Abstract
Introduction
The Rutgers Acquired Equivalence Test is a visually guided equivalence learning paradigm that involves rule acquisition and generalization. Earlier we found impaired performance in this paradigm among adult migraine patients without aura. The aim of the study was to investigate if similar impairments can be found already in the pediatric form of the disease and to compare the performance of the pediatric study population with that of an adult study population. We hypothesized that the deficits observed in adults would be observable already in the pediatric population.
Methods
Twenty-seven children and adolescents newly diagnosed with migraine without aura and 27 age- and sex-matched healthy controls were tested with the Rutgers Acquired Equivalence Test. Their performance data were compared to each other and those of an earlier adult study population involving 22 patients and 22 age- and sex-matched healthy controls. Four parameters characterizing performance in the two main phases of the paradigm were calculated for each of the four groups. Performance parameters were compared with Mann-Whitney U test.
Results
In contrast to the decreased performance of the adult patients in the Rutgers Acquired Equivalence Test, no significant difference was found between pediatric patients and controls in any phase of the paradigm.
Conclusion
Children living with migraine without aura do not exhibit the same cognitive deficits in the Rutgers Acquired Equivalence Test as their adult counterparts. It can be hypothesized that the deficit of equivalence learning is not an inherent feature of the migrainous cognitive profile, rather the result of the interference of the disease with normal development.
Introduction
While the issue of the cognitive effects of migraine crops up in the literature from time to time (1–3), the area is still relatively under-investigated, especially regarding the child/adolescent patient population. The available studies suggest that this patient population might be affected by various cognitive deficits, including but not limited to poor perceptual organization, impaired visuospatial memory, slower information processing and impaired executive functions (4,5). Our own research group came to similar conclusions (6–8). Of the most recent studies, Margari and colleagues showed that both migraine and tension-type headache affect non-verbal cognitive abilities in children and adolescents (9). Termine and co-workers, themselves corroborating the presence and seriousness of the problem in their review, add that “…migraine-related cognitive deficits may interfere with functioning levels across several settings”, and suggest that “[a] careful analysis of cognitive impairment in the context of migraine is pivotal for making informed decisions on the most appropriate care pathways” (10).
The Rutgers Acquired Equivalence Test (RAET) investigates visually guided equivalence learning, a specific kind of associative learning (11). Acquired equivalence is a form of learning in which generalization is induced between two seemingly different stimuli that participants are trained to treat as equivalent in terms of the same associated responses. This is referred to as functional equivalence because the basis of grouping is not some inherent characteristic of the stimuli, but the outcome associated with them (12). RAET consists of two main parts. In the acquisition part, the participants must learn to associate pairs of visual stimuli (faces and colored fish) and the equivalence testing or transfer part includes new, hitherto not presented pairings of the same stimuli. If the functional equivalence has been successfully established, the participants will have no difficulty with these new pairings.
The two main parts are divided into three phases. The efficiency of pair learning is assessed in the acquisition phase, which involves feedback-guided learning of face-fish associations. The efficiency of the recall of these previously acquired associations is evaluated in the retrieval part of the test phase, without feedback. The success of equivalence acquisition is assessed in the generalization part of the test phase.
Studying patients with selective damage to the basal ganglia or the hippocampi, Myers and co-workers were able to demonstrate that the named structures play a role in different phases of the test (11). Compared to healthy controls, persons living with Parkinson’s disease (basal ganglia dysfunction) showed slower pair learning in the acquisition phase, and performed well in the test phase; however, patients with hippocampal atrophy had difficulties primarily in the test phase. The validity of the link between these subcortical areas and performance in the different phases of the paradigm has been borne out by several studies since the publication of the original paper (13–17).
In 2017, we demonstrated deficits in this paradigm in a group of adults with migraine with a long disease history (18). In another study, we investigated a cohort of 265 healthy subjects aged 3–52 years and found a significant association between age and associative learning/retrieval, but no significant association between age and generalization (19). With the present study, we sought to examine migraine-related deficits in RAET from a different perspective and asked if the deficits observed in adulthood are already present in pediatric migraine. To give a better characterization of the performance of the pediatric population, we compared their performance to that of the adult study population of the 2017 study. We hypothesized that the deficits observed in adults would be observable already in the pediatric population. In other words, the study tested the hypothesis that poor performance in this paradigm is an inherent feature of migraine, possibly indicating some inherent structural or functional alteration of the underlying neural structures.
