Abstract
Background
Erenumab (erenumab-aooe in the US) effectively reduces monthly migraine days in episodic and chronic migraine. This traditional outcome does not capture the intensity of headache pain on days with migraine.
Methods
This post hoc analysis of two pivotal randomized, placebo-controlled studies in patients with episodic migraine and chronic migraine examined the effect of erenumab 70 and 140 mg on migraine pain. Cumulative monthly migraine pain intensity is the sum of the peak pain intensity scores (0 = no migraine to 3 = migraine day with severe pain) on migraine days. Change from baseline in cumulative monthly migraine pain and average monthly pain intensity was assessed over months 4 to 6 for episodic migraine and month 3 for chronic migraine; change in average monthly pain intensity was assessed among monthly migraine days responders/non-responders.
Results
Efficacy analysis included 946 patients for the episodic migraine study and 656 patients for the chronic migraine study. Cumulative monthly migraine pain decreased significantly with erenumab versus placebo (p < 0.001, for episodic migraine and chronic migraine). In addition, monthly average migraine pain intensity decreased significantly with erenumab versus placebo for episodic migraine (p < 0.01); decreases were non-significant for chronic migraine. In comparison with placebo-treated patients, a greater proportion of erenumab-treated patients were pain intensity responders regardless of threshold used. Episodic migraine and chronic migraine patients with a ≥50% reduction in monthly migraine days (responders) had a greater reduction in monthly average pain intensity than non-responders.
Conclusions
Erenumab reduced cumulative monthly migraine pain in episodic migraine and chronic migraine patients and significantly reduced monthly average migraine pain in episodic migraine, demonstrating treatment benefit beyond reduction in migraine frequency.
Introduction
The frequency and intensity of migraine symptoms contribute to the impact of migraine on health-related quality of life (1,2); headache pain is one of the key diagnostic criteria of migraine (3). The European Federation of Neurological Societies recommend that preventive treatment should be considered for patients with migraine if quality of life is severely impaired, at least two migraines occur per month, there is no response to acute treatment, or auras occur frequently (4). Further, the American Headache Society recommends preventive treatment be considered based on a combination of monthly headaches and functional disability (5). The goals of preventive treatment include reducing the number of migraine attacks or days as well as the intensity of pain on those days (5). Reduced pain intensity is an important patient goal for acute treatment (6). While the effect of preventive treatments on reducing the number of monthly migraine days (MMD) or headache days has been studied extensively, the potential effect of treatment on headache pain intensity merits further study.
Erenumab (erenumab-aooe in the US) is a fully human monoclonal antibody that selectively targets and blocks the canonical calcitonin gene-related peptide (CGRP) receptor and was developed specifically for the treatment of migraine (7). Clinical trials have confirmed the safety and efficacy of erenumab in the preventive treatment of migraine (8–10), and it is approved for use in adults (11,12). While the benefit of erenumab treatment has been demonstrated based on various frequency-based outcome measures, there is a need to evaluate additional treatment benefits (e.g. reduction in pain intensity) that is not captured by these outcome measures alone. Indeed, assessment of pain intensity, which is a meaningful measure to patients, is a recommended secondary endpoint in controlled clinical studies of migraine preventive treatment (13).
Given the association of migraine pain intensity and migraine-related disability, we sought to examine the effect of erenumab treatment on migraine pain intensity. This post hoc analysis of two pivotal randomized, placebo-controlled studies of erenumab in patients with episodic migraine (EM) or chronic migraine (CM) (9,10) evaluated the impact of erenumab on a series of pain-related outcomes defined below, including cumulative monthly migraine pain, monthly average migraine pain, pain intensity response, and monthly average pain of the breakthrough migraine headaches in treatment responders (i.e. those who achieved at least a 50% reduction from baseline in MMD averaged over months 4 to 6 in the EM study and for month 3 of the CM study) versus those who achieved less than 50% MMD reduction. While cumulative measures reflect aggregate pain burden, these estimates are influenced by the number of migraine days. The importance of monthly average pain intensity is that it indexes pain per attack and is less influenced by number of attacks.
