Abstract
Background
Occipital nerve stimulation is a potential treatment option for medically intractable short-lasting unilateral neuralgiform headache attacks. We present long-term outcomes in 31 patients with short-lasting unilateral neuralgiform headache attacks treated with occipital nerve stimulation in an uncontrolled open-label prospective study.
Methods
Thirty-one patients with intractable short-lasting unilateral neuralgiform headache attacks were treated with bilateral occipital nerve stimulation from 2007 to 2015. Data on attack characteristics, quality of life, disability and adverse events were collected. Primary endpoint was change in mean daily attack frequency at final follow-up.
Results
At a mean follow-up of 44.9 months (range 13–89) there was a 69% improvement in attack frequency with a response rate (defined as at least a 50% improvement in daily attack frequency) of 77%. Attack severity reduced by 4.7 points on the verbal rating scale and attack duration by a mean of 64%. Improvements were seen in headache-related disability and depression. Adverse event rates were favorable, with no electrode migration or erosion reported.
Conclusion
Occipital nerve stimulation appears to offer a safe and efficacious treatment for refractory short-lasting unilateral neuralgiform headache attacks with significant improvements sustained in the long term. The procedure has a low adverse event rate when conducted in highly specialised units.
Keywords
Introduction
Short-lasting unilateral neuralgiform headache attacks (SUNHA) are a rare form of primary headache syndrome characterized by attacks of unilateral pain occurring in the trigeminal distribution. The pain is of moderate to severe intensity and is accompanied by at least one cranial autonomic symptom or sign, ipsilateral to the pain (1). Attacks can last 1–600 seconds, with attack frequency ranging between 1 and over 200 a day (1,2). The International Classification of Headache Disorders (ICHD-3beta) describes two subsets of the syndrome defined by the presence of ipsilateral tearing and conjunctival injection: Short-lasting unilateral neuralgiform headache attacks with conjunctival injection and tearing (SUNCT) (where both lacrimation and conjunctival injection are present) and short-lasting unilateral neuralgiform headache attacks with autonomic features (SUNA) (where only one of or neither of lacrimation or conjunctival injection are present) (1). The chronic form of SUNHA is defined as attacks occurring for over one year with remission periods of less than one month. Seventy percent of patients suffer the chronic variant (2).
First line treatment for the condition is lamotrigine, but other drugs such as topiramate, oxcarbazepine and gabapentin have been reported to be effective (3–7). Temporary response to lidocaine infusions and greater occipital nerve blocks have also been reported (8–10). Short-lasting unilateral neuralgiform headache attacks can be difficult to treat medically, and some patients prove intractable to treatment. In the past, such patients have been subject to destructive procedures of the trigeminal nerve with poor long-term outcomes (11). However, recent meta-analysis suggests that microvascular decompression of the ipsilateral trigeminal nerve in selected patients with intractable SUNHA and neurovascular contact may be useful (12). Long-term outcome data from this procedure is awaited.
Peripheral (occipital nerve stimulation) and central (ventral tegmental area deep brain stimulation) neurostimulation techniques have been carried out with some success for the syndrome. Our group have recently reported a cohort of 11 patients who have undergone deep brain stimulation for SUNHA, with a median improvement in daily attack frequency of 78% with an at least 50% reduction in attack frequency seen in nine patients (13). Deep brain stimulation is, however, highly invasive and associated with a small risk of fatal intracerebral hemorrhage (14). Occipital nerve stimulation (ONS) appears potentially useful for chronic migraine based on a small number of randomized trials and for chronic cluster headache based on a number of open-label series (15–22). The safety profile has been dominated by high lead migration and infection rates. The experience of ONS in the treatment of SUNHA is limited to a single series of nine patients (23). In this prospective open-label series, we report the long-term follow-up of 31 medically-intractable chronic SUNHA patients treated with ONS who had tried numerous other treatment options available to them within the UK’s National Health Service.
Methods
Patients
Under the supervision of our institution’s Clinical Effectiveness Supervisory Committee (CESG), with arrangements for clinical governance, consent and audit, we offered ONS to medically-intractable SUNHA. The procedure was provided on the basis of a “humanitarian intervention”. In addition, ethic board approval for data collection and publication was granted by Northwick Park Hospital Research Ethics Committee, Hampstead, London, UK.
