Abstract
Aim
Little is known about short-lasting unilateral neuralgiform headache attacks with conjunctival injection and tearing (SUNCT) and short-lasting unilateral neuralgiform headache attacks with cranial autonomic symptoms (SUNA). We present our experience with SUNCT/SUNA patients to aid identification and management of these disorders.
Methods
A retrospective review of patient records of one orofacial pain clinic was performed. Inclusion criteria was a diagnosis of SUNCT/SUNA confirmed with at least one follow-up visit.
Results
Six of the 2464 new patients seen between 2015–2018 met the selection criteria (SUNCT n = 2, SUNA n = 4). Gender distribution was one male to one female and average age of diagnosis was 52 years (range 26–62). Attacks were located in the V1/V2 trigeminal distributions, and five patients reported associated intraoral pain. Pain quality was sharp, shooting, and burning with two patients reporting “numbness”. Pain was moderate-severe in intensity, with daily episodes that typically lasted for seconds. Common autonomic features were lacrimation, conjunctival injection, rhinorrhea, and flushing. Frequent triggers were touching the nose or a specific intraoral area. Lamotrigine and gabapentin were commonly used as initial therapy.
Conclusions
Differentiating between SUNCT/SUNA does not appear to be clinically relevant. Presenting symptoms were consistent with those published, except 5/6 patients describing intraoral pain and two patients describing paresthesia.
Keywords
Introduction
Trigeminal autonomic cephalalgias (TACs) are rare primary headache disorders; therefore, knowledge is limited. This is especially true with regard to short-lasting unilateral neuralgiform headache attacks with conjunctival injection and tearing (SUNCT) and short-lasting unilateral neuralgiform headache attacks with cranial autonomic symptoms (SUNA), collectively known as short lasting unilateral neuralgiform headache attacks (SUNHA, or SUN) (1,2). Given that many publications addressing presenting features of SUNHA have been based on patients seeking care within neurology and/or headache clinics, little is known about how these patients present in other settings. It can be expected that patient complaints and presentation of symptoms may be different in an orofacial pain clinic, and it is valuable to understand these nuances to aid clinicians in efficient diagnosis.
Understanding patient and pain characteristics within dental care settings is important because there are reports of patients receiving misguided dental treatment due to a missed diagnosis of SUNHA. This often includes unnecessary dental interventions, such as endodontic and restorative procedures, periodontal surgery, and tooth extraction, all without symptom resolution and not without significant time, cost, and risk of adverse events for the patient (3–6). Contrarily, there are reports of cracked teeth mimicking TACs (7) and of SUNCT exacerbated by myofascial pain (3). Therefore, it is important for information regarding how these disorders present to dental and orofacial pain clinics be reported to improve patient care.
Another difficulty in management of SUNHA is its phenotypical similarities to trigeminal neuralgia (TN) (8). All three disorders are characterized by unilateral, paroxysmal attacks confined within the distribution of the trigeminal nerve. SUNHA has conventionally been distinguished from TN by the presence of distinct cranial autonomic features present during attacks, by attack duration (1–600 sec), and by attacks occurring as single stabs, a series of stabs, or in a sawtooth pattern (9). Trigeminal neuralgia can present with autonomic features (10); however, they are not considered distinguishing diagnostic criteria. Furthermore, classic TN has a maximum attack duration of 2 minutes and often exhibits a refractory period during which time subsequent attacks cannot be triggered (9). Because distinguishing between SUNHA and TN presents diagnostic challenges, it is important to accumulate as much data as possible to elucidate similarities and differences.