Methods
Study design and patient population
The study designs and results from two randomized, double-blind, and placebo-controlled erenumab studies have been reported previously (9,10). Briefly, a pivotal EM study was conducted across 121 sites in North America, Europe, and Turkey (9), and a pivotal CM study was conducted across 69 sites in North America and Europe (10). Both studies included a 4-week baseline phase. Patients in the EM study (9) were randomized (1:1:1) to placebo, erenumab 70 mg, or erenumab 140 mg once monthly for 6 months during the double-blind treatment phase (DBTP), and patients in the CM study (10) were randomized (3:2:2) to placebo, erenumab 70 mg, or erenumab 140 mg once monthly for 3 months.
Adults aged 18 to 65 years were eligible for participation in the EM study (≥4 and <15 migraine days per month and < 15 headache days per month) and CM study (≥15 headache days per month and ≥8 migraine days per month). Patients were excluded from both studies if they were older than 50 years at migraine onset and if they had a history of hemiplegic migraine or cluster headache. Additionally, patients were excluded if they had shown no therapeutic response to more than two (EM study) or three (CM study) migraine-preventive treatment categories and if continuous pain was present for the CM group. No therapeutic response was defined as an adequate trial of at least 6 weeks at the recommended dose without any benefit in terms of frequency, duration, or intensity of the attacks, as assessed by the investigator. Patients with concurrent use of preventive treatment for migraine were excluded from the CM study; however, patients using one migraine-preventive drug at a stable dose could enroll in the EM study. In both studies, an independent ethics committee at each trial center approved the study protocols (9,10). All patients provided written informed consent before study initiation.
Outcome measures
The primary endpoint in each study was the change in mean MMD from baseline for the last 3 months (mean over months 4–6, EM study (9)) or for the last month (month 3, CM study (10)) of the DBTP. Patients reported the date of migraine onset and peak pain intensity (mild, moderate, or severe) in daily electronic diaries throughout the baseline period and the DBTP. The criteria used to define a migraine day included a headache (with or without aura) lasting 30 min or longer for EM and 4 h or longer for CM, the presence of at least two pain features (unilateral, throbbing, moderate-to-severe intensity, or aggravation by exercise or physical activity) and one of two non-pain symptoms (nausea or vomiting or photophobia and phonophobia), or treatment with an acute migraine-specific medication (9,10).
In this post hoc analysis, we define a series of pain intensity-related outcomes and endpoints of interest.
Statistical analysis
The efficacy analysis was conducted separately for each study among patients who received at least one erenumab dose and had at least one post-baseline outcome assessment. The 70 and 140 mg dosage groups were separately evaluated for each study. For continuous endpoints, the least squares (LS) mean, associated standard error, 95% confidence interval, and p-value for treatment comparison were calculated using a generalized linear mixed-effect model assuming normal distribution with identity based on observed data. For dichotomous responder endpoints, the adjusted odds ratio compared to placebo, associated 95% confidence interval, and p-value were estimated using a stratified Cochran-Mantel-Haenszel test after missing data were imputed as non-response. p-values were nominal values without multiplicity adjustment.
Results
Patient demographics and characteristics
Patient demographics and baseline characteristics were previously reported and balanced between groups in both studies (Table 1) (9,10). Briefly, 955 patients were enrolled in the EM study, 946 patients were included in the efficacy analysis, and 858 patients (89.8%) completed treatment; mean age was 40.9 years, and patients had a mean MMD at baseline of 8.3 (9). In the CM study, 667 patients were randomized to treatment, 656 patients were included in the efficacy analysis, and 637 (95.5%) completed treatment. For the full population (n = 667), the mean age was 42.1 years, and patients had a mean of 18.0 MMD at baseline (10). Mean pain intensity was 2.2 (out of a maximum of 3) at baseline in both EM and CM study populations. Most patients in both studies were female (85.2% and 82.8%, EM and CM, respectively). Mean age at migraine onset was similar in both studies (21.1 years and 20.9 years). Prior preventive treatment failures were reported by 370 patients (38.7%) in the EM study and 453 patients (67.9%) in the CM study.
