Abstract
Introduction and objective
Monoclonal antibodies targeting the calcitonin gene-related peptide pathway (anti-CGRP mAbs) have shown promising efficacy in randomised clinical trials for the prevention of episodic and chronic migraine, but no head-to-head comparisons with established treatments are available. We aimed to examine absolute differences in benefit-risk ratios between anti-CGRP mAbs, topiramate and propranolol for the prevention of episodic migraine and between anti-CGRP mAbs, topiramate and onabotulinumtoxinA for the prevention of chronic migraine using a likelihood to help versus harm analysis.
Methods
The number of patients needed to be treated for a patient to achieve ≥ 50% reduction in migraine days (NNTB50%) was used as an effect size metric of efficacy. The number of patients needed to be treated for a patient to experience an adverse event that led to treatment discontinuation (NNTHD-AE) was used as a measure of risk. Likelihood to help versus harm values – which are the ratios of NNTH:NNTB – were calculated using data from phase 3 randomised clinical trials.
Results
All agents tested were more likely to be beneficial than harmful (likelihood to help versus harm > 1) with the exception of topiramate at 200 mg per day for the prevention of episodic migraine. Anti-CGRP mAbs in all tested doses had higher LHH values than propranolol or topiramate for episodic migraine and onabotulinumtoxinA or topiramate for chronic migraine prevention. Fremanezumab had the highest LHH ratio in episodic migraine and galcanezumab in chronic migraine.
Conclusion
This analysis showed that anti-CGRP mAbs exhibit a more favourable benefit-risk ratio than established treatments for episodic and chronic migraine. Head-to-head studies are needed to confirm these results.
Keywords
Introduction
Migraine is a chronic brain disease found to be the most common cause of disability below the age of 50 (1) and affecting more than one billion people worldwide (2). Although symptomatic treatment is important, the role of prophylactic treatment, which prevents chronification, is crucial in migraine management. Current prophylactic treatment includes only repurposed drugs, such as anti-epileptics, beta-blockers, anti-depressants, calcium channel blockers and onabotulinumtoxinA (3–5). Recently, disease-specific and mechanism-based treatments for the prophylaxis of migraine became available. Four monoclonal antibodies targeting the calcitonin gene-related peptide pathway (anti-CGRP mAbs) have been tested extensively in several randomised clinical trials (RCTs) for both episodic (EM) and chronic migraine (CM), showing promising efficacy and good safety/tolerability (6,7). The introduction of the anti-CGRP mAbs in migraine treatment raises reimbursement and prioritising issues for health policymakers and practitioners. The clinical dilemma of choosing the right pharmaceutical agent for the right patient, who may also suffer from comorbidities, should ideally be answered with evidence coming from direct head-to-head RCTs along with predictors for treatment response. However, this type of data is not currently available and migraine-related health policies and therapeutic decisions may have to be based on evidence from indirect cross-trial comparisons (8) and data from patient preferences surveys (9).
The number of patients needed to be treated (NNT) to achieve a specific beneficial outcome (NNTB) (e.g. the 50% responder rate in RCTs for migraine prophylaxis) and its counterpart, the number needed to treat to harm (NNTH), are typical effect size metrics and can be used to quantify the effect sizes of clinically relevant benefits and harms in clinical trials (10). NNTB is used as a measure of efficacy with the defined outcome being beneficial and NNTH as a measure of safety with the defined outcome being harmful (e.g. percentage of patients exhibiting any adverse event, a serious adverse event, or drop-out due to an adverse event). Lower NNTB and higher NNTH values are associated with a more favorable treatment profile (e.g. when NNTB is lower than the NNTH, benefits are encountered more frequently than harms) (10). Thus, NNTB, and NNTH can be used as means to communicate easily translatable information to healthcare providers and patients. In this context, the NNTH:NNTB ratio, known as the likelihood to help or harm (LHH) ratio, facilitates the evaluation of the benefit-risk profile of a treatment. The LHH for a specific beneficial treatment response (e.g. the 50% responder rate for migraine trials) versus harmful outcomes (e.g. adverse events (AEs), serious AEs or drop-outs due to AEs) can be compared across treatments and studies (11). In addition, LHH ratios are a tool to present the results of risk-benefit trade-off analyses, and can be used to help health policy makers, clinicians and patients to evaluate potential treatments during decision-making processes (11). Previously published NNTB and LHH values of anti-CGRP mAbs in migraine prophylaxis, have been calculated from phase-2 RCTs data (12,13), providing evidence for a more favorable benefit-risk ratio of anti-CGRP mAbs compared to the established preventive treatments for migraine. In this study, we evaluated the benefit-risk profiles of anti-CGRP mAbs for the prophylaxis of EM and CM in phase 3 trials and compared them with topiramate and propranolol for the prophylaxis of EM or with onabotulinumtoxinA and topiramate for the prophylaxis of CM.
Methods
Search strategy
Two independent reviewers (LK and KD) performed the literature search according to PRISMA recommendations (http://www.prisma-statement.org). Six separate computer-based literature searches were conducted on PubMed and the US National Library of Medicine clinicaltrials.gov, using ‘‘[AMG 334] AND [migraine]”, “[TEV-48125] AND [migraine]”, “[LY2951742] AND [migraine]”, “[ALD403] AND [migraine]”, “[topiramate] AND [migraine]” and “[onabotulinumtoxinA] AND [migraine]” as search phrases, respectively. Electronic publication date was set from 1 January 2000 to 31 May 2020.