Finally, there has been a collaborative effort to establish a classification system for facial pain, known as the International Classification of Orofacial Pain (ICOP), which is aligned with the International Classification of Headache Disorders, 3rd edition (ICHD-3) system for headaches (9,11). The goal of this organization is to more accurately classify facial pains to facilitate precise research to better understand ambiguous disorders such as SUNHA. They have suggested updating the terminology to be more descriptive and proposed the term short-lasting unilateral neuralgiform facial attacks with autonomic signs (SUNFA). While the committees have been discussing the development of these classification systems, it is apparent that multiple disorders that present most often in the “head” (i.e. forehead and temple regions) have different clinical characteristics than when they present in the “face” (i.e. midface and jaws). Furthermore, there are gaps in knowledge about whether there are even meaningful differences between some of the rarer disorders, specifically SUNCT/SUNA. Therefore, the aim of this case series was to contribute to the limited body of knowledge on these disorders by presenting characteristics of patients with SUNCT/SUNA seeking care in an orofacial pain clinic, which will aid future studies and improve patient care.
Methods
We performed a retrospective record review of all patients seen in the TMD, Orofacial Pain, and Dental Sleep Medicine Clinic at the University of Minnesota School of Dentistry from 1 October 2015 until 21 November 2018. Approval to conduct this study (FWA00000312) was provided by The Human Subjects Committee of the University of Minnesota Institutional Review Board.
A preliminary diagnosis of SUNHA was made by orofacial pain residents in a CODA-accredited training program based on ICHD-3 beta criteria (9). Initial patient exams consisted of a 90-min appointment that included a comprehensive health history, review of systems, a detailed clinical examination, and diagnostic testing as indicated. Final diagnosis was made by an orofacial pain specialist with 20 years of experience overseeing the residents (DRN) who re-interviewed and independently examined the patient to verify findings.
