Abstract

Epidemiology, comorbidity, outcomes and classification including big data
MTV20-DP-001
REDUCTIONS IN MIGRAINE FREQUENCY AND DURATION IN PATIENTS WITH CHRONIC MIGRAINE TREATED WITH ERENUMAB: INTERIM RESULTS FROM A REAL-WORLD MULTICENTER CHART-REVIEW STUDY OF US HEADACHE CENTERS
Elizabeth Faust1, Irina Pivneva2, Karen Yang1, Keith A. Betts3, Zubair Ahmed4, Rebecca Hogan5, Andrew Blumenfeld6, Jack Schim6, Alexander Feoktistov7, Kenneth Carnes8, Mark Bensink9, Eric Q. Wu10, Denise E. Chou9 and David Chandler9*
1Analysis Group, New York City, United States
2Analysis Group, Montreal, Canada
3Analysis Group, Los Angeles
4Center for Neurological Restoration, Cleaveland, OH
5DENT Neurologic Institute, Amherst, NY
6The Headache Center of Southern California, Carlsbad, CA
7Diamond Headache Clinic, Chicago, IL
8Raleigh Neurology Associates, Raleigh, NC
9Amgen, Thousand Oaks
10Analysis Group, Boston, MA, United States
Introduction
Erenumab (erenumab-aooe in US; FDA approval May 2018), the first-in-class monoclonal antibody targeting the CGRP pathway approved for the prevention of migraine in adults, significantly decreased the number of monthly migraine days versus placebo in clinical trials (Tepper 2017, Goadsby 2017). Since this is a recent approval, limited data is available on its impact in real-world settings.
Objectives
To characterize the real-world treatment profiles and clinical outcomes of patients prescribed erenumab from select US headache centers.
Methods
This study was a center-based, retrospective, chart review of patients with migraine at 5 major US headache centers. Adult patients with chronic or episodic migraine were included if they were treated with erenumab for ≥3 consecutive months. Baseline was defined as the 3 months before erenumab initiation; study period was defined as ≥3 months of erenumab treatment.
An electronic case report form was used to collect patient and clinical characteristics as well as acute and preventive migraine medication use. These interim results are based on charts collected from April to November 2019.
Results
Here we report results for the 469 patients in the study with chronic migraine. The mean age was 49 years; 86% were female and 74% were white. The most common comorbidities were depression (27%) and anxiety (25%). Prior to erenumab initiation, patients averaged 9.2 years of disease duration and the majority were treatment refractory, with a mean of 5 preventive treatment failures. At baseline, 46% had physician-assessed severe migraine. The most commonly prescribed preventive treatments during baseline were antiepileptics (48%), onabotulinumtoxin (47%), and antidepressants (42%); the most commonly prescribed acute treatments were triptans (69%) and NSAIDs (34%). The mean baseline number of headache/migraine days per month was 16.6, with an average duration of 26.8 hours per attack.
A majority (365; 78%) of patients remained on erenumab at the end of the study period (mean follow-up 8.7 months). 39% of patients discontinued ≥1 baseline preventive treatment and 31% discontinued ≥1 baseline acute treatment, although the aggregate treatment profile remained largely similar between baseline and study period. After treatment with erenumab, mean headache/migraine days per month decreased by 5.5 days, with 39% of patients having ≥50% reduction in the mean number of headache/migraine days per month. Headache/migraine duration per attack was reduced on average by 6.0 hours.
Conclusion
In patients with chronic migraine, many of whom were refractory to preventive therapies, erenumab reduced the mean number of headache/migraine days per month and the average duration of migraine/headache attacks. After erenumab initiation, patients treated in 5 US headache centers largely continued to be managed via a polypharmacy approach. Further research is needed to better understand patient outcomes and treatment patterns in this population.
Disclosure of Interest
E. Faust Conflict with: mployed by Analysis Group, which provides consulting services to Amgen., I. Pivneva Conflict with: employee of Analysis Group, Inc., which provides consulting support to Amgen for this study, K. Yang Conflict with: employee of Analysis Group, Inc., which provides consulting support to Amgen for this study, K. Betts Conflict with: employee of Analysis Group, Inc., which provides consulting support to Amgen for this study, Z. Ahmed Conflict with: Zubair A. Ahmed has received consulting fees from Eli Lilly, Amgen, Allergan. He has served as a speaker for Allergan and has received research funding from Teva, R. Hogan: None Declared, A. Blumenfeld: None Declared, J. Schim Conflict with: Consultant – Aeon, Allergan, Amgen, electroCore, Impel, Lilly, Lundbeck, Novartis, Teva, Upsher-Smith Research – Aeon, Allergan, Amgen, electroCore, Lilly, Lundbeck, Satsuma, Teva Speaker – Allergan, Amgen, electroCore, Lilly, Lundbeck, Novartis, Teva, Upsher-Smith, A. Feoktistov Conflict with: Speaker bureau: Amgen, Allergan, Teva, Lilly, Biohaven. Consultant/Advisory: Amgen, Teva, Lilly, Biohaven, K. Carnes: None Declared, M. Bensink Conflict with: Employment & Stock Ownership with Amgen Inc, E. Wu Conflict with: employee of Analysis Group, Inc., which provides consulting support to Amgen for this study, D. Chou Conflict with: Employment & Stock Ownership with Amgen Inc, D. Chandler Conflict with: Employment & Stock Ownership with Amgen Inc.
References
MTV20-DP-002
CGRP MONOCLONAL ANTIBODY USE AND PATIENT-REPORTED IMPROVEMENT OF MIGRAINE: RESULTS OF THE OVERCOME STUDY
Robert E. Shapiro1*, Karen H. Samaan2, Robert A. Nicholson2, Anthony J. Zagar2, Yongin Kim2, Dawn C. Buse3, Sait Ashina4, Michael L. Reed5, Bert B. Vargas2, Susan Hutchinson6 and Richard B. Lipton7
1Department of Neurological Sciences, University of Vermont, Burlington
2Eli Lilly and Company, Indianapolis
3Department of Neurology, Albert Einstein College of Medicine, Bronx
4Department of Neurology and Department of Anesthesia, and Harvard Medical School, Beth Israel Deaconess Medical Center, Boston
5Vedanta Research, Chapel Hill
6Orange County Migraine and Headache Center, Irvine
7Albert Einstein College of Medicine, Bronx, United States
Introduction
Real-world patient-reported outcome data are limited for people who use a calcitonin gene-related peptide (CGRP) targeted monoclonal antibody (mAb) for the preventive treatment of migraine.
Objectives
To assess patient characteristics and patient-reported improvement among current CGRP mAb users.
Methods
Data were obtained in Q4, 2019 from the ObserVational survey of the Epidemiology, tReatment and Care Of MigrainE (OVERCOME), a web-based survey conducted in a representative US sample. Respondents were identified via a validated migraine diagnostic screener for ICHD-3 criteria (95.1%) and/or self-reported healthcare professional migraine diagnosis (59.0%). This analysis included those who had used a CGRP mAb (erenumab, fremanezumab, galcanezumab) for the preventive treatment of migraine within the past 3 months.
Current CGRP mAb users completed the Patient Global Impression of Improvement (PGI-I; range 1 = very much better to 7 = very much worse) to assess perceived improvement in their “migraine condition” since starting the current CGRP mAb. Individuals reported duration of current CGRP mAb use and any additional recommended migraine preventive medication use in the past 3 months, and completed the Migraine Disability Assessment (MIDAS) scale. Unadjusted percentages of patients (95% CI) with PGI-I responses (better, no change, worse) are reported overall and by monthly headache day categories (0–3, 4–7, 8–14, ≥15). Other data are described using central tendencies.
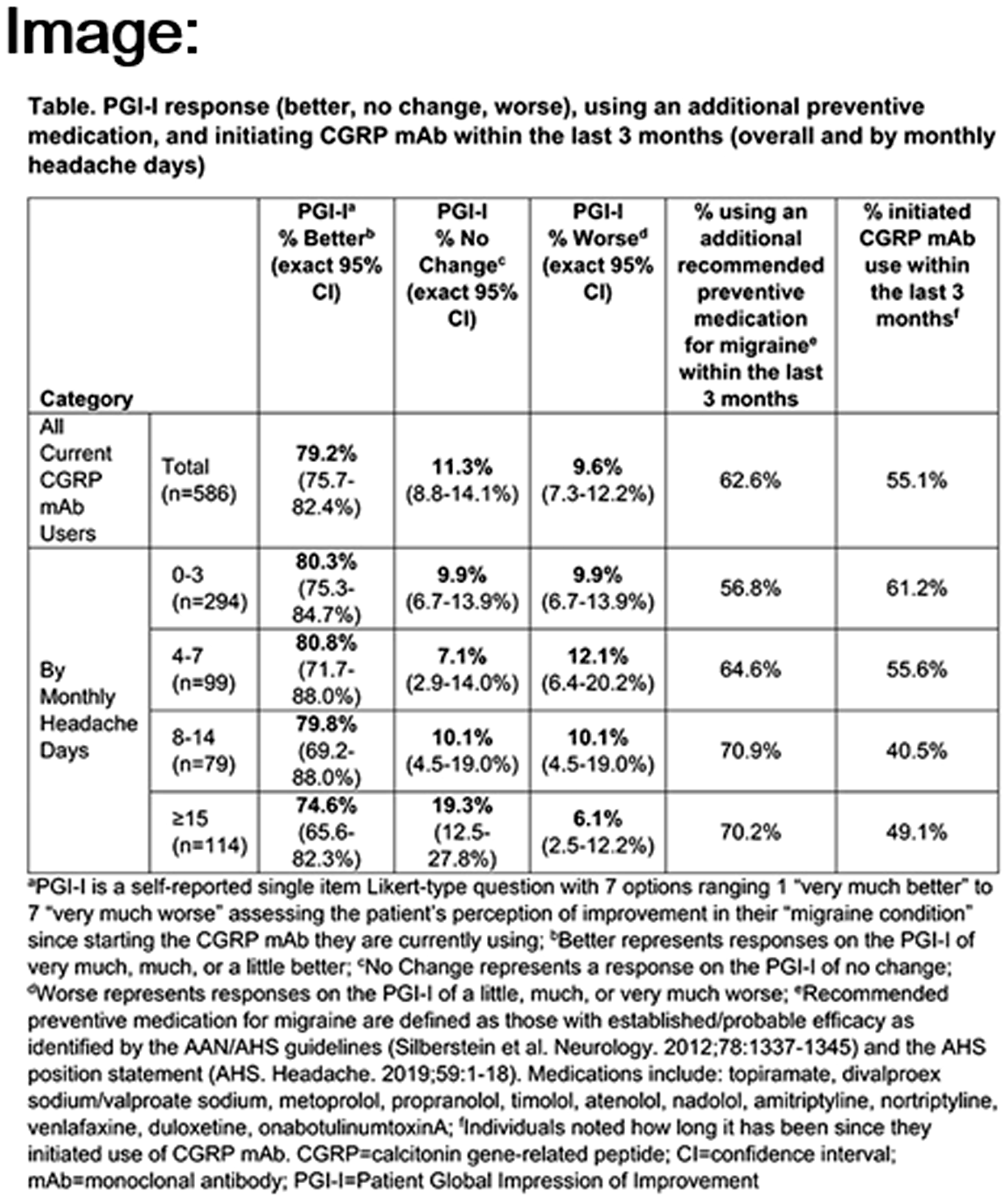
Results
Of 20,782 surveyed, 950 (4.6%) reported ever using a CGRP mAb. Among the 586 (2.8%) currently using a CGRP mAb, mean (SD) age was 38.9 (12.9) years, 64.0% were female, and 73.5% were white. Mean monthly headache days was 7.6 (8.4). Nearly half (49.5%) had severe migraine-related disability (MIDAS score ≥21), 22.9% moderate (11–20), 15.0% mild (6–10), and 12.6% little to none (≤5). Over half had initiated use of the current CGRP mAb within the past 3 months (55.1%) and 62.6% had used an additional migraine preventive medication (concomitant or switching) in the past 3 months.
Overall, most (79.2%; 95% CI 75.7–82.4%) reported their migraine condition as “better” based on the PGI-I since starting the CGRP mAb. This was consistent across monthly headache day categories (Table).
Conclusion
Most patients taking a CGRP mAb for the preventive treatment of migraine reported their migraine as better since starting this medication. These are among the first real-world population-based patient-reported outcomes from those using CGRP mAbs and suggest that these novel medications may increase patient-perceived improvement. Future analyses should consider how migraine-related (eg, baseline headache frequency, symptom severity, cyclic disease fluctuations), medication-related (eg, duration of therapy, other preventive medication use, acute treatment optimization, switching between CGRP mAbs) and healthcare-related (eg, care location, medication access) factors may influence patient-reported outcomes when using CGRP mAbs.
Disclosure of Interest
R. Shapiro Conflict with: Eli Lilly and Company, K. Samaan Conflict with: Eli Lilly and Company, R. Nicholson Conflict with: Eli Lilly and Company, A. Zagar Conflict with: Eli Lilly and Company, Y. Kim Conflict with: Eli Lilly and Company, D. Buse Conflict with: Amgen, Conflict with: Allergan, Biohaven, Eli Lilly and Company, Dr. Reddy’s, Teva, Amgen, S. Ashina Conflict with: Allergan, Amgen, Novartis, Eli Lilly and Company, Biohaven, Satsuma, Percept, Theranica, Supernus, M. Reed Conflict with: Promius/Dr Reddy’s, Allergan, Conflict with: Eli Lilly and Company, B. Vargas Conflict with: Eli Lilly and Company, S. Hutchinson Conflict with: Alder/Lundbeck, Allergan, Amgen, Biohaven, Currax, ElectroCore, Eli Lilly and Company, Novartis, Theranica, Teva, Upsher-Smith, R. Lipton Conflict with: Allergan, American Headache Society, Amgen, Avanir, Biohaven, Biovision, Boston Scientific, Dr. Reddy’s (Promius), Electrocore, Eli Lilly, eNeura Therapeutics, Equinox, GlaxoSmithKline, Lundbeck (Alder), Merck, Pernix, Pfizer, Supernus, Teva, Trigemina, Vector, Vedanta
MTV20-DP-003
PATIENTS’ REASONS FOR STARTING, SWITCHING, AND STOPPING CGRP TARGETED MONOCLONAL ANTIBODIES: RESULTS OF THE OVERCOME STUDY
Dawn C. Buse1*, Kory Schuh2, Robert A. Nicholson2, Michael L. Reed3, Yongin Kim2, Anthony J. Zagar2, Bert B. Vargas2, Robert E. Shapiro4, Sait Ashina5, Susan Hutchinson6 and Richard B. Lipton1
1Albert Einstein College of Medicine, Bronx
2Eli Lilly and Company, Indianapolis
3Vedanta Research, Chapel Hill
4Department of Neurological Sciences, University of Vermont, Burlington
5Department of Neurology and Department of Anesthesia, and Harvard Medical School, Beth Israel Deaconess Medical Center, Boston
6Orange County Migraine and Headache Center, Irvine, United States
Introduction
There is little real world, population-based evidence from patients for their reasons for starting, switching, and stopping use of calcitonin gene-related peptide (CGRP)-targeted monoclonal antibody (mAb) antagonists prescribed for the preventive treatment of migraine.
Objectives
To assess patient-reported reasons for starting, switching, and stopping CGRP mAbs.
Methods
Data were obtained in Q4 2019 from a web-based survey of a representative US sample (ObserVational survey of the Epidemiology, tReatment and Care Of MigrainE [OVERCOME]). The present sample included 20,782 people with migraine identified using a validated migraine diagnostic screener based on ICHD-3 criteria (95.1%) and/or self-reported healthcare professional migraine diagnosis (59.0%). Branching logic within the survey identified respondents who started (ever taken a CGRP mAb [erenumab, fremanezumab, galcanezumab]), switched (taken more than one CGRP mAb and taken a CGRP mAb within the last 3 months), and/or stopped (previously taken a CGRP mAb, but have not taken within the last 3 months) using a CGRP mAb. Respondents were asked to select all applicable reasons for these behaviors. The reasons were then categorized into domains labeled as: efficacy, dosing/delivery, recommendation/request, tolerability, access/economic, new treatment, disease resolution, or stigma.
Image:

Results
Of 20,782 respondents, 950 (4.6%) had used one or more mAb(s). mAb users had a mean of 6.8 monthly headache days, a mean age of 36.9 years, and 61.4% were female. The majority had first started a mAb(s) in the past 12 months (89.3%); 25.9% had moderate and 46.8% had severe migraine-related disability based on the Migraine Disability Assessment Scale (MIDAS). Of 950 people sampled, 11.8% had switched and 38.3% had stopped (Table). The three most common reasons for: starting: pursuit of greater efficacy (64.2%), dosing/delivery advantages (50.8%), and recommendations from others (45.7%); switching: efficacy (50.9%), access/economic issues (48.2%), and recommendations from others (42.0%); and stopping: tolerability (40.1%), access/economic issues (39.3%), and recommendations from others (33.2%).
Conclusion
The findings from this analysis suggest that the reasons why patients start, switch, or stop CGRP mAbs can vary. Prescribers should consider this and not assume the reasons are homogeneous when discussing initiating or changing medications with their patients pertaining to migraine prevention.
Disclosure of Interest
D. Buse Conflict with: Amgen, Conflict with: Allergan, Biohaven, Eli Lilly and Company, Dr. Reddy’s, Teva, Amgen, K. Schuh Conflict with: Eli Lilly and Company, R. Nicholson Conflict with: Eli Lilly and Company, M. Reed Conflict with: Promius/Dr Reddy’s, Allergan, Conflict with: Eli Lilly and Company, Y. Kim Conflict with: Eli Lilly and Company, A. Zagar Conflict with: Eli Lilly and Company, B. Vargas Conflict with: Eli Lilly and Company, R. Shapiro Conflict with: Eli Lilly and Company, S. Ashina Conflict with: Allergan, Amgen, Novartis, Eli Lilly and Company, Biohaven, Satsuma, Percept, Theranica, Supernus, S. Hutchinson Conflict with: Alder/Lundbeck, Allergan, Amgen, Biohaven, Currax, ElectroCore, Eli Lilly and Company, Novartis, Theranica, Teva, Upsher-Smith, R. Lipton Conflict with: Allergan, American Headache Society, Amgen, Avanir, Biohaven, Biovision, Boston Scientific, Dr. Reddy’s (Promius), Electrocore, Eli Lilly, eNeura Therapeutics, Equinox, GlaxoSmithKline, Lundbeck (Alder), Merck, Pernix, Pfizer, Supernus, Teva, Trigemina, Vector, Vedanta
MTV20-DP-004
EPIDEMIOLOGICAL STUDY ON CLINICAL AND FUNCTIONAL DISABILITY OF MIGRAINE PATIENTS VISITING NEUROLOGICAL CENTERS IN NEPAL
Ayush Chandra1*
1Clinical Medicine, Tianjin Medical University, Tianjin, China
Introduction
Migraine is a neurological disabling condition which is prevalent globally and it is a major reason sought for the medical care. A large number of population evidenced by 1 billion people around the world are affected due to migraine disorder. It is the sixth most disabling illness in the world and fifth leading cause of year of life lived with disability in south Asia. Very few studies are reported in Nepal regarding this disorder. This study is conducted in order to explore the clinical and functional disability of patient with migraine among Nepali residents.
Objectives
The general objective was to determine the clinical features and influence of migraine headaches on the everyday functioning of patient. Special objectives are to assess the socio-demographic characteristics of patient with migraine, to investigate the clinical manifestation of migraine and to determine the functional disability caused due to migraine using Migraine Disability Assessment (MIDAS) format.
Methods
Quantitative research methods were used to carry out this study. MIDAS questionnaire was used. Data was collected under Likert scale format. Data were analyzed by using descriptive statistical method by SPSS (version 20).
Image:

Results
About 219 patients were diagnosed with migraine in the period of 3 months. Majority of participants were in the age group of 20–29 years (29.7%) followed by age group of 30–39(27.9%) with the majority of female participants (79%).
The various symptoms experienced by participants were throbbing pain (87.2%), photophobia (9.2%), Phonophobia (86.3%), Nausea (54.3%) and vomiting (38.8%). About 36.52% of patients were in the category of episodic migraine and 63.47% were in the category of chronic migraine. Most of the subjects (84.5%) experienced the severe level of functional disability.
Conclusion
It is concluded that chronic migraine was found to be 3.4 times higher in disability called as level IV in comparison with level I also called as lower disability level and this relationship was statistically significant (OR = 3.4, 95%CI = 1.01-11.48) as shown in Table. The study was conducted on the group of patient who came in Out-patient department (OPD) based in the hospital within the certain period of time.
Disclosure of Interest
None Declared
References:
None
MTV20-DP-005
IMPACT OF POOR SELF-RATED MIGRAINE ON PUBLIC HEALTH SERVICES UTILISATION IN SLOVENIA
Marjan Zaletel*, Bojana Žvan1 and Lijana Zaletel-Kragelj1
1Department of Vascular Neurology, University Clinical Centre, Ljubljana, Slovenia
Introduction
Human health is a complex concept with multiple dimensions (1). Undoubtedly, physical dimension is extremely important, however, mental and social dimensions are important as well. Consequently, self-rated health (SRH) became important indicator in obtaining a rough assessment of health at both, individual and population levels.
Objectives
Recent Global Burden of Disease 2016 study confirmed that among different types of headache particularly migraine is a major public health problem, especially in young and middle-aged women. Among others, direct costs are attributed to medical care for emergency department (ED) visits, hospitalizations (H), and specialist visits (SV). In general, patients with poor self-rated health (pSRH), represents a sub-group of patients with greater utilization of health services. So far studies, few in number, have shown the relationship between migraine and pSRH, but they were not systematically controlled for comorbidity and socioeconomic factors. In addition, the relationship between pSRH migraine and public health institution utilization has not been systematically studied yet. The aim of our study was to assess the relationships between migraine and ED visits, H, and SV in subgroup of migraineurs with pSRH in Slovenia.
Methods
The survey, conducted from August to December 2014, included 6262 adults, aged 15 years or older. Binary logistic regression was used in univariate as well as in multivariate analysis to established relationships between health services (ED, H, SV) migraine, comorbidities related to physical and mental dimension of health and additionally to demographic and socioeconomic factors. pSRH was introduced to substitute physical and mental comorbidities.
Results
In univariante models of study population we found associations between migraine and ED (OR = 1.74; p < 0.001), SV (OR = 1.40; p < 0.001) but not H (OR = 1.11; p = 0.330). In multivariante models we did not observe significant relationships between migraine and H (p = 0.843), ED (p = 0.210) and SV (p = 0.152). Univariante models of subpopulation showed associations of pSRH migraineurs with H (OR = 4.03; < 0.001), ED (OR = 20.00; p < 0.001) and SV (OR = 2.38; p < 0.001). Multivariate models showed relationships between pSRH migraineurs and H (3.48; p < 0.001), ED (14.15; p < 0.001) and SV (OR = 2.27; p < 0.001).
Conclusion
Migraine is not independently associated with public health services utilization. The subgroup of migraine with pSRH is independently associated with public health services visits. Thus the health policy has to be focused on proper management of pSRH migraine.
Disclosure of Interest
None Declared
MTV20-DP-006
WHEN PILLS DO NOT WORK : GASTROPARESIS IN MIGRAINE
Sheena Aurora1*, Linda Nguyen2, Sutapa Ray1 and Stephen Shrewsbury1
1Impel NeuroPharma, Seattle
2Stanford University, Palo Alto, United States
Introduction
An understanding of gastric dysmotility is important to patients and physicians since it has implications in treatment of migraine.
Objectives
Gastric stasis has long been implicated in association with migraine. In this abstract we review the current state of scientific evidence that exists for migraine and gastric stasis.
Methods
Key words, gastric stasis, migraine, autonomic dysfunction were used to obtain relevant studies in a literature search.
Results
Early experimental studies by Volans et al. reported a delay in effervescent aspirin absorbtion in 19 out of 42 migrainuers during an attack, but not during the headache free period. These studies suggested delayed gastric emptying occurs during spontaneous migrane attacks. This hypothesis was questioned when studies demonstrated delay in gastric emptying during visually induced migraines and during the headache free interictal period. Using gastric scintigraphy the rate of gastric emptying was measured in 10 migraineurs and 10 age and sex matched controls. The time of half emptying after an induced migraine attack was delayed 78% ictally and 80% interictally in migrainuers and compared to non-migraine controls the time to half emptying was significantly longer at 188.8 minutes compared to 111.8 min. Subsequently, Aurora and colleagues confirmed findings of delayed half emptying during spontaneous migraine attacks as well. Another study found contradictory results in migraineurs without interictal dyspepsia symptoms. Compared to migraineurs, subjects with functional dyspepsia had more delayed gastric emptying (Yu 2012). However, a study evaluating liquid phase gastric emptying observed delayed ictal but not interictal gastric emptying in migrainuers compared to controls (Yalcin 2012). In a survey of 516 patients diagnosed with gastroparesis in the NIH Gastroparesis consortium, migraine was the most common extra-intestinal comorbidity (36.6%). This presence of migraines was associated with more severe gastroparesis symptoms.
Conclusion
The association between gastroparesis and migraine may be under recognized. Conflicting findings in studies may be due to test methodology or the variability in migraine phenotype. Attention to this comorbidity may be particularly important if patients have symptoms of nausea, vomiting and/or abdominal pain who do not experience relief of migraine symptoms using oral abortive treatment. Route of administration and formulation may have an impact on absorption and hence efficacy of migraine therapies.
Disclosure of Interest:
S. Aurora Conflict with: Employee and stock holder of Impel NeuroPharma, L. Nguyen: None Declared, S. Ray Conflict with: Employee and stock holder of Impel NeuroPharma, S. Shrewsbury Conflict with: Employee and stock holder of Impel NeuroPharma
MTV20-DP-007
IDIOPATHIC INTRACRANIAL HYPERTENSION – A STUDY OF THE INCIDENCE AND DEMOGRAPHICS IN NORTHERN IRELAND
Martin Harley1* and Thomas Peukert
1Neurology, Royal Victoria Hospital Belfast, Belfast, United Kingdom
Introduction
Idiopathic Intercranial Hypertension (IIH) is a common neurological condition which often initially presents to acute services. Incidence is normally reported at 1–2 per 100,000 however in practice the number often feels larger than this. Building on previous work we have recorded three years incidence data for the condition in Northern Ireland.
Objectives
The Northern Irish population is well suited to incidence studies due to low levels of migration, a single emrergency care provider (NHS) and the presence of comprehansive region wide electronic care systems. We aimed to identify all newly diagnoised patients with IIH within a three year time frame. We also used thses lectronic care records to look at key demographics including patient BMI, imaging findings and initial ophthalmological examination.
Methods
We made use of networked imaging systems as the main information source cross referenced with clinical coding data form admissions units and eye casualty. We sampled the time period March 2017 to February 2020 inclusive and used National Office of Statistics population estimates to calculate incidence.
Results
We have demonstrated an incidence of IIH within the Northern Irish population which is much higher than is commonly reported in the literature but which is in line with similar studies published by our colleagues in the North West of Ireland1. Demographic data shows the condition to affect a predominently female population who are overweight and between the ages of 15 to 45. This is in keeping with the published body of literature.
Conclusion
Is IIH a Northern Irish problem or one systematically under reported in the literature? We postulate that the previously published literature on incidence is out of date and does not reflect current incidence. The increasing obesity epidemic is likly a major contributing factor to increasing incidence.
Disclosure of Interest
None Declared
Reference
MTV20-DP-008
TRACKING THE EVOLUTION OF NON-HEADACHE SYMPTOMS THROUGH THE MIGRAINE ATTACK
Roberta Messina*, Ilaria Cetta1, Bruno Colombo1 and Massimo Filippi1
1San Raffaele Hospital, Milan, Italy
Introduction
Previous studies have highlighted non-headache symptoms associated with migraine occurring during the prodrome or postdrome phase. However, non-headache symptoms can start before the onset of the pain, persist during the headache phase, and can go on after migraine headache resolution.
Objectives
The aim of this study was to track the evolution of non-headache symptoms through the different phases of the migraine attack.
Methods
One hundred and eight migraine patients (81 females, mean age: 47 years) were enrolled. Patients were asked to recall retrospectively whether non-headache symptoms occurred during the prodrome, headache and postdrome phase of their migraine attacks. The Cochran’s Q test was performed to assess any significant differences in the frequency of non-headache symptoms across the three phases of the migraine attack. The co-occurrence of symptoms during the different phases was tested using Cohen’s and Fleiss’ kappa (k) with 95% confidence intervals (CI).
Results
The frequency of most non-headache symptoms was significantly different throughout the three phases of the migraine attack, being higher during the headache phase than during the prodrome and postdrome phase. The rate of thirst and food craving did not significantly change over the migraine attack. The symptoms with the highest co-occurrence through all three phases were food craving (k = 0.35, 95% CI 0.34–0.35), neck stiffness (k = 0.34, 95% CI 0.33–0.34) and thirst (k = 0.34, 95% CI 0.33–0.34). If thirst occurs during the headache phase it is highly likely that it will persist during the postdrome phase (k = 0.59, 95% CI 0.59–0.60), while if neck stiffness occurs during the prodrome phase it will probably occur also during the headache phase (k = 0.46, 95% CI 0.46–0.47). We also found that if yawning is present during the prodrome phase it is highly likely that it will occur also during the postdrome phase (k = 0.47, 95% CI 0.46–0-47).
Conclusion
Although cognitive, mood and homeostatic changes are characteristics of the prodrome and postdrome phase, migraine patients most often recall having these symptoms in association to the headache pain. There is evidence showing that thirst, food craving and neck stiffness can be mediated by the hypothalamus. Thirst, food craving and neck stiffness occurred throughout all three phases, suggesting a constant involvement of the hypothalamus during the migraine attack.
Disclosure of Interest
None Declared
MTV20-DP-009
EXPLORING THE BOUNDARIES BETWEEN EPISODIC AND CHRONIC MIGRAINE: RESULTS FROM THE CAMEO STUDY
Richard B. Lipton1*, Michael L. Reed2, Kristina M. Fanning2, Dawn C. Buse1, Peter J. Goadsby3, David W. Dodick4 and Aubrey Manack Adams5
1Albert Einstein College of Medicine, Bronx, NY
2Vedanta Research, Chapel Hill, NC, United States
3King’s College, London, United Kingdom
4Mayo Clinic, Phoenix, AZ
5Allergan, an AbbVie Company, Irvine, CA, United States
Introduction
Differences in migraine burden between people with high-frequency episodic migraine (HFEM) and low-frequency chronic migraine (LFCM) have not been well characterized. We evaluated migraine burden across monthly headache day (MHD) subgroups in the web-based Chronic Migraine Epidemiology and Outcomes (CaMEO) Study population.
Objectives
To characterize the burden of migraine across monthly headache day (MHD) subgroups in the web-based Chronic Migraine Epidemiology and Outcomes (CaMEO) Study population by assessing headache-related disability and burden.
Methods
Eligible respondents who met modified International Classification of Headache Disorders, 3 rd edition criteria for migraine answered web-based survey questions on demographics, headache features, disability, interictal burden, and depression. Four subgroups were defined based on self-reported MHDs: low-frequency episodic migraine (LFEM; 0–7), HFEM (8–14), LFCM (15–23), and high-frequency chronic migraine (HFCM; ≥24). Trends by MHDs were assessed by the linear-by-linear association test and 2-sided chi statistics.
Results
Data were provided by 16,789 respondents (13,473 LFEM, 1840 HFEM, 1035 LFCM, and 441 HFCM). Of the respondents in the 4 MHD subgroups, 12.8%, 51.9%, 66.3%, and 70.1%, respectively, had severe disability (grade 4) based on the Migraine Disability Assessment Scale (MIDAS), and at least moderate disability (MIDAS grade 3 or 4) was reported by 29.6%, 74.1%, 81.1%, and 78.9% of respondents. Severe scores (≥5) on the Migraine Interictal Burden Scale (MIBS) were reported by 19.2%, 38.2%, 46.7%, and 58.3% of respondents in the 4 MHD groups, respectively, and moderate or severe scores (MIBS ≥3) were reported by 32.3%, 56.1%, 63.7%, and 71.9%, respectively. Moderate to severe depression (score of ≥10 on the 9-item Patient Health Questionnaire) occurred in 27.6%, 47.3%, 54.9%, and 60.8% of respondents in the 4 MHD subgroups, respectively. Moderate to severe anxiety (score of ≥10 on the 7-item Generalized Anxiety Disorder [GAD] scale) was reported in 26.2%, 41.9%, 46.9%, and 52.2% of respondents in the 4 MHD subgroups. All changes were statistically significant for the linear-by-linear association test (P < 0.001).
Conclusion
These data indicate substantial overlap in these measures of burden and depression among CaMEO respondents with HFEM (8–14 MHDs) and those with LFCM (15–23 MHDs).
Disclosure of Interest :
R. Lipton Conflict with: serves on the editorial boards of Neurology and Cephalalgia and as senior advisor to Headache. He has received research support from the NIH. He also receives support from the Migraine Research Foundation and the National Headache Foundation. He has reviewed for the NIA and NINDS, and has served as consultant or advisory board member for or has received honoraria from Alder, AbbVie, Amgen, Autonomic Technologies, Avanir, Biohaven, Biovision, Boston Scientific, Dr. Reddy’s, Electrocore, Eli Lilly, eNeura Therapeutics, GlaxoSmithKline, Merck, Novartis, Pernix, Pfizer, Supernus, Teva, Trigemina, Vector, and Vedanta. He receives royalties from Wolff’s Headache (8th Edition, Oxford University Press), Informa, and Wiley. He holds stock options in eNeura Therapeutics and Biohaven., M. Reed Conflict with: Managing Director of Vedanta Research, which has received research funding from AbbVie, Amgen, Dr. Reddy’s Laboratories, Eli Lilly, GlaxoSmithKline, Merck & Co., Inc., Novartis and the National Headache Foundation. Vedanta Research has received funding directly from AbbVie for work on the CaMEO Study., K. Fanning Conflict with: employee of Vedanta Research, which has received research funding from AbbVie, Amgen, Dr. Reddy’s Laboratories, Eli Lilly, GlaxoSmithKline, Merck & Co., Inc., Novartis, and the National Headache Foundation. Vedanta has received funding directly from AbbVie for work on the CaMEO Study., D. Buse Conflict with: received grant support and honoraria from AbbVie, Avanir, Amgen, Biohaven, Eli Lilly and Company, Promius, Teva and for work on the editorial board of Current Pain and Headache Reports., P. J. Goadsby Conflict with: reports personal fees from AbbVie and related grants and personal fees from Amgen and Eli Lilly and Company; personal fees from Alder Biopharmaceuticals, Autonomic Technologies Inc., Dr. Reddy’s Laboratories, electroCore LLC, eNeura, Novartis, Scion, Teva Pharmaceuticals, and Trigemina Inc.; personal fees from MedicoLegal work, Massachusetts Medical Society, Up-to-Date, Oxford University Press, and Wolters Kluwer; and a patent Magnetic stimulation for headache assigned to eNeura without fee., D. Dodick Conflict with: reports personal fees for consulting from Amgen, University Health Network, Daniel Edelman Inc., Autonomic Technologies, Axsome, AbbVie, Alder, Biohaven, Charleston Laboratories, Promius, Eli Lilly, eNeura, Neurolief, Novartis, Ipsen, Impel, Satsuma, Supernus, Theranica, Teva, WL Gore, Nocira, XoC, Zosano, Upjohn (Division of Pfizer), Pieris, Revance, Equinox, Salvia, and Amzak Health. Honoraria: Foresite Capital, ZP Opco, Oppenheimer, Association of Translational Medicine, Healthlogix, Medicom Worldwide, Medlogix Communications, Mednet, Electrocore, Miller Medical, PeerView, WebMD Health/Medscape, Chameleon, Academy for Continued Healthcare Learning, Sun Pharma (India), Universal Meeting Management, Haymarket, Global Scientific Communications, Global Life Sciences, Global Access Meetings, UpToDate (Elsevier), Oxford University Press, Cambridge University Press, and Wolters Kluwer Health. Research Support: Department of Defense, National Institutes of Health, Henry Jackson Foundation, Sperling Foundation, American Migraine Foundation, and Patient Centered Outcomes Research Institute (PCORI). He reports stock options/shareholder/patents/board of directors from Aural Analytics, Healint, Theranica, Second Opinion/Mobile Health, Epien (Options/Board), Nocira, Matterhorn/Ontologics (Options/Board), King-Devick Technologies (Options/Board), and Precon Health (Options/Board). Patent 17189376.1–1466:vTitle: Botulinum Toxin Dosage Regimen for Chronic Migraine Prophylaxis., A. Manack Adams Conflict with: employee of AbbVie and may hold AbbVie stock.
MTV20-DP-010
THE RELATIONSHIP BETWEEN PAIN CATASTROPHIZING LEVEL, EXTREME SENSORY PROCESSING PATTERNS AND HEADACHE SEVERITY AMONG ADOLESCENTS WITH MIGRAINE
Jacob Genizi*
Ayelet Halevy, Mitchell Schertz and Batya Engel-Yeger
Introduction
The incidence of primary headaches and specifically migraine increases during adolescence. Catastrophizing about pain is a critical variable in how we understand adjustment to pain and has a unique contribution in predicting pain intensity. It may enhance disability and negatively affect daily function. Recent reports found that migraine is also related to enhanced sensory sensitivity. However, the relationship between pain severity, pain catastrophizing level and sensory sensitivity requires greater study especially among adolescents.
Objectives
To evaluate the relationship between pain catastrophizing level sensory processing patterns as expressed in daily life, and headache severity among adolescents with episodic migraine.
Methods
Participants were 92 adolescents aged 13–18 years, 40 with episodic migraine and 52 healthy controls. The migraine patients were prospectively recruited from outpatient pediatric neurology clinics. All participants completed the Short Sensory Profile (SSP), and the Pain Catastrophizing Scale for children (PCS-ch). The migraine groups also completed the PedMIDAS, which measures Headache related disability.
Table:
Results
Adolescents with migraine had significantly higher pain catastrophizing level than healthy controls, as seen in enhanced rumination (p ≤ 0.001) and helplessness (p ≤ 0.05). They also had significantly lower tendency to seek sensory input than healthy controls. Elevated rumination and helplessness correlated with higher migraine pain severity. Tendency to avoid sensory input predicted the migraine related disability level.
Conclusion
Sensory processing difficulties are common among adolescents with episodic migraine. Sensory avoidance may be related to pain experience, and pain catastrophizing and disability level.
Disclosure of Interest
None Declared
MTV20-DP-011
HEADACHE EPIDEMIOLOGICAL STUDIES IN CROATIA: A SYSTEMATIC REVIEW
Lukrecija Jakuš1*, Darija Mahović Lakušić2 and Matea Bracic3
1University of Applied Health Sciences
2Department of Neurology, University Hospital Centre Zagreb, School of Medicine Zagreb
3School of Medicine, University of Zagreb, Zagreb, Croatia
Introduction
Recently, we have witnessed the growth of publishing activity in the research of headaches. Despite numerous scientific studies, researchers gathered in the Global Headache Campaign point out that the extent and scale of headaches in the population and the consequent burdens are still underestimated.
Objectives
The main aim of this systematic review was to do an update on studies on primary headache epidemiology in Croatia.
Methods
A literature search was performed with PubMed using the expressions “migraine epidemiology”, “headache epidemiology” and “migraine prevalence” or “headache prevalence” for Croatia. Only studies published as original scientific papers from 1992 to 2020 in English or Croatian were considered. A systematic search of the PubMed identified 8 studies reporting the prevalence, incidence and/or frequency of migraine, tension-type headache, or chronic headache among children, adolescent and adults in Croatia. The information extracted was the region of origin, year of publication, sample characteristics and the prevalence estimates for headache, migraine, tension-type headache, and chronic headache.
Results
Headache epidemiology has been reported in 8 studies conducted since 1999 to 2015: seven studies reported cross-sectional survey, four of them conducted epidemiological population-based survey. Four studies reported about headaches among children and youth (age range 11–18), headache prevalence among high school students (migraine: 16.5%>17% for girls and 8.1%>11.8% for boys; TTH: 18.4%>40.6% for girls and 13.4%>35.7% for boys), headache characteristic, therapy, self-reported causes of headaches, absenteeism from school and quality of life. Those studies were performed on a representative sample of the whole population within a certain age range in ten cities of the Republic of Croatia, a mix of urban, suburban and rural settings, using self-structured questionnaires, and diagnoses were made according to ICHD-II or the earlier ICHD-I or/and Lipton criteria. Four studies reported about headaches among adults (age range 15–65); two were population-based, “face-to-face, door-to-door” epidemiological survey reported: 65.2% headache screen-positive responders (13.92% migraine and 25.5% TTH), and the prevalence of migraine (1-year prevalence: 20.2% and lifetime prevalence: 22.9%) in the adult residents of Bakar. One study reported the 1-year prevalence of chronic headache (2.4%).
Conclusion
There is a lack of population-based studies of headache disorders in Croatia. There are major methodological differences and variable quality among the published studies.
Disclosure of Interest
None Declared
MTV20-DP-012
THE IMPACT OF CORONAVIRUS DISEASE 2019 (COVID‐19) PANDEMIC ON MIGRAINE MANAGEMENT (FACE-TO-FACE VS. TELEHEALTH): PATIENTS’ PERSPECTIVE.
Faraidoon Haghdoost1*, Candice Delcourt2, Cheryl Carcel2, Deepak Chandrasekhar3, Weijie Poh3, François Cadiou3 and Anthony Rodgers1
1The George Institute for Global Health, University of New South Wales, Sydney, Australia
2The George institute for Global Health, The University of NSW, Royal Prince Alfred Hospital, The University of Sydney., Sydney, Australia
3Healint Pte Ltd, Singapore, Singapore, Singapore
Introduction
Migraine is one of the most prevalent neurological disorders globally. Coronavirus disease 2019 (COVID‐19) is a pandemic and has affected the management of other disorders.
Objectives
To evaluate the impact of COVID‐19 era on migraine management from patients` point of view regarding the face-to-face consultations versus telehealth visits.
Methods
This cross-sectional study was a survey of five questions. Questionnaire is reported in table 1 and was sent to users of a migraine tracking mobile phone application named [Migraine Buddy]. Users based in English speaking countries were invited to respond to the survey. Data was self-reported and migraine characteristics are based on records collected through [Migraine Buddy].
Results
1689 users participated in the survey from 16th to 30th of July 2020. Table 1 shows a summary of the responses to each question.
Conclusion
Based on the results of the survey, migraine patients had fewer face-to-face and more telehealth consultations during the COVID-19 pandemic period. Even though users prefer the face-to-face method to telehealth, most users mentioned that the change in the method of consultation had no effect on their headache frequency or on the quality of healthcare.
Disclosure of Interest
F. Haghdoost: None Declared, C. Delcourt: None Declared, C. Carcel: None Declared, D. Chandrasekhar Conflict with: Employee of Healint Pte Ltd. (owner of the Migraine Buddy application) and provided the data., W. Poh Conflict with: Employee of Healint Pte Ltd. (owner of the Migraine Buddy application) and provided the data., F. Cadiou Conflict with: Employee of Healint Pte Ltd. (owner of the Migraine Buddy application) and provided the data., A. Rodgers: None Declared
COVID-19 impact on migraine management.

