Abstract
Background
Headache in patients with moyamoya disease is an under-addressed topic in the medical literature. Delay in the diagnosis of moyamoya disease or inappropriate treatment of headache could lead to devastating cerebrovascular outcome. With the evolving understanding of moyamoya disease, migraine pathophysiology, and various migraine-specific medications that have become available, it is crucial to provide an updated overview on this topic.
Methods
We searched PubMed for keywords including moyamoya disease, moyamoya syndrome, headache in moyamoya, surgical revascularization, surgical bypass, migraine and moyamoya, and calcitonin gene-related peptide (CGRP). We summarized the literature and provide a comprehensive review of the headache presentation, possible mechanisms, the impact of various surgical revascularizations on headache in patients with moyamoya disease, and the medical management of headache incorporating novel migraine-specific treatments.
Abbreviations
MMD = moyamoya disease
NRS = numeric rating scale
CGRP = calcitonin gene-related peptide
STA-MCA = superficial temporal artery to middle cerebral artery (bypass)
EMS = encephalo-myo-synangiosis
NSAIDs = nonsteroidal anti-inflammatory drugs
Introduction
Moyamoya disease (MMD) is a chronic progressive vasculopathy characterized by bilateral occlusion or narrowing of the supraclinoid, terminal internal carotid arteries and proximal portions of the middle and anterior cerebral arteries, the development of small collateral vessels in the vicinity of the occlusive arterial lesions, and the compensatory development of arterial collaterals supplying the brain via the external carotid artery and/or the vertebrobasilar system (1,2). The abnormal wispy collaterals lead to the characteristic smoky angiographic appearance of older conventional angiogram studies, giving its name “moyamoya,” meaning “puff of smoke” in Japanese (2). When the characteristic vasculopathy is unilateral or when it is associated with an acknowledged predisposing condition, such as sickle cell disease, Down’s syndrome, cranial irradiation, and neurofibromatosis type 1, it is often categorized as moyamoya syndrome (3). The categorization of moyamoya as a disease or syndrome obscures the likelihood that in most cases there is an interplay of environmental factors and genetic susceptibility, so in this paper we will use the term MMD for all. The overall incidence of MMD is reported to be 0.086 per 100,000 person-years in the USA, with the incidence highest among Asian Americans (0.28/100,000 person-years), followed by African Americans (0.13), Caucasians (0.06), and Hispanics (0.03) (4).
The pathophysiology of MMD is not completely understood. It involves hyperplasia of the arterial wall, thickened internal elastic lamina with proliferation of smooth muscle cells (5), resulting in narrowing of the major anterior circulation arteries (6). Ensuing hypoperfusion of brain parenchyma leads to compensatory development of collateral circulation, including vascular angiogenesis and progressive dilation of pre-existing small perforator vessels. As a result, the vessel wall becomes more fragile and prone to developing micro-aneurysm and aneurysmal dissection, increasing the risk of hemorrhage in patients with MMD (5,6).
The most common presentation of MMD is ischemic stroke and transient ischemic attacks, especially in the pediatric population (7). Intracerebral hemorrhage is rarely seen in children but is commonly seen in adults with MMD (1). Headache, seizures, and movement disorders are other commonly reported symptoms in patients with MMD (1,8). Headache is reported in 20 to 76.4% of patients with MMD (1,9,10). However, there is a dearth of literature addressing the description and burden of headache symptoms accompanying MMD. Furthermore, with the advancement of surgical techniques for MMD and the availability of many new migraine-specific treatments over the past 2 years, it is important to provide an updated overview of the headache phenotypes, proposed mechanisms, the medical treatments of headache, and the impact/association of various surgical managements on headache in patients with MMD.
Diagnosis and phenotypes of headache in patients with MMD
Based on the International Classification of Headache Disorders, 3rd edition, Headache attributed to moyamoya angiopathy can be defined as a recurrent headache occurring in temporal relationship to a diagnosis of MMD, in parallel with worsening symptoms or worsening radiographic signs of MMD, or headache that improves after revascularization surgery for MMD (Table 1) (11). In clinical practice, it might be difficult to establish such direct causation in all cases as many patients with MMD might also have primary headache disorders, such as migraine and tension-type headache, given the high prevalence in the general population. However, it remains important to keep in mind that headache could be a presenting symptom of MMD. Additionally, it is clinically important to rule out hemorrhage when patients with MMD develop new or worsening headache, especially in adults. It has recently been reported that de novo intracranial hemorrhage can occur in the previously unaffected non-hemorrhagic hemisphere with an overall annual risk of 2% in patients with MMD who had hemorrhagic strokes in the past, and the risk is higher in patients with choroidal collaterals (12).