Patients were reviewed in a multidisciplinary clinic and were implanted over a period from 2007 to 2013. All patients fulfilled International Classification of Headache Disorders 2nd edition at time of diagnosis (24). Although four patients had longer duration attacks (between 15–45 minutes) than in the ICHD criteria, this has been described in other large clinical series, and all had had some attacks less than 10 minutes during the course of the syndrome (2). Patients with longer lasting attacks underwent indometacin trials (oral or intramuscular) to rule out indometacin-sensitive headaches (25). Unlike chronic cluster headache or migraine, intractability is not defined for SUNHA and so local criteria were devised. Patients were considered for ONS if they had had disabling chronic SUNHA for at least two years and had failed adequate trials of lamotrigine, topiramate, gabapentin, pregabalin and one of either carbamazepine or oxcarbazepine (23). Agents were selected on the basis of reported efficacy and local experience (2,26). Failed trials were defined as a lack of response, intolerable side effects or contraindications to use of medication. Microvascular decompression was not available to the patient group during the study period. Given the lack of evidence to support its use as a predictor of ONS outcome, response to greater occipital nerve block was not considered in patient selection (27).
Surgical procedure
Demographic data.
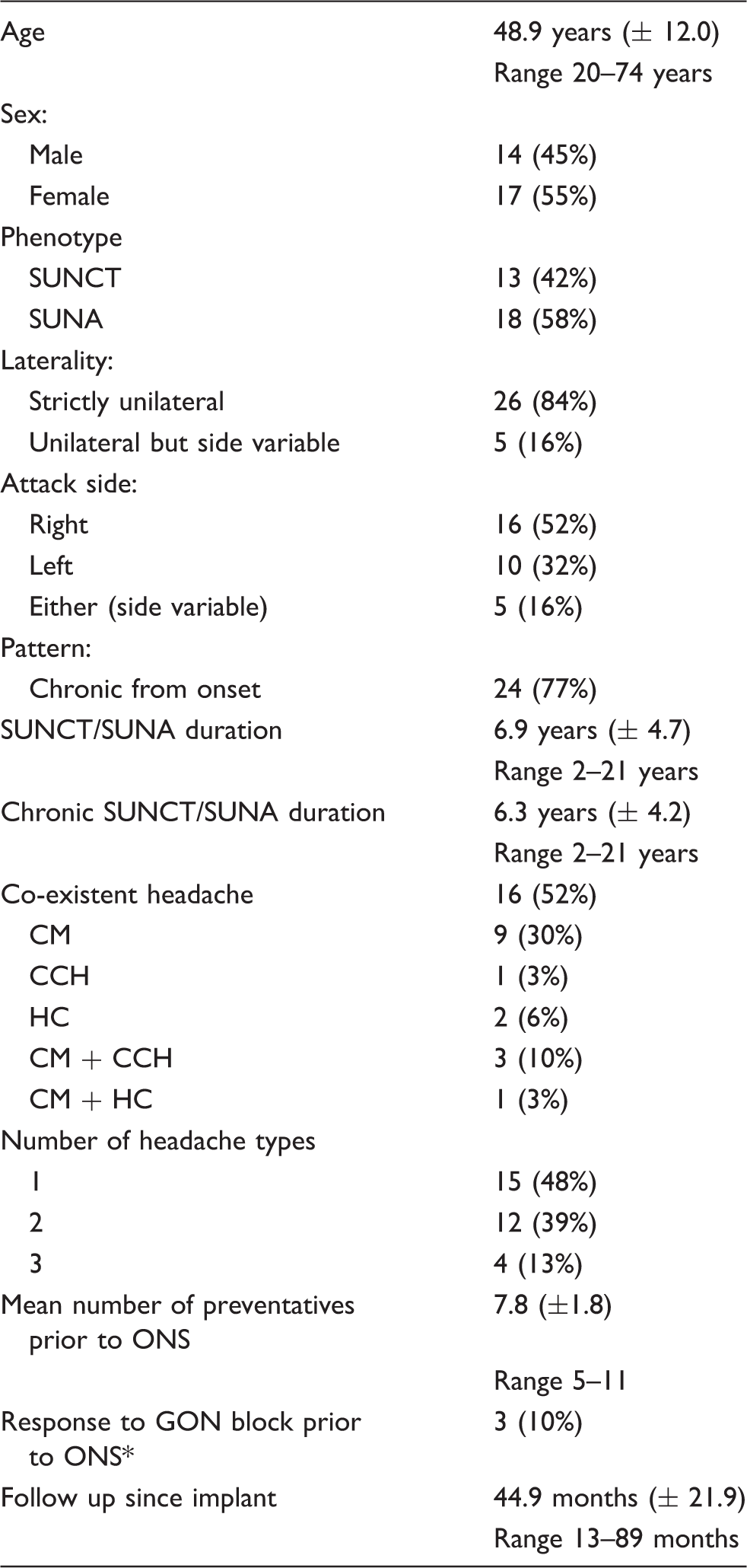
CM: chronic migraine; CCH: chronic cluster headache; GON: greater occipital nerve; HC: hemicrania continua; ONS: occipital nerve stimulation; SUNA: short lasting unilateral neuralgiform headache attacks with autonomic features; SUNCT: short lasting unilateral neuralgiform headache attacks with conjunctival injection and tearing.
Response to GON block defined as a more than 50% reduction in daily attack frequency lasting at least two weeks.
Patients were provided with remote controls allowing them to adjust their stimulation amplitude, but were asked to use continuous stimulation where possible. Polarity of the electrodes was adjusted during follow-up visits to ensure comfortable bilateral paresthesia in the occipital region. Stimulation settings and changes were recorded at each visit. Medications were changed as needed at the discretion of the headache specialist.
Data collection
The primary outcome measure was the change in mean daily attack frequency. Secondary measures included the clinical response to ONS (defined as a more than 50% reduction in attack frequency), changes in attack severity and duration, headache-specific disability scores, quality of life scores and affect scores.