MTV20-DP-013
A TWENTY-YEAR COMPARISON OF THE BURDEN OF HEADACHE DISORDERS
Tatiana R. F. Cabral1 and Isabel P. Martins2
1Faculdade de Medicina da Universidade de Lisboa
2Faculdade de Medicina da Universidade de Lisboa e Centro de Estudos de Cefaleias do Centro de Estudos Egas Moniz, Lisboa, Portugal
Introduction
Headache is one of the most pervasive disorders worldwide and a significant cause of personal and socioeconomic burden, considering they affect individuals during their most productive years of life. Addressing headache’s repercussions on patients’ quality of life (QoL) is challenging, by its complexity and issues connected to the different experiences and responsibilities involved in patients’ life.
Objectives
To evaluate QoL in patients with headache, understand the contributing factors to the QoL, including the accessibility to information, and to compare the burden of headache disorders and patients QoL with a twenty-year time gap on a defined patient population.
Methods
The study is a prospective observational cross-sectional on adult patients with primary headache, observed in a Headache outpatient clinic of a University Hospital. Surveys were conducted by a structured questionnaire and disease-specific instruments targeting QoL, depression, disability and headache’s impact. A comparative analysis was held between the results obtained and data of a previous project from the year of 2000, led under the same variables.
Results
Among 51 eligible patients, with a mean age of 43.2 ± 13.9 years, most were females (84.3%). The majority of the patients had Episodic Headache (EH) (80.4%), and 19.6% had Chronic Headache (CH), of which 92% had migraine (episodic in 73% and chronic in 19%). CH patients reported superior clinical severity, disability, and poorer QoL than EH patients. Analysis using both groups revealed as the best model of predictors of lower Migraine Specific Quality of Life measure’s (MSQOL) total score, an older age, severe depressive symptoms and disability, headache chronicity and a higher headache frequency. What is more, the epoch factor was shown to be a QoL predictor, when included in this six-variable best predictors model, proposing that contemporary patients have better MSQOL scores compared to those from the control sample.
Conclusion
The headache-related burden is still substantial. Clinical and sociodemographic factors were associated with, and likely predictors of reduced QoL in headache sufferers. More in- depth investigations on this essential topic are needed.
Disclosure of Interest
None Declared
References
MTV20-DP-014
THE ASSOCIATION BETWEEN MIGRAINE IN MIDLIFE AND DEMENTIA IN LATER LIFE – A NATIONAL REGISTER-BASED STUDY
Sabrina Islamoska*, Jakob M. Hansen1, Åse M. Hansen2, Anne H. Garde2, Gunhild Waldemar3 and Kirsten Nabe-Nielsen2
1Danish Headache Center, Rigshospitalet
2Department of Public Health, University of Copenhagen
3Danish Dementia Research Centre, Rigshospitalet, Copenhagen, Denmark
Introduction
Migraine and dementia are prevalent neurological disorders and leading causes of disability. Several pathophysiological mechanisms linking migraine and dementia have been proposed. Indeed, previous studies have reported a higher risk of dementia among individuals with migraine, particularly, migraine with aura. However, no previous studies investigated the risk of dementia among migraine cases identified by both clinical diagnoses and migraine medication.
Objectives
The main objective of our study was to investigate the association between migraine in midlife and the risk of dementia in later life using information on migraine diagnoses and redeemed migraine medication to define migraine cases. The second objective was to investigate whether the risk of dementia was associated with migraine without aura and migraine with aura in combination with the simultaneous use of migraine medication.
Methods
This longitudinal study used national register data obtained for birth cohorts born between 1934 and 1960. Migraine cases were matched (1:5) on sex and birthdate with individuals without migraine leading to 543,025 eligible individuals. Migraine was defined by hospital-based diagnoses of migraine in patient registers and redemption of migraine medication from a prescription register. Dementia was defined by hospital-based diagnoses of dementia, diagnoses from death certificates, and dementia medication after the age of 60 years from patient, prescription and mortality registers.
Results
We observed 94,879 migraine cases and 7,477 dementia cases in our population. The association between migraine and dementia depended on how migraine was identified: hospital-based diagnoses of migraine were associated with a higher rate of dementia (HR = 1.36; 95% CI: 1.14–1.63), whereas migraine identified by redemption of migraine medication was associated with a lower rate of dementia (HR = 0.91; 95% CI: 0.85–0.98). Among individuals with a hospital-based diagnosis of migraine without aura, those who were simultaneously registered as using migraine medication had a non-significant higher rate of dementia (HR = 1.32; 95% CI: 0.92-1.90), which was also the case for those without registered medication (HR = 1.35; 95% CI: 0.79–2.28). Among individuals with a hospital-based diagnosis of migraine with aura, those who were simultaneously registered as using migraine medication had a higher rate of dementia (HR = 1.66; 95% CI: 1.00–2.77), which was also the case for those without registered medication (HR = 1.86; 95% CI: 1.18–2.93).
Conclusion
We observed that migraine identified by patient data was associated with a higher rate of dementia in later life, while migraine identified though prescription data was associated with a lower rate of dementia in later life compared with individuals without registered migraine. Our findings may inspire future studies investigating whether early intervention and treatment of particularly migraine with aura reduce the risk of dementia in later life.
Disclosure of Interest :
None Declared
General aspects of headache care
MTV20-DP-015
PAPER OR ELECTRONIC HEADACHE DIARY, THAT IS THE QUESTION
Teresa Catarci1*
1headache outpatient clinic, azienda sanitaria locale Roma 1, Rome, Italy
Introduction
Headache diary is the most important tool used in headache centres. Preference for electronic (palm-device) versus paper based headache diaries have been reported in headache patients [1] and menstrual related disorders [2]. One study on general adolescent population reported that filling in the internet-based diaries was more bothersome [3]. At the best of our knowledge there are no studies investigating patients’ choice -rather then preference- between the two types of diaries.
Objectives
To investigate the choice of patients with primary headaches when offered a paper or an electronic based headache diary. We also aimed at understanding the possible explanations for the choice of one of the two options.
Methods
All adult consecutive patients with diagnosis of either migraine, tension type headache or both had been selected. They had been included from May 2019 to January 2020 for a first or a follow-up visit, that is when the study had to be interrupted due to the Sars-Cov 2 outbreak. We collected: diagnosis, occupation, education, no. of headache years, no. of headache days per month, choice of type of diary (cellular phone app, paper based, both, none, do not know).
Results
One hundred-thirty-eight patients had been enrolled in the study, 109 females and 29 males, mean age 40,5 ± 13,3 years-old, 78% seen as a first visit. One hundred-one patients reported less then 10 days of headache per month while 28 had more than 15 headache days per month, mean headache days per month was 9,1 ± 8,7. Forty-nine (35,5%) patients choose the app, 76 (55,1%) the paper based diary, 10 (7,3%) both, 2 (1,4%) did not know and 1 (0,7%) did not want to choose (λ2 test p < 0,01). There was a trend for more patients with chronic headache choosing the paper diary (67,8% vs 49,5% – λ2 test p = 0.1856). Also, there were no statistically different rates between younger and older patients and between males and females. No differences were also found between patients seen for the fist time or for a follow-up visit.
Conclusion
In our study more patients choose a paper based diary to record their headaches in contrast with previous preference data [1-2]. Most of the patients were seen as first visit and therefore not biased with prior use of diaries, nevertheless, there were no differences in the choice between the two groups of patients. Probably, a higher number of headache days per month is an important factor to drive the choice toward the paper diary. Our data show that the paper based diary is still the first choice among patients with primary headache irrespective of age and gender. It will be interesting to further study the second choice done by the patients in the follow-up visits and their ultimate preference.
Disclosure of Interest
None Declared
Patients’ choice between paper based and mobile phone app headache diary

References:
MTV20-DP-016
THE ORIGIN OF NECK PAIN IN MIGRAINE: CERVICAL OR NOT?
Zhiqi Liang1*, Gwendolen Jull1, Lucy Thomas1 and Julia Treleaven1
1The School of Health and Rehabilitation Sciences, The University of Queensland, St Lucia, Australia
Introduction
Around 80% of migraineurs report neck pain1 and often seek local treatment of the neck.2 This may or may not be indicated. Neck pain could be due to local nociception from a cervical disorder but it can also be part of migraine symptomology with no relationship to the neck.3 Previous studies of cervical musculoskeletal dysfunction in migraine associated neck pain have failed to acknowledge this disparity in potential causes in their design and treated migraineurs as a homogenous group.4 This does not help to distinguish if the neck pain has a cervical origin or not in individual migraineurs.
Objectives
To determine cervical musculoskeletal function in migraineurs by identifying how episodic and chronic migraineurs, persons with idiopathic neck pain and pain free controls cluster on the basis of cervical function or dysfunction.
Methods
Migraineurs (total n = 124: episodic migraine n = 106, chronic migraine = 18), healthy controls (n = 32) and persons with idiopathic neck pain (n = 21) were assessed using a suite of typical measures for a cervical musculoskeletal disorder, including cervical movement range and accuracy, segmental joint dysfunction, neuromuscular and sensorimotor measures. Migraineurs with diagnoses of comorbid neck disorders were excluded. Cluster analysis was performed with all subjects based on cervical function measures.
Results
Two distinct clusters were found: i) cervical function similar to healthy controls (n = 108), ii) cervical dysfunction similar to persons with neck disorder (n = 69). Sixty-one percent of all migraineurs (n = 76) were clustered as having normal cervical function, whilst the remaining 39% (n = 48) had cervical dysfunction comparable to a neck disorder.
Conclusion
Just over 60% of our cohort of migraineurs with neck pain had normal neck function providing no indication for local neck interventions. Treatment to the neck could be indicated in the 40% who had cervical musculoskeletal dysfunction indicative of a comorbid neck disorder. Further research on this distinct group is required to determine if local treatment has effects on neck pain and or the migraine itself.
Disclosure of Interest
Z. Liang Conflict with: Research Grant from the Physiotherapy Research Foundation, Australian Physiotherapy Association, G. Jull Conflict with: Research Grant from the Physiotherapy Research Foundation, Australian Physiotherapy Association, L. Thomas Conflict with: Research Grant from the Physiotherapy Research Foundation, Australian Physiotherapy Association, J. Treleaven Conflict with: Research Grant from the Physiotherapy Research Foundation, Australian Physiotherapy Association
References
MTV20-DP-017
UNDERTREATMENT OF CHRONIC MIGRAINE PATIENTS IN PRIMARY CARE IN FRANCE, GERMANY, ITALY, SPAIN AND THE UNITED KINGDOM
Angel L Guerrero1*, Andrea Negro2, Philippe Ryvlin3, Kiril Skorobogatykh4, Christina Sundal5, Rainel Sanchez-De La Rosa6 and E. Anne MacGregor7
1Neurology Dpt, Hosp Clinico Univ, Valladolid, Spain
2Clinical & Molecular Med Dpt, Sapienza Univ, Rome, Italy
3Clinical Neurosciences Dpt, CHUV, Lausanne, Switzerland
4Univ Headache Clinic, Moscow, Russian Federation
5Clinical Neuroscience Dpt, Neuroscience and Physiology Inst, Gothenburg, Sweden
6Region Europe Medical Dpt, Novartis Pharma AG, Basel, Switzerland
7Barts Health NHS Trust, London, United Kingdom
Introduction
Despite its substantial burden, migraine and particularly chronic migraine (CM), remains underdiagnosed and undertreated worldwide.
Objectives
To identify how CM patients are managed in primary care in 5 European countries.
Methods
A pan-European scientific committee was created to discuss the needs of non-experts on the management of CM. An online survey was developed and conducted among 200 European GPs from France (FR), Germany (DE), Italy (IT), Spain (ES) and the United Kingdom (UK).
Results
201 GPs participated in the survey (41 from FR and 40 from the other countries, difference occurred during the enrolment process). 64% of the GPs reported using an anamnesis guide for diagnosis, either published or developed by themselves. Some differences were observed among countries, an anamnesis guide that included red flags or warning features to rule out secondary headache was used by 93% and 89% of the participants from UK and ES respectively, compared to 52%, 48% and 19% of the participants from FR, DE and IT respectively (p < 0.001). 85% of the participants reported asking their patients to complete a patient diary mainly in the follow-up visits.
Although only 13% of participants considered they had received enough training on CM, 82% of them were in charge of CM patients’ treatment. 55% of their CM patients had been prescribed a specific acute migraine treatment and 55% of them were taking a preventive treatment. The evaluation of acute or preventive treatment efficacy was mainly done through the patient perception (75% of the participants) and frequency of attacks (60% for the acute treatment evaluation and 50% for the prophylactic treatment).
Among GPs reporting being in charge of patient treatment, the main reason for patient referral to a specialist was: diagnostic confirmation (69% of the participants), lack of efficacy of preventive treatment (67%) or acute treatment (65%), or when migraine was highly disabling (64%).
Conclusion
Considering that the European Headache Federation recommends that patients with CM are referred to specialists, the low referral rate obtained in this study would indicate that the majority of CM patients may not be receiving optimal management, reinforcing the need of more training and guidance in the primary care setting with respect to the management of CM patients.
Disclosure of Interest
A. L. Guerrero Conflict with: consultant and speaker fees from Allergan, Exeltis, Eli Lilly, Novartis and Teva, A. Negro Conflict with: consultant and speaker fees from Allergan, Eli Lilly and Novartis, P. Ryvlin Conflict with: consultant and speaker fees from Eli Lilly and Novartis, K. Skorobogatykh Conflict with: consultant and speaker fees from Novartis and Teva, C. Sundal Conflict with: consultant fees from Allergan and Novartis, R. Sanchez-De La Rosa Conflict with: employee and stakeholder of Novartis Pharma AG, E. A. MacGregor Conflict with: consultant fees from Eli Lilly and Novartis
Genetics and biomarkers of headache disorders
MTV20-DP-018
THE ROLE OF MTHFR GENETIC POLYMORPHISM IN THE SUSCEPTIBILITY TO ACUTE ISCHEMIC STROKE WITHIN THE MIGRANE PATIENTS
Dmytro Sotnikov1* and Olexander Potapov1
1SUMY STATE UNIVERSITY, Sumy, Ukraine
Introduction
The presence of migraine, especially with aura, increases the relative risk of acute ischemic stroke (AIS) at young age. Common to these diseases is the assosiation of C677T polymorphism of the methylenetetrahydrofolate reductase gene (MTHFR).
Objectives
The purpose of this research is to study the influence of the C677T MTHFR gene polymorphism to the risk of AIS within migraine patients.
Methods

Results
Among all AIS patients who had a migraine, the most common was homozygous C677C genotype – 43.2%.
This is comparable to the number of this genotype within AIS patients without migraine – 40%. Among men with migraine, the homozygous T677T genotype was dominated by 42.9%. This genotype was about twice as common for both men and women with migraine, as compared to persons without migraine and carring this genotype.Persons with a homozygous T677T genotype suffering from migraine with aura have risk of AIS 2.5 times more than without migraine (OR = 3.95; p < 0.05) and 2.1 times more than patients with migraine with aura and heterozygote for C677T (OR = 0,29; p < 0.05). Migraine without aura in carriers of homozygote C677C increases the risk of AIS by 2-fold than heterozygotes (OR = 3,18; p < 0.05), and 1.3 times more in people without migraine and same genotype (OR = 1,58; p < 0.05).
Among all AIS patients who had a migraine, the most common was homozygous C677C genotype – 43.2%.This is comparable to the number of this genotype within AIS patients without migraine – 40%. Among men with migraine,the homozygous T677T genotype was dominated by 42.9%. This genotype was about twice as common for both menand women with migraine, as compared to persons without migraine and carring this genotype. Persons with ahomozygous T677T genotype suffering from migraine with aura have risk of AIS 2.5 times more than without migraine(OR = 3.95; p < 0.05) and 2.1 times more than patients with migraine with aura and heterozygote for C677T (OR = 0,29; p < 0.05). Migraine without aura in carriers of homozygote C677C increases the risk of AIS by 2-fold thanheterozygotes (OR = 3,18; p < 0.05), and 1.3 times more in people without migraine and same genotype (OR = 1,58; p < 0.05).
Conclusion
Detection of MTHFR homozygous T677T polymorphism in migraine patients with aura can be used topredict the development of acute ischemic stroke and early prevention in migraine patients, especially in men with riskfactors such as smoking, using ergotamine, arterial hypertension, obesity. However, studies should continue toinvestigate the impact of other MTHFR gene polymorphisms on the development of acute ischemic stroke and migraine.
References:
Detection of MTHFR homozygous T677T polymorphism in migraine patients with aura can be used to predict the development of acute ischemic stroke and early prevention in migraine patients, especially in men with risk factors such as smoking, using ergotamine, arterial hypertension, obesity. However, studies should continue to investigate the impact of other MTHFR gene polymorphisms on the development of acute ischemic stroke and migraine.
Disclosure of Interest
None Declared
MTV20-DP-019
INTERICTAL AMYLIN LEVELS IN CHRONIC MIGRAINE PATIENTS. A CASE-CONTROL STUDY
Pablo Irimia1, Ane Minguez-Olaondo2*, Iván Martínez-Valbuena1, Clara Domínguez-Vivero3, Juan Antonio ánchez-Arias1, Eduardo Martínez-Vila1, Rosario Luquin1 and Rogelio Leira3
1Neurology, Clínica Universidad de Navarra, Pamplona
2Neurology, Hospital Donostia, San Sebastián
3Neurology, Hospital Clínico Universitario de Santiago de Compostela, Santiago de Compostela, Spain
Introduction
Recently, amylin and its receptors were found in different structures involved in migraine pathophysiology.
Objectives
Here, we evaluate interictal concentrations of amylin and calcitonin gene-related peptide (CGRP) in peripheral blood as biomarkers for chronic migraine (CM).
Methods
We prospectively recruited patients with episodic migraine (EM), CM and healthy controls (HC). Interictal amylin and CGRP levels were assessed in blood samples using ELISA.
Results
We assessed plasma samples from 249 participants (58 with EM and 191 with CM) and on 68 HC. Interictal plasma amylin levels were higher in CM patients (47.1 pg/mL) than in the EM patients (28.84 pg/mL, p < 0.0001) and HC (24.74 pg/mL, p < 0.0001). Plasma CGRP levels were increased (20.01 pg/mL) in CM patients when compared to HC (11.37 pg/mL, p = 0.0016), but not to EM patients (18.89 pg/mL, p = 0.4369). Applying a cut-off concentration of 39.68 pg/mL plasma amylin, the sensitivity to differentiate CM from HC was 57.6% and the specificity was 88.2%. Variables such as age, analgesic overuse, depression, allodynia, use of preventive medication or a history of aura, did not influence the plasma concentrations of amylin or CGRP.
Conclusion
Interictal plasma amylin levels are higher in patients with CM and may serve as a diagnostic biomarker for CM
Disclosure of Interest
None Declared
References
MTV20-DP-020
TWO NOVEL BIOMARKERS FOR MIGRAINE IN PERIPHERAL BLOOD MONONUCLEAR CELLS
Paul Rolan1*, Kelsi Dodds1, Florence Lees1, Vasiliki Staikopoulos1, Martin Gosnell2, Ewa Goldys3 and Mark Hutchinson4
1University of Adelaide, Adelaide
2Quantitative Pty Ltd
3University of New South Wales, Sydney
4Nanoscale Biophotonics, University of Adelaide, Adelaide, Australia
Introduction
Biomarkers for migraine could help with diagnosis and treatment selection and monitoring.
Objectives
To determine whether two novel biomarkers can distinguish migraine form non-migraine populations.
Methods
20 patients with episodic migraine (EM), 20 healthy controls (HC) and 8 patients with chronic migraine (CM) were recruited. Peripheral blood mononuclear cells (PBMCs) were isolated from blood. The PainCELL test involved constructing concentration-response relationships to lipopolysaccharide (LPS) in cell culture, measuring the resulting concentration of IL-1b. EC50, Emaxand Hill slope were calculated using R and R studio. The Pain HS (“hyperspectral”) test involved analysis of the native fluorescence of PBMCs to 11 excitation frequencies with analysis of emissions over 18 spectral channels captured by digital camera. After image pre-processing and correction for uneven illumination, 70–90 cells were randomly selected from each image. After each cell was segmented from the background; quantitative features were calculated to parameterise the cell data. This data were used for supervised discriminatory analysis which provided a set of seven feature variables maximising the separation between the sample populations. Receiver operating curves (ROC) were plotted to assess sensitivity and specificity.
Image:

Results
For PainCELL, EM patients were more sensitive to LPS and CM patients less sensitive than HC: EC5033.4, 18.2 and 15.3 pg/ml. CM patients had lower Emaxthan EM or HC : 1467, 2194 and 2258 ng/ml p = 0.0075 for combined analysis. Pain HS was able to discriminate migraine from non-migraine (Figure) with the area under the ROC of 0.92. Discriminant analysis where each segmentation region (cell) is represented as a point in a feature space derived from 7 features. Pairwise statistics are as follows; Healthy-Episodic (Canonical stat distance is 1.17, p-288), Healthy-Chronic (Canonical stat distance is 3.14, p < 5.48e-279), Episodic-Chronic (Canonical stat distance is 0.42, p
Conclusion
A peripheral blood biomarker based on an exaggerated inflammatory profile in PBMCs was able to discriminate migraine populations from healthy controls. This supports the concept of migraine being a neuro inflammatory condition with mirroring of inflammation in the central compartment with that in the periphery. An alternative non-mechanistic approach which interrogates complex biology, and which is suitable for widespread use should be further investigated, especially with regard to potential utility to guide treatment.
Disclosure of Interest :
None Declared
MTV20-DP-021
INVESTIGATING THE LINK BETWEEN CLUSTER HEADACHE AND GENETIC VARIANTS IN THE BIOLOGICAL CLOCK GENE PER3
Felicia M. E. Jennysdotter Olofsgård*, Caroline Ran1, Carmen Fourier1, Christina Sjöstrand2, Elisabet Waldenlind2, Anna Steinberg2 and Andrea Carmine Belin1
1Neuroscience, Karolinska Institutet
2Clinical Neuroscience, Karolinska University Hospital, Stockholm, Sweden
Introduction
A recent study performed by our lab found that 66.7% of a Swedish cluster headache (CH) cohort experienced diurnal rhythmicity connected to their attacks. The biological core clock protein period circadian regulator 3 (PER3) interacts with other key biological clock genes to regulate circadian rhythm. Three PER3 genetic variants rs228697, rs57875989, and rs10462020 have been previously linked to circadian rhythm disturbances such as delayed sleep phase syndrome. For that reason, these genetic variants of PER3 were screened in a Swedish CH case control material to investigate if an association could partially explain the rhythmicity of the attacks.
Objectives
The aim of this study was to investigate possible links between three genetic variants in PER3 and CH while stratifying it for diurnal rhythmicity of attacks.
Methods
DNA was extracted from whole blood in over 700 CH patients and nearly 800 controls. The indel rs57875989 was part of a variable number tandem repeat (VNTR) and genotyped using polymerase chain reaction (PCR) and run on a 3.5% agarose gel. The single nucleotide polymorphisms (SNPs) rs10462020 and rs228697 were genotyped using TaqMan® quantitative PCR.
Results
CH was not found to be associated with either the two SNPs’ genotypes (rs228697; p = 0.845, rs10462020; p = 0.97) or allele frequency (rs228697; p = 0.521, rs10462020; p = 0.916). In addition, there was no association between the indel rs57875989 and CH either in genotype (p = 0.578) or allele frequency (p = 0.847). Patients were further grouped depending on whether they reported a rhythmicity linked to their attacks or not. When stratified for diurnal rhythmicity, there was still no significant difference in genotype between the groups (rs228697; p = 0.920, rs10462020; p = 0.565, rs57875989; p = 0.746).
Conclusion
This study indicates that the three genetic variations of PER3 investigated did not show an association with CH in a Swedish cohort. Findings regarding rs57875989 were in line with results from a Norwegian study (Ofte et al. 2016). Though these genetic variants were not linked with CH, PER3 and other biological clock genes cannot be ruled out as playing a role in the pathophysiology of CH.
Disclosure of Interest
None Declared
Reference
MTV20-DP-022
INSIGHTS FROM WHOLE-EXOME SEQUENCING IN PORTUGUESE FAMILIES WITH PRIMARY HEADACHES
Andreia Dias1*,2, José Pereira-Monteiro1, Jorge Sequeiros1, Maria Paço3, Teresa Pinho3, Alda Sousa1,2, Carolina Lemos1,2, Miguel Alves-Ferreira1,2
1UnIGENe; i3S – Instituto de Investigação e Inovação em Saúde
2ICBAS – Instituto Ciências Biomédicas Abel Salazar, Porto
3IINFACTS, CESPU – Instituto de Investigação e Formação Avançada em Ciências e Tecnologias da Saúde, Gandra, Portugal
Introduction
Primary headaches comprises migraine with or without aura (MA/MO) and cluster headache (CH). During the last 17 years, our group has been able to clinically characterize more than a thousand patients, relatives and controls and we performed several candidate-gene association studies in different pathways. Moreover, we found several variants involved in the vascular component, trigeminal nociceptive plasticity, neurogenic inflammation and in the release of neurotransmitters. However, these complex diseases are caused by several genetic factors and a genome variation study represents a potential solution, but also a big challenge. Whole-exome sequencing (WES) is a powerful approach to explore coding regions, particularly low-frequency variants. As we intend to study the hereditary transmission intra- and inter-families, we will focus mainly on variants with a predicted high impact.
Objectives
To perform a WES in three families to unravel the genetic factors in migraine and CH susceptibility.
Methods
We gathered clinical information and DNA samples from 3 families with primary headaches and associated phenotypes. Afterwards, a WES and bioinformatics analysis were performed.
Results
We analysed 20 patients from the 3 families and, after applying some variant filters, it was possible to find differences between individuals with MA vs. MO. Furthermore, we found common and rare variants in genes already associated with migraine subtypes as CACNA1A and PRRT2 and in new genes that may open new pathways of study.
Conclusion
These preliminary results need to be further explored and interactions among these variants need to be understood to deepen the pathophysiological pathways of migraine. Understanding the mechanisms underlying primary headache pathophysiology could lead to the development of more effective and better-tolerated therapeutic approaches to avoid the overlay of painful events.
Disclosure of Interest
None Declared
MTV20-DP-023
CLINICAL FEATURES OF CADASIL IN FAMILY WITH THE Y189C NOTCH3 MUTATION
Marta Kowalska1*, Tomasz Grzegorski2,3, Dorota Dziewulska4,5, Katarzyna Karmelita-Katulska6, Anna-Maria Barciszewska7, Michal Prendecki1, Wojciech Gorczynski2, Wojciech Kozubski2 and Jolanta Dorszewska1
1Laboratory of Neurobiology, Department of Neurology
2Department of Neurology
3Department of Clinical Neuroimmunology, Poznan University of Medical Sciences, Poznan
4Department of Experimental and Clinical Neuropathology, Mossakowski Medical Research Centre, Polish Academy of Sciences
5Department of Neurology, Medical University of Warsaw, Warsaw
6Department of General Radiology and Neuroradiology
7Department and Clinic of Neurosurgery and Neurotraumatology,, Poznan University of Medical Sciences, Poznan, Poland
Introduction
Cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL) is a hereditary, progressive disease of small cerebral arteries. The disease is characterized by migraine with aura (MA), recurrent subcortical ischemic episodes, vascular dementia and psychiatric disorders. In 20–40% of patients, the first symptom is MA, often with atypical aura, started in the second-third decade of life. CADASIL is caused by mutations in the NOTCH3 gene. We provided the first clinical manifestation of the NOTCH3 Y189C mutation.
is a hereditary, progressive ischemic disease of small vessels of the brain characterized by migraine with aura (MA), recurrent subcortical ischemic episodes, cognitive decline and psychiatric disorders. CADASIL is caused by mutations in the NOTCH3 gene. We identified the NOTCH3 Y189C mutation as a genetic cause of CADASIL in a Polish family and provided its first clinical manifestation.
Objectives
The study included twelve subjects from a Polish family with CADASIL.
The study included twelve subjects from one family. The NOTCH3 mutation, APOE and MTHFR polymorphisms were determined by high-resolution melting analyses (HRMA) and Sanger sequencing. Neuroimaging included CT and MRI. Ultrastructural examination of skin-muscle biopsy material of the proband was performed.
Methods
The NOTCH3 mutation was determined by high-resolution melting analyses (HRMA) and Sanger sequencing. Neuroimaging included CT and MRI. Ultrastructural examination of skin-muscle biopsy material of the proband was performed.
Results
The NOTCH3 Y189C mutation was present in a 36-year-old woman and her two sisters (aged 40 and 27) from 6 siblings. The MA was found in all of them, and started or became more severe after childbirth. The numerous T2/FLAIR hyperintense lesions were shown in the brain MRI. The deposition of granular osmiophilic material in the wall of small vessels of the proband observed in histopathological analysis confirmed the CADASIL diagnosis.
The NOTCH3 Y189C mutation was present in a 36-year-old woman and her two sisters (aged 40 and 27) from 6 siblings. The MA was found in all of them, and started or became more severe after childbirth. The numerous T2/FLAIR hyperintense lesions were shown in the brain MRI. The deposition of granular osmiophilic material in the wall of small vessels of the proband observed in histopathological analysis confirmed the high degree of CADASIL severity
Conclusion
Patients with the Y189C mutation of NOTCH3 from the same family display a similar phenotype of CADASIL.
Disclosure of Interest
None Declared
Headache pathophysiology: basic science
MTV20-DP-024
SRC FAMILY KINASES REGULATE NEUROINFLAMMATION IN A RAT MIGRAINE MODEL
Lingdi Nie1*,2, John Quinn2, Minyan Wang1,2
1Department of Biological Sciences, Centre for Neuroscience, Xi’an Jiaotong-Liverpool University, Suzhou, China
2Department of Molecular and Clinical Pharmacology, University of Liverpool, Liverpool, United Kingdom
Introduction
Cortical spreading depression (CSD) is a key pathophysiological event triggering migraine [1] and is widely used as a preclinical model of migraine [2]. Src family kinases (SFK), a group of non-receptor tyrosine kinases, have an emerging role in migraine pathogenesis [3-6]. Nevertheless, how SFK play their role in this pathological condition is not fully understood.
Objectives
This study aimed to explore whether SFK are involved in CSD-induced neuroinflammatory process, which is essential for central and peripheral sensitization in migraine.
Methods
A single CSD was induced by topical application of potassium chloride on the cerebral cortex of rats and was recorded using electrophysiology. The SFK inhibitor, 3-(4-chlorophenyl) 1-(1,1-dimethylethyl)-1H-pyrazolo[3,4-d]pyrimidin-4-amine (PP2), was administrated to rats by intraperitoneal injection (i.p.) 1 day before CSD induction. Quantitative PCR was used to detect the gene expression levels of the pro-inflammatory cytokines IL-1β and TNFα in the ipsilateral cortices of rats.
Results
The data showed that systematic inhibition of SFK activity prolonged the latency of CSD propagation, which is accompanied by reduced gene expression levels of cortical IL-1β and TNFα.
Conclusion
In conclusion, SFK activation promotes cortical susceptibility to CSD but also the CSD-induced cortical neuroinflammation in rats, suggesting a critical role of SFK in CSD-associated migraine pathogenesis. These data highlight the therapeutic potential of SFK inhibitors in migraine pathophysiology.
Disclosure of Interest
None Declared
References
Headache pathophysiology: clinical
MTV20-DP-025
PLACEBO-CONTROLLED INTRAVENOUS NITROGLYCERIN PHENOTYPING OF ACUTE ATTACKS OF CLUSTER HEADACHE
Diana Y. Wei1,2* and Peter J. Goadsby1,2
1NIHR Wellcome Trust King’s Clinical Research Facility, King’s College Hospital
2Headache Group, Department of Basic and Clinical Neuroscience, King’s College London, London, United Kingdom
Introduction
Cluster headache (CH) is characterised by attacks of unilateral excruciating headache, ipsilateral cranial autonomic symptoms (CAS) and/or agitation. Nitroglycerin (NTG) is known to induce CH. This is the first single blind placebo-controlled study using intravenous NTG in CH to characterise comprehensively NTG induced CH attacks.
Objectives
To investigate in detail the stages of CH attacks: non-headache symptoms, CAS symptoms and attack onset.
Methods
CH patients attended three study visits in total. During the first visit they were unblinded and received intravenous NTG 0.5mcg/kg/min over 20 minutes. If an attack was triggered, they were asked to return for blinded visits either receiving NTG or normal saline, in random order. The clinical phenotype was recorded. The study was approved by an NHS Research Ethics Committee.
Results
Thirty-three participants underwent the unblinded NTG visit, of which blinded NTG visits (n = 25) and blinded placebo infusions (n = 24) were then performed. NTG triggered CH attacks in 79% of patients in the unblinded NTG visit and in 76% in the blinded NTG visit. NTG also brought on non-headache symptoms: 91% unblinded NTG visit, 84% blinded NTG visit, and cranial autonomic symptoms: 94% unblinded NTG visit and 97% blinded NTG visit, with the attacks. Time until CH attacks was shorter in the episodic cluster headache group compared with the chronic cluster headache group.
Conclusion
We demonstrate that weight-adjusted intravenous NTG effectively triggers CH attacks, as well as cranial autonomic symptoms and non-headache symptoms, highlighting potential phenotypical differences between episodic and chronic cluster headache attacks.
Disclosure of Interest
None Declared
MTV20-DP-026
THALAMO-CORTICAL NETWORK ACTIVITY IN SUBGROUPS OF MIGRAINE WITH AURA PATIENTS: INSIGHTS FROM MRI-BASED MICROSTRUCTURAL AND FUNCTIONAL RESTING-STATE NETWORK CORRELATION ANALYSIS
Gianluca Coppola*, Antonio Di Renzo1, Emanuele Tinelli2, Barbara Petolicchio2, Vincenzo Parisi1, Mariano Serrao3, Marco Fiorelli2, Francesca Caramia2, Vittorio Di Piero2 and Francesco Pierelli3
1IRCCS – Fondazione Bietti
2Department of Human Neurosciences, Sapienza University of Rome, Rome
3Department of Medico-surgical Sciences and Biotechnologies, Sapienza University of Rome Polo Pontino, Latina, Italy
Introduction
Due to this variability in clinical presentation, it has been suggested that migraine with aura is a condition characterized by a broad spectrum of clinical subtypes that are likely to differ in pathophysiological mechanisms.
Objectives
We were aimed to study resting-state functional connectivity (FC) between brain networks and its relationship with the microstructure of the thalamus in migraine patients with pure visual auras (MA), and in patients with complex neurological auras (MA+), i.e. with the addition of at least one of sensory and language symptoms.
Methods
3T MRI data from 20 patients with MA and 15 with MA+ were collected and compared with data from 19 healthy controls (HCs). We collected resting state data among independent component networks. Diffusivity metrics values of bilateral thalami were calculated and correlated with resting state ICs Z-scores.
Results
As compared to HCs, both patients with MA and MA+ showed disrupted FC between the default mode network (DMN) and the right dorsal attention system (DAS). MA+ subgroup of patients showed lower microstructural metrics than those of both HCs and MA, and peculiar correlation with the strength of DMN. Despite the microstructural metrics of MA patients did not differ from those of HCs, they did not show the same correlations with the strength of DAS than HCs.
Conclusion
Present findings suggest that clinical heterogeneity of migraine with aura MRI profiles is associated with common and specific morpho-functional features of the nodes of the thalamo-cortical network.
Disclosure of Interest :
None Declared
MTV20-DP-027
THALAMOCORTICAL MORPHOFUNCTIONAL INTEGRITY AND RESTING STATE NETWORKS HAEMODYNAMIC ACTIVITY CHARACTERIZATION BY FRACTAL ANALYSIS IN PATIENTS WITH CHRONIC MIGRAINE
Camillo Porcaro1, Antonio Di Renzo2, Emanuele Tinelli3, Giorgio Di Lorenzo4, Vincenzo Parisi2, Francesca Caramia3, Vittorio Di Piero3, Francesco Pierelli5, Gianluca Coppola*
1Institute of Cognitive Sciences and Technologies (ISTC), National Research Council (CNR)
2IRCCS – Fondazione Bietti
3Department of Human Neurosciences, Sapienza University of Rome
4Laboratory of Psychophysiology and Cognitive Neuroscience, University of Rome Tor Vergata, Rome
5Department of Medico-surgical Sciences and Biotechnologies, Sapienza University of Rome Polo Pontino, Latina, Italy
Introduction
Chronic migraine (CM) can be associated with aberrant long-range connectivity of MRI-derived resting-state networks (RSNs).
Objectives
Here, we investigated how the fractal dimension (FD) of blood oxygenation level dependent (BOLD) activity may be used to estimate the complexity of RSNs, reflecting flexibility and/or efficiency in information processing in CM patients respect to healthy controls (HC).
Methods
Resting-state MRI data were collected from 20 untreated CM without history of medication overuse and 20 HC. On both groups, we estimated the Higuchi’s FD. On the same subjects, fractional anisotropy (FA) and mean diffusivity (MD) values of bilateral thalami were retrieved from diffusion tensor imaging and correlated with the FD values.
Results
CM showed higher FD values within dorsal attention system (DAS) and the anterior part of default-mode network (DMN), and lower FD values within the posterior DMN compared to HC. Although FA and MD were within the range of normality, both correlated with the FD values of DAS.
Conclusion
FD of DAS and DMN may reflect disruption of cognitive control of pain in CM. Since the normal microstructure of the thalamus and its positive connectivity with the cortical networking found in our CM patients reminds similar results obtained assessing the same structures but with the methods of neurophysiology, in episodic migraine during an attack, this may be yet another evidence in supporting CM as a never-ending migraine attack.
Disclosure of Interest
None Declared
Migraine – acute therapy
MTV20-DP-028
COMPARING THE EFFECT OF HEAT AND COLD THERAPY ON THE SEVERITY OF NITRATE INDUCED MIGRAINE TYPE HEADACHE IN CARDIAC INPATIENTS: A RANDOMIZED CONTROLLED TRIAL
Haleh Ghavami*, Aynaz Bagerzadi1, Roghiyeh Emani1, Hamid R. Khalkhali; Student Research Committee of Urmia University of Medical Sciences
1Urmia university of Medical sciences University, Urmia, Iran, Islamic Republic Of
Introduction
Organic nitrates are potent vasodilators and are the most widely used antianginal agents during acute events. They selectively dilate epicardial coronaries and also enhance collateral flow; they also inhibit platelet aggregation. However, headache and tolerance being the major deterrents. NTG-induced headache is the most prominent side effect of nitrate therapy in patients with chest pain and has a detrimental effect on the quality of life.
Objectives
This study aimed to compare the effect of heat and cold therapy on the severity of nitroglycerine induced migraine type headache in cardiac inpatients.
Methods
This randomized controlled trial was conducted on a total of 75 cardiac inpatients in three groups design (heat or cold therapy, and control group) as pre-test and post-test. Patients in the intervention group received heat or cold therapy for25 minutes, two times (at 1-hour interval), Patients in the control group did not receive any heat or cold therapy. Headache severity was measured by the numeric rating scale for pain (NRS Pain), in three groups of study for 3 times (just before the study, at the end of applying the first therapy, and at the end of applying the second therapy).
Results
No baseline differences existed among the three groups for the mean pain scale score (P = 0.781) just before the study; but the difference between three groups after applying heat and cold therapy was statistically significant (p = 0.000).
Conclusion
This study demonstrated applying heat and cold therapy may reduce the severity of nitrate induced migraine type headache in cardiac inpatients. Considering this fact that approximately 10% of patients cannot tolerate nitrate therapies due to unbearable headache, applying heat or cold therapy in patients with nitrate induced migraine type headache is recommended to improve patient’s adherence to treatment.
Disclosure of Interest
None Declared
Comparison of mean and standard deviation of headache intensity in each of the studied groups in the first, second, and third measurements

References
MTV20-DP-029
A PHASE 1, OPEN-LABEL, SINGLE-DOSE PHARMACOKINETIC STUDY OF LASMIDITAN IN PAEDIATRIC PATIENTS WITH MIGRAINE
Max Tsai1*, Emel Serap Monkul Nery1, Lisa Kerr1, Rashna Khanna1, Mika Komori1, Ellen B. Dennehy1 and Darren Wilbraham1
1Eli Lilly and Company, Indianapolis, United States
Introduction
Lasmiditan is a selective serotonin (5-HT1F) receptor agonist approved in the United States (US) for acute treatment of migraine with or without aura in adults.
Objectives
This Phase 1, multicentre, open-label, single-dose study (NCT03988088) aimed to determine pharmacokinetics (PK), safety, and tolerability of lasmiditan in paediatric patients 6 to < 18 years of age with migraine.
Methods
The study was conducted in the US and Japan. Cohort 1 (15 kg to ≤40 kg) received a dose of 100 mg lasmiditan and cohort 2 (>40 kg to ≤55 kg) received a dose of 200 mg lasmiditan. This weight-based dosing was expected to elicit exposures in paediatric patients comparable to adults receiving a 200 mg dose. As PK is generally linear over a clinical dose range of 50 to 200 mg, and 200 mg is the highest approved dose in adults, this exposure was selected to maximize collection of safety and tolerability information. Study procedures were conducted while patients were migraine-free. Blood samples for assessment of PK and safety parameters were collected over a 24-hour (h) period. Parameters included maximum concentration (Cmax), time to maximum concentration, and area under the concentration curve (AUC). Follow-up was ∼14 days after dosing.
Results
Eighteen patients received lasmiditan (11 in cohort 1 and 7 in cohort 2). Seventeen patients completed the study (11 in cohort 1 and 6 in cohort 2). One patient discontinued due to adverse events (AEs).
Following administration, the time course of lasmiditan concentrations was similar between patients receiving 100 mg and 200 mg. Plasma concentrations peaked at a median Cmax time of 1.5 to 2.0 h postdose and then declined with a geometric mean terminal half-life of ∼4.0 h, similar to adult subjects. While the exposure to lasmiditan expressed by Cmax and AUC was generally similar between cohorts, geometric mean values for apparent total body clearance of drug calculated after extra-vascular administration and apparent volume of distribution during the terminal phase after extra-vascular administration were greater for the 200-mg cohort, relative to the 100-mg cohort, as this reflects differences in both dose and body weight in the 2 cohorts.
No deaths or serious AEs were reported. Frequency and severity of AEs (including somnolence, dizziness, and fatigue) were similar to those in adult studies.
Conclusion
The PK results support weight-based dosing of lasmiditan in paediatric patients with migraine 6 to < 18 years of age. Safety and tolerability were similar to that observed in adult studies; no new safety issues were identified in paediatric patients with migraine. These findings support further investigation of lasmiditan as a potential treatment in paediatric patients.
Disclosure of Interest
M. Tsai Conflict with: employee and shareholder at Eli Lilly and Company, E. S. M. Nery Conflict with: employee and shareholder at Eli Lilly and Company, L. Kerr Conflict with: employee and shareholder at Eli Lilly and Company, R. Khanna Conflict with: employee and shareholder at Eli Lilly and Company, M. Komori Conflict with: employee and shareholder at Eli Lilly and Company, E. Dennehy Conflict with: employee and shareholder at Eli Lilly and Company, D. Wilbraham Conflict with: employee and shareholder at Eli Lilly and Company
MTV20-DP-030
LASMIDITAN EFFICACY IN MILD VERSUS MODERATE OR SEVERE MIGRAINE HEADACHES
Mario F. Peres1,2, Raghavendra Vasudeva3, Simin K. Baygani3, Ellen B. Dennehy3,4, Maurice Vincent3 and Deborah I. Friedman5
1Hospital das Clínicas da Faculdade de Medicina
2Hospital Israelita Albert Einstein, São Paulo, Brazil
3Eli Lilly and Company, Indianapolis
4Purdue University, West Lafayette
5University of Texas Southwestern Medical Center, Dallas, United States
Introduction
Early treatment, when pain is mild, is recommended for acute treatment of migraine attacks. Lasmiditan (LTN), a novel, selective 5-HT1F receptor agonist, was superior to placebo in treating moderate to severe migraine attacks in two phase 3 trials. There were few mild attacks studied in the phase 3 program.
Objectives
To evaluate the efficacy of LTN 100 mg and 200 mg in treating migraine attacks of mild versus moderate or severe pain severity.
Methods
This analysis assessed data (intent to treat) from a prospective, randomized, open-label, phase 3 study (GLADIATOR [NCT02565186]) in which patients were randomized 1:1 to LTN 100 mg or 200 mg. Efficacy measures included proportion of attacks with 2-hour pain freedom (PF), 2-hour most bothersome symptom (MBS) freedom, and 24-hour sustained pain freedom (SPF). The proportion of patients with PF, SPF, or MBS freedom among mild, moderate, or severe attacks was compared using Fisher’s exact test.
Results
This analysis includes data from 1,981 patients who treated 17,878 migraine attacks. A total of 273 (1.5%), 11,644 (65.1%), and 5,948 (33.3%) attacks were treated when pain was mild, moderate, and severe, respectively. Average (mean) time from headache onset to treatment administration was 1.3 hours. The table depicts the relative greater proportion of mild attacks with PF, SPF and MBS freedom. Overall, ≥1 treatment-emergent adverse event (TEAE) was reported in 16.8%, 14.3%, and 10.2% of mild, moderate, and severe migraine attacks, respectively. The most frequent TEAE was dizziness, irrespective of headache intensity across the attacks.
Conclusion
Treating migraine attacks at mild pain showed a tendency to relatively better efficacy outcomes, suggesting that earlier dosing is beneficial. Further research is needed to better understand the relationship of LTN outcomes to time of administration in the course of migraine attacks.
Disclosure of Interest
M. Peres Conflict with: advisory board member, and/or speaker, and/or consultant for Allergan, Eurofarma, Libbs, Lilly, Pfizer, Sanofi, Teva, and Novartis. M. Peres serves on the editorial board for Headache Medicine (Editor), Arquivos de Neuro-Psiquiatria (Editorial Board), and Journal of Headache and Pain (Editorial Board). M. Peres also declares a patent (BR2020170233532) and the following research support, government entities, foundations and societies: CNPq, FAPESP, IHS, and Abraces, R. Vasudeva Conflict with: employee and stockholder of Eli Lilly and Company, S. Baygani Conflict with: employee and stockholder of Eli Lilly and Company, E. Dennehy Conflict with: employee and stockholder of Eli Lilly and Company and employee of Purdue University, M. Vincent Conflict with: employee and stockholder of Eli Lilly and Company, D. Friedman Conflict with: advisory board fees from Allergan, Amgen/Novartis, Biohaven Pharmaceuticals, electroCore, Impel, Invex, Lilly, Lundbeck, Revance, Teva, Theranica, and Zosano; grant support (clinical trial) from Allergan, Lilly, and Zosano; grant support (investigator-initiated) from Merck; and consultant fees from electroCore. D. Friedman serves on the editorial board for Neurology Reviews, medical advisory board for Spinal CSF Leak Foundation and HealthyWomen and is a contributing author to MedLink Neurology and Medscape.
Proportion of Migraine Attacks with Pain and Most Bothersome Symptom Freedom