The International Classification of Diseases – 9th and 10th editions (ICD-9 and ICD-10) codes for SUNCT (ICD-9: 339.05, ICD-10: G44.051/G44.059) and Other Trigeminal Autonomic Cephalalgias (TAC) (ICD-9: 339.09, ICD-10: G44.091, G44.099) were pulled from the database of patients seen within the clinic. Generic headache codes were also screened: ICD-9: 784.0 and ICD-10: R51and G44.1. Inclusion criteria required a diagnosis of SUNHA with a subsequent appointment where the diagnosis was confirmed (see Figure 1: Flow chart describing inclusion criteria for eligible patients).
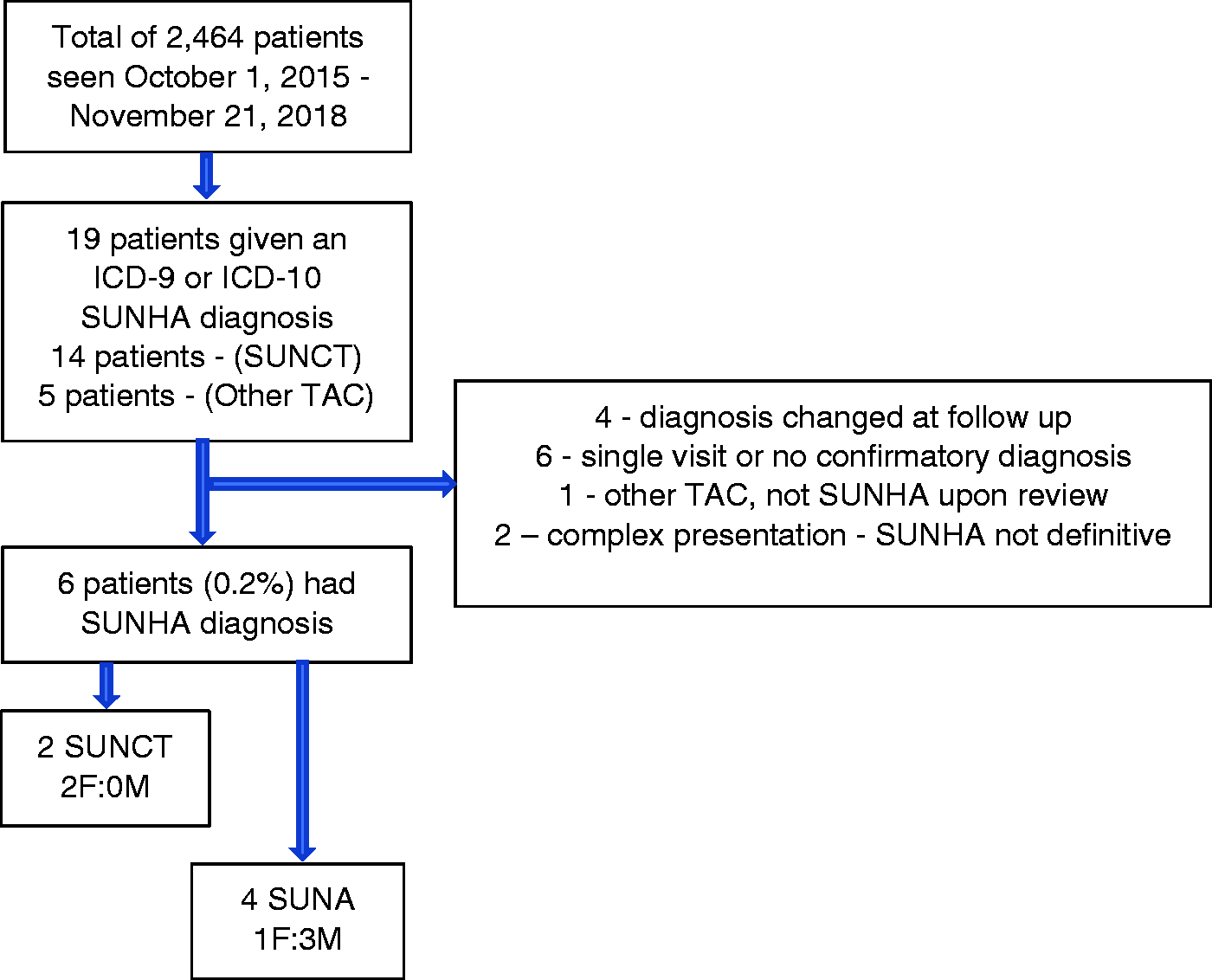
Flow chart describing inclusion criteria.
Results
SUNCT/SUNA diagnosis and prevalence
Six of the 2464 new patients seen between 1 October 2015 to 21 November 2018 met the selection criteria (SUNCT: n = 2, SUNA: n = 4), for a combined clinic prevalence of 0.2% (SUNCT: 0.08%, SUNA: 0.16%). All patients had brain MRIs performed during the diagnostic workup, often before reporting to our clinic. (Table 1: Demographics and neuroimaging).
Demographics and neuroimaging.

Pain characteristics.

Age, gender, race, chief complaint, onset, referral source
Gender distribution was three males and three females (3M:3F). Average age at presentation and years since symptom onset ± standard deviation was 52 ± 15 years of age (range: 26–62 years) and 9 ± 8 years (range: 0.2–20 years) respectively. All patients were Caucasian. Five presented with a chief complaint of “facial pain” and one with “jaw pain”. Four reported no attributable event for pain onset; however, one reported a nasal fracture immediately prior to onset and another reported infrequent pain that had become chronic following an extended period of increased emotional stress.
Patients were referred to us from multiple sources. One was sent from a multidisciplinary facial pain center, one from an oral surgeon, two from a neurosurgeon, and two from their general dentist (Table 1: Demographics and neuroimaging).
Pain characteristics
SUNHA attacks were primarily confined to the V1/V2 trigeminal distributions. Four reported right-sided symptoms and two indicated left-sided pain. Five patients reported intraoral pain, and three of these indicated that the intraoral locations were the primary origin for their pain. For these patients, touching the gingiva overlaying specific maxillary teeth could evoke an attack with no dental disease noted. Pain most often radiated to the ipsilateral orbital or periorbital areas. Two patients indicated radiating auricular pain, one described temporal and intraorally radiating pain, and another masseter muscle pain (Figure 2: Pain patterns of SUNHA patients).

Pain patterns of SUNHA patients. Black area denotes perceived origin(s) of pain. Gray area shows associated referral pattern.