Outcome data was collected and recorded prospectively. Prospective headache diaries were kept for one month prior to implant and then two weeks prior to each follow-up visit. All patients were shown how to keep a paper diary recording the duration and pain intensity of their attacks and asked to record all daily attacks. Patients were seen at three-monthly intervals post-implant for one year and at least six to 12 monthly thereafter, depending on clinical condition. Data was collected on demographics, diagnosis, treatments, attack frequency, severity and duration, headache disability scores, quality of life scales, affective scores, ONS settings and adverse events. Although specific tools for measuring the associated disability of SUNHA have yet to be validated, Migraine Disability Assessment Scores (MIDAS) and Headache Impact Test-6 (HIT-6) scores were conducted. These scores have been used extensively in the assessment of primary headache disorders and previously used to monitor the response of trigeminal autonomic cephalalgias, including SUNHA, to ONS (18,23). Euro-QoL (EQ5D), Short Form-36 questionnaires (SF36), Beck Depression Inventory (BDI-II), Hospital Anxiety (HAD-A) and Depression (HAD-D) scores were used to monitor quality of life and affect pre-and post-implant.
Data was collected prospectively from 2007 until 2015 and entered onto a clinical database (Microsoft Excel, Microsoft Corporation, Redmond, WA, USA).
Statistics
All statistical analyses were conducted using IBM SPSS Statistics version 22 (IBM Corp. Int.). A last observation carried forward technique was used in the case of missing data. Descriptive statistics were summarised as appropriate. Data is presented as mean ± standard deviation or range where appropriate. Paired and independent t-tests were used to compare treatment effect. All statistical tests were two-sided with a significance level of 95%.
Results
Patient demographics
Thirty-one patients (14 male) with SUNHA were implanted between August 2007 and December 2013. Provisional data from some of these patients had been previously included in a publication from 2014 (23). Patient demographics and baseline headache characteristics are shown in Table 1. Mean age at implant was 48.9 years (range 20–74). Seventy-seven percent were chronic from onset, with mean chronic phase duration of 6.3 years (range 2–21 years). Patients had tried a mean of 7.8 preventative medications prior to ONS (range 5–11), summarised in Supplementary Table 1.
