Abstract
Objective
The objectives of this article are to compare interictal and ictal visual sensitivity between migraine and controls using two published questionnaires, and to correlate responses with a physiologic measure of visual cortex activation.
Methods
Migraine with (MWA, n = 51) and without (MwoA, n = 45) aura and control individuals (n = 45) were enrolled and underwent BOLD fMRI with a visual stimulus. The visual discomfort score (VDS) assessed interictal and the migraine photophobia score (MPS) assessed ictal visual sensitivity.
Result
VDS was significantly higher both in MWA and MwoA vs controls (both p < 0.0001). MPS was greater in MWA vs MwoA (p = 0.008). Ictal and interictal visual sensitivity strongly correlated in MWA (p = 0.004) but not MwoA patients (p = 0.12). BOLD activation in visual cortex was greater in MWA vs controls (2.7% vs 2.3%, p = 0.003) but similar between MwoA and controls. Increasing VDS was associated with greater BOLD signal change in MWA (p = 0.03) but not MwoA (p = 0.65) or controls (p = 0.53). MPS did not correlate with BOLD activation in either group.
Conclusion
Increased interictal visual sensitivity is present both in MWA and MwoA. However, the correlation with ictal visual sensitivity and with cortical hyper-responsivity varies between MWA and MwoA, suggesting underlying differences between groups.
Introduction
Visual sensitivity is a common clinical complaint in migraine, and may take a variety of forms. This may range from typical photophobia occurring during migraine headache, to a variety of more subtle disturbances in the interictal, headache-free period, such as lower discomfort thresholds to bright light, aversion to specific wavelengths and patterns of light stimulus, and susceptibility to visual illusions (1–5). In addition to being considered a key diagnostic criterion for migraine, visual sensitivity is important because it may substantially contribute to the discomfort and disability experienced by those with migraine (6).
A number of psychometric studies have used complex testing paradigms with a variety of visual stimuli to demonstrate increased visual sensitivity in migraine (2,7,8). While useful at elucidating specific aspects of visual dysfunction, the resource-intensive nature of these tools is limited for large-scale research and in clinical practice. In contrast, questionnaire-based scales that use patients’ self-report of visual sensitivity offer an economical and practical approach to gathering this information. We aimed to investigate the usefulness of two previously published scales, one focused on interictal visual sensitivity and one on ictal migraine-associated photophobia, in differentiating between migraineurs with (MWA) and without aura (MwoA) and headache-free controls (9,10). We also aimed to explore the correlation between these scales and a physiologic measure of visual cortex functional activation.
Methods
Study design and participants
We performed a secondary analysis of data collected from the Anatomy and Cerebral Hemodynamic Evaluation of Migraine (ACHE-M) study (11). The ACHE-M study is a prospective case-control study using magnetic resonance imaging (MRI) to compare vascular structure and function between headache-free controls and MWA and MwoA patients. Participants were recruited from the neurology clinic at the University of Pennsylvania and by advertisements in the wider community. Eligible participants were 25–50 years old, had a diagnosis of MWA or MwoA using International Classification of Headache Disorders criteria, or were headache-free controls (6). Participants under age 25 were excluded to minimize misclassification of subjects who might harbor migraine but not yet have become symptomatic; this was based on epidemiologic data suggesting the large majority of individuals who will develop migraine become symptomatic by age 25 (12,13). Participants over age 50 were excluded based on prior data showing acquired changes in vascular structure and function in older people (14). Individuals with any history of cerebrovascular or cardiovascular disease or other neurologic illness were excluded. Family history of migraine was not used to exclude headache-free people. The study aimed to enroll MWA, MwoA, and control individuals in a 1:1:1 ratio. Control participant recruitment was targeted to match the migraine cohort by age and sex. Participants were screened and examined by a single study neurologist to ensure that they met inclusion/exclusion criteria, and were enrolled between March 2008 and June 2012. The study was approved by the University of Pennsylvania Institutional Review Board; all participants provided written informed consent.