Abbreviations: lasmiditan, LTN
P<0.001;
MTV20-DP-031
REMOTE ELECTRICAL NEUROMODULATION FOR THE ACUTE TREATMENT OF MIGRAINE IN ADOLESCENTS
Andrew D. Hershey1*, Tamar Lin2, Yaron Gruper3 and Frank Berenson4
1Division of Neurology, Cincinnati Children’s Hospital Medical Center, Cincinnati, United States
2Theranica Bio-Electronics, Nertanya
3Theranica Bio-Electronics, Netanya, Israel
4Atlanta Headache Specialists, Atlanta, United States
Introduction
Migraine is a common disabling neurologic disorder in adolescents (12 to 17 years of age). Current acute treatments for migraine in adolescents are mostly pharmacological and may have limited effectiveness, can cause side effects, and may lead to medication overuse. Thus, there is a great unmet need for alternative acute treatments for migraine that are both effective and well tolerated.
Objectives
Remote electrical neuromodulation (REN) is a novel acute treatment of migraine which stimulates upper arm peripheral nerves to induce conditioned pain modulation (CPM) – an endogenous analgesic mechanism in which conditioning stimulation inhibits pain in remote body regions. The REN device (Nerivio®, Theranica Bio-Electronics LTD., Israel) is FDA-authorized for acute treatment of migraine in adults. The current study assessed the efficacy and safety of REN in adolescents with migraine.
Methods
This was an open-label, single-arm, multi-center study in adolescents (ages 12–17 years) with migraine. Participants underwent a 4-week run-in phase during which the headaches were treated according to usual care and recorded in an electronic migraine diary. Eligible participants continued to an 8-week treatment phase in which they were asked to treat their headaches with the device. Pain severity levels, associated symptoms and functional disability were recorded at treatment initiation, 2- and 24-hours post-treatment.
Results
Sixty participants were enrolled, of these 14 failed to meet the run-in criteria and 1 was lost to follow-up. 39 participants completed a test treatment with REN. Pain relief and pain-free at 2 hours were achieved by 71.8% (28/39) and 35.9% (14/39) participants, respectively. Pain relief and pain-free responses were sustained at 24 hours in 90.9% (20/22) of participants and 90.9% (10/11) of participants, respectively. Nausea, photophobia, and phonophobia disappeared at 2 hours in 54.5% (12/22), 41.9% (13/31), and 40.0% (10/25) of participants, respectively. 69.7% (23/33) participants experienced improvement in functional ability at 2 hours. One device-related adverse events were reported in which a temporary feeling of pain in the arm was felt.
Conclusion
REN provides clinically meaningful relief of migraine pain and associated symptoms, offering a safe and effective non-pharmacological alternative for acute treatment in adolescents.
Disclosure of Interest
A. Hershey Conflict with: Andrew Hershey has advised and/or received funding to his institution or himself for this Alder/Lundbeck, Allergan, Amgen, Biohaven, Curelator, Depomed/Assertio, Electrocore, Impax, Lilly, Supernus, Teva, Theranica, Upsher‐Smith, Migraine Research Foundation, and NIH., T. Lin Conflict with: Tamar Lin is employee of Theranica, Y. Gruper Conflict with: Yaron Gruper is employee of Theranica, F. Berenson Conflict with: Frank Berenson has received speaking honorarium and/or serves on the advisory boards of Lilly, Biohaven, Teva, Lundbeck, Currax, and Allergan
MTV20-DP-032
PATIENT ACCEPTABILITY OF A NOVEL UPPER NASAL DELIVERY SYSTEM FOR DHE – USING THE PRECISION OLFACTORY DELIVERY (POD®) DEVICE (INP104)
Stephen Shrewsbury1*, Sheena Aurora1, John Hoekman1 and Maria Jeleva1
1Impel NeuroPharma, Seattle, United States
Introduction
DHE is an effective acute treatment for episodic migraine, but from 1946–1996 was only available as an injection. Nasal DHE (Migranal® ) introduced in 1996 as a much desired needle-free alternative has shown variable efficacy which may be due to loss of the drug product out of the nose onto the upper lip, or down the throat, leading to inconsistent absorption of the remaining drug from the lower/anterior nasal space. Delivery of drugs to the upper nasal space may provide greater, more consistent absorption, reduce response variability and provide more reliable relief similarly without the need for an injection. Assessing the safety and tolerability of delivery to this previously unexplored area is important – but so too is understanding how acceptable patients find drug delivery to this space with the POD device.
Objectives
To assess patient acceptability of INP104 (POD DHE) over 24/52 weeks in the pivotal STOP 301, safety trial.
Methods
In an open label, Phase 3 safety study of INP104 (STOP 301), conducted at 38 centers in the US, patients were asked to complete a questionnaire (PAQ) at the end of 24/52 weeks after using the INP104 product, a self-administered, single use, propellant-enabled device delivering a previously approved formulation of DHE mesylate as required for self-recognized migraine. Subjects were asked to score Strongly Disagree; Disagree; Neutral; Agree; Strongly Agree to the following series of 6 questions once they had completed the study: (1) Study drug is easy to carry, (2) Study drug allows me to return to normal faster, (3) Study drug more consistently treats my migraine, (4) Study drug works faster than previous treatment, (5) Study drug keeps my migraine from coming back and (6) Study drug is easy to use.
Results
324 subjects who entered the 24-week treatment period completed the PAQ. Results are presented in a tabular format. Neutral, Agree or Strongly Agree was noted by: 65% that the device was easy to carry; 78% that it allowed them to return to normal activities faster than previous medication; 76% that it worked more consistently than their previous best usual care; 77% that it worked faster than their previous best usual care; 82% that it kept their migraine from coming back; and 94% that it was easy to use.
Conclusion
The results suggest DHE delivered to the upper nasal space with POD may provide an effective, well tolerated alternative to acute oral treatments for migraine. The majority of study subjects reported INP104 was easy to carry and use, faster and more consistently provided benefit with longer lasting relief and allowed them to return to normal activities faster than previous medications they had used.
Disclosure of Interest :
S. Shrewsbury Conflict with: Employee and stock holder of Impel NeuroPharma, S. Aurora Conflict with: Employee and stock holder of Impel NeuroPharma, J. Hoekman Conflict with: Employee and stock holder of Impel NeuroPharma, M. Jeleva Conflict with: Employee and stock holder of Impel NeuroPharma
MTV20-DP-033
DOES DIHYDROERGOTAMINE TREAT THE “WHOLE MIGRAINE”?
Sheena Aurora*, Sutapa Ray1, Kelsey Satterly1, Stephen Shrewsbury1 and John Hoekman1
1Impel NeuroPharma, Seattle, United States
Introduction
Migraine is recognized as a complex, multifactorial brain network disease associated with 40 different gene loci. Despite a growing body of scientific evidence about the spectrum of migraine pathophysiology including autonomic dysfunction, advances in migraine therapeutics have become increasingly focused on headache pain which may be achieved by targeting a very narrow set of receptors i.e. 5HT1b/d or F or CGRP. Migraine however encompasses a spectrum of symptoms i.e. premonitory phase, aura, photo/phono and osmophobia as well as a postdrome. When a narrow receptor profile is targeted patients do not achieve a holistic migraine relief with lack of consistency, recurrence and accompanying nausea are the prominent reasons for discontinuation.
DHE has demonstrated a larger therapeutic gain compared to triptans [24hr sustained relief, central sensitization, no treatment window therefore it is likely that other receptor targets are involved at the various stages of the migraine cycle. A closer look at the receptor pharmacology of these agents in the context of the migraine cycle may provide insight into the total migraine benefits of treating with a broad receptor binding agent such as DHE versus the limitations of treating with a narrowly targeted therapy. This may further explain why DHE has such a high response rate even in the most difficult to treat migraines.
Objectives
To review the comparative pharmacology of acute treatments for migraine.
Methods
A literature review was conducted to compare the pharmacology and biological activity of new and existing migraine specific treatments.
Results
Comparative receptor binding of current and new migraine specific therapies is presented in tabular format. A model was created to show where in migraine progression each acute migraine specific therapeutic acts to address migraine symptoms.
Conclusion
Unlike other migraine therapeutics, DHE interacts with several different receptor subtypes and therefore is able to exert a greater influence over the pathophysiology of the migraine cycle (premonitory thirst, aura, allodynia, hypersensitivity, withdrawal, ictal pain, vasoconstriction, central sensitization, postdrome and interictal period). Moreover, the slow dissociation of DHE from target receptors is thought to sustain its anti-migraine effects therefore reducing headache recurrence rates and medication overuse headaches. Achieving dose-to-dose consistency and optimal plasma concentrations of DHE has been demonstrated to maximize therapeutic gain while providing safety and tolerability in addressing acute migraine. Advances in delivery systems for DHE will address these issues.
Disclosure of Interest
S. Aurora Conflict with: Employee and stock holder of Impel NeuroPharma, S. Ray Conflict with: Employee and stock holder of Impel NeuroPharma, K. Satterly Conflict with: Employee and stock holder of Impel NeuroPharma, S. Shrewsbury Conflict with: Employee and stock holder of Impel NeuroPharma, J. Hoekman Conflict with: Employee and stock holder of Impel NeuroPharma
MTV20-DP-034
USE NEUROIMAGING FOR PREDICTING SUMATRIPTAN TREATMENT RESPONSE IN PATIENTS WITH MIGRAINE
Jr-Wei Wu1*, Shu-Ting Chen2, Yen-Feng Wang1, Jiing-Feng Lirng2, Kuan-Lin Lai1, Yung-Lin Chen3, Pi-Yi Lai3, Wei-Ta Chen1, Yu-Te Wu3, Shuu-Jiun Wang1,4
1Department of Neurology
2Department of Radiology, Taipei Veterans General Hospital
3Institute of Biophotonics
4Brain Research Center, National Yang-Ming University, Taipei City, Taiwan, Province of China
Introduction
Sumatriptan is a 5-HT1B/1D receptor agonist, peripherally-acting, migraine-specific treatment for acute migraine attacks. However, about 20 to 40% of patients with migraine are not responsive to sumatriptan, and currently, predictors for treatment response is still lacking. In this study, we hypothesized that the response to sumatriptan might not merely reflect pure pharmacological effects but also depend on central modulations. Therefore, we analyzed the neurological substrate of regional brain structures associated with the responsiveness of sumatriptan in migraine patients.
Objectives
To identify the neuroimaging predictors for the responsiveness of sumatriptan.
Methods
We analyzed the brain magnetic resonance imaging of patients with episodic migraine in a headache center. The brain volume of regions of interest (ROI) was calculated, including bilateral amygdala, anterior cingulated cortex, caudate, putamen, precuneus, orbitofrontal cortex, superior frontal gyri, middle frontal gyri, hippocampus, and parahippocampus. Regional brain volumes were automatically calculated using FreeSurfer version 5.3. A sumatriptan-responder was defined as headache free within 2 hours after intake of sumatriptan during migraine attacks.
Results
During the study period, a total of 49 patients with episodic migraine were recruited, including 33 sumatriptan-responders (67.3%) and 16 non-responders (32.7%). Among 20 ROIs, the left hippocampus volume is larger in sumatriptan-responders (responders vs. non-responders: 3914.8 ± 437.4 mm3 vs. 3571.3 ± 420.7 mm3, p = .012). Patients with a larger left hippocampus volume (> 4036.2 mm3 versus ≤ 4036.2 mm3) have a higher response rate for sumatriptan (93.3% versus 55.9%, p = .001). Moreover, the right hippocampus volume correlates with headache frequency (r = 0.368, p = .009) and frequencies of moderate-to-severe headache (r = 0.851, p < .001).
Conclusion
Our study showed that left hippocampus volume could help to predict the treatment response to sumatriptan in patients with episodic migraine.
Disclosure of Interest
None Declared
MTV20-DP-035
EFFECTS OF GALCANEZUMAB ON ACUTE MEDICATION USE AND HEALTH CARE RESOURCE UTILIZATION IN PATIENTS WITH TREATMENT-RESISTANT MIGRAINE: RESULTS FROM A RANDOMIZED, DOUBLE BLIND, PLACEBO-CONTROLLED CLINICAL TRIAL (CONQUER)
Janet Ford1, Julio Pascual Gómez2*, Chad Stroud1, Mallikarjuna Rettiganti1, Emad Estemalik3, Kathleen Day1 and Anna Ambrosini4
1Eli Lilly & Company Corporate Centre, Indianapolis, United States
2Hospital Universitario Marqués de Valdecilla, Santander, Spain
3Cleveland Clinic, Cleveland, United States
4Istituto di Ricovero e Cura a Carattere Scientifico, Pozzilli, Italy
Introduction
CONQUER was a 3-month double-blind (DB) study of patients with episodic or chronic migraine and 2–4 migraine preventive category failures due to lack of effectiveness or safety/tolerability.
Objectives
Assess acute headache medication use (AHM) and health care resource utilization (HCRU) in patients with protocol-defined treatment-resistant migraine treated with galcanezumab (GMB).
Methods
In the 3-month double-blind (DB) study phase, patients with episodic or chronic migraine and 2–4 migraine preventive category failures due to lack of effectiveness or safety/tolerability, received GMB 120 mg/month (following initial 240 mg loading dose) or placebo (PBO); an optional 3month open-label (OL) GMB treatment followed. AHM was self-reported daily with eDiary and paper-forms. HCRU was reported at baseline (retrospectively for previous 6 months) and at monthly visits.
Results
Of the 462 patients (GMB n = 232, PBO n = 230), baseline mean (±SD) days/month of AHM was 12.3 (±6.0); 44.8% had AHM overuse. The percentage of patients reporting migraine-specific HCRU at baseline in the GMB and PBO groups were respectively: 40% and 50% healthcare-professional visits (HCP), 6% and 5% emergency-room (ER) visits, and in each, 2% hospitalizations. LS mean reductions from baseline in the mean number of days/month with AHM in the DB was greater for the GMB group (3.9 to 4.5 days) compared to PBO (0.4 to 1.0 days) in each of the first 3 months; change difference, 3.1 to 3.5, p < 0.001 at each month during Months 1–3. During the OL, reductions from baseline ranged 4.7 to 5.3 days; prior PBO group reductions were comparable to that observed in GMB. During the DB, reductions from baseline of migraine-specific HCP (per 100 person-years) were numerically greater with GMB than PBO (215.5 vs 155.3); during OL, the prior PBO group reductions (212.9) were similar to GMB (222.6). For both groups, migraine-specific ER visits were < 13 and hospitalizations were < 2 per 100 person-years during the DB and OL.
Conclusion
GMB-treated patients with treatment-resistant migraine had clinically meaningful reductions in days with AHM and numerically greater reductions in migraine-specific HCP.
Disclosure of Interest
J. Ford Conflict with: is an employee and a minor stockholder of Eli Lilly and Company, J. Pascual Gómez Conflict with: Julio Pascual Advisory Boards of Allergan, Biohaven, Lilly and Novartis., C. Stroud Conflict with: Chad Stroud is an employee of Eli Lilly and Company, M. Rettiganti Conflict with: Mallikarjuna Rettiganti is an employee of Eli Lilly and Company, E. Estemalik Conflict with: Advisory Boards/Committees: None; Consulting: Lilly; Speakers Bureau/Training: Allergan, Biohaven, Teva, Lilly; Board Memberships: None; Research Grants: None; Ownership interest: None, K. Day Conflict with: Kathleen Day is an employee of Eli Lilly and Company, A. Ambrosini Conflict with: Anna Ambrosini functions as Research Support for Novartis; a consultant for Eli Lilly, Teva, Novartis; Honorarium for Novartis
MTV20-DP-036
PHARMACOKINETICS, SAFETY, AND TOLERABILITY OF UBROGEPANT FOR THE ACUTE TREATMENT OF MIGRAINE FOLLOWING COADMINISTRATION WITH PREVENTIVE MONOCLONAL ANTIBODY TREATMENT
Abhijeet Jakate1*, Andrew M. Blumenfeld2, Ramesh Boinpally1, Matthew Butler1, Lisa Borbridge3, Janette Contreras-De Lama3, Danielle McGeeney1, Antonia Periclou1 and Richard B. Lipton4
1AbbVie, Madison, NJ
2Headache Center of Southern California, Carlsbad, CA
3AbbVie, Irvine, CA
4Albert Einstein College of Medicine, Bronx, NY, United States
Introduction
Ubrogepant, an approved acute treatment for migraine in adults, is used to treat breakthrough attacks in patients receiving preventive treatment.
Objectives
To examine pharmacokinetics (PK), safety, and tolerability of ubrogepant when administered to participants before and after treatment with 1 of 2 monoclonal antibody (mAb) preventive treatments, erenumab or galcanezumab.
Methods
This open-label, multicenter trial enrolled adults diagnosed for ≥1 year with migraine not on preventive treatment. Participants were assigned to an erenumab arm or galcanezumab arm. In each arm, participants received ubrogepant 100 mg on day 1 (without a mAb). On day 8, participants received the assigned mAb: single subcutaneous (SC) erenumab 140 mg injection or galcanezumab 240 mg (2 SC 120 mg injections). Ubrogepant 100 mg was administered once daily on days 12–15. Participants were followed for 30 days. PK parameters, derived from plasma ubrogepant concentrations in blood collected at prespecified timepoints over 24 h postdose on days 1 and 12, included area under the concentration-time curve from time 0 to time t (AUC0-t) or to infinity (AUC0-∞); peak plasma concentration (Cmax); time to Cmax (Tmax); and apparent terminal half-life (t1/2). Safety outcomes included incidence of adverse events (AEs; daily and through follow-up), and laboratory values, vital signs, and electrocardiogram (ECG) results (at prespecified timepoints pre-/postdose). Log-transformed Cmax, AUC0-t, and AUC0-∞ parameters of ubrogepant with mAb vs ubrogepant alone were compared using a linear mixed-effects model with treatment as fixed effect and participant as random effect. Ratios of least squares mean values for Cmax, AUC0-t, and AUC0-∞ for ubrogepant with/without erenumab or galcanezumab with 90% CI were constructed.
Results
Forty participants enrolled (20 per arm). Ubrogepant Cmax, AUC0-t, and AUC0-∞ were minimally elevated after erenumab administration
Conclusion
Ubrogepant PK was unchanged when coadministered with erenumab or galcanezumab. No safety concerns were identified.
Disclosure of Interest
A. Jakate Conflict with: was an employee of AbbVie at the time of the study and may hold AbbVie stock., A. Blumenfeld Conflict with: served on advisory boards and/or has consulted for AbbVie, Depomed, Pernix, Supernus, and Teva; and has received funding for travel, speaking, and/or royalty payments from AbbVie., R. Boinpally Conflict with: employee of AbbVie and may hold AbbVie stock., M. Butler Conflict with: employee of AbbVie and may hold AbbVie stock., L. Borbridge Conflict with: employee of AbbVie and may hold AbbVie stock., J. Contreras-De Lama Conflict with: employee of AbbVie and may hold AbbVie stock., D. McGeeney Conflict with: employee of AbbVie and may hold AbbVie stock., A. Periclou Conflict with: was an employee of AbbVie at the time of the study and may hold AbbVie stock., R. Lipton Conflict with: serves on the editorial boards of Neurology and Cephalalgia and as senior advisor to Headache. He has received research support from the NIH. He has served as consultant or advisory board member for or has received honoraria from AbbVie, Alder, Amgen, Biohaven, Dr. Reddy’s, electroCore, Eli Lilly, eNeura Therapeutics, GlaxoSmithKline, Merck, Novartis, Pernix, Pfizer, TevaVector, and Vedanta. He receives royalties from Wolff’s Headache (8th Edition, Oxford University Press), Informa, and Wiley. He holds stock options in Biohaven and eNeura Therapeutics.

AUC0-t, area under concentration-time curve from time 0 to time t; AUC0-∞, area under concentration-time curve from time 0 to infinity; Cmax, maximum plasma concentration; CI, confidence interval; GLSM, geometric least squares mean; GMR, geometric means ratio; PK, pharmacokinetic.
MTV20-DP-037
EFFICACY AND SAFETY OF UBROGEPANT IN PARTICIPANTS TAKING CONCOMITANT PREVENTIVE MEDICATION
Andrew M. Blumenfeld1*, Kerry Knievel2, Aubrey Manack Adams3, Lawrence Severt4, Matthew Butler4, Hongxin Lai4 and David W. Dodick5
1Headache Center of Southern California, Carlsbad, CA
2Barrow Neurological Institute, Phoenix, AZ
3AbbVie, Irvine, CA
4AbbVie, Madison, NJ
5Mayo Clinic, Phoenix, AZ, United States
Introduction
Ubrogepant is an oral, small-molecule calcitonin gene–related peptide receptor antagonist FDA approved for the acute treatment of migraine with or without aura in adults.
Objectives
The goal of this analysis was to evaluate the potential impact of concomitant preventive medication use on the efficacy and safety of ubrogepant for the acute treatment of migraine.
Methods
ACHIEVE I (NCT02828020) and ACHIEVE II (NCT02867709) were multicenter, double-blind, single-attack, phase 3 trials. The trials included adults with a history of migraine with or without aura. Participants were randomized 1:1:1 in a blinded fashion to placebo or ubrogepant (50 mg or 100 mg, ACHIEVE I; 25 mg or 50 mg, ACHIEVE II). Data from ubrogepant 50 mg and placebo treatment groups were pooled for this analysis. Co-primary endpoints (pain freedom at 2 hours and most bothersome symptom [MBS] freedom at 2 hours) and pain relief at 2 hours postdose were evaluated in the subgroups with and without self-reported use of concomitant preventive medication. After completion of the ACHIEVE trials, participants could enroll in an open-label, 52-week extension trial and were randomized 1:1 in a blinded fashion to ubrogepant 50 mg or 100 mg. Participants treated up to 8 migraine attacks of any pain severity every 4 weeks. Safety and tolerability were assessed.
Results
The analysis included 2246 participants from the ACHIEVE trials; 417 (18.6%) reported use of preventive treatments. In the long-term extension trial, 813 participants were included in the safety population; 143 (17.6%) reported preventive use. No significant differences were observed for pain freedom, MBS freedom, or pain relief at 2 hours between those who did and did not report use of concomitant preventive medication (P>0.05). A numerically greater therapeutic gain was found for those who reported concomitant preventive medication use compared with those who did not; percentages of placebo responders for all outcome measures were greater among those not reporting use of preventive medication. In the long-term extension study, treatment-emergent adverse events (TEAEs) in any dose group were reported by 74% and 68% of participants who did and did not report concomitant preventive medication use, respectively. Treatment-related TEAEs were reported in 8% and 11% between those who reported preventive medication use and those who did not, respectively.
Conclusion
Concomitant preventive medication use did not impact the efficacy of ubrogepant for the acute treatment of migraine and was not associated with additional safety concerns.
Disclosure of Interest
A. Blumenfeld Conflict with: within the past 12 months, has served on advisory boards for, consulted for, and/or been a speaker or contributing author for AbbVie, Alder, Amgen, Biohaven, Lilly, Novartis, Teva, Theranica, and Zoscano. He has received grant support from AbbVie and Amgen., K. Knievel Conflict with: has served as a consultant for AbbVie, Amgen, Eli Lilly, and Biohaven; conducted research with AbbVie, Amgen, and Eli Lilly; and is on speaker programs with AbbVie and Amgen., A. Manack Adams Conflict with: employee of AbbVie, and may hold AbbVie stock., L. Severt Conflict with: employee of AbbVie, and may hold AbbVie stock., M. Butler Conflict with: employee of AbbVie, and may hold AbbVie stock., H. Lai Conflict with: employee of AbbVie, and may hold AbbVie stock., D. Dodick Conflict with: reports the following conflicts: Personal fees: AbbVie, AEON, Alder BioPharmaceuticals, Amgen, Amzak Health, Association of Translational Medicine, Autonomic Technologies, Axsome, Biohaven, Cerecin, Charleston Laboratories, Clexio, Daniel Edelman Inc., Dr Reddy’s Laboratories/Promius, electroCore LLC, Eli Lilly, eNeura, Equinox, Foresite Capital, Impel, Ipsen, Neurolief, Nocira, Novartis, Oppenheimer, Pieris, PSL Group Services, Revance, Salvia, Satsuma, Sun Pharma (India), Supernus, Teva, Theranica, University Health Network, Upjohn (Division of Pfizer), Vedanta, WL Gore, XoC, Zosano, and ZP Opco; Speaking fees: Amgen, Eli Lilly, Lundbeck, and Novartis Canada; CME fees or royalty payments: Academy for Continued Healthcare Learning, Cambridge University Press, Catamount, Chameleon, Global Access Meetings, Global Life Sciences, Global Scientific Communications, Haymarket, HealthLogix, Medicom Worldwide, MedLogix Communications, Mednet, Miller Medical, Oxford University Press, PeerView, Universal Meeting Management, UpToDate (Elsevier), WebMD Health/Medscape, and Wolters Kluwer Health; Stock options: Aural Analytics, Epien, Healint, King-Devick Technologies, Matterhorn, Nocira, Ontologics, Precon Health, Second Opinion/Mobile Health, and Theranica; Consulting without fee: Aural Analytics, Epien, Healint, Second Opinion/Mobile Health; Board of Directors: Epien, King-Devick Technologies, Matterhorn, Ontologics, Paranet North America, and Precon Health; Patent: 17189376.1–1466:vTitle: Botulinum Toxin Dosage Regimen for Chronic Migraine Prophylaxis without fee; Research funding: American Migraine Foundation, Henry Jackson Foundation, PCORI, and US Department of Defense; Professional society fees or reimbursement for travel: American Academy of Neurology, American Brain Foundation, American Headache Society, American Migraine Foundation, Canadian Headache Society, and International Headache Society.
MTV20-DP-038
TRIGEMINAL ALPHA6 SUBUNIT CONTAINING GABAA RECEPTORS AS A NOVEL ANTI-MIGRAINE TARGET: A PROOF-OF-CONCEPT STUDY
Ming Tatt Lee1,2,3, Hung-Ruei Tzeng1, Pi-Chuan Fan4, Daniel E. Knutson5, Tzu-Hsuan Lai4, Werner Sieghart6, James Cook5, Lih-Chu Chiou1,2
1Graduate Institute of Pharmacology
2Graduate Institute of Brain and Mind Sciences, National Taiwan University College of Medicine, Taipei, Taiwan, Province of China
3Faculty of Pharmaceutical Sciences, UCSI University, Kuala Lumpur, Malaysia
4Department of Pediatrics, National Taiwan University College of Medicine, Taipei, Taiwan, Province of China
5Department of Chemistry and Biochemistry, University of Wisconsin-Milwaukee, WI, United States
6Center for Brain Research, Medical University Vienna, Vienna, Austria
Introduction
Hyperactivity of trigeminal ganglion (TG) mediates the activation of the trigeminovascular system (TGVS), the histopathological hallmark of migraine. The α6 subunit-containing GABAA receptor (α6GABAAR) has been reported to be expressed in TG, but its role in migraine was not identified until we demonstrated that an α6GABAAR-selective positive allosteric modulator (PAM), the pyrazoloquinolinone Compound 6, was effective in a rat model of migraine.1
Objectives
We recently developed a Compound 6 derivative, DK-I-56-1 which retained the α6GABAAR-selective PAM property and showed satisfactory pharmacokinetics profiles and a wide therapeutic window in preliminary in vivo studies. Here, we employed DK-I-56-1 to investigate the therapeutic role of α6GABAARs in animal models of migraine.
Methods
The antimigraine effect of DK-I-56-1 was tested histopathologically in a TGVS activation model induced by intracisternal (ic.) capsaicin in rats and behaviorally in a repeated nitroglycerin (NTG)-induced grimace model in mice. The possible psychoactive and addictive properties of DK-1-56-1 were examined in open field and conditioned place preference (CPP) tests, respectively.
Results
Immunohistochemistry studies in ic. capsaicin-treated rats indicated that DK-I-56-I (3–10 mg/kg, i.v.) significantly inhibited the trigeminal nucleus caudalis neuron activation, TG CGRP elevation, and dural CGRP depletion, the signatures of TGVS activation. In mice, DK-I-56-1 (3 and 10 mg/kg, i.p.) exhibited migraine abortive and prophylaxis effects by suppressing the manifestation of migraine-like grimaces, eg. orbital tightening, nose & cheek bulge, and ear & whisker position changes, induced by repeated injections of NTG once every two days for 5 times. Notably, these effects of DK-I-56-1 were comparable with clinically used abortive and preventive anti-migraine agents, sumatriptan and topiramate, respectively. The effects of DK-I-56-1 were prevented by i.p. furosemide, a brain-impermeable α6GABAAR antagonist, suggesting the site-of-action is peripherally located. Importantly, oral administration of DK-I-56-1 (10 mg/kg) achieved a similar efficacy in suppressing NTG-induced grimaces in mice. Lastly, DK-I-56-1 at 10 mg/kg (i.p.) did not affect the spontaneous locomotor activity of mice, nor induced significant CPP when administered repeatedly for 5 times.
Conclusion
These results suggest that TG α6GABAARs are novel therapeutic targets for migraine treatment and the α6GABAR-selective PAM, DK-I-56-1, is a potential candidate devoid of psychoactive and addictive potentials.
Disclosure of Interest
M. T. Lee: None Declared, H.-R. Tzeng: None Declared, P.-C. Fan Conflict with: A patent application has been jointly filed by PCF, DEK, JC, WS and LCC as inventors for the composition and use patents of α6GABAAR-modulating pyrazoloquinolinone compounds for multiple indications. The patent right is co-owned by the inventors’ institutions., D. Knutson Conflict with: A patent application has been jointly filed by PCF, DEK, JC, WS and LCC as inventors for the composition and use patents of α6GABAAR-modulating pyrazoloquinolinone compounds for multiple indications. The patent right is co-owned by the inventors’ institutions., T.-H. Lai: None Declared, W. Sieghart Conflict with: A patent application has been jointly filed by PCF, DEK, JC, WS and LCC as inventors for the composition and use patents of α6GABAAR-modulating pyrazoloquinolinone compounds for multiple indications. The patent right is co-owned by the inventors’ institutions., J. Cook Conflict with: A patent application has been jointly filed by PCF, DEK, JC, WS and LCC as inventors for the composition and use patents of α6GABAAR-modulating pyrazoloquinolinone compounds for multiple indications. The patent right is co-owned by the inventors’ institutions., L.-C. Chiou Conflict with: A patent application has been jointly filed by PCF, DEK, JC, WS and LCC as inventors for the composition and use patents of α6GABAAR-modulating pyrazoloquinolinone compounds for multiple indications. The patent right is co-owned by the inventors’ institutions.
Reference
MTV20-DP-039
LONG-TERM CARDIOVASCULAR SAFETY OF LASMIDITAN FOR THE ACUTE TREATMENT OF MIGRAINE FOR UP TO ONE YEAR: INTERIM RESULTS OF AN OPEN-LABEL PHASE 3 STUDY (GLADIATOR)
Andrew Buchanan1, Simin K. Baygani1, Noah Rosen2, Paul G. Mathew3, Helen Hochstetler1* and Rashna Khanna1
1Eli Lilly and Company, Indianapolis
2Hofstra Northwell School of Medicine, Hempstead
3Harvard Medical School, Boston, United States
Introduction
Lasmiditan, a selective serotonin (5-HT) 1F receptor agonist, lacks vasoconstrictive activity of other acute migraine treatments. Previous studies exhibited that the drug has low risk of associated cardiovascular (CV) treatment-emergent adverse events (TEAEs); no difference in CV TEAEs frequency in subjects with/without CV risk factors.
Objectives
Long-term CV safety for acute migraine treatment was assessed by analyzing interim results from Phase 3 study GLADIATOR.
Methods
GLADIATOR patients who previously participated in Phase 3, placebo-controlled, single-attack lasmiditan studies were randomized 1:1; open-label lasmiditan 100/200 mg taken as 1 dose (within 4 hrs of moderate/severe pain onset); 2 doses (within 2–24 hrs). Previous studies included patients with CV risk factors, coronary artery disease (CAD), arrhythmia, uncontrolled hypertension. CV adverse event rates were calculated for 3 time periods – treatment-emergent ( < 48 hrs postdose), intermediate (48 hrs–1 week postdose), remote (>1 week postdose) – as some CV events were likely not reported as treatment emergent/were identified later. CV events occurring >48 hrs postdose were analyzed to determine whether they increased in treatment-emergent/intermediate periods versus remote period.
Results
1978 patients received ≥1 dose of lasmiditan. 19058 migraine attacks were treated. Study median duration was 288 days. 81% patients had ≥1 CV risk factor, 0.3% had CAD, 35% had low high-density lipoprotein cholesterol. No vasoconstriction-related CV events occurred in treatment-emergent period; such events were rarely observed (between 1–3, all in remote period, limiting clinical interpretation). Treatment-related CV events (palpitations, tachycardia) were more frequent in treatment-emergent period versus intermediate and remote periods (Table 1).
Conclusion
Interim analysis of long-term GLADIATOR study found no vasoconstriction-related CV events occurred during treatment-emergent period. CV safety of lasmiditan was generally consistent with data from single-attack studies.
Disclosure of Interest
A. Buchanan Conflict with: Andrew Buchanan is an employee and a minor stockholder of Eli Lilly and Company, S. Baygani Conflict with: Simin Baygani is an employee and a minor stockholder of Eli Lilly and Company, N. Rosen Conflict with: Noah Rosen has received research support from Allergan, Eli Lilly, and Theranica; has served on a speaker panel for Allergan; has received compensation as a consultant for Alder, Allergan, Amgen, Avanir, Biohaven, Eli Lilly, Impel, Promius, Revance, Supernus, and Teva; has received honoraria from the American Headache Society and the American Academy of Neurology; and has received honoraria from Springer for his role as assistant editor for Current Pain and Headache Reports, P. Mathew Conflict with: Paul Mathew served as an uncompensated consultant and not in his capacity as a member of the HMS/BWH Faculty. He has served as a consultant for Allergan, Amgen, Avanir, BioMobie, Biohaven, ElectroCore, Ipsen, Lilly, Novartis, Promius, Revance, Satsuma, Stealth BioTherapeutics, Supernus, Takeda, Theranica, Teva, and Upsher-Smith, H. Hochstetler Conflict with: Helen Hochstetler is an employee and a minor stockholder of Eli Lilly and Company, R. Khanna Conflict with: Rashna Khanna is an employee and a minor stockholder of Eli Lilly and Company
Event rates (person–time-adjusted)* for CV events in patients with migraine treated with lasmiditan 100/200 mg over 3 time periods in Phase 3 study GLADIATOR.

Time-adjusted cardiovascular event rate/1000 patient-years of exposure.
MTV20-DP-040
TOLERABILITY OF LASMIDITAN IN ELDERLY PATIENTS WITH MIGRAINE
Andrew Buchanan1, Vincent Martin2, Zubair Ahmed3, Simin Baygani1, Helen Hochstetler1, Rashna Khanna1 and Emel Serap Monkul Nery1*
1Eli Lilly and Company, Indianapolis
2University of Cincinnati, Cincinnati
3Cleveland Clinic, Cleveland, United States
Introduction
Tolerability of lasmiditan, a selective serotonin (5-HT) 1F receptor agonist, was evaluated in elderly and non-elderly patients in a pooled analysis of 2 Phase 3 studies of lasmiditan in acute treatment of migraine.
Objectives
Evaluate treatment-emergent adverse events (TEAEs) occurring within 48 hours of the first dose in elderly versus non-elderly lasmiditan-treated patients.
Methods
SAMURAI and SPARTAN were randomized, double-blind, controlled studies of adults who took placebo or lasmiditan 50, 100 or 200 mg for a single migraine attack, within 4 hours of the onset of moderate or severe pain. This post-hoc analysis of pooled data evaluated TEAEs, defined as adverse events occurring within 48 hours of the first dose (regardless of whether a second dose was taken), in elderly (aged ≥65 years) versus non-elderly ( < 65 years) lasmiditan-treated patients. Treatment-by-age subgroup interactions were examined using logistic regression analysis.
Results
Analysis included 3177 lasmiditan-treated patients of whom, 132 (4.2%) were elderly; 1262 placebo-treated patients of whom, 54 (4.3%) were elderly. The percentage of patients with at least 1 TEAE was similar for elderly (34.8%) and non-elderly (35.7%) lasmiditan-treated patients. No significant treatment-by-age subgroup interactions were seen in patients who reported at least 1 TEAE overall or for any individual TEAE. There were some numerical differences in incidences of individual TEAEs between elderly and non-elderly patients treated with lasmiditan (Table 1). However, no trend was identified nor any safety signals observed in the elderly age group.
Conclusion
In a pooled analysis of 2 Phase 3 studies, the overall incidence of TEAEs with lasmiditan was similar for elderly and non-elderly patients with migraine. However, numerical differences in the incidence of some specific TEAEs were seen.
Disclosure of Interest
A. Buchanan Conflict with: Andrew Buchanan is an employee and a minor stockholder of Eli Lilly and Company, V. Martin Conflict with: Vincent Martin has served as a speaker for Amgen, a consultant for Alder and Theranica, and a speaker and consultant for Allergan, Biohaven, Eli Lilly and Company, and Teva Pharmaceuticals, Z. Ahmed Conflict with: Zubair Ahmed has received consulting fees from Allergan, Amgen, and Eli Lilly; has served as a speaker for Allergan; and has received research funding from Teva, S. Baygani Conflict with: Simin Baygani is an employee and a minor stockholder of Eli Lilly and Company, H. Hochstetler Conflict with: Helen Hochstetler is an employee and a minor stockholder of Eli Lilly and Company, R. Khanna Conflict with: Rashna Khanna is an employee and a minor stockholder of Eli Lilly and Company, E. S. M. Nery Conflict with: Emel Serap Monkul Nery is an employee and a minor stockholder of Eli Lilly and Company
MTV20-DP-041
WHO IS ELIGIBLE FOR NOVEL MEDICATIONS DESIGNED FOR ACUTE TREATMENT OF MIGRAINE AND WHAT ARE THEIR UNMET NEEDS? RESULTS OF OVERCOME STUDY
Gerhardt Pohl1, Richard Lipton2, Andre Araujo3, Robert Nicholson1, Dawn Buse2*, Robert Shapiro4, Sait Ashina5, Michael Reed6, Dena Jaffe7, Janelle Cambron-Mellott7, Vicky Li7, Anthony Zagar1, Erin Doty1 and Eric Pearlman1
1Eli Lilly and Company, Indianapolis
2Albert Einstein College of Medicine, Bronx
3Formerly of Eli Lilly & Company Corporate Centre, Indianapolis
4Larner College of Medicine, Burlington
5Harvard Medical School, Beth Israel Deaconess Medical Center, Boston
6Vedanta Research, Chapel Hill
7Kantar Health, New York, United States
Introduction
A recent American Headache Society (AHS) Consensus Statement (Headache 2019;59:1-18) indicates that novel medications for acute migraine treatment are to be considered in “ patients who have contraindications to the use of triptans or who have failed to respond to or tolerate at least 2 oral triptans.”
Objectives
Estimate percentage of those with migraine meeting these criteria; characterize their unmet medical needs.
Methods
Data were obtained in 2018 from web-based survey of representative US sample. Present sample included 21143 people with migraine. To understand those with greatest care needs, people with ≥4 migraine headache days (MHDs)/month were oversampled. Survey included migraine Treatment Optimization Questionnaire (mTOQ: measures adequacy of treatment for current acute therapy). We examined population size of those potentially eligible for novel medications by forming 3 groups: people not currently on oral triptan but with history of using, discontinuing at least 2 oral triptans [Group A]; people currently using oral triptan with mTOQ sum score < 6 (poor/very poor treatment efficacy), history of failing at least 1 prior oral triptan [Group B]; people with potential contraindications to triptans [Group C]. We examined medication use, healthcare visits among these groups.
Results
Sample mean(SD) age:42(15)years; female:74%. 12299(58.2%) people with migraine reported 0–3 MHDs/month; 8844(41.8%) reported ≥4 MHDs/month. Combining Groups A, B, C, 2594(21.1%) people with 0–3 MHDs and 2448(27.7%) with ≥4 MHDs are eligible for novel medications. 30.4% of eligible population with 0–3 MHDs were using oral triptan (contraindicated/reporting poor/very poor treatment efficacy). Oral triptan use among those with ≥4 MHDs (39.3%) was higher. Similar pattern was seen for opioids and barbiturates. Regarding healthcare utilization in the past year, 57.7%(0–3 MHDs) and 75%(≥4 MHDs) of eligible people sought migraine care; 23% and 30.3% sought emergency room care.
Conclusion
Among people with migraine, regardless of MHDs/month, roughly 1/4 may qualify for novel medications to be used acutely for migraine attack, according to AHS Consensus Statement. Use of non-preferred treatments and emergency room care highlight substantial opportunities to improve care in this population.
Disclosure of Interest
G. Pohl Conflict with: Gerhardt Pohl is an employee and a minor stockholder of Eli Lilly and Company, R. Lipton Conflict with: Alder, Allergan, American Academy of Neurology, American Headache Society, Amgen, Autonomic Technologies, Avanir, Biohaven, Boston Scientific, Cephalalgia, Dr. Reddy’s, Electrocore, Eli Lilly, eNeura Therapeutics, GlaxoSmithKline, Headache, Informa, Merck, Migraine Research Foundation, National Headache Foundation, Neurology, NIH, Novartis, Pfizer, Teva, Vedanta, A. Araujo Conflict with: Andre Araujo is a former employee of Eli Lilly and Company, R. Nicholson Conflict with: Robert Nicholson is an employee and a minor stockholder of Eli Lilly and Company, D. Buse Conflict with: Dawn Buse has received research support from the FDA-NIH, Amgen, Allergan, Dr. Reddy’s and Lilly and is a consultant to Amgen, Allergan, Dr. Reddy’s, Eli Lilly and Teva. She is a section editor for Current Pain and Headache Reports, R. Shapiro Conflict with: Robert Shapiro is a consultant for Eli Lilly and Company, S. Ashina Conflict with: Sait Ashina does Consulting, Speaking and Teaching, Honoraria for Allergan, Amgen, Eli Lilly and Company, Novartis, Promius, Teva, M. Reed Conflict with: Michael Reed does Consulting, and has Research Grants for Allergan, Amgen, Eli Lilly and Company, Promius, Vedanta, D. Jaffe Conflict with: Nothing to disclose, J. Cambron-Mellott Conflict with: Nothing to disclose, V. Li Conflict with: Nothing to disclose, A. Zagar Conflict with: Anthony Zagar is an employee and a minor stockholder of Eli Lilly and Company, E. Doty Conflict with: Erin Doty is an employee and a minor stockholder of Eli Lilly and Company, E. Pearlman Conflict with: Eric Pearlman is an employee and a minor stockholder of Eli Lilly and Company
MTV20-DP-042
MULTIPLE-ASCENDING DOSE, SAFETY, TOLERABILITY, PHARMACOKINETIC, AND DRUG-DRUG INTERACTION STUDY WITH LASMIDITAN
Darren Wilbraham1, Debra Luffer-Atlas1, Max Tsai1* and Dustin D. Ruff1
1Eli Lilly and Company, Indianapolis, United States
Introduction
Lasmiditan is a highly selective, potent serotonin 1F receptor agonist approved in US for acute treatment of migraine with/without aura in adults.
Objectives
- Evaluate safety, tolerability, pharmacokinetics (PK) in healthy subjects after multiple doses of 200 mg and 400 mg lasmiditan.
- Assess effect of multiple doses of 200 mg dose on cytochrome P450 (CYP) activity, based on in vitro data suggesting CYP induction by lasmiditan.
Methods
This was an investigator-and-subject-blinded, randomized, placebo (PBO)-controlled multiple ascending-dose study in 2 cohorts of healthy subjects. Cohort 1 (C1) subjects received probe drug cocktail of CYP substrates (100 mg caffeine, 500 mg tolbutamide, 2 mg midazolam) on day -3 then daily oral doses of 200 mg lasmiditan/PBO (7:3 lasmiditan to PBO) on day 1 for 7 days. On day 7, drug cocktail was co-administered with lasmiditan. Cohort 2 (C2) subjects received daily oral doses of 400 mg lasmiditan/PBO (1:1 lasmiditan to PBO) for 7 days. Serial blood samples for lasmiditan were collected for 24 hours (hr) postdose on days 1 and 7 plus pre-dose on days 3–5. Serial blood samples from C1 subjects were collected for 48 hr after drug cocktail dosing to assess probe drug PK. Log-transformed Cmax and area under the curve (AUC) estimates for probe drugs were evaluated in linear mixed-effects analysis of variance model with fixed effect for treatment and random effect for subject.
Results
70 subjects (57.1% male) aged 20–63 participated in the study. No deaths/other serious adverse events (AEs) occurred. 2 subjects discontinued due to folliculitis (PBO) and nodal rhythm (lasmiditan). 22 of 40 C1 subjects reported treatment-emergent AEs (TEAE). 9 of 30 C2 subjects reported TEAEs. Most common TEAE (≥10%) was dizziness. More subjects reported TEAEs, all causality AEs in C1 versus C2. More subjects reported TEAEs when receiving lasmiditan alone [42.9% (C1), 26.7% (C2)] versus PBO alone [8.3% (C1), 13.3% (C2)]; most TEAEs occurred on day 1 for subjects receiving lasmiditan. No increase in AEs was noted on administration of lasmiditan with probe drug cocktail on day 7. Lasmiditan was orally absorbed (median tmax of 2 hr) and eliminated (geometric mean t1/2 of ∼4 hr); no accumulation of lasmiditan was observed. Daily administration of 200 mg lasmiditan for 7 days had no clinically relevant effects on PK of caffeine, tolbutamide, or midazolam; 90% confidence intervals for Cmax and AUC parameters were within 0.8 to 1.25 range.
Conclusion
Multiple doses of 200 mg and 400 mg lasmiditan were well-tolerated in healthy subjects, no new safety issues were identified. No accumulation of lasmiditan was observed with daily dosing. Lasmiditan does not appear to inhibit or induce the activities of CYPs 1A2, 2C9, or 3A.
Disclosure of Interest
D. Wilbraham Conflict with: Darren Wilbraham is an employee and a minor stockholder of Eli Lilly and Company, D. Luffer-Atlas Conflict with: Debra Luffer-Atlas is an employee and a minor stockholder of Eli Lilly and Company, M. Tsai Conflict with: Max Tsai is an employee and a minor stockholder of Eli Lilly and Company, D. Ruff Conflict with: Dustin D Ruff is an employee and a minor stockholder of Eli Lilly and Company
Migraine – preventive therapy
MTV20-DP-043
INSIGHTS INTO THE PERSONAL EXPERIENCE OF MIGRAINE PATIENTS AND THE POSITIVE IMPACT ON THEIR QUALITY OF LIFE DURING ERENUMAB TREATMENT
Katrin Schuh1*, Heike Israel-Willner2, Mirja Koch1 and Charly Gaul3
1Clinical Research Neuroscience, Novartis Pharma GmbH, Nueremberg
2NFZB, Specialized Center of Neurology Berl, Berlin
3Migraine and Headache Clinic,, Koenigsstein/Taunus, Germany
Introduction
Erenumab (Aimovig) is a fully human monoclonal antibody against the calcitonin gene-related peptide (CGRP) receptor approved for the preventive treatment of migraine in adults. Up until now, “reduction of monthly migraine days” has served as a crucial factor to evaluate therapeutic success of prophylactic migraine treatment. From the patient´s perspective however, other factors can have a high impact on their contentment with the achieved therapeutic outcome.
Objectives
PERISCOPE evaluates the benefit of the human monoclonal antibody Erenumab on the quality of life from the patients’ point of view.
Methods
29,042 patients suffering from migraine completed an online survey from July to December 2019 on their migraine history and their experience with pharmacological and non-pharmacological therapies. Among the participants, 155 patients were undergoing erenumab treatment and were asked to provide insights into their perception of therapeutic outcomes.
Results
89.9% of all erenumab treated patients reported improved quality of life, mostly related to more pain free days (81%) and reduced pain intensity (51.2%). Of patients not reaching a 50% reduction in monthy migraine days (n = 33), 75.8% reported an improvement in quality of life. After three months of erenumab therapy monthly migraine days were reduced from an average of 15.4 days (prior to treatment) to 7.0 days. Mean acute medication use was reduced by −5.47 monthly days on average. 96.1% of all patients reported they would recommend erenumab to their friends and relatives.
Conclusion
PERISCOPE provides real-word evidence from the migraine patients’ perspective. This data builds upon our knowledge of erenumab´s benefit as assessed in controlled trials, capturing the therapeutic impact as perceived by the patient.
Disclosure of Interest
K. Schuh: None Declared, H. Israel-Willner Conflict with: Heike Israel-Willner received honoraria for consulting and lectures within the past 3 years from Allergan Pharma, Novartis Pharma, Desitin Arzneimittel and Teva. She does not hold any stocks of pharmaceutical companies or medical device companies., M. Koch Conflict with: employee of Novartis Pharma GmbH, C. Gaul Conflict with: Charly Gaul has received personal compensation for consulting, serving on a scientific advisory board, speaking, or other activities with Allergan Pharma, Ratiopharm, Boehringer Ingelheim Pharma, Lilly, Novartis Pharma, Desitin Arzneimittel, Cerbotec, Bayer vital, Hormosan Pharma, electroCore, Reckitt Benckiser, and Teva.
MTV20-DP-044
REAL-WORLD EVIDENCE DATA REVEALS THE PHYSICIANS’ PERCEPTION ON ERENUMAB THERAPY IN GERMAN HEADACHE CENTERS
Mirja Koch1*, Katrin Schuh1, Charly Gaul2, Philipp Stude3 and Andreas Straube4
1Clinical Research Neuroscience, Novartis Pharma GmbH, Nuremberg
2Migraine and Headache Clinic, Koenigstein am Taunus, Koenigstein
3Practice for Neurology, Bochum
4Clinic for Neurology, Ludwig-Maxilmilians-University, Munich, Germany
Introduction
Erenumab (Aimovig) is a first-in-class calcitonin gene-related peptide (CGRP) receptor inhibitor approved for the preventive treatment of migraine in adults. Real-world evidence data is highly valued and has become increasingly important in providing evidence of treatment effectiveness in clinical practice.
Objectives
TELESCOPE aimed to collect real-world data on erenumab treatment outcome from the physicians’ perception in daily clinical routine.
Methods
45 German headache centers were surveyed from July to December 2019. Physicians with a broad range of migraine patients provided information on their therapy decisions and therapeutic effects of erenumab. In addition, physicians reported therapeutic outcomes of individual erenumab recipients, in total 542 patients.
Results
Restricted quality of life (100%) and number of monthly migraine days (98.8%) were main reasons for treatment initiation with erenumab as stated by the 45 neurologists. According to the treating physicians’ reports for all erenumab patients in the last quarter, erenumab reduced headache intensity (77.4% of patients), improved quality of life (75.5% of patients) and reduced monthly migraine days by half (66% of patients). For 69.4% of patients, treating physicians saw a response already after the first injection. Mean change in monthly migraine days was -6.2 (12.1 vs. 5.9 ) and mean change in acute medication days was -6.4 (11.5 vs. 5.1) for 542 documented patients.
Conclusion
The TELESCOPE project reveals real-world evidence data for erenumab in German headache centers. According to physicians’ perception, erenumab treatment showed therapeutic benefit for the majority of their patients with improvement in quality of life in more than 75% of patients.
Disclosure of Interest
M. Koch Conflict with: employee of Novartis Pharma GmbH, K. Schuh Conflict with: employee of Novartis Pharma GmbH, C. Gaul Conflict with: Charly Gaul has received personal compensation for consulting, serving on a scientific advisory board, speaking, or other activities with Allergan Pharma, Ratiopharm, Boehringer Ingelheim Pharma, Lilly, Novartis Pharma, Desitin Arzneimittel, Cerbotec, Bayer vital, Hormosan Pharma, electroCore, Reckitt Benckiser, and Teva., P. Stude Conflict with: Phillip Stude has received honoraria for consulting, speaking or other activities from Allergan, Eli Lilly, Hormosan, Novartis, TEVA., A. Straube Conflict with: Andreas Straube has received personal compensation for consulting, serving on a scientific advisory board, speaking, or other activities with Allergan, Allergosan, Bayer, electroCore, Eli Lilly, Novartis, Sanofi, TEVA. Dr. Straube has received research support from Novartis Pharma
MTV20-DP-045
PATIENT‐REPORTED OUTCOMES IN PATIENTS WITH MIGRAINE AND PRIOR PROPHYLACTIC TREATMENT FAILURE: A SUBGROUP ANALYSIS FROM THE BECOME STUDY
Patricia Pozo-Rosich1*, Christian Lucas2, Charly Gaul3, David Watson4, Paolo Martelletti5, Shannon Ritter6 and Josefin Snellman7
1Headache Unit, Neurology Department, Vall d’Hebron University Hospital, Barcelona, and Headache and Neurological Pain Research Group, Vall d’Hebron Institute of Research (VHIR), Universitat Autònoma de Barcelona, Barcelona, Spain
2Pain Clinic, Service de Neurochirurgie, Hô pital Salengro, CHRU de Lille, Lille Cedex, France
3Migraine and Headache Clinic Königstein, Königstein im Taunus, Germany
4Hamilton Medical Group, Aberdeen, Scotland, United Kingdom
5Department of Clinical and Molecular Medicine, Sapienza University of Rome, Sant’Andrea Hospital, Via di Grottarossa, Rome, Italy
6Novartis Pharmaceuticals Corporation, East Hanover, NJ, United States
7Novartis Pharma AG, Basel, Switzerland
Introduction
Migraine-induced disability impairs daily functioning, quality of life and presents substantial societal burden. BECOME study assessed the
Objectives
To report the subgroup analysis from the BECOME study of patient‐reported outcomes (PROs) in patients with migraine and PPTF (1, 2, 3, ≥4 medication categories) due to lack of efficacy and/or poor tolerability.
Methods
The BECOME study assessed disease characteristics of all patients with migraine visiting headache specialist centres (Part 1), and burden of disease using PROs and healthcare resource utilisation questionnaires in patients with
Image:
Results
Overall, 2419 patients were included in Part 2 analysis. The WPAI-headache scores indicated a high level of impairment due to migraine on absenteeism (15.6%), presenteeism (48.6%), and overall work impairment (52.6%) domains (
Conclusion
This subgroup analysis of the BECOME study confirms the burden of migraine on work productivity, anxiety, and quality of life in migraine patients, and an incremental increase with the number of PPTFs.
Disclosure of Interest
P. Pozo-Rosich Conflict with: received honoraria as a consultant and speaker during the last 5 years from Allergan, Almirall, Chiesi, Eli Lilly, Novartis and Teva. Her research group has received research grants from Allergan and funding for clinical trials from Alder, Boehringer Ingelheim, MSD, electroCore, Eli Lilly, Janssen Cilag, and Novartis., Conflict with: She is a trustee member of the board of the International Headache Society and a Member of the Council of the European Headache Federation. She is on the editorial board of Revista de Neurologia. She is an editor for Frontiers of Neurology and The Journal of Headache and Pain. She is a member of the Clinical Trials Guidelines Committee of the International Headache Society. She has edited the Guidelines for the Diagnosis and Treatment of Headache of the Spanish Neurological Society. She does not own stocks from any pharmaceutical company., C. Lucas Conflict with: collaboration as an expert, investigator or coordinator of clinical trials with Novartis, Teva, Sanofi, Grünenthal, Eli Lilly, Biogen, and Ethypharm., C. Gaul Conflict with: received honoraria for consulting and lectures within the past 3 years from Allergan Pharma, Ratiopharm, Boehringer Ingelheim Pharma, Eli Lilly, Novartis, Desitin Arzneimittel, Cerbotec, Bayer Vital, Hormosan Pharma, electroCore, Grünenthal, Reckitt Benckiser, and Teva. He does not hold any stocks of pharmaceutical companies or medical device companies., D. Watson Conflict with: Received honoraria from Novartis, Teva and Allergan in the last 12 months for consultancy and educational work., P. Martelletti Conflict with: Section Editor, Medicine, Springer Nature Comprehensive Clinical Medicine; Editor-in-Chief, The Journal of Headache and Pain; Headache Books Series Editor, Springer; EU Expert, European Medicine Agency. Past-President of European Federation, Chairman of School of Advanced Studies of European Headache Federation. He does not hold any stocks of any pharmaceutical companies or medical device companies, S. Ritter Conflict with: Employees and stocks: Novartis, J. Snellman Conflict with: Employees and stocks: Novartis

