Abstract
Purpose
To define the melanopsin and cone luminance retinogeniculate pathway contributions to photophobia in healthy controls and migraineurs.
Methods
Healthy controls and migraineurs were categorized according to the International Classification of Headache Disorders criteria. Photophobia was measured under full-field illumination using electromyography in response to narrowband lights spanning the melanopsin and cone luminance action spectra. Migraineurs were tested during their interictal headache-free period. Melanopsin-mediated post-illumination pupil responses quantified intrinsically photosensitive Retinal Ganglion Cell (ipRGC) function.
Results
A model combining the melanopsin and cone luminance action spectra best described photophobia thresholds in controls and migraineurs; melanopsin contributions were ∼1.5× greater than cone luminance. In the illumination range causing photophobia, migraineurs had lower photophobia thresholds (∼0.55 log units; p < 0.001) and higher post-illumination pupil response amplitudes (p = 0.03) than controls.
Conclusion
Photophobia is driven by melanopsin and cone luminance inputs to the cortex via the retino-thalamocortical pathway. In migraineurs, lower photophobia thresholds reflect hypersensitivity of ipRGC and cone luminance pathways, with the larger and prolonged post-illumination pupil response amplitude indicative of a supranormal melanopsin response. Our findings inform artificial lighting strategies incorporating luminaires with low melanopsin excitation and photopic luminance to limit the lighting conditions leading to photophobia.
Keywords
Introduction
Photophobia (1) occurs in around 80% of migraineurs during the ictal period (2) and can exacerbate headache (3). Hypersensitivity to light is known to involve cone and rod photoreceptor inputs to conventional retinogeniculate visual pathways (4) and in migraine-related photophobia, the neuroretinal cone and rod responses are supranormal during the ictal period (5,6), then recover during the interictal period (7). Patients with retinal dystrophies affecting cones also experience photophobia and present with supranormal neuroretinal rod responses (8–10). Evidence that melanopsin only, rod-cone knockout mice show light aversion (11), and people with complete loss of rod and/or cone photoreceptors experience photophobia (12,13), supports a role for melanopsin expressing intrinsically photosensitive retinal ganglion cells (ipRGCs) in this process. The contribution of ipRGC pathways to photophobia in healthy participants and migraineurs has not however, been directly established.
An initial estimate of the photophobia action spectrum was neither completely described by cone luminance (photopic Vλ) or the rod spectral sensitivity (scotopic V’λ) (14). In contrast, human brightness estimation is mediated by ipRGCs and cone (luminance) pathways (15,16). The intrinsic melanopsin response of ipRGCs has higher sensitivity to shorter wavelength lights (∼480 nm; bluish/cyanish appearing lights) (17–19) that cause more ocular discomfort (lacrimation) than luminance-matched longer wavelengths (reddish lights) (20), and short-wavelength attenuating lenses reduce lacrimation in people who experience photophobia (21). Primate ipRGCs receive cone and rod inputs (17), drive the pupil light reflex (PLR) (18,19,22) and input to image forming vision (23,24) and brightness perception (15,16). Here we determine the action spectrum of photophobia to evaluate if it is mediated independently via the intrinsic melanopsin pathway, or in combination with the cone luminance pathway. Conventional electroretinographic measures of neuroretinal function do not quantify melanopsin (25) and so we use pupillometry protocols designed to isolate the melanopsin-mediated PLR (19,22) in healthy controls and migraineurs. We hypothesized that if photophobia involves melanopsin signalling, then hypersensitivity to light might be accompanied by supranormal melanopsin-mediated pupil amplitudes.