Behavioral data on visual sensitivity and photophobia
At study enrollment, participants completed two previously published and validated survey instruments in the following order: 1) the Visual Discomfort Scale, designed to measure interictal visual discomfort irrespective of headache status, and 2) a migraine photophobia scale, designed to assess photophobia related specifically to headache, which was administered only to migraineurs (9,10). The Visual Discomfort Scale consists of 23 questions assessing somatic, perceptual, and performance difficulties experienced in visual tasks. A four-point rating system is used for each question (never, occasionally, often, almost always), with increasing scores indicating increased visual discomfort. The survey instrument was presented in paper format and each question was formatted with an equal distance between the four choices. A total visual discomfort score (VDS) for each subject was calculated by adding the individual point responses for all questions, with each question thus producing a score range of 0–3, and the total score varying from 0 to 69. As the Visual Discomfort Scale includes two questions referencing headaches triggered by light (questions 4 and 5), we also performed an analysis using a VDS subscore (VDS noHA) excluding these two questions. A prior analysis of the Visual Discomfort Scale has suggested that it measures different domains of visual sensitivity—movement/fading (questions 10, 13, 15, 16, 18, 21, 22), blur/diplopia (questions 9, 11, 12, 14, 17, 20), headache/eye soreness (questions 1– 5), glare (question 19), rereading (questions 6–8), and slow reading (question 23) (15). We performed a separate analysis of each of these subscores. The migraine photophobia scale consists of eight yes-no questions relating to specific features of photophobia during a migraine attack; it also includes one question on light as a headache trigger and one question on interictal photophobia. A “yes” answer to any of the first seven questions on this scale was considered evidence of photophobia by the original creators of the scale. A total migraine photophobia score was generated by adding the total number of yes responses to any of questions 1 to 7 in the scale. Administration of both survey instruments was added to the ACHE-M study procedures after study enrollment had begun, thus data were not available for the entire ACHE-M cohort. Both survey instruments were administered in the interictal period and prior to MRI scanning. Immediately after completion of the MRI scan, subjects were asked about headache occurring during the MRI. They were also instructed to notify the research center if they experienced migraine later on the same day as the MRI was performed. Longitudinal follow-up beyond this period to ascertain headache occurrence was not performed.
Imaging data
Full details of the imaging protocol and methods used have been published previously (16). The data presented in the present study are inclusive of the patient data presented previously, but include additional subject data from further patient enrollment to study completion. Briefly, MRI scans were acquired at 3 Tesla using a Siemens Trio and an eight-channel Siemens head coil. Blood-oxygen-level-dependent functional MRI (BOLD fMRI) and T1-weighted magnetization-prepared rapid gradient echo (3D MPRAGE) images were obtained. All scans were performed interictally when subjects were free of complaints of headache. A visual stimulus was generated on an Apple MacBook and presented using a liquid-crystal display (LCD) projector. While the patient maintained central fixation, a 5 Hz flickering square-wave checkerboard (0.5 cycles per degree fundamental spatial frequency) was presented in 15-second, randomly ordered blocks either on the participant’s left, right or bilateral visual field. A blank period of no visual stimulation was also presented randomly for 15 seconds between the different blocks of visual stimulation. Contralateral and bilateral visual stimulation were combined and compared to periods without visual stimulation. Luminance modulation between the white and black checks was 1750 cd/m2. The boundary of the V1 cortex was defined for each participant by anatomical features of the cortical surface, and further constrained to the predicted location of the central 10 degrees of visual field representation. This region of interest (ROI) was then projected back to the volumetric space for each individual. The average BOLD percentage signal change across voxels within each ROI for each participant and for each stimulus condition was then extracted, and this measurement of BOLD activation in the visual cortex was used as the primary measure of physiologic visual response.
Statistical analysis
Groups of participants were compared using the chi-squared or Fisher’s exact test for dichotomous or categorical variables and the t-test or Wilcoxon ranked-sum tests for continuous variables as appropriate. Means and standard deviations or medians and interquartile ranges (IQR) are presented as appropriate. Associations between continuous variables were tested using multivariable linear regression, adjusting for factors that were associated in the univariate analysis at a significance level of p < 0.10. For all reported linear regression analyses, analysis of residuals confirmed the assumption of linearity. Similarly, multivariable logistic regression was used to test associations between categorical variables. An association was considered significant if p < 0.05. All tests were two sided. As recommended by biostatistical experts, corrections for multiple comparisons were not performed (17). Statistical analyses were performed using JMP (Version 9, SAS Institute Inc., Cary, NC).