MMD: monthly migraine days; MSMD: monthly acute migraine-specific medication days.
*Only assessed in evaluable patients who received at least one erenumab dose and had at least one post baseline outcome assessment.
Cumulative monthly migraine pain
Cumulative monthly migraine pain was the sum of peak pain intensities on migraine days for the critical pre-specified time period for assessing primary efficacy. In the EM study, at baseline, mean (SD) cumulative monthly migraine pain was 19.8 (7.4) for the placebo group, 19.8 (7.0) for the 70 mg erenumab group, and 19.6 (7.4) for the erenumab 140 mg group. Over the last 3 months of treatment in the EM study, cumulative monthly migraine pain decreased significantly in patients receiving either erenumab 70 mg (LS mean [SE], −9.1 [0.4]) or erenumab 140 mg (LS mean [SE], −9.9 [0.4]) compared with placebo (LS mean [SE], −6.6 [0.4]; Figure 1(a)). The difference in LS mean (95% CI) change from baseline over months 4 to 6 in cumulative monthly migraine pain, compared with placebo, was −2.5 (−3.5, −1.4) for erenumab 70 mg (p < 0.001) and −3.2 (−4.3, −2.2) for erenumab 140 mg (p < 0.001; Figure 1(a)). Mean (SD) baseline values in the CM study were 41.5 (13.9), 41.3 (12.6), and 41.3 (13.7) for placebo, erenumab 70 mg, and erenumab 140 mg, respectively. Cumulative monthly migraine pain was decreased in patients with CM receiving either erenumab 70 mg (LS mean [SE], −19.7 [1.0]) or erenumab 140 mg (LS mean [SE], −20.2 [1.0]) compared with placebo (LS mean [SE], −15.3 [0.8]; Figure 1(b)). The difference in LS mean (95% CI) change from baseline to month 3, compared to placebo, was −4.3 (−6.8, −1.9) for erenumab 70 mg (p < 0.001) and −4.8 (−7.3, −2.4) for erenumab 140 mg (p < 0.001; Figure 1(b)).

Change from baseline in cumulative monthly migraine pain with erenumab compared with placebo in the (a) episodic migraine study and (b) chronic migraine study.
Monthly average migraine pain
The impact of erenumab on migraine pain intensity, independent of frequency, was evaluated by calculating monthly average pain intensity of breakthrough migraine headaches, using the pain intensity score ranging up to 3. EM patients receiving erenumab at 70 mg and 140 mg showed significant decreases in monthly average migraine pain intensity over the last 3 months of treatment. Compared to placebo, the difference in LS mean (95% CI) change from baseline over months 4 to 6 in monthly average migraine pain intensity was −0.13 (−0.21, −0.05) for erenumab 70 mg (p = 0.002) and −0.13 (−0.21, −0.04) for erenumab 140 mg (p = 0.003; Figure 2(a)). Slight decreases in monthly average migraine pain were observed for CM patients receiving erenumab, although these differences were not significant. After 3 months of treatment, the difference in LS mean (95% CI) change from baseline compared with placebo was −0.04 (−0.11, −0.02) for erenumab 70 mg (p = 0.20) and −0.06 (−0.13, 0.01) for erenumab 140 mg (p = 0.076; Figure 2(b)).
