Abstract
Background
Hormonal and menstrual factors are known to influence migraines in women. However, studies in the postmenopausal period are relatively insufficient for clinical translation. This study investigated the influence of endogenous and exogenous hormonal factors on migraines in spontaneous menopausal women.
Methods
We obtained and analyzed the data related to hormonal factors from the Korean Health Examination database. A migraine diagnosis was identified using the Korean National Health Insurance Service database between 2009 and 2018. We observed migraine occurrence in spontaneous postmenopausal women. Study populations were divided into two groups depending on new diagnosis of migraine during the follow up periods. We investigated the association between endogenous and exogenous hormonal factors and migraine.
Results
1,114,742 spontaneous postmenopausal women were enrolled. Migraine risk tended to increase in the shorter lifetime number of years of menstruation group compared to the group with lifetime number of years of menstruation ≥40 years. All of the hormone replacement therapy (HRT) groups showed higher risk compared with the non-HRT group. Migraine risk tends to increase with greater postmenopausal years compared to the postmenopausal <5 years group.
Conclusion
Our study suggests that female hormonal factors, including endogenous and exogenous estrogen exposure, may be associated with migraine occurrence in spontaneous menopausal women.
Introduction
Migraine is one of the common disorders in the population, and it can bring disability into a patient’s daily life. The incidence of migraine is more prevalent in females than males, and the difference is more pronounced after puberty (1,2). In women, the decrease in estrogen induces migraine attacks during the late-luteal-phase. The relationship between migraine and estrogen is known as the “estrogen withdrawal hypothesis” (1). Women experience various sex hormonal states throughout their lives, and menopause is a physiologically significant period in terms of rapid hormonal changes (3). During the perimenopausal period, follicle stimulating hormone (FSH) secretion increases, which excessively stimulates the remaining follicles, causing estrogen fluctuation. According to the estrogen withdrawal hypothesis, estrogen fluctuation leads to frequent migraine attacks in the perimenopausal period (4).
As life expectancy increases, interest in the quality of life after menopause is increasing. Several studies have reported headaches and migraines in postmenopausal women. However, most studies have focused on migraine in the perimenopausal period. Previous studies reported the incidence of migraine is ∼10–29% during the postmenopausal periods (5–8). Although there are methodological differences in the research, some studies showed that migraine risk might decrease after menopause (4,9). However, other studies reported that migraine incidence is not different or aggravated during the postmenopausal period compared to the premenopausal period (7,10,11). The risk of migraine is still unclear in spontaneous postmenopausal women.
Hormone replacement therapy (HRT) is used to reduce perimenopausal symptoms and has many benefits, including improving quality of life and decreasing the risk of osteoporosis. However, the effect of HRT on coronary heart disease, stroke, and breast cancer still has been debated (12). Several studies reported that current HRT might aggravate migraine frequency and prevalence in postmenopausal women (5,13,14). However, another study reported that HRT has beneficial effects for migraine, and 77.4% of postmenopausal women who take HRT experience migraine improvement or remission (10). HRT is used as a treatment option for menstrual migraine prophylaxis (15). Considering these results, the influence of HRT on migraines is still unclear.
Our study investigated migraine risk and its association with female hormonal factors related to menstruation and HRT in spontaneous postmenopausal women. We used a population-based cohort that included medical information obtained from the Korean National Health Insurance Service Database.
Methods
Data source and ethical approval
We obtained data from the Korean Health Examination (KHE) database and the Korean National Health Insurance (KNHI) Service database. In Korea, regular national health examination is mandatory for adults over 40 years of age every two years. KHE items include routine laboratory tests, work-up for malignancy, physical measurements, and self-report health questionnaire. The KHE data includes age, a history of comorbid diseases (i.e., hypertension, dyslipidemia, stroke, ischemic heart disease, diabetes mellitus), and health-related behavior, including alcohol consumption, smoking, and regular physical activity for each individual. In the part related to women’s menstruation, the KHE data includes menarche age, menopause age, history of hysterectomy, and duration of HRT. The KNHI program provides medical services for the entire Korean population. Therefore, the KNHI database has a sample size of over 50 million individuals (16). The KNHI database includes all claims data for the NHI program and lists diagnoses by International Classification of Disease, Tenth Revision (ICD-10) codes.