Diagnostic criteria for Headache attributed to moyamoya angiopathy (MMA), section 6.8.3 in ICHD-3 (11).

As for the headache characteristics in patients with MMD, a case series reported recurrent migraine-like headache was the initial symptom of MMD for 20% of patients (10). A cohort study investigated headache in Caucasian patients with MMD and reported that 37 out of 55 patients with MMD (67.3%) had headaches. The headache intensity was mild to moderate (average 3.2; median: 3; range: 1–6, using a numeric rating scale of 0 to 10). Patients in this cohort experienced about six headache days per month. Among those patients, 48% had migraine-like headaches, and of those 27% had migraine with aura-like attacks. Most of the aura like attacks were visual or sensory (47%), followed by speech or motor (24%) symptoms. Headache with tension-type features was reported in 27%, and a combination of migraine-like and headache with tension-type features was found in 27% patients. Of note, the majority of patients (73%) reported migraine-like headache either alone or combined with headache with tension-type features (9).
In addition to this cohort study, several case reports included detailed descriptions of headache phenotypes in patients with MMD. An analysis of a 4-month headache diary of a patient diagnosed with MMD described a mixture of probable migraine without aura and probable frequent episodic tension-type headache (13). Others reported headache phenotypes including migraine with aura or hemiplegic migraine (14,15), hemiplegic migraine leading to ischemic infarct (16–18), and cluster headache (19). In all cases, patients had other neurological symptoms in addition to headache, such as paresis, seizures, dysarthria, ptosis, and unilateral restless leg syndrome, which are not common presenting features of primary headache disorders.
Pathophysiology of headache in patients with MMD
The pathogenesis of headache in patients with MMD is complex and likely multifactorial. In this section, we discuss the hypotheses that have been proposed (9,13).
The intracranial pain-sensitive structures include vessels (arteries, venous sinuses) and dura. The trigeminal nerve and its branches travel alongside and innervate major intracranial arteries, including the internal carotid artery, proximal vessels of the circle of Willis, and meningeal vessels including the middle meningeal artery. The trigeminal nerve also innervates the dura itself. Several studies have shown that dilatation or other external stimulation on the intracranial and extracranial arteries produces pain (20,21). MMD involves narrowing of the intracranial arteries, abnormal angiogenesis, and dilation of pre-existing small perforator vessels, either at the skull base or at the leptomeninges. Additionally, MMD is characterized by a high level of cerebrovascular plasticity, as vascular growth factors, including vascular endothelial growth factor and hepatocyte growth factor were highly expressed in patients with MMD (22). Angiopoietin-2, a pro-angiogenic cytokine, was significantly overexpressed on the middle cerebral artery in patients with MMD compared to their hemodynamically matched patients with atherosclerotic disease (23). The dynamic deformation of the vessels could lead to activation of cerebral arterial nociceptors and cause headache. Other than these mechanical perturbations, additional putative hypotheses include the release of angiogenic or neuropeptide substances that may act on perivascular nociceptors during episodes of cerebral ischemia or in response to angiogenesis.
MMD is also characterized by the dilated leptomeningeal collaterals, a result of the compensatory development of trans-dural/trans-cranial anastomosis from the external carotid artery system as a response to the steno-occlusive changes at terminal internal carotid artery (24). Preclinical migraine models demonstrate that stimulating dural or trigeminal ganglion with electrical, inflammatory, or chemical mediators triggers the release of neuropeptides such as calcitonin gene-related peptide (CGRP), nitro-oxide and pituitary adenylate cyclase-activating polypeptide, causing neuroinflammatory responses including mast cell degranulation, plasma protein extravasation, and vasodilation (25,26). Previous human studies suggested stimulation of dura mater near its arteries produces migraine-like headache, nausea and phonophobia (21,25). Recent studies also suggested that dilation of the middle meningeal artery is a surrogate marker for activation of dural perivascular nociceptors, emphasizing the meningeal contribution to migraine pain (25,27). The leptomeningeal collaterals in MMD could stimulate the dural perivascular nociceptors, resulting in activation of the trigeminovascular system, neuroinflammatory responses, and trigger migraine-like headache (9,13).