Pain was often described as sharp, shooting, and burning. Several patients were elaborate with their descriptions using terms such as: “dagger-like,” “being in an electric chair,” and “fibrillations” of pain. Two patients reported “numbness” associated with their attacks. Pain was moderate-to-severe with the lowest intensity using a 0–10 numeric rating scale (NRS) of 2 for morning attacks with increasing intensity as the day progressed. Two patients exceeded NRS values, rating their pain as “11/10” and “12/10”. Two patients reported 10/10 pain, and one 5–8/10. All patients reported daily attacks. Most experienced clusters of pain 2–4 times/day, and one described a consistent presentation of 1–5 attacks per hour. Attack duration varied from 1–2 to 206 seconds. Five of the six patients indicated blowing or rubbing the nose, and four indicated toothbrushing as pain triggers. Other triggers are noted in Table 3: Pain characteristics. Four of the six patients reported sleep disturbances associated with their pain. Three stated pain would wake them from sleep periodically, and two specifically would wake when their trigger area was touched at night. The final patient noted an increased frequency of attacks following a night of non-restorative sleep. Two patients reported a prodrome prior to the onset of their SUNHA symptoms. One described this as needing to become very still when they felt their pain begin to “ramp up” to minimize the shocking sensation. Another noted tingling in the involved eye prior to attack onset. Further details can be found in Table 2.
Autonomic symptoms.

Autonomic features
Common autonomic features were lacrimation, conjunctival injection, rhinorrhea, and facial flushing. Most patients presented with multiple autonomic features. All but one patient experienced lacrimation. Two patients specifically reported lacrimation with conjunctival injection, thus meeting the criteria for SUNCT; both also reported rhinorrhea. One of these additionally reported facial flushing. The remaining patients exhibited various symptoms that did not include conjunctival injection and thus led to a diagnosis of SUNA (Table 3: Autonomic symptoms).
Comorbid diagnoses
Patients presented with a varied and complex list of medical comorbidities, often including concurrent or previous diagnosis of trigeminal neuralgia and temporomandibular disorder(s), which were carefully screened for during our diagnostic process. Half of the patients indicated a history of cancer, arthritis, and hypertension. Four had a history of depression (Table 4: Comorbid diagnoses).
Comorbid diagnoses.

Treatment and response
Lamotrigine and gabapentin were found to be the most effective in management of SUNCT/SUNA symptoms. The first patient reported reduced pain intensity from 11/10 to 7–8/10 with lamotrigine (100 mg) and gabapentin (600 mg). The second patient found lamotrigine (400 mg), gabapentin (100 mg), baclofen (40 mg), ice pack application, and onabotulinumtoxinA (100 U left masseter muscle, 100 U maxillary left buccal vestibule) helpful in minimizing symptoms to a “tolerable” level. The third patient reported 75% improvement with lamotrigine (300 mg) and gabapentin (3000 mg). The fourth patient perceived 50% improvement with lamotrigine (50 mg) and pregabalin (600 mg). She had previously reported 75% improvement with verapamil (240 mg) and pregabalin (600 mg) but had to discontinue verapamil due to intolerable side effects. The fifth patient was placed on verapamil (80 mg) as first-line therapy due to co-morbid vestibulocochlear disturbances and risk of vertigo with lamotrigine. She experienced decreased attack frequency. This patient self-discontinued verapamil 1 year later as her pain became “tolerable” without the medication and changed its presentation to one daily attack of 30-min duration and 3/10 intensity. The sixth patient reported complete resolution of symptoms with oxcarbazepine (600 mg).
Discussion
SUNHA disorders are rare, so definitive data for incidence and prevalence are unknown. Studies report a prevalence ranging from 6.6/100,000 to 109/100,000 (12). In comparison, within the same time frame, we had an incidence of TN of 1.8% (44 of the same 2464 new patients). Given that our clinic is a tertiary pain center, it would be anticipated that the SUNHA prevalence of 0.2% within our clinic would be higher than that estimated for the general population. The literature reports SUNCT is more common than SUNA (13), although there is debate as to whether SUNCT is merely a subtype of SUNA (2). In contrast to this, within our population, we had four SUNA patients and two SUNCT patients, although our sample size was small. Additionally, based on the SUNFA terminology suggested by ICOP, which describes the same type of attacks but presenting in the face, 5/6 of our SUNHA patients meet the criteria for SUNFA. This supports the idea that SUNFA and SUNHA are the same disorder with two different anatomical presentations.