Methods
Participants
Experiments were conducted in accordance with the Declaration of Helsinki and the Queensland University of Technology (QUT) Human Research Ethics Committee approval (#1800000843). Participants were recruited through email advertisements sent to staff and students in the QUT Faculty of Health. Informed consent was obtained from participants before the experiment. The comprehensive ophthalmic testing included fundus examination with indirect ophthalmoscopy, optical coherence tomography (RS-3000 OCT RetinaScan Advance; Nidek Co. Ltd., Aichi, Japan), fundus photography (Canon Non Mydriatic retinal camera, CR-DGi; Canon Inc., Tokyo, Japan), visual acuity (Bailey-Lovie), slit-lamp examination (Takagi SM-70N 330-2 Iwafune; Nakano-shi, Nagano-ken, Japan), intraocular pressure measurements (iCare TA01, Helsinki, Finland) and color vision assessment (Ishihara pseudoisochromatic plates; Kanehara Trading Inc., Tokyo, Japan; Lanthony desaturated panel D-15; Richmond Products, Albuquerque, NM, USA). All participants were naïve to the purpose of the experiment.
Twelve healthy controls [mean (± SEM) age = 29.75 ± 1.92 years, six females] and 11 migraineurs (mean (± SEM) age = 35.91 ± 2.85 years, nine females) with normal trichromatic color vision, crystalline lens opacification (LOCS III) grade ≤ 1 (grade = 0 in 22/23 participants indicating the absence of cortical, subcapsular and nuclear opacification) and no ophthalmic disease were included in the study sample. There was no significant age difference between the groups (t21 = 1.82, p = 0.08). All control participants were free from headaches (in total ≤ four headaches in the past year) and had no history of migraine. Eligible migraine participants met the International Classification of Headache Disorders criteria (26) and were diagnosed by their general practitioner or neurologist. All migraine participants were tested during their interictal headache-free period, with the last headache episode at least one week prior to the study, and completed a photophobia questionnaire (Supplementary Table S1) and the Migraine Disability Assessment Score (MIDAS) questionnaire to assess the headache characteristics, including the grades of migraine severity (Table 1).
Migraine characteristics.

Optical setup and calibration
The test stimuli (Supplementary Table S2) were six narrowband primary lights (λmax (full-width at half maximum): 461 nm (21 nm), 499 nm (29 nm), 509 nm (28 nm), 525 nm (32 nm), 592 nm (15 nm), and 630 nm (13 nm)) presented in full-field illumination to the left eye through a 2.54 cm aperture of an 8.38 cm diameter Ganzfeld integrating sphere (Newport Corporation, Fountain Valley, CA, USA). The output of the primary lights was controlled by an Arduino based stimulation system with an LED driver (TLC5940) and microcontroller (Arduino Uno SMD R3, Model A000073) with custom-designed software programmed in X-code (version 3.2.3) driven by an Apple Mac Pro Quad-Core Intel computer (64-bit, Apple Inc., Cupertino, CA, USA) (15). The TLC5940 had 16 independent channels with 12-bit resolution per channel and controlled the LED output by the pulse width modulation frequency which generates stimuli up to ∼488Hz (23), beyond the critical flicker fusion frequency of human vision. The spectral power distributions of the lights were measured with a spectroradiometer (StellarNet EPC200C, Tampa, FL, USA) and the radiometric (Watts.cm−2.s−1 converted to log quanta.cm−2.s−1) and photometric (cd·m−2, Lux) outputs measured with an ILT1700 Research Radiometer (International Light Technologies, Inc., Peabody, MA, USA) as a function of voltage; linearization coefficients were computed for each primary light (15,16).
Electromyography (EMG)
Photophobia thresholds were estimated at all stimulus wavelengths using electromyography (EMG) according to established protocols (14,27). Detailed descriptions of the EMG recordings and digital signal processing are provided in the Supplementary Information (Figure S1). To span the light levels commonly inducing photophobia, the stimulus irradiances ranged between 13.0 and 15.7 log quanta.cm−2.s−1 (2.5 to 5,011 cd.m−2) (Supplementary Table S2). The photophobia threshold was estimated from each average EMG recording using a criterion signal-noise ratio (14) (Supplementary Figure S1(e)). Subjective photophobia thresholds were also estimated using verbal reports of the stimulus level that caused discomfort.
