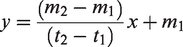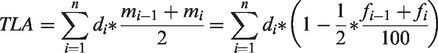Abstract
Objective
We propose a new outcome measure to assess the efficacy of migraine treatments translating the approach of the Global Burden of Disease studies from a societal to an individual level: Instead of calculating “years lived with disability”, we suggest estimating “time lost due to an attack”.
Methods
Time lost due to an attack is calculated by multiplying the duration and the degree of impaired functioning during an attack.
Results
Time lost due to an attack, different from other outcome measures, does not just focus on the short-term analgesic effects of treatments, but rather on the improvement of all migraine symptoms and restoration of functioning, also considering therapy-related impairment. Importantly, time lost due to an attack measures the entire time patients are not functioning normally, from onset to complete resolution.
Conclusions
Time lost due to an attack represents a new paradigm to assess migraine burden in single patients for a patient-centered evaluation of both acute and prophylactic treatments.
Introduction
Since the triptan era, changes in pain intensity have been assessed to provide evidence of acute efficacy of novel treatments. Generally, the proportion of patients with a reduction from moderate or severe to “no or only mild pain” or “no pain at all” at 2 h post-dose was the preferred primary outcome measure in clinical trials (1,2). More recently, the Food and Drug Administration has added “resolution of the most bothersome associated feature at 2 hours post-dose” as co-primary endpoint, acknowledging that migraine attacks are more than just painful episodes (1,3).
It is important to realise that in clinical trials, patients are required to treat migraine attacks only when the headache has reached moderate or severe intensity (2,4,5). Although somewhat artificial and arbitrary, there are a number of methodological advantages to this strategy. When the headache reaches a moderate or severe intensity, there are usually other associated features as well (6), ensuring that migraine attacks are being treated and not featureless and usually milder tension-type headaches. This should minimise the placebo response and increase the validity of the results. Moreover, it also simplifies and standardises the assessment procedure, as measurements are always made from a similar baseline pain intensity rather than from different levels. After all, improvement from severe to moderate pain is not the same as improvement from mild to no pain.
However, the recommended endpoint also has important disadvantages, preventing straightforward extrapolation of the results to clinical practice, where patients usually prefer treating attacks as soon as possible to limit the ictal burden (4,7,8). Moreover, when treating early, prior to development of central sensitisation, efficacy might also be higher (4,5,9,10). In addition, measuring efficacy only until 2 h post-dose does not take into account that approximately one third of patients relapse within 24 h after initial improvement (1). Sustained pain freedom (i.e. pain-free by 2 h post-dose and for the subsequent 22 h, without recurrence of the headache) would be a clinically more relevant endpoint (4,5,11).
Migraine attacks are more than just pain. The headache phase is typically preceded, accompanied, and/or followed by other – often also highly disabling – features; examples are photophobia, phonophobia, nausea, vomiting, fatigue, and mood and cognitive changes (6,12,13). These non-pain symptoms also contribute to the overall burden of migraine attacks and may incapacitate patients for longer periods than just the headache phase (6,12,14). Recent studies have tested these symptoms as secondary endpoints but have not yet used a standardised approach (15). Finally, pain endpoints do not take into account possible treatment-related adverse events.
While sensitive and specific in assessing efficacy of putative new acute migraine treatments versus placebo (16–20) the recommended endpoint (1) has been much less useful in differentiating the effectiveness of active acute treatments. For instance, direct comparator trials using the recommended outcome measure have nearly all failed in detecting significant anti-migraine differences between triptans and simple analgesics (21), yet many experts and patients would agree that triptans are clinically more effective.
In brief, the strategy to assess anti-migraine efficacy by focusing on short-term analgesic effects in patients instructed to wait until the pain is moderate or severe does not seem to reflect good clinical practice. Potentially relevant effect differences between two active treatments may easily be missed. The resolution of pain from a moderate or severe intensity has proven a good primary endpoint to provide evidence of acute efficacy of a new agent against placebo; to compare agents with already proven efficacy, we propose to estimate the “time lost due to an attack”. This novel patient-reported outcome measure takes the aforementioned considerations into account and is likely to be more sensitive in detecting clinically relevant differences between different treatment approaches.