This study was approved by the Institutional Review Board of the Korean National Institute for Bioethics Policy (NHIS-2019-1-454). The study was also approved by the Institutional Review Board of the Catholic University of Korea (no. UC19EESI0001). The informed consent was waived by boards.
Study population and study design
To investigate the influence of endogenous and exogenous hormonal factors on migraine in spontaneous postmenopausal women, we used linked KHE and KNHI data for the same subjects to evaluate migraine occurrence. The KNIH data did not include detailed information on the clinical symptoms of migraine. In the KNIH database, the migraine diagnosis consists of both migraine-specialist and non-expert diagnoses of all medical institutions, which reduces the diagnosis’s accuracy. We defined migraine diagnosis as a woman without migraine history before the observation period and given a migraine diagnosis (ICD-10 code: G430, G431, G433, G438, G439) at least once during the observation period while taking medication for migraine treatment (Triptan derivatives, Ergotamine, Topiramate, Valproate, Propranolol, Metoprolol, Timolol, Flunarizine, Amitriptyline, and Venlafaxine). We selected the medication list based on migraine treatment recommendations and medical insurance coverage in Korea (17). Spontaneous menopause is defined as menopause onset without a history of hysterectomy as based on the standard KHE questionnaire. We extracted KNHIS claims that had an ICD-10 code diagnosis for migraine, and associated medication history. We excluded women who had a history of previous migraine diagnosis (ICD-10 code) during washout period (between 2005 and 2009). Study populations were divided into a migraine group, and a control group according to migraine diagnosis during the follow-up period (between 2009 and 2018).
In this study, we observed migraine occurrence in spontaneous postmenopausal women. We investigated the association between endogenous and exogenous hormonal factors, and migraine attack. Endogenous hormonal factors were menarche age, menopause age, postmenopausal years, and the lifetime number of years of menstruation (LNYM). We divided the menarche age group by 12 years. Previous study reported the mean Korean menarche age was 12.9 ± 1.18 years (18). Menopause age was divided by 50 years because the mean Korean menopause age was 49.3 ± 3.5 years (19). Postmenopausal years are defined as the total years of life after menopause. LNYM represents the cumulate duration of total lifetime menstruation cycle. Generally, LNYM defined as the number of years between menarche and menopause, minus duration of pregnancy, duration of oral contraceptive and breastfeeding (20,21). However, KHE questionnaire did not include the exact period of pregnancy, oral contraceptive and breastfeeding. So, we defined LNYM as the total number of years between menarche and menopause. The LNYM is used as a proxy for cumulative exposure to endogenous hormones during a women’s life (20). The LNYM is divided into four categories (<30 years, <35 years, <40 years, and ≥40 years). We set the exogenous hormonal factor as the duration of HRT, divided into five categories by the total years of HRT use (none, <2 years, ≥2–<5 years, ≥5 years, and unknown).
Statistical analysis
We used the Student's t-test for continuous variables and the chi-square test for binary and categorical variables. All subjects were divided by LNYM, duration of HRT, and total postmenopausal years. The incidence rate was calculated as 1,000 person-years in each group. The hazard ratio was analyzed using Cox's proportional hazards regression models, with a 95% confidence interval (CI) for each factor. To control for confounding factors, we constructed Model 1, which included age, and Model 2, which included age, smoking, alcohol, physical activity, and body mass index (BMI).
Using Cox proportional hazards regression models for each of three consecutive age intervals (<50 years, <60 years, and <70 years), we analyzed the relationship between HRT duration and migraine risk according to age. The association between postmenopausal years and migraine risk was analyzed with the same method for two menopause age groups (<50 years, and ≥50 years). All statistical analyses were performed using SAS software (ver. 9.4; SAS Institute, Cary, NC, USA).
Data availability statement
Data cannot be shared publicly due to identity protection. Raw data are available from the National Health Insurance Date Sharing Service (https://nhiss.nhis.or.kr/bd/ab/bdaba000eng.do) for researchers who meet the requirements for access to confidential data.