Pupillometry
The dark-adapted consensual pupil light reflex (PLR) was measured using a Ganzfeld in response to narrowband 1 s stimuli pulses presented in the illumination range causing photophobia. The stimuli were either short-wavelength light (λmax = 461 nm, corneal irradiance: 15.5 log quanta.cm−2.s−1) with high melanopsin excitation (3.0 log µWatts.cm−2) or long-wavelength light (λmax = 630 nm, corneal irradiance: 15.5 log quanta.cm−2.s−1) with low melanopsin excitation (−0.3 log µWatts.cm−2). The PLR for each participant was analyzed in accordance with the international standards in pupillography (22). The post-illumination pupil response (PIPR) amplitude provides the direct measure of melanopsin function (18,19,22). Our dark-adapted pupillometry measures the melanopsin-mediated PIPR to the same spatio-temporal stimulus pulse as in the photophobia threshold experiment under conditions affording higher signal-to-noise ratios than with light-adapted protocols (22). Light-adapted paradigms desensitise the photoreceptors and their post-receptoral mechanisms and inadvertently limit the available dynamic range by attenuating the baseline pupil diameter and afferent pupil light response (28). Of the 23 recruited participants (12 controls and 11 migraineurs), five were unable to provide reliable pupillometry recordings due to frequent blinking and/or intolerance to the test stimulus intensity (two controls and three migraineurs).
Experimental protocol
We performed photophobia threshold measurements using EMG, then quantified melanopsin function with pupillometry. Testing sessions were 2.0–2.5 hours duration including electrode set-up, a 10 min dark adaptation (<1 Lux) to minimize any effect of pre-testing light exposure and breaks. The participant’s head position was stabilized with a chin rest. For each threshold measurement, participants were instructed to look inside the Ganzfeld with their left eye and were adapted for 60 s to dim white light (3.0 cd.m−2; 1931CIE x = 0.332, y = 0.267; CCT = 3973°K) to standardize the pre-adaptation light exposure and desensitize the rod photoreceptors (29). Participants completed all the measurements with natural pupils. The mean pupil diameter with the adapting background light (3.0 cd.m−2) was 5.50 ± 0.18 mm (±SEM). Each trial included a 1 s pre-stimulus baseline recording period followed by 1 s stimulus presentation (Supplementary Figure S1(d)). An 80 s inter-stimulus interval (ISI) ensured that pupil size returned to baseline diameter and the after-image had dissipated. The test began at a sub-threshold photophobic stimulus irradiance and then increased based on the participants' response (i.e. no photophobia) by using pre-determined increment step sizes (0.4 log unit) until photophobia was experienced. Photophobia thresholds for three wavelengths (461 nm, 525 nm, 630 nm) were measured in 12 control participants and 11 participants with migraine. An additional three wavelengths (499 nm, 509 nm, 592 nm) were measured in a subsample of control participants (n = 3).
Modeling the retinal photoreceptor contributions to photophobia
To quantify the relative photoreceptor contributions to photophobia, the threshold (corneal) irradiances obtained by the objective method (EMG) for each stimulus was converted to individual log sensitivities (1/thresholdphotophobia; log quanta.cm−2.s−1) and the relative average photophobia sensitivity was plotted as a function of wavelength (nm) and normalized to the peak (525 nm). Given that the perception of brightness in photopic illumination is mediated by the combined contributions of melanopsin expressing ipRGCs and the cone luminance pathways (15), we implemented an analytical model given in Equation 1 to describe photophobia using the combined action spectra of the opn4 melanopsin photopigment (M) (19) and the photopic 10° luminosity function that is the sum of the L- and M-cone inputs to the magnocellular pathway (30,31), where photophobia sensitivity = [m*M - c*Vλ + (z* (M+Vλ)] + k (Equation 1), and m is the melanopsin contribution and c is the cone contribution (the negative sign indicates an adaptation effect). Although a simpler form exists (photophobia sensitivity = (z + m)M + (z – c) Vλ + k), Equation 1 was chosen to emphasise the relative variations in contribution of the two photoreceptor sensitivity functions (32–34), while maintaining necessarily positive output values. By including a grouped single-scale addition component (M + Vλ) multiplied by the coefficient z, the individual-contribution coefficients, m and c, will focus on the region of intersection. The scaling factor, k, accounts for differences in photophobia sensitivity between the control and migraine groups. The cone luminance pathway has two contributions, one that is subtractive (an adaptation response inherent to the magnocellular pathway (35)) and a second that is additive with melanopsin (15). S-cones are not included in the model because of their negligible input to luminosity (36–38). The photophobia data were modeled using Equation 1 by adjusting the free parameters to minimize the sum of squared error differences (SSE) between the data and model using a least-square regression procedure. The quality of fit of the model to the data was assessed using Chi-squared goodness of fit (p > 0.05) and the SSE differences between the data and model. The Supplementary Methods (Modeling) detail the application of models incorporating the rhodopsin action spectrum.