Time lost due to an attack
Experiencing a severe migraine attack is highly disabling (14,22,23). During attacks, patients cannot function normally and lose time that they would rather have spent differently. We propose translating the approach chosen in the Global Burden of Disease studies (14) from a societal to an individual level. Instead of calculating “years lived with disability”, we suggest estimating “time lost due to an attack” (TLA) (Supplement 1), using the following formula:
Here,
Subtracting
We believe that it is easier for patients to rate functioning (a positive concept) than disability (a negative concept).
We suppose that most people are intuitively able to estimate their level of functioning. A more precise definition could be the following: The level of functioning is measured on a scale from 0–100 and corresponds to the proportion of planned activities actually executed. Zero implies that one has been unable to do anything because of pain or associated features; 100 implies that everything has been done as planned.
This approach only leads to a rough estimate, as a constant level of impairment is assumed for the whole attack. Precision increases if patients assess their functioning at multiple time points, which – for practical reasons – should be assessed using digital diaries.
Although single records are possible, they might not reflect the variability of functioning during an attack. Ideally, the number of assessments with TLA for each attack ranges from an essential minimum of 4 (baseline, time of acute drug administration, 2 h post-dose, 48 h post-dose) to a higher number, which may be signal-driven (e.g. onset of a relapse) or pre-planned (e.g. every 2 h for 48 h).
According to current international consensus, “disability lies on a continuum from no disability (full functioning) to complete disability” and fluctuates during a person’s life in different domains and to varying degrees (24). This issue is even more important in migraine attacks, during which the level of disability is likely to vary considerably, because of both the disease and its treatment. Hence, we are aiming to measure an intrinsically variable phenomenon (i.e. disability) in patients affected by a disease that intrinsically induces sudden and relevant variations of functioning (i.e. migraine).
In that case, we assume that impairment
Here,
TLA can then be calculated using the following formula:
Changes of the level of functioning are recorded until the end of the attack, which we define as the return of the level of functioning to the baseline value (SQA).
We propose assessing functioning at fixed time-points; nonetheless, measurements may also be adapted to specific needs. For example, for research purposes, the investigators may set specific time-points for the recording of functioning according to the specific aims of the study. Alternatively, in clinical practice, physicians may fix individual time-points for each patient, taking into consideration their attack profile.
Sleep
During sleep, again, we assume a linear increase or decrease of the level of functioning. Consequently, patients are required to record their level of functioning before dozing off and after waking.
For example, if a patient suffering a migraine attack slept for 8 h and recorded a level of functioning of 50 just before going to sleep and 80 shortly after waking up, during that period, 2.8 h were lost due to the attack.
Should a patient notice a new-onset headache-related decreased functioning on waking up (i.e. an attack that has not been present before going to bed), we advise recording the level of functioning for the evening before retrospectively. Again, we assume a linear decrease of the level of functioning and recommend using the above-mentioned formula.
Impact of premonitory symptoms
If researchers are also interested in impairment caused by premonitory symptoms, which may occur up to 48 h before the attack (13), only minor adaptations are needed. In this case, patients are asked to record impairment retrospectively as soon as they have realised that they have a migraine attack. Alternatively, patients may be asked to register their level of functioning at specific times throughout the study period, irrespective of presence of pain. This would certainly prevent recall bias, but would increase the effort on the part of the participants.
A possible concern may emerge regarding the retrospective assessment of pre-attack functioning. However, we suspect that the time lag from the onset of an attack to the beginning of the recording of functioning will be, in most cases, quite short. In addition, in research settings, periodic recordings of functioning could be planned to prevent any recall bias almost completely.
Relapse
Studies assessing pain as the primary endpoint revealed that pain might return within 46 to 70 h after initial complete resolution 2 h post-dose (“relapse”, previously termed “recurrence”) (1). In this case, the attack cannot be considered as having ceased at 2 h. Conceptually, “relapse” implies that drugs may suppress symptoms of a migraine attack, while the underlying processes continue; symptoms reappear as soon as the effect of the medication fades.
When using TLA as the endpoint, a transient return to the SQA would suggest the end of an attack although more time was to be lost due to the attack. Therefore, we recommend assuming a relapse if the level of functioning drops within 48 h after reaching the baseline value (SQA) at 2 h post-dose. When impairment reaches the baseline value and does not rise any more for the remainder of the 48-h period, the attack has ended. Consequently, in order to detect a possible relapse, patients who reached SQA at 2 h post-dose should be encouraged to continue recording for a further 46 h.