Another possible cause of headache in MMD is chronic cerebral hypoperfusion, or borderline perfusion, as a result of progressive stenosis and occlusion of the major anterior circulation arteries (9). Microvascular ischemia could also trigger cortical-spreading depression, a mechanism known to cause migraine aura (9). This theory is supported by several case series that showed improvement in headache after augmenting cerebral perfusion by surgical revascularization (9,28,29) and by a clinical case observation that new onset migraine with visual aura resolved after an occipital stroke, indicating that borderline perfusion in the occipital lobe can lower the threshold for developing cortical spreading depression, triggering migraine aura (18).
Additionally, the vascular endothelial damage in MMD and related collagen exposure could also promote platelet aggregation, which not only correlates with the risk of ischemic infarct but also was recently shown to correlate with intractable headache in patients with MMD (30). The theory is supported by study results showing that the platelet aggregation test value is associated with a higher risk of developing intractable headache in pediatric MMD patients, that the risk of developing headache decreased with aspirin administration, and 85% of all patients in the study with intractable headaches improved with aspirin, a medication that suppresses collagen-mediated platelet aggregation (30). Given the small sample size (35 patients), more studies are needed to explore the role of platelet aggregation and headache in MMD.
As described, vascular structural abnormalities in MMD may mechanistically relate to headache; however, not all patients with MMD develop headache. Additionally, many patients with other vascular abnormalities, such as aneurysms or stenosis of the internal carotid artery from atherosclerosis, do not suffer from headache. These observations raise the possibility that other disease-related factors like severity, rate of progression and collateral formation or genetic susceptibility may play an important role in the development of headache in patients with MMD, in addition to the above-mentioned structural abnormalities (18). Furthermore, the chronic stress and anxiety related to MMD could also trigger and worsen headache in genetically and structurally susceptible individuals with MMD.
Surgical management of MMD and its impact on headache
Surgical management to optimize cerebral blood flow by using different bypass techniques and reconstructing the vasculature plays a key role in reducing stroke rate and improving prognosis (3). The surgical techniques can be broadly classified into two major subtypes, direct and indirect revascularization. Direct revascularization involves surgical creation of a bypass between the superficial temporal artery to middle cerebral artery (STA-MCA) and is associated with improvement in focal cerebral perfusion and angiographic abnormalities. However, it can be technically difficult to perform, especially in pediatric patients with small blood vessels (31). Indirect revascularization, aimed to promote the development of collateral vascular networks over time, is preferred in pediatric patients, and it is also often combined with direct bypass. Indirect revascularization requires less surgical expertise, shorter duration of surgery, and has fewer complications than direct revascularization. The main drawback with this approach is the time lag between the procedure and desired improvement in perfusion (32).
The primary goal of surgical revascularization is to improve cerebral blood flow to prevent ischemic and hemorrhagic strokes. However, an array of headache outcomes following surgical revascularization have been reported in both the pediatric (Table 2) (8,15,28,29,33–38). and adult (Table 3) (9,13,18,19,39–43) age groups.
Literature review of headache in MMD in pediatric age group and their outcomes.