Data analysis
Data are presented using boxplots showing the median, interquartile range and range. Statistical analyses were conducted using GraphPad Prism (GraphPad Software, v7, Inc., CA, USA). All data were normally distributed (Shapiro-Wilk normality test, p > 0.05) and showed equal variances (Levene’s test) for each stimulus wavelength (control: F2,33 = 0.583, p = 0.56; migraine: F2,30 = 0.941, p = 0.40); parametric tests were therefore applied. Independent t-tests were used to detect differences between the control and migraine groups in age, photophobia threshold, the PLR and PIPR metrics. To determine if photophobia thresholds were dependent on stimulus wavelength, a one-way ANOVA (Mean ± SD) was conducted. Bonferroni post-hoc testing was used for multiple pairwise comparisons in the presence of a significant main effect. Significance level was set to 0.05. Bland Altman analysis assessed the agreement of the single, binary and tertiary combination models with the photophobia spectral sensitivity (Supplementary Figure S4) and between the photophobia thresholds estimated using objective (EMG responses) and subjective methods (verbal responses).
Results
Objective EMG photophobia thresholds (log quanta.cm−2.s−1) were wavelength dependent in both the migraine (F1,3 = 21.51, p < 0.001) and control groups (F1,3 = 19.07, p < 0.001) (Figure 1). Thresholds to 630 nm red stimuli were significantly higher than to 461 nm blue stimuli (Bonferroni post-hoc: migraine, mean difference = 0.57, p < 0.001; control, mean difference = 0.50, p < 0.001) and 525 nm green stimuli (migraine, mean difference = 0.67, p < 0.001; control, mean difference = 0.59, p < 0.001), consistent with past reports (14,39,40); there were no significant differences in either group between the EMG photophobia threshold for blue and green lights (migraine, mean difference = 0.10, p = 0.33; control, mean difference = 0.09, p = 0.33). Between-group comparisons revealed that photophobia thresholds in migraineurs were 0.58 log units lower than control participants for blue and green lights (461 nm, t21 = 5.428, p < 0.001; 525 nm, t21 = 5.429, p < 0.001) and 0.50 log units lower for red light (630 nm, t21 = 4.71, p < 0.001) (Figure 1). Bland-Altman analysis determined the between-method bias was 0.04 log units (subjective self-reports were higher than objective EMG photophobia thresholds) with 95% limits of agreement ranging from −0.15 to 0.23.

Photophobia thresholds in controls and migraineurs. Objective EMG photophobia thresholds for control participants (n = 12, unfilled circles) and people with migraine (n = 11, filled circles) at three wavelengths (blue: 461 nm; green: 525 nm; red: 630 nm). Boxplots show the median, interquartile range, and range. One-way ANOVA, significance level ***p < 0.001.