MRI brain imaging is recommended for patients suspected of having SUNHA due to its recorded occurrence secondary to other factors (1,2,14). In accordance, five of our patients had recommended imaging prior to arriving in our clinic, and we ordered imaging for the sixth. Three of the imaging studies reported no abnormal findings, one report was unavailable for review, and two showed intracranial abnormalities not thought to be associated with their SUNHA diagnosis. Intracranial pathology has been reported in up to 23% of patients with SUNHA in the literature; therefore it is important to rule this out as an underlying etiology (2,15,16).
Overall, our clinic population demographics were similar to those reported. Prior studies show a male:female (M:F) predilection of 1.5:1 for SUNCT and 1:1.7 for SUNA (14). Overall, we observed a 1:1 male to female ratio for SUNHA; however, we found a M:F ratio of 0:2 for SUNCT and 3:1 for SUNA, likely due to the small sample size. Average age and standard deviation of symptom onset for our patients was 46 ± 12 years, which is similar to that previously reported: 48 ± 17 years (14), 43 ± 9 years (17), and 46 ± 13 for SUNCT/45 ± 16 for SUNA (18). Average age of SUNHA diagnosis within our clinic was 52 ± 15 years, and the patients were undiagnosed an average of 9 years, which is higher than the 2 years reported in a 2019 study of German patients at a medical center (17), and the 7 years reported at a neurology clinic in the UK (19). This delay in diagnosis may be due to differences in healthcare systems between the United States and European countries, differences in clinic type, or to more time by this subset of patients being spent seeing dental providers initially, who may be less familiar with rare headache disorders than primary care physicians.
Presenting chief complaints were similar to prior reports, with five patients describing “facial pain” and one “jaw pain” (14,19). Four of our patients reported no known trigger for the onset of their initial symptoms. The remaining two reported trauma and stress as possible instigating events, both of which have been reported (19–21). Our patients presented with symptoms primarily within the second division of the trigeminal nerve. Five of our six patients (83%) reported intraoral pain, and three (50%) indicated it as the primary location for their pain, thus fitting the terminology of SUNFA. Of note, half of the patients who presented felt the gingiva overlaying a tooth was the primary source of their pain, despite no identified dental pathology. Two of our patients perceived SUNHA pain referral to the tooth. Only one patient in our orofacial pain clinic presented with more conventional ocular pain that radiated to the ear without oral involvement. Odontogenic symptoms have been previously reported in only 6% of cases (19). The higher percentage in our clinic is logical, given that orofacial pain specialists are typically dentists and would be likely to receive referrals for patients with a tooth complaint. Interestingly, one study specifically mentioned the maxillary right premolar as a painful area, which was a common region for pain within our population (4). Another case report cited extraction of a lower central incisor in an attempt to resolve SUNHA symptoms (3). Otherwise, details regarding specific intraoral site manifestations of SUNHA are lacking in the literature which suggests it is rare, not recognized, or a combination.
Affected nerve distributions in our population were slightly different than in prior reports, possibly due to the SUNHA terminology that has historically not included the face until the recently proposed SUNFA criteria. In our population, two SUNHA patients (33%) perceived primary pain in the orbital region, while three perceived referred pain to the ophthalmic distribution of the trigeminal nerve. Two (33%) reported pain radiating to the auricular area. In contrast, ear symptoms were previously only reported in 5% of SUNCT and 11% of SUNA patients (19). This may be explained by the fact that orofacial pain practitioners are familiar with treating temporomandibular disorders (TMD); therefore, auricular symptoms may have been referred for evaluation in our clinic. Four of our six patients (67%) had right-sided pain. Such a side predominance is not typically noted. In an analysis of 222 SUNCT/SUNA patients, 45% had left-sided attacks, 44% had right-sided symptoms, 2% bilateral, 6% side-shifting, and 3% unilateral but not specified (14). Our discrepancy is likely due to small sample size.