Criteria for selection of studies and comparators
Study eligibility for analysis required fulfilment of the following criteria: i) phase-3 randomised controlled trial design for migraine prophylaxis; ii) use of inert placebo as at least one of the comparators; iii) trials of pharmacological agents only; iv) trials that are completed or at least their double-blind phase is completed; v) evaluating the efficacy and safety of tested agents by reporting at least one of the following outcomes: The number or percentage of patients who had at least 50% reduction from baseline in migraine-days per month and the number or percentage of patients who discontinued because of AEs; vi) study drugs administered by oral, intravenous, subcutaneous, or intramuscular routes; vii) studies in which only adult patients (>18 years old) were participating; viii) studies that did not exclusively recruit migraine patients with previous treatment failures; ix) studies with overall low risk of bias according to Cochrane Revised Risk of Bias (RoB-2) tool (14) and x) studies written in English.
The selection of anti-CGRP mAbs comparators was based on three criteria: The quality and size of their clinical evaluation program, the timing of clinical testing, and finally their frequency of use in current clinical practice. Topiramate met most of the criteria for the prevention of EM and CM, as propranolol did for the prevention of EM. For the prevention of CM, onabotulinumtoxinA met all set criteria. In addition, onabotulinumtoxinA is a unique FDA and EMA-approved treatment for CM.
Data extraction
Data were extracted from phase-3, double blind, placebo-controlled studies of anti-CGRP mAbs for both EM and CM, propranolol for EM, topiramate for EM and CM and onabotulinumtoxinA for CM. The number and the proportion of patients with at least 50% response (= 50% responder rate) in migraine days per month, the proportion of patients with any AE, any SAE and the proportion of patients who dropped out due to AEs in the placebo and active treatment arms were extracted from the articles and expressed as percentages. Additional data extracted consisted of the time of assessment of safety and efficacy outcomes, mean patient age, race, mean patient body mass index, mean patient age at the onset of migraine, percentage of females in the trial, disease duration, percentage of patients with aura, percentage of patients having used or still using migraine prophylaxis medication, percentage of patients with history of preventive treatment failure, assessment of migraine mean severity during baseline phase using international scores and scales (e.g. MIDAS, HIT-6).
Definitions
The LHH values were calculated as a measure of benefit for an at least 50% percent reduction in migraine days per month versus the risk of an AE, an SAE or an AE leading to treatment discontinuation (D-AE). In most of the studies included in our analysis, the 50% responder rate was a secondary efficacy endpoint. PREEMPT-1 (15) and PREEMPT-2 (16) RCTs of onabotulinumtoxinA for CM prophylaxis, and one study of topiramate for EM prophylaxis (INTREPID study) (17) did not report this outcome. For onabotulinumtoxinA the number of patients who achieved the 50% responder rate was calculated from the proportion presented in the pooled analysis of PREEMPT-1 and PREEMPT-2 (18). The safety parameters chosen across the studies were the incidence of AEs, SAEs and D-AE.
Statistical analysis: Calculation of NNTs and LHH
The number needed to treat for a patient to achieve at least 50% reduction in their migraine days per month (NNTB50%) was calculated as the inverse of the absolute risk difference between the proportion of patients who achieved a 50% responder rate in the active arm (Iactive) and the proportion of patients who achieved a 50% responder rate on placebo treatment (Iplacebo), (NNTB50% = 1/(Iactive − Iplacebo). Accordingly, the NNT to exhibit an AE (NNTHAE), an SAE (NNTHSAE) and an AE that resulted in discontinuation (NNTHD-AE) were calculated as the inverse of the absolute risk difference between the proportion of patients who experienced the AEs on the active drug, and the proportion of patients who exhibited these AEs on placebo. All NNTB and NNTH values were rounded up to whole numbers. The LHH values were calculated as the ratio of NNTH:NNTB (19). No confidence intervals are reported for LHH ratios, because this metric is not amenable to statistical analysis (see clinical implications). MedCalc software was used to calculate the 95% confidence intervals (95% CIs) of NNTB and NNTH.
Ethics
No ethics approval or patient consent were obtained because all data used in this study were collected from publications with no identifiable patient-level data.
Results
Study and patient characteristics
Using the keywords and inclusion criteria described above, our literature search yielded 2227 records in total, 2091 from PubMed and 136 from the US National Library of Medicine ClinicalTrials.gov (Figure 1). Articles that appeared in both PubMed and clinicaltrials.gov searches are counted as two records. From the 2227 articles retrieved, 20 were included in the final analysis: Six RCTs of anti-CGRP mAbs in EM (20–25), three RCTs of anti-CGRP mAbs in CM (26–28), five topiramate RCTs in EM (17,29–32), one RCT of propranolol in EM (30) two RCTs of topiramate in CM (33,34) and two RCTs of onabotulinumtoxinA in CM (15,16). All studies were published between 2000 and 2020. The characteristics of the studies and patients included in the analysis are shown in Supplemental Tables 1–5.