Sixteen patients (51.6%) reported other headache phenotypes in addition to SUNHA: Nine had chronic migraine, three had both chronic migraine and chronic cluster headache, two had hemicrania continua, one had chronic migraine and hemicrania continua, and one had chronic cluster headache (Table 1). All kept separate diaries for each phenotype throughout follow-up.
Whole cohort
Mean follow-up for all patients was 44.9 months (range 13–89 months). At final follow-up, mean daily attack frequency had reduced by 69% (± 38.2%) from 98.5 to 31.2 attacks (p = 0.012). Figure 1(a) shows the change in daily attack frequency over the follow-up period. A 50% or more reduction in daily attack frequency was observed in 77% (n = 24) of patients. Twelve patients were pain free at final-follow up with a mean complete remission time of 36.5 months (10–78). Significant improvements were also seen in attack severity (5.5 points on verbal response scale (VRS); p < 0.001) and attack duration (64%; p = 0.001) (Table 2). At final follow-up, reductions in both MIDAS (40.6 points; p = 0.053) and HIT-6 (4.7 points; p = 0.017) were observed, although only HIT-6 reduction was significant. Affect scores showed a significant reduction in HAD-D but non-significant reductions in HAD-A and BDI-II. The SF36-P, SF36-M and EQ-5D all showed non-significant improvement, but EQ-VAS showed a significant improvement of 13.1 points (p = 0.028) (Table 3). Mean patient estimated improvement across all patients was 72% (± 34.0)
Reduction in daily attack frequency following occipital nerve stimulation. (a) Reduction in mean daily attack frequency of the whole cohort; (b) Reduction in mean daily attack frequency of those with short lasting unilateral neuralgiform headache attacks alone compared to those with multiple phenotypes. Summary of attack outcome measures. CI: confidence interval; ONS: occipital nerve stimulation; SD: standard deviation; SUNHA: short lasting unilateral neuralgiform headache attacks; VRS: verbal rating scale; mins: minutes p value < 0.05; ‡Wilcoxon Signed Rank Test Table accompanies Figure 1(b) in showing the reduction in mean daily attack frequency of those with short lasting unilateral neuralgiform headache attacks alone compared to those with multiple phenotypes. *p value < 0.05

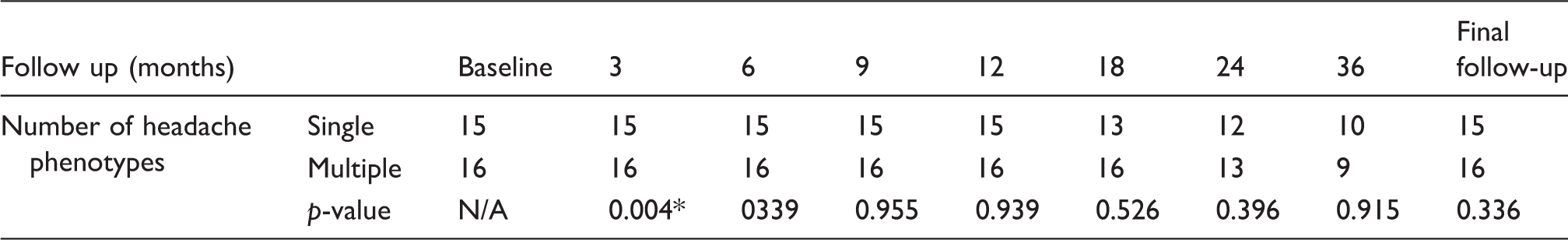
Short lasting unilateral neuralgiform headache attacks alone
In the 15 patients with SUNHA alone, the mean follow-up was 44.2 months (range 13–81). A 50% reduction in daily attack frequency was seen in 67% (n = 10) at final follow-up. Mean daily attack frequency reduced by 61.9% (±43.1) from 152.3 to 57.5 (p = 0.025). Figure 1(b) shows the reduction in daily attack frequency over the follow-up period in this cohort. Reductions were also seen in attack intensity (5.0 points on VRS; p = 0.001) and attack duration (68%; p = 0.011). Significant improvements were seen in MIDAS (71.4 points, p = 0.023) but not in HIT-6. Affect scores showed significant improvements in HAD-A, HAD-D and BDI-II (Table 3) but no significant changes were seen in any of the other quality of life scores. Mean patient estimated improvement was 70% (± 38.4).