Triggering areas for SUNHA attacks were in similar locations for our patients compared to others. Triggers of touching or washing the face, chewing, brushing teeth, talking, and cervical motion were reported. A review noted 72.7% of patients indicated similar triggering events (14). Fifty-six percent of SUNCT patients and 86% of SUNA patients had cutaneous triggers in a separate study (18): 100% of our patients reported triggering areas. This could be due to small sample size. Prior literature reports that SUNHA attacks may be totally spontaneous, totally evoked, or a combination thereof (2,22). All attacks in our study were able to be triggered, with special note made of the autonomic features present in three cases. It was not clear from our data whether some patients experienced spontaneous attacks as well.
Pain descriptors were similar to those frequently reported. The most common adjective for pain quality was “sharp” (67%), “shooting” (50%), and “burning” (33%). The most common term in the medical literature is “stabbing” (2,14,19,23). “Burning” has also been noted (23). Patients within our study provided vivid descriptions to convey their pain. It is not reported if this was common in the other studies. Paresthesia was noted with two patients, and this has rarely been reported elsewhere. One case series of four reported one patient experiencing paresthesia prior to an attack (24), while a review of 222 SUNHA patients in 2013 did not reference any such symptoms (14).
Pain intensity was moderate-to-severe. Historically, pain intensity has been reported to be 10/10 or worst imaginable in 84% of SUNCT patients and 33% of SUNA patients (19). Two of our SUNCT patients exceeded the traditional 10/10 level using the NRS. In our population, the two patients who reported moderate pain intensity ranges (5–8/10 and 2–9/10) were both SUNA patients suggesting that SUNA may be a slightly milder form of SUNHA than SUNCT.
All our patients experienced daily pain attacks. Most experienced a series of attacks, which was reported in 36% of SUNCT and 62% of SUNA patients in one recent RCT (18). Only one SUNA patient described the classic “saw-tooth” pattern of attacks previously reported with 34% of SUNCT and 24% of SUNA patients (18). Attack duration in our population was consistent with that required for diagnosis based on the ICDH-3 criteria: Attacks lasting 1–600 seconds (9), the longest of which in our population was recorded at 206 seconds.
Aggravating factors were consistently related to A-beta fiber triggering. This is similar to that found in trigeminal neuralgia (TN), and there has been debate about whether SUNHA and TN are variations on a spectrum of disorders (8). To differentiate, two patients described attacks lasting longer than the 2 min indicated as the maximum attack length in the diagnostic criteria for TN. One record specifically noted a lack of refractory period. Another patient clearly described the characteristic “saw tooth” pattern of pain described in SUNHA versus TN. None of our patients with characteristics more similar to TN had vascular compression noted on MRI with the exception of the patient without brain MRI data available who had undergone bilateral MVD prior to arriving in our clinic and retained diagnosis of both TN and SUNHA. This patient initially had difficulty articulating the different pains he was experiencing. Alternating trials of oxcarbazepine and lamotrigine would alternately control a portion of the patient’s pain. It was only when the patient and team realized that the oxcarbazepine was managing one set of symptoms and the lamotrigine a distinct set of symptoms and the patient was placed on both medications that both of his pains were sufficiently improved.
Poor sleep quality was also correlated with SUNHA symptoms. As opposed to the case series of 52 SUNHA patients in which none of their nine SUNA cases exhibited symptoms during sleep, two of our three patients reporting sleep disturbance due to attacks were diagnosed with SUNA. The third patient who correlated sleep dysfunction with SUNCT attacks experienced more frequent attacks when her sleep was disturbed; however, she did not specify if attacks would arouse her or if she was already sleeping poorly and then experienced an increase in symptoms.