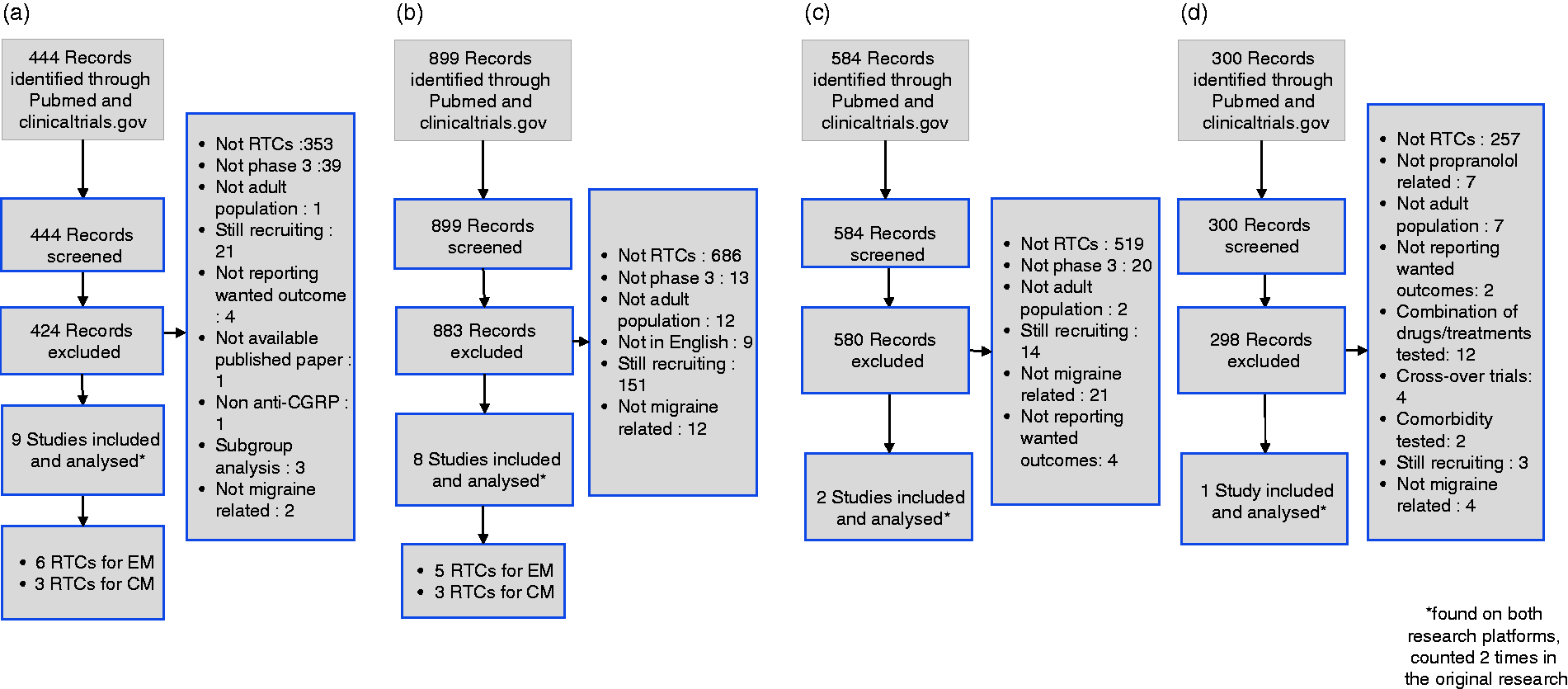
Article selection flow charts (PRISMA). (a) Search for anti-CGRP prophylactic treatment trials for episodic (EM) and chronic migraine (CM); (b) search for topiramate prophylactic treatment trials for EM and CM; (c) search for onabotulinumtoxinA (OBTA), trials for CM; (d) search for propranolol trials in EM.
Efficacy in episodic migraine
All anti-CGRP mAbs displayed similar NNTB50% with 95% CIs ranging from 3–41 in EM (Table 1). NNTB50% of erenumab, fremanezumab, and eptinezumab varied from 5 to 10, 5 to 6 and 6 to 8, respectively. The NNTB50% of galcanezumab was 5 in all studies. The NNTB50% of propranolol was 5 (95% CI: 3–9) and the NNTB50% of topiramate varied from 4 to 18 (95% CIs: 2–59) (Table 2).
Efficacy, safety and benefit/risk outcomes in trials with anti-CGRP mAbs in the prophylaxis of episodic migraine.
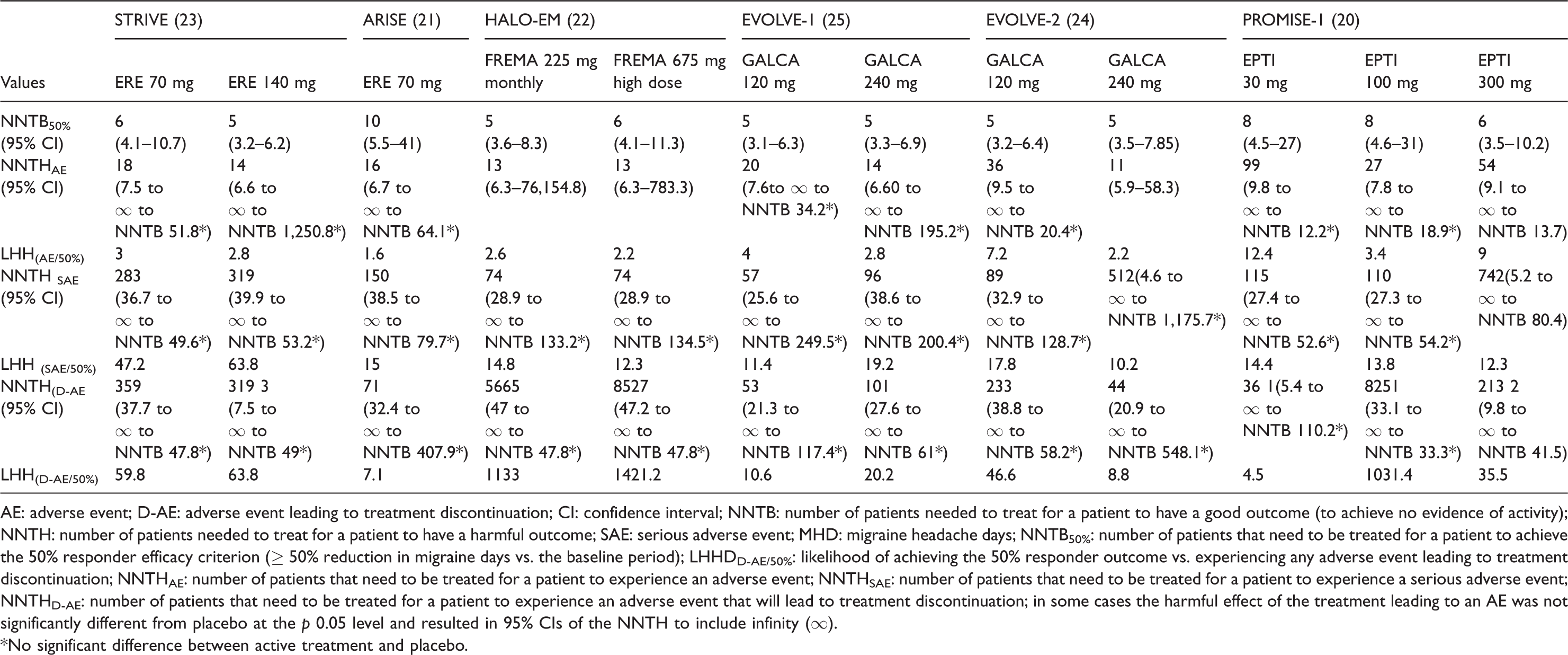
AE: adverse event; D-AE: adverse event leading to treatment discontinuation; CI: confidence interval; NNTB: number of patients needed to treat for a patient to have a good outcome (to achieve no evidence of activity); NNTH: number of patients needed to treat for a patient to have a harmful outcome; SAE: serious adverse event; MHD: migraine headache days; NNTB50%: number of patients that need to be treated for a patient to achieve the 50% responder efficacy criterion (≥ 50% reduction in migraine days vs. the baseline period); LHHDD-AE/50%: likelihood of achieving the 50% responder outcome vs. experiencing any adverse event leading to treatment discontinuation; NNTHAE: number of patients that need to be treated for a patient to experience an adverse event; NNTHSAE: number of patients that need to be treated