MTV20-DP-046
HEALTHCARE RESOURCE UTILISATION AMONG MIGRAINE PATIENTS FROM THE EU5 WHO HAVE FAILED PREVIOUS PROPHYLACTIC TREATMENTS: FINDINGS FROM THE BECOME STUDY
Christian Lucas1*, Paolo Martelletti2, David P. Watson3, Patricia Pozo-Rosich4,5, Charly Gaul6, Shannon Ritter7 and Josefin Snellman8
1Pain Clinic, Service de Neurochirurgie, Hô pital Salengro, CHU de Lille, Lille Cedex, France
2Department of Clinical and Molecular Medicine, Sapienza University of Rome, Sant’Andrea Hospital, Rome, Italy
3Hamilton Medical Group, Aberdeen, United Kingdom
4Headache Unit, Neurology Department, Vall d’Hebron University Hospital
5Headache Research Group, VHIR, Universitat Autonoma de Barcelona, Barcelona, Spain
6Migraine and Headache Clinic Königstein, Königstein im Taunus, Germany
7Novartis Pharmaceuticals Corporation, New Jersey, United States
8Novartis Pharma AG, Basel, Switzerland
Introduction
Limited data on healthcare resource utilisation (HRU) in migraine patients with prior prophylactic treatment failure (PPTF) in the EU5 (France, Germany, Italy, Spain, and the United Kingdom [UK]) are available to date.
Objectives
To report HRU in migraine patients from the BECOME study visiting headache specialised centres in the EU5 and the impact of disease duration on HRU.
Methods
The BECOME study was conducted in two concurrent parts (Part 1, cumulative hospital data; Part 2, patient-level data). Part 2 examined HRU in patients with ≥1PPTF and ≥4monthly migraine days from the EU5.
Image:
Results
Data from 1013 patients were analysed for the EU5. In the three months prior to their visit to the study site, the proportion of patients visiting a neurologist (53.5%) (highest: UK [96.1%]; lowest: Italy [33.9%]) was higher than those visiting a general practitioner (GP) (38.0%) (highest: France [49.1%]; lowest: Italy [28.1%]). In the UK and Spain, the proportion of patients visiting neurologists was more than double of those visiting a GP (UK [96.1% vs 41.4%]; Spain [67.8% vs 30.8%], respectively). Although the proportion of patients visiting neurologists was higher, the mean number of visits/patient were higher for the GP vs neurologist across the EU5, regardless of the disease duration (
Conclusion
Analysis of HRU reveals differences in healthcare systems and migraine management in the EU5 regardless of disease duration. The proportion of patients visiting neurologists was overall high, especially in the UK. The mean visits/patient were higher to a GP vs a neurologist, indicating that patients probably visited GPs more often. Also notable are the high number of ED visits, the high proportions of patients requiring hospitalization for their migraine regardless of disease duration and the high proportion of patients visiting at least 3 or more different specialists. Differences among countries might reflect different health care systems. Overall, there is a need for improved treatment options for difficult-to-treat migraine patients with PPTF.
Disclosure of Interest
C. Lucas Conflict with: collaboration as an expert, investigator or coordinator of clinical trials with Novartis, Teva, Sanofi, Grünenthal, Eli Lilly, Biogen, and Ethypharm., P. Martelletti Conflict with: Section editor, Medicine, Springer Nature Comprehensive Clinical Medicine; editor-in-chief, The Journal of Headache and Pain; Headache Books Series editor, Springer; and EU expert, European Medicine Agency. Former president of the European Headache Federation and chairman of the School of Advanced Studies of the European Headache Federation. He does not hold stocks of any pharmaceutical or medical device companies., D. Watson Conflict with: received honoraria from Novartis, Teva, and Allergan in the last 12 months for consultancy and educational work., P. Pozo-Rosich Conflict with: received honoraria as a consultant and speaker during the last 5 years for Allergan, Almirall, Chiesi, Eli Lilly, Novartis, and Teva. Her research group has received research grants from Allergan and has received funding for clinical trials from Alder, Boehringer Ingelheim, MSD, electroCore, Eli Lilly, Janssen Cilag, and Novartis. She is a trustee member of the board of the International Headache Society and a member of the Council of the European Headache Federation. She is on the editorial board of Revista de Neurologia. She is an editor for Frontiers of Neurology and The Journal of Headache and Pain. She is a member of the Clinical Trials Guidelines Committee of the International Headache Society. She has edited the Guidelines for the Diagnosis and Treatment of Headache of the Spanish Neurological Society. She does not own stocks from any pharmaceutical company., C. Gaul Conflict with: received honoraria for consulting and lectures within the past 3 years from Allergan, Ratiopharm, Boehringer Ingelheim Pharma, Eli Lilly, Novartis Pharma, Desitin Arzneimittel, Cerbotec, Bayer Vital, Hormosan Pharma, electroCore, Grünenthal, Reckitt Benckiser, and Teva. He holds no stocks of pharmaceutical companies or medical device companies., S. Ritter Conflict with: employee of and owns stocks in Novartis, J. Snellman Conflict with: employee of and owns stocks in Novartis.

MTV20-DP-047
SUSTAINED EFFICACY AND SAFETY OF ERENUMAB IN EPISODIC MIGRAINE PATIENTS FAILING 2–4 PRIOR PREVENTIVE TREATMENTS: 2-YEAR INTERIM RESULTS OF THE LIBERTY OPEN-LABEL EXTENSION STUDY
Uwe Reuter1*, Peter J. Goadsby2, Michel Lanteri-Minet3,4, Tracy Stites5, Shihua Wen5, Nadia Tenenbaum5, Michel D. Ferrari6 and Shaloo Pandhi7
1Department of Neurology, Charité Universitätsmedizin Berlin, Berlin, Germany
2NIHR-Wellcome Trust, King’s Clinical Research Facility, London, United Kingdom
3Pain Department, CHU Nice
4Université Côte d’Azur, Nice, France
5Novartis Pharmaceutical Corporation, East Hanover, NJ, United States
6Department of Neurology, Leiden University Medical Center, Leiden, Netherlands
7Novartis Pharma AG, Basel, Switzerland
Introduction
The LIBERTY study (NCT03096834) demonstrated efficacy of erenumab 140 mg in episodic migraine (EM) patients with 2–4 prior preventive treatment failures.
Objectives
To assess the efficacy and safety of erenumab at Week 112 of the 3-year open-label treatment phase (OLTP) of the LIBERTY study (NCT03096834).
Methods
Patients completing the 12-week double-blind treatment phase (DBTP) of the LIBERTY study (N = 240) initially randomised to placebo and erenumab 140 mg (1:1) were enrolled into the OLTP to receive monthly erenumab 140 mg for 3 years. Outcomes measured included proportion of patients who achieved ≥50%/≥75%/100% reduction from the DBTP baseline in monthly migraine days (MMD), change from the DBTP baseline in MMD, Headache Impact Test total score (HIT-6), Migraine Physical Function Impact Diary (MPFID, Everyday Activities and Physical Impairment) scores and safety.
Image:
Results
Both groups of patients: on continuous erenumab and those who initiated erenumab in the OLTP, demonstrated improvement through 2 years of treatment (
Conclusion
Efficacy of erenumab was sustained over long-term treatment in EM patients with 2–4 PPTF both in patients continuously treated with erenumab and those initiating erenumab during the OLTP. Erenumab was well tolerated with no new safety signals.
Disclosure of Interest
U. Reuter Conflict with: received consulting fees, speaking/teaching fees, from Allergan, Amgen, Autonomic Technologies, CoLucid, ElectroCore, Eli Lilly, Medscape, Novartis, StreamMedUp, Teva and research grants from Allergan, Amgen, Autonomic Technologies, CoLucid, ElectroCore, Eli Lilly, Medscape, Novartis, StreamMedUp, Teva., P. J. Goadsby Conflict with: received personal fees from Amgen and Eli Lilly, Alder Biopharmaceuticals, Allergan, Autonomic Technologies, Clexio, ElectroCore, eNeura, Impel Neuropharma, Mundipharma, Novartis, Teva, Trigemina, MedicoLegal work, Journal Watch, Up-to-Date, Oxford University Press, Massachusetts Medical Society, and Wolters Kluwer., M. Lanteri-Minet Conflict with: received honoraria for advisory boards, speaker panels or investigation studies from Allergan, Amgen, Astellas, ATI, BMS, Boehringer Ingelhei, Boston Scientific, CoLucid, Convergence, Glaxo-SmithKline, Grünenthal, Eli Lilly, Medtronic, Menarini, MSD, Novartis, Pfizer, Reckitt Benckiser, Saint-Jude, Sanofi-Aventis, Teva, UCB, Zambon., T. Stites Conflict with: employees and own stocks in Novartis, S. Wen Conflict with: employees and own stocks in Novartis., N. Tenenbaum Conflict with: employees and own stocks in Novartis., M. D. Ferrari Conflict with: consultancy from Medtronic, ElectroCore, Amgen, Eli Lilly, Teva, and Novartis, and independent support from the European Community, NWO, NIH and the Dutch Heart Foundation and grants, trial support from Medtronic, ElectroCore, Amgen, Eli Lilly, Teva, and Novartis., S. Pandhi Conflict with: employees and own stocks in Novartis.

MTV20-DP-048
INTERIM BASELINE PATIENT CHARACTERISTICS FOR PARTICIPANTS OF THE PREVENTIVE TREATMENT OF MIGRAINE: OUTCOMES FOR PATIENTS IN REAL-WORLD HEALTHCARE SYSTEMS (TRIUMPH) STUDY
Miguel J. Lainez1*, Michel Lanteri-Minet2,3, Manjit Matharu4, Tobias Kurth5, Shivang Joshi6, Carlos Vallarino, Linda Wietecha, Chad Stroud and J Scott Andrews
1Hospital Clínico Universitario, Universidad Católica de Valencia, Valencia, Spain
2Pain Department CHU Nice and FHU InovPain Côte Azur University, Nice
3INSERM/UdA, U1107, Neuro-Dol, Auvergne University, Clermont-Ferrand, France
4UCL Institute of Neurology and National Hospital for Neurology and Neurosurgery, London, United Kingdom
5Institute of Public Health, Charité Universitätsmedizin Berlin, Berlin, Germany
6DENT Neurologic Institute, Amherst
7Eli Lilly and Company, Indianapolis, United States
Introduction
Migraine has a significant impact on a patient’s health-related quality of life, and effects on family, work, and social relationships. Current treatment guidelines are based on clinical trial data and vary among countries. Data from observational studies are needed to evaluate effectiveness of migraine treatments available to patients and inform optimal treatment strategies for patients receiving care.
Objectives
Describe baseline and clinical characteristics of patients with migraine in routine clinical care who are switching or initiating pharmacologic treatment for migraine prevention.
Methods
TRIUMPH is an ongoing, prospective, multicenter, international, 2-stage noninterventional observational study. Eligible patients are 18 years or older, have a diagnosis of migraine and are switching or initiating a new pharmacologic migraine preventive treatment. Stage 1 is comprised of a cross-sectional, single-day assessment of demographics, disease characteristics and baseline, and historical migraine treatment patterns. Stage 2 is a 24-month longitudinal follow-up assessment.
Results
Currently 57 patients are enrolled in the study and are treated with galcanezumab (N = 9), other calcitonin gene-related peptide monoclonal antibodies (N = 7, erenumab N = 3, fremanezumab N = 2, unspecified N = 2), and oral standard of care preventive treatment (N = 39) including anticonvulsants (N = 18), tricyclic-antidepressants (N = 18), calcium channel blockers (N = 1), angiotensin II receptor antagonist (N = 2), and other [locally approved treatment (N = 1) or not assigned (N = 1)]. The mean age of current participants is 44 years old, 86% are female and have an average of 12 monthly migraine headache days.
Conclusion
The study has enrolled approximately 2% of the target sample size of 2850 patients. Conclusions for Stage 1 will be available once this stage is completed.
Disclosure of Interest
M. Lainez Conflict with: Honoraria, consultation fees and research grants from: Allergan, Amgen, ATI, Bial, Boehringher, Chiesi, ElectroCore, Eli Lily, Medtronic, Novartis, Otsuka, Roche, Teva and UCB, M. Lanteri-Minet Conflict with: Personal fee for advisory boards, speaker panels or investigation studies from Allergan, Amgen, Astellas, ATI, BMS, Boehringer, Boston Scientific, CoLucid, Convergence, GlaxoSmithKline, Grunenthal, Eli Lilly, Lundbeck, Medtronic, MSD, Novartis, Pfizer, ReckittBenckiser, Saint-Jude, Sanofi-Aventis, Teva, UCB, and Zambon., M. Matharu Conflict with: Abbott, Medtronic and ElectroCore, Conflict with: Honoraria from Allergan, Novartis, Eli Lilly, TEVA, Autonomic Technologies Inc, ElectroCore and Medtronic., T. Kurth Conflict with: Honoraria from Eli Lilly, Newsenselab, Total S.A., and the BMJ., S. Joshi Conflict with: Advisory: Eli Lilly, Amgen/Novartis, Teva, Biohaven, Currax, Assertio, Abvie. Speaking: Eli Lilly, Amgen/Novartis, Teva, Biohaven, Abvie, C. Vallarino Conflict with: Full-time employee of Eli Lilly and Company, L. Wietecha Conflict with: Full-time employee of Eli Lilly and Company, C. Stroud Conflict with: Full-time employee of Eli Lilly and Company, J. S. Andrews Conflict with: Full-time employee of Eli Lilly and Company
MTV20-DP-049
SUSTAINED EFFICACY AND LONG-TERM SAFETY OF ERENUMAB IN PATIENTS WITH EPISODIC MIGRAINE: RESULTS OF A 5-YEAR, OPEN-LABEL EXTENSION STUDY
Messoud Ashina1*, Peter J. Goadsby2,3, Uwe Reuter4, Stephen Silberstein5, David W. Dodick6, Fei Xue7, Feng Zhang7, Sunfa Cheng7, Denise E. Chou7 and Gabriel Paiva da Silva Lima7
1Dept Neurology, Danish Headache Center, Rigshospitalet Glostrup, Faculty of Health and Medical Sciences, University of Copenhagen, Copenhagen, Denmark
2Department of Neurology, University of California, Los Angeles, Los Angeles, United States
3NIHR-Wellcome Trust King’s Clinical Research Facility, King’s College, NIHR-Wellcome Trust King’s Clinical Research Facility, King’s College, London, United Kingdom
4Dept Neurology, Charité Universitätsmedizin Berlin, Berlin, Germany
5Jefferson Headache Center, Thomas Jefferson University, Philadelphia
6Dept of Neurology, Mayo Clinic, Scottsdale
7Amgen Inc, Thousand Oaks, United States
Introduction
Migraine, a long-lasting disorder, sometimes requires preventive therapy for many years. Although erenumab has demonstrated significant reduction in migraine frequency and improved quality of life in studies lasting 3–12 months, little is known about long-term therapy.
Objectives
To evaluate the efficacy and safety of erenumab (erenumab-aooe in the US) in a 5-year open-label treatment period (OLTP) following a preceding 12-week double-blind treatment period (DBTP) in patients with episodic migraine (EM).
Methods
Patients with EM received open-label erenumab monthly, 70 mg initially and 140 mg after protocol amendment at ∼2 years. Descriptive summaries of efficacy assessments, including changes from double-blind baseline in monthly migraine days (MMD) and in acute migraine-specific medication days (AMSM), and adverse events (AEs), were based on observed data.
Results
Of 383 patients enrolled in the OLTP, 250 switched to 140 mg and 216 (56.4%) completed open-label treatment. Very few discontinuations were due to adverse events (5%) or lack of efficacy (3%).
Mean (SD) change in MMD from DBTP baseline of 8.7 (2.7) was –5.0 (4.0) at week 64 and –5.3 (3.9) at week 268 for an average reduction of 61.7% at end of the OLTP. The proportion of patients with ≥50%/≥75%/100% reduction in MMD were 65.7%/42.2%/25.2% at week 64 and 69.3%/48.2%/35.8% over the last 4-week period at week 268. Among patients using AMSM at baseline (6.2 [2.8] treatment days/month), mean change in AMSM was –3.5 (3.3) days at week 64 and –4.4 (3.3) days at week 268.
Median (Q1, Q3) erenumab exposure for patients receiving ≥1 dose of open-label erenumab was 251 (68, 256) weeks. Exposure-adjusted AE incidence rates were 123.0/100-patient-years (100PY) compared with 326.2/100PY for the erenumab 70-mg arm in the DBTP. Nasopharyngitis (10.6/100PY vs 23.3/100PY in 70 mg DBTP), upper respiratory tract infection (6.7/100PY vs 11.4/100PY in 70 mg DBTP), and influenza (4.6/100PY vs 3.8/100PY in 70 mg DBTP) were the most frequent AEs. SAEs were reported by 49 patients; an incidence rate of 3.8/100PY (vs 3.9/100PY in 70 mg DBTP). Most SAEs were single occurrence. Two fatal adverse events were reported: previously reported arteriosclerosis (70 mg) and death of unknown cause, autopsy not performed (140 mg). Safety signals were consistent with those observed in the DBTP without increases in AEs/SAEs over 5 years of OLE vs DBTP.
Conclusion
Long-term erenumab treatment up to 5 years demonstrated sustained reductions in migraine frequency without new safety signals.
Disclosure of Interest
M. Ashina Conflict with: personal fees from Alder BioPharmaceuticals, Allergan, Amgen, Eli Lilly, Lundbeck, Novartis, and Teva. Dr. Messoud Ashina currently is a principal investigator on clinical trials for Allergan, Amgen, Eli Lilly, Lundbeck and Novartis. Dr. Messoud Ashina has no ownership interest and does not own stocks of any pharmaceutical company. Dr. Messoud Ashina also serves as an Associate Editor of Cephalalgia, Associate Editor of Headache, and Associate Editor of the Journal of Headache and Pain. Dr. Messoud Ashina reports research grants from Lundbeck Foundation, Novo Nordisk Foundation, and Novartis, P. J. Goadsby Conflict with: consulting fee, speaking/teaching fee, and/or research grants: Alder Biopharmaceuticals, Allergan, Amgen, Autonomic Technologies, Celgene, Clexio, electroCore, Inc., Eli Lilly, eNeura, Inc., Epalex, Impel, Mundipharma, Journal Watch, Massachusetts Medical Society, Medico-Legal Journal, Novartis, Oxford University Press, Pfizer, Teva Pharmaceuticals, Trigemina, Inc., UpToDate, WL Gore, Wolters Kluwer, U. Reuter Conflict with: consulting fee, speaking/teaching fee, and/or research grants from Allergan, Amgen, Autonomic Technologies, CoLucid, ElectroCore, Novartis, Pharm Allergan, Eli Lilly, and Teva Pharmaceuticals, S. Silberstein Conflict with: consultant and/or advisory panel member for and/or honoraria from Alder, Allergan, Amgen, Avanir, Dr. Reddy’s, eNeura, electroCore Medical, Medscape, Medtronic, Mitsubishi Tanabe Pharma America, NINDS, Supernus, Trigemina, and Teva, D. Dodick Conflict with: Consulting: AEON, Amgen, Clexio, Cerecin, Allergan, Alder, Biohaven, Linpharma, Promius, Eli Lilly, eNeura, Novartis, Impel, Theranica, WL Gore, Nocira, XoC, Zosano, Upjohn (Division of Pfizer), Pieris, Revance, Equinox. Honoraria: CME Outfitters, Curry Rockefeller Group, DeepBench, Global Access Meetings, KLJ Associates, Majallin LLC, Medlogix Communications, Miller Medical Communications, Southern Headache Society (MAHEC), WebMD Health/Medscape, Wolters Kluwer, Oxford University Press, Cambridge University Press. Research Support: Department of Defense, National Institutes of Health, Henry Jackson Foundation, Sperling Foundation, American Migraine Foundation, Patient Centered Outcomes Research Institute (PCORI). Stock Options/Shareholder/Patents/Board of Directors: Aural analytics (options), ExSano (options), Palion (options), Healint (Options), Theranica (Options), Second Opinion/Mobile Health (Options), Epien (Options/Board), Nocira (options), Ontologics (Options/Board), King-Devick Technologies (Options/Board), Precon Health (Options/Board). Patent 17189376.1–1466:vTitle: Botulinum Toxin Dosage Regimen for Chronic Migraine Prophylaxis, F. Xue Conflict with: employee of and stockholder in Amgen Inc, F. Zhang Conflict with: employee of and stockholder in Amgen Inc, S. Cheng Conflict with: employee of and stockholder in Amgen Inc, D. Chou Conflict with: employee of and stockholder in Amgen Inc, G. Paiva da Silva Lima Conflict with: employee of and stockholder in Amgen Inc
MTV20-DP-050
ERENUMAB IN EPISODIC AND CHRONIC MIGRAINE: POST HOC ANALYSES OF EFFICACY DATA BY HIGH VERSUS LOW-FREQUENCY MIGRAINE IN PHASE 2 AND PHASE 3 STUDIES
Uwe Reuter1*, David W. Dodick2, Stewart J. Tepper3, Shannon Ritter4, Gabriel P. D. S. Lima5, Denise E. Chou5, Ronald Zielman6 and Messoud Ashina7
1Department of Neurology, Charité Universitätsmedizin Berlin, Berlin, Germany
2Department of Neurology, Mayo Clinic, Scottsdale, AZ
3Geisel School of Medicine at Dartmouth, Hanover, NH
4Novartis Pharmaceutical Corporation, East Hanover, NJ
5Amgen Inc., Thousand Oaks, CA, United States
6Novartis Pharma B.V., Amsterdam, Netherlands
7Department of Neurology, Danish Headache Center, Rigshospitalet Glostrup, Faculty of Health and Medical Sciences, University of Copenhagen, Copenhagen, Denmark
Introduction
Patients with high-frequency episodic migraine (HFEM) may have a similar disease burden as patients with chronic migraine (CM).
Objectives
We evaluated the monthly migraine days (MMD) in subgroups of patients on erenumab defined by their baseline MMD from Phase 3 STRIVE EM (NCT02456740) and Phase 2 CM (NCT02066415) studies.
Methods
In this post hoc analysis, subgroups were defined by baseline MMD and included low frequency EM (LFEM; 4–7 MMD), HFEM (8–14 MMD), low frequency CM (LFCM; ≥15 monthly headache days [MHD], of which 8–14 are MMD) and high frequency CM (HFCM; ≥15 MHD, of which >15 are MMD). Change from baseline in MMD and 50% responder rates in MMD reduction were assessed at Months 4–6 for the STRIVE study, and at Weeks 9–12 for the CM study.
Image:
Results
Erenumab at both doses, significantly reduced MMD across all subgroups compared with placebo in the double blind treatment phase of both studies (Table). The ≥50% response rate was significantly higher with erenumab when compared with placebo in all subgroups; the exception was LFCM subgroup on erenumab 70 mg which had a numerically higher number of responders compared to placebo (Table). Erenumab 140 mg was numerically superior to 70 mg in the LFEM, HFEM, and HFCM subgroups.
Conclusion
Erenumab is effective across the entire migraine spectrum (LFEM, HFEM, LFCM and HFCM). The 140 mg dose showed numerically higher reduction in MMD than 70 mg in all subgroups except LFCM, where 70 mg showed slightly higher reduction than 140 mg. However, 140 mg still showed higher 50% response rate than 70 mg in the LFCM subgroup.
Disclosure of Interest
U. Reuter Conflict with: received consulting fees, speaking/teaching fees, and/or research grants from Allergan, Amgen, Autonomic Technologies, CoLucid, ElectroCore, Eli Lilly, Medscape, Novartis, StreamMedUp and Teva Pharmaceuticals, D. Dodick Conflict with: within the last 12 months, consulting: AEON, Amgen, Clexio, Cerecin, Allergan, Alder, Biohaven, Linpharma, Lundbeck, Promius, Eli Lilly, eNeura, Novartis, Impel, Theranica, WL Gore, Nocira, XoC, Zosano, Upjohn (Division of Pfizer), Pieris, Revance, Equinox. Honoraria: CME Outfitters, Curry Rockefeller Group, DeepBench, Global Access Meetings, KLJ Associates, Majallin LLC, Medlogix Communications, Miller Medical Communications, Southern Headache Society (MAHEC), WebMD Health/Medscape, Wolters Kluwer, Oxford University Press, Cambridge University Press. Research Support: Department of Defense, National Institutes of Health, Henry Jackson Foundation, Sperling Foundation, American Migraine Foundation, Patient Centered Outcomes Research Institute (PCORI). Stock Options/Shareholder/Patents/Board of Directors: Aural analytics (options), ExSano (options), Palion (options), Healint (Options), Theranica (Options), Second Opinion/Mobile Health (Options), Epien (Options/Board), Nocira (options), Ontologics (Options/Board), King-Devick Technologies (Options/Board), Precon Health (Options/Board). Patent 17189376.1–1466:vTitle: Botulinum Toxin Dosage Regimen for Chronic Migraine Prophylaxis., S. Tepper Conflict with: Grants for research (no personal compensation): Alder, Allergan, Amgen, Dr. Reddy’s, ElectroCore, Eli Lilly, eNeura, Neurolief, Novartis, Scion Neurostim, Teva, Zosano; Consultant and/or Advisory Boards (honoraria): Acorda, Alder, Alexsa, Align Strategies, Allergan, Alphasights, Amgen, Aperture Venture Partners, Aralez Pharmaceuticals Canada, Axsome Therapeutics, Becker Pharmaceutical Consulting, BioDelivery Sciences International, Biohaven, Charleston Labs, Decision Resources, DeepBench, ElectroCore, Eli Lilly, eNeura, Equinox, ExpertConnect, GLG, GSK, Guidepoint Global, Healthcare Consultancy Group, Impel, M3 Global Research, Magellan Rx Management, Marcia Berenson Connected Research and Consulting, Medicxi, Navigant Consulting, Neurolief, Nordic BioTech, Novartis, Pfizer, Pulmatrix, Reckner Healthcare, Relevale, Revance, Satsuma, Scion Neurostim, Slingshot Insights, Sorrento, Spherix Global Insights, Sudler and Hennessey, Synapse Medical Communications, Teva, Theranica, Thought Leader Select, Trinity Partners, XOC, Zosano; Salary: Dartmouth-Hitchcock Medical Center, American Headache Society; Stock options: Percept; CME honoraria: American Academy of Neurology, American Headache Society, Cleveland Clinic Foundation, Diamond Headache Clinic, Elsevier, Forefront Collaborative, Hamilton General Hospital, Ontario, Canada, Headache Cooperative of New England, Henry Ford Hospital, Detroit, Inova, Medical Learning Institute Peerview, Miller Medical Communications, North American Center for CME, Physicians’ Education Resource, Rockpointe, WebMD/Medscape., S. Ritter Conflict with: employee of and own stocks in Novartis, G. Lima Conflict with: employee of and own stocks in Amgen, D. Chou Conflict with: employee of and owns stocks in Amgen, R. Zielman Conflict with: employee of Novartis, M. Ashina Conflict with: Consultant or scientific advisor for Allergan, Amgen, Alder, ATI, Novartis, and Eli Lilly; primary investigator for Amgen, and GM-11 gamma-Core-R trials; grants from Lundbeck Foundation, Research Foundation of the Capital Region of Copenhagen, Danish Council for Independent Research-Medical Sciences and Novo Nordisk Foundation.
MTV20-DP-051
ERENUMAB IS COST-EFFECTIVE FOR THE PREVENTION OF EPISODIC AND CHRONIC MIGRAINE AMONG PATIENTS WITH PRIOR TREATMENT FAILURES IN SWEDEN
Ronan Mahon1*, Andrea Lang2, Jasper Huels3, Philip Cooney1, Andrii Danyliv1, Umakanth Vudumula4, Sreelatha Vadapalle4, Pamela Vo3 and Peter J. Goadsby5
1NBS CONEXTS, Novartis Ireland Limited, Dublin, Ireland
2Market Access, Novartis Sverige AB, Stockholm, Sweden
3Global Patient Access, Novartis Pharma AG, Basel, Switzerland
4NBS CONEXTS, Novartis Healthcare Private Limited, Hyderabad, India
5NIHR-Wellcome Trust King’s Clinical Research Facility, SLaM Biomedical Research Centre, King’s College London, London, United Kingdom
Introduction
Migraine is a highly debilitating neurological disorder affecting approximately 13% of people in Sweden. Erenumab is a fully human monoclonal antibody targeting the canonical calcitonin gene-related peptide receptor and is approved for the prevention of migraine.
Objectives
The aim of this study was to evaluate the cost-effectiveness of erenumab versus best supportive care only for the preventive treatment of migraine in Sweden.
Methods
A hybrid decision-tree plus Markov model was developed to evaluate the cost-effectiveness of erenumab as a migraine treatment compared to placebo (both in conjunction with best supportive care) for patients experiencing at least 4 monthly migraine days (MMDs) who have failed at least two prior preventive treatments. Analysis was performed on a combined episodic (25%) and chronic (75%) migraine population from the Swedish societal perspective with a 10-year time horizon. The model was designed based on inputs from clinical expert consultations, published literature, and previous modelling approaches for preventive treatments of migraine. Clinical efficacy data were based on results from the four randomized controlled trials of erenumab against placebo (the 295 study, STRIVE, ARISE and LIBERTY trials). Utility inputs were derived by mapping Migraine Specific Quality-of-life questionnaire v2.1 scores to EQ-5D. Costs and outcomes were discounted at 3%. The primary outcomes were costs, migraine days and quality-adjusted life years (QALY). An incremental cost-effectiveness ratio (ICER) was estimated as the cost per QALY gained. The cost per migraine day avoided was also estimated, as well as disaggregated direct and indirect costs.
Results
In the base-case deterministic analysis, against placebo over a 10-year time horizon, erenumab treatment resulted in ICER of SEK 34,696 per QALY gained, with an incremental cost of SEK 10,690 per patient, incremental QALY of 0.3081, and a reduction of 249 migraine days. The cost per migraine day avoided with erenumab treatment was SEK 43. Results indicated that indirect costs and resource utilisation costs were much lower for patients on erenumab in comparison to placebo. The total net monetary benefit to society of erenunab treatment was estimated to be SEK 81,739 per patient assuming a willingness to pay threshold of SEK 300,000/QALY. Under a lifetime horizon erenumab dominates placebo (is both cost-saving more effective).
Conclusion
From a Swedish societal perspective, erenumab is cost-effective in migraine patients who have failed two or more prior preventive treatments.
Disclosure of Interest
R. Mahon Conflict with: Ronan Mahon is an employee of Novartis, A. Lang Conflict with: Andrea Lang is an employee of Novartis, J. Huels Conflict with: Jasper Huels is an employee of Novartis, P. Cooney Conflict with: Philip Cooney is an employee of Novartis, A. Danyliv Conflict with: Andrii Danyliv is an employee of Novartis, U. Vudumula Conflict with: Umakanth Vudumula is an employee of Novartis, S. Vadapalle Conflict with: Sreelatha Vadapalle is an employee of Novartis, P. Vo Conflict with: Pamela Vo is an employee of Novartis, P. J. Goadsby Conflict with: Personal fees from Alder Biopharmaceuticals, personal fees from Allergan, grants and personal fees from Amgen, grant from Celgene, personal fees from Autonomic Technologies Inc., personal fees from Biohaven Pharmaceuticals Inc., personal fees from Clexio, grants and personal fees from Eli Lilly and Company, personal fees from Electrocore LLC, personal fees from eNeura Inc, personal fees from Impel Neuropharma, personal fees from MundiPharma, personal fees from Novartis, personal fees from Teva Pharmaceuticals, other from Trigemina, personal fees from WL Gore, outside the submitted work. In addition, PG has a patent Magnetic stimulation for headache licensed to eNeura without fee and Fees for publishing from Oxford University Press, Massachusetts Medical Society, Wolters Kluwer and for medico-legal work.
MTV20-DP-052
PREGNANCY OUTCOMES FOLLOWING EXPOSURE TO ONABOTULINUMTOXINA UPDATE: 29 YEARS OF SAFETY OBSERVATION
Mitchell F. Brin1, Russell S. Kirby2, Anne Slavotinek3, Aubrey Adams4, Lori Parker5, Ahunna Ukah5, Lavinia Radulian6, Larisa Yedigarova4* and Irina Yushmanova5
1BOTOX® & Neurotoxins, Allergan, an AbbVie company, Irvine
2College of Public Health, University of South Florida, Tampa
3Division of Medical Genetics, Department of Pediatrics, University of California, San Francisco, San Francisco
4Medical Affairs Migraine
5Global Patient Safety & Epidemiology, Allergan, an AbbVie company, Irvine, United States
6Global Patient Safety & Epidemiology, Allergan, an AbbVie company, Bucharest, Romania
Introduction
A previous publication of pregnancy outcomes in onabotulinumtoxinA-exposed mothers (Brin et al. Pharmacoepidemiol Drug Saf. 2016; 25:179-187) demonstrated that the prevalence of fetal defects (2.7%; 3/110, 95% CI: 0.6–8.0%) was comparable with background rates in the general population.
Objectives
The present analysis evaluated pregnancy outcomes following onabotulinumtoxinA exposure to provide a cumulative 29-year update.
Methods
The Allergan Global Safety Database contains reports of onabotulinumtoxinA administration before/during pregnancy, including prospective (reported before outcome known) and retrospective (outcome known when reported) cases. The database was searched (January 1, 1990 to December 31, 2018) for eligible cases, where treatment (across various indications, including migraine) occurred during pregnancy or ≤3 months prior to conception. A conservative approach was taken to minimize reporting bias, where prevalence rates for overall and major birth defects were estimated from prospective cases of live births only.
Results
Of 913 pregnancies, 397 (43.5%) were eligible with known outcomes. Maternal age was known in 215 cases, 45.6% were ≥35 years. Indication was known in 340 cases, with the most frequent indications being cosmetic (35.3%), migraine/headache (30.3%), and movement disorders (12.1%). Timing of exposure was known in 318 cases: 16.0% were prior to conception, 78.6% during the first trimester, 3.8% during the second trimester, and 1.6% during the third trimester. OnabotulinumtoxinA dose information was known in 242 cases: 32.2% were < 50 U, 11.2% 50 U to < 100 U, 40.1% 100 U to < 200 U, and 16.5% ≥200 U. Of 195 prospective pregnancy cases with 197 fetuses, there were 152 (77.2%) live births and 45 (22.8%) fetal losses (32 spontaneous abortions, 13 elective abortions). Of the 152 live births,148 (97.4%) had normal outcomes and 4 had abnormal outcomes. Among the 4 abnormal birth outcomes, there were 1 major birth defect, 2 minor fetal defects, and 1 birth complication. The prevalence rate for overall fetal defects was 2.6% (4/152, 95% CI: 1.0–6.6%) and 0.7% (1/152, 95% CI: 0.1–3.6%) for major fetal defects (3–6% in general population).
Conclusion
A 29-year retrospective analysis of safety data in onabotulinumtoxinA-exposed mothers, which included over 30% of women with migraine, demonstrated that prevalence rates of abnormal birth outcomes were within/below those reported in the general population, with no new safety signals identified.
Disclosure of Interest
M. Brin Conflict with: Employee of AbbVie and receives salary and stock/stock options as compensation., R. Kirby Conflict with: Received grant funding from the Centers for Disease Control and Prevention, Florida Department of Health, and Maternal and Child Health Bureau/Health Resources and Services Administration., Conflict with: Employee of University of South Florida, A. Slavotinek Conflict with: None declared as relevant to this publication., A. Adams Conflict with: Employee of AbbVie and receives salary and stock/stock options as compensation., L. Parker Conflict with: Employee of AbbVie and receives salary as compensation., A. Ukah Conflict with: Employee of AbbVie and receives salary and stock/stock options as compensation., L. Radulian Conflict with: Employee of AbbVie and receives salary and stock/stock options as compensation., L. Yedigarova Conflict with: Employee of AbbVie and receives salary and stock/stock options as compensation., I. Yushmanova Conflict with: Employee of AbbVie and receives salary and stock/stock options as compensation
MTV20-DP-053
CHARACTERIZATION OF INJECTION-SITE REACTIONS FROM THE CONQUER STUDY OF GALCANEZUMAB IN PATIENTS WITH TREATMENT-RESISTANT MIGRAINE
Virginia L. Stauffer1*, Michel Lanteri-Minet2, David García-Azorín3, Phebe Kemmer1, Angelo Camporeale4 and Richard Wenzel1
1Eli Lilly and Company, Indianapolis, United States
2Pain Department CHU Nice and FHU InovPain Côte Azur University, Nice, France; INSERM/UdA, U1107, Neuro-Dol, Auvergne University, Nice, France
3Headache Unit, Department of Neurology, Hospital Clinico Universitario, Valladolid, Spain
4Eli Lilly Italia, Sesto Fiorentino, Italy
Introduction
In the Phase 3 galcanezumab studies, injection-site reactions (ISRs) were the most frequently reported treatment emergent adverse events (TEAEs).
Objectives
Characterize ISRs in galcanezumab-treated patients 18–75 years with episodic or chronic migraine, from the CONQUER study.
Methods
The galcanezumab-treated patients received a 240mg loading dose of galcanezumab followed by 120mg per month. Patients were treated for up to 6 months with galcanezumab (3 months double-blind/3 months open-label). ISRs were defined using the Medical Dictionary for Regulatory Activities high level term of ‘injection site reactions.’ Each ISR was characterized based on a series of follow-up questions such as timing of the event, when signs and symptoms resolved, location, size and site of the erythema, induration, pain, pruritus, and swelling or edema. Staff were encouraged to offer comfort measures such as cold compress, ice pack, or topical anesthetic cream to the injection site prior to or after the injection.
Results
Demographics included mean age of 45.8 years; 85.9% were female and 81.7% were white. At baseline, patients averaged 13.2 migraine headache days per month, and failed an average of 3.3 migraine preventive treatments over the past 10 years. Among galcanezumabtreated patients (N = 457), 10.9% had ≥1 TEAE related to injection sites. ISRs occurring in >1.0% of patients were injection site pain (4.4%), injection site erythema (4.2%), injection site pruritis (2.0%), injection site reaction (unspecified) (1.8%), and injection site induration (1.1%). There were no serious adverse events (SAEs) related to injection sites. One patient discontinued due to an injection site erythema TEAE. A severe TEAE of injection site pain as assessed by the injection site follow-up form was reported by 1 patient. Most TEAEs related to injection sites occurred on the same day as the injection, and most TEAEs of injection site pain occurred during injection administration. Comfort measures were used before and after 1.8% (ice pack [1.7%], cold compress [0.1%]) and 2.4% (ice pack [2.3%], cold compress [0.2%]) of injections, respectively.
Conclusion
ISRs may occur with galcanezumab. Pain and erythema were the most common injection site events, and 1 patient discontinued due to injection site erythema. No SAEs related to injection sites were reported. Five percent of patients used comfort measures (ice pack most frequently).
Disclosure of Interest
V. Stauffer Conflict with: Employee of Eli Lilly and Company, M. Lanteri-Minet Conflict with: Personal fee for advisory boards, speaker panels or investigation studies, D. García-Azorín: None Declared, P. Kemmer Conflict with: Employee of Eli Lilly and Company, A. Camporeale Conflict with: Employee of Eli Lilly and Company, R. Wenzel Conflict with: Employee of Eli Lilly and Company
MTV20-DP-054
UNMET NEEDS IN MIGRAINE PREVENTION: AN INDIAN PERSPECTIVE
Sneha Thakur* and Joy DesaiPankaj Gupta1
1Novartis, Mumbai, India
Introduction
According to Global Burden of Disease study 2016, migraine is the second leading cause of years lived with disability (YLD), responsible for 5.6% of all YLDs in the world in the age group 15–49 years. [1] It is important to note that these are the productive years in a life of an individual and the impact of migraine-attributed disability during these years is amplified. [1] In spite of the high prevalence and burden migraine is under-recognized and undertreated
Objectives
This review aims to highlight the burden of unmet needs in migraine prevention from an Indian perspective.
Methods
Literature searches were performed using PubMed and Google Scholar. Articles published in ’English’ from the period 2002 to Dec 2019 were selected that also included keywords like burden of migraine, migraine prophylaxis, chronic migraine, episodic migraine, diagnosis, barriers, medication overuse headache, topiramate, propranolol, amitriptyline, patient education, trigger factors, anti-CGRPS, gepants.
Image:

Results
The results of our search highlighted that one of the reasons for inadequate migraine management and prevention is the delay in diagnosis due to multiple visits to GPs/CPs before approaching neurologists or headache specialists.2 An important reason for this is that in a vast geography like India, there is a disproportionate distribution of neurologists which mandates consulting GPs/CPs. Several drug classes like beta-blockers, triptans, anti-epileptics, etc are available for migraine prophylaxis; however, their efficacy is limited which leads to poor patient adherence.3-5 Our literature search also highlighted that patients lack sufficient awareness on trigger factors and lifestyle modification to control migraine attacks.6
Conclusion
A faster and accurate diagnosis would lead to timely management and prevention of future attacks. Target-specific therapies with established efficacy and tolerability should be used in combination with patient education and awareness measures.
Disclosure of Interest
None Declared
References
Migraine – preventive therapy
MTV20-DP-055
BASELINE DATA OF A REAL-WORLD-EVIDENCE-STUDY CHARACTERIZING PRESCRIPTION PATTERNS IN EPISODIC AND CHRONIC MIGRAINE PATIENTS STARTING TREATMENT WITH ERENUMAB IN GERMANY (SPECTRE)
Charly Gaul1*, Mirja Koch2 and Caroline Baufeld2
1Migraine and Headache Clinic Königstein, Königstein
2Novartis Pharma GmbH, Nürnberg, Germany
Introduction
Collection of information on monoclonal antibodies as a prophylactic treatment in the clinical routine of migraine specialists outside randomised controlled trials is an important issue. Erenumab, a Calcitonin Gene-Related Peptide (CGRP) – receptor antagonist, was approved with two monthly dosages: 70 mg and 140 mg.
Objectives
The aim of the SPECTRE study is to understand the choice of the starting dose as well as dose switching based on migraine characteristics and comorbidities.
Methods
This is an observational, non-interventional, multicenter, open label, single arm study comprising migraine patients receiving erenumab treatment. The study is conducted at 150 centers in Germany and aims to enroll 1960 adult migraine patients. Patients either can be new on the treatment or have started treatment recently, but not more than 3 months before entering the study. Apart from a headache diary, the patient-reported-outcome questionnaires HIT-6 and TSQM are used to assess the efficacy of erenumab and the satisfaction of the patients with the drug.
Results
The results of an early interim analysis with 111 erenumab patients are presented here. Ninety-nine (89%) were female with a median age of 43.8 (±12.0) years and a migraine history of 24.9 (±13.8) years. About two thirds of the patients (n = 69, 62.2%) were diagnosed with chronic migraine. 70.3% (n = 78) of patients had at least one comorbidity. The most frequent comorbidities were psychiatric disorders (n = 34, 30.6%), endocrine disorders and musculoskeletal and connective tissue disorders (for both: n = 23, 20.7%).
Further patients’ baseline migraine characteristics as well as the percentage of patients on each starting dose of erenumab stratified by the major reasons for prescription and comorbidities are discussed.
Conclusion
The SPECTRE study will give valuable insights into the clinical routine of erenumab prescriptions in Germany. Characterization of the prescription pattern and analysis of the respective therapy response will possibly allow for development of individual treatment strategies for each patient.
Disclosure of Interest
C. Gaul Conflict with: honoraria for consulting and lectures within the past three years from Allergan Pharma, Bayer vital, Boehringer Ingelheim Pharma, Cerbotec, Desitin Arzneimittel, electroCore, Grünenthal, Hormosan Pharma, Lilly Germany, Novartis Pharma, Ratiopharm, Reckitt Benckiser, Sanofi Aventis and TEVA., M. Koch Conflict with: employee of Novartis Pharma, C. Baufeld Conflict with: employee of Novartis Pharma
MTV20-DP-056
EFFICACY AND SAFETY OF ERENUMAB IN PATIENTS WITH EPISODIC MIGRAINE FROM THE EMPOWER STUDY
Shuu-Jiun Wang1*, Artemio Roxas Jr2, Bibiana Saravia3, Byung-Kun Kim4, Debashish Chowdhury5, Naji Riachi6, Mei-Ling Sharon Tai7, Surat Tanprawate8, Tai Ngọc Tran9, Zhao Yi Jing10, Daniel Mikol, Shaloo Pandhi12, Shihua Wen13, Subhayan Mondal14, Nadia Tenenbaum13 and Peggy Hours-Zesiger12
1Taipei Veterans General Hospital, Taipei, Taiwan, Province of China
2The Medical City, Pasig, Philippines
3Mautalen Salud e Investigación, Buenos Aires, Argentina
4Nowon Eulji Medical Center, Eulji University, Seoul, Korea, Republic Of
5Govind Ballabh Pant Institute of Postgraduate Medical Education and Research, New Delhi, India
6Lebanese American University Medical Center, Rizk Hospital, Beirut, Lebanon
7University of Malaya, Kuala Lumpur, Malaysia
8Faculty of Medicine, Chiang Mai University, Chiang Mai, Thailand
9University Medical Center, University of Medicine and Pharmacy, Ho Chi Minh City, Viet Nam
10National Neuroscience Institute, Singapore General Hospital Campus, Singapore, Singapore
11Amgen Inc., Thousand Oaks, CA, United States
12Novartis Pharma AG, Basel, Switzerland
13Novartis Pharmaceuticals Corporation, East Hanover, NJ, United States
14Novartis Healthcare Pvt. Ltd., Hyderabad, India
Introduction
EMPOwER study enrolled patients from regions not adequately represented in the Phase 2/3 trials of erenumab
Objectives
EMPOwER (NCT03333109), a 12-week, double-blind, randomised study, evaluated efficacy and safety of erenumab (70 mg and 140 mg) in adult patients with episodic migraine (EM) from Asia, the Middle East, and Latin America.
Methods
Patients (N = 900) were randomized (3:3:2) to receive monthly doses of placebo, erenumab 70 mg or 140 mg at Weeks 4, 8, and 12. The primary endpoint was the change from baseline in monthly migraine days (MMD). Secondary endpoints assessed achievement of ≥50% reduction in MMD, change in headache impact test (HIT-6™), change in monthly acute migraine-specific medication treatment days (MSMD), and safety/tolerability. These outcome measures were assessed over the last four weeks (Weeks 9–12) of the double-blind treatment phase.
Image:
Results
At baseline, mean (SD) age was 37.5(9.9) years, 81.9% were women; mean MMD was 8.2(2.81). Change in MMD from baseline at Week 12 was −3.1 with placebo, −4.2 and −4.8 for erenumab 70 mg and 140 mg respectively, and achieved statistical significance vs placebo (p: 0.002 [70 mg] and < 0.001 [140 mg]) (Table). Proportion of patients achieving ≥50% reduction in MMD at Week 12 was significantly higher in erenumab 70 mg and 140 mg vs placebo (53% and 64% vs 45%; p: 0.007 [70 mg] and < 0.001 [140 mg]). Change in HIT-6™ from baseline at Week 12 was −6.6 with placebo, −8.4 and −9.3 with erenumab 70 mg and 140 mg, respectively, reaching statistical significance vs placebo (p: 0.004 [70 mg] and < 0.001 [140 mg]). At Week 12, change in MSMD from baseline for patients with at least one MSM at baseline was −0.5 with placebo, −1.8 and −2.4 for erenumab 70 mg and 140 mg, respectively, reaching statistical significant vs placebo (both p < 0.001). In general, safety and tolerability profiles of erenumab 70 mg or 140 mg were comparable with placebo, and there were no newly emergent safety signals.
Conclusion
The EMPOwER study confirms the efficacy and safety of erenumab 70 mg and 140 mg in adult patients with EM from the regions of Asia, the Middle East, and Latin America.
Disclosure of Interest
S.-J. Wang Conflict with: Received research grants from the Taiwan Minister of Technology and Science, Brain Research Center, National Yang-Ming University from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan, Taipei Veterans General Hospital, and Taiwan Headache Society, Conflict with: Served on the advisory boards of Eli Lilly and Taiwan Novartis. He has received honoraria as a moderator from Allergan, Pfizer, Eli Lilly, Bayer, and Eisai, A. Roxas Jr Conflict with: Part of the advisory board and speaker of Novartis for erenumab, and involved in Novartis erenumab trials for episodic migraine (EMPOwER) and chronic migraine (DRAGON), B. Saravia: None Declared, B.-K. Kim Conflict with: Consulting fees and honoraria from Center for New Health Technology Assessment, National Pension Service, Eli Lilly, Allergan, Teva, Lundbeck, Sanofi Korea, SK Pharm, Yuyu Pharma, and Ildong Pharm, D. Chowdhury Conflict with: Funding received from Novartis for the conduct of the study, N. Riachi Conflict with: Funding received from Novartis for the conduct of the study, M.-L. S. Tai Conflict with: Funding received from Novartis for the conduct of the study, S. Tanprawate Conflict with: Received research grants from the Faculty of Medicine, Chiang Mai University, and National Innovation Agency of Thailand., Conflict with: Received honoraria as a lecturer from Allergan, Pfizer, Eli Lilly, Novartis and Eisai., T. N. Tran Conflict with: Received consultation and/or honoraria/lecture fees from Abbott, Boehringer-Ingelheim, Ipsen Pharmaceuticals, Medtronic, and Novartis, and funding from Novartis for the conduct of this study, Z. Y. Jing: None Declared, D. Mikol Conflict with: Employee of and owns stock in Amgen, S. Pandhi Conflict with: Employee of Novartis and may own Novartis stocks, S. Wen Conflict with: Employee of Novartis and may own Novartis stocks, S. Mondal Conflict with: Employee of Novartis and may own Novartis stocks, N. Tenenbaum Conflict with: Employee of Novartis and may own Novartis stocks, P. Hours-Zesiger Conflict with: Employee of Novartis and may own Novartis stocks

MTV20-DP-057
BASELINE DEMOGRAPHICS AND DISEASE CHARACTERISTICS FROM THE PHASE 3 EMPOWER STUDY IN PATIENTS WITH EPISODIC MIGRAINE IN ASIA, MIDDLE EAST AND LATIN AMERICA
Shuu-Jiun Wang1*, Artemio Roxas Jr2, Bibiana Saravia3, Byung-Kun Kim4, Debashish Chowdhury5, Naji Riachi6, Mei-Ling Sharon Tai7, Surat Tanprawate8, Tai Ngoc Tran9, Zhao Yi Jing10, Daniel Mikol11, Shaloo Pandhi12, Shihua Wen13, Subhayan Mondal14, Nadia Tenenbaum13 and Peggy Hours-Zesiger12
1Taipei Veterans General Hospital, Taipei, Taiwan, Province of China
2The Medical City, Pasig, Philippines
3Mautalen Salud e Investigación, Buenos Aires, Argentina
4Nowon Eulji Medical Center, Eulji University, Seoul, Korea, Republic Of
5Govind Ballabh Pant Institute of Postgraduate Medical Education and Research, New Delhi, India
6Lebanese American University Medical Center, Rizk Hospital, Beirut, Lebanon
7University of Malaya, Kuala Lumpur, Malaysia
8Faculty of Medicine, Chiang Mai University, Chiang Mai, Thailand
9University Medical Center, University of Medicine and Pharmacy, Ho Chi Minh City, Viet Nam
10National Neuroscience Institute, Singapore General Hospital Campus, Singapore, Singapore
11Amgen Inc., Thousand Oaks, CA, United States
12Novartis Pharma AG, Basel, Switzerland
13Novartis Pharmaceuticals Corporation, East Hanover, NJ, United States
14Novartis Healthcare Pvt. Ltd., Hyderabad, India
Introduction
EMPOwER (NCT03333109) study enrolled patients from regions (Asia, Middle East and Latin America) not adequately represented in the phase 2/3 trials of erenumab.
Objectives
EMPOwER was a 12-week double-blind, randomised study comparing the efficacy and safety of erenumab (70 mg and 140 mg) in adult patients aged 18–65 years with episodic migraine (EM).
Methods
Eligible patients were randomised (3:3:2) to receive monthly doses of placebo, erenumab 70 mg or erenumab 140 mg at weeks 4, 8 and 12. Demographics and disease characteristics assessed during the 4 week baseline period prior to randomisation included monthly migraine days (MMD), monthly headache days (MHD), acute headache medication (AHM) use, and monthly migraine-specific medication days (MSMD).
Results
Of the 900 patients randomised, most patients were women (81.9%) and of Asian origin (81.2%). The mean±SD age was 37.5±9.9 years; BMI 24.6±4.4 kg/m2; and migraine duration 11.7±9.8 years. 67.9% of patients had never failed prior prophylactic treatment (PPT) and 32.1% failed ≥1 PPT. Baseline MMD, MHD and MSMD (mean±SD) were 8.2±2.8, 9.3±3.1 and 1.9±2.9 days, respectively. A total of 92.0% of patients used any AHM, and 36.8% used migraine-specific medications. The three treatment groups were well balanced in terms of demographics and baseline characteristics.
Conclusion
The majority of the study population consisted of female patients in their productive years of life, as expected from the disease epidemiology. However, migraine-specific medications were underused and a high proportion of patients had not previously used prophylactic treatment.
Disclosure of Interest
S.-J. Wang Conflict with: Received research grants from the Taiwan Minister of Technology and Science, Brain Research Center, National Yang-Ming University from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan, Taipei Veterans General Hospital, and Taiwan Headache Society., Conflict with: Served on the advisory boards of Eli Lilly and Taiwan Novartis. He has received honoraria as a moderator from Allergan, Pfizer, Eli Lilly, Bayer and Eisai., A. Roxas Jr Conflict with: Part of the advisory board and speaker of Novartis for erenumab, and involved in Novartis erenumab trials for episodic migraine (EMPOwER) and chronic migraine (DRAGON)., B. Saravia: None Declared, B.-K. Kim Conflict with: Consulting fees and honoraria from Center for New Health Technology Assessment, National Pension Service, Eli Lilly, Allergan, Teva, Lundbeck, Sanofi Korea, SK Pharm, Yuyu Pharma, and Ildong Pharm., D. Chowdhury Conflict with: Funding received from Novartis for the conduct of the study, N. Riachi Conflict with: Funding received from Novartis for the conduct of the study, M.-L. S. Tai Conflict with: Funding received from Novartis for the conduct of the study, S. Tanprawate Conflict with: Received research grants from the Faculty of Medicine, Chiang Mai University, and National Innovation Agency of Thailand., Conflict with: Received honoraria as a lecturer from Allergan, Pfizer, Eli Lilly, Novartis and Eisai., T. N. Tran Conflict with: Received consultation and/or honoraria/lecture fees from Abbott, Boehringer-Ingelheim, Ipsen Pharmaceuticals, Medtronic, and Novartis, and funding from Novartis for the conduct of this study, Z. Y. Jing: None Declared, D. Mikol Conflict with: Employee of and owns stock in Amgen, S. Pandhi Conflict with: Employee of Novartis and may own Novartis stocks., S. Wen Conflict with: Employee of Novartis and may own Novartis stocks., S. Mondal Conflict with: Employee of Novartis and may own Novartis stocks., N. Tenenbaum Conflict with: Employee of Novartis and may own Novartis stocks., P. Hours-Zesiger Conflict with: Employee of Novartis and may own Novartis stocks.
MTV20-DP-058
DOES WEARING OFF OF EFFICACY OCCUR IN GALCANEZUMAB-TREATED PATIENTS AT THE END OF THE MONTHLY TREATMENT CYCLE: A POST HOC ANALYSIS OF FOUR PHASE 3 RANDOMIZED TRIALS
Jessica Ailani1*, Dulanji K. Kuruppu2, Mallikarjuna Rettiganti2, Tina M. Oakes2, Linda A. Wietecha2, Martha D. Port2 and Andrew M. Blumenfeld3
1MedStar Georgetown University Hospital, Washington DC
2Eli Lilly and Company, Indianapolis
3The Neurology Center of Southern California, Carlsbad, CA, United States
Introduction
Anecdotal clinical reports suggest that some patients experience an increase in migraine headache frequency towards the end of their monthly CGRP monoclonal antibody (mAb) treatment cycle. This ‘wearing off’ is more commonly reported in patients with high frequency episodic migraine (HFEM) or chronic migraine (CM).
Objectives
To develop a threshold of ‘wearing off’ of efficacy at the end of a month with CGRP mAbs and determine the percentage of patients in galcanezumab (GMB) clinical trials who met this threshold.
Methods
This post hoc analysis examined data from 4 double-blind, placebo-controlled phase 3 studies in adult patients with migraine. Patients received monthly subcutaneous GMB 120mg (with 240mg loading dose), GMB 240mg, or placebo (PBO) for 6 months (EVOLVE-1 and -2; N = 1176) or 3 months (REGAIN; N = 1113; CONQUER; N = 391). Patients with an increase of ≥2 migraine headache days/week from Week 2 to Week 4 (the 7 days prior to next dose) during at least 2 of months 4–6 in EVOLVE-1 and -2 were considered to have ‘wearing off’ of efficacy. In REGAIN and CONQUER, an increase of ≥2 migraine headache days/week from Week 2 to Week 4 of both months 2 and 3 was considered as ‘wearing off’. P-values comparing rates between treatment groups were calculated using two-sided Fisher’s exact test.
Image:
Results
In the HFEM populations, rates of wearing off as defined herein were comparable among PBO- and GMB-treated patients, with 4% to 7% of patients experiencing an increase of ≥2 migraine headache days/week during Week 4 for 2 months. In EVOLVE-1 and -2 patients, for whom 3 months of data was available, fewer than 1% of patients met the defined threshold for all 3 months. In the CM populations, rates were slightly higher with GMB treatment compared to PBO, with 3% to 9% of PBO- and GMB-treated patients showing an increase of ≥2 migraine headache days/week during Week 4 for 2 months (Table 1).
Differences in wearing off between patient groups treated with 120mg and 240mg GMB were not statistically significant (all p>0.05), nor were differences between GMB treatments and PBO (all p>0.05) (Table 1).
Conclusion
In this exploratory analysis, rates of patients meeting the threshold of ‘wearing off’ were similar among the PBO, GMB 120mg, and GMB 240mg treatment groups. In each group, a small percentage of patients (≤7% in HFEM and ≤9% in CM) met the threshold during the final week of the treatment cycle.
Disclosure of Interest
J. Ailani Conflict with: Clinical trial grants have been paid to Jessica Ailani’s institution from American Migraine Foundation, Allergan, Biohaven, Eli Lilly and Company, Satsuma, and Zosano, Conflict with: Jessica Ailani has received honoraria for consulting from Amgen, Allergan/Abbvie, Biohaven, Eli Lilly and Company, Lundbeck, Teva, Impel, Satsuma, Theranica, Neurodiem, and Axsome. She has received CME programming-related fees from Miller Communications (CME content and speaker fee), Avent (CME content and speaker fee), Peer View (CME speaker fee), Pri-Med (CME content and speaker fee), Forefront (CME content and speaker fee), Medscape (CME content and speaker fee), Clinical Care Options (CME content and speaker fee), Academia for continued health care learning (CME content and speaker fee), and Answers in CME (CME content). She received honoraria for speaking from Allergan, Amgen, Biohaven, Eli Lilly and Company, Lundbeck, and Teva. She also received honoraria for editorial services from Current Pain and Headache Reports, Section editor, Unusual Headache Syndromes, D. Kuruppu Conflict with: Employee of Eli Lilly and Company, M. Rettiganti Conflict with: Employee of Eli Lilly and Company, T. Oakes Conflict with: Employee of Eli Lilly and Company, L. Wietecha Conflict with: Employee of Eli Lilly and Company, M. Port Conflict with: Employee of Eli Lilly and Company, A. Blumenfeld Conflict with: Andrew Blumenfeld has received grant support from Allergan and Amgen., Conflict with: Andrew Blumenfeld has received honoraria for serving on advisory boards of Allergan, Aeon, Alder, Amgen, Biohaven, Lilly, Novartis, Revance, Teva, Theranica, and Zoscano. He has received fees for speaking from Allergan, Amgen, Lilly, and Teva and as a consultant for Allergan, Alder, Amgen, Biohaven, Lilly, Novartis, Teva, Theranica, Zoscano. He has been a contributing author in partnership with Novartis, Teva, Allergan, and Biohaven.
MTV20-DP-059
HEALTHCARE RESOURCE UTILISATION IN MIGRAINE PATIENTS STRATIFIED BY PRIOR PROPHYLACTIC TREATMENT FAILURE, FREQUENCY OF MONTHLY MIGRAINE DAYS AND MEDICATION OVERUSE: EVIDENCE FROM THE BECOME STUDY
Charly Gaul1*, Christian Lucas2, Patricia Pozo-Rosich3,4, David PB Watson5, Paolo Martelletti6, Shannon Ritter7 and Josefin Snellman8
1Migraine and Headache Clinic Königstein, Königstein im Taunus, Germany
2Pain Clinic, Service de Neurochirurgie, Hô pital Salengro, CHU de Lille, Lille Cedex, France
3Headache Unit, Neurology Department, Vall d’Hebron University Hospital
4Headache Research Group, VHIR, Universitat Autonoma de Barcelona, Barcelona, Spain
5Hamilton Medical Group, Aberdeen, Scotland, United Kingdom
6Department of Clinical and Molecular Medicine, Sapienza University of Rome, Sant’Andrea Hospital, Rome, Italy
7Novartis Pharmaceuticals Corporation, East Hanover, NJ, United States
8Novartis Pharma AG, Basel, Swaziland
Introduction
Limited healthcare resource utilisation (HRU) data is available about migraine patients who had experienced prior prophylactic treatment failure (PPTF) in Europe.
Objectives
To report HRU in patients with migraine stratified by PPTF (1 to ≥4), frequency of monthly migraine days (MMD) and medication overuse headache (MOH) from the BECOME study.
Methods
BECOME was a prospective, multicentre, non-interventional, cross-sectional study conducted in
2 concurrent parts: Part 1 (cumulative hospital data) and Part 2 (patient-level data). Part 2 assessed migraine-related HRU in adult patients attending headache centres as outpatients/inpatients with ≥4 MMD and ≥1 PPTF in the previous 5 years.
Results
Patients with ≥4 PPTFs were more likely to have visited a neurologist in the past 3 months (64.4%) as compared to patients with 1 PPTF (53.4%), while both groups had equal proportions of patients visiting a general practitioner (GP, 31.8% vs. 32.3%). On average, patients in all PPTF groups were more likely to have visited a neurologist compared to a GP; higher in PPTFs (3 and ≥4) subgroups compared to those with 1 PPTF (
Conclusion
Overall, the number of patients visiting a neurologist for migraine were twice as high as those visiting a GP, while the mean GP visits were higher compared to neurologist visits throughout, indicating that patients probably visited GPs more often during early years of diagnosis of migraine. Migraine-related hospitalisations more than doubled with disease advancement. Although, differences in healthcare systems and migraine management exist, high HRU was observed individually across the 18 participating countries, indicating a need for improved treatment options for difficult-to-treat migraine patients with PPTF, higher MMD and MOH.
Disclosure of Interest
C. Gaul Conflict with: received honoraria for consulting and lectures within the past 3 years from Allergan, Teva-Ratiopharm, Boehringer Ingelheim Pharma, Eli Lilly, Novartis Pharma, Desitin Arzneimittel, Cerbotec, Bayer Vital, Hormosan Pharma, Grünenthal, and Reckitt Benckiser. He holds no stocks of pharmaceutical companies or medical device companies., C. Lucas Conflict with: collaboration as an expert, investigator or coordinator of clinical trials with Novartis, Teva, Sanofi, Grünenthal, Eli Lilly, Biogen, and Ethypharm., P. Pozo-Rosich Conflict with: received honoraria as a consultant and speaker during the last 5 years for Allergan, Almirall, Chiesi, Eli Lilly, Novartis, and Teva. Her research group has received research grants from Allergan and has received funding for clinical trials from Alder, Boehringer Ingelheim, MSD, electroCore, Eli Lilly, Janssen Cilag, and Novartis. She is a trustee member of the board of the International Headache Society and a Member of the Council of the European Headache Federation. She is on the editorial board of Revista de Neurologia. She is an Editor for Frontiers of Neurology and Journal of Headache and Pain. She is a member of the Clinical Trials Guidelines Committee of the International Headache Society. She has edited the Guidelines for the Diagnosis and Treatment of Headache of the Spanish Neurological Society. She does not own stocks from any pharmaceutical company., D. PB Watson Conflict with: received honoraria from Novartis, Teva, and Allergan in the last 12 months for consultancy and educational work., P. Martelletti Conflict with: Section Editor, Medicine, SpringerNature Comprehensive Clinical Medicine; Editor-in-Chief, The Journal of Headache and Pain; Headache Books Series Editor, Springer; EU Expert, European Medicine Agency. Former President of the European Federation, and Chairman of the School of Advanced Studies of the European Headache Federation. He does not hold stocks of any pharmaceutical or medical device companies., S. Ritter Conflict with: employees and own stocks in Novartis., J. Snellman Conflict with: employees and own stocks in Novartis.

MTV20-DP-060
TREATMENT SATISFACTION WITH ERENUMAB IN THE US FROM THE PATIENT PERSPECTIVE: A CROSS-SECTIONAL ANALYSIS OF THE NATIONAL HEALTH AND WELLNESS SURVEY DATA
Purnima Pathak1*, Jennifer Ken-Opurum2, Leiyu Yue2, Shaloo Gupta2, Juanzhi Fang3, Andy Cheadle3, Santosh Tiwari4, Matias Ferraris5, Roshani Shah3 and Pamela Vo5
1Novartis Ireland Ltd, Dublin, Ireland
2Kantar, New York, NY
3Novartis Pharmaceuticals Corporation, East Hanover, NJ, United States
4Novartis Healthcare Pvt. Ltd, Hyderabad, India
5Novartis Pharma AG, Basel, Switzerland
Introduction
Erenumab (erenumab-aooe in the United States [US]; Aimovig®) was approved in the US in May 2018 for the preventive treatment of migraine in adults. While the therapeutic benefits of erenumab are well established in clinical trials, real-world data is needed to understand the potential use of erenumab in addressing the unmet needs of patients with migraine in clinical practice.
Objectives
To assess patient-reported treatment satisfaction with erenumab.
Methods
This retrospective, cross-sectional study used data from the 2019 US National Health and Wellness Survey collected during March-July 2019 (NHWS; N = 74,994). Respondents self-reporting a physician diagnosis of migraine and current use of erenumab were analyzed. Treatment satisfaction was measured on a seven-point Likert scale (1 = extremely dissatisfied to 7 = extremely satisfied). Respondents were stratified into dissatisfied (percentage of respondents reporting 1 = extremely dissatisfied to 4 = neither dissatisfied nor satisfied), and satisfied (the percentage of patients reporting 5 = somewhat satisfied to 7 = extremely satisfied). Data were further stratified by duration of treatment.
Results
At the time of the survey, 67 respondents reported using erenumab treatment for up to 1 year. The mean age was 46.7±12.9 years, and 86.6% were female. Respondents were predominantly white (74.6%), commercially-insured (67.2%), and married or living with a partner (59.7%). Approximately 42.0% were former or current smokers, 71.6% were occasional drinkers, and 40.3% had at least one comorbidity based on the Charlson Comorbidity Index. Approximately half of respondents were college graduates and employed (49.3% each). Out of 67 respondents, 11 (16.4%), 23 (34.3%), and 33 (49.3%) received treatment for < 3 months, 3 to < 6 months, and 6 to 12 months, respectively. Among 67 respondents, 46 received erenumab exclusively with no other preventives. Of these, 8 (17.4%), 14 (30.4%), and 24 (52.2%) respondents received treatment for < 3 months, 3 to < 6 months, and 6 to 12 months, respectively. Across both groups, the percentage of respondents satisfied with erenumab was slightly higher among those with longer treatment duration (Figure).
Conclusion
Results demonstrated high levels of patient-reported treatment satisfaction with erenumab. These initial findings support erenumab as a valuable preventive treatment option for migraine in real-world practice.
Disclosure of Interest
P. Pathak Conflict with: Purnima Pathak is an employee of Novartis, J. Ken-Opurum Conflict with: Jennifer Ken-Opurum is an employee of Kantar, L. Yue Conflict with: Leiyu Yue is an employee of Kantar, S. Gupta Conflict with: Shaloo Gupta is an employee of Kantar, J. Fang Conflict with: Juanzhi Fang is an employee of Novartis and holds shares of Novartis, A. Cheadle Conflict with: Andy Cheadle is an employee of Novartis and holds shares of Novartis, S. Tiwari Conflict with: Santosh Tiwari is an employee of Novartis, M. Ferraris Conflict with: Matias Ferraris is an employee of Novartis and holds shares of Novartis, R. Shah Conflict with: Roshani Shah is an employee of Novartis, P. Vo Conflict with: Pamela Vo is an employee of Novartis and holds shares of Novartis

MTV20-DP-061
A COLLABORATIVE FIVE-YEAR JOURNEY DEMONSTRATING SERVICE IMPROVEMENT & EFFICIENCY IN A NURSE-LED BOTULINUM TOXIN TYPE-A (BOTOX) CLINIC FOR CHRONIC MIGRAINE
Paul Button, Siobhan Jones*
Introduction
Migraine is a very common condition affecting 12–15 % of the UK population. The World health Organisation recognises migraine as being amongst the most disabling lifetime conditions. Chronic migraine is defined as having 15 headache days or more per month, 8 of which are migraine. To enable us to meet the increasing demand for Botox® treatment for chronic migraine patients we developed a continuous process improvement program in partnership with a management-efficiency company [ProcEx Solutions Ltd] in 2015. As previously reported, we standardised the configuration of our clinic set up by utilising lean techniques and categorised patients as ‘runners’ (uncomplicated/routine), ‘repeaters’ (more time-consuming, complex) and ‘strangers’ (new patients). This enabled a standardised patient pathway with fixed appointment times. The time allocated depended on the type of patient and whether the patient could be treated in a 15 minute or a 30-minute time slot.
Objectives
To measure and monitor the value created by the continuous improvement process to refine service delivery, efficiency and capacity of the chronic migraine clinic.
Methods
Data was collected at the end of each month and a trend analysis of the number of chronic migraine patient treatments was carried out. Corrective actions were taken if the number of treatments fell below the anticipated demand. We calculated the increased capacity over the five-year period based on the data collected. To determine if the increased capacity required additional human resources, we also reviewed the headache specialist nurse resources allocated for chronic migraine treatments over the same period.
Results
In 2015 we had a waiting list of 35 patients with a wait time of 4–6 months. At that time we only had capacity to accommodate 28 new patient referrals for Botox. During the period between 2015 and 2019, the total number of yearly treatments almost doubled from 791 to 1,634. The clinic at this point was conducted by 1 headache specialist nurse. An additional nurse was then employed to support the increasing demand in early 2019. After implementing the improved process we were able to eliminate the waiting list. In 2019 we generated capacity to accommodate
Conclusion
The service efficiency program implemented in 2015 has enabled us to increase capacity to meet the increasing demands in the administration of Botox® treatment. For the first four years this was achieved without incurring any additional nursing costs. We also conclude that because of the success of the program, more patients with chronic migraine have benefited from accessing Botox® treatment. The service continues in its mission to grow and provide a superior patient experience.
Disclosure of Interest
P. Button Conflict with: The ProcEx pilot was funded by Allergan. After completion the authors continued to collect data for 5 years. Writing and editorial assistance was provided to the authors by Paul Button, LRSC of ProcEx Solutions Ltd.UK and funded by Allergan (now AbbVie) at the request of the investigator. All authors met the ICMJE authorship criteria, S. Jones Conflict with: ProcEx pilot was funded by Allergan. All authors met the ICMJE authorship criteria
MTV20-DP-062
SHIFT FROM CHRONIC TO EPISODIC MIGRAINE STATUS AND HIGH- TO LOW-FREQUENCY EPISODIC MIGRAINE STATUS AMONG PATIENTS WITH TREATMENT-RESISTANT MIGRAINE IN A PHASE 3 GALCANEZUMAB STUDY
Kathleen A. Day1, Mallik Rettiganti1, Margaret B. Ferguson1, Chad E. Stroud1, Benjamin P. Trewin2, Takao Takeshima3 and William S. Kingston4*
1Eli Lilly and Company, Indianapolis, United States
2Sharp Neurology, Sydney, New South Wales, Australia
3Headache Center, Osaka, Japan
4Sunnybrook Health Sciences Centre, Toronto, Canada
Introduction
Relative to low frequency episodic migraine (LFEM), chronic migraine (CM) and high frequency episodic migraine (HFEM) have a higher disease burden owing to higher disability, comorbidity and healthcare resource utilization.
Objectives
To assess the shift from chronic migraine (CM) to episodic migraine (EM) status and high-frequency EM (HFEM) to low-frequency EM (LFEM) status following treatment with galcanezumab (GMB) in the CONQUER study.
Methods
Patients aged 18–75 years with 2–4 migraine-preventive medication category failures over the past 10 years were randomized 1:1 to receive GMB 120mg (loading dose: 240mg) or placebo (PBO) during the 3-month, double-blind (DB) treatment period. Patients who completed the DB period entered an optional 3-month, open-label (OL) treatment period, wherein patients previously on PBO also received GMB 120mg (loading dose: 240mg). Results from patients who received GMB during the DB and OL period (GMB/GMB and PBO/GMB) are reported here. On a given month, patients were considered as shifted from CM (≥15 headache days/month, of which ≥8 are migraines) to EM status if they had < 8 migraine headache days/month or < 15 headache days/month. In addition, patients who shifted from HFEM (8 to < 15 migraine headache days/month with < 15 headache days/month) to LFEM (4 to < 8 migraine headache days/month) and to very-low-frequency EM (VLFEM; < 4 migraine headache days/month) were also reported.
Results
At baseline, 193 patients had CM (PBO = 98; GMB = 95) and 198 patients had HFEM (PBO = 96, GMB = 102). The mean (SD) number of migraine headache days/month were CM: 18.7 (4.7); EM: 9.3 (2.8) and the number of headache days/month were CM: 20.9 (4.4); EM: 10.8 (2.8). During the DB period, a significantly greater proportion of patients receiving GMB vs PBO shifted from CM to EM status: Month 1, 49.0 vs 29.1 [P = .008]; Month 2, 50.1 vs 25.7 [P = .001]; Month 3, 61.9 vs 32.1 [P≤.001]. At Month 3, significantly greater proportion of patients treated with GMB vs PBO shifted from CM to EM and sustained that shift over each of the three consecutive months of DB period, 35.8% vs 12.4% [P < .001]. By Month 3, proportions of patients treated with GMB vs PBO shifted from HFEM to LFEM and VLFEM and sustained that shift at each of the three consecutive months of DB period, HFEM to LFEM: 39.9% vs 20.5%; HFEM to VLFEM: 9.8% vs 0%. By Month 6 (3rd month of OL), 51.5% of patients in the GMB/GMB vs 46.7% [P = .540] in PBO/GMB group had ≥3 consecutive months of shift from CM to EM; 60.2% vs 66.1% [P = .413] shifted from HFEM to LFEM and 18.2% vs 22.5% [P = .437]) shifted from HFEM to VLFEM.
Conclusion
Among patients with treatment-resistant migraine, treatment with GMB resulted in a significant shift from CM to EM status as well as from HFEM to LFEM. The sustained shift to EM and LFEM observed in this study, suggests that GMB may result in reduced rates of disability and consequent lowering of the disease burden associated with CM and HFEM.
Disclosure of Interest
K. Day Conflict with: Employee of Eli Lilly and Company, M. Rettiganti Conflict with: Employee of Eli Lilly and Company, M. Ferguson Conflict with: Employee of Eli Lilly and Company, C. Stroud Conflict with: Employee of Eli Lilly and Company, B. Trewin: None Declared, T. Takeshima: None Declared, W. Kingston Conflict with: Consulting and advisory board fees from Eli Lilly and Company
MTV20-DP-063
REAL-WORLD EFFECTIVENESS OF ERENUMAB IN CHRONIC MIGRAINE PROPHYLAXIS IN CROATIA: A PROSPECTIVE STUDY
Matea Bracic1*, Darija Mahović Lakušić2 and Lukrecija Jakuš3
1School of Medicine, University of Zagreb
2Department of Neurology, University Hospital Centre Zagreb, School of Medicine Zagreb
3University of Applied Health Sciences, Zagreb, Croatia
Introduction
Erenumab is a monoclonal antibody which inhibits the calcitonin gene-related peptide (CGRP) receptor. The safety and efficacy of erenumab as a specific prophylactic medication in patients with chronic migraine have been demonstrated in clinical trials. However, real-world data for chronic migraine are still insufficient.
Objectives
Our aim was to evaluate the effectiveness of erenumab in a real-world setting in patients with chronic migraine after a 6-month treatment period.
Methods
This study included patients with chronic migraine who failed to respond to three or more classes of prophylactic migraine medication. All patients were recruited in the University Hospital Centre Zagreb, Croatia between March 2019 and November 2019 and received monthly erenumab 70 mg for 6 months. Patient interviews were conducted at the beginning of treatment and after 6 months in order to assess clinical improvement. The clinical parameters chosen for analysis were monthly migraine days (MMD), monthly non-migraine headache days (MND), monthly headache days (MHD), pain intensity measured by the visual-analogue scale (VAS) and monthly acute migraine medication intake. Statistical analysis of the data was performed using the Wilcoxon test.
Results
Out of the 57 participants who entered the study, 54 received at least one dose of erenumab 70 mg and were included in the analysis. Fifty-one patients (94.44%) were female. The mean age of the patients was 46.5±10.4 years (range 21–72). After 6 months, 70.37% of patients had a >50% reduction in monthly migraine days, while 40.74% had a >75% reduction in MMD. The mean (±SE) value of MMD was reduced from 10.37±0.38 (median 10) to 4.59±0.43 days (median 3) (p < 0.001), while the mean value of MND was reduced from 11.78±0.72 (median 10) to 5.15±0.60 days (median 4) (p < 0.001). The mean (±SE) number of MHD was also reduced from 22.24±0.70 (median 22) to 9.74±0.91 days (median 8) (p < 0.001). Furthermore, mean (±SE) migraine pain intensity measured by the VAS score decreased from 10.00±0 (median 10) to 6.69±0.24 (median 6.5) (p < 0.001). Mean (±SE) monthly acute migraine medication intake decreased from 22.78±0.74 (median 20) to 9.72±1.02 (median 6.5) (p < 0.001) tablets per month.
Conclusion
The data from this real-world study show that erenumab is an effective prophylactic therapeutic option in patients with chronic migraine.
Disclosure of Interest :
None Declared
MTV20-DP-064
LONG-TERM EFFICACY AND SAFETY OF FREMANEZUMAB IN PATIENTS WITH EPISODIC AND CHRONIC MIGRAINE WHO HAD INADEQUATE RESPONSE TO 2–4 PRIOR MIGRAINE PREVENTIVE MEDICATION CLASSES: OPEN-LABEL EXTENSION OF THE PHASE 3B FOCUS STUDY
Messoud Ashina1*, Joshua M. Cohen2, Maja Galic3, Verena Ramirez Campos2, Evelyn Du2, Xiaoping Ning2, Egilius L. H. Spierings4 and Hans-Christoph Diener5
1Danish Headache Center, Department of Neurology, Rigshospitalet, Denmark
2Teva Branded Pharmaceutical Products R&D, Inc, West Chester, PA, United States
3Teva Pharmaceuticals Industries, Amsterdam, Netherlands
4Boston Headache Institute, Boston PainCare, Waltham, MA, United States
5Universitätsklinikum Essen, Essen, Denmark
Introduction
Results from the 12-week, double-blind, placebo (PBO)-controlled, phase 3b FOCUS study showed that fremanezumab, a fully-humanized monoclonal antibody (IgG2Δa) that selectively targets calcitonin gene-related peptide (CGRP), was effective and well tolerated for the preventive treatment of episodic and chronic migraine (EM and CM) in patients (pts) with documented inadequate responses to 2–4 classes of migraine preventive medications.
Objectives
To evaluate the long-term efficacy and tolerability of fremanezumab during the open-label extension (OLE) of the phase 3b FOCUS study.
Methods
The FOCUS study included a 12-week double-blind period (DBP) and 12-week OLE. Pts were initially randomized (1:1:1) to quarterly (qtly) fremanezumab (Month 1/2/3: 675mg/PBO/PBO), monthly (mtly) fremanezumab (Month 1/2/3: 675mg [CM], 225mg [EM]/225mg/225mg), or matched mtly PBO for 12 weeks. All pts completing the DBP entered the OLE and received 3 mtly fremanezumab doses of 225mg. Efficacy and tolerability were evaluated during the OLE and summarized by double-blind randomization group.
Results
Of 838 pts randomized, 807 completed the DBP and entered the OLE; 772 completed the OLE. In the PBO, qtly fremanezumab, and mtly fremanezumab groups, respectively, mean (SD) changes from baseline in mtly average migraine days during the 12-week OLE period were −4.7(5.41), −5.1(4.71), and −5.5(4.96), and mean (SD) changes in mtly average headache days of at least moderate severity were −4.5(5.04), −4.8(4.47), and −5.2(4.94). Proportions of pts achieving ≥50% reduction from baseline in mtly average migraine days during the 12-week OLE period were similar in pts continuing on fremanezumab (qtly, 45%; mtly, 46%) and rolling over from PBO (38%). Substantial reductions in disability were observed with fremanezumab treatment. Sustained reductions in migraine-related symptoms (nausea or vomiting, photophobia, and phonophobia) were also observed. Adverse events were reported for similar proportions of pts during the OLE regardless of DBP treatment, and no safety signals were identified.
Conclusion
Fremanezumab demonstrated sustained efficacy up to 6 months and long-term tolerability in pts with EM or CM who had inadequate response to 2–4 classes of prior migraine preventive medications.
Disclosure of Interest
M. Ashina Conflict with: serves as a consultant and/or scientific advisor for Alder BioPharmaceuticals, Allergan, Amgen, Biohaven, Eli Lilly, Lundbeck, Novartis, Teva Pharmaceuticals; a principal investigator for Alder, Allergan, Amgen, Eli Lilly, Novartis, Teva Pharmaceuticals; an associate editor for Cephalalgia and associate editor for Headache; and a co-editor for the Journal of Headache and Pain, J. Cohen Conflict with: Employee of Teva Pharamaceuticals, M. Galic Conflict with: employee of Teva Pharmaceuticals, V. Ramirez Campos Conflict with: employee of Teva Pharmaceuticals, E. Du Conflict with: employee of Teva Pharmaceuticals, X. Ning Conflict with: employee of Teva Pharmaceuticals, E. L. Spierings Conflict with: Teva Pharmaceuticals, Conflict with: member of Teva’s Speakers Bureau, H.-C. Diener Conflict with: honoraria for participation in clinical trials, contribution to advisory boards, or oral presentations from Abbott, Allergan, AstraZeneca, Bayer Vital, Boehringer Ingelheim, Bristol-Myers Squibb, CoAxia, Corimmun, Covidien, Daiichi Sankyo Pharma, DPharm, Fresenius, GlaxoSmithKline, Janssen-Cilag, Johnson & Johnson, Knoll, Lilly, Medtronic, Merck Sharp & Dohme Corp., MindFrame, Neurobiological Technologies, Novartis, Novo Nordisk, Paion, Parke-Davis, Pfizer, Sanofi Aventis, Schering-Plough, Servier, Solvay, St Jude Medical, Syngis, Talecris, Thrombogenics, WebMD Global, Wyeth, and Yamanouchi. Financial support for research projects was provided by AstraZeneca, GlaxoSmithKline, Boehringer Ingelheim, Lundbeck, Novartis, Janssen-Cilag, Sanofi Aventis, Syngis, and Talecris.
MTV20-DP-065
EFFICACY OF FREMANEZUMAB IN PATIENTS WITH MIGRAINE AND COMORBID DEPRESSION
Dawn C. Buse1*, Joshua M. Cohen2, Xiaoping Ning2, Evelyn Du2, Verena Ramirez Campos2 and Richard B. Lipton1
1Department of Neurology, Albert Einstein College of Medicine, The Bronx, NY
2Teva Branded Pharmaceutical Products R&D, Inc, West Chester, PA, United States
Introduction
Patients (pts) with migraine and comorbid depression experience reduced quality of life and may experience reduced efficacy with treatment. Fremanezumab, a fully humanized monoclonal antibody (IgG2Δa) that selectively targets calcitonin gene-related peptide (CGRP), is approved for the preventive treatment of migraine.
Objectives
The efficacy of fremanezumab in adults with migraine and comorbid moderate to severe depression was evaluated in subgroup analyses in a randomized, double-blind study and a long-term treatment study.
Methods
In a 12-week, placebo (PBO)-controlled study, pts with episodic or chronic migraine (EM or CM) and inadequate response to 2–4 prior migraine preventive medication classes were randomized (1:1:1) to quarterly fremanezumab (Month 1/2/3: 675mg/PBO/PBO), monthly fremanezumab (Month 1/2/3: 225[EM],675[CM]/225/225mg), or matched PBO. In a separate 52-week study, CM patients received monthly fremanezumab (225mg monthly [675mg starting dose] or quarterly fremanezumab (675mg every 3 months). For both studies, post hoc subgroup analyses evaluated efficacy in pts with comorbid moderate to severe depression (based on a Patient Health Questionnaire [PHQ]-9 score ≥10) at baseline (BL). Changes from BL in monthly average migraine days (MMDs), headache days of at least moderate severity (HDs), and PHQ-9 scores were evaluated in these subgroups.
Results
In the 12-week study, 154 of 838 randomized pts had moderate to severe depression. In the 52-week study, 219 of 1103 of CM pts had moderate to severe depression. In the 12-week study, mean reductions from BL in MMDs were significantly greater with fremanezumab versus PBO at 12 weeks (quarterly, −3.2; monthly, −3.9 vs 0.2; P < 0.01), as were reductions in HDs (quarterly, −4.3; monthly, −4.7 vs −0.8; P < 0.01). In that 12-week study, fremanezumab treatment was also associated with greater mean reductions from BL in the PHQ-9 score versus PBO, although differences were not statistically significant (quarterly, −6.7; monthly, −7.8 vs −4.9; P = 0.3357 and 0.0943, respectively). In the 52-week study, mean reductions from BL at Month 12 with quarterly and monthly fremanezumab, respectively, were –7.2 and –8.6 in MMDs and –6.3 and –7.0 in HDs. In that 52-week study, mean reductions in the PHQ-9 score from BL at Month 12 were –10.3 with quarterly fremanezumab and –10.5 with monthly fremanezumab.
Conclusion
Fremanezumab was effective in migraine pts with comorbid moderate to severe depression and was associated with improvement in depressive symptoms, as measured by PHQ-9 scores, over long-term treatment and in pts with difficult-to-treat migraine.
Disclosure of Interest
D. Buse Conflict with: honoraria from Allergan, Amgen, Avanir, Biohaven, Lilly, Promeius and Teva, on the editorial board of Current Pain and Headache Reports, J. Cohen Conflict with: employee of Teva Pharmaceuticals, X. Ning Conflict with: employee of Teva Pharmaceuticals, E. Du Conflict with: employee of Teva Pharmaceuticals, V. Ramirez Campos Conflict with: employee of Teva Pharmaceuticals, R. Lipton Conflict with: Edwin S. Lowe Professor of Neurology at the Albert Einstein College of Medicine in New York. He receives research support from the NIH: 2PO1 AG003949 (mPI), 5U10 NS077308 (PI), RO1 NS082432 (Investigator), 1RF1 AG057531 (Site PI), RF1 AG054548 (Investigator), 1RO1 AG048642 (Investigator), R56 AG057548 (Investigator), K23 NS09610 (Mentor), K23AG049466 (Mentor), 1K01AG054700 (Mentor). He also receives support from the Migraine Research Foundation and the National Headache Foundation. He serves on the editorial board of Neurology, senior advisor to Headache, and associate editor to Cephalalgia. He has reviewed for the NIA and NINDS, holds stock options in eNeura Therapeutics and Biohaven Holdings; serves as consultant, advisory board member, or has received honoraria from: American Academy of Neurology, Alder, Allergan, American Headache Society, Amgen, Autonomic Technologies, Avanir, Biohaven, Biovision, Boston Scientific, Dr. Reddy’s, Electrocore, Eli Lilly, eNeura Therapeutics, GlaxoSmithKline, Merck, Pernix, Pfizer, Supernus, Teva, Trigemina, Vector, Vedanta. He receives royalties from Wolff’s Headache 7th and 8th Edition, Oxford Press University, 2009, Wiley and Informa.
MTV20-DP-066
HEALTH-RELATED QUALITY OF LIFE OUTCOMES WITH FREMANEZUMAB IN PATIENTS WITH EPISODIC AND CHRONIC MIGRAINE WHO HAD INADEQUATE RESPONSE TO 2–4 PRIOR MIGRAINE PREVENTIVE MEDICATION CLASSES: OPEN-LABEL EXTENSION OF THE PHASE 3B FOCUS STUDY
Dawn C. Buse1*, Xiaoping Ning2, Verena Ramirez Campos2, Joshua Cohen2, Evelyn Du2 and Laszlo Mechtler3
1Department of Neurology, Albert Einstein College of Medicine, New York, NY
2Teva Branded Pharmaceutical Products R&D, Inc., West Chester, PA
3Dent Neurologic Institute, Buffalo, NY, United States
Introduction
Fremanezumab, a fully-humanized monoclonal antibody (IgG2Δa) that selectively targets calcitonin gene-related peptide (CGRP), demonstrated efficacy and tolerability as a preventive treatment in adult patients (pts) with episodic and chronic migraine (EM and CM) and documented inadequate responses to 2–4 classes of migraine preventive medications in the 12-week double-blind period (DBP) of the phase 3b FOCUS study. Significant improvements were also observed in migraine-specific and overall health-related quality of life (HRQoL) with fremanezumab versus placebo (PBO) at the end of the DBP.
Objectives
To evaluate long-term HRQoL outcomes with fremanezumab in the 12-week open-label extension (OLE) of the phase 3b FOCUS study.
Methods
The FOCUS study consisted of a 12-week DBP and 12-week OLE. Pts were initially randomized (1:1:1) to quarterly fremanezumab (Month 1/2/3: 675mg/PBO/PBO), monthly fremanezumab (Month 1/2/3: 675mg [CM], 225mg [EM]/225mg/225mg), or matched monthly PBO for 12 weeks. All pts completing the DBP entered the OLE and received 3 monthly doses of fremanezumab (225mg). Mean changes from baseline in migraine-specific quality of life (MSQOL) and work productivity and activity impairment (WPAI) were measured at the end of the DBP and OLE and summarized using descriptive statistics. Patient Global Impression of Change (PGIC; moderately better, better, or great deal better) and Patient Health Questionnaire (PHQ-9) were also assessed.
Results
Of 838 pts randomized, 807 completed the DBP and entered the OLE; 772 pts completed the OLE. Scores for all 3 domains of MSQOL questionnaire showed continued improvement during the OLE across treatment groups. Improvements in WPAI observed during the DBP were maintained in all 4 domains and across all randomization groups during the OLE. The proportion of responders based on the PGIC increased with longer duration of fremanezumab treatment, with the majority of pts (PBO, 69%; quarterly fremanezumab, 77%; monthly fremanezumab, 75%) considered responders at the end of the OLE. Improvements in depression symptomology based on the PHQ-9 were also maintained during the OLE across all double-blind treatment groups.
Conclusion
In pts with EM or CM and inadequate response to 2–4 classes of prior migraine preventive medications, long-term administration of fremanezumab maintained improvements in HRQoL, as measured by the MSQOL, WPAI, PGIC, and PHQ-9 questionnaires.
Disclosure of Interest
D. Buse Conflict with: D.C. Buse received grant support and honoraria from Allergan, Amgen, Avanir, Biohaven, Eli Lilly, Promius, and Teva Pharmaceuticals. D.C. Buse is on the editorial board of Current Pain and Headache Reports., X. Ning Conflict with: X. Ning is an employee of Teva Pharmaceuticals., V. Ramirez Campos Conflict with: V. Ramirez Campos is an employee of Teva Pharmaceuticals., J. Cohen Conflict with: J. Cohen is an employee of Teva Pharmaceuticals., E. Du Conflict with: E. Du is an employee of Teva Pharmaceuticals., L. Mechtler Conflict with: L. Mechtler served as an investigator on this study for Teva Pharmaceuticals.
MTV20-DP-068
IMPACT OF PREVENTIVE TREATMENT ON TESTS OF ATTENTION, VIGILANCE AND EXECUTIVE FUNCTIONS IN MIGRAINE: A CLINIC-BASED STUDY FROM NORTH INDIA
Debashish Chowdhury*, Debabrata Datta1 and Ankit Mundra1
1Neurology, G B Pant Institute of Post Graduate Medical Education and Research, New Delhi, India, New Delhi, India
Introduction
Several clinic-based studies have shown interictal deficits of attention, vigilance and executive functions in migraine patients. However, it is unknown whether these dysfunctions improve following successful treatment of migraine.
Objectives
To compare the tests of attention, vigilance and executive functions in migraine patients during the inter-ictal phase before and after the institution of preventive treatment.
Methods
Consecutive migraine patients aged >18 years were recruited prospectively. None of the patients received migraine-specific preventive drugs in the past. Frontal assessment battery, digit span forward and backward, Stroop word, Stroop color, and Stroop word & color interference tests, trail making test A and B scores were generated at the baseline and optimal oral preventive treatment for migraine was instituted according to the standard of care. Treatment outcome and cognitive functions were assessed at 6 months.
Results
Out of 150 patients enrolled, 120 patients remained on follow up till 6 months. There were 26 males and 94 females and the mean age of migraine onset was 29.1±8.5 years. 51.5% received a single preventive drug, 35.8% received combinations and 12.7% were on acute symptomatic treatment only. Fifty percent or more reduction of migraine days was observed in 74 patients (62%). Significant reduction of mean monthly headache days (11.5±9.7 vs 5.5±6.2; p = 0.0253), migraine days (7.0±4.4 vs 3.4±3; p = 0.0004), headache severity by visual analog scale score (7.1±1.4 vs 5.6±1.5; p = 0.007) and headache impact test-6 score (61.2±6.0 vs 54.8±8.2; p = 0.0131) was observed at the end of 6 months of treatment. All the cognitive function tests improved significantly at 6 months as compared to baseline (table).
Conclusion
Successful treatment resulted in improvement in tests of attention, vigilance and executive functions in migraine patients. These results therefore suggest a possible pathophysiological role of recurrent migraine headaches in causing these cognitive dysfunctions.
Disclosure of Interest
None Declared

