hours is described (30,33,34). In a human brain, it could persist for as long as 6 h (35). This could explain why even if the MRI is performed more than 4 h after the end of the aura, the CVS is still visible in 21% of patients in this series. Some characteristics of CVS are less concordant with CSD models: CVS are frequently bilateral or concern large territories, sometimes the whole hemisphere (extension to the frontal lobe in 28% of patients). These areas are more extensive than suggested by clinical symptoms, and classically, CSD (4) does not cross major sulci. These findings raise the question of whether the hypoperfusion found is solely related to the CSD. Moreover, some studies also demonstrated bilateral posterior hypoperfusion in migraine without aura (36–38). One hypothesis is that the primary event could be an extensive oligemia triggered by the activation of brainstem nuclei in migraine both with and without aura; this hypoperfusion could generate a CSD in some patients, worsening this pre-existing hypoperfusion in posterior areas and generating clinical symptoms of aura.
Our study has some limitations. First, this is a retrospective clinical study of patients with migraine with aura attacks, with a control group of patients having neurological symptoms not involving the CNS. This design allows the pathological nature of the CVS to be confirmed and the characteristics of the sign in this population to be described, but it is impossible to exclude that such radiologic patterns could be described in the case of transient ischemic attack (TIA) without diffusion lesions or post-epileptic deficit. The specificity of this sign will have to be studied in a population of TIA and stroke mimics. Secondly, we recruited our patients from the stroke unit, which can lead to the recruitment of patients with more severe or worrying symptoms of aura. This could be an explanation for the high frequency of aphasic symptoms and left hemisphere involvement. We cannot exclude that for less severe MA, the CVS would be less frequent. The high rate of MA patients presenting with motor symptoms might be explained by the fact that the onset of an acute motor deficit is part of our triage selection criteria to organise a direct referral to our acute stroke unit, which might have created a selection bias. The third limitation is that our patients did not have a systemically associated multimodal MRI exploration with perfusion sequences, which would have been useful for a better understanding of underlying mechanisms. If our results suggest that CVS might be associated with MA, its prevalence in other causes of transient neurological focal deficit remains to be defined in order to establish the yield of CVS to guide the etiological diagnosis of transient neurological symptoms. To address this question, we have run a prospective cohort study (Interest of the T2 * Sequence in MRI for the Diagnosis of Migraine Aura in the Acute Phase. (MARIE) NCT03689361), in which 60 consecutive patients referred for transient focal neurological symptoms were systematically evaluated by multimodal MRI including T2* and perfusion imaging. Discharge diagnoses were compared to the results of acute MRI assessed by independent investigators blinded of clinical history.
Conclusion
The cortical veins sign, a dilated appearance of cortical veins on T2*-GRE MRI, is frequent and easily identifiable in the acute setting of severe migraine with aura attack. This imaging marker has a characteristic spatial distribution, and may help clinicians identify underlying migraine with aura attack in the early management of acute neurological symptoms. Further prospective studies are needed to assess the specificity of the CVS for the diagnosis of migraine with aura in patients with transient neurological symptoms of various origins.
Clinical implications
In the acute phase of migraine aura, the cortical veins sign (CVS), a marked hypointensity and apparent increased diameter of cortical veins can be detected on T2*-GRE MRI in 42% of patients. The CVS is associated with increased severity of aura and shorter delay to MRI. This imaging marker can help clinicians identify underlying migraine aura.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: NR was supported by a Fulbright Scholarship and received an Arthur Sachs Scholarship from the Harvard University committee on General Scholarship, and a Philippe Foundation research grant.