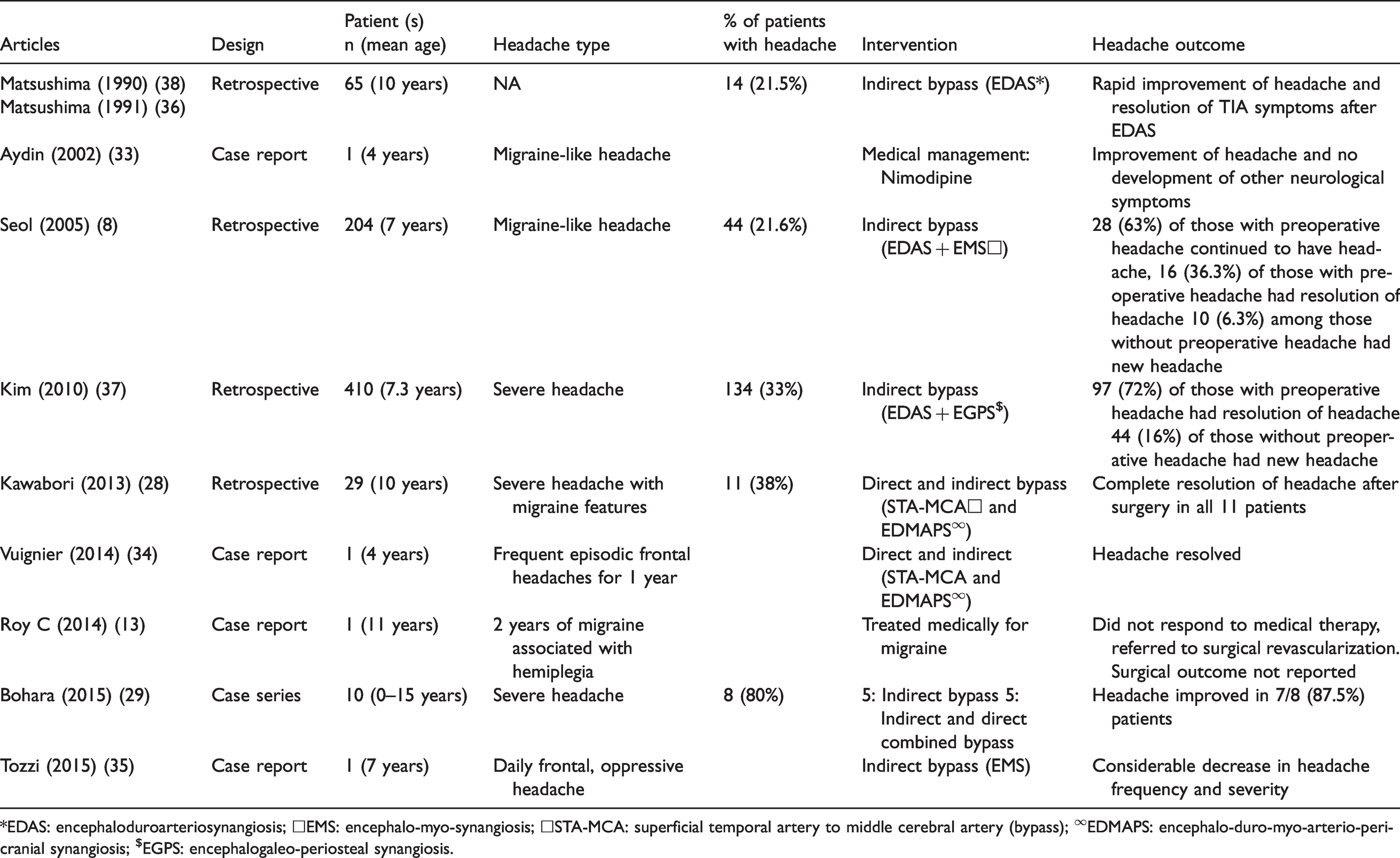
*EDAS: encephaloduroarteriosynangiosis; □EMS: encephalo-myo-synangiosis; □STA-MCA: superficial temporal artery to middle cerebral artery (bypass); ∞EDMAPS: encephalo-duro-myo-arterio-pericranial synangiosis; $EGPS: encephalogaleo-periosteal synangiosis.
Literature review of headache in MMD in adult age group and their outcomes.

STA-MCA: superficial temporal artery to middle cerebral artery (bypass).