Results
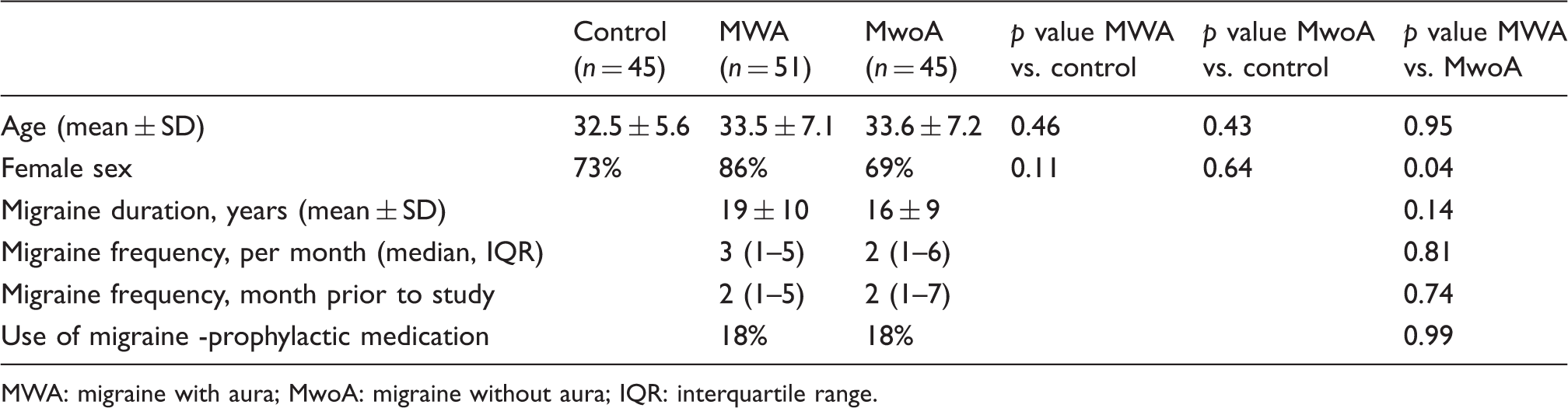
A total of 141 participants were included. Characteristics of enrolled patients are presented in Table 1. Participants were well-matched across groups, with the exception being a greater percentage of women in the MWA compared to MwoA group. All MWA patients had visual aura; 41% of the MWA patients described having aura with headaches ≥50% of the time. VDS were significantly higher both in MWA and MwoA groups compared to controls (Figure 1). There was no significant difference between MWA and MwoA groups. Similar results were seen for the VDS-noHA score excluding the two questions that asked about headache, as well as for the various VDS subscores (Table 2). There was no significant association between total VDS or the VDS subscores and age, sex, age of migraine onset, average monthly migraine frequency, migraine frequency in month prior to study, migraine duration in years, or use of migraine prophylactic medications. Including female sex in a multivariable model comparing MWA and MwoA did not substantively change these results. Analysis of individual VDS items demonstrated 10 questions for which a significant difference was seen between both MWA and MwoA subjects compared to controls (questions 1, 2, 9, 14, 16–18, 20, 21, 23). A short-form VDS created using just these questions resulted in similar overall results to the full VDS comparing MWA vs control (p < 0.0001) and MwoA vs control (p < 0.0001).
Distribution of visual discomfort scores (VDS) across groups. Clinical characteristics of groups. MWA: migraine with aura; MwoA: migraine without aura; IQR: interquartile range. Visual discomfort score (VDS) and subscores across groups. All values are presented as medians (interquartile range). MWA: migraine with aura; MwoA: migraine without aura; HA: headache.

Migraine photophobia scale question responses in migraine groups.

MWA: migraine with aura; MwoA: migraine without aura. ap values adjusted for sex.