Multiple phenotypes including SUNHA
Summary of headache related disability, affect and quality of life scores with occipital nerve stimulation.
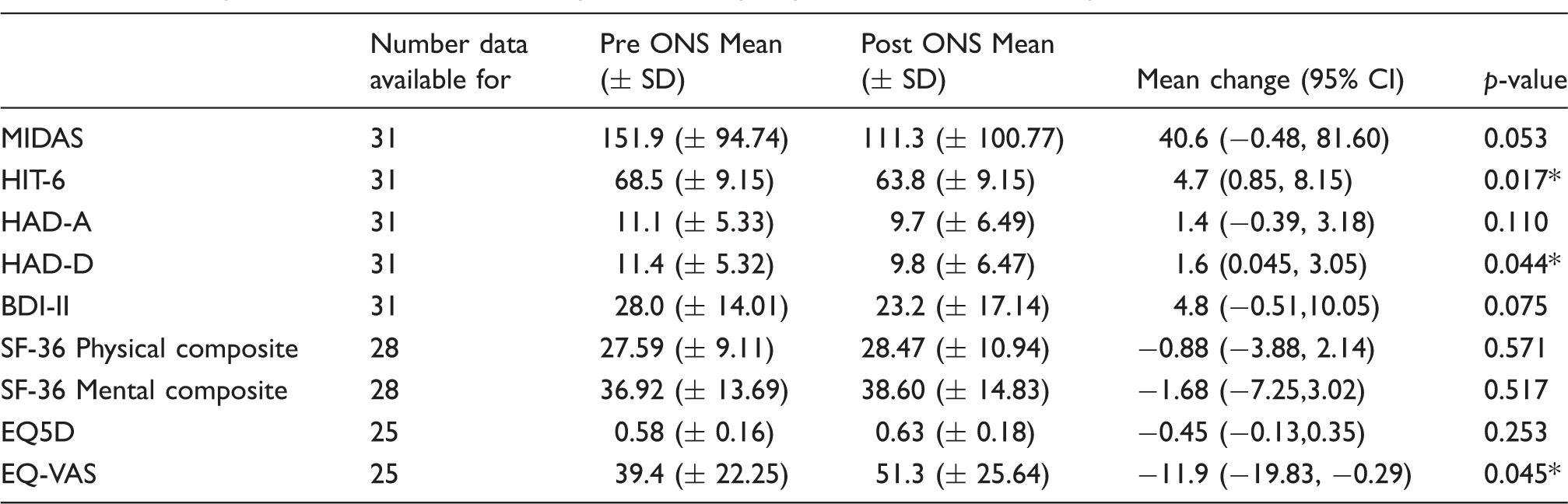
ONS: occipital nerve stimulation; MIDAS: Migraine Disability Assessment Scale; HIT-6: Headache Impact Test; HAD-A: Hospital Anxiety and Depression scores – anxiety specific; HAD-D: Hospital Anxiety and Depression scores – depression specific; BDI-II: Becks Depression Inventory; SF-36: Short Form 36; EQ5D: Euro-QoL 5D Index; EQ-VAS: Euro-QoL visual analogue score; CI: confidence interval *p value < 0.05
In those whose SUNHA responded to ONS, 6/11 chronic migraines, 1/3 chronic cluster headaches and 2/3 hemicrania continua also showed improvement (defined as a more than 50% reduction in daily attack frequency for CCH and a more than 30% reduction in moderate-to-severe headache days for chronic migraine and hemicrania continua). In those SUNHA non-responders, 1/2 of the co-existent chronic migraines responded to ONS.
Concomitant drug use
Twenty-five patients were taking preventative medication at time of implant (mean two drugs). At follow-up, six patients previously on preventative drugs had stopped all medication and 10 had reduced the dose or number of drugs taken from baseline. In responders to ONS, 18/24 had medications started during follow-up; however, in only three cases was this medication for SUNHA and in one patient medication for SUNHA was started after the ONS was removed.