To identify the photoreceptor contributions to photophobia, the relative photophobia sensitivity data in the control and migraine groups were described using the single, binary and tertiary action spectra models (Figure 2 and Supplementary Figures S2 and S3). For the control data, the scaling factor was set to zero (k in Equation 1; Figures 2(a),(e)). The goodness of fit of the combined binary model (melanopsin + Vλ) in the control group had smaller deviations between the data and model compared to other binary combinations (∼57.1 times better than melanopsin and V’λ and ∼23.7 times better than Vλ and V’λ). The tertiary model (melanopsin + Vλ + V’λ) also provided a satisfactory goodness of fit (χ2 = 0.07, p = 0.99); however, the deviations (SSE = 0.03) were higher (∼4.1 times) than the binary model (Supplementary Figure S2(f)). The agreement of the single, binary, and tertiary combination models with the photophobia relative sensitivity showed that the binary melanopsin and Vλ model had the lowest bias and narrowest 95% limits of agreement (Supplementary Figure S3(a)). We therefore conclude that the binary melanopsin and Vλ action spectra (Equation 1) best describes the retinal pathway mediating photophobia in healthy control participants. In this best-fitting binary model (Figure 2(a)), the relative photoreceptor contribution weights (parameters m and c in Equation 1) were 0.47 ± 0.04 (average ± 95% CI) for melanopsin (Figure 2(c), unfilled squares) and −0.32 ± 0.04 (average ± 95% CI) for the cone luminance pathway (Figure 2(c), unfilled circles); the melanopsin to cone luminance weighting ratio (Figure 2(d)) indicates the melanopsin weighting parameter (m in Equation 1) is ∼1.50× higher than cone luminance parameter (c in Equation 1). The weighting of the z-coefficient (Equation 1) was 0.41 ± 0.03 (average ± 95% CI).

Photophobia spectral sensitivity and the relative photoreceptor contributions to photophobia in control and migraine groups. Relative photophobia sensitivity as a function of wavelength in (a) control participants (n = 12, mean ± 95% CI, unfilled symbols) and (b) migraineurs (n = 11). Panels (a) and (b) show the combined binary action spectra model (Equation 1, solid black line ± 95% confidence limits), the melanopsin (opn4, blue dashed line) and 10° photopic spectral luminous efficiency function (Vλ, green dashed line). The left ordinate specifies the average sensitivity of the melanopsin and cone luminance pathways relative to the peak at 525 nm. The right ordinate specifies the average absolute photophobia threshold irradiance (log quanta.cm−2.s−1); note the migraineur data have a different right ordinate scale to account for their lower photophobia threshold. (c) Boxplots showing the median, interquartile range and range of the model parameters (m and c in Equation 1). (d) Boxplot showing melanopsin to cone luminance weightings (m:c ratio). (e) Photophobia sensitivity (normalized to the peak at the 525 nm test wavelength) was higher (p < 0.001) in migraineurs than controls.
The photoreceptor contributions to photophobia threshold in the migraine group (Figure 2(b)) were modeled using the same process as for the control group; photophobia thresholds were significantly lower in the migraine (i.e. 0.55 log unit higher sensitivity) than the control group (parameter k; t21 = 98.43, p < 0.001, Figure 2(b), left y-axis; Figure 2(e)). The combined binary model (melanopsin + Vλ) provided a satisfactory goodness of fit (χ2 = 0.00, p = 0.99) and smaller deviations between the data and model (SSE = 0.0001) compared to other binary combinations (682 times better than melanopsin + V’λ and 1140 times better than the Vλ + V’λ). The tertiary model (melanopsin + Vλ + V’λ) also returned a satisfactory goodness of fit (χ2 = 0.01, p = 0.99, SSE = 0.004; Supplementary Figures S3(f)); however, the SSE deviations were 41 times higher than the binary model. Therefore, the binary melanopsin and Vλ action spectra provides the best description of the retinal pathway mediating photophobia in migraine. The relative photoreceptor contribution weights (parameters m and c in Equation 1) were 0.48 ± 0.04 (average ± 95% CI) for melanopsin (Figure 2(c), filled squares) and −0.32 ± 0.05 for the cone luminance pathway (Figure 2(c), filled circles); the melanopsin to cone ratio (Figure 2(d)) indicates the melanopsin weighting is 1.51× higher than cone luminance (parameter c in Equation 1). Neither of these parameters were significantly different to the control parameters (parameter m: t21 = 0.59, p = 0.56; parameter c: t21 = 0.10, p = 0.92). The weighting of the z-coefficient (Equation 1) was 0.39 ± 0.03 (average ± 95% CI) and not significantly different to the controls (z-coefficient: t21 = 0.98, p = 0.34).