Methods
Recruitment
The study population was recruited from the Department of Pediatrics, Faculty of Medicine, University of Szeged, Hungary. The study period spanned 18 months altogether, from January 2018 to November 2019 (the 3 months of the summer holiday season were excluded in each year). Inclusion criteria were a new diagnosis of migraine without aura as determined by the same pediatric neurologist (JK) according to ICHD-3 beta criteria and age in the pediatric range (maximum age: 18 years). Only patients with no other neurological, psychiatric or ophthalmological disorder were eligible for this study. A negative history was verified from the patients’ files. A further exclusion criterion was color vision deficiency, which was tested using Ishihara plates before testing. For all patients, at least 5 days had passed since the last attack at the time of testing. To allow an optimal matching of controls, the patients were also tested for intelligence with Raven’s progressive matrices (PM). In the pediatric population, both patients and controls were free of psychotropic medication, including any migraine treatment at time of testing.
The sample size was determined by the rigorous application of the diagnostic and inclusion/exclusion criteria. In total, 29 newly diagnosed pediatric patients with migraine were approached in the study period. One was excluded for signs of excessive anxiety during the procedure, and another failed to cooperate. This left us with the final sample of 27 patients.
The study was carried out according to a one case-one control design. Controls were recruited from primary schools and secondary schools in Szeged, Hungary. Members of the control group had no history of any kind of headache, and they were also free of any kind of neurological, psychiatric, or ophthalmological disorder. Similarly to the study population, only controls with intact color vision were eligible, as assessed with the Ishihara plates, and they were also tested with PM. Matching of cases and controls was based on age (with a tolerance limit of ±6 months), sex, and score achieved on PM.
The details of the recruitment of the adult patients and controls is described in Őze et al. (18).
The study protocol conformed to the ethical principles of the Declaration of Helsinki in all aspects. Prior to testing, participants and their guardians were informed about the background, aims and procedures of the study both orally and in written form. None of the subjects received any compensation for their involvement, and they were informed that the study has merely scientific purposes without direct diagnostic or therapeutic use and they are free to leave at any time. All recruitment and protocols were conducted with written informed consent and with the approval of the Regional Research Ethics Committee for Medical Research at the University of Szeged, Hungary (Reg. No. 53/2015.)
The visual associative learning paradigm
Testing was carried out according to Myers and co-workers (11). The testing software (originally written for iOS) was used and rewritten in Assembly (for Windows) with the written permission of Myers and colleagues at Rutgers University, NJ, USA. Stimuli were presented and responses were recorded with a personal computer with a CRT screen. The testing sessions took place in a quiet room with the subjects sitting at a comfortable distance from the computer screen (114 cm). One subject was tested at a time. To ensure that the participants could concentrate on the task, no time limit was set either for the individual trials (see below) or the test as a whole.
The visual stimuli referred to as antecedents were cartoon faces of a woman (A1), a girl (A2), a man (B1) and a boy (B2). The stimuli referred to as consequents were yellow (X1), red (X2), green (Y1) and blue (Y2) fish.
During a trial, the participant was presented with an antecedent (a face) and two consequents (a pair of fish of different color) and asked to choose one of the latter by pressing one of two buttons on the keyboard. The trials were organized into two main phases: Acquisition and test. The test phase was further broken down to retrieval and generalization (see below). Depending on the actual testing phase, the choice was, or was not followed by feedback on the correctness of the choice.