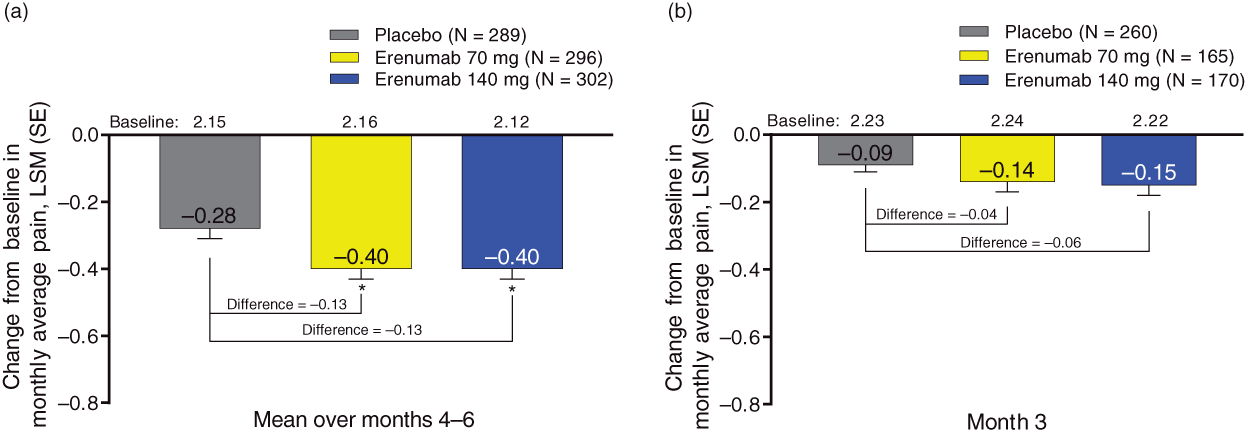
Change from baseline in the monthly average migraine pain on days with migraine with erenumab compared with placebo in the (a) episodic migraine study and (b) chronic migraine study.
A greater proportion of patients receiving erenumab 70 and 140 mg versus placebo achieved a reduction in monthly average pain intensity at the various thresholds assessed (Supplementary Figure 1). Further, there was a clear separation between placebo and erenumab groups in the cumulative distribution of change in monthly average pain intensity from baseline (Supplementary Figure 2).
Monthly average pain intensity for breakthrough headaches among different levels of MMD reduction
We also explored whether the average monthly pain intensity for breakthrough migraine headaches differed by the degree of reduction in MMD. Using a threshold of at least a 50% reduction in MMD from baseline, MMD responders in the EM and CM studies had a greater reduction in monthly average pain intensity than those who had <50% reduction in MMD (Figure 3). Reduction in average pain intensity was intermediate for partial responders (≥30% to <50% reduction in MMD) and lowest in the marginal response (>0% to <30% reduction in MMD) and no response or worsening headache groups (≤0% reduction in MMD). Among ≥50% MMD responders, the mean reduction in pain intensity was greater for EM (∼0.6) than CM patients (∼0.2) (Figure 3(a),(b)).

Change from baseline in monthly average migraine pain (not cumulative) among subgroups based on MMD response/non-response in the (a) EM study and (b) CM study. Response status determined based on percent reduction in MMD from baseline.
Discussion
The efficacy of migraine-specific preventive medication is often established based on reduction in migraine frequency (i.e. MMD) (9,10,14), yet frequency alone may not capture the full benefits of treatment, and so secondary measures such as impact on physical function and everyday activities may also be assessed (9,14). In addition, a measure of change in MMD does not capture reductions in the maximum intensity of pain associated with migraine days when they occur. In this post hoc analysis, we defined monthly cumulative migraine pain as a composite measure that takes into account both migraine frequency and the peak pain intensity on each migraine day. In patients with EM or CM, treatment with erenumab resulted in a greater reduction in monthly cumulative migraine pain compared with placebo. Based on this measure alone, we cannot determine if there was a reduction in MMD, in pain intensity or both. While baseline pain intensity was similar for EM and CM populations, in general, reductions in pain intensity were less pronounced for patients with CM compared to patients with EM. This difference could reflect the longer treatment period for patients with EM (6 months) versus patients with CM (3 months). Alternatively, the degree of treatment-related reduction in pain intensity could depend upon the baseline migraine frequency, with CM patients possibly being less treatment-responsive for pain on days with migraine than EM patients.