for a patient to experience a serious adverse event; NNTHD-AE: number of patients that need to be treated for a patient to experience an adverse event that will lead to treatment discontinuation; in some cases the harmful effect of the treatment leading to an AE was not significantly different from placebo at the p 0.05 level and resulted in 95% CIs of the NNTH to include infinity (∞).
*No significant difference between active treatment and placebo.
Efficacy, safety and benefit/risk outcomes in trials with topiramate and propranolol in the prophylaxis of episodic migraine.
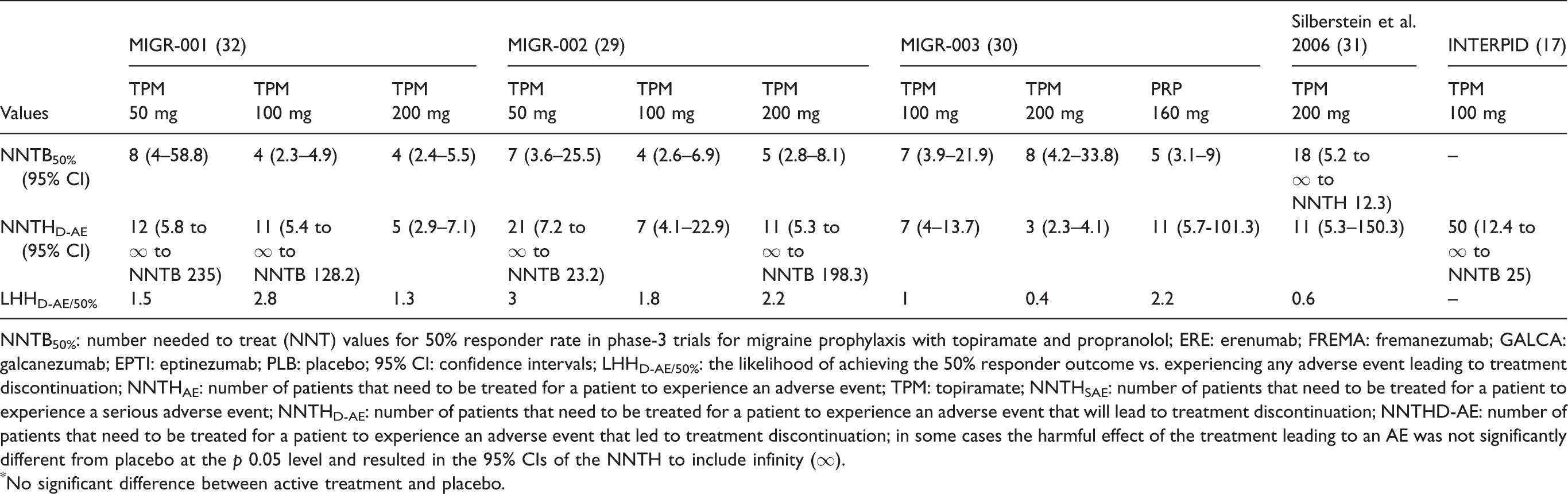
NNTB50%: number needed to treat (NNT) values for 50% responder rate in phase-3 trials for migraine prophylaxis with topiramate and propranolol; ERE: erenumab; FREMA: fremanezumab; GALCA: galcanezumab; EPTI: eptinezumab; PLB: placebo; 95% CI: confidence intervals; LHHD-AE/50%: the likelihood of achieving the 50% responder outcome vs. experiencing any adverse event leading to treatment discontinuation; NNTHAE: number of patients that need to be treated for a patient to experience an adverse event; TPM: topiramate; NNTHSAE: number of patients that need to be treated for a patient to experience a serious adverse event; NNTHD-AE: number of patients that need to be treated for a patient to experience an adverse event that will lead to treatment discontinuation; NNTHD-AE: number of patients that need to be treated for a patient to experience an adverse event that led to treatment discontinuation; in some cases the harmful effect of the treatment leading to an AE was not significantly different from placebo at the p 0.05 level and resulted in the 95% CIs of the NNTH to include infinity (∞).