During the acquisition phase, the participants learned a series of antecedent-consequent pairs via trial and error. When face A1 or face A2 were shown, the correct choice was fish X1 over fish Y1; however, when face B1 or face B2 appeared on the screen, the correct answer was fish Y1, instead of fish X1. Visual feedback on the correctness of the subject’s choice was always given immediately in the form of the words CORRECT (in green) and INCORRECT (in red) displayed on the screen under the actual antecedent-consequent pair. This way, besides the face-fish associations, the participants also learned that the face A1 is equivalent to face A2 in terms of their relation to the consequents (fish). New associations were introduced gradually (see Figure 1 and Table 1), and they were presented mixed with trials of previously learned associations. The subjects had to achieve a certain number of consecutive correct answers after the presentation of each new association to be allowed to proceed. This number was four when the first association was presented, and it was increased by two upon the presentation of each association that followed (up to a maximum of 12). Thus, the length of the acquisition phase varied among the participants, depending upon how efficiently they learned.
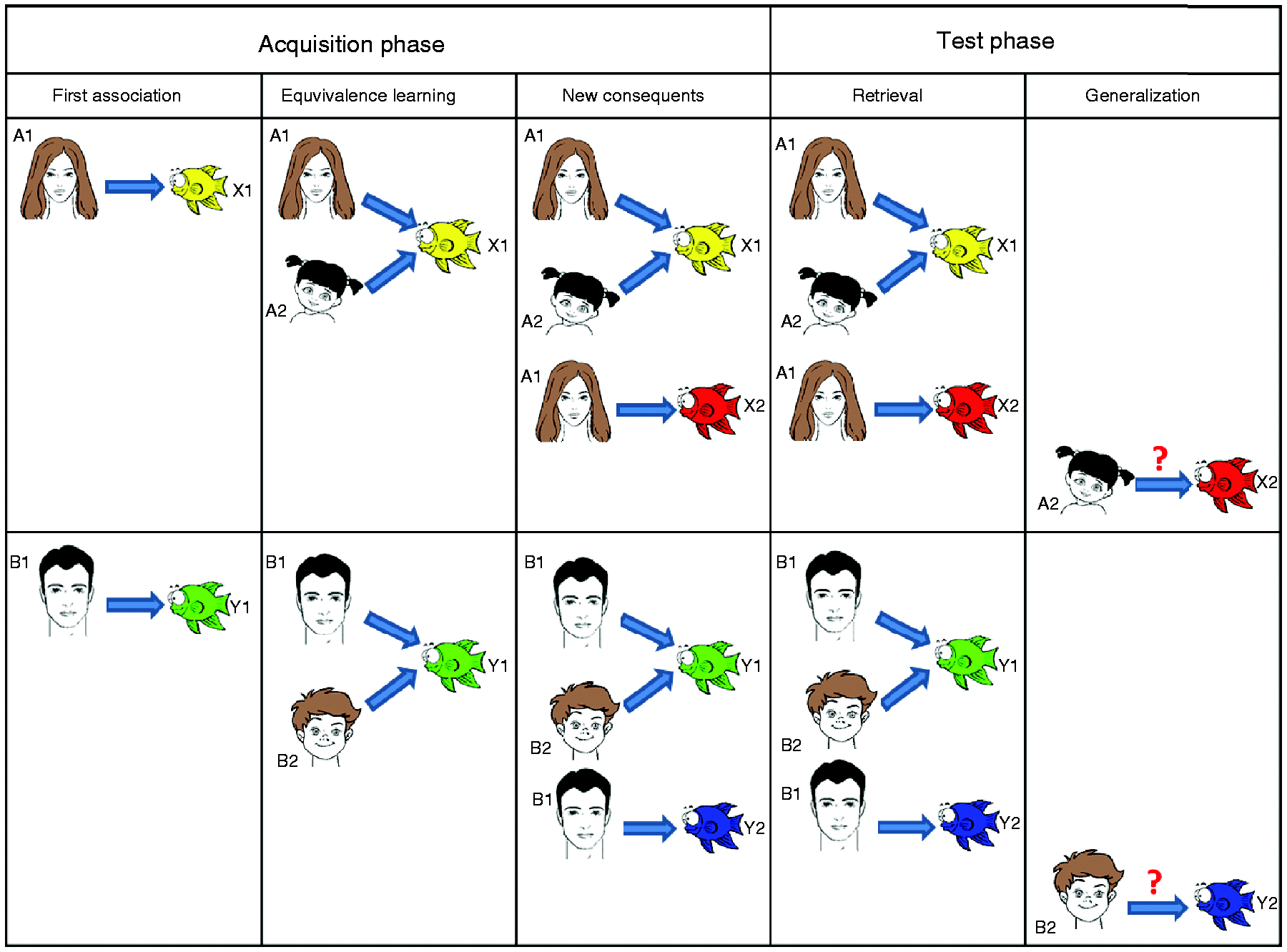
The antecedent–consequent pairs of the test. The antecedents were cartoon faces of a woman (A1), a girl (A2), a man (B1) and a boy (B2). The consequents (responses) were drawings of fish of yellow (X1), red (X2), green (Y1) and blue (Y2) colors.