Time to effect and recurrence of attacks
The mean time of all patients to reach a 50% improvement was 5.7 months (range 1–19.0). In responders, this time was 5.3 months (range 1–12.0). A significant difference was seen in daily attack frequency between baseline and three months in responders (85.3 attacks; p = 0.031), but no such change was observed between month three and any other time point.
Twelve patients had their ONS turned off for a period of time (median eight months, range 2–60 months) – eight due to battery depletion, two due to lack of efficacy, one due to explantation, and one due to technical reasons. In 10 of these, SUNHA attacks worsened with ONS off within a mean time of 1.2 months (range 1–3).
Stimulation settings
All patients were encouraged to use continuous stimulation. A range of settings was used to achieve the widest area of occipital paresthesia possible. As a group, the range for amplitude was 0.20–4.65 volts (mean 1.4 volt), pulse width 294–480µs (mean 441.5µs) and frequency 30–170 Hz (mean 78.3 Hz).
Adverse events
Adverse events.

ONS: Occipital nerve stimulator; IPG: implantable pulse generator
A total of 11 events required surgical intervention, the majority being revisions of the ONS system to rechargeable batteries.
Discussion and conclusions
This cohort is the largest reported for ONS in intractable chronic SUNHA. In line with previous open-label series of chronic cluster headache and the single series of SUNHA patients, we suggest that ONS may be a beneficial treatment with sustained effects for refractory patients (18–23). Our series of 31 complex patients reported a significant reduction in daily attack frequency of 69%, with 12 patients (39%) recording complete pain freedom at follow-up. Overall, 77% of patients had a more than 50% reduction in daily attack frequency with ONS. Significant improvements were seen in the headache-specific disability score HIT-6 and quality of life component EQ-VAS, but not in other quality of life measures or MIDAS.
Occipital nerve stimulation was first used for presumed occipital neuralgia, but has since been used for intractable chronic migraine and chronic trigeminal autonomic cephalalgias (28,29). Controlled trials in chronic migraine have shown limited and conflicting evidence of efficacy (15–17). Although controlled trials of ONS in trigeminal autonomic cephalalgias have not been completed, reviews of the open label series of chronic cluster headache report a clinical response in 67% of patients (29). In the single published series of ONS for refractory SUNHA, a cohort of nine patients was reported using headache load as primary outcome (23). In that small series, 89% of patients recorded a headache load reduction of at least 50% at a median of 38 months.
Clinically and statistically important reductions in HIT-6, HAD-D and EQ5D-VAS scores were seen following ONS, but no significant improvements were seen in other quality of life measures or migraine specific scores. Reasons for this may include sample size, long duration of chronic pain (seven years), co-existent headache disorders, and the usefulness of generic scales in measuring quality of life in headache patients. The cohort continued to suffer a mean total of nearly 90 minutes of pain per day, which, although a significant reduction from the pre-ONS value of 9.5 hours is still a considerable burden. In those with multiple headache types, not all other headaches improved. The impact of continued disabling chronic headaches in this sub-population may negatively influence quality of life at follow-up, and this is supported by a more favorable quality of life profile in those with SUNHA alone than in multiple phenotype patients (Table 3).
In keeping with previous ONS series for other headache conditions, our cohort reported an average delay of six months before reporting a clinical response and a return of attacks after an average of one month when stimulation was stopped (16,18–20,22,23,29). The delay and reversibility of effect reflects the slow but reversible neuroplastic response proposed to underlie ONS treatment.
The adverse event profile of ONS for headache has been a major cause of criticism regarding the treatment. Previous ONS series reported high rates of lead migration (7–50%), lead fracture (10–15%) and infection (10–24%) (15,18–21,30). Our cohort has a favorable adverse event profile with no episodes of lead migration, fracture or erosion and only one patient (3%) suffering a superficial wound infection requiring medical management only. The major need for surgical intervention was battery replacement, and with developments in neuromodulation technology leading to the use of longer lasting rechargeable batteries the rates of battery replacement should decrease in the future.