*No significant difference between active treatment and placebo.
Efficacy in chronic migraine
All anti-CGRP mAbs displayed similar NNTB50% with 95% CIs ranging from 3 to 16 in CM trials (Table 3). The NNTB50% of galcanezumab was 9 and of both fremanezumab and eptinezumab varied from 5 to 6 and 5 to 6, respectively. The NNTB50% of topiramate in studies for CM prevention varied from 4 to 13 (95% CIs: 2–36) (Table 4) and the NNTB50% of onabotulinumtoxinA in studies for CM prevention was 9 (95% CI 6–15) (Table 5). In Figure 2 the efficacy analysis for both EM and CM is presented.
Efficacy, safety and benefit/risk outcomes in trials with anti-CGRP mAbs in the prophylaxis of chronic migraine.

Note: NNTB50%: number needed to treat (NNT) values for 50% responder rate in phase-3 and trials for migraine prophylaxis (only for erenumab, the study was phase 2b).
ERE: erenumab; FREMA: fremanezumab; GALCA: galcanezumab; EPTI: eptinezumab; PLB: placebo; 95% CI: confidence intervals; LHHD-AE/50%: the likelihood of achieving the 50% responder outcome vs. experiencing any adverse event leading to treatment discontinuation; NNTHAE: number of patients that need to be treated for a patient to experience an adverse event; NNTHSAE: number of patients that need to be treated for a patient to experience a serious adverse event; NNTHD-AE: number of patients that need to be treated for a patient to experience an adverse event that will lead to treatment discontinuation; NNTHD-AE: number of patients that need to be treated for a patient to experience an adverse event that led to treatment discontinuation; in some cases the harmful effect of the treatment leading to an AE was not significantly different from placebo at the p 0.05 level and resulted in the 95% CIs of the NNTH to include infinity (∞).
*No significant difference between active treatment and placebo.
Efficacy, safety and benefit/risk outcomes in trials with topiramate in the prophylaxis of chronic migraine.

Note: NNTB50%: number needed to treat (NNT) values for 50% responder rate in phase-3 trials for migraine prophylaxis with topiramate.
AE: adverse event; SAE: severe adverse event; PLB: placebo; 95% CI: confidence intervals; NNTB50% NNT for 50% responder rate; NNTHAE: number of patients that need to be treated for a patient to experience an adverse event; NNTHSAE: number of patients that need to be treated for a patient to experience a serious adverse event; NNTHD-AE: number of patients that need to be treated for a patient to experience an adverse event that will lead to treatment discontinuation.
Efficacy, safety and benefit/risk outcomes in trials with onabotulinumtoxinA in the prophylaxis of chronic migraine.

Note: NNTB50%: number needed to treat (NNT) values for 50% responder rate in phase-3 trials for migraine prophylaxis with onabotulinumtoxinA (pooled analysis, no data from the individual PREEMPT-1 and PREEMPT-2 trials have been published).
AE: adverse event; SAE: severe AE; 95% CI: confidence intervals; NNTB50%: NNT for 50% responder rate; NNTHAE: number of patients that need to be treated for a patient to experience an adverse event; NNTHD-AE: number of patients that need to be treated for a patient to experience an adverse event that will lead to treatment discontinuation.

NNT values for the prophylaxis of episodic and chronic migraine. Number needed to treat (NNT) values for 50% responder rate in phase-3 RCTs for migraine prevention; bars represent 95% confidence intervals. Different bars of the same agent within the trial represent different doses, the lower dose being at the left. ERE: erenumab; FREMA: fremanezumab; GALCA: galcanezumab; EPTI: eptinezumab.
Safety analysis in episodic migraine
AEs were assessed throughout the double-blind phase of all included trials. In several cases the incidence of AEs in the active treatment arm was not significantly different from placebo at the p < 0.05 level of statistical significance and resulted in the 95% CIs of the NNTH to include infinity (∞). In EM trials of anti-CGRP mAbs, the NNTHAE varied from 11 to 99, the NNTHSAE from 51 to 319 and the NNTHD-AE from 36 to 8527 (Table 1). In EM trials with topiramate, the NNTHAE-D varied from 3 to 50. No data were available to calculate the NNTHAE and the NNTHSAE from topiramate and propranolol trials (Table 2). The estimated NNTHD-AE for propranolol in EM was 11 (95% CI 6–101).
Safety analysis in chronic migraine
In CM trials of anti-CGRP mAbs, the NNTHAE varied from 13 to 31, the NNTHSAE from 95 to 4344 and the NNTHD-AE from 58 to 25,854 (Table 3). The estimated NNTHAE for topiramate in CM RCTs varied from 3 to 10 (95% CI 1.6–203), the NNTHSAE from 173 to 13,446 (95% CI 10–84) and the NNTHD-AE from 13 to 21 (95% CI 23–73) (Table 4). The estimated NNTHAE for onabotulinumtoxinA in CM RCTs was 10 (95% CI 6–18), the NNTHSAE 41 (95% CI 22–184) and the NNTHD-AE 38 (95% CI 23–100) (Table 5). Supplemental Table 6 presents the three most common AEs recorded in all studies included in the safety analysis.