Two of our patients described being able to “sense” when an attack was to occur. Prodromes have been reported very infrequently in the literature (12,24). This manifestation occurred in our patients who did not have migraine with aura and suggests a possible area for further study.
According to a 2013 review (14), lacrimation, conjunctival injection, and rhinorrhea were the most common presenting symptoms in 100%, 100%, and 48.7% of patients, respectively. These were also the most common presenting symptoms in our population; however, they occurred with frequency of 83%, 33%, and 50% respectively. Flushing was noted in only 7.6% of the review participants but was present in two of our six patients (33%). Of note, none of our patients presented with eyelid edema, fullness of the ear, or miosis and/or ptosis as suggested as part of the diagnostic criteria of the ICHD-3. However, two patients reported subjective facial “numbness” accompanying their attacks. Otherwise, presenting autonomic features in our population was consistent with that reported.
Comorbidities are common in those diagnosed with SUNHA. Some of the more common diagnoses include migraine, trigeminal neuralgia, and pituitary tumors (2,23). Interestingly, none of the patients in our primary study experienced migraine. The literature only has one case report, to our knowledge, describing the association of temporomandibular disorders (TMD) with SUNHA (3), which was present in 5/6 (83%) of our patients, although an incidental finding in two of these cases. Another article described a “background” pain that was not otherwise identified, with features that appear consistent with undiagnosed TMD (24). Given that TMD is a known, common comorbidity with other headache syndromes, it would be expected for patients with another chronic facial pain condition to also present with TMD (25).
Patients generally reported improvement of SUNHA symptoms with prescribed medications; however, complete resolution of symptoms was not reported for any of our cases. Treatment for TACs has been reported with mixed success. Successful treatment for SUNCT with a combination of lamotrigine and topiramate and for SUNA with lamotrigine and gabapentin was reported in one study (18). This combination of medications is reported frequently in the literature; however, other authors indicate that SUNHA appears to be intractable in many cases (26,27). Other medications more frequently reported to treat SUNHA include carbamazepine, corticosteroids, lidocaine, and pregabalin (2,14,28). Case reports are also available regarding onabotulinumtoxinA used to treat SUNHA (29,30), hormone replacement therapy (31), infra- and supraorbital nerve blocks (32), and non-invasive vagal nerve stimulation (33). Surgical options are reported, including occipital nerve stimulation (34), microvascular decompression (35), and deep brain stimulation (36). A recent review regarding treatment options found only lidocaine infusions followed by occipital nerve stimulation to be the more effective than use of lamotrigine for SUNHA (37). Until there is a better scientific understanding of the disorder itself and tailored therapeutic options available, treatment will continue to be palliative and based on symptomology and is increasingly becoming more invasive based on trends in the literature.
A limitation is that our study is retrospective in nature with non-standardized data collection and documentation by different providers, thus limiting some details regarding data such as past medications trialled and phenotypic changes over time. Another limitation is our small sample size; however, given that SUNHA are rare disorders to begin with, our data provides a valuable perspective to the body of knowledge, with the focus on SUNHA symptom presentation in orofacial pain settings.
Conclusions
Based on our clinical experience and review of the literature, differentiating between SUNCT/SUNA does not appear to be clinically relevant; and the terms SUNHA, or SUNFA, are preferred. Presenting symptoms in our orofacial pain clinic were consistent with those previously published, with the exception of a majority of patients describing concomitant intraoral pain as a portion of their chief complaint.
Clinical implications
Patients with SUNHA are encountered in TMD and orofacial pain clinics Differentiating between SUNCT/SUNA does not appear to be clinically relevant Similar to trigeminal neuralgia, triggering of A-beta fibers appears to be a consistent trigger for SUNCT/SUNA Presenting symptoms are typically consistent with those reported in the literature with the exception that those presenting in an orofacial pain practice report intraoral pain more frequently, likely due to referral bias.
Footnotes
Acknowledgements
The authors thank Dr. Aaron Nisley for providing data regarding numbers of patients with trigeminal neuralgia.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