Results
Demographics and menstruation characteristics
We enrolled 1,368,400 spontaneous menopausal women aged 40–60 years who completed the health examination questionnaire using KHE data between 2009 and 2018. We excluded 96,928 women who had a history of migraine diagnosis. A total of 156,730 were removed due to unanalyzable or missing data. Finally, 1,114,742 spontaneous postmenopausal women were enrolled and assessed for a diagnosis of migraine from 2009 to 2018. (Figure 1) The migraine group (150,802) showed older (migraine vs. control; 58.7 ± 5.89 vs. 58.5 ± 5.82 years, p < 0.001) and higher BMI (migraine vs. control; 24.25 ± 3.07 vs. 24.23 ± 3.11 kg/m2, p = 0.001) than the control group (940,963). hypertension (HBP), dyslipidemia, and ischemic heart disease (IHD) were more frequent in controls than the migraine group. Diabetes (DM) was more common in the control group than in the migraine group. Stroke was not significantly different between the two groups (Table 1).

Flow chart of the study process.
Demographics and population characteristics according to migraine occurrence.

Data are presented as mean ± standard deviation (SD) or n (%).
Menarche age was older in the migraine group (migraine vs. control; 16.44 ± 1.84 vs. 16.32 ± 1.84 years, p < 0.0001) but there were no significant differences in the categorical analysis based on a menarche age of 12 years. The menopause age was younger in the migraine group than the control group (migraine vs. control; 50.06 ± 3.94 years, 50.24 ± 3.85 years, p < 0.0001). The LNYM was longer in the control group than the migraine group (migraine vs. control; 33.93 ± 42. vs. 33.62 ± 4.29 years, p < 0.0001). The postmenopausal years were longer in the migraine group than the control group (migraine vs. control; 8.63 ± 6.62 vs. 8.26 ± 6.45 years, p < 0.0001). The distribution of menopause age, LYNM, postmenopausal years, and duration of HRT were significantly different between groups according to migraine occurrence (p < 0.0001). (Table 2).
Characteristics of endogenous and exogenous estrogen exposure in control and migraine group.

Data are presented as mean ± standard deviation (SD) or n (%).
Risk of migraine according to endogenous hormonal factor and postmenopausal years
Table 3 shows the hazard ratio (HR) with a 95% confidence interval (CI) of migraine risk according to hormonal factor. Model 2 presents migraine risk after adjusting for confounding factors that included age, BMI, smoking status, alcohol consumption, and physical activity. Migraine risk tended to significantly increase for shorter LNYMs. The HRs were significantly higher in short LNYM groups that included LNYM<30 (HR, 1.25; 95% CI, 1.219–1.281), LNYM<35 (HR, 1.152; 95% CI, 1.127–1.178) and LNYM<40 (HR, 1.06; 95% CI, 1.037–1.084) compared to the longer ≥40 years LNYM group. Migraine risk significantly increased with longer postmenopausal years. The HR was higher in longer postmenopausal groups that included postmenopausal ≥15 years (HR, 1.196; 95% CI, 1.169–1.224), <15 years (HR, 1.09; 95% CI, 1.069–1.111), <10 years (HR, 1.042; 95% CI, 1.027–1.058) compared to the <5 years group.
Multivariate Cox’s proportional hazard regression model of the relationship between hormonal factors and risk of migraine.

The incidence rates were calculated per 1,000 person-years. Model 1 Adjusted for age. Model 2 Adjusted for age, BMI, smoking status, alcohol consumption, and physical activity.
We present the HR and 95% CI for migraine according to postmenopausal years for each menopausal age group (<50 and ≥50 years) (Figure 2). Our analysis showed the HR for migraine increased in both menopause age groups.

The hazard ratio for migraine according to postmenopausal year for each menopause age group after adjusting for age, BMI, income, smoking status, alcohol consumption, physical activity, hypertension, dyslipidemia, and diabetes. (* indicates statically significant result)
Risk of migraine according to HRT duration
All HRT groups that included HRT <2 years (HR, 1.194, 95% CI, 1.176–1.213), HRT <5 years (HR, 1.165, 95% CI, 1.137–1.193), and HRT ≥5 years (HR, 1.194, 95% CI, 1.162–1.227) showed higher migraine risk compared with the non-HRT group. (Table 3) In the analysis to determine whether the effect of HRT on the occurrence of migraine was different according to age, the HR for migraine in HRT groups was higher than the non-HRT for all age groups (Figure 3).