The pupil light response (PLR) metrics and melanopsin-mediated PIPR amplitudes in the control participants were within the expected normal range (19). The mean (± SEM) baseline pupil diameter in the control (6.25 ± 0.34 mm) and migraine groups (6.48 ± 0.24 mm) were not significantly different (t16 = 0.54, p = 0.60). The PLR metrics during presentation of the stimulus light were not significantly different between groups with high melanopsin excitation stimuli (transient PLR, t16 = 1.25, p = 0.23; peak pupil constriction amplitude, t16 = 1.20, p = 0.25) or low melanopsin excitation stimuli (transient PLR, t16 = 0.14, p = 0.89; peak pupil constriction amplitude, t16 = 0.27, p = 0.79). In the migraine group however, the PIPR amplitude to high melanopsin excitation stimuli (51.53 ± 1.43%, mean ± SEM; Figure 3(a), black dashed line) was significantly larger (supranormal) (t16 = 2.48, p = 0.03) and more prolonged than the healthy control group (44.35 ± 2.31%) (Figure 3(b)). As expected, the PIPR amplitude to low melanopsin excitation stimuli (Figure 3(a)) was not significantly different between groups (t16 = 0.53, p = 0.60). The rhodopsin-mediated early PIPR amplitudes were not significantly different between the groups for stimuli with high (t16 = 1.27, p = 0.22) or low melanopsin excitation (t16 = 0.66, p = 0.52) (Figure 3(a)).
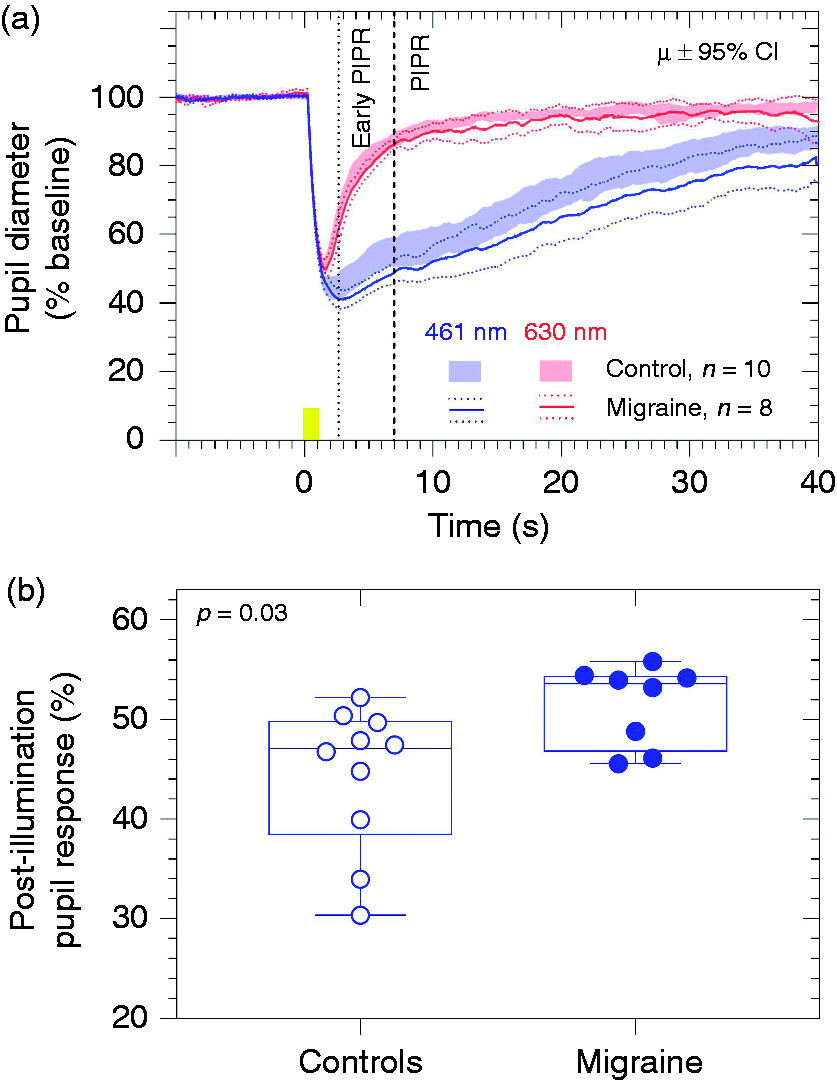
Melanopsin-mediated pupil function. (a) Mean pupil light reflex (PLR) (solid lines) and the melanopsin-mediated post-illumination pupil response (PIPR) (black dashed line) in response to stimuli with high melanopsin excitation (λmax = 461 nm; blue lines) or low melanopsin excitation (λmax = 630 nm; red lines) stimuli of participants with migraine (95% CI indicated by dotted lines) compared to the control group (boundaries of the 95% CI indicated by the solid area), (b) The melanopsin-mediated PIPR with high melanopsin excitation (blue line) at 6 s post-stimulus offset (black dashed line) showed a significantly larger amplitude (difference to baseline) in the migraine group compared to healthy controls.
Discussion