A summary of the applied paradigm. A, B: antecedents; X, Y: consequents. See also Figure 1.

Demographic and basic migraine-specific characteristics of the studied groups.

In the test phase, the task remained the same, but visual feedback was no longer provided. In contrast to the acquisition phase, the test phase always involved 48 trials (12 new and 36 previously learned associations). Subjects were not informed that new associations would have to be formed, only that their task remained the same, but without feedback.
Statistical analyses
Statistical analysis was performed in SPSS 21.0 (IBM, USA). The level of significance was set at p = 0.05.
Participants were divided into four groups: Pediatric patient (PP), pediatric control (PC), adult patient (AP) and adult control (AC). The performance of the participants was characterized with four parameters: The number of trials necessary for the completion of the acquisition phase (NAT), association learning error ratio (the ratio of incorrect choices during the acquisition trials, ALER), retrieval error ratio (RER), and generalization error ratio (GER).
Before the hypothesis tests, extreme outliers were removed from the dataset, as suggested by Tukey (20): For each variable in each group, the 25th and 75th percentile limits (Q1 and Q3) were calculated, the value of Q1 was subtracted from the value of Q3, the result was multiplied by 1.5, and the product was subtracted from Q1 and added to Q3 to modify the limits (Q1’ and Q3’). Values under Q1’ and over Q3’ were removed. After data cleaning, medians, minima and maxima were calculated. As the normality and homogeneity assumptions were not fulfilled in all groups and all parameters, we decided to use the Mann-Whitney U test for hypothesis testing (PP vs. PC and AP vs. AC). To determine if sex should be considered as a factor in the analysis of the pediatric groups, Mann-Whitney U tests were conducted with sex as a categorical variable for controls and patients separately and for all four target variables. As the adult population consisted almost entirely of females (which is quite normal for an adult migraine population), such an analysis could not be meaningfully conducted in the adult groups. Post-hoc power tests for the Mann-Whitney U test were conducted in G*Power 3.1.9 (Universität Kiel, Germany) to determine if the sample sizes of the pediatric population were large enough to allow comparison with the adult population.
To reduce bias, those who actively participated in testing were excluded from the analysis and the statistical analysis was blinded (the analyst was not aware of the meaning of the group codes).
Results
The pediatric population
A total of 54 children aged 8 to 17.5 years participated in the study. The pediatric patient group consisted of 27 participants and the control group involved 27 healthy subjects matched by age, sex and level of intelligence as indicated by PM. The mean age in the pediatric patient group was 14.1 ± 2.9 years, and the female-to-male ratio was 15 to 12. As for the control group, the mean age was 14.2 ± 2.9 years, and the female-to-male ratio was the same as in the patient group.
The adult population
Forty-four adult subjects aged 20–65 years were recruited for the study of Őze and co-workers (18). The adult patient group consisted of 22 patients, and the control group involved 22 healthy individuals matched by age, sex and level of education. The mean age in both groups was 40 ± 11.76 years. The female-to-male ratio was 20 to 2. The demographic and migraine-specific characteristics of the investigated groups are summarized in Table 2.
Data cleaning
Altogether 18 entries were removed out of the 392 (4.5%). All removed entries exceeded Q3’. Four entries were removed from GER and one from RER in AC, four from NAT in PC, three from NAT, one from ALER and two from RER in PP, and three from NAT in AP. The removed entries were excluded from the analyses.