Physiologic correlates of behavioral data
In 135 participants for whom both imaging and VDS data were available, there was significantly greater BOLD activation (defined as the amplitude change of signal in primary visual cortex in response to photic stimulation) in the MWA compared to control group (MWA 2.70 ± 0.79% vs control 2.26 ± 0.54%; p = 0.003), but no significant difference between the MwoA and control groups (MwoA 2.24 ± 0.68% vs control 2.26 ± 0.54%; p = 0.85). BOLD activation decreased modestly but significantly with age (0.03% reduction in BOLD activation per year of age, p = 0.004), but was not associated with sex, age of migraine onset, average monthly migraine frequency, migraine frequency in month prior to study, migraine duration in years, or use of migraine prophylactic medications. The difference in BOLD activation by migraine status was not significantly affected by adjustment for age and sex.
VDS was significantly associated with BOLD signal change after adjustment for age in the MWA cohort (0.2% increase in BOLD activation per 10-point increase in VDS, p = 0.03), but there was no association in the MwoA (0.06% decrease in BOLD activation per 10-point increase in VDS, p = 0.65), or controls (0.2% decrease in BOLD activation per 10-point increase in VDS, p = 0.53). The relationship between BOLD activation and VDS across groups is presented graphically in Figure 2. Examining individual VDS subscores, the VDS movement-fading (p = 0.04) and VDS-rereading (p = 0.007) subscores were associated with BOLD activation after adjustment for age in the MWA patients, but no significant association was present in the MwoA or controls. There were no significant associations between the BOLD response and other VDS subscores in any of the groups. There was also no association between migraine photophobia score and BOLD activation in either migraine group in unadjusted analysis or in analysis adjusted for age and sex (MWA: p = 0.70, MwoA: p = 0.67), nor with any of the individual migraine photophobia questions. In participants who endorsed light as a headache trigger (question 7 of the migraine photophobia scale), BOLD activation was not significantly greater (2.65% vs 2.42%, p = 0.23), even after adjustment for age and sex (p = 0.11).
Association between VDS and BOLD signal change in visual cortex in response to a visual stimulus by group.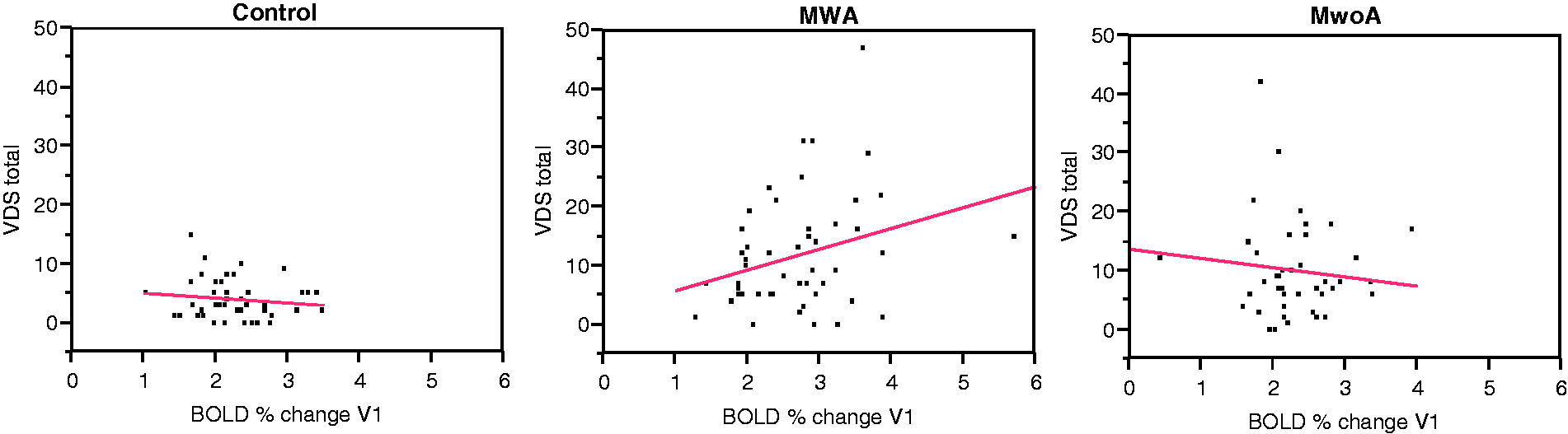
Visual stimuli-induced headache
Of individuals with migraine, six (6%) developed migraine headache during or shortly after the visual stimuli administered during the MRI scan. No controls developed migraine. Those with visual stimuli-induced headache had higher total migraine photophobia scores (median 7 (IQR 7–7) vs 5 (IQR 4–6), p = 0.001), higher total VDS scores (median 21 (IQR 7–31) vs 7 (IQR 3–12), p = 0.01), and were more likely to endorse light as a migraine trigger on question 7 of the migraine photophobia scale (83% vs 20%, p = 0.003). Five participants with MWA had visual stimuli-induced headache compared to one patient with MwoA, but this was not significant (p = 0.21). One MWA individual had typical aura with the visual stimuli-induced headache, one MWA participant had an attenuated version of typical aura, and the other patients did not experience aura. There was no difference in age, sex, or BOLD activation between those with and without visual stimuli-induced headache. Exploratory analysis excluding the six participants with visual stimuli-induced headache did not alter the overall group differences in BOLD activation (BOLD signal change 2.67% MWA, 2.24% MwoA, 2.26% controls).