Most case series reported improvement of headache after surgical revascularization, both in the pediatric and adult population. Kim et al. reported a series of 410 children with moyamoya disease who underwent indirect revascularization with encephaloduroarteriosynangiosis (EDAS) or EDAS with bifrontal encephalogaleo-/periosteal synangiosis (EGPS) (37). A total of 134 (33%) had headache at presentation, defined as the presence of severe headache that disturbed daily activity, requiring rest or medication, and occurring at least once a month. After indirect bypass surgery, 81% patients had favorable clinical outcome, defined as the symptoms having totally disappeared with no, or mild, fixed neurological deficits. The mean time from surgery to disappearance of transient ischemic attack (TIA) symptoms was 4.3 months (range 0–87). Preoperative headache disappeared in 97 out of 134 (72%) patients with a mean duration of 16 months. Newly developed headache was reported in 16% of the 276 patients without preoperative headache during follow-up. Seizure control was excellent in that 91% patients who were on anti-epileptics were able to successfully withdraw medications after surgery. Of note, 25% of the patients had unfavorable magnetic resonance imaging (MRI) changes after surgery, defined as new formation or an increase in the infarction size. Revascularization of MCA, ACA and PCA territory was good, as opposed to fair or poor, in 34%, 55% and 69% out of 526, 494 and 29 hemispheres. Whether the presence of newly developed headache correlates with unfavorable MRI changes, degree of successful revascularization, location of the affected artery, or other cerebrovascular outcomes were not specifically reported (37). A pediatric cohort of 65 patients reported 14 (21.5%) patients with long-standing headache before surgery experienced near resolution of the headache and other symptoms, including seizures and involuntary movements, after indirect bypass (36,38). In another series of childhood moyamoya (28), 38% (11 of 29) of the patients presented with severe frontal or temporal headaches, and surgical revascularization with combined direct and indirect technique led to improvement of headache in all. These observations support the hypothesis that altered cerebral hemodynamics and cerebral hypoperfusion could explain headache improvement after effective revascularization. Bohara et al. also reported improvement in headache after surgical revascularization in 87.5% of pediatric patients (29). Various other case reports in the pediatric population echoed these findings (Table 2).
Similar observations have been reported in adults. A large retrospective study of 117 adults showed that 25 (21%) patients presented with severe headache; improvement was noted in most patients who underwent direct bypass (STA-MCA) (40). Kraemer et al. also found that out of 55 patients with adult MMD, 37 (67.3%) had headache. Among the 34 patients who received STA-MCA bypass combined with encephalomyosynangiosis (EMSA), 19 (56%) patients reported significant improvement of headache intensity and frequency after surgery. However, nine patients reported new onset of headache following bypass (9). Similar headache improvement was also found in Gao et al. (43) and various other case reports in adults (Table 3) (13,18,19,41,42).
Despite these encouraging surgical outcomes, persistent or newly developed headache after surgery have been reported. In a retrospective analysis of 204 children with MMD who underwent indirect bypass surgery, 44 (21.6%) patients had preoperative headache. Nausea and/or vomiting were present in 12 patients. Persistent postoperative headache was observed in 28 (63%) of the 44 patients with preoperative headache, and new-onset postoperative headache occurred in 10 (6.3%) of the 160 patients without preoperative headache. In these 10 patients without preoperative headache, headache was first noticed at a mean of 15 months after surgery. The headache improved and lasted for <36.1 months in eight patients but lasted for >70 months in two patients. Of the 44 patients with pre-operative headache, the majority (59%) experienced improvement in headache within the first month after surgery. The headache eventually resolved in all but three patients, who experienced persistent headache. The authors reported no significant association between the presence of headache and worsening MRI or single photon emission computed tomography (SPECT) results after surgery, defined as worsening infarct size or perfusion defect, decreased perfusion, and decreased cerebrovascular reactivity to acetazolamide (8). A retrospective study reported that post-operative increase rate of STA diameter was associated with post-operative headache (44). A long-term observational cohort studying 143 patients 21 years old or younger who underwent indirect bypass (pial synangiosis) also reported that headache of varying intensity was common postoperatively (specific numbers were not provided) (45). Though the improvement of headache in most patients could support hypoperfusion as the cause of headache in MMD pre-operatively, the post-operative headache is unlikely to be explained by decreased regional cerebral blood flow itself given the lack of correlation (8). The dynamic change and reorganization of the cerebral vasculature, the dilation of certain collateral vessels that progress even after surgery, the progressive recruitment of synangiosis stimulating the dural trigeminal nociceptors (46), and the neurogenic inflammation associated with the structural changes of the vasculature may be possible explanations of the continuous, or newly developed headaches post-operatively (8,44). Other possibilities of post-operative headache include mechanisms similar to ICHD 5.5 and 5.6 Headache attributed to craniotomy, as both direct and indirect revascularization involves craniotomy or craniectomy which inevitably creates necessary trauma of the scalp, skull and meninges (47). Given the manipulation of major intracranial arteries with direct bypass, mechanisms similar to the development of 6.5.2 Post-endarterectomy headache and 6.5.3 Headache attributed to carotid or vertebral artery angioplasty (48), including altered cerebral autoregulation and activation of the trigeminovascular afferent, are possible explanations as well.