While in the past studies using “pain at two hours” as the primary endpoint often monitored for recurrence during 22 h (25), we feel that this period is too short for the endpoint proposed in this article. Impairment during a migraine attack is not solely due to pain; functional impairment may occur during postdromes as well as because of side effects of the treatment (12,26). In accordance with guidelines published by the International Headache Society (1), we propose monitoring for relapses for 48 h post-dose.
Other measurements
We recommend including relapses of pain as a secondary endpoint. Furthermore, additional information (pain localisation, character, and intensity as well as associated features) may be recorded to validate the diagnosis of a migraine attack.
Finally, we would like to stress the relevance of the TLA for special populations (i.e. patients with chronic headache, medication overuse headache, or psychiatric comorbidities such as anxiety or depression), in which the outcome marker may be a better real-world measure of the improvement with acute medications. For example, in patients with medication overuse headache, drug consumption may decrease when it becomes apparent that acute treatment reduces pain but does not improve functioning.
We believe that assessing the TLA is likely more sensitive and more intuitive to use than the verbal numerical scale (0 = no disability; 1 = mild disability; 2 = moderate disability; 3 = severe disability), the 24-h MSQoL or the Minor Symptoms Evaluation Profile recommended by IHS Guidelines (1). These advantages may help to delineate a more realistic profile of the evolution of a migraine attack; identify small changes, which are particularly relevant when symptoms are mild; and detect fading drug effects or relapses. In addition, the IHS-recommended Global Impact measurement of functioning requires patients to transform the level of functioning into the level of disability and refers to a general concept of normal daily living. By contrast, we suggest considering planned activities that the patient has been unable to do because of the migraine attack as reference to rate the functioning.
To define the relative value of different approaches, it would be useful to compare them in properly designed trials evaluating the advantages and disadvantages of each available measure. In addition, the 24-h MSQoL and the Minor Symptoms Evaluation Profile comprise more questions and undoubtedly require more time and thought. It is likely that even repeated assessments of the level of functioning are less burdensome to patients than answering many different questions. From this point of view, TLA has potential strengths.
Conclusions
Migraine attacks result in reduced fitness for personal, professional, and social activities by limiting the ability to function normally. Often these constraints are not only due to pain, but also to other features of migraine attacks such as fatigue. Therefore, acute migraine outcome measures should not just focus on the short-term analgesic effects of a treatment, but rather on improvement of all migraine symptoms and restoration of normal functioning, in addition to possible impairment caused by the therapy.
We propose TLA, calculated by multiplying the duration and the degree of impaired functioning as the primary endpoint for future studies assessing the efficacy of acute migraine treatments. Of course, this approach is not limited to the evaluation of an acute treatment. If TLA is measured for every attack over the course of several weeks or months, changes in the total amount of lost time after the initiation of a preventive treatment correlate with its efficacy and tolerability.
An additional advantage of TLA as a clinical trial endpoint is that, like in clinical practice, patients may treat attacks as soon as possible and are not required to wait until the headache has worsened to moderate or severe intensity. Finally, while the current 2-h endpoint does not account for relapse, TLA will measure the entire time patients are not functioning normally, from onset to complete resolution.
Although TLA has several strengths, validation is necessary before promoting its use for research purposes and in clinical practice. Accordingly, we have planned a validation study and the development of an open-access web app automatically calculating TLA.
Clinical implications
Current endpoints for acute treatments focus on short-term analgesic effects, not assessing the entire treatment impact. TLA summarises, from attack onset to complete resolution, the status of all migraine symptoms and functioning, including therapy-related impairment. TLA is a new patient-centered paradigm to assess migraine burden and evaluate acute and prophylactic treatments.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024211006048 - Supplemental material for Time lost due to an attack – a novel patient-reported outcome measure for acute migraine treatments
Supplemental material, sj-pdf-1-cep-10.1177_03331024211006048 for Time lost due to an attack – a novel patient-reported outcome measure for acute migraine treatments by Heiko Pohl, Silvia Benemei, David Garcia-Azorin, Joanna Dixon, Elizabeth Huzzey and Michel D Ferrari in Cephalalgia
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.