A numerically greater proportion of patients treated with erenumab had improvements in pain intensity scores regardless of the threshold used, with a clear separation observed between placebo and erenumab in the cumulative distribution of change. The benefits of preventive treatment include reducing MMD frequency and reduction in the maximum pain intensity of breakthrough headaches. It should be kept in mind that the measure of average pain intensity for migraine was to be included independent of any effect on reduction in frequency of migraine days. Patients who achieved at least a 50% reduction in MMD also showed a greater reduction in monthly average pain intensity compared to those who did not achieve this level of reduction in MMD. Partial responders (≥30% to < 50% reduction in MMD) also had substantial reduction in pain intensity for breakthrough headaches. The association of the degree of reduction in MMD and the degree of reduction in pain intensity on days with breakthrough headaches suggests that these two benefits of treatment may have a common mechanism. In clinical practice, patients with < 50% MMD response are labelled as “non-responders” based on MMD reduction; however, they may benefit in other meaningful ways, including pain reduction for breakthrough headaches and reduced time spent with moderate-severe pain. The effect of erenumab on headache duration of at least moderate pain intensity will be assessed in the upcoming phase 4 “Erenumab - Comprehensive Assessment of Efficacy in (High-Frequency) Episodic Migraine” (EMBRACE) study (15).
The most recent IHS guidelines on controlled clinical trials recommend change in migraine days as a primary endpoint and migraine intensity as a secondary endpoint (13). The primary endpoint of reduction in MMD was met in both the EM and CM studies included in this post hoc analysis of erenumab treatment (9,10). In this analysis, we demonstrated that migraine pain intensity significantly decreased in patients with EM who received erenumab. These results suggest that erenumab-treated patients may experience additional benefits beyond the numerical reduction in MMD that is typically measured in migraine preventive clinical trials.
Limitations of this analysis include the limited range of the 0 to 3 point scale used to assess pain intensity. In addition, when we compute mean pain scores we are implicitly assuming that this is an interval scale. We do not know that a change from 3 to 2 points is the same as a change from 2 points to 1 point on this scale. Our decision to treat pain score as a continuous interval scale was based on the US Food and Drug Administration (FDA) guidance in analgesic indications and the absence of a better alternative (16). Though the FDA guidance acknowledges that a mean of pain scores (numerical or categorical) among patients may not be a meaningful quantity, they also note that comparing mean pain scores may be used as a “valid, sensitive test for systematic differences between groups in individual scores.” Recent successful regulatory applications included such statistical treatment of the numerical rating scales for pain (17,18). Thus, in this manuscript, we use mean pain scores to describe systematic differences between treatment groups and use the endpoint of change from baseline in the monthly average migraine pain intensity to assess treatment effects of erenumab versus placebo on attacks that continue to occur. In addition, our ordinal scale (i.e. ordered categories with uncertain spacing between categories) makes it challenging to identify a clinically meaningful pain reduction. Nonetheless, results of our threshold analyses were directional and pointed to a potential magnitude of response that could be further explored in future analyses. In our study, meaningful changes in pain may have been difficult to detect because pain was measured on a 3-point scale. To address this limitation, change from baseline in monthly average pain intensity scores was also assessed, with cumulative percentages also presented. Researchers may consider alternative pain rating scales, such as a 0–10 point numerical rating scale or a 100 mm visual analogue scale in future clinical trials. Neither of these methods has been shown to be an interval scale, so the issues in computing averages remain. Another limitation is that we captured only peak pain intensity per headache episode for EM and per headache day for CM. Assessing least intense and most intense pain, average pain or multiple time points using ecological momentary assessment approaches may provide a more robust measure of the effects of treatment on pain (19–21). We did not assess the benefits of erenumab on the intensity of migraine features other than pain, including associated symptoms and functional status during the prodrome, headache phase, and post-drome. Additionally, the impact of acute medications on pain intensity was not accounted for in the analyses.
Conclusion
In this post hoc analysis of patients with episodic or chronic migraine, treatment with erenumab led to a reduction in cumulative monthly migraine pain, suggesting a benefit of treatment beyond reduction in migraine frequency. Future studies of migraine pain should consider use of a broader and more sensitive rating scale and analysis of the duration of time spent with moderate or severe pain to better assess the impact of treatment on migraine pain intensity.