The major weakness of this study is the lack of placebo or blinded stimulation. However, this has been an issue throughout ONS research and with such a rare syndrome as SUNHA it is unlikely a large randomized trial will ever be completed. Although there is undoubtedly a placebo effect in headache treatment it is unlikely our findings are explained by placebo alone. The intractable nature of the group, the delay to clinical response in keeping with other trigeminal autonomic cephalalgia cohorts, the stable long-term response and the return of attacks when stimulation is stopped would argue against a pure placebo response. It has also been reported that the placebo response rates for ONS in migraine are low (6%, 17% and 20%), and there is no evidence to suggest placebo response is different in other headache conditions (15–17).
The patient group included a high proportion of patients (52%) with multiple chronic headache phenotypes. These patients had all been carefully phenotyped by headache specialists, and patients were able to differentiate the different types, recording them in separate diaries to allow outcomes to be compared. Although this percentage may seem excessive, other SUNHA cohorts have also described similar patterns (2,31). It is speculated that those with multiple headaches fare worse with treatment; however, our data, albeit limited, does not support this. Occipital nerve stimulation has been used to treat a number of headache conditions and we view it as a potentially useful treatment for those with multiple headache types, as a single treatment modality can improve multiple conditions. More work is needed on the outcomes of such complex patients to all treatments not just ONS, but our data suggests that they should not be deprived of ONS on the assumption that complex patients do not respond well.
Strengths of the study include the sample size, prolonged follow-up, prospective collection of data and the clinical relevance of the data. The cohorts were not selected by vigorous study inclusion criteria, but were patients in a specialist centre implanted due to clinical need when ONS was only available as a last-line treatment option. The cohort will be similar to patients in other units conducting implants for headache conditions, and so our findings should be widely and clinically applicable.
Microvascular decompression and deep brain stimulation have also been reported in open-label series to have efficacy in SUNHA disorders of around 75%, similar to the response rate in this ONS series (12,29,32–34). The surgical experience of the team, the invasiveness of surgery, the associated risks, the need for implanted hardware and cost of treatment will all influence treatment choice. Bearing these factors in mind, our current pathway is that patients with intractable SUNHA first undergo microvascular decompression (if there is evidence of trigeminal neurovascular contact on neuroimaging), and ONS be reserved for those with no neurovascular compromise or who fail to respond to microvascular decompression. This order is based on our own clinical experience, technical expertise and treatment availability.
Our series suggests that ONS may provide a significant and sustained reduction in attack frequency in intractable short-lasting unilateral neuralgiform headache attacks even after four years follow-up. Adverse event rates are low when implants are conducted in specialist centers. However, given the invasiveness and cost of treatment, ONS should be reserved for those who fail all other appropriate treatment options.
Clinical implications
Short lasting unilateral neuralgiform headache disorders are intensely painful and highly disabling. A significant minority of these patients are intractable to medical treatments. Occipital nerve stimulation (ONS) is associated with sustained reduction in daily attack frequency, severity and duration over a four-year period in some patients with intractable short lasting unilateral neuralgiform headache disorders. When undertaken by highly specialised centres, the complication rates associated with ONS implantation are relatively low.
Footnotes
Acknowledgements
We would like to thank our Headache Specialist Nurses, especially Susie Lagrata, for their help with completion of the clinical database and management of the patients. We also thank the patients and their families for their help with this project.
Contributors
SM: Recruitment of subjects, analysis and interpretation of data, drafting and revision of manuscript. LW: Performed surgery and manuscript revision. MSM: Study concept, recruitment of subjects, interpretation of data and manuscript revision.
Ethics approval
Ethical approval for the collection, storage and publication of data was obtained from subjects undergoing ONS implantation. Ethic board approval for data collection and publication was granted by Northwick Park Hospital Research Ethics Committee, Hampstead, London, UK.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SM has received educational and travel grants from St Jude Medical and Medtronic. LW declares no competing interests. MSM serves on the advisory board for Allergan, St Jude Medical and Medtronic and has received payment for the development of educational presentations from Allergan, Merck Sharpe and Dohme Ltd, Medtronic and electroCore.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