LHH analysis in episodic migraine
The LHAE/50% of different anti-CGRP mAbs in EM RCTs varied from 1.6 to 12.4, the LHHSAE/50% from 10.2 to 63.8 and the LHHD-AE/50% from 4.5 to 1421.2 (Table 1). In particular, the LHHD-AE/50% of erenumab, fremanezumab, galcanezumab and eptinezumab varied from 7.1 to 63.8, 1333 to 1421.2, 8.8 to 20.2 and 4.5 to 1031.4, respectively. Eptinezumab had the highest LHHAE/50% (12.4), erenumab the highest LHHSAE/50% (63.8) and fremanezumab the highest LHHD-AE/50% (1421.2). The LHHD-AE/50% of propranolol was 2.2 and the LHHD-AE/50% of topiramate varied from 0.4 to 2.8 (Table 2). Figure 3 presents the LHH values of treatments for EM included in the analysis.

LHH values for the prophylaxis of episodic migraine. The likelihood of achieving the 50% responder outcome versus experiencing any severe adverse event (LHHSAE/50%) values in phase-3 RCTs for prophylaxis of episodic migraine. In the MIG-003 (30) and Silberstein et al. 2006 (31) studies, topiramate did not reach an LHHSAE/50% value ≤ 1 at doses of 100 and 200 mg daily. Different bars of the same agent in a trial represent different doses; the low dose of the agent is represented by the upper bar and the high dose by the lower.
LHH analysis in chronic migraine
The LHHAE/50% of different anti-CGRP mAbs in CM RCTs varied from 1.4 to 5.1, the LHHSAE/50% from 10.6 to 724 and the LHHD-AE/50% from 11.6 to 2872.7 (Table 2). In particular, the LHHD-AE/50% of fremanezumab, galcanezumab and eptinezumab varied from 20.8 to 70, 31.4 to 2872.7 and 11.6 to 2085.8, respectively. Eptinezumab had the highest LHHAE/50% (5.1) and LHHSAE/50% (724) and galcanezumab the highest LHHD-AE/50% (2872.7). In CM trials of topiramate the LHHAE/50% was 0.7 in both trials, the LHHSAE/50% varied from 43.2 to 1034 and the LHHD-AE/50% from 1.6 to 3.2 (Table 4). In CM trials of onabotulinumtoxinA, the LHHAE/50% value was 1.1, the LHHSAE/50% was 4.6 and the LHHD-AE/50% was 4.2 (Table 5). Figure 4 presents the LHH values of treatments for chronic migraine included in the analysis.

LHH values for the prophylaxis of chronic migraine. The likelihood of achieving the 50% responder outcome versus experiencing any severe adverse event (LHHSAE/50%) values in phase-3 RCTs for prophylaxis of chronic migraine. Different bars of the same agent in a trial represent different doses; the low dose is represented by the upper bar and the high dose by the lower.
Discussion
Summary of the results
To our knowledge, this is the first efficacy, safety and benefit-risk analysis of data from phase-3 RCTs of anti-CGRP mAbs, topiramate, propranolol and onabotulinumtoxinA for migraine prophylaxis. All migraine treatments tested were generally more likely to help than harm, as demonstrated by their estimated LHH > 1, with the exception of topiramate at 200 mg per day. Even at a dose of 100 mg per day, which is commonly used in daily practice, the LHH value for topiramate was equal to one in a study controlled with placebo and propranolol (30), indicating that a participant in this study had an equal chance to achieve a 50% reduction in migraine days and/or to discontinue the treatment due to an AE. The efficacy metrics for propranolol were comparable to those of topiramate. The observed magnitudes of the benefit-risk evaluation of the prophylactic anti-migraine treatments, as reflected by the LHH ratios, varied greatly. However, in all cases the benefit-risk evaluation favoured the anti-CGRP mAbs over all comparators for the prophylaxis of both EM and CM.
The calculated NNTB values of anti-CGRP mAbs reflect a moderate to large efficacy comparable to topiramate, propranolol and onabotulinumtoxinA in prophylactic EM and CM trials. On the contrary, the high NNTH values of anti-CGRP mAbs suggest a significantly improved safety profile, indicating an advantage of anti-CGRPs over topiramate, propranolol and onabotulinumtoxinA safety profiles and resulting in higher benefit-risk ratios (LHHs) for the anti-CGRP mAbs compared to topiramate, propranolol and onabotulinumtoxinA. Although both efficacy and safety metrics varied considerably among the four anti- CGRP mAbs, their differences were not statistically significant. Fremanezumab had the highest benefit-risk ratio in all EM trials and galcanezumab had the highest benefit-risk ratio in all CM trials. Overall, the present assessment is indicative of a more favorable benefit-risk profile for anti-CGRP mAbs compared to established prophylactic migraine treatments.
Methodology
Due to significant heterogeneity across RCTs (see limitation section) data from anti-CGRP mAbs trials and the topiramate trials could not be pooled. The NNT, NNH and LHH metrics were therefore presented individually for each RCT. Thus, a pooled analysis of topiramate trials was excluded from our study (35) to avoid potential masking of trends due to a Yule-Simpson effect, as a recent LHH analysis of the pooled topiramate trials (36) failed to show the low benefit-risk values of the two topiramate studies at a dose of 100 or 200 mg per day (30,31). However, pooled data was analysed for onabotulinumtoxinA, as the required data from PREEMPT-1 and PREEMPT-2 have not been published separately (18). Although a similar effect might also be hidden in the pooled analysis for onabotulinumtoxinA, this is less likely because in both PREEMPT-1 and PREEMPT-2 trials the same onabotulinumtoxinA dose was used. Two more published pooled analyses of PREEMPT-1 and PREEMPT-2 trials (37,38) were excluded from this study, because the first reports data for the 50% responder rate of headache days rather than migraine days (37) and the second included phase-2 onabotulinumtoxinA studies (38). The phase 2b study for CM prophylaxis of erenumab (13) was not included in the analysis because it did not fulfil the eligibility criteria. On the other hand, data from the MIG-003 trial (30) was used to assess the efficacy and safety metrics for propranolol, despite propranolol being the active comparator for topiramate and not the treatment in question in this trial, because this trial fulfilled the eligibility criteria. The anti-CGRP trials in patients with previous treatment failures have been excluded from our analysis because they did not fulfil the eligibility criteria.