Discussion
We found that both the VDS and the migraine photophobia scale represent useful tools for evaluating visual sensitivity in migraineurs. The VDS is able to robustly discriminate between individuals with migraine and headache-free controls, and the migraine photophobia scale demonstrated substantial differences between MWA and MwoA patients. While it is widely recognized that visual sensitivity is common in subjects with migraine, identifying and quantifying this through the use of a standardized survey instrument represents a valuable tool for studies assessing visual disturbances and photophobia in migraine.
The creators of the migraine photophobia scale demonstrated that light sensitivity is more accurately identified by their survey instrument compared to the single question about photophobia used in the migraine screening tool ID-Migraine (10,18). This suggests that patients may be misclassified unless presented with a detailed series of questions exploring different facets of visual sensitivity. Indeed, the concept of visual sensitivity likely subserves multiple domains including soreness at the level of the eye itself (photo-oculodynia), worsening of headache in the setting of light, susceptibility to visual illusions, and alterations in contrast sensitivity or motion perception. Considering visual sensitivity as a single construct, therefore, is almost certainly an oversimplification. Given this, it is likely that different mechanisms underlie different aspects of visual sensitivity. One such example is recent data suggesting ictal photophobia in particular may be mediated by dura-sensitive thalamocortical neurons modulated by the non-image-forming melanopsin visual pathway (19). As both the VDS and the migraine photophobia scale include items addressing multiple different areas of visual sensitivity, it might be possible to use them to categorize the specific types of visual discomfort experienced by an individual. To this end we analyzed different VDS subscores based on the type of visual sensitivity referenced in specific questions, but our results were broadly similar across all subscores.
Our study offers several important insights into the differences in interictal and ictal visual sensitivity between MWA and MwoA patients. We have previously demonstrated that compared to MwoA or control participants, MWA individuals have increased activation in the primary visual cortex in response to a visual stimulus (16). This is present even though interictal visual sensitivity, as assessed by the VDS, does not differ between MWA and MwoA patients. These findings are confirmed in the expanded population presented here, and analysis comparing VDS and BOLD fMRI activation suggests a further notable difference between groups in that interictal visual sensitivity appears to correlate with cortical hyper-responsivity in MWA subjects, but not in MwoA or control groups. This raises the possibility that different types of visual sensitivity, or perhaps different mechanisms underlying visual sensitivity, are present in MWA compared to MwoA subjects. Further supporting this possibility, we also found that interictal visual sensitivity (as measured by the VDS) significantly correlates with ictal photophobia (as measured by the migraine photophobia scale) only in MWA patients. While our evaluation suggests that ictal photophobia is more significant in MWA compared to MwoA individuals, more notable is the dramatic difference between the two groups in endorsing light as a headache trigger. This was the case for 40% of the MWA patients, compared to only 5% of the MwoA patients. The pathophysiological mechanisms underlying these various differences remain uncertain. It is possible that hyper-responsive visual cortex modulates the interictal sensitivity noted by MWA individuals, and also is related to the triggering of aura itself. Related to this is the possibility that the experience of visual aura contributes to the heightened perception of light as a headache trigger, and possibly of greater ictal photophobia, in individuals with MWA. Other mechanisms may modulate light sensitivity in MwoA patients.