Medical management of headache in patients with MMD
Adequate medical treatment of headache is crucial to improve quality of life of patients with MMD. Currently, no studies are specifically dedicated to the medical treatment of headaches in patients with MMD. The mainstay of medical therapy in such cases is to treat according to the headache phenotype and avoid vasoactive agents, if possible, especially those with the potential to alter blood pressure or cause vasoconstriction.
Antiplatelet therapy is often used to prevent ischemic stroke and transient ischemic attack in patients with MMD. As previously stated, a recent retrospective study also demonstrated the benefit of aspirin for headache in patients with MMD (30). Of note, other studies have suggested the potential role of aspirin (49–51), clopidogrel (52,53), prasugrel, and ticagrelor (54) in migraine prevention (55–57). Further studies are needed to confirm the role of antiplatelet therapy in the treatment of headache in patients with MMD.
As for abortive treatment of headache, acetaminophen, and nonsteroidal anti-inflammatory drugs (NSAIDs) are the most used medications. However, acetaminophen has variable effects, and concerns exist for drug-drug interactions between NSAIDs and aspirin for patients who are already on aspirin for stroke prevention (58). Triptans, serotonin 5HT 1B/1D agonists, are the first class of abortive medications designed specifically for the acute treatment of migraine. Immunohistochemistry studies of human vessel segments showed that 5-HT1B receptors present in the smooth muscle layer in the middle meningeal artery, coronary artery, pial and uterine arteries and saphenous vein correlated with their sumatriptan-induced vasoconstrictor responses (59,60). Triptans are contraindicated in patients with a history of ischemic stroke and should generally be avoided in patients with MMD due to the potential of vasoconstriction in a vascular network compromised by stenosis of the major anterior circulation arteries that relies heavily on the leptomeningeal collateral vessels. Similarly, dihydroergotamine should be avoided due to the vasoconstrictive effects.
Antiemetics with dopamine antagonism can be used for acute treatment of headache and nausea, either in combination with NSAIDs or alone. Metoclopramide has the strongest evidence for the acute treatment of migraine. Prochlorperazine, promethazine, and domperidone (not available in the USA) can also be used (61,62). However, clinicians should be mindful of the potential side effects including QTc prolongation, sedation, and extrapyramidal symptoms.
Lasmiditan, a selective 5-HT1F receptor agonist that decreases the release of neuropeptides and pain transmission in the trigeminovascular system, could be a good alternative for the treatment of migraine in patients with MMD as studies showed that it does not constrict blood vessels (63,64). The post-hoc analysis of the pooled results from two randomized, double-blind, phase 3 trials showed no statistical difference in lasmiditan efficacy or the frequency of likely cardiovascular-emergent adverse events by the presence of cardiovascular risk factors (63,65,66). Lasmiditan received FDA approval for the abortive treatment of migraine in adults in October 2019. However, it is currently not approved for patients under the age 18, although clinical studies are under way. Additionally, as driving impairment has been reported, it is recommended to avoid driving 8 h following medication intake.
The most used migraine-preventive oral medications supported by evidence include topiramate, sodium valproate, beta-blockers including propranolol, metoprolol, atenolol, and nadolol, and antidepressants including amitriptyline and venlafaxine. Other medications, such as nortriptyline, verapamil, flunarizine (not available in the USA), candesartan, lisinopril, gabapentin, tizanidine, memantine, and cyproheptadine, are used as well with weaker evidence or conflicting data (67). OnabotulinumtoxinA injection is FDA-approved for the treatment of chronic migraine in adults but not the pediatric population. For patients with MMD, blood pressure-lowering medications should be used with caution, as lowering blood pressure could potentially affect cerebral perfusion in patients who might already have chronic cerebral hypoperfusion due to MMD (68). Otherwise, consideration of the above preventive medications should be based on other comorbidities of the patients and side-effect profiles of the medications.