Comparisons with other benefit-risk studies
In a previous benefit-risk analysis, data from phase-2 RCTs of erenumab, propranolol and topiramate were examined (36). However, data from different sources for topiramate and onabotulinumtoxinA were extracted, avoiding comparisons. Conversely, there are several methodological similarities with these two assessments; for example, variables with comparable clinical relevance were used in both analyses: The NNTB analysis was based on the 50% responder rate and the NNTH analysis on treatment discontinuation due to AEs, both of which are widely accepted and frequently used parameters that inform clinical decision-making. Other safety outcomes such as the occurrence of any AE or SAE were used, which are also clinically relevant (36). Our results are consistent with the above studies. First, the NNTB values of all agents tested were comparable for both EM and CM prophylaxis. Second, robust differences in the NNTH values among the various anti-CGRP mAbs treatments were detected, implying that anti-CGRP mAbs may differ in their safety and/or that the likelihood of observing a discontinuation due to an AE varied significantly depending on trial setting, treatment, or dose. In this context, it is difficult to analyse the reasons why fremanezumab had the best benefit/risk performance in EM and galcanezumab in CM, when the safety criterion was the treatment discontinuation due to AEs. Also, anti-CGRP mAbs displayed different performances depending on the safety criterion; for example, when the safety criterion was AEs, eptinezumab showed the highest LHH in both EM and CM, but when the criterion was SAEs erenumab had a better performance in EM and eptinezumab in CM. Therefore, no particular treatment consistently performed better in all three benefit/risk assessments. However, the anti-CGRP mAbs, as a class treatment and individually, performed significantly and consistently better in all three benefit/risk assessments versus standard anti-migraine treatments.
Clinical implications
In the absence of data from head to head trials, cross-trial comparisons may be used to inform treatment decisions in practice. The LHH values provide balanced information to physicians and patients alike, including evidence of efficacy and safety (10,11). Thus, the findings of the present assessment address some of the clinical needs and concerns related to the use of the anti-CGRP mAbs in migraine prevention; for example, to whom and when these novel treatments should be prescribed. In addition, the remarkably higher NNTHD-AE values of anti-CGRP mAbs reflect their potential for greater treatment adherence, which remains the most common cause of migraine management failure (39). Our evidence suggests that with the advent of anti-CGRP mAbs, treatment selection is more likely to be guided by the risk of intolerance to a prophylactic anti-migraine treatment rather than its risk of efficacy failure. In this context, two RCTs of anti-CGRP mAbs in patients with prior treatment failure have been conducted and several other post-hoc analyses of the subgroup of patients with prior treatment failure in phase-3 RCTs have shown benefit from anti-CGRP mAbs treatment. Along with treatment tolerance and efficacy, it should be noted that in practice additional parameters are considered in therapeutic decision-making, such as comorbidities and patient preferences.
Limitations
Our study has a number of limitations that should be acknowledged. NNT and LHH are metrics for indirect cross-trial comparisons but they are no substitutes for head-to-head trials. The use of NNTB, NNTH, and LHH for cross-trial comparisons assumes homogeneity across trials regarding populations and designs. Although all anti-CGRP mAbs, topiramate, propranolol and onabotulinumtoxinA RCTs included in our study were reasonably similar in terms of demographics, clinical features and study designs, inhomogeneities can be sources of bias. Generally, neither LHH nor NNT values are suitable for statistical analysis. Both are means for communicating efficacy and safety of interventions and drug treatments. Thus, there are several limitations and concerns for their interpretation in clinical practice. In addition, NNTs and LHHs have been estimated from clinical trials, which means that they apply to populations that may differ from those of daily clinical practice and specific time periods depending on the assessment period of the clinical trial. Thus, NNTs are population and time specific and may vary over time, meaning that in chronic diseases like migraine the emergence of AEs may be postponed rather than completely avoided (40). The predictive value of the NNTB, NNTH and LHH metrics is limited to a time frame equal to the duration of the clinical trial from which they have been calculated. Serious AEs may appear for the first time when a medication is already marketed (41) and the incidence and severity of an AE may be estimated more accurately in the post-marketing setting as in the case of hypersensitivity reaction and constipation, which have led to FDA updates of the erenumab label.
Apart from the general potential pitfalls that are inherent to the nature of NNT and LHH analyses, the following limitations specific to our study should also be mentioned. NNTB was based on the 50% responder rate, which was not the primary endpoint in the RCTs included in the analysis. Not all studies assessed the monthly migraine days as primary endpoint, but some assessed the monthly headache days (HALO for CM (28)). In addition, the assessment period at which the 50% responder rate is calculated in the double-blind phase of each of the selected anti-CGRP mAb studies varied between 1 month (month 3 in the ARISE study (21)), 3 months (4th to 6th month in the STRIVE study (23), 1st to 3rd month in HALO-EM (22), HALO- CM (28), PROMISE-1 (20), REGAIN (26) and PROMISE-2 studies (27)), and 6 months (1st to 6th month in EVOLVE-1 (25) and EVOLVE-2 (24)) (Tables 1 and 2). Differences in treatment and assessment periods appear to have significant impact on the total favourable and unfavourable outcomes. The incidence of AEs throughout the studies included in the analysis is reported as a proportion of the total number of patients, without a time variable included. This could pose a limitation in reporting AEs with delayed appearance after exposure to an intervention (42). Finally, no data were available to calculate the NNTH values for AEs and SAEs in topiramate and propranolol trials. Thus, our analysis was focused on tolerability versus efficacy for these treatments, without providing an additional safety analysis.