The effect of sex in the pediatric groups
We observed no significant differences between the two genders in both the control and the patient groups in any of the investigated parameters (for the pediatric patient group NAT: p = 0.619, U = 61.00; ALER: p = 0.471, U = 69.50; RER: p = 0.360, U = 58.00; GER: p = 1.000, U = 90.00), so this parameter was omitted from the further analyses.
Power analysis
The input effect sizes (Cohen’s d) were derived from the adult population (NAT: 0.5, ALER: 0.88, RER: 0.74, GER: 1.72). The statistical power achieved from the pediatric population was 0.55 for NAT, 0.88 for ALER, 0.69 for RER and 0.99 for GER; that is, the hypothesis tests have acceptable power (over 0.8) for ALER and GER and were under-powered for NAT and RER.
Test performance
Performance data of 98 participants altogether were analyzed.
In the adult population, significant difference was found between patients and controls in ALER (p = 0.043, U = 155.50,) and GER (p < 0.001, U = 66.00). The difference was not significant in NAT (p = 0.080, U = 141.50) and RER (p = 0.395, U = 195.50). Figure 2 shows the comparisons of the adult groups.
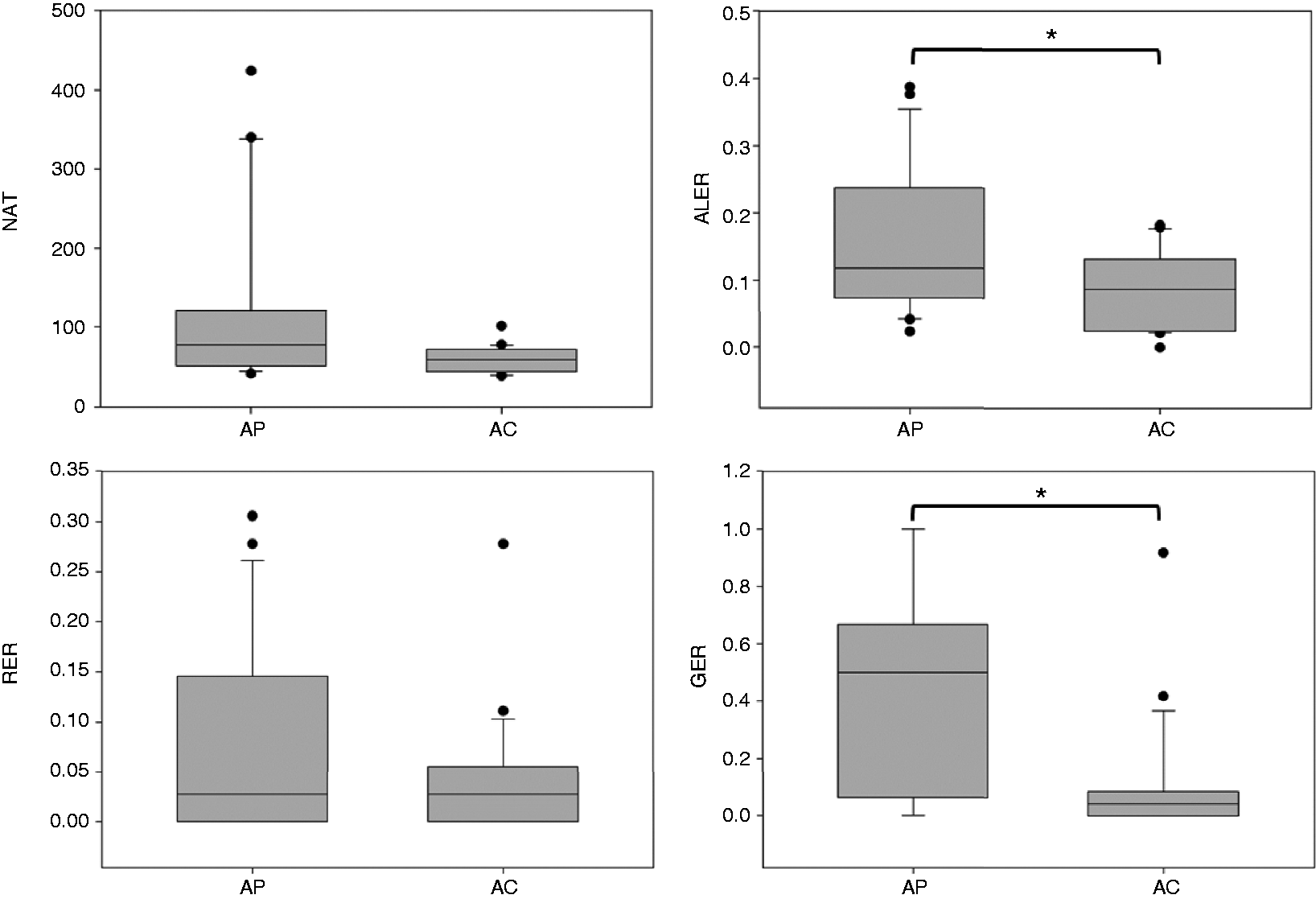
Performance of the adult population. The lower margin of the boxes shows the 25th percentile, the line within the boxes marks the median, and the upper margin of the boxes indicates the 75th percentile. The error bars (whiskers) above and below the boxes indicate the 90th and 10th percentiles. The black dots represent the outliers.
In the pediatric population, no significant differences could be found in any of the variables (NAT: p = 0.395, U = 235.50; ALER: p = 0.369, U = 300.00; RER: p = 0.621, U = 310.00; GER: p = 0.484, U = 323.50). Figure 3 shows the comparisons of the pediatric groups.

Performance of the pediatric population. The lower margin of the boxes shows the 25th percentile, the line within the boxes marks the median, and the upper margin of the boxes indicates the 75th percentile. The error bars (whiskers) above and below the boxes indicate the 90th and 10th percentiles. The black dots represent the outliers.
Table 3 gives a descriptive summary of the performance of all groups.
Performance by the studied parameters.

NAT: number of acquisition trials; ALER: association learning error ratio (the ratio of incorrect choices during the acquisition trials); RER: retrieval error ratio; GER: generalization error ratio.
Discussion