The retinal pathway mediating photophobia is best described by a photoreceptor action spectrum requiring the combined contributions of the melanopsin expressing ipRGCs and the photopic spectral luminous efficiency function (Vλ) of the cone luminance pathway (Figure 2(a),(b)), with a higher melanopsin weighting (∼1.5× cone luminance weighting). The relative melanopsin and cone-opsin weights were similar for both cohorts (Figure 2(c),(d)), with migraineurs having a ∼0.55 log unit lower photophobia threshold (Figures 1 and 2(e)). The melanopsin-mediated PIPR amplitude was supranormal in migraineurs (Figure 3(b)), indicative of a hypersensitivity of the melanopsin pathway. We propose that a retino-geniculate pathway with the intrinsic melanopsin response of the ipRGCs and the cone luminance signal carried by the parasol retinal ganglion cells of the magnocellular pathway together combine in a higher cortical center to mediate photophobia.
Earlier studies inferred that photophobia is driven by cone and rod photoreceptor inputs (5,6,12) to a retino-cortical pathway (14). In addition to ipRGCs, we identify the cone luminance pathway (Vλ) as the second retino-cortical pathway subserving photophobia. Electrophysiological recordings in primate retina show that rod photoreceptor inputs to parasol ganglion cells of the magnocellular pathway are weaker at illuminances > 20 Td (41), and so are unlikely to provide meaningful signals for photophobia. Similarly, the retinal illuminances in this study were all greater than 100 Td and on average, the photophobia threshold was evident at ∼7300 Td (5.5 mm average pupil diameter), an illumination level in the upper range of observable rod contributions to human vision (29). When incorporating the rod action spectrum (V’λ) in the single, binary and tertiary models, it didn’t provide a satisfactory improvement to the descriptions of the photophobia action spectrum (Supplementary Figures S2, S3 and S4). It is known that rods and cones share common retinogeniculate pathways (29,41) and so rod signalling, if involved in photophobia, is likely to be mediated via the luminance and melanopsin pathways, as shown for human brightness estimations in dim scotopic and mesopic illumination (16).