Conclusions
Our study provides evidence of a superior benefit-risk profile of anti-CGRP mAbs compared to the three standard prophylactic treatments currently available for episodic and chronic migraine (topiramate, propranolol and onabotulinumtoxinA). It also predicts a better adherence profile for anti-CGRP mAbs. Notably, these conclusions are based on comparisons of clinical studies, but new AEs may later emerge in clinical practice. The level of acceptance and inclusion of these novel treatments in migraine patient preferences will soon be explored. Patient satisfaction and quality of life data along with the anti-CGRP mAbs’ promising compliance and safety profile should guide health policymakers and facilitate the reimbursement process of these novel treatments.
Clinical implications
The remarkably higher NNTHD-AE values of anti-CGRPmAbs reflect their potential for greater treatment adherence, which remains the most common cause of migraine management failure (39). With the advent of anti-CGRPmAbs treatment, selection is more likely to be guided by the risk of intolerance to a prophylactic anti-migraine treatment, rather than its risk of efficacy failure. Along with treatment tolerance and efficacy, additional parameters should be considered in therapeutic decision-making in practice, such as comorbidities and patient preferences.
Supplemental Material
sj-pdf-1-cep-10.1177_0333102421989601 - Supplemental material for Anti-CGRP monoclonal antibodies for migraine prevention: A systematic review and likelihood to help or harm analysis
Supplemental material, sj-pdf-1-cep-10.1177_0333102421989601 for Anti-CGRP monoclonal antibodies for migraine prevention: A systematic review and likelihood to help or harm analysis by Konstantina Drellia, Lili Kokoti, Christina I Deligianni, Dimitrios Papadopoulos and Dimos D Mitsikostas in Cephalalgia
Supplemental Material
sj-pdf-2-cep-10.1177_0333102421989601 - Supplemental material for Anti-CGRP monoclonal antibodies for migraine prevention: A systematic review and likelihood to help or harm analysis
Supplemental material, sj-pdf-2-cep-10.1177_0333102421989601 for Anti-CGRP monoclonal antibodies for migraine prevention: A systematic review and likelihood to help or harm analysis by Konstantina Drellia, Lili Kokoti, Christina I Deligianni, Dimitrios Papadopoulos and Dimos D Mitsikostas in Cephalalgia
Supplemental Material
sj-pdf-3-cep-10.1177_0333102421989601 - Supplemental material for Anti-CGRP monoclonal antibodies for migraine prevention: A systematic review and likelihood to help or harm analysis
Supplemental material, sj-pdf-3-cep-10.1177_0333102421989601 for Anti-CGRP monoclonal antibodies for migraine prevention: A systematic review and likelihood to help or harm analysis by Konstantina Drellia, Lili Kokoti, Christina I Deligianni, Dimitrios Papadopoulos and Dimos D Mitsikostas in Cephalalgia
Supplemental Material
sj-pdf-4-cep-10.1177_0333102421989601 - Supplemental material for Anti-CGRP monoclonal antibodies for migraine prevention: A systematic review and likelihood to help or harm analysis
Supplemental material, sj-pdf-4-cep-10.1177_0333102421989601 for Anti-CGRP monoclonal antibodies for migraine prevention: A systematic review and likelihood to help or harm analysis by Konstantina Drellia, Lili Kokoti, Christina I Deligianni, Dimitrios Papadopoulos and Dimos D Mitsikostas in Cephalalgia
Supplemental Material
sj-pdf-5-cep-10.1177_0333102421989601 - Supplemental material for Anti-CGRP monoclonal antibodies for migraine prevention: A systematic review and likelihood to help or harm analysis
Supplemental material, sj-pdf-5-cep-10.1177_0333102421989601 for Anti-CGRP monoclonal antibodies for migraine prevention: A systematic review and likelihood to help or harm analysis by Konstantina Drellia, Lili Kokoti, Christina I Deligianni, Dimitrios Papadopoulos and Dimos D Mitsikostas in Cephalalgia
Supplemental Material
sj-pdf-6-cep-10.1177_0333102421989601 - Supplemental material for Anti-CGRP monoclonal antibodies for migraine prevention: A systematic review and likelihood to help or harm analysis
Supplemental material, sj-pdf-6-cep-10.1177_0333102421989601 for Anti-CGRP monoclonal antibodies for migraine prevention: A systematic review and likelihood to help or harm analysis by Konstantina Drellia, Lili Kokoti, Christina I Deligianni, Dimitrios Papadopoulos and Dimos D Mitsikostas in Cephalalgia
Footnotes
Author contributions
All authors contributed to the study conception and design. Material preparation, literature search, data collection and statistical analysis were performed by KD and LK. The first draft of the manuscript was written by KD and LK and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript. Methodology: DDM and DP; formal analysis and investigation: KD and LK; writing – original draft preparation: KD and LK; writing – review and editing: CD; supervision: DDM and DP.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: KD, LK and CID report no disclosures. DP has received consulting, speaking fees and travel grants from Bayer, Genesis Pharma, Merck, Mylan, Novartis, Roche, Sanofi-Aventis, Specifar and Teva. DDM has received consulting, speaking fees and travel grants from Allergan, Amgen, Bayer, Biogen, Cefaly, Genesis Pharma, GlaxoSmithKline, ElectroCore, Eli Lilly, Merck-Serono, Merz, Mylan, Novartis, Roche, Sanofi- Genzyme, Specifar and Teva.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