MTV20-DP-069
DEMOGRAPHICS, METHODOLOGY AND DISEASES CHARACTERISTICS OF A RANDOMIZED TRIAL STUDYING EFFICACY OF GREATER OCCIPITAL NERVE BLOCK AS AN ADD-ON THERAPY TO TOPIRAMATE FOR THE PREVENTIVE TREATMENT OF CHRONIC MIGRAINE
Ankit Mundra*, Debashish Chowdhury1; GB Pant Headache Group
1Neurology, GB Pant Institute of Postgraduate Medical Education and Research, New Delhi, India
Introduction
Efficacy of greater occipital nerve block (GON-block) as an add-on therapy to topiramate for the preventive treatment of chronic migraine (CM) has not been studied previously.
Objectives
This was a randomized study comparing the efficacy and safety of GON-block as an add-on therapy to topiramate in adult CM patients against topiramate alone.
Methods
Study population consisted of adult patients (>18 years) of CM. Eligible patients were randomized to receive oral topiramate alone 100mg /day (group A), oral topiramate plus GON-block with 40mg (2ml) 2% lidocaine + 80 mg (2ml) methylprednisolone as the first injection followed by monthly injections of 40mg (2ml) 2% lidocaine for 2 months(group B) and oral topiramate plus GON- block with 40mg (2ml) 2% lidocaine monthly for 3 months (group C). For each patient, one month baseline period prior to randomization captured monthly migraine and headache days (MMD, MHD), days of acute headache medication use, average headache severity, headache impact and disability scores. Efficacy assessments were done at the end of 3 months. The sample size was estimated to be 40 patients for each group.
Results
A total of 125 patients were randomized (41 for group A, 44 for group B, and 40 for group C) [table]. Overall, the study population included 11 males and 114 females. The mean age of onset and duration of migraine was 32.9±9.7 years and 67.5±59.2 months respectively. The mean duration of CM was 4.4±1.3 months. Ninety-five patients also had medication overuse headache (MOH). Mean MMD and MHD were 14.1±5.1, and 23.5±5.3 respectively. The average monthly days of acute headache medication use was 12.8±5.4. Mean VAS and mean HIT-6 scores were 7.8±0.8 and 64.9±3.6 respectively. All patients had MIDAS grade 4 disabilities.
Conclusion
The treatment groups were well balanced in terms of demographics, headache profiles, and baseline characteristics. The majority of the study population consisted of female patients. About two-third patients also had co-existent MOH.

MTV20-DP-070
EARLY EFFICACY OF FREMANEZUMAB IN PATIENTS WITH EPISODIC AND CHRONIC MIGRAINE AND INADEQUATE RESPONSE TO ≥1 CLASS OF MIGRAINE PREVENTIVE MEDICATION DUE TO LACK OF EFFICACY: RESULTS OF THE INTERNATIONAL, MULTICENTER PHASE 3B FOCUS STUDY
Amaal J. Starling1*, Jan Lewis Brandes2,3, Verena Ramirez Campos4, Evelyn Du4, Joshua M. Cohen4, Maja Galic5, Xiaoping Ning4 and Christina Treppendahl6
1Department of Neurology, Mayo Clinic, Scottsdale, AR, United States
2Nashville Neuroscience Group, Nashville, TN
3Department of Neurology, Vanderbilt University, Nashville, TN, United States
4Teva Branded Pharmaceutical Products R&D, Inc, West Chester, PA, United States
5Teva Pharmaceuticals Industries, Amsterdam, Netherlands
6The Headache Center, Ridgeland, MS, United States
Introduction
Fremanezumab, a fully-humanized monoclonal antibody (IgG2Δa) that selectively targets calcitonin gene-related peptide (CGRP), has demonstrated efficacy in adults with episodic (EM) or chronic migraine (CM), with documented previous inadequate responses to 2–4 classes of migraine preventive medications.
Objectives
This analysis evaluated early onset of efficacy for fremanezumab in the FOCUS study in patients (pts) with inadequate response to ≥1 prior preventive medication due to lack of efficacy.
Methods
In the 12-week, double-blind study, pts were randomized (1:1:1) to quarterly (qtly) fremanezumab (Month 1/2/3: 675mg/placebo[PBO]/PBO), monthly (mtly) fremanezumab (Month 1/2/3: 675mg [CM], 225mg [EM]/225mg/225mg), or matched PBO. Efficacy outcomes evaluated over the first 4 weeks in pts with inadequate response to ≥1 prior migraine preventive medication due to lack of efficacy were changes from baseline (BL) in mtly average migraine days (MMDs) and headache days of at least moderate severity (MHDs), and proportions of pts with ≥50% reduction in mean mtly MMDs.
Results
Of 838 pts randomized, 741 demonstrated inadequate response to prior preventive migraine medication classes due to lack of efficacy. Changes from BL in MMDs over 4 weeks was significantly greater with qtly and mtly fremanezumab, respectively, vs PBO (least-squares mean difference [LSMD] vs PBO: −3.5 and −3.5; P < 0.0001). Changes from BL in MHDs were also significantly greater with fremanezumab vs PBO (LSMD: qtly, −3.6; mtly, −3.9; P < 0.0001). Changes from BL in MMDs and MHDs were significantly greater with qtly and mtly fremanezumab vs PBO across all classes of prior preventive medications. Over weeks 1–4, the proportion of pts with ≥50% reduction in MMDs was significantly greater with qtly and mtly fremanezumab vs PBO overall, and trended greater vs PBO across all prior preventive medication classes.
Conclusion
In migraine pts with documented inadequate response due to lack of efficacy to ≥1 prior migraine preventive medication, fremanezumab rapidly reduced the number of MDs and HDs and improved 50% responder rates vs PBO within the first 4 weeks.
Disclosure of Interest
A. Starling Conflict with: consultant for Alder, Allergan, Amgen, Axsome Therapeutics, eNeura, Eli Lilly & Company, Impel, Lundbeck, Med-IQ, Medscape, Novartis, Teva, and Theranica; research funding from Migraine Research Foundation and Mayo Clinic; and honoraria from World Health Education Foundation, J. L. Brandes Conflict with: speaker’s bureaus/advisory boards for Amgen, Teva Pharmaceuticals, Eli Lilly, Promius Pharma, Supernus Pharmaceuticals, Novartis, Alder, Valeant, and electroCore; receives research support from Amgen, Teva Pharmaceuticals, Lilly/CoLucid, Biohaven, Allergan, Clinvest, and Alder, V. Ramirez Campos Conflict with: employee of Teva Pharmaceuticals, E. Du Conflict with: employee of Teva Pharmaceuticals, J. Cohen Conflict with: employee of Teva Pharmaceuticals, M. Galic Conflict with: employee of Teva Pharmaceuticals, X. Ning Conflict with: employee of Teva Pharmaceuticals, C. Treppendahl Conflict with: speaker’s bureaus/advisory boards for Allergan, Amgen, Avanir Pharmaceuticals, Depomed, Iroko Pharmaceuticals, Nautilus Neurosciences, Novartis, Pernix Therapeutics Holdings, Promius Pharma, Supernus Pharmaceuticals, Teva Pharmaceuticals, Zogenix, electroCore, Eli Lilly, Alder, Impel Neuropharma, and Upsher-Smith Laboratories
MTV20-DP-071
THE COMPARATIVE EFFICACY OF SPENOPALATINUM GANGLION BLOCKES IN PROPHYLACTIC TREATMENT OF CHRONIC MIGRAINE
Dmytro Sotnikov1* and Olexander Potapov1
1Sumy State University, Sumy, Ukraine
Introduction
A study of organized population groups in Ukraine revealed a prevalence of chronic migraine within 0.9%.
Objectives
We set the goal to study the comparative efficacy of the two local effects on spenopalatinum ganglion (SPG) as an important modulator of headache.
Methods
The study involved 90 patients with chronic migraine, aged 18 to 60 (avarage 32.68 ± 12.4) years. The majority of patients were women – 79 (87.8%). Taking into account the transformation of headache during chronic migraine, we differentiated 2 types of cephalagia: the intensity of 5 or more points for visual analogue scale (VAS) which limits the daily activities was considered as an attack, and in other cases, the headache was regarded as "background" (bilinear, moderate). By random allocation, the patients were divided into 3 groups 30 people each. All patients for prophylaxis used a combination of Amitriptyline and Propranolol that proved to be effective in our previous studies. The average daily dose of Amitriptyline was 35.5 ± 4.8 mg, Propranolol 78.5 ± 6.8 mg. The first group of patients received only pharmacological treatment. Patients in the second group were additionally treated with a bilateral SPG block throgh transnasal approach involving usage of a cotton tipped applicator soaked in Lidocaine and Betamethason every two weeks. In the third group of patients combination of Amitriptyline with Propranolol and a bilateral SPG block via lateral infratemporal approach with the injection of Lidocaine and Betamethasone were used.
Results
According to the study the number of days with headache decreased by more than 50%: in the first group by 44.5% of patients, in the second group – by 55.6%, in the third – by 61.8%. At the same time the number of days with “background” cephalgia in the groups decreased by 54.1%, 62.2% and 68.9% accordingly. The intensity of the “background” headache decreased by 22.5% in the first group, by 19.5% in the second and by 23.1% in the third. The frequency of attacks in groups decreased by 45.8%, 50.0% and 54.1% accordingly. The intensity of cephalgia during attacks decreased in all groups approximately equally: by 21.2%, 21.8% and 25.3%. The difference in the decrease in the duration of attacks between the groups also showed insignificant: by 40.4%, 44.0% and 44.3%.
Conclusion
The usage of SPG blockes as an addition to the pharmacological preventive treatment of chronic migraine has been shown to be effective in reducing the number of days with a headache, especially with regard to background headaches. The injection of Lidocaine with Betamethasone lateral infratemporal approach showed better results than the transnasal approach. However, taking to account not such a significant advantage of the lateral infratemporal approach and certain technical difficulties in implementing of this blockade, when having a necessity to potentiate the action of Amitriptyline with Propranolol combination we would recommend to start with SPG block via transnasal approach.
Disclosure of Interest
None Declared
MTV20-DP-072
POOLED ANALYSIS OF CARDIOVASCULAR SAFETY WITH FREMANEZUMAB TREATMENT IN PATIENTS WITH MIGRAINE AND CONCOMITANT CARDIOVASCULAR MEDICATION OR TRIPTAN USE AT BASELINE
Gianluca Coppola1*, Xiaoping Ning2, Yoel Kessler2, Joshua M. Cohen2, Evelyn Du2 and Piero Barbanti3
1Sapienza University of Rome Polo Pontino, Department of Medico-Surgical Sciences and Biotechnologies, Latina, Italy
2Teva Branded Pharmaceutical Products R&D, Inc., West Chester, PA, United States
3Headache and Pain Unit, IRCCS San Raffaele Pisana, Rome, Italy
Introduction
Fremanezumab, a fully-humanized monoclonal antibody (IgG2Δa) that selectively targets calcitonin gene-related peptide (CGRP), has proven efficacy for the preventive treatment of migraine in adults.
Objectives
These pooled analyses evaluated the cardiovascular (CV) safety of fremanezumab in a subgroup of patients with episodic migraine (EM) and chronic migraine (CM) and concomitant use of CV medications or triptans at baseline.
Methods
These pooled analyses of CV safety of fremanezumab included data from 3 international, multicenter, randomized, double-blind, placebo (PBO)-controlled trials: the 2 HALO studies (1 in EM, 1 in CM) and the FOCUS study in patients with EM or CM who had inadequate response to 2–4 classes of migraine preventive medications. In all 3 trials, patients were randomized 1:1:1 to quarterly fremanezumab (Month 1/2/3: 675mg/PBO/PBO), monthly fremanezumab (Month 1/2/3: 675mg [CM], 225mg [EM]/225mg/225mg), or matched PBO for 12 weeks. CV adverse events (CVAEs) were evaluated in patients with baseline CV medication use and in patients with and without concomitant triptan use.
Results
Of the total pooled population of 2,842 patients across these 3 studies, 280 patients were receiving CV medications at baseline (most commonly agents acting on the renin-angiotensin system or beta-blockers) and 1,123 patients used triptans during the studies. For patients receiving CV medications at baseline, cardiac disorder AEs were infrequent across all treatment groups (PBO, < 1%; quarterly fremanezumab, 0%; monthly fremanezumab [675/225/225mg], 2%; monthly fremanezumab [225/225/225mg], 0%), as were vascular disorder AEs (0%, 1%, 6%, and 0%, respectively). In patients with and without triptan use, the incidence of CVAEs was similar (2% vs 3%) and consistently low across all treatment groups. The only CVAE with >1 occurrence in the PBO and fremanezumab groups was hypertension (PBO, n = 2; quarterly fremanezumab, n = 2; monthly fremanezumab [675/225/225mg], n = 1; monthly fremanezumab [225/225/225mg], n = 0). No safety signals were identified.
Conclusion
This pooled analysis demonstrates that fremanezumab treatment over 12 weeks was well tolerated; the incidence of CVAEs was low and similar to PBO in patients with migraine using CV medications at baseline or in patients with concomitant triptan use.
Disclosure of Interest
G. Coppola Conflict with: served as an advisor for Teva Pharmaceuticals, Novartis, and Eli Lilly, X. Ning Conflict with: employee of Teva Pharmaceuticals, Y. Kessler Conflict with: employee of Teva Pharmaceuticals, J. Cohen Conflict with: employee of Teva Pharmaceuticals, E. Du Conflict with: employee of Teva Pharmaceuticals, P. Barbanti Conflict with: consultant, advisory board member, investigator on clinical studies or has received honoraria from Amgen, Alder, Allergan, Bayer, Electrocore, Eli Lilly, GSK, Lusofarmaco, New Penta, Novartis, Teva Pharmaceuticals, Visufarma.
MTV20-DP-073
EFFICACY IN PATIENTS SWITCHING FROM QUARTERLY TO MONTHLY FREMANEZUMAB OR MAINTAINED ON MONTHLY FREMANEZUMAB TREATMENT OVER 6 MONTHS IN THE PHASE 3B FOCUS STUDY
Jonathan Gladstone1*, Hans-Christoph Diener2, Xiaoping Ning3, Joshua M. Cohen3, Verena Ramirez Campos3, Evelyn Du3 and Egilius L. H. Spierings4
1Gladstone Headache Clinic, Toronto, ON, Canada
2Universitätsklinikum Essen, Essen, Germany
3Teva Branded Pharmaceutical Products R&D, Inc., West Chester, PA
4Boston Headache Institute, Boston PainCare, Waltham, MA, United States
Introduction
In the 12-week, double-blind, placebo (PBO)-controlled, phase 3b FOCUS study, fremanezumab, a fully-humanized monoclonal antibody (IgG2Δa) that selectively targets calcitonin gene-related peptide (CGRP), dosed quarterly (qtly) or monthly (mtly) was effective and well tolerated in migraine preventive treatment in patients (pts) with episodic or chronic migraine (EM or CM) and inadequate response to 2–4 prior classes of migraine preventive medications. In a 12-week open-label extension (OLE), all pts received mtly fremanezumab.
Objectives
To assess efficacy in pts who switched from qtly fremanezumab during the double-blind period (DBP) to mtly fremanezumab (switch subgroup) in the OLE and in pts who received mtly fremanezumab (maintenance subgroup) throughout the DBP and OLE.
Methods
FOCUS included a 12-week DBP and 12-week OLE. Pts were initially randomized 1:1:1 to qtly fremanezumab (Month 1/2/3: 675mg/PBO/PBO), mtly fremanezumab (Month 1/2/3: 675mg [CM], 225mg [EM]/225mg/225mg), or matched PBO for DBP. All pts completing DBP entered the OLE and received 3 mtly fremanezumab doses (225 mg). Efficacy measures during OLE were summarized by DB randomization group.
Results
Of 838 pts randomized, 807 completed DBP and entered OLE: 271 were in the switch subgroup and 272 in the maintenance subgroup. For pts in the switch and maintenance subgroups, respectively, comparable mean changes from baseline (BL) during the OLE were observed in mtly average migraine days (−5.1 and −5.5), headache days of at least moderate severity (−4.8 and −5.2), and days of any acute headache medication use (−4.9 and −4.8). The proportion of pts who achieved ≥50% reduction from BL in mtly average migraine days during the OLE was also comparable in the switch (45%) and maintenance (46%) subgroups. During the OLE, mean changes from BL in disability outcomes were also similar for pts in the switch and maintenance subgroups, respectively, including the 6-item Headache Impact Test (−8.4 and −8.3) and Migraine Disability Assessment (−29.9 and −33.9).
Conclusion
Efficacy, based on reductions in migraine days, headache days of at least moderate severity, and days of acute headache medication use, proportion of pts achieving clinically meaningful reduction in migraine days, and reductions in disability outcomes, was comparable in pts switching from qtly to mtly fremanezumab dosing during the OLE and those maintained on mtly dosing throughout the DBP and OLE.
Disclosure of Interest
J. Gladstone Conflict with: consulted for or served on an advisory board or speakers bureau for Allergan, Amgen, Aralez, Lilly, Novartis, Nuvo, and Teva Pharmaceuticals, H.-C. Diener Conflict with: honoraria for participation in clinical trials, contribution to advisory boards, or oral presentations from Alder, Allergan, Amgen, Autonomic Technologies, Bristol-Myers Squibb, CoLucid, electroCore, Ipsen Pharma, Lilly, Medtronic, MSD, Novartis, Pfizer, Schaper & Brümmer, Teva Pharmaceuticals, and Weber & Weber; and has received financial support for research projects from Allergan, electroCore, MSD, and Pfizer; serves on the editorial boards of Cephalalgia and Lancet Neurology; chairs the Clinical Guidelines Committee of the German Society of Neurology; is a member of the Clinical Trials Committee of the HIS; and has no ownership interest and does not own stocks of any pharmaceutical company. H.-C. Diener’s headache research at the Department of Neurology in Essen is supported by the German Research Council, the German Ministry of Education and Research, and the European Union, X. Ning Conflict with: employee of Teva Pharmaceuticals, J. Cohen Conflict with: employee of Teva Pharmaceuticals, V. Ramirez Campos Conflict with: employee of Teva Pharmaceuticals, E. Du Conflict with: employee of Teva Pharmaceuticals, E. L. Spierings Conflict with: FOCUS study investigator and has received research grants from and served on a speakers bureau for Teva Pharmaceuticals.
MTV20-DP-074
COADMINISTRATION OF SINGLE THERAPEUTIC ORAL DOSES OF ATOGEPANT AND SUMATRIPTAN PRODUCES NO CLINICALLY RELEVANT DRUG-DRUG INTERACTIONS
Ramesh Boinpally1*, Abhijeet Jakate1, Matthew Butler1 and Antonia Periclou1
1AbbVie, Madison, NJ, United States
Introduction
Atogepant is an oral calcitonin gene‒related peptide receptor antagonist in development for migraine prevention. The serotonin receptor antagonist, sumatriptan, is the most commonly used acute treatment for migraine attacks in the US.
Objectives
Because there is a possibility that people with migraine will take atogepant and sumatriptan together, this single-center, open-label, randomized, phase 1, pharmacokinetic (PK) study examined the potential for PK drug-drug interaction between the 2 medications.
Methods
Healthy adults (N = 30) aged 18–45 years were randomized to 1 of 6 sequences in which they received 3 single-dose oral treatments: atogepant 60 mg, sumatriptan 100 mg, and atogepant 60 mg plus sumatriptan 100 mg. This 3-way crossover design included study drug administration on days 1, 8, and 15, a 7-day washout period between treatments, and a safety visit 30 days after the last treatment. Pre- and post-dose blood samples were used to evaluate the potential for PK drug-drug interaction as the primary endpoint. For the comparison of study drug in combination vs study drug alone, geometric mean ratios and 90% confidence intervals were calculated for the area under the plasma drug concentration-time curve (AUC) and maximum plasma concentration (Cmax). Safety and tolerability were also assessed.
Results
Of 30 participants enrolled, 27 (90%) completed the study. Three participants discontinued due to positive urine drug screens. For atogepant, the median time to achieve maximum plasma concentration (Tmax) was delayed by 1.5 hours when coadministered with sumatriptan than when administered alone, while the mean apparent terminal half-life (t½) was generally similar (
Conclusion
Atogepant mean Cmax was reduced by 22% when coadministered with sumatriptan, while AUC remained unchanged. This change is not expected to be clinically relevant. The systemic exposure to sumatriptan was unchanged when coadministered with atogepant. Coadministration of atogepant with sumatriptan was safe and well tolerated, with no safety concerns identified.
Disclosure of Interest
R. Boinpally Conflict with: employee of AbbVie and may hold AbbVie stock., A. Jakate Conflict with: employee of AbbVie at the time of the study and may hold AbbVie stock., M. Butler Conflict with: employee of AbbVie and may hold AbbVie stock., A. Periclou Conflict with: employee of AbbVie at the time of the study and may hold AbbVie stock.
Mean (± SD) PK Parameters of Atogepant and Sumatriptan Following Single-Dose Oral Administration of Each Medication Alone or in Combination With the Other in Healthy Adult Participants

Median (range).
AUC, area under the plasma drug concentration-time curve from time 0 to time t; AUC0-∞, AUC from time 0 to infinity; CL/F, apparent total clearance of the drug from plasma after oral administration; Cmax, maximum plasma concentration; GMR, geometric means ratio; PK, pharmacokinetic; h, hour; SD, standard deviation; t½, terminal half-life; Tmax, time to achieve maximum plasma concentration; Vz/F, apparent volume of distribution during terminal phase after nonintravenous administration.
MTV20-DP-075
SINGLE THERAPEUTIC DOSES OF ATOGEPANT ARE NOT ASSOCIATED WITH A CLINICALLY RELEVANT DRUG-DRUG INTERACTION WHEN COADMINISTERED WITH ACETAMINOPHEN OR NAPROXEN
Ramesh Boinpally1*, Abhijeet Jakate1, Matthew Butler1 and Antonia Periclou1
1AbbVie, Madison, NJ, United States
Introduction
Atogepant is an oral calcitonin gene‒related peptide (CGRP) receptor antagonist in development for migraine prevention. If approved, atogepant may be coadministered with acetaminophen and naproxen, over-the-counter analgesics commonly used to treat headache pain.
Objectives
The goal of this study was to assess the drug-drug interaction potential of atogepant and acetaminophen or naproxen.
Methods
Pharmacokinetic (PK) interactions between atogepant 60 mg and acetaminophen 1000 mg or naproxen 500 mg were assessed in a phase 1, open-label, randomized, single-center, 5-way crossover study. Healthy adults aged 18–45 years were randomized to 1 of 10 sequences of 5 treatment periods—atogepant alone, acetaminophen or naproxen alone, and atogepant plus acetaminophen or naproxen (all single doses)—with 7-day washout between periods. For the comparison of each study medication in combination vs study medication alone, geometric mean ratios and 90% CIs were calculated for the area under the plasma drug concentration-time curve and maximum plasma concentration (Cmax). No statistically significant interaction was concluded if the 90% CIs for the ratios of the PK parameters of the drugs coadministered vs alone were within 80% to 125%. Safety and tolerability were also assessed.
Results
Of 40 participants enrolled, 35 (87.5%) completed the study. Three participants withdrew consent and 2 were lost to follow-up. Participants in the statistical analyses of the PK parameters were as follows: n = 38, atogepant alone; n = 39, acetaminophen alone; n = 37, coadministration of atogepant and acetaminophen; n = 36, naproxen alone; and n = 38, coadministration of atogepant and naproxen. Mean plasma atogepant PK parameters when administered with or without acetaminophen or naproxen are summarized in the
Conclusion
Coadministration of atogepant 60 mg and acetaminophen 1000 mg or naproxen 500 mg resulted in no statistically significant or clinically relevant changes in Cmax or overall systemic exposure to either drug. Coadministration of atogepant with either acetaminophen or naproxen was safe and well tolerated.
Disclosure of Interest
R. Boinpally Conflict with: employee of AbbVie and may hold AbbVie stock., A. Jakate Conflict with: employee of AbbVie at the time of the study and may hold AbbVie stock., M. Butler Conflict with: employee of AbbVie and may hold AbbVie stock., A. Periclou Conflict with: employee of AbbVie at the time of the study and may hold AbbVie stock.
Mean (± SD) Pharmacokinetic Parameters of Atogepant, Acetaminophen, or Naproxen Following Single-Dose Oral Administration of Each Medication Alone or in Combination With the Other in Healthy Adult Participants

Median (range).
AUC, area under the plasma drug concentration-time curve from time 0 to time t; AUC0-∞, AUC from time 0 to infinity; CL/F, apparent total clearance of the drug from plasma after oral administration; Cmax, maximum plasma concentration; GMR, geometric means ratio; h, hour; PK, pharmacokinetic; SD, standard deviation; t½, terminal half-life; tmax, time to achieve maximum plasma concentration; VZ/F, apparent volume of distribution during terminal phase after nonintravenous administration.
MTV20-DP-076
HEALTHCARE RESOURCE UTILIZATION AND HEALTH-RELATED QUALITY OF LIFE IN ADULT PATIENTS WITH CHRONIC MIGRAINE: RESULTS FROM THE PREDICT STUDY
Guy Boudreau1, Ian Finkelstein2, Corrie Graboski3, May Ong-Lam4, Suzanne Christie5, Katherine Sommer6*, Meetu Bhogal7, Goran Davidovic7 and Werner J. Becker8
1Centre Hospitalier Universitaire de Montréal (CHUM), Montréal, QC
2Toronto Headache & Pain Clinic, Toronto, ON
3Island Health, Brentwood Bay, BC
4St. Paul Hospital, Vancouver, BC
5University of Ottawa, Ottawa, ON, Canada
6Allergan, an AbbVie Company, Marlow, Buckinghamshire, United Kingdom
7Allergan, an AbbVie Company, Markham, ON
8University of Calgary, Calgary, AB, Canada
Introduction
Chronic migraine (CM) is a neurological disease associated with substantial individual, societal, and economic burden.
Objectives
The PREDICT (NCT02502123) study aimed to assess real-world, long-term health-related quality of life (HRQoL) in Canadian patients with CM treated with onabotulinumtoxinA. This analysis evaluates outcomes including HRQoL, healthcare resource utilization (HRU), and safety in the PREDICT study.
Methods
PREDICT was a 2-year, Canadian, single-arm, open-label, multicenter, post-authorization, prospective study in adults naïve to botulinum toxin(s) for CM. OnabotulinumtoxinA was administered per the Canadian product monograph. The primary endpoint was mean change in Migraine-Specific Quality of Life Questionnaire (MSQ) at treatment 4 vs baseline. HRU data were collected at baseline and at each treatment session.
Results
A total of 184 enrolled participants received ≥1 onabotulinumtoxinA treatment. Patients were, on average, aged 45 years, Caucasian, and 85% female; 60% had a family history of migraine. Statistically significant improvements from baseline were observed in MSQ domains (Restrictive, 21.5; Preventive, 19.5; Emotional, 22.9) after treatment 4 and at the final study visit (Restrictive, 21.3; Preventive, 19.2; Emotional, 27.4), all P < 0.0001 and exceeding minimal important differences (MID: Restrictive, 10.9; Preventive, 8.3; Emotional, 12.2). At baseline, 96.2% of patients reported an HA-related visit to an HCP within the past 6 months, and 17.9% reported an HA-related visit to an ER or urgent care clinic within the past 6 months, both of which decreased to 76.8% and 8.4% at the final study visit (2 years), respectively. HA-related hospital admissions within the past 6 months were reported by 3.8% of patients at baseline and only 1.4% at the final study visit. At baseline, the median (range) days for length of overnight hospital admissions for headache in the past 6 months was 2.5 (1.0–5.0; n = 6); this decreased to 1.0 (1.0–1.0; n = 1) at the final study visit. At baseline, 37.5% of patients reported any HA-related diagnostic testing within the past 6 months; this decreased to 9.9% at the final study visit. The most common diagnostic tests were blood tests, MRIs, and CAT/CT scans. A total of 168 TEAEs were reported by 41.8% of patients; of those, 38 were considered treatment related in 12.0% of patients. Six serious TEAEs were reported in 2.2% of patients; none was considered treatment related. No new safety signals were identified.
Conclusion
Real-world findings from the PREDICT study demonstrate that treatment with onabotulinumtoxinA in the Canadian population is associated with improvements in health-related quality of life and reductions in HRU.
Disclosure of Interest
G. Boudreau Conflict with: received consulting, honoraria, and/or research grants from AbbVie, Amgen, Lilly, and Teva., I. Finkelstein Conflict with: received grants/honoraria from AbbVie, Amgen, Eli Lilly, J&J, Merck, Purdue, Eva, and Tribute; was a speaker for AbbVie, Amgen, Eli Lilly, Teva, and Aralez; and received consulting fees from AbbVie, Amgen, Eli Lilly, Tribute, and Novartis., C. Graboski Conflict with: received research and education grants from AbbVie and speaking honoraria from Purdue, Lilly, Tribute, Genzyme, AbbVie, and Janssen., M. Ong-Lam Conflict with: received research support from AbbVie and Lilly., S. Christie Conflict with: received consulting fees, honoraria, and/or grants/research support from Biogen, Novartis, Lilly, Amgen, AbbVie, and Tribute., K. Sommer Conflict with: employee of AbbVie and may hold AbbVie stock., M. Bhogal Conflict with: employee of AbbVie and may hold AbbVie stock., G. Davidovic Conflict with: employee of AbbVie and may hold AbbVie stock., W. Becker Conflict with: received clinical trial research support from AbbVie and Amgen; speaker’s honoraria from AbbVie and Amgen; and consulting fees from AbbVie, Amgen, Lundbeck, Aralez, and Novartis.
MTV20-DP-077
ONABOTULINUMTOXINA TREATMENT IMPROVED HEALTH-RELATED QUALITY OF LIFE IN ADULTS WITH CHRONIC MIGRAINE: RESULTS FROM A PROSPECTIVE, OBSERVATIONAL STUDY (PREDICT)
Guy Boudreau1, Ian Finkelstein2, Corrie Graboski3, May Ong-Lam4, Suzanne Christie5, Katherine Sommer6, Meetu Bhogal7, Goran Davidovic7* and Werner J. Becker8
1Centre Hospitalier Universitaire de Montréal (CHUM), Montréal, QC
2Toronto Headache & Pain Clinic, Toronto, ON
3Island Health, Brentwood Bay, BC
4St. Paul Hospital, Vancouver, BC
5University of Ottawa, Ottawa, ON, Canada
6Allergan, an AbbVie Company, Marlow, Buckinghamshire, United Kingdom
7Allergan, an AbbVie Company, Markham, ON
8University of Calgary, Calgary, AB, Canada
Introduction
Chronic migraine (CM) can adversely affect health-related quality of life (HRQoL) and daily functioning, resulting in social and economic burdens.
Objectives
PREDICT aimed to assess real-world long-term HRQoL in Canadian patients with CM treated with onabotulinumtoxinA.
Methods
Two-year, Canadian, multicentre, prospective, observational study (NCT02502123) in adults naïve to botulinum toxin(s) for CM. 155–195 U onabotulinumtoxinA was recommended, per the Canadian product monograph, for injection every 12 weeks (≤7 treatments). Primary endpoint: mean change in Migraine-Specific Quality of Life (MSQ) at Tx4 vs. baseline. Secondary endpoints: mean change in MSQ at final visit vs. baseline, onabotulinumtoxinA treatment utilization, headache days, and moderate/severe days (daily headache diary). Unless noted, data presented as mean (SD).
Results
A total of 184 participants with an average age of 45 years, predominantly female (84.8%), and Caucasian (94.6%) received ≥1 treatment with onabotulinumtoxinA. Mean onabotulinumtoxinA dose per treatment cycle 171(18) U; mean treatment interval 13.2(1.8) weeks. At baseline, patients reported 20.9(6.7) headache days/month, which decreased over time by -3.5(6.3) days at Tx1, -6.5(6.6) days at Tx4, and -6.3(7.3) days at Tx7; all timepoints versus baseline, p < 0.0001. Moderate/severe headache days also decreased from 12.9(7.7) days/month at baseline by -3.4(7.1) days at Tx1, -5.6(7.1) days at Tx4, and -4.7(7.7) days at Tx7; all timepoints versus baseline, p < 0.0001. Significant increases in MSQ post-Tx4 (restrictive: 21.5, preventive: 19.5, emotional: 22.9) and at the final visit (restrictive: 21.3, preventive: 19.2, emotional: 27.4) were observed versus baseline, exceeding minimal clinically important differences (all, p < 0.0001). Overall, 77 patients (41.8%) reported 168 treatment-emergent adverse events (TEAEs); 38 TEAEs in 22 patients (12.0%) were considered treatment-related. 4 patients (2.2%) reported 6 serious TEAEs, none were considered treatment-related. No new safety signals were identified.
Conclusion
Real-world data from PREDICT demonstrate that long-term onabotulinumtoxinA treatment for CM reduced headache days and improved HRQoL. These results add to the body of evidence on the safety and effectiveness of onabotulinumtoxinA for CM.
Disclosure of Interest
G. Boudreau Conflict with: received consulting, honoraria, and/or research grants from AbbVie, Amgen, Lilly, and Teva., I. Finkelstein Conflict with: received grants/honoraria from AbbVie, Amgen, Eli Lilly, J&J, Merck, Purdue, Eva, and Tribute; was a speaker for AbbVie, Amgen, Eli Lilly, Teva, and Aralez; and received consulting fees from AbbVie, Amgen, Eli Lilly, Tribute, and Novartis., C. Graboski Conflict with: received research and education grants from AbbVie and speaking honoraria from Purdue, Lilly, Tribute, Genzyme, AbbVie, and Janssen., M. Ong-Lam Conflict with: received research support from AbbVie and Lilly., S. Christie Conflict with: received consulting fees, honoraria, and/or grants/research support from Biogen, Novartis, Lilly, Amgen, AbbVie, and Tribute., K. Sommer Conflict with: employee of AbbVie and may hold AbbVie stock., M. Bhogal Conflict with: employee of AbbVie and may hold AbbVie stock., G. Davidovic Conflict with: employee of AbbVie and may hold AbbVie stock., W. Becker Conflict with: received clinical trial research support from AbbVie and Amgen; speaker’s honoraria from AbbVie and Amgen; and consulting fees from AbbVie, Amgen, Lundbeck, Aralez, and Novartis.
MTV20-DP-078
PERSISTENCE TO ONABOTULINUMTOXINA AND CALCITONIN GENE–RELATED PEPTIDE MONOCLONAL ANTIBODIES (CGRP MABS) AMONG MIGRAINE PATIENTS IN A US ELECTRONIC HEALTH RECORD DATABASE
Amy Tung1*, Jamie Ta2,3, Alina N. Bogdanov4, Kevin Lavelle4 and Patrick Gillard2
1Allergan, an AbbVie Company, Irvine, CA, Canada
2Allergan, an AbbVie Company, Irvine, CA
3Comparative Health Outcomes, Policy, and Economics (CHOICE) Institute, University of Washington, Seattle, WA
4Veradigm, San Francisco, CA, United States
Introduction
Limited evidence exists for real-world treatment patterns of onabotulinumtoxinA and calcitonin gene‒related peptide (CGRP) monoclonal antibodies (mAbs) for migraine.
Objectives
Describe persistence among adults with migraine treated with onabotulinumtoxinA and CGRP mAbs.
Methods
The Practice Fusion ambulatory electronic health record database was used to identify adults ≥18 years with migraine initiating onabotulinumtoxinA or CGRP mAbs (erenumab, fremanezumab, or galcanezumab) between March 1, 2018 and September 22, 2019 (index date = earliest prescription date). Patients were required to have a migraine diagnosis prior to index and an office visit ≥6 months before and after the index date (or prescription ≥6 months after). Patients were excluded if they had a diagnosis for a nonmigraine onabotulinumtoxinA indication. Persistence at 6 months was defined as length of therapy without a ≥30-day gap. In addition, patients with an index prescription for a length of therapy ≥6 months were required to have an additional prescription or office visit with documentation of index medication up to 7 months post-index. Chi-squared tests were used to assess differences in persistence rates.
Results
Among 1976 patients meeting study criteria, 333 (17%) initiated onabotulinumtoxinA and 1643 (83%) initiated a CGRP mAb (erenumab: 1134 [57%]; fremanezumab: 298 [15%]; galcanezumab: 211 [11%]). Mean (SD) age was 48 (13) years and 86% were female. The most common comorbidities were anxiety (22%) and depression (19%). Most patients received an acute (54%) or preventive (57%) treatment in the pre-index period, most commonly triptans (39%) or anticonvulsants (36%), respectively. Most patients initiating onabotulinumtoxinA received prescriptions from neurologists and pain specialists (85%) compared to patients initiating CGRP mAbs (55%‒70%). More patients initiating onabotulinumtoxinA were persistent at 6 months (77%) compared with CGRP mAbs (erenumab: 49%; fremanezumab: 55%; galcanezumab: 45%; all P < 0.001). Similar trends were observed in patients with chronic migraine (CM; onabotulinumtoxinA: 81%; erenumab: 51%; fremanezumab: 58%; galcanezumab: 44% [all P < 0.001]) and episodic migraine (EM; onabotulinumtoxinA: 68%; erenumab: 46% [both P < 0.001]; fremanezumab: 53% [P>0.05]; galcanezumab: 46% [P < 0.05]).
Conclusion
In real-world practice, patients show higher levels of persistence on onabotulinumtoxinA compared to CGRP mAbs. This finding holds true among patients with CM and those with EM. Future research is warranted to confirm these findings over a longer period of follow-up.
Disclosure of Interest
A. Tung Conflict with: employee of AbbVie and may hold AbbVie stock., J. Ta Conflict with: supported by a training grant from Allergan, an AbbVie company, at the time the study was conducted., A. Bogdanov Conflict with: employee of Veradigm., K. Lavelle Conflict with: employee of Veradigm., P. Gillard Conflict with: employee of AbbVie and may hold AbbVie stock.
MTV20-DP-079
REAL-WORLD PERSISTENCE IN PATIENTS TREATED WITH ONABOTULINUMTOXINA AND CALCITONIN GENE–RELATED PEPTIDE MONOCLONAL ANTIBODIES (CGRP MABS) FOR MIGRAINE: A LARGE US ADMINISTRATIVE CLAIMS DATABASE STUDY
Jamie Ta1*,2, Amy Tung1, Weiying Wang3, David Oliveri3 and Patrick Gillard1
1Allergan, an AbbVie Company, Irvine, CA
2Comparative Health Outcomes, Policy, and Economics (CHOICE) Institute, University of Washington, Seattle, WA
3Genesis Research LLC, Hoboken, NJ, United States
Introduction
Limited evidence exists for real-world treatment patterns of onabotulinumtoxinA and calcitonin gene‒related peptide (CGRP) monoclonal antibodies (mAbs) for migraine.
Objectives
Describe characteristics and persistence in adults with migraine treated with onabotulinumtoxinA and CGRP mAbs.
Methods
A retrospective cohort study used administrative claims data from the IBM® MarketScan® Commercial and Medicare Supplemental databases from July 1, 2017 to October 31, 2019. Adults ≥18 years were included if they had ≥1 claim for onabotulinumtoxinA or CGRP mAb (erenumab, fremanezumab, or galcanezumab; index date = date of first treatment claim), an ICD-10-CM diagnosis code for migraine (G43.xx) on or 6 months prior to index date, and continuous medical and pharmacy coverage for 6 months pre- and post-index. Patients with a diagnosis for an onabotulinumtoxinA indication other than migraine at any time were excluded. Baseline demographic and clinical characteristics were described in the 6-month pre-index period. Persistence was defined as length of therapy without a ≥30-day gap and was assessed at 6 months (post-index). Chi-squared or Fisher’s exact tests were used to assess differences in persistence rates.
Results
Among 8593 patients who met all inclusion criteria, 3110 (36%) initiated onabotulinumtoxinA and 5483 (64%) initiated a CGRP mAb (erenumab: 2955 [54%]; fremanezumab: 1197 [22%]; galcanezumab: 1331 [24%]). Mean (SD) age was 44 (12) years and most were female (86%). The most common comorbidities were anxiety (24%), depression (18%), and hypertension (18%). In the pre-index period, triptans (64%) and NSAIDs (32%) were the most commonly used acute medications, while antidepressants (57%) and anticonvulsants (51%) were the most commonly used preventive agents. Overall, more patients were persistent to onabotulinumtoxinA at 6 months (65%) compared to CGRP mAbs (erenumab: 42%; fremanezumab: 48%; galcanezumab: 51%; all P < 0.001). Persistence rates for onabotulinumtoxinA were higher compared to CGRP mAbs in patients with chronic migraine (CM; onabotulinumtoxinA: 66%; erenumab: 40%; fremanezumab: 47%; galcanezumab: 49%; all P < 0.001) and episodic migraine (EM; onabotulinumtoxinA: 55%; erenumab: 44%; fremanezumab: 48%; galcanezumab: 52%; all P < 0.001).
Conclusion
Patients initiating onabotulinumtoxinA had better persistence compared to patients initiating CGRP mAbs for migraine. These results were observed in both CM and EM populations. Future research is warranted to confirm these findings as more long-term data become available.
Disclosure of Interest
J. Ta Conflict with: supported by a training grant from Allergan, an AbbVie company, at the time the study was conducted., A. Tung Conflict with: employee of AbbVie and may hold AbbVie stock., W. Wang Conflict with: employee of Genesis Research, which provides consulting services to Allergan, an AbbVie Company., D. Oliveri Conflict with: employee of Genesis Research, which provides consulting services to Allergan, an AbbVie Company., P. Gillard Conflict with: employee of AbbVie and may hold AbbVie stock.
MTV20-DP-080
EFFECTIVENESS OF ERENUMAB AND ONABOTULINUMTOXINA ON ACUTE MEDICATION USAGE AND HEALTHCARE RESOURCE UTILIZATION AS MIGRAINE PREVENTION IN THE UNITED STATES
Stewart J. Tepper1*, Juanzhi Fang2, Pamela Vo3, Ahmad Abdrabboh2, Ying Shen4, Lujia Zhou4, Mrudula Glassberg2 and Matias Ferraris3
1Geisel School of Medicine at Dartmouth, Hanover, NH
2Novartis Pharmaceuticals Corporation, East Hanover, NJ, United States
3Novartis Pharma AG, Basel, CH-4002, Switzerland
4KMK Consulting Inc., Morristown, NJ, United States
Introduction
Erenumab (erenumab-aooe in the US; Aimovig®), a calcitonin gene-related peptide (CGRP) receptor antagonist, was approved by FDA for the preventive treatment of migraine in adults in May 2018. Although pivotal trials have established potential clinical efficacy and safety of erenumab in placebo-controlled studies, it is necessary to have an in-depth understanding of real-world comparative effectiveness of erenumab.
Objectives
To evaluate the effectiveness of erenumab and onabotulinumtoxinA (onabot) on acute medication usage and health care resource use (HCRU) among migraine patients in the US.
Methods
Adult migraine patients initiating erenumab or onabot between 1 May, 2018 and 30 September, 2019 were identified from the Optum’s de-identified Clinformatics® Data Mart Database. Index date was the date of first erenumab or onabot claim. Erenumab and onabot cohorts were matched 1:1 using the propensity score (PS) method with stratification. Use of any acute medications, HCRU and a composite endpoint of emergency room (ER), in-patient (IP), and outpatient (OP) visits with acute medication use during the 6-month post index period were evaluated for both cohorts with two models: the PS matching and the inverse probability of treatment weighting (IPTW) models with baseline covariates adjustment. Logistic regression was used for dichotomous variables, and a negative binomial model was used for count variables, with odds ratios (OR) or rate ratios (RR) and 95% confidence intervals (CI) calculated.
Results
Data for 1,338 matched patients from each cohort (erenumab N = 3,171, onabot N = 3,100) were analyzed. PS matching with baseline covariates adjustment model showed that at 6 months, use of acute medications and HCRU were significantly lower for erenumab users compared with onabot users for most of endpoints (Table 1). For the composite outcome, a significantly lower proportion of patients with any of the three events was also observed (31.7% vs. 44.3%; OR [95% CI]: 0.59 [0.49-0.70], P < 0.0001). The same was true for the mean number of events (0.44 vs. 0.69; RR [95% CI]: 0.63 [0.56-0.71], P < 0.0001). IPTW with covariates adjustment model showed consistent results.
Conclusion
This retrospective study highlights that erenumab significantly reduces acute medication use and HCRU among migraine patients in the real world, greatly reducing the burden of disease. The magnitude of reductions was higher in erenumab than onabot-treated patients in this population and time period.
Disclosure of Interest
S. Tepper Conflict with: Alder, Allergan, Amgen, Dr. Reddy’s, ElectroCore, Eli Lilly, eNeura, Neurolief, Novartis, Scion Neurostim, Teva, Zosano, Conflict with: Consultant and/or Advisory Boards (honoraria): Acorda, Alder, Alexsa, Align Strategies, Allergan, Alphasights, Amgen, Aperture Venture Partners, Aralez Pharmaceuticals Canada, Axsome Therapeutics, Becker Pharmaceutical Consulting, BioDelivery Sciences International, Biohaven, Charleston Labs, Decision Resources, DeepBench, ElectroCore, Eli Lilly, eNeura, Equinox, ExpertConnect, GLG, GSK, Guidepoint Global, Healthcare Consultancy Group, Health Science Communications, Impel, Lundbeck, M3 Global Research, Magellan Rx Management, Marcia Berenson Connected Research and Consulting, Medicxi, Navigant Consulting, Neurolief, Nordic BioTech, Novartis, Pfizer, Pulmatrix, Reckner Healthcare, Relevale, Revance, SAI MedPartners, Satsuma, Scion Neurostim, Slingshot Insights, Sorrento, Spherix Global Insights, Sudler and Hennessey, Synapse Medical Communications, Teva, Theranica, Thought Leader Select, Trinity Partners, XOC, Zosano; Salary: Dartmouth-Hitchcock Medical Center, American Headache Society; Stock options: Nocira, Percept; CME honoraria: American Academy of Neurology, American Headache Society, Cleveland Clinic Foundation, Diamond Headache Clinic, Elsevier, Forefront Collaborative, Hamilton General Hospital, Ontario, Canada, Headache Cooperative of New England, Henry Ford Hospital, Detroit, Inova, Medical Learning Institute Peerview, Miller Medical Communications, North American Center for CME, Physicians’ Education Resource, Rockpointe, WebMD/Medscape, J. Fang Conflict with: Employed by, and own stocks in, Novartis, P. Vo Conflict with: Employed by, and own stocks in, Novartis, A. Abdrabboh Conflict with: Employed by, and own stocks in, Novartis, Y. Shen Conflict with: Employed by KMK Consulting Inc., L. Zhou Conflict with: Employed by KMK Consulting Inc., M. Glassberg Conflict with: Employed by, and own stocks in, Novartis, M. Ferraris Conflict with: Employed by, and own stocks in, Novartis