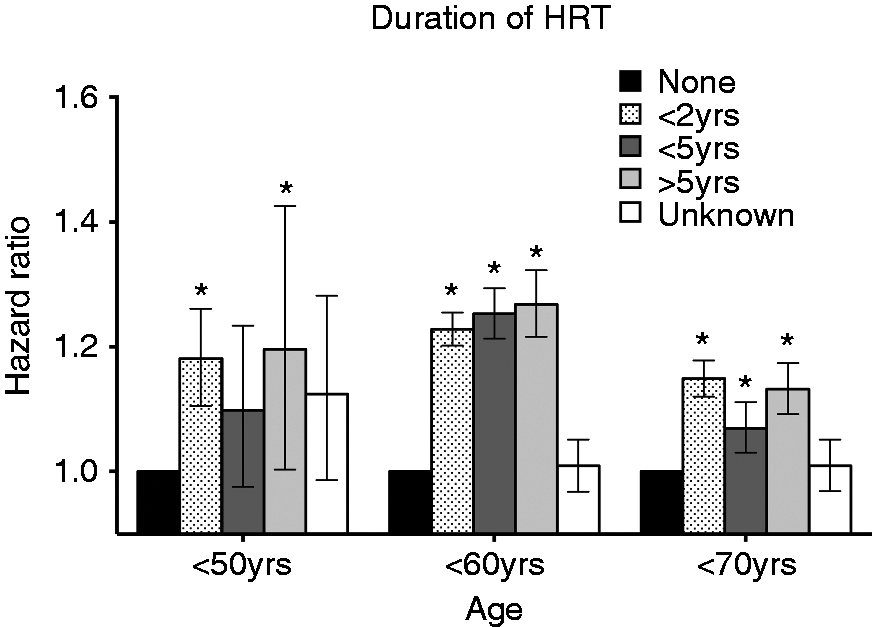
The hazard ratio for migraine according to 10-year age group by HRT duration after adjustment for menstrual duration, age, age, BMI, income, smoking status, alcohol consumption, physical activity, hypertension, dyslipidemia, and diabetes. (* indicates statically significant result)
Discussion
This is a nationwide population-based retrospective cohort study that investigated migraine risk in spontaneous menopausal women without previous migraines. The major outcomes of the present study are as follows. Shorter LNYM was related to a higher risk of migraine. HRT increased the risk of migraine, and the effect persisted even into older age. The risk of migraine increased after menopause in spontaneous menopausal women.
Endogenous estrogen and migraine
Longer LNYM represent longer endogenous estrogen exposure. Longer LNYM increases the risk of breast and endometrial cancer (20,22). Estrogen is related to migraine pathogenesis by modulation of pain-related excitatory and inhibitory neurotransmission (23). Animal studies reveal that estrogen enhances structural changes in the excitatory glutaminergic synapse secondary to the sprouting of dendritic spines and NMDA receptors. This neuroexcitation is associated with migraine aggravation in the trigeminal nucleus caudalis (TNC) (24–26). Also, a high level of sex hormone enhances activation of inhibitory GABA-A receptors located at extrajunctional sites that effectively inhibits neuroexcitation (26,27). During high estrogen states (late follicular and early to mid-luteal phases), neuroexcitation is decreased by activation of the inhibitory system. When estrogen decreases (late luteal and early follicular phase), the inhibitory system rapidly reverses, but the neuroexcitation structural change (glutamate and NMDA receptors) may not reverse as fast as the inhibitory system. So perimenstrual phase and estrogen fluctuation are related to a migraine attack (26). Women with migraine experience had fewer migraine attacks during pregnancy that worsened during the postpartum period (28,29). During pregnancy, the plasma estrogen level did not fluctuate and constantly remained 30–40 times higher than the peak level of the natural menstruation period (26). However, migraine risk increases in early artificial menopause, such as that due to hysterectomy or oophorectomy (4,8,30). After artificial menopause, estrogen’s abrupt decline occurs initially, but after that, the estrogen level remains continuously low without fluctuations. Our results showed that longer endogenous estrogen exposure might be related to low migraine risk. These results suggest that adequate endogenous estrogen exposure also has a significant impact on migraine risk in addition to estrogen fluctuation.