Using chromatic pupillometry with stimulus lights in the illumination range causing photophobia, we detected supranormal and prolonged melanopsin activity in migraineurs with no difference in outer retinal rod- and cone-mediated photoreceptor function (Figure 3(a)). Our observations of normal PLR metrics (transient and peak constriction amplitudes) in the migraine group is to be expected during the interictal period (42), with reduced pupil constriction amplitudes evident within 2 days following a migraine event (43) due to changes in autonomic tone (44). The post-illumination pupil response amplitudes therefore reveal a difference in melanopsin function between the two groups. The supranormal and prolonged melanopsin-mediated PIPR (Figure 3) might reflect inner retinal hyperexcitability. In migraineurs, the relative weightings for the melanopsin and cone luminance pathway inputs to photophobia were similar to the healthy controls (Figure 2); however, the migraineurs were hypersensitive to light stimuli as evidenced by the lower photophobia threshold (Figure 1). It is well established that migraineurs can experience higher photophobia sensitivity (39,42), although the pathways are incompletely defined. Supranormal neuro-retinal responses are evident in associated conditions where photophobia is experienced, such as in albinism (8) and cone dystrophies (9,10). Melanopsin activation also enhances cone-mediated visual thresholds, including luminance processing (24). A signal for cortical hypersensitivity and the enhancement of the discomfort to light might therefore originate in the retina, which is subject to amplification at thalamic and/or cortical loci. Accordingly, neuroimaging of the primary visual cortex shows hypersensitivity to light stimuli (45) and the pattern visual evoked potentials demonstrate increased amplitudes during the interictal period (7). During the interictal period, migraineurs exhibit normal neuro-retinal function (7) and the recovery from transient visual field loss experienced during the attack (46), which indicates there is no retinal pathology, consistent with our findings of a normal PLR in migraineurs (Figure 2(a)).
By testing patients during the interictal period we cannot detect differences that may be attributed to changes occurring during the migraine attack. Between the ictal and interictal periods, the relative sensitivities and time-courses of recovery of the melanopsinergic and cone-opsin pathways could therefore vary. In addition to the afferent melanopsinergic projections to the olivary pretectal nucleus (OPN) to drive the pupil light reflex (47,48), ipRGCs also project to the dorsal lateral geniculate nucleus in non-human primates (17,49) to mediate light-dependent visual functions through the thalamocortical pathway (15,23) that we infer are involved in the photophobia pathway. Differences in activity within the geniculo-striate feedback loop during the ictal and interictal periods may modulate the amplitude and timing of the (supranormal and prolonged) melanopsin signals.
The melanopsinergic and cone-opsin pathways may not be the only conduits of photophobia. Light discomfort and photophobic sensations during migraine can be driven by changes in visual-cortical excitability modulated by brain stem nuclei (50), by neuronal pathways through the hypothalamus to neurons that regulate autonomic function (51) and nociceptive signals relayed via trigemino-vascular pathways (13). Non-visual (affective) brain areas are also involved in photophobia (51) and these brain areas might utilise melanopsinergic pathways through their projections to mood centres in the brain (medial amygdala and lateral habenula) as evidenced in animal models (52,53). Our identification of the involvement of ipRGCs and conventional retinal ganglion cell pathways subserving cone luminance during photophobia can inform the implementation of artificial lighting strategies incorporating luminaires having both a low photopic cone luminance and low melanopsin excitation. Consequently, such artificial lights would advantageously attenuate those retinal signals that may be combined and amplified in post-retinal brain regions when experiencing discomfort to light.
Article highlights
The retinogeniculate pathways mediating photophobia in healthy controls and migraineurs include the melanopsin and cone luminance pathways, with a higher melanopsin weighting. The supranormal and prolonged melanopsin mediated post-illumination pupil response in migraineurs indicates hypersensitivity signalled via intrinsically photosensitive retinal ganglion cells.
Supplemental Material
sj-pdf-1-cep-10.1177_0333102420963850 - Supplemental material for Melanopsin hypersensitivity dominates interictal photophobia in migraine
Supplemental material, sj-pdf-1-cep-10.1177_0333102420963850 for Melanopsin hypersensitivity dominates interictal photophobia in migraine by Andrew J Zele, Ashim Dey, Prakash Adhikari and Beatrix Feigl in Cephalalgia
Footnotes
Acknowledgements
We thank Drew Carter for software design and development.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
Experiments were conducted in accordance with the Declaration of Helsinki and the Queensland University of Technology (QUT) Human Research Ethics Committee approval (#1800000843).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article is supported by the Australian Research Council Discovery Projects ARC-DP170100274 (BF, AJZ) and an Australian Research Council Future Fellowship ARCFT180100458 (AJZ).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