MTV20-DP-081
EFFICACY AND IMPACT ON ACUTE MEDICATION USE AND DISABILITY WITH FREMANEZUMAB IN PATIENTS WITH MIGRAINE WHO HAD INADEQUATE RESPONSE TO ≥3 PRIOR MIGRAINE PREVENTIVE MEDICATION CLASSES: RESULTS OF THE RANDOMIZED, DOUBLE-BLIND PHASE 3B FOCUS STUDY
Giorgio Lambru1*, Dalbir Dhiraj2, Xiaoping Ning3, Verena Ramirez Campos3, Evelyn Du3, Joshua M. Cohen3 and Manjit Matharu4
1The Headache Centre, Guy’s and St Thomas NHS Foundation Trust, London, United Kingdom
2Teva UK Limited, Harlow, United Kingdom
3Teva Branded Pharmaceutical Products R&D, Inc., West Chester, PA, United States
4Headache and Facial Pain Group, UCL Queen Square Institute of Neurology and The National Hospital for Neurology and Neurosurgery, Queen Square, London, United Kingdom
Introduction
The FOCUS study evaluated fremanezumab, a fully-humanized monoclonal antibody (IgG2Δa) that selectively targets calcitonin gene-related peptide (CGRP), for preventive treatment of episodic (EM) and chronic migraine (CM) in patients (pts) with documented inadequate responses to 2–4 classes of migraine preventive medications.
Objectives
To evaluate the efficacy of fremanezumab in a subgroup of EM and CM pts with inadequate response to ≥3 prior preventive medication classes (≥3 inadequate responses subgroup).
Methods
Pts were randomized (1:1:1) to quarterly (qtly) fremanezumab (Month 1/2/3: 675mg/placebo ([PBO]/PBO), monthly (mtly) fremanezumab (Month 1/2/3: 675mg [CM], 225mg [EM]/225mg/225mg), or matched mtly PBO for 12 weeks. Changes from baseline (BL) in mtly average migraine days (MMDs) and mtly days of acute headache medication use were evaluated in the EM and CM ≥3 inadequate responses subgroups. In addition, post hoc analyses examined disability outcomes (Migraine Disability Assessment [MIDAS] for EM pts; 6-item Headache Impact Test [HIT-6] for CM pts) in the ≥3 inadequate responses subgroups.
Results
Of the 838 pts randomized, 127 and 293 pts were in the EM and CM ≥3 inadequate response subgroups, respectively. In the EM ≥3 inadequate responses subgroup, changes from BM in MMDs over 12 weeks were significantly greater with fremanezumab (LSMD[SE] vs PBO: qtly, −4.0[0.86] and mtly, −4.0[0.82]; P < 0.0001); significant reductions were observed in mtly days of acute medication use (qtly, −3.8[0.77] and mtly, −3.9[0.73]; P < 0.0001) and MIDAS scores (qtly, −18.8[4.66]; mtly, −18.6[4.43]; P < 0.0001). In the CM ≥3 inadequate responses subgroup, changes from BL in MMDs over 12 weeks were significantly greater with fremanezumab (LSMD[SE] vs PBO: qtly, −3.0[0.65]; mtly, −3.1[0.64]; P < 0.0001); and significant reductions were observed in mtly days of acute medication use (qtly, −3.2[0.59] and mtly, −3.4[0.58]; P < 0.0001) and HIT-6 scores (qtly, −2.2[0.83]; mtly, −3.3[0.83]; P < 0.01).
Conclusion
Based on this subgroup analysis, fremanezumab is effective in patients with EM and CM and inadequate response to ≥3 migraine preventive medication classes, with significant reductions in MMDs, acute medication use, and disability score versus PBO.
Disclosure of Interest
G. Lambru Conflict with: received speaker honoraria, funding for travel, and honoraria for participation in advisory boards by Allergan, Novartis, Teva Pharmaceuticals, Eli Lilly, electroCore, Nevro Corp., and Autonomic Technologies, D. Dhiraj Conflict with: employee of Teva Pharmaceuticals, X. Ning Conflict with: employee of Teva Pharmaceuticals, V. Ramirez Campos Conflict with: employee of Teva Pharmaceuticals, E. Du Conflict with: employee of Teva Pharmaceuticals, J. Cohen Conflict with: employee of Teva Pharmaceuticals, M. Matharu Conflict with: serves on the advisory board for Allergan, Eli Lilly, Medtronic, Novartis, Salvia, and Teva Pharmaceuticals; and has received payment for the development of educational presentations from Allergan, Eli Lilly, Medtronic, Novartis, Salvia, Teva Pharmaceuticals, and electroCore
MTV20-DP-082
SUSTAINED CLINICAL BENEFITS FOLLOWING ONABOTULINUMTOXINA TREATMENT IN PATIENTS WITH CHRONIC MIGRAINE: A POST HOC ANALYSIS OF THE POOLED PREEMPT STUDIES
David W. Dodick1, Hans-Christoph Diener2, Stephen D. Silberstein3, Larisa Yedigarova4* and Richard B. Lipton5
1Mayo Clinic, Phoenix, AZ, United States
2University of Duisburg-Essen, Essen, Germany
3Thomas Jefferson University, Philadelphia, PA
4Allergan, an AbbVie Company, Irvine, CA
5Albert Einstein College of Medicine, Bronx, NY, United States
Introduction
We evaluated the sustained benefits of onabotulinumtoxinA treatment in patients with chronic migraine (CM) through 56 weeks.
Objectives
The goal of the study was to evaluate the sustained benefits of onabotulinumtoxinA treatment on clinically meaningful reductions in headache days and headache impact in patients with chronic migraine (CM) through 56 weeks.
Methods
Post hoc analysis of pooled data from the 24-week, placebo-controlled PREEMPT (NCT00156910, NCT00168428) clinical trials, including the 32-week open-label phase, during which all patients received onabotulinumtoxinA. The proportions of patients treated with onabotulinumtoxinA who achieved clinically meaningful improvement at week 24 (primary endpoint; double-blind period) in headache day reductions (≥50% or ≥75% from baseline) and 6-item Headache Impact Test score (HIT-6; ≥5-point improvement) were calculated. A sustained responder was defined as a patient who experienced clinically meaningful improvement at week 24 that continued through 56 weeks of treatment. Only patients who achieved a clinically meaningful change at week 24 and had ≥1 available assessment after 24 weeks were included in this analysis.
Results
A total of 1384 patients were randomized to onabotulinumtoxinA (n = 688) or placebo (n = 696) in the PREEMPT trials; only those treated with onabotulinumtoxinA were included in this analysis. At week 24, 254/539 (47%) patients experienced a ≥50% reduction in headache days; 163 of the 254 (64%) sustained benefits for the remaining 32 weeks of the study. A ≥75% reduction in headache days was experienced by 123/539 (23%) patients at week 24, and 69 of the 123 (56%) sustained the reduction for the remaining 32 weeks of the study. At week 24, 248/599 (41%) patients experienced a clinically meaningful improvement on HIT-6; 169 of the 248 (68%) sustained benefits over the remaining 32 weeks.
Conclusion
In the PREEMPT trials, a majority of patients treated with onabotulinumtoxinA experienced clinically meaningful improvements at week 24 that were sustained throughout the entire 56-week treatment period with regard to headache days and headache impact.
Disclosure of Interest
D. Dodick Conflict with: Personal fees: Amgen, AEON, Association of Translational Medicine, University Health Network, Daniel Edelman Inc., Autonomic Technologies, Axsome, AbbVie, Alder BioPharmaceuticals, Biohaven, Charleston Laboratories, Clexio, Dr. Reddy’s Laboratories/Promius, Electrocore LLC, Eli Lilly, eNeura, Neurolief, Novartis, Ipsen, Impel, Satsuma, Supernus, Sun Pharma (India), Theranica, Teva, Vedanta, WL Gore, Nocira, PSL Group Services, XoC, Zosano, ZP Opco, Foresite Capital, Oppenheimer; Upjohn (Division of Pfizer), Pieris, Revance, Equinox, Salvia, and Amzak Health. Speaking fees: Eli Lilly, Novartis Canada, Amgen, and Lundbeck. CME fees or royalty payments: HealthLogix, Medicom Worldwide, MedLogix Communications, Mednet, Miller Medical, PeerView, WebMD Health/Medscape, Chameleon, Academy for Continued Healthcare Learning, Universal Meeting Management, Haymarket, Global Scientific Communications, Global Life Sciences, Global Access Meetings, Catamount, UpToDate (Elsevier), Oxford University Press, Cambridge University Press, and Wolters Kluwer Health; Stock options: Precon Health, Aural Analytics, Healint, Theranica, Second Opinion/Mobile Health, Epien, Nocira, Matterhorn, Ontologics, and King-Devick Technologies; Consulting without fee: Aural Analytics, Healint, Second Opinion/Mobile Health, and Epien; Board of Directors: Precon Health, Epien, Matterhorn, Ontologics, and King-Devick Technologies. Patent: 17189376.1–1466:vTitle: Botulinum Toxin Dosage Regimen for Chronic Migraine Prophylaxis without fee; Research funding: American Migraine Foundation, US Department of Defense, PCORI, and Henry Jackson Foundation; Professional society fees or reimbursement for travel: American Academy of Neurology, American Brain Foundation, American Headache Society, American Migraine Foundation, International Headache Society, and Canadian Headache Society., H.-C. Diener Conflict with: received honoraria for participation in clinical trials, contribution to advisory boards or oral presentations from: Alder, AbbVie, Amgen, Autonomic Technology, Bristol-Myers Squibb, CoLucid, Electrocore, Ipsen Pharma, Lilly, Medtronic, MSD, Novartis, Pfizer, Sanofi, Schaper and Brümmer, Teva, and Weber & Weber. Financial support for research projects was provided by AbbVie, Electrocore, MSD, and Pfizer. Headache research at the Department of Neurology in Essen is supported by the German Research Council (DFG), the German Ministry of Education and Research (BMBF), and the European Union. HCD has no ownership interest and does not own stocks of any pharmaceutical company. HCD serves on the editorial boards of Cephalalgia and Lancet Neurology. HCD chairs the Clinical Guidelines Committee of the German Society of Neurology and is a member of the Clinical Trials Committee of the International Headache Society., S. Silberstein Conflict with: consultant and/or advisory panel member for and has received honoraria from Alder Biopharmaceuticals, AbbVie, Amgen, Avanir, eNeura, ElectroCore Medical, Labrys Biologics, Medscape, Medtronic, Neuralieve, NINDS, Pfizer, and Teva. His employer receives research support from AbbVie, Amgen, Cumberland Pharmaceuticals, ElectroCore Medical, Labrys Biologics, Eli Lilly, Mars, and Troy Healthcare., L. Yedigarova Conflict with: employee of AbbVie and may own stock in AbbVie., R. Lipton Conflict with: serves on the editorial boards of Neurology and Cephalalgia and as senior advisor to Headache. He has received research support from the NIH. He also receives support from the Migraine Research Foundation and the National Headache Foundation. He has reviewed for the NIA and NINDS; serves as consultant, advisory board member, or has received honoraria from Alder, AbbVie, Amgen, Autonomic Technologies, Avanir, Boston Scientific, Dr. Reddy’s, ElectroCore, Eli Lilly, eNeura Therapeutics, GlaxoSmithKline, Merck, Novartis, Teva, and Vedanta. He receives royalties from Wolff’s Headache, 8th Edition (Oxford University Press, 2009), and Informa. He holds stock options in eNeura Therapeutics and Biohaven.
MTV20-DP-083
IMPACT OF FREMANEZUMAB ON DISABILITY SEVERITY IN PATIENTS WITH EPISODIC AND CHRONIC MIGRAINE: POOLED RESULTS OF 3 RANDOMIZED, DOUBLE-BLIND, PLACEBO-CONTROLLED PHASE 3 STUDIES
Abraham J. Nagy1*, Peter McAllister2, Joshua M. Cohen3, Xiaoping Ning3, Verena Ramirez Campos3, Evelyn Du3 and Messoud Ashina4
1Nevada Headache Institute, Las Vegas, NV, United States
2New England Institute for Neurology and Headache, Stamford, CT, United States
3Teva Branded Pharmaceutical Products R&D, Inc., West Chester, PA, United States
4Danish Headache Center, Department of Neurology, Rigshospitalet, Glostrup, Denmark
Introduction
Migraine is a leading cause of disability worldwide. Fremanezumab, a fully-humanized monoclonal antibody (IgG2Δa) has demonstrated efficacy in migraine prevention. Guidelines for determining response to CGRP monoclonal antibodies for migraine were provided in the American Headache Society (AHS) Consensus Statement, which include measuring disability with validated patient-reported outcomes.
Objectives
This pooled analysis evaluated disability response to fremanezumab using the Migraine Disability Assessment (MIDAS) and the 6-item Headache Impact Test (HIT-6).
Methods
This analysis of fremanezumab in subjects with episodic (EM) or chronic migraine (CM) included data from 3 double-blind phase 3 trials (HALO EM, HALO CM, and FOCUS), in which subjects were randomized 1:1:1 to subcutaneous quarterly or monthly fremanezumab or matched placebo for 12 weeks. MIDAS and HIT-6 were used to assess subjects in HALO EM/FOCUS and HALO CM/FOCUS, respectively. Measurements of disability response were defined per AHS guidelines: for MIDAS, a reduction of ≥5 points when baseline score is 11–20 or ≥30% when baseline score is >20; for HIT-6, a reduction of ≥5 points. Proportions of subjects who demonstrated shifts in severity for each disability outcome were also evaluated.
Results
For subjects with a baseline MIDAS score of 11–20, the proportion achieving a 5-point reduction from baseline in MIDAS scores was significantly higher with fremanezumab (quarterly, 71%; monthly, 70%) versus placebo (49%; P < 0.01 for both). For subjects with a baseline MIDAS score >20, the proportion achieving a ≥30% reduction in points was significantly higher with fremanezumab (quarterly, 69%; monthly, 79%) versus placebo (58%; P < 0.001 for both). The proportion of subjects achieving a 5-point reduction from baseline in HIT-6 scores was also significantly higher with fremanezumab (quarterly, 53%; monthly, 55%) versus placebo (39%; P < 0.0001 for both). Likewise, the proportion of subjects who experienced a shift of 1, 2, or 3 grades down in disability severity, according to MIDAS or HIT-6 scores, were significantly greater with quarterly and monthly fremanezumab versus placebo (all P < 0.0001).
Conclusion
This pooled analysis demonstrated that fremanezumab treatment resulted in clinically meaningful improvements in headache- and migraine-related disability severity, further supporting the efficacy of fremanezumab in subjects with difficult-to-treat migraine.
Disclosure of Interest
A. Nagy Conflict with: received research support from Amgen/Novartis, Lilly, Teva Pharmaceuticals, Allergan, Zosano, and Alder Pharmaceuticals; serves as a consultant for Lilly, Teva Pharmaceuticals, Zosano, Electrocore, Biohaven, and Alder, P. McAllister Conflict with: received research support from and serves as a consultant for Alder Pharmaceuticals, Amgen, Eli Lilly, Novartis, and Teva Pharmaceuticals, J. Cohen Conflict with: employee of Teva Pharmaceuticals, X. Ning Conflict with: employee of Teva Pharmaceuticals, V. Ramirez Campos Conflict with: employee of Teva Pharmaceuticals, E. Du Conflict with: employee of Teva Pharmaceuticals, M. Ashina Conflict with: received personal fees from Allergan, Amgen, Biohaven, Eli Lilly, Lundbeck, Novartis and Teva. Dr. Ashina is currently the principal investigator for Allergan, Amgen, Eli Lilly, Lundbeck and Novartis trials. Dr. Ashina serves as associate editor of Cephalalgia, associate editor of Headache, and associate editor of the Journal of Headache and Pain. Dr. Ashina is President of the International Headache Society
MTV20-DP-084
EFFICACY AND SAFETY OF FREMANEZUMAB IN PATIENTS WITH MIGRAINE AND INADEQUATE RESPONSE TO MULTIPLE MIGRAINE PREVENTIVE MEDICATION CLASSES DUE TO LACK OF EFFICACY OR POOR TOLERABILITY
Abraham J. Nagy1*, Verena Ramirez Campos2, Evelyn Du2, Joshua M. Cohen2, Maja Galic3, Xiaoping Ning2 and Ladislav Pazdera4
1Nevada Headache Institute, Las Vegas, NV, United States
2Teva Branded Pharmaceutical Products R&D, Inc., West Chester, PA, United States
3Teva Pharmaceuticals, Amsterdam, Netherlands
4Vestra Clinics, Rychnov nad Kněžnou, Jiráskova, Czech Republic
Introduction
Fremanezumab, a fully-humanized monoclonal antibody (IgG2Δa) that selectively targets calcitonin gene-related peptide (CGRP), has demonstrated efficacy for migraine prevention in adults with documented inadequate response (lack of efficacy, tolerability, or contraindicated/not suitable) to 2–4 migraine preventive medication classes in the phase 3b FOCUS study.
Objectives
This analysis evaluated efficacy of fremanezumab in subjects with inadequate response to ≥2 preventive medications, with ≥1 inadequate response due to lack of efficacy (lack of efficacy subgroup) and safety in subjects with ≥1 inadequate response due to poor tolerability (poor tolerability subgroup).
Methods
In the 12-week, double-blind period of FOCUS, subjects were randomized (1:1:1) to quarterly fremanezumab (Month 1/2/3: 675mg/placebo[PBO]/PBO), monthly fremanezumab (Month 1/2/3: 675mg[CM], 225mg[EM]/225mg/225mg), or matched PBO. Monthly migraine days (MMDs) and proportions of subjects with ≥50% reduction in MMDs (≥50% responders) were assessed in the lack of efficacy subgroup overall and by prior therapeutic class. Adverse events (AEs) were assessed in the poor tolerability subgroup.
Results
Of 838 subjects randomized, 741 and 448 were included in the lack of efficacy and poor tolerability subgroups, respectively. In the lack of efficacy subgroup, changes from baseline in average MMDs over 12 weeks were significantly greater with quarterly and monthly fremanezumab, respectively, vs PBO (least-squares mean difference vs PBO: −3.1 and −3.4; P < 0.0001), as were ≥50% responder rates at 12 weeks (34% and 34% vs 9%; P < 0.0001). Across all prior preventive medication classes in lack of efficacy subgroup, changes from baseline in MMDs and ≥50% responder rates were significantly greater with fremanezumab vs PBO (P < 0.05). For subjects in the poor tolerability subgroup, ≤1% across all treatment groups had AEs leading to discontinuation or serious AEs. The most common AEs were injection-site erythema, injection-site induration, nasopharyngitis, and insomnia.
Conclusion
In EM or CM subjects with inadequate response to ≥2 prior preventive medications, including ≥1 inadequate response due to lack of efficacy, fremanezumab demonstrated efficacy vs PBO, regardless of prior therapeutic class. In subjects with ≥1 inadequate response due to poor tolerability, fremanezumab demonstrated similar, low incidences of AEs leading to discontinuation and serious AEs vs PBO.
Disclosure of Interest
A. Nagy Conflict with: received research support from Amgen/Novartis, Lilly, Teva Pharmaceuticals, Allergan, Zosano, and Alder Pharmaceuticals; and serves as a consultant for Lilly, Teva Pharmaceuticals, Zosano, Electrocore, Biohaven, V. Ramirez Campos Conflict with: employee of Teva Pharmaceuticals, E. Du Conflict with: employee of Teva Pharmaceuticals, J. Cohen Conflict with: employee of Teva Pharmaceuticals, M. Galic Conflict with: employee of Teva Pharmaceuticals, X. Ning Conflict with: employee of Teva Pharmaceuticals, L. Pazdera Conflict with: received consultation fees from GL Pharma, Mylan, Sandoz and Vipharm; educational grants and support from Vipharm, GP Pharma and Mylan; honoraria and speaking fees for symposia and other events sponsored by Astellas, Lundbeck, Teva, Eisai and GL Pharma; served as Principal Investigator for more than 50 Sponsor-initiated studies; was a National Coordinator and Key Opinion Leader for Astellas; and an expert witness and consultant for lawsuits, providing deposition and testimony including analysis of medical documents.
MTV20-DP-085
REAL-LIFE USE OF ONABOTULINUMTOXINA FOR SYMPTOM RELIEF IN PATIENTS WITH CHRONIC MIGRAINE: REPOSE STUDY GERMAN POPULATION
Katja Kollewe1, Charly Gaul2, Astrid Gendolla3 and Katherine Sommer4*
1Hannover Medical School, Hannover
2Migraine and Headache Clinic, Königstein
3Praxis fur Neurologie, Essen, Germany
4Allergan, an AbbVie Company, Marlow, Buckinghamshire, United Kingdom
Introduction
In Germany, chronic migraine (CM) has been associated with substantial disability and increased healthcare resource utilization (HRU) and costs.
Objectives
To assess healthcare resource utilization and patient-reported reductions in headache days in patients treated with onabotulinumtoxinA for CM in a sample of German patients from the REPOSE study.
Methods
REPOSE, a 2-year, prospective, noninterventional, observational, open-label study, described real-world use of onabotulinumtoxinA in adults with CM. Patients received onabotulinumtoxinA ∼every 12 weeks. Patient-estimated mean headache-day frequency in the last month was collected at each treatment visit. HRU data, including family doctor or specialist visits, inpatient acute treatment, acute treatment for headache, acupuncture, technical investigations (CT, MRI, X-ray, ECG, ultrasound), and use of nonpharmacologic remedies, were collected at baseline (occurrence within prior 3 months) and at 6, 12, 18, and 24 months (events since prior visit).
Results
Of 641 enrolled patients, 633 received ≥1 onabotulinumtoxinA dose. Patients were, on average, aged 45 years, 85% were female, and 60% (n = 377) were from Germany. German patients had an average of 18.9 monthly headache days at baseline, declining to 8.6 at 12 months and 7.6 at 24 months; significant reductions were observed at all follow-up visits (range: 11.3–6.0; P < 0.0001). The percentage of patients who saw a family doctor declined from 41.7% at baseline to 12.2% at 12 months and 9.2% at 24 months. The percentage of patients who saw a specialist also declined from 61.7% at baseline to 8.9% at 12 months and 4.6% at 24 months. Inpatient acute treatment declined from 6.4% of patients at baseline to 0.8% at 12 months and 0.0% at 24 months. Acute treatment for headache also declined from 71.2% of patients at baseline to 65.9% at 12 months and 55.4% at 24 months. Use of acupuncture declined from 15.5% at baseline to a range of 2.2%–0.0%. Technical investigations and use of remedies decreased from 19.7% and 30.6% at baseline to ranges of 1.6%–0.0% and 15.4%–4.6%, respectively.
Conclusion
In German REPOSE participants, onabotulinumtoxinA treatment for CM is associated with reduced monthly headache days and decreased HRU. Due to drop-outs, a population of responders remained in later months of the study.
Disclosure of Interest
K. Kollewe Conflict with: received travel grants and honoraria for lectures from AbbVie, Biogen, Ipsen, Lilly, Merz, Novartis, and Teva., C. Gaul Conflict with: received honoraria for lectures or advisory boards from AbbVie, TEVA-Ratiopharm, Boehringer Ingelheim Pharma, Lilly, Novartis Pharma, Desitin Arzneimittel, Cerbotec, Bayer vital, Hormosan Pharma, Grüenthal, and Reckitt Benckiser. CG does not hold stocks of any pharmaceutical or medical device companies., A. Gendolla Conflict with: received fees for lectures and consulting in the last 3 years from AbbVie, Reckitt Benckiser, St. Jude Medical, Bayer, Grünenthal, Mundipharma, Zahnärztekammer KVNO, GAF, Novartis, Lilly, and Teva., K. Sommer Conflict with: employee of AbbVie and may own stock in AbbVie.
MTV20-DP-086
CARDIOVASCULAR SAFETY OF FREMANEZUMAB IN PATIENTS WITH MIGRAINE AND CARDIOVASCULAR MEDICAL HISTORY OR RISK FACTORS: A POOLED ANALYSIS OF PHASE 3 STUDIES
Stephanie J. Nahas1*, Tim P. Jurgens2, Yoel Kessler3, Xiaoping Ning3, Joshua M. Cohen3, Verena Ramirez Campos3, Evelyn Du3 and Stephen D. Silberstein1
1Jefferson Headache Center, Thomas Jefferson University, Philadelphia, PA, United States
2Dept. of Neurology, Headache Center North-East, University Medical Center of Rostock, Rostock, Germany
3Teva Branded Pharmaceutical Products R&D, Inc., West Chester, PA, United States
Introduction
Understanding cardiovascular (CV) safety of medications targeting the calcitonin gene-related peptide (CGRP) pathway is important given the vasodilatory properties of CGRP. Fremanezumab, a fully-humanized monoclonal antibody (IgG2Δa) that selectively targets CGRP, has proven efficacy for the preventive treatment of migraine in adults in three phase 3 studies.
Objectives
To evaluate the CV safety of fremanezumab in patients with episodic migraine (EM) and chronic migraine (CM) based on CV medical history and CV/cerebrovascular risk factors (CVRFs).
Methods
This analysis of CV safety of fremanezumab included data from the 2 HALO studies (1 in EM, 1 in CM) and the FOCUS study in patients with EM or CM who had inadequate response to 2–4 classes of migraine preventive medications. In all 3 trials, patients were randomized 1:1:1 to quarterly fremanezumab (Month 1/2/3: 675mg/placebo [PBO]/PBO), monthly fremanezumab (Month 1/2/3: 675mg [CM], 225mg [EM]/225mg/225mg), or matched PBO for 12 weeks. CV adverse events (CVAEs) were evaluated in patients with and without CV medical history and by number of CVRFs at baseline.
Results
Among patients with CV medical history (fremanezumab, n = 325; PBO, n = 153), CVAEs occurred in similar, low proportions of patients across treatment groups (3%–6%); the most common CVAE was hypertension (0%–2%). Among patients without CV medical history (fremanezumab, n = 1,572; placebo, n = 792), CVAEs also occurred in similar, low proportions of patients across treatment groups (1%–2%); the most common CVAE was hypertension (0.2%–0.6%). In total, 499 of 2,842 pooled patients had ≥2 CVRFs (0 CVFRs, n = 1,350; ≥1 CVRF, n = 1,492; ≥2 CVRFs, n = 499; ≥3 CVRFs, n = 183, ≥4 CVRFs, n = 55); of these, 66% had CV medical history. CVAEs were infrequent in patients with ≥2 or ≥3 CVRFs (0%–2%); no CVAEs were reported in patients with ≥4 CVRFs. No CV safety signals were identified.
Conclusion
This pooled analysis demonstrates a favorable CV safety profile for fremanezumab in patients with CV medical history or with ≥2 CVRFs. No CV safety signals were identified. These data support the strong safety profile of this preventive treatment targeting the CGRP pathway.
Disclosure of Interest
S. Nahas Conflict with: serves on the advisory board for Allergan, Amgen/Novartis, Biohaven, Eli Lilly, Impel, Supernus, Teva, Theranica, and Zosano; serves as a consultant for Allergan, Amgen/Novartis, Biohaven, Eli Lilly, Impel, Supernus, Teva, Theranica, Zosano; serves as a contributing author for Springer and Woulters-Klouwer; and has received speaker honoraria for Amgen/Novartis, electroCore, Eli Lilly, Supernus, and Teva Pharmaceuticals, T. Jurgens Conflict with: received honoraria for participation in advisory boards or oral presentations from Allergan, Hormosan Pharma, Lilly, Novartis, Sanofi, and Teva Pharmaceuticals, Y. Kessler Conflict with: employee of Teva Pharmaceuticals, X. Ning Conflict with: employee of Teva Pharmaceuticals, J. Cohen Conflict with: employee of Teva Pharmaceuticals, V. Ramirez Campos Conflict with: employee of Teva Pharmaceuticals, E. Du Conflict with: employee of Teva Pharmaceuticals, S. Silberstein Conflict with: received honoraria as a consultant and/or advisory panel member of Abide Therapeutics, Alder Biopharmaceuticals, Allergan, Inc., Amgen, Avanir Pharmaceuticals, Inc., Biohaven Pharmaceuticals, Cefaly, Curelator, Inc., Dr. Reddy’s Laboratories, Egalet Corporation, GlaxoSmithKline Consumer Health Holdings, LLC., eNeura Inc., electroCore Medical, LLC, Impel NeuroPharma, Inc., Lilly USA, LLC, Medscape, LLC, Novartis, Inc., Satsuma Pharmaceuticals, Supernus Pharmaceuticals, Inc., Teva Pharmaceuticals, Theranica, and Trigemina, Inc.
MTV20-DP-087
REAL-WORLD EVIDENCE FOR CONTROL OF PATIENTS WITH CHRONIC MIGRAINE WHO RECEIVED CGRP MONOCLONAL ANTIBODY THERAPY ADDED TO ONABOTULINUMTOXINA TREATMENT
Andrew M. Blumenfeld1*, Benjamin M. Frishberg1, Jack D. Schim1, Ashley Iannone2, Gary Schneider2, Larisa Yedigarova3 and Aubrey Manack Adams3
1Headache Center of Southern California, The Neurology Center, Carlsbad, CA
2ICON plc, Boston, MA
3Allergan, an AbbVie Company, Irvine, CA, United States
Introduction
Concomitant use of onabotulinumtoxinA and monoclonal antibodies (mAbs) directed against the calcitonin gene–related peptide (CGRP) or its receptor has the potential to be more effective than monotherapy for chronic migraine (CM) prevention.
Objectives
To collect real-world data to gain improved understanding of the safety, tolerability, and potential benefits of adding a CGRP mAb to onabotulinumtoxinA treatment in patients with CM.
Methods
This retrospective, longitudinal chart review included adults with CM treated at 1 clinical site (October 2018–November 2019) with at least 2 consecutive cycles of onabotulinumtoxinA prior and at least 1 month of onabotulinumtoxinA and CGRP mAb (erenumab, fremanezumab, or galcanezumab) concomitant treatment. Charts at the time of first CGRP mAb prescription (index date) and up to 4 visits at approximately 3, 6, 9, and 12 months post-index were reviewed for safety and tolerability (adverse events [AEs], discontinuations) and outcome measures (headache day frequency, intensity, migraine-related disability, headache impact, and depression).
Results
Of 300 charts reviewed, 257 patients met eligibility criteria (mean age: 50 years; 82% women). The most frequently prescribed CGRP mAb was erenumab (78%), followed by galcanezumab (16%) and fremanezumab (6%). Patients discontinued the CGRP mAb more often than onabotulinumtoxinA (23% vs 3%). AEs were reported in 28% of patients; the most common AE was constipation (9%). The average headache day frequency was 21.5 days/month before initiation of onabotulinumtoxinA and 12.1 days/month before starting CGRP mAb as add-on therapy. After approximately 6 months of concomitant use, positive changes from baseline were noted: 82% of patients had decreased headache day frequency, 70% had reduced headache intensity, and 57% had improved migraine-related disability. Statistically significant decreases in headache day frequency from baseline occurred at all visits, with clinically significant mean decreases of between 3.5 and 4.0 days/month following 6, 9 and 12 months of concomitant treatment.
Conclusion
In this real-world study of CM patients, clinically meaningful treatment benefits were demonstrated with onabotulinumtoxinA alone with additive benefits observed after add-on therapy with a CGRP mAb. Concomitant treatment with onabotulinumtoxinA and CGRP mAbs was well tolerated with no new safety signals identified. Additional real-world and prospective, randomized-controlled trials should be considered to further quantify the benefits of this multimodal treatment paradigm for those with CM.
Disclosure of Interest
A. Blumenfeld Conflict with: served on advisory boards for Aeon, AbbVie, Amgen, Alder, Biohaven, Teva, Supernus, Promius, Eaglet, and Lilly; and has received funding for speaking from AbbVie, Amgen, Pernix, Supernus, Depomed, Avanir, Promius, Teva, Eli Lilly and Company, Lundbeck, Novartis, and Theranica., B. Frishberg Conflict with: received compensation for speaking from Teva, Lilly, Biohaven, Amgen, Novartis, and AbbVie, and served on advisory boards for Lundbeck, Upsher-Smith, and Theranica., J. Schim Conflict with: served on advisory boards for Aeon, AbbVie, Amgen, Biohaven, electroCore, Impel, Lilly, Lundbeck, Novartis, Promius, Revance, Teva, and Upsher-Smith; and has received compensation for speaking from AbbVie, Amgen, Biohaven, electroCore, Lilly, Lundbeck, Novartis, Promius, Teva, and Upsher-Smith., A. Iannone Conflict with: employee of ICON plc., G. Schneider Conflict with: prior employee of ICON plc., L. Yedigarova Conflict with: employee of AbbVie and may hold AbbVie stock., A. Manack Adams Conflict with: employee of AbbVie and may hold AbbVie stock.
MTV20-DP-088
IMPACT OF ERENUMAB ON ACUTE MEDICATION USAGE AND HEALTHCARE RESOURCE UTILIZATION AMONG MIGRAINE PATIENTS: A US CLAIMS DATABASE STUDY
Stewart J. Tepper1*, Juanzhi Fang2, Pamela Vo3, Ahmad Abdrabboh2, Matias Ferraris3, Ying Shen4, Lujia Zhou4 and Mrudula Glassberg2
1Geisel School of Medicine at Dartmouth, Hanover, NH
2Novartis Pharmaceuticals Corporation, East Hanover, NJ, United States
3Novartis Pharma AG, Basel, CH-4002, Switzerland
4KMK Consulting Inc., Morristown, NJ, United States
Introduction
Erenumab (erenumab-aooe in the U.S.; Aimovig®), a calcitonin gene-related peptide (CGRP) receptor antagonist, was approved by FDA for the preventive treatment of migraine in adults in May 2018. Although pivotal trials have adequately established potential clinical efficacy and safety of erenumab in controlled studies, it is necessary to have an in-depth understanding of the real-world effectiveness of erenumab.
Objectives
To evaluate real-world impact of erenumab on acute medication usage and health care resource use (HCRU) among migraine patients in the US.
Methods
Adult migraine patients initiating erenumab between 1 May, 2018 and 30 September, 2019 were identified from US Optum’s de-identified Clinformatics® Data Mart Database. Index date was defined as the date of first erenumab claim, and patients who received any other anti-CGRP drugs during the 12-month pre-index or 6-month post index period were excluded. Use of acute medications overall and at different drug class level, and HCRU were compared during 6 months pre vs. post index period. Impact of erenumab on a
Results
Data for 3,171 patients were analyzed (mean [SD] age: 50.74 [13.60] years, 84.8% female, and 62.5% with chronic migraine without aura). Following initiation of erenumab, use of acute medications and HCRU decreased significantly at 6 months (Table 1). For the composite outcome, a significant reduction in proportion of patients with any of the three events was also observed (52.7% vs. 39.5%, P < 0.0001). Moreover, the mean (SD) number of events decreased from 1.03 (1.53) to 0.77 (1.48) (rate ratio: 0.75, 95% CI: 0.71 to 0.79, P < 0.0001).
Conclusion
This retrospective study highlights that erenumab significantly reduces acute medication use and HCRU in real-world setting, hence significantly reducing the burden of the disease. A composite endpoint could be used as a proxy to evaluate migraine attacks, while further research is needed to validate the algorithm.
Disclosure of Interest
S. Tepper Conflict with: Alder, Allergan, Amgen, Dr. Reddy’s, ElectroCore, Eli Lilly, eNeura, Neurolief, Novartis, Scion Neurostim, Teva, Zosano, Conflict with: Consultant and/or Advisory Boards (honoraria): Acorda, Alder, Alexsa, Align Strategies, Allergan, Alphasights, Amgen, Aperture Venture Partners, Aralez Pharmaceuticals Canada, Axsome Therapeutics, Becker Pharmaceutical Consulting, BioDelivery Sciences International, Biohaven, Charleston Labs, Decision Resources, DeepBench, ElectroCore, Eli Lilly, eNeura, Equinox, ExpertConnect, GLG, GSK, Guidepoint Global, Healthcare Consultancy Group, Health Science Communications, Impel, Lundbeck, M3 Global Research, Magellan Rx Management, Marcia Berenson Connected Research and Consulting, Medicxi, Navigant Consulting, Neurolief, Nordic BioTech, Novartis, Pfizer, Pulmatrix, Reckner Healthcare, Relevale, Revance, SAI MedPartners, Satsuma, Scion Neurostim, Slingshot Insights, Sorrento, Spherix Global Insights, Sudler and Hennessey, Synapse Medical Communications, Teva, Theranica, Thought Leader Select, Trinity Partners, XOC, Zosano; Salary: Dartmouth-Hitchcock Medical Center, American Headache Society; Stock options: Nocira, Percept; CME honoraria: American Academy of Neurology, American Headache Society, Cleveland Clinic Foundation, Diamond Headache Clinic, Elsevier, Forefront Collaborative, Hamilton General Hospital, Ontario, Canada, Headache Cooperative of New England, Henry Ford Hospital, Detroit, Inova, Medical Learning Institute Peerview, Miller Medical Communications, North American Center for CME, Physicians’ Education Resource, Rockpointe, WebMD/Medscape, J. Fang Conflict with: Employed by, and own stocks in, Novartis, P. Vo Conflict with: Employed by, and own stocks in, Novartis, A. Abdrabboh Conflict with: Employed by, and own stocks in, Novartis, M. Ferraris Conflict with: Employed by, and own stocks in, Novartis, Y. Shen Conflict with: Employed by KMK Consulting Inc., L. Zhou Conflict with: Employed by KMK Consulting Inc., M. Glassberg Conflict with: Employed by, and own stocks in, Novartis