This study tested a simple hypothesis. We hypothesized that the pediatric patient population would show significantly poorer performance in at least one phase of our learning paradigm compared with age- and sex-matched controls. In other words, we hypothesized that deficits observed in adults (18) would be observable already in the pediatric population, indicating some sort of inherent structural or functional alteration of the underlying neural structures. The discussion is confined to this hypothesis, especially as we do not wish to deal with questions that have been addressed in other studies better designed to answer those specific questions. Please note that a detailed discussion of the comparisons between the adult groups is found in Őze et al. (18). Similarly, the development of the studied parameters from 3 to 52 years of age was analyzed in another study by Braunitzer et al. (19), where the number of subjects (n = 265) allowed statistically supported, detailed developmental conclusions. The number of subjects in this study does not allow such conclusions.
The current study failed to support the hypothesis. The difference between the pediatric groups was not significant in any of the studied parameters. It must be emphasized that even if a null result should always be interpreted with caution (especially with a small sample size), the study was adequately powered to allow conclusions regarding ALER and GER, the key variables. Thus, these results indicate that equivalence acquisition is not one of the functions affected by the disease from an early age onwards.
In contrast, as already discussed in our previous study (18), adult patients show significantly lower performance in both acquisition and generalization than matched controls: Adults living with migraine acquired visual stimulus pairings and the pairing rule with greater difficulty than matched controls (ALER); but having acquired them, their recall performance for already-seen stimulus pairs was on par with that of controls (RER). When it came to generalizing the pairing rule (GER), though, they performed significantly worse. To put it simply, no difference in childhood turns into a large gap by adulthood, and this is especially true for generalization. The question logically arises: What happens in the meantime? This is a question only a large longitudinal study supported by instrumental measurements could exactly answer. The available literature offers some clues, though. What follows is a brief summary of how the (mostly imaging and histological) findings of other research groups could coherently explain our present results. Please note that this hypothetical explanation does not follow directly from our measurements. While, to our knowledge, this is the most probable explanation, this is not the only possible explanation.