There has been limited prior study of the VDS in headache. While the original publication describing and validating the Visual Discomfort Scale included some data suggesting that scores were higher in patients with headache, the primary focus was on reading and perceptual difficulty (9). No details about headache type, frequency, or severity were provided. More recently, two small studies in migraineurs have used the VDS. The first examined the relationship between color and spatial frequency and visual pattern sensitivity in 12 controls, 12 individuals with MWA, and 12 MwoA patients and found results quite similar to ours, with both migraine subjects showing higher VDS scores compared to headache-free controls, and both MWA and MwoA patients being similar (7). In this cohort, higher VDS correlated with visual migraine triggers, also similar to the results we found, where individuals endorsing light as a migraine trigger had higher VDS. A second small study by the same group also found a relationship between VDS and visual motion processing alterations in migraine subjects (20).
The original publication describing the migraine photophobia scale included data on 103 migraineurs, 82% of whom had MwoA (10). For most questions, 50%–60% of participants answered “yes,” with the exceptions of question 4 (During your headache, do you have to wear sunglasses even in normal daylight?) for which only 10% agreed, and question 7 (Is your headache triggered by bright lights?) for which only 14% agreed. Data were not presented comparing the MWA to MwoA subgroups. Relatively similar results were found in a subsequent study investigating pattern-induced visual discomfort in 62 migraine patients (95% MwoA) (21). In our population, although the MWA group endorsed greater ictal photophobia than the MwoA group, both groups had an almost uniformly higher prevalence of “yes” answers to each question. However, the relative difference between questions was quite similar, with a substantially lower number of participants answering “yes” to questions 4 and 7 compared to the other questions.
Several limitations of our study should be noted. First, absolute VDS values appear to vary significantly across studies. In the small migraine cohort studies noted above, the mean VDS reported were around 10 for controls and 22 to 25 for migraine patients. In a large cohort of 594 healthy college students (in which headache status was not determined), the overall mean VDS was 15.4 (15). In our cohort, there was a dramatic skew in VDS with a preponderance of lower scores, making median values the more appropriate way of expressing the data. However, simply for comparison purposes, the mean values we obtained were 4.1 in controls, 11.5 in MWA, and 10.1 in MwoA. This indicates that the VDS must be used in conjunction with a control group within studies, and that further research to determine what contributes to variability across populations is important before it could be used as an assessment tool in clinical practice. Second, while our study is one of the larger correlating fMRI and behavioral data in migraine, we were underpowered to fully examine the correlation between cortical activation and specific aspects of the survey data we collected. Third, roughly 20% of our migraine patients were using prophylactic medications. Whether these medications might affect cortical activation or visual sensitivity is unknown. Fourth, while participants were required to be headache free at the time of MRI, we did not specify a particular time interval during which individuals were required to be headache free prior to scan acquisition. Given the relatively low headache frequency and large number of subjects included, it is unlikely this affected our results, however. Finally, the analysis described here should be recognized as a secondary, post-hoc exploratory analysis and will need replication in independent cohorts.
In conclusion, our results suggest that the VDS and migraine photophobia scale may be useful tools in the assessment of patients with migraine. Further work to investigate longitudinal changes in these scales in response to therapeutic interventions, to determine specific population factors that contribute to variability in the scores, and to further correlate participant responses with physiologic alterations in visual processing or activation will be critically important.
Clinical implications
Interictal visual sensitivity can be easily quantified using the Visual Discomfort Scale, and migraine patients have substantially greater Visual Discomfort Scale scores than headache-free controls. Visual cortex hyper-responsiveness, as measured by blood-oxygen-level-dependent functional magnetic resonance imaging (BOLD fMRI), is present in individuals with migraine with aura but not migraine without aura. Interictal visual sensitivity correlates with increased visual cortex activation only in patients with migraine with aura.
Footnotes
Funding
This work was supported by grants from the National Institute of Neurological Disorders and Stroke (NS061572 to B.C; NS058386, NS045839, RR002305 to J.D.).
Conflict of interest
None declared.