Exogenous estrogen and migraine; HRT
In postmenopausal women, ovarian follicular function decreases, and estrogen variability may be stabilized. However, intermittent oral HRT induces artificial hormonal variability, which could increase the risk of migraine attack (9). Although these results vary depending on the study, most large population-based studies report that HRT aggravates migraines in postmenopausal women. A population-based study of 17,107 postmenopausal women reported that current HRT significantly increases migraine risk (OR 1.42; 95% CI 1.24–1.62) compared to the non-HRT population. Odds of migraine attack were similar among women with 1–4 and ≥5 years of HRT use (14). In a population-based cross-sectional study of 6,007 postmenopausal women, the HRT group showed a higher migraine risk than the non-HRT group. There was no association with administration method (local or systemic administration) (13). In male-to-female transsexuals, estrogen administration is related to the occurrence and worsening of headaches (31). The present study showed that HRT is associated with higher migraine risk, regardless of HRT duration. This tendency was also observed in elderly menopausal women. However, the women who have migraines as perimenopausal symptoms may choose HRT more, and our study did not include the information of the perimenopausal period. So there needs a cautious interpretation. Our results support that HRT may have a negative influence on migraine risk.
Migraine in postmenopausal women
There have not been enough investigations of the risk and prevalence of migraine in postmenopausal women. Migraine risk increases during the perimenopausal period due to estrogen concentration instability (4). Perimenopausal women showed higher estrogen levels and greater fluctuations compared to young premenopausal women (32). A cross-sectional study reported that migraine prevalence increases during perimenopause and decreases after spontaneous menopause. However, this study investigated the prevalence of migraine among women aged 40 to 54 years (8). One study reported that migraine risk increased in perimenopausal (OR 1.42, 95% CI 1.03–1.94) but not significantly in postmenopausal women (OR 1.27, 95% CI 0.83–1.92) compared to premenopausal women (33). Other population-based studies reported that the risk of migraine did not have a significant association with menopause. The early period after menopause was more strongly associated with the occurrence of migraine without aura. However, this study analyzed the association between menopause and migraine in a restricted age group (45–59 years, n = 375) (34). These studies show an increase in migraine risk during the perimenopausal period. However, the long-term migraine risk is still inconclusive in postmenopausal women. The present study showed that migraine risk tends to increase over time after menopause. These findings seem to be counterintuitive as it is a common assumption that migraine prevalence decreases with menopause (4). There is a need for caution in interpreting our results. Our study investigated migraine risk only after spontaneous menopause. Therefore, our results could not be interpreted as an increased risk of postmenopausal migraine compared to the premenopausal age. Previous studies for postmenopausal menopause were investigated in a small population or short observation period compared to our study (4,8,9). In line with the role of LNYM in migraine, the present results suggest that adequate endogenous estrogen exposure may be associated with migraine risk. However, further investigations are needed to clarify the association between hormones and migraine in postmenopausal women.
This study is a large population-based study using the KNHI Database but there are some limitations to our study. We enrolled migraine participants by using ICD-10 codes in the KNHIS database. Also, we enrolled participants who took migraine-specific abortive and preventive medications. Since medical insurance data did not include clinical symptoms, inevitably, we used these migraine diagnostic criteria. Therefore, it is possible that our study did not include women with migraines who take simple analgesics only and may enroll the more severe migraineurs. It is known that the different patterns of HRT can have a different effect on migraine (35). Our study could not investigate the relationship between the type of HRT and migraine because the KHE data did not include information about the type of HRT. Also, mimic migraine symptoms could be over diagnosed and enrolled in the study group. So, there is a need for cautious interpretation of our result. The present study design did not contain perimenopausal period information, so it is difficult to clarify migraine impacts during this period.
In conclusion, this study found that short LNYM was related to high migraine risk. HRT had a negative influence on migraine risk regardless of HRT duration in postmenopausal women. In spontaneous postmenopausal women, female hormonal factors, including endogenous and exogenous estrogen exposure, may be related to migraine occurrence.
Clinical implications
The exogenous hormonal factor (HRT) and short endogenous hormonal exposure (LMYN) negatively influenced postmenopausal women’s migraine risk. Migraine risk tends to increase over time after menopause. The female hormonal factors, including endogenous and exogenous estrogen exposure, may be related to migraine occurrence via various mechanisms.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors wish to acknowledge the financial support of The Catholic University of Korea Uijeongbu St. Mary’s Hospital Clinical Research Laboratory Foundation made in the program year of 2019