An increasing number of studies have come to the conclusion that migraine affects both areas that are of key importance for the successful completion of the studied task: The basal ganglia and the hippocampi (21–26).
In their seminal 2011 fMRI study, Maleki and colleagues concluded that ‘migraine attacks the basal ganglia’ (21) and added that the impairment was proportional to attack frequency. Petrusic and colleagues demonstrated that volumes of the right globus pallidus, left globus pallidus, and left putamen were significantly smaller in migraine patients (with aura) than in healthy controls (22). The research group of Yuan examined persons living with migraine without aura and concluded that the volume of the nucleus accumbens and caudate nucleus of patients was significantly smaller than that of controls. The authors also found dysfunctional interictal dynamics within the basal ganglia (27).
As for the hippocampi, Maleki’s group found significantly larger bilateral hippocampal volume in migraine patients with low attack frequency than in the high-frequency and control groups. They proposed that this finding was suggestive of an initial adaptive plasticity that may then become dysfunctional with increased frequency (28). Yu and colleagues performed voxel-based morphometric analysis and found that the left hippocampus/parahippocampal gyrus volume of their healthy control group was smaller than that of the episodic migraine group but was larger than that of the chronic migraine group (29). Liu and colleagues conducted a review and found that “factors including headache frequency, accumulative number of migraine attacks, anxiety score, depression score, and genetic variants are related to hippocampal morphology and functional changes in people with migraine” (25). The same group also published an MRI-based volumetric study, where they found that the hippocampus and amygdala displayed a structural plasticity linked to both headache frequency and clinical outcome of migraine (26). These findings seem to support the plasticity-related hypothesis of Maleki and colleagues mentioned above (28). What is common in these studies is that they all emphasize the role of attack frequency and/or the cumulative number of attacks in the structural changes. To sum it up, according to our present knowledge, migraine attacks not only the basal ganglia but also the hippocampi. How does that affect the brain in development?
Developmental studies tell us that the human striatum shows protracted development, well into adolescence (30), while the hippocampi never really seem to finish development; rather, they are in a constant state of dynamic remodeling (31). Thus, both structures are vulnerable for a long period. In fact, it is safe to assume that the hippocampi – due to their extreme plasticity – remain sensitive to insults throughout life. The other side of the hippocampal plasticity coin is, of course, that dynamic plasticity offers a way to remodel and regenerate once the insult is gone. Unfortunately, no study has ever examined this question in the context of migraine, but it would be intriguing to see if hippocampus-related cognitive functions improve after a prolonged attack-free period.
Looking at the results of the present study, the influence of migraine appears to be obvious. While we do not wish to draw firm developmental conclusions (for reasons explained at the beginning of this section), it cannot escape attention that the median generalization error ratio of pediatric patients is 8%, while that of adult patients is 50%. Normally, generalization reaches adult level by the age of 6, and remains relatively constant thereafter (19). The numbers suggest that in migraine there is massive deterioration.
The limitations of this study include the relatively low number of participants (due to and partially offset by the strict application of the diagnostic criteria) and that the adult dataset used for comparison was originally compiled for another study (which is not re-discussed here). In our opinion, these limitations do not interfere with the validity of the inference that the equivalence learning deficit observed in adult migraine patients is not present in pediatric patients.
Conclusions
Our results seem to indicate that the deficit of equivalence learning is not an inherent feature of the migrainous cognitive profile. Developmental interference of migraine appears to be a plausible explanation, but this is only a hypothesis at this point.
Key findings
Pediatric migraine patients were not characterized by the same deficit of equivalence learning as described in a sample of previously recruited adult patients. Pediatric patients performed just as well as controls in each phase of the learning paradigm. The observations suggest that deficient equivalence acquisition is not an inherent feature of migraine.
Footnotes
Acknowledgements
Attila Nagy and Gábor Braunitzer contributed equally to this study. The authors would like to express their gratitude to Márton Edelmayer for his help with data collection.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the University of Szeged Grant SZTE-ÁOK-KKA (Grant No. 2019/270-62-2.)