MTV20-DP-089
PATIENT PERCEPTIONS OF THE IMPACT AND BURDEN OF MIGRAINE: AN INTERNATIONAL SURVEY OF 12,545 PATIENTS ACROSS 16 COUNTRIES
Dimos Dimitrios Mitsikostas1*, Oved Daniel2, Joshua M. Cohen3, Tamar Lengil3, Leonidas Lyras3, Matthias Dusselier3, Magali Geens3 and Hans-Christoph Diener4
1First Neurology Department, Aeginition Hospital, Medical School, National and Kapodistrian University of Athens, Athens, Greece
2Headache & Facial Pain Clinic, Ramat Aviv Medical Center, Ramat Aviv, Israel
3Teva Branded Pharmaceutical Products R&D, Inc., West Chester, PA, United States
4Universitätsklinikum Essen, Essen, Denmark
Introduction
billion people worldwide. As the second leading cause of years lived with disability, migraine is associated with a substantial personal burden for patients, their families, social circles, and employers.
Objectives
To evaluate patient perceptions of the impact and burden of migraine, as well as patient perceptions of migraine diagnosis and treatment, stigma, and migraine awareness and support, across 16 countries worldwide.
Methods
This was a brief digital survey (approximately 12 minutes in duration) that was completed by adult patients (≥18 years of age) diagnosed with migraine who self-reported ≥4 days of migraine per month. Data were collected between November and December of 2019 across 16 countries in Europe, South America, Asia, and Australia.
Results
A total of 12,545 patients completed this survey. The majority of patients were 25 to 54 years of age (74%) and women (73%). A total of 70% of patients reported that their migraine symptoms had progressed over time. Only 59% of patients reported being satisfied with their prescription treatment for migraine, while 61% of patients reported being satisfied with the physician treating their migraine. Patients most commonly reported that migraine impacted the following areas of their lives: overall health and wellbeing (69%), social life (60%), work/career (56%), or relationship with family (39%). Patients reported that the following people in their lives were the most impacted by their migraine: their partner (68%), children (55%), parents (22%), employer (22%), friends (19%), and other family members (19%). Almost half of patients (46%) reported having hidden their migraine, most commonly from their employer (63%), family (49%), or friends (37%). Patients (44%) felt that the healthcare community could play the greatest role in raising awareness of migraine.
Conclusion
These survey results from a large population of patients worldwide indicate that migraine has a substantial negative impact on patients’ lives and their familial, social, and professional environment and represents a substantial burden across many life roles.
Disclosure of Interest :
D. D. Mitsikostas Conflict with: D.D. Mitsikostas has received honoraria, research and travel grants from Allergan, Amgen, Biogen, Cefaly, Eli Lilly, electroCore, Mertz, Novartis, Roche, Sanofi, Specifar and Teva Pharmaceuticals., O. Daniel Conflict with: Oved Daniel has received honoraria, research and travel grants from Eli Lilly, Novartis, and Teva Pharmaceuticals., J. Cohen Conflict with: J.M. Cohen is an employee of Teva Pharmaceuticals., T. Lengil Conflict with: T. Lengil is an employee of Teva Pharmaceuticals., L. Lyras Conflict with: L. Lyras is an employee of Teva Pharmaceuticals., M. Dusselier Conflict with: M. Dusselier is an employee of InSites Consulting, and worked on the current analyses, funded by Teva Pharmaceuticals., M. Geens Conflict with: M. Geens is an employee of InSites Consulting, and worked on the current analyses, funded by Teva Pharmaceuticals., H.-C. Diener Conflict with: H.C. Diener has received honoraria for participation in clinical trials, contribution to advisory boards, or oral presentations from Alder, Allergan, Amgen, Electrocore, Ipsen, Lilly, Medtronic, Novartis, Pfizer, Teva and Weber & Weber.
MTV20-DP-090
COMORBIDITIES IN LOW AND HIGH FREQUENCY EPISODIC AND CHRONIC MIGRAINE: A SUBGROUP ANALYSIS OF THE BECOME STUDY
David Watson1*, Charly Gaul2, Christian Lucas3, Patricia Pozo-Rosich4, Paolo Martelletti5, Shannon Ritter6 and Josefin Snellman7
1Hamilton Medical Group, Aberdeen, Scotland, United Kingdom
2Migraine and Headache Clinic Königstein, Königstein im Taunus, Germany
3Pain Clinic, Service de Neurochirurgie, Hô pital Salengro, Lille Cedex, France
4Vall d’Hebron University Hospital; Vall d’Hebron Institute of Research, Universitat Autònoma de Barcelona, Barcelona, Spain
5Sapienza University of Rome, Sant’Andrea Hospital, Via di Grottarossa, Rome, Italy
6Novartis Pharmaceuticals Corporation, East Hanover, NJ, United States
7Novartis Pharma AG, Basel, Switzerland
Introduction
BECOME is the largest real-world cross-sectional study on prevalence and burden of migraine from patients visiting specialist headache centres in Europe and Israel.
Objectives
In the sub-analysis of BECOME we examined a broad range of comorbidities associated with migraine such as musculoskeletal and connective tissue disorders, vascular disorders, endocrine disorders, nervous system disorders and psychiatric disorders (anxiety and/or depression disorders) in the subgroup of patients with low- and high-frequency episodic and chronic migraine (EM and CM).
Methods
BECOME was conducted in two concurrent parts: Part 1, cumulative hospital data and Part 2, patient-level data. In the sub-analysis of Part 2 we examined the effect of comorbidities in patients with migraine. Patients with migraine (EM and CM) were grouped based on low- and high-frequency of monthly migraine days (MMD): LFEM (4–7 MMD), HFEM (8–14 MMD), LFCM (≥15 MHD with 8–14 MMD), and HFCM (≥15 MHD with ≥15 MMD).
Image:

Results
Overall, 2419 patients (mean age: 43 years) were included in the Part 2 analysis, and majority were women (86.9%). The proportion of patients with comorbidities was greater in the CM subgroups, with an average of 1.6 (HFCM) and 1.4 (LFCM) comorbidities per patient and 1.1 each in the LFEM and HFEM subgroups. Psychiatric disorders (n = 181[7.5%]) were the most common comorbidities in the HFCM subgroup compared with other three subgroups (Table). The most frequent psychiatric disorder across all four subgroups was depression (followed by anxiety and insomnia), with the highest prevalence among patients in the HFCM subgroup than other three subgroups (depression: n = 111[4.6%]) (Table).
Conclusion
In migraine, comorbidities per patient increased with increase in disease severity; and as the severity of disease increased the prevalence of psychiatric comorbidities also increased, with depression being the most frequent and highly prevalent in the HFCM subgroup of patients in particular.
Disclosure of Interest
D. Watson Conflict with: Received honoraria from Novartis, Teva and Allergan in the last 12 months for consultancy and educational work., C. Gaul Conflict with: Received honoraria for consulting and lectures within the past 3 years from Allergan Pharma, Ratiopharm, Boehringer Ingelheim Pharma, Eli Lilly, Novartis Pharma, Desitin Arzneimittel, Cerbotec, Bayer Vital, Hormosan Pharma, electroCore, Grünenthal, Reckitt Benckiser, and Teva. He does not hold any stocks of pharmaceutical companies or medical device companies., C. Lucas Conflict with: Collaboration as an expert, investigator or coordinator of clinical trials with Novartis, Teva, Sanofi, Grünenthal, Eli Lilly, Biogen, and Ethypharm., P. Pozo-Rosich Conflict with: Her research group has received research grants from Allergan and funding for clinical trials from Alder, Boehringer Ingelheim, MSD, electroCore, Eli Lilly, Janssen Cilag, and Novartis., Conflict with: Received honoraria as a consultant and speaker during the last 5 years from Allergan, Almirall, Chiesi, Eli Lilly, Novartis and Teva. She is a trustee member of the board of the International Headache Society and a Member of the Council of the European Headache Federation. She is on the editorial board of Revista de Neurologia. She is an editor for Frontiers of Neurology and The Journal of Headache and Pain. She is a member of the Clinical Trials Guidelines Committee of the International Headache Society. She has edited the Guidelines for the Diagnosis and Treatment of Headache of the Spanish Neurological Society. She does not own stocks from any pharmaceutical company., P. Martelletti Conflict with: Section Editor, Medicine, Springer Nature Comprehensive Clinical Medicine; Editor-in-Chief, The Journal of Headache and Pain; Headache Books Series Editor, Springer; EU Expert, European Medicine Agency. Past-President of European Federation, Chairman of School of Advanced Studies of European Headache Federation. He does not hold any stocks of any pharmaceutical companies or medical device companies., S. Ritter Conflict with: Employee and stocks: Novartis, J. Snellman Conflict with: employee and stocks: Novartis
MTV20-DP-091
BURDEN OF COMORBID DEPRESSION AND ANXIETY ON MIGRAINE-SPECIFIC HEALTH-RELATED QUALITY OF LIFE AND WORK PRODUCTIVITY IN ADULT MIGRAINE PATIENTS IN THE UNITED STATES
Richard B. Lipton1,2*, Ravi Iyer3, Joshua M. Cohen3, James Jackson4, Verena Ramirez Campos3, Sarah Cotton4, Gary Milligan4 and Dawn C. Buse1
1Albert Einstein College of Medicine, Bronx, New York
2Montefiore Medical Center, Bronx, New York, United States
3Teva Branded Pharmaceutical Products R&D, Inc., West Chester, PA, United States
4Adelphi Real World, Bollington, Cheshire, United Kingdom
Introduction
Depression and anxiety are comorbid with migraine and are associated with negative outcomes, including reduced health-related quality of life (HRQoL) and increased disease burden.
Objectives
To examine relationships with comorbid depression and/or anxiety on migraine-specific HRQoL outcomes and work productivity in adult migraine patients (pts) in the US.
Methods
Data were drawn from the 2017 US Adelphi Migraine Disease Specific Programme. Pts completed validated questionnaires, including the Migraine Disability Assessment Scale (MIDAS), the Migraine-Specific Quality of Life Questionnaire (MSQ), work productivity and activity impairment (WPAI), and the EuroQol 5-Dimension 5-Level (EQ-5D-5L). Depression/anxiety was derived from an EQ-5D domain, and categorized as none, slight, or moderate-to-severe (moderate+) depression/anxiety. Multivariate regressions were conducted on outcomes that included number of monthly headache days (MHDs), depression/anxiety, and potential confounding factors, including age, gender, body mass index, time since diagnosis, and selected comorbidities. Linear regression analyses were performed on the EQ-5D visual analogue scale (VAS) and MSQ domains to assess relationships between anxiety/depression severity and MHDs on these HRQoL outcomes. Logistic regression was run on WPAI absenteeism dichotomised yes/no variable. Poisson regressions were performed using the square root of MIDAS as the outcome and on the WPAI overall and presenteeism (impairment in work) domains to assess relationship between anxiety/depression severity and MHDs on work productivity outcomes.
Results
873 pts provided depression/anxiety information. Activity impairment was assessed for all pts (n = 801 responded) and work productivity for employed pts (n = 613 responded). Worse anxiety/depression and increasing number of MHDs were associated with poorer MSQ scores (P < 0.01); with more severe disability, based on MIDAS scores (P < 0.001); and with significantly greater overall activity and work impairment and presenteeism on the WPAI (P < 0.05). Higher anxiety/depression scores were associated with reductions in overall HRQoL, based on EQ-5D VAS scores (P < 0.001). Worsening anxiety/depression and increasing numbers of HDs were not significantly associated with an increase in work time missed (P>0.05).
Conclusion
As MHDs and anxiety/depression increase, HRQoL declines and productivity losses (absenteeism and presenteeism) rise.
Disclosure of Interest
R. Lipton Conflict with: Edwin S. Lowe Professor of Neurology at the Albert Einstein College of Medicine in New York. Dr. Lipton receives research support from the NIH: 2PO1 AG003949 (mPI), 5U10 NS077308 (PI), RO1 NS082432 (Investigator), 1RF1 AG057531 (Site PI), RF1 AG054548 (Investigator), 1RO1 AG048642 (Investigator), R56 AG057548 (Investigator), K23 NS09610 (Mentor), K23AG049466 (Mentor), 1K01AG054700 (Mentor). Dr. Lipton also receives support from the Migraine Research Foundation and the National Headache Foundation. Dr. Lipton serves on the editorial board of Neurology, senior advisor to Headache, and associate editor to Cephalalgia. Dr. Lipton has reviewed for the NIA and NINDS, holds stock options in eNeura Therapeutics and Biohaven Holdings; serves as consultant, advisory board member, or has received honoraria from: American Academy of Neurology, Alder, Allergan, American Headache Society, Amgen, Autonomic Technologies, Avanir, Biohaven, Biovision, Boston Scientific, Dr. Reddy’s, Electrocore, Eli Lilly, eNeura Therapeutics, GlaxoSmithKline, Merck, Pernix, Pfizer, Supernus, Teva, Trigemina, Vector, Vedanta. Dr. Lipton receives royalties from Wolff’s Headache 7th and 8th Edition, Oxford Press University, 2009, Wiley and Informa, R. Iyer Conflict with: employee of Teva Pharmaceuticals, J. Cohen Conflict with: employee of Teva Pharmaceuticals, J. Jackson Conflict with: employee of Adelphi Real World, and worked on the current analyses, funded by Teva Pharmaceuticals. D.C., V. Ramirez Campos Conflict with: employee of Teva Pharmaceuticals, S. Cotton Conflict with: employee of Adelphi Real World, and worked on the current analyses, funded by Teva Pharmaceuticals. D.C., G. Milligan Conflict with: employee of Adelphi Real World, and worked on the current analyses, funded by Teva Pharmaceuticals. D.C., D. Buse Conflict with: received grant support and honoraria from Allergan, Amgen, Avanir, Biohaven, Lilly, Promeius and Teva. D.C. Buse is on the editorial board of Current Pain and Headache Reports.
MTV20-DP-092
NUMBER NEEDED TO TREAT/HARM FOR FREMANEZUMAB IN PATIENTS WHO HAD INADEQUATE RESPONSE TO 2–4 PRIOR MIGRAINE PREVENTIVE MEDICATION CLASSES
Messoud Ashina1*, Joshua M. Cohen2, Maja Galic3, Verena Ramirez Campos2, Evelyn Du2, Xiaoping Ning2 and Hans-Christoph Diener4
1Danish Headache Center, Department of Neurology, Rigshospitalet, Glostrup, Denmark
2Teva Branded Pharmaceutical Products R&D, Inc., West Chester, PA, United States
3Teva Pharmaceuticals, Amsterdam, Netherlands
4Universitätsklinikum Essen, Essen, Germany
Introduction
Fremanezumab, a fully-humanized monoclonal antibody (IgG2Δa) that selectively targets calcitonin gene-related peptide (CGRP), has demonstrated efficacy and safety in adults with episodic (EM) or chronic migraine (CM), with documented previous inadequate response to 2–4 classes of migraine preventive medications.
Objectives
This analysis determined the number needed to treat (NNT), number needed to harm (NNH), and likelihood of being helped or harmed (LHH) in the FOCUS study.
Methods
In the 12-week, double-blind, placebo (PBO)-controlled study, patients (pts) were randomized (1:1:1) to quarterly fremanezumab (Month 1/2/3: 675mg/PBO/PBO), monthly fremanezumab (Month 1/2/3: 675mg[CM], 225mg[EM]/225mg/225mg), or matched monthly PBO. NNT was based on responder analysis defined as ≥30% and ≥50% reduction in monthly average number of migraine days at 12 weeks for CM and EM patients, respectively. NNH was based on discontinuations due to adverse events (AEs); if NNH ≤0, it was imputed as 1000. LHH is an indirect measure of effect size and determined as NNH divided by NNT.
Results
Among CM pts (N = 509), response rates were: quarterly fremanezumab, 49% (83/169); monthly fremanezumab, 54% (93/173); PBO, 19% (32/167), and discontinuations due to AEs were: quarterly fremanezumab, 0; monthly fremanezumab, 2% (n = 3); PBO, < 1% (n = 1). These results translated to NNT of 3.3 and 2.9, NNH of 1000 and 71, and LHH of 300 and 25 for quarterly and monthly fremanezumab, respectively. Among EM pts (N = 328), response rates were: quarterly fremanezumab, 47% (50/107); monthly fremanezumab, 43% (47/110); PBO, 10% (11/111), and discontinuations due to AEs were < 1% (n = 1) each in all 3 groups. These results translated to NNT of 2.7 and 3.0, NNH of 1000 and 1000, and LHH of 370 and 330 for quarterly and monthly fremanezumab, respectively. Combined data for CM and EM pts translated to NNT of 3.0 and 2.9, NNH of 1000 and 333, and LHH of 330 and 113 for quarterly and monthly fremanezumab, respectively.
Conclusion
The NNT of 3.0 and 2.9, minimum NNH of 1000 and 333, and LHH of 330 and 113 for quarterly and monthly fremanezumab, respectively, compare favorably with those for topiramate (NNT = 4; NNH = 11 [100 mg] and 5 [200 mg]; LHH = 2.75 [100 mg] and 1.25 [200 mg] [Linde M et al. Cochrane Database Syst Rev. 2013]), valproate (NNT = 4 [Linde M et al. Cochrane Database Syst Rev. 2013]), and propranolol (NNT = 4.5 [Jackson JL, et al. PLoS One. 2019]).
Disclosure of Interest
M. Ashina Conflict with: serves as a consultant and/or scientific advisor for Alder BioPharmaceuticals, Allergan, Amgen, Biohaven, Eli Lilly, Lundbeck, Novartis, Teva Pharmaceuticals; a principal investigator for Alder, Allergan, Amgen, Eli Lilly, Novartis, Teva Pharmaceuticals; an associate editor for Cephalalgia and associate editor for Headache; and a co-editor for the Journal of Headache and Pain, J. Cohen Conflict with: employee of Teva Pharmaceuticals, M. Galic Conflict with: employee of Teva Pharmaceuticals, V. Ramirez Campos Conflict with: employee of Teva Pharmaceuticals, E. Du Conflict with: employee of Teva Pharmaceuticals, X. Ning Conflict with: employee of Teva Pharmaceuticals, H.-C. Diener Conflict with: received honoraria for participation in clinical trials, contribution to advisory boards, or oral presentations from Alder, Allergan, Amgen, Electrocore, Ipsen, Lilly, Lundbeck, Medtronic, Novartis, Pfizer, Teva and Weber & Weber. Electrocore provided financial support for research projects. The German Research Council (DFG), the German Ministry of Education and Research (BMBF) and the European Union support his headache research.
MTV20-DP-093
ECONOMIC IMPACT OF MIGRAINE IN EMPLOYED ADULT PATIENTS WITH MIGRAINE FROM THE UNITED STATES: RESULTS FROM THE MEDICAL EXPENDITURE PANEL SURVEY (MEPS)
Janet Ford1, Shivang Joshi2*, Jeffrey Scott Andrews1, Rohit Bhandari1, Rose Okonkwo1 and Wenyu Ye1
1Eli Lilly & Company Corporate Centre, Indianapolis
2DENT Neurologic Institute, Amherst, United States
Introduction
Migraine poses a high financial burden in patients with migraine.
Objectives
This study evaluated annual societal burden (direct + indirect costs) associated with migraine among employed adults.
Methods
MEPS panel data between 2012–2014 were included in patients with a migraine diagnosis (ICD-9 346) and non-migraine ages 18–65 years and who were employed. Continuous and categorical variables were summarized using descriptive statistics applying survey sample weights. Comparisons among total migraine cohort and two identified migraine subgroups, acute medication overuse (AMO; threshold indicative of insufficient treatment) and stigmatized-risk (concurrent anxiety/depression), versus non-migraine were conducted using propensity scores (PS) matching.
Results
The total study cohort was 16,495 employed participants; 556 in the migraine cohort (mean age: 39.5 years; women: 81.6%) and 15,939 (mean age: 40.7 years; women: 44.6%) in the non-migraine cohort. After PS matching, the annualized societal burden was significantly higher for patients with migraine (total and subgroups) versus non-migraine, mean difference: total migraine, $3392; AMO, $7859; stigmatized-risk, $5031 (Table 1).
Conclusion
Migraine is associated with substantially higher annualized societal cost versus patients without migraine; which is notably higher for specific subgroups.
Disclosure of Interest
J. Ford Conflict with: Janet Ford is an employee and a minor stockholder of Eli Lilly and Company, S. Joshi Conflict with: Shivang Joshi served on the advisory board of Eli Lilly and Company, J. S. Andrews Conflict with: Jeffrey Scott Andrews is an employee and a minor stockholder of Eli Lilly and Company, R. Bhandari Conflict with: Rohit Bhandari is an employee and a minor stockholder of Eli Lilly and Company, R. Okonkwo Conflict with: Rose Okonkwo is an employee and a minor stockholder of Eli Lilly and Company, W. Ye Conflict with: Wenyu Ye is an employee and a minor stockholder of Eli Lilly and Company
Annual societal burden of migraine among employed adults (propensity score matched population with ratio 1:5)

≤0.0001; ***≤0.001; **≤0.01; *≤0.05; All values are weighted mean (95% confidence interval)
Stigmatized-risk constitutes patients who are at greater risk of stigma due to their neurological disease, i.e., patients with a concurrent diagnoses of anxiety and/or depression
MTV20-DP-094
EFFECTIVENESS OF DUAL MIGRAINE THERAPY WITH CGRP ANTAGONISTS AND ONABOTULINUMTOXINA INJECTIONS: EXPERIENCE FROM A SINGLE MIGRAINE CENTER IN HAWAII
Tahlia Toni1*,2, Rayce Tamanaha1,3, Bashak Newman1,4, Yutong Liang1,5, James Lee1,6, Enrique Carrazana1,7, Vimala Vajjala1, Jason Viereck1,7, Kore Kai Liow1,7
1Headache & Facial Pain Center, Hawaii Pacific Neuroscience, Honolulu
2Indiana University, Bloomington
3Gonzaga University, Spokane
4University of South Florida, Tampa
5Brown University, Providence
6Dartmouth College, Hanover
7John A. Burns School of Medicine, University of Hawaii, Honolulu, United States
Introduction
Calcitonin gene-related peptide (CGRP) antagonists are a novel class of drugs for migraine prevention, but clinical trials excluded the use of onabotulinumtoxinA. Therefore, there is no clear evidence demonstrating a synergistic effect, nor is there evidence on the safety of the combined therapies.
Objectives
Our study aims to examine the effectiveness of anti-CGRP monoclonal antibodies (mAbs) with onabotulinumtoxinA by evaluating migraine reductions in headache days and severity.
Methods
A retrospective chart review was conducted at a migraine center in Hawaii to identify patients who had a partial or poor response to onabotulinumtoxinA, and were placed on dual therapy with an anti-CGRP mAb: fremanezumab, erenumab, or galcanezumab. Patients’ initial headache days and severity ratings were recorded and compared to their final values taken 1–6 months after adding the CGRP antagonist to their treatment regime.
Results
Of 17 patients (16F/1M), n = 9 were taking fremanezumab, n = 4 were taking erenumab, and n = 4 were taking galcanezumab. The average age of onset in 16 of 17 patients was 45.8 years old, with n = 8 reporting childhood-onset (≤18 years) and n = 8 reporting adult-onset (≥19 years). Variables were recorded for anxiety (n = 4), depression (n = 4), and sleep disorders (bruxism n = 1, hypersomnia n = 1, insomnia n = 4). Patients experienced an initial average of 27.44 ± 4.78 headaches per month, which was reduced to 18.56 ± 9.37 headache days post-treatment. 10 patients experienced reductions in headache days, and 7 patients experienced no change in headache days. Patients also experienced an initial average pain level of 8.38 ± 1.39 out of 10, which was reduced to 5.52 ± 2.59 post-treatment. 11 patients improved, 4 patients experienced no change, and 2 patients worsened. No serious adverse side effects were reported from patients on dual therapy, only mild concerns.
Conclusion
A significant portion of patients with sub-optimal response to onabotulinumtoxinA may respond to dual therapy with CGRP antagonists. Placebo-controlled, randomized studies are suggested to corroborate and elaborate on this observation.
Disclosure of Interest
None Declared
MTV20-DP-095
A SURVEY ASSESSING THE BURDEN AND IMPACT OF MIGRAINE ACROSS 6 COUNTRIES IN SOUTH AMERICA, ASIA, AND AUSTRALIA
Marcio Nattan Portes Souza1, Joshua M. Cohen2, Tamar Lengil2, Matthias Dusselier3 and Magali Geens3
1Neurology Department, Hospital das Clínicas, Universidade de São Paulo, São Paulo, Brazil
2Teva Branded Pharmaceutical Products R&D, Inc., West Chester, PA, United States
3InSites Consulting, Ghent, Belgium
Introduction
Migraine is a common and disabling disease, affecting over 1 billion people worldwide. Migraine is associated with a substantial negative impact on patients’ day-to-day lives, careers, and relationships with family and friends.
Objectives
To evaluate the patient perception of the impact and burden of migraine, its diagnosis, treatment, stigma, as well as migraine awareness and support across six countries in South America, Asia, and Australia.
Methods
This was a digital survey (approximately 12 minutes in duration) completed by adult patients (≥18 years of age) who self-reported a diagnosis of migraine with ≥4 days of migraine per month. Data were collected between 19 November and 8 December 2019 across 6 countries: Australia, Russia, Argentina, Brazil, Israel, and Mexico.
Results
Across the 6 included countries, 5,024 patients completed this survey. Most patients were women (72%) and 25 to 44 years of age (65%). 71% of patients reported that their migraine symptoms had progressed over time. The proportion of patients who reported satisfaction with their prescription migraine treatment was 51% for preventive medications and 58% for acute medications. Overall, 60% of patients were satisfied with the physician treating their migraine, most commonly a general practitioner (45%), neurologist (39%), or family physician (18%). Although 75% of patients felt their physician understood their migraine condition well, 55% felt that healthcare professionals who are better educated about migraine would make the biggest difference to the migraine community. Patients most commonly reported that their overall health and wellbeing (73%), work/career (59%), and relationship with family (44%) were impacted by their migraine, with overall health and wellbeing cited as the most impacted aspect of life by 48%. Patients most commonly reported that their partner (66%) and children (54%) were most impacted by their migraine, followed by their parents (25%). Overall, 49% of patients reported having hidden their migraine, most commonly from their employer (62%), friends (37%), family (29%), partner (27%), or children (21%). Nearly half of the patients surveyed (47%) reported that the healthcare community should have the primary responsibility for highlighting the impact of migraine and supporting patients with migraine.
Conclusion
These survey results from a large population across six different countries in Asia, South America, and Australia indicate that the burden and impact of migraine on patients and their families, friends, and employers is substantial.
Disclosure of Interest
M. N. Portes Souza Conflict with: received honoraria for oral presentations from Teva, Lilly, Novartis, Allergan, and Libbs, J. Cohen Conflict with: employee of Teva Pharmaceuticals, T. Lengil Conflict with: employee of Teva Pharmaceuticals, M. Dusselier Conflict with: employee of InSites Consulting, and worked on the current analyses, funded by Teva Pharmaceuticals, M. Geens Conflict with: employee of InSites Consulting, and worked on the current analyses, funded by Teva Pharmaceuticals.
MTV20-DP-096
THE PATIENT-PERCEIVED BURDEN AND IMPACT OF MIGRAINE ACROSS 10 EUROPEAN COUNTRIES
Uwe Reuter1*, Joshua M. Cohen2, Leonidas Lyras2, Matthias Dusselier3, Magali Geens3 and Patricia Pozo-Rosich4
1Department of Neurology, Charité Universitätsmedizin Berlin, Berlin, Germany
2Teva Branded Pharmaceutical Products R&D, Inc., West Chester, PA, United States
3InSites Consulting, Ghent, Belgium
4Headache Unit, Neurology Department, Vall d’Hebron University Hospital, Barcelona, and Headache and Neurological Pain Research Group, Vall d’Hebron Institute of Research (VHIR), Universitat Autònoma de Barcelona, Barcelona, Spain
Introduction
Migraine is a common and disabling disease, with a substantial negative impact on patients’ health-related quality of life. Migraine may negatively affect patients’ lives and careers, as well as the lives of friends and family.
Objectives
To evaluate the patient-perceived impact and burden of migraine; patient perceptions of migraine diagnosis and treatment, stigma, and migraine awareness and support, across 10 European countries.
Methods
This was a 12-minute digital survey completed by adult patients (≥18 years of age) diagnosed with migraine who self-reported ≥4 days of migraine per month. Data were collected between 19 November and 6 December 2019 across 10 countries in Europe: Belgium, the Netherlands, France, Germany, Spain, the Czech Republic, Italy, Poland, Sweden, and the United Kingdom.
Results
A total of 7,521 patients across the 10 European countries completed this survey. The majority of patients were 25 to 54 years of age (70%) and women (73%). Nearly half (47%) of patients reported that their migraine diagnosis occurred 3 or more years after their initial symptoms, while 31% reported 3 or more years between diagnosis and receiving a prescription treatment for migraine. Only 58% of patients reported being satisfied with their prescription treatment for migraine, while 61% reported being satisfied with the physician treating their migraine. Patients most commonly reported that migraine impacted the following areas of their lives: overall health and wellbeing (65%), social life (61%), and work/career (54%). Overall, 69% and 57% of patients reported that their partner and children, respectively, were most impacted by their migraine. Almost half of patients (44%) reported having hidden their migraine, most commonly from their employer (63%). For information related to migraine, patients most commonly consulted their doctor (66%), search engines (39%), medical websites (37%), or a pharmacist (35%). Patients (41%) most commonly reported that the healthcare community was most responsible for raising awareness of the impact of migraine and providing patient support.
Conclusion
These survey results from a large European population indicate that the personal burden of migraine is substantial and that patients feel that their migraine has a negative impact on their own lives and careers, as well as the lives of their family and friends.
Disclosure of Interest:
U. Reuter Conflict with: received personal compensation from Allergan, Amgen, Eli Lilly, Medscape, StreaMedUp, Novartis, and Teva Pharmaceuticals, J. Cohen Conflict with: employee of Teva Pharmaceuticals, L. Lyras Conflict with: employee of Teva Pharmaceuticals, M. Dusselier Conflict with: employee of InSites Consulting, and worked on the current analyses, funded by Teva Pharmaceuticals, M. Geens Conflict with: employee of InSites Consulting, and worked on the current analyses, funded by Teva Pharmaceuticals, P. Pozo-Rosich Conflict with: received honoraria as a consultant and speaker for: Allergan, Almirall, Chiesi, Eli Lilly, Novartis and Teva Pharmaceuticals. P. Pozo-Rosich’s research group has received research grants from Allergan and has received funding for clinical trials from Alder, electroCore, Eli Lilly, Novartis and Teva Pharmaceuticals.
MTV20-DP-097
MIGRAINE ASSOCIATIONS WITH MYOFASCIAL PAIN, TREATMENT OUTCOME DEPENDS ON PATIENT`S PHENOTYPE
Rostyslav Bubnov1* and Olga Golubnitschaja2
1Diagnostic and interventional ultrasound, Clinical hospital “Pheophania”, Kyiv, Ukraine
2Predictive, Preventive and Personalised (3P) Medicine, Department of Radiation Oncology, Friedrich-Wilhelms University of Bonn, Bonn, Germany
Introduction
Myofascial trigger point (MTrP) is a pillar pathophysiological unit in development of myofascial pain and postural imbalance. Dry needling (DN) of MTrP under ultrasound (US) guidance is prioritized method for treatment myofascial pain. MTrP are spastic hypovascularized hypoxic low energy areas that can produce organismic hypoxic signaling, associated with patients` phenotype [5].
Objectives
Hypothesis: we hypothesize that muscle posture might have crucial effect on migraine; additionally to mechanical stimuli cumulative signaling of hypoxic MTrPs, stress can shorten periods of relapse. This effect likely more expressed in specific constitutional characteristics. The aim was to study the associations of myofascial pain, altered posture, activity of MTrP and FS phenotype with migraine activity.
Methods
We included 17 patients with cases of chronic headache (13 females, aged 23–56 y.o.). We retrospectively precisely analyzed course of disease, measured BMI and received responses on Flammre syndrome phenotype questionnaire. All patients underwent general exam, MRI, precise physical tests, extensive functional multiparameter neuromuscular US including M-mode, elastography (SWE), B-Flow (LOGIC E9 GE). Then patients received DN of detected MTrPs under US guidance: MTrPs were identified via clinical examination, referred pain pattern, US identification; single fine (28G) steel needle DN under US guidance was applied to elicit LTR and/or `needle grasp`. Outcome assessments were performed before, during and after treatment according to VAS and patients diary reports including the number of migraine days, attack duration, pain intensity, time of relapse and need the next DN session or use of analgesic medication. The total follow-up period was 6–12 months.
Results
Pain relevant to myofascial pain was diagnosed in all patients, migraines confirmed in 9 individuals, among them 7 patients demonstrated FS phenotype (15/15 positive responses); one patients had anomaly of cervical spine. We successfully detected MTrP as hypoechoic, stiff and hypovascular small areas with different patterns of decreasing motility. We found MTrP with more expressed hypovascular pattern, higher sensitivity and excessive responses (fasciculations and `needle grasp` over 30 min) in lean individuals and with FS phenotype and high activity of migraine. DN resulted immediately in decreasing pain clinically relevant to tension type headache and to migraine attacks. We detected decreases the number of migraine days after normalization posture in all cases. Posture imbalance provoked relapse, MTrPs activity was higher after recent stress.
Conclusion
Trigger points inactivation is crucial for migraine treatment, continuous management requires sophisticated individualized approach. Application of individualized patient profiling is strongly recommended for follow-up investigations in context of presented hypothesis.
Disclosure of Interest
None Declared
Paediatric headaches
MTV20-DP-098
PEDIATRIC HEADACHE – WHEN TO WORRY ?
Eugenia Roza1,2*, Diana Epure1, Oana Vladacenco1,2, Evelina Carapancea1,2, Raluca I. Teleanu1,2
1Dr Victor Gomoiu Children’s Hospital
2UMF Carol Davila, Bucharest, Romania
Introduction
Headache is a common symptom within the pediatric population and a frequent reason for referral towards the child neurologist’s office. The ethiological spectrum is wide, starting from benign non-structural causes to life-threatening conditions.
Objectives
The objectives of our study were to demonstrate the vast ethiology of headache in children and highlight the importance of a complete evaluation in order to provide an accurate diagnosis and adequate therapeutic options.
Methods
We present our clinical experience in the pediatric neurology department of Dr Victor Gomoiu Children’s Hospital between 2016–2020 regarding children with headache episodes.
Results
The most frequent reason for headache in our study population was non-structural with a very important emotional component, followed by migraine, CNS infections and stroke. The patients included in the study presented “headache red flags” for a possible structural abnormality such as: nausea/vomiting, vertigo, focal neurological deficits, VAS score of 7 and higher, nocturnal headache, persistent headache, occipital pain localization etc. The evaluation consisted of video-EEG, brain imaging studies, ENT evaluations, psychological and/or psychiatric assessment and cardiological evaluation
Conclusion
In our study the vast majority of headache episodes were attributed to an emotional cause such as stress, anxiety or other mental health problems. However, we also encountered cases with more severe ethiology such as migraine, CNS infections or stroke. The “headache red flags” were present in both cases and this finding highlights the importance of complete evaluations in cases of pediatric headache and early recognition of possible life-threatening conditions. It is also paramount to acknowledge the importance of psychological factors in pediatric headache and to to offer the patient the proper care and support as soon as possible.
Disclosure of Interest
None Declared
References
Psychological and behaviour factors
MTV20-DP-099
IS THE BURDEN OF SMOKING HISTORY ASSOCIATED WITH HEADACHE AMONG FORMER SMOKERS?
Jonathan H. Smith1, Wu Chunyi2 and Ehab Y. K. Harahsheh1*
1Department of Neurology, Mayo Clinic, Scottsdale
2Institute of Gerontology, University of Micihigan Ann Arbor, Ann Arbor, United States
Introduction
Cigarette smoking has been associated with headache in prior studies, where an incremental association between likelihood of headache and both pack-years and cigarettes smoked daily has been observed. However, the impact of smoking history on headache among former smokers is unknown. This cross-sectional study addressed this question, with the hypothesis that an increased burden of smoking history would remain associated with headache even after smoking cessation.
Objectives
To assess whether the burden of smoking among former smokers is associated with headache even after smoking cessation.
Methods
We examined cross-sectional association between elements of smoking history and headache among former smokers from the National Health and Nutrition Examination Survey (NHANES) from 1999–2004 using a multiple regression model. The existence of headache was determined by an affirmative response to this question, “During the past 3 months, did you have severe headaches or migraines?” The ages participants first and last smoked regularly, duration of smoking, number of years since quitting, number of cigarettes smoked daily when quit, and the presence of household smokers were compared between those reporting or not reporting headache.
Results
Among 2503 former smokers, 375 (14.9%) reported headache. After adjustment for age, sex, body mass index (BMI), race/ethnicity, educational attainment, poverty index and alcohol use, statistically significant associations were not observed between any element of the smoking history and headache. However, individuals reporting smoking regularly before the age of 16 were noted to have significant association (OR: 1.38; 95% CI: 1.05–2.44; P = 0.027).
Conclusion
The burden of the smoking history was not associated with headache among former smokers. However, cigarette smoking in young adults may portend unfavorable longitudinal headache outcomes.
Disclosure of Interest
None Declared
References:
Secondary headaches
MTV20-DP-100
CERVICAL VESTIBULAR EVOKED MYOGENIC POTENTIALS IN IDIOPATHIC INTRACRANIAL HYPERTENSION
Feray G. UyaroğluRoza UçarGülgün ŞengülNeşe Çelebisoy1
1Neurology, Ege University Medical School, İzmir, Turkey
Introduction
Idiopathic intracranial hypertension (IIH) is raised intracranial pressure without any identifiable etiology. The inner ear structures are susceptible to cerebrospinal fluid (CSF) pressure changes due to connections between the CSF space and the labyrinth to explain the audio-vestibular symptoms such as pulsatile tinnitus or dizziness reported in 50–60% of these patients.
Objectives
To investigate the vestibular functions by using cervical vestibular evoked myogenic potentials (cVEMPs) in IIH.
Methods
cVEMPs were recorded in 30 patients with IIH prior to lumbar puncture. Thirty healthy volunteers constituted the control group. The latencies of peaks p13 and n23 and peak-to-peak amplitude of p13–n23 were measured.
Results
Responses were gathered bilaterally from all healthy controls. In 30 patients with IIH, 49 responses could be gathered from 60 tests (81.7 %). The potential was absent bilaterally in five and unilaterally in one patient. When recorded, the latency and amplitude values of the responses of the patients were not significantly different from the healthy controls (p>0.005). A correlation between CSF pressure and response persistence could not be determined.
Conclusion
cVEMPs are affected in patients with IIH and the main finding is the absence of the responses. Increased intracranial pressure causing sound transmission changes within the inner ear can affect the saccular afferents and may end up with absent responses on air conducted cVEMP recordings. To comment on the correlation between the CSF pressure and cVEMP changes successive cVEMP recordings with longitudinal CSF pressure monitoring seem necessary.
Disclosure of Interest
None Declared
Mean p13, n23 latencies and p13-n23 amplitudes of the healthy controls and IIH patients with recordable responses

p values show the right-left comparison of each group.
References
Secondary headaches
MTV20-DP-101
HEADACHE AT ONSET OF THE FIRST EVER ISCHEMIC STROKE
Elena R. Lebedeva1*, Anton V. Ushenin2, Natalia M. Gurary3, Denis V. Gilev4 and Jes Olesen5
1Neurology, International Headache Center “Europe-Asia”, the Ural State Medical University
2Neurology, The Ural State Medical University
3Neurology, Medical Union “New Hospital”
4Department of Econometrics and Statistics, the Graduate school of Economics and Management, the Ural Federal University, Yekaterinburg, Russian Federation
5Neurology, Danish Headache Center, Rigshospitalet-Glostrup, University of Copenhagen, Copenhagen, Denmark
Introduction
Despite several studies in this field, no studies investigated headache at onset of the first-ever ischemic stroke and many studies did not evaluate the previous history of headache, did not diagnose according to the International classification of headache disorders (ICHD) and did not have a sizeable simultaneous control group.
Objectives
Two important questions need to be elucidate. 1) Is headache at stroke onset causally related to the stroke? 2) What are typical clinical characteristics of these headaches, can they represent only new type of headache or also headache with altered characteristics?
Methods
Study populations consist of 550 patients (mean age 63,1, 54% males) with ischemic stroke and 192 control patients (mean age 58.7, 36% males). Eligible patients with stroke had first-ever acute ischemic stroke with presence of new infarction on MRI with DWI (n = 469) or on CT (n = 81). As a control group we studied in parallel patients who were admitted to the emergency room without acute neurological deficits or serious neurological or somatic disorders. All data were collected prospectively, using a standardized case-report form during face-to-face interviews by two neurologists during admission to the emergency room in both groups of patients.
Results
Headache at onset of ischemic stroke was present in 82 (14.9%) of 550 patients with stroke. In 64 of 82 patients (78.0%) they developed simultaneously with stroke, in 10 patients (12.2%) during first hour after stroke, in 8 (9.8%) in the period from 1 hour to 24 hours after.
A new type of headache at stroke onset was observed in 46 of 82 patients (56.1%). No controls had a new type of headache. 29 of the 46 patients had migraine-like headache, 10 patients had tension-type-like headache and 7 patient thunderclap-like headache. All these new types of headache developed simultaneously with stroke onset.
A previous headache with an altered characteristics was found in 30 of 82 patients (36.6%) and two control patients (p < 0.009). The typical changes included: prolonged duration (70%), change of localization (26.7%), increased intensity (36.7%), development of new accompanying symptoms (36.7%), increased frequency (13.3%) and refractoriness to usual treatment (30%). Among these patients 9 (30%) had migraine-like headache and 21 (70%) tension-type-like headache. In 14 patients these headaches developed at the same time as stroke and in 16 patients during 24 hours after stroke onset.
Six of 550 patients (1.1%) and 9 controls (4.6%) had a usual headaches without any changes. Four patients had these headaches at the time of stroke onset and two patients during the following 24 hours.
Conclusion
Headache of a new type did not occur in the control groups and headache with altered characteristics was highly significantly more common in stroke than in controls. These headaches are therefore causally related to ischemic stroke. In the terminology of the ICHD-3 they are “attributed to ischemic stroke”.
Disclosure of Interest
None Declared
Tension-type headaches
MTV20-DP-102
PRECISE DRY NEEDLING OF TRIGGER POINTS IN NECK, SHOULDER AND PTERYGOID MUSCLES IS EFFECTIVE TO TREAT MIGRAINE AND HEADACHE AND RESTORE POSTURE
Rostyslav Bubnov1* and Lev Kalika2
1Diagnostic and interventional ultrasound, Clinical hospital “Pheophania”, Kyiv, Ukraine
2NY Dynamic Neuromuscular Rehabilitation & Physical Therapy https://nydnrehab.com/, New York, United States
Introduction
Myofascial pain is widespread, can be reliable cause of headaches; Myofascial Trigger Points (MTrPs) treatment of the head and neck muscles was reported to be effective for tension-type headache and migraine [1]. We proposed a new approach of trigger point therapy, performing precise muscle dry needling under ultrasound (US) guidance [2- 3], effective for treatment numeral pain localizations and widespread pain. Suboccipital triangle has complex vascularity and innervation network with sympathetic signaling, together with oral and TMJ dysfunction influences on the posture, muscular system and headache development.
Objectives
Methods
We included to the study 25 patients (16 females, avarage age was 37±7 years old). All patients suffered from intensive chronic persistent or recurrent one- or two-sided pain in temporal and/or occipital areas; sometimes radiation to neck, arm and teeth, TMJ dysfunction symptoms. All patients underwent general exam, MRI, precise physical tests, extensive functional multiparameter neuromuscular US. Then patients received DN of detected MTrPs under US guidance according to R.Bubnov [1]: MTrPs were identified via clinical examination, referred pain pattern, US identification; single fine (28G) steel needle DN under US guidance was applied to elicit the local twitch response (LTR) and/or `needle grasp`. Visual analogue scale data (VAS 0–10) was used to measure pain, physical exam and functional ultrasound for monitoring movement. Further evaluation of patients` reports was completed after 1 month.
Results
The active and latent MTrPs were diagnosed in rectus and obliquus capitis muscles, shoulder rotators and medial, lateral pterygoid muscles. Most patients demonstrated individually specific patterns of MTrPs mapping in these areas. In one session 1–3 needles were inserted, 1–2 sessions applied to each patient. Retention of needles depended of muscle LTR duration. Shoulder dysfunction (impingement) with active trigger points in shoulder rotator muscles was detected and successfully treated in 23 patients. DN resulted immediately in decreasing pain clinically relevant to tension type headache and to migraine attacks (80% decreasing by VAS, p < 0.01), and sustainable pain relief outcome after one month observation (18 patients did not need another session); seven patient received another session after 2–3 weeks after first procedure. After one month all patients reported significant decreasing of pain levels and range of headache attacks.
Conclusion
Dry needling trigger points in suboccipital pterygoid muscles under ultrasound guidance is effective to treat headache and restore regional posture. Shoulder rotator muscles are important in postural imbalance leading to headaches, evoked by myofascial disorders.
Disclosure of Interest:
None Declared