Clinical implications
In patients taking erenumab, in addition to reducing the number of monthly migraine days, there was also a reduction in pain intensity for breakthrough migraine attacks. Reduction in pain intensity was seen in both chronic and episodic migraine patients taking erenumab. The ability of a treatment to reduce migraine pain intensity should be considered when evaluating treatment options for patients with migraine, as migraine pain intensity is associated with increased disability and reduced health-related quality of life.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024211028966 - Supplemental material for Reduction in migraine pain intensity in patients treated with erenumab: A post hoc analysis of two pivotal randomized studies
Supplemental material, sj-pdf-1-cep-10.1177_03331024211028966 for Reduction in migraine pain intensity in patients treated with erenumab: A post hoc analysis of two pivotal randomized studies by Richard B Lipton, David W Dodick, David Kudrow, Uwe Reuter, Nadia Tenenbaum, Feng Zhang, Gabriel Paiva da Silva Lima, Denise E Chou and Daniel D Mikol in Cephalalgia
Footnotes
Acknowledgements
The authors thank Allison Gillies, PhD (ICON, North Wales, PA), whose work was funded by Amgen Inc., and Annalise Nawrocki, PhD (Amgen Inc., Thousand Oaks, CA) for medical writing assistance in the preparation of this manuscript.
Data sharing
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RBL reports serving on the editorial board of Neurology, senior advisor to Headache, and associate editor to Cephalalgia; holding stock options in Biohaven Holdings; serving as consultant or advisory board member for or receiving honoraria from AEON, AbbVie/Allergan, Amgen, Biohaven, Biovision, Boston Scientific, Dr. Reddy’s (Promius), electroCore, Eli Lilly, GlaxoSmithKline, Lundbeck (Alder), Merck, Teva, Vector, and Vedanta; and receiving royalties for Wolff’s Headaches.
DD reports consulting fees from AEON, Alder, Allergan, Amgen, Amzak Health, Association of Translational Medicine, Autonomic Technologies, Axsome, Biohaven, Charleston Labs, Clexio, Daniel Edelman Inc., Dr Reddy's Laboratories (Promius), ElectroCore, Eli Lilly, eNeura, Equinox, Foresite Capital, Impel, Ipsen, Neurolief, Nocira, Novartis, Oppenheimer, Pieris, PSL Group Services, Revance, Salvia, Satsuma, Sun Pharma (India), Supernus, Teva, Theranica, University Health Network, Upjohn (Division of Pfizer), Vedanta, WL Gore, XoC, Zosano, and ZP Opco; personal fees to develop/deliver educational content for continuing medical education projects from the Academy for Continued Healthcare Learning, Catamount, Chameleon, Global Access Meetings, Global Life Sciences, Global Scientific Communications, Haymarket, HealthLogix, Medicom Worldwide, MedLogix Communications, Mednet, Miller Medical, PeerView, Universal Meeting Management, UpToDate (Elsevier), and WebMD Health/Medscape; personal fees, royalties from Cambridge University Press, Oxford University Press, and Wolters Kluwer Health; board of directors, received stock options from Aural Analytics, Epien, Healint, King-Devick Technologies, Matterhorn, Nocira, Ontologics, Precon Health, Second Opinion/Mobile Health, and Theranica; no personal fees or royalties from Allergan for Patent 17189376.1-1466:vTitle: Botulinum Toxin Dosage Regimen for Chronic Migraine Prophylaxis; research funding to institution for salary support from the American Migraine Foundation, Henry Jackson Foundation, PCORI, and US Department of Defense; reimbursement for travel from the American Academy of Neurology, American Brain Foundation, American Headache Society, American Migraine Foundation, Canadian Headache Society, and International Headache Society; personal fees, speaking (not speakers bureau) from Amgen, Lilly, Lundbeck, and Novartis. DK reports serving as consultant for Alder, Amgen, Biohaven, Eli Lilly, Nerivio, and Satsuma, and receiving research grants from Alder, Allergan, Amgen, Axsome, Biohaven, Eli Lilly, Satsuma, and Teva. UR discloses consulting fees, speaking/teaching fees, and/or research grants from Allergan, Amgen, Autonomic Technologies, CoLucid, ElectroCore, Novartis, Pharm Allergan, Eli Lilly, and Teva Pharmaceuticals. NT is an employee and stockholder of Novartis Pharma AG. FZ, GL, DEC, and DDM are employees and stockholders of Amgen Inc.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Amgen Inc. and Novartis.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