Since 2018, CGRP-targeted therapies received FDA approval for the treatment of migraine, including four CGRP monoclonal antibodies for migraine prevention – erenumab, fremanezumab, galcanezumab, and eptinizumab, and two CGRP receptor antagonists, ubrogepant and rimegepant for the abortive treatment of migraine. Rimegepant and a third CGRP receptor antagonist, atogepant, have also recently been found in randomized phase 2/3 trials to be efficacious in prevention of migraine (69,70). CGRP is the most abundant neuropeptide in the trigeminal system and plays a key role in the pathophysiology of migraine and cluster headache. Both CGRP monoclonal antibodies and CGRP receptor antagonists have extremely limited ability to cross the blood brain barrier (BBB) into the central nervous system and were presumed to work in the dura and meninges. The trigeminal ganglion is, however, also outside the BBB, and it has been postulated that this is more likely to be the primary site targeted by the novel CGRP drugs for migraine (71). Phase 3 clinical trials and real-world studies have demonstrated and validated the efficacy and safety of CGRP-targeted therapy for patients with migraine (72,73). Even the combination of rimegepant for acute attacks in persons on CGRP mAb for migraine prevention was well tolerated and without safety issues, when tested in a 13 person, 12-week sub-study nested within a multi-center, open-label, long-term safety study of CGRP mAb use (69). However, despite the favorable safety profile to date, the knowledge that CGRP is a potent vasodilator with a crucial role in regulating vascular resistance and regional blood flow (74,75) prompts concern that CGRP-targeted therapy might block compensatory vasodilation or cerebral autoregulation, in the setting of vessel occlusion or vasoconstriction. One animal study showed extensive neuronal cell loss and less compensatory capillary growth in CGRP knock-out mice than wild-type (75). Another study showed that in brief middle cerebral artery occlusion, exposure to CGRP small molecule receptor blockers increased the rate of cerebral infarction in mice as compared to placebo and produced larger infarcts in longer duration occlusion events, suggesting that while blocking CGRP receptors may not produce stroke on its own, it may worsen outcomes during ischemic events (76).
To further investigate the effect of CGRP mAb on cerebral hemodynamics, a study evaluated the cerebral vasomotor reactivity and endothelial reactivity at baseline, 2 and 18 weeks after initiating erenumab therapy in 60 patients with migraine without aura and did not show any significant changes after treatment. Vasomotor reactivity is the potential of intracranial arterioles to dilate in response to vasodilatory stimulation, such as hypercapnia, and was measured by breath-holding index and pulsatility index of the middle and posterior cerebral arteries measured by transcranial doppler. Endothelial reactivity was measured peripherally by brachial flow mediated dilation (77). However, the results measured under physiologic condition might not apply to situations of acute ischemia, such as myocardial infarction or ischemic strokes. Additionally, the study did not include patients with migraine with aura, known to have an increased risk of stroke potentially related to endothelial dysfunction (57,78).
The package insert of erenumab has been amended due to post-market reports of de novo or worsening hypertension (79). Extreme caution is also recommended when considering CGRP-targeted therapy in patients with potentially compromised blood-brain barrier, including patients with vascular malformations such as MMD; status epilepticus with diffuse infarct 2 days after erenumab injection has been reported in a patient with known cerebral proliferative angiopathy (80).
Conclusion
Headache in patients with MMD remains an important but under-addressed topic in the medical literature. Given the complexity of the pathophysiology and the disease course, a multidisciplinary approach tackling both surgical and medical treatment of headache could significantly improve the quality of life of patients. More prospective studies are warranted to better understand and manage headache in patients with MMD.
Clinical implications
Headache in patients with moyamoya disease remains an important but under-addressed topic in the medical literature. The most common headache phenotype is migraine; tension-type headache, hemiplegic migraine, and cluster headache have also been reported. Most patients experience improvement of headache after surgical revascularization, though some patients report worsening, or new-onset headache after surgery. Given the complexity of moyamoya disease, careful consideration of different types of medical therapy for headache is necessary to improve the quality of life while not increasing the risk of adverse cerebrovascular events.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: GL is a consultant for Superior Medical Editing. GET owns common stock in Johnson & Johnson. The other authors report nothing to disclose.